Introduction
The World Health Organization (WHO) baseline report on the Decade of Healthy Ageing (2021–2030) sets the goal of optimizing older people’s functional ability and mobility (World Health Organization, 2020). The report discusses the negative impact that medications can have on functional ability, particularly in terms of movement impairment and balance interference (World Health Organization, 2020). Cumulative exposure to anticholinergic and sedative medications measured with the Drug Burden Index (DBI) (Hilmer et al., Reference Hilmer, Mager, Simonsick, Cao, Ling, Windham, Harris, Hanlon, Rubin, Shorr, Bauer and Abernethy2007) has been associated with impaired physical function across a range of measures including decreased gait speed (Cao et al., Reference Cao, Mager, Simonsick, Hilmer, Ling, Windham, Crentsil, Yasar, Fried and Abernethy2008; Gnjidic et al., Reference Gnjidic, Cumming, Le Couteur, Handelsman, Naganathan, Abernethy and Hilmer2009, Reference Gnjidic, Bell, Hilmer, Lonnroos, Sulkava and Hartikainen2012; Hilmer et al., Reference Hilmer, Mager, Simonsick, Ling, Windham, Harris, Shorr, Bauer and Abernethy2009), decreased Barthel Index score (Bostock et al., Reference Bostock, Soiza and Mangoni2013; Gnjidic et al., Reference Gnjidic, Bell, Hilmer, Lonnroos, Sulkava and Hartikainen2012; Lowry et al., Reference Lowry, Woodman, Soiza, Hilmer and Mangoni2012), as well as reduced performance on the chair stand test (Cao et al., Reference Cao, Mager, Simonsick, Hilmer, Ling, Windham, Crentsil, Yasar, Fried and Abernethy2008; Gnjidic et al., Reference Gnjidic, Bell, Hilmer, Lonnroos, Sulkava and Hartikainen2012; Wouters et al., Reference Wouters, Hilmer, Gnjidic, Van Campen, Teichert, Van Der Meer, Schaap, Huisman, Comijs, Denig, Lamoth and Taxis2020) and on the Timed Up and Go (TUG) test (Gnjidic et al., Reference Gnjidic, Bell, Hilmer, Lonnroos, Sulkava and Hartikainen2012). Cumulative exposure to anticholinergic medications has also been associated, although inconsistently, with impaired cognition (Welsh et al., Reference Welsh, van derWardt, Ojo, Gordon and Gladman2018).
The WHO International Classification of Functioning, Disability, and Health (ICF) defines capacity as the assessment, for example, by the aforementioned tests, of what a person can do in a standardized environment (World Health Organization, 2001). The ICF emphasizes the importance of additionally measuring what a person can do in a usual or real-life setting, referred to as performance. Our ability to collect refined mobility metrics from patients has been greatly enhanced by smartphones, wearable devices, and other linked devices (Huckvale et al., Reference Huckvale, Venkatesh and Christensen2019; Montero-Odasso et al., Reference Montero-Odasso, Almeida, Bherer, Burhan, Camicioli, Doyon, Fraser, Muir-Hunter, Li, Liu-Ambrose, McIlroy, Middleton, Morais, Sakurai, Speechley, Vasudev, Beauchet, Hausdorff and Rosano2019). Patient-reported outcomes (PROs) instruments such as the Late-Life Function and Disability Instrument (LLFDI) (Haley et al., Reference Haley, Jette, Coster, Kooyoomjian, Levenson, Heeren and Ashba2002; Jette et al., Reference Jette, Haley, Coster, Kooyoomjian, Levenson, Heeren and Ashba2002) provide information on a wide range of everyday activities that complement the direct assessment of capacity and performance.
Clinical and research interest in the negative impact that medications have on older adults is increasingly focused on deprescribing anticholinergic and sedative medications due to the mounting evidence of the physical impairment (Phutietsile et al., Reference Phutietsile, Fotaki, Jamieson and Nishtala2023) associated with the cumulative use of these medications in older adults. The refined mobility metrics captured by wearable devices during the deprescribing process can contribute to a better understanding of how these medications impact mobility. The study’s primary objective is to evaluate the feasibility of assessing the impact of reducing the anticholinergic and sedative medication burden on physical function measured by wearable devices in the laboratory and in real-life settings, in support of the development of a future study. The study’s secondary objective is to explore the impact of reducing the anticholinergic and sedative medication burden on physical function.
Methods
Study design, population, and setting
This prospective longitudinal pilot study was conducted from July 2021 to March 2023 in two primary care clinics (PCC) and at the mobility laboratory of the Research Centre on Aging of the CIUSSS de l’Estrie-CHUS in Sherbrooke, Quebec, Canada. PCC pharmacists referred eligible patients to the research team for consenting. The included patients were as follows: 1) 60 years and older, 2) community-dwelling, 3) having a DBI score ≥ 1, and 4) agreeing to a deprescribing plan targeting a reduction in their DBI score of ≥ 0.5 points. Patients presenting with dementia or unstable medical condition(s) (≥ 1 unplanned hospitalization or ≥ 2 emergency department visits in the previous month) or requiring a walking frame as a mobility assistive device were excluded from the study. All of this study’s procedures have been successfully implemented separately in previous studies of our group (Cossette et al., Reference Cossette, Taseen, Roy-Petit, Villemure, Grondin, Ricard, Goyer, Blanchard, Joly-Mischlich and Éthier2019; Walha, Dagenais, et al., Reference Walha, Dagenais, Gaudreault, Beaudoin-Côté and Boissy2022; Walha, Gaudreault, et al., Reference Walha, Gaudreault, Dagenais and Boissy2022). The conduct of the study during the pandemic proved challenging. A cohort of 5 patients was judged sufficient to assess the feasibility of the procedure’s combined implementation, even though the initial sample size was set at 10. The study was approved by the Ethics Research Board of the CIUSSS de l’Estrie-CHUS (MP-2022-4235).
Study interventions and deprescribing
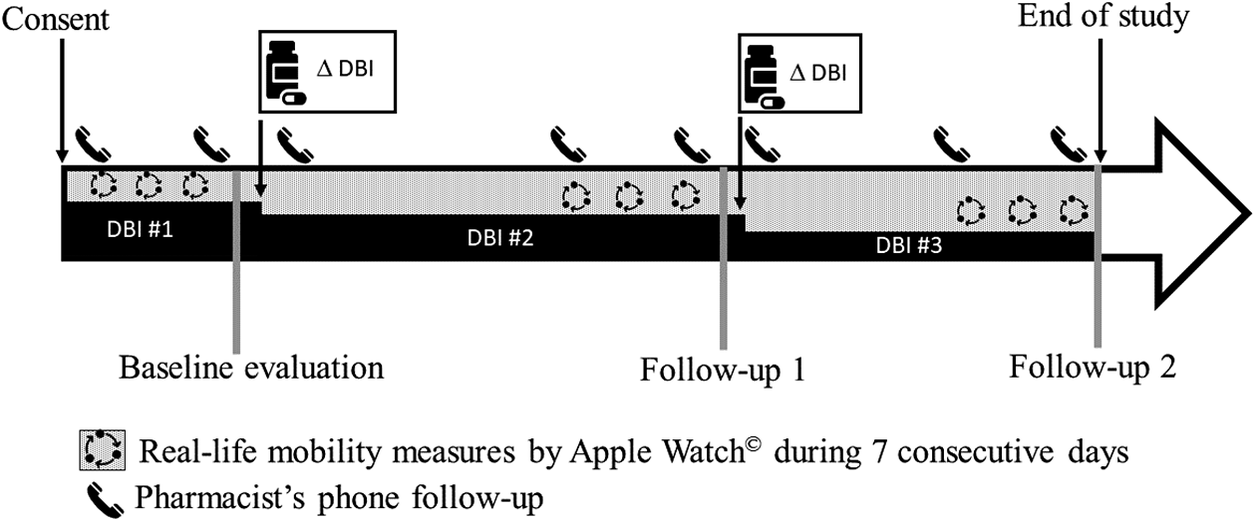
The participants’ physical function was assessed during three (baseline, follow-up 1, and follow-up 2) laboratory visits and in real-life environments in the days preceding these visits (Figure 1). As part of usual care, the PCC pharmacists developed the individualized deprescribing plans in collaboration with the participant and the PCC physicians and scheduled the laboratory visits according to the progress of these plans. The first follow-up visit was scheduled once the DBI score decreased by 0.25 from the baseline visit, while the second follow-up was planned after a further 0.25 decrease or increase (re-prescribing). Re-prescribing was allowed when medication tapering led to the return of the medical condition symptoms treated by the deprescribed drug or to withdrawal symptoms (Reeve et al., Reference Reeve, Moriarty, Nahas, Turner, Kouladjian O’Donnell and Hilmer2018; Scott et al., Reference Scott, Hilmer, Reeve, Potter, Le Couteur, Rigby, Gnjidic, Del Mar, Roughead, Page, Jansen and Martin2015). Providing optimal clinical care to the participants always took precedence over research objectives.
Study procedures.
Note. DBI, Drug Burden Index.

Exposure measurement: Drug Burden Index score
The DBI Calculator© (Kouladjian et al., Reference Kouladjian, Gnjidic, Chen and Hilmer2016), adapted for the Canadian context (Dearing et al., Reference Dearing, Bowles, Isenor, Kits, Kouladjian O’Donnell, Neville, Hilmer, Toombs, Sirois, Hajizadeh, Negus, Rockwood and Reeve2020), was used to calculate the DBI score for each participant throughout the deprescribing process. The DBI score is based on a dose–response relationship assuming linear additive effects (Hilmer et al., Reference Hilmer, Mager, Simonsick, Cao, Ling, Windham, Harris, Hanlon, Rubin, Shorr, Bauer and Abernethy2007). A DBI score of 0.5 points corresponds to exposure to one anticholinergic or sedative medication at the minimum licensed dose, which has been associated with clinically meaningful changes in physical function (Hilmer et al., Reference Hilmer, Mager, Simonsick, Cao, Ling, Windham, Harris, Hanlon, Rubin, Shorr, Bauer and Abernethy2007; Kouladjian et al., Reference Kouladjian, Gnjidic, Chen, Mangoni and Hilmer2014). The WHO Anatomical Therapeutic Chemical (ATC) classification was used to categorize medications (World Health Organization, s.d.).
Outcome measurement
Laboratory evaluations
The laboratory evaluations were selected from the mobility core-battery tests of the 2019 consensus on shared measures of mobility and cognition from the Canadian Consortium on Neurodegeneration in Aging (CCNA) (Montero-Odasso et al., Reference Montero-Odasso, Almeida, Bherer, Burhan, Camicioli, Doyon, Fraser, Muir-Hunter, Li, Liu-Ambrose, McIlroy, Middleton, Morais, Sakurai, Speechley, Vasudev, Beauchet, Hausdorff and Rosano2019). The 10-meter walk test, the Short Physical Performance Battery (SPPB), and the mini-BESTest were completed by the participants in that order during laboratory visits. Gait speed (meters/second (m/s)) was calculated during the 10-meter walk test. Small meaningful changes in gait speed have been shown to range from 0.04 to 0.06 m/s, whereas substantial changes varied from 0.08 to 0.14 m/s (Perera et al., Reference Perera, Mody, Woodman and Studenski2006). The SPPB consists of three tests: balance, gait speed, and chair stand, each scoring up to four points for a total score of 12; where a higher score indicates a better lower extremity function (Guralnik et al., Reference Guralnik, Simonsick, Ferrucci, Glynn, Berkman, Blazer, Scherr and Wallace1994). While small meaningful changes in SPPB ranged from 0.27 to 0.55 points, substantial changes varied from 0.99 to 1.34 points (Perera et al., Reference Perera, Mody, Woodman and Studenski2006). The mini-BESTest is a 14-item clinical balance assessment tool evaluating four dimensions: anticipatory postural adjustments, reactive postural control, sensory orientation, and dynamic gait with a maximum score of 28 and a higher score indicating better balance (Franchignoni et al., Reference Franchignoni, Horak, Godi, Nardone and Giordano2010). For the mini-BESTest, a minor detectable change of 3.5 points and a minimal important change of 4 points have been reported (Godi et al., Reference Godi, Franchignoni, Caligari, Giordano, Turcato and Nardone2013).
Participants underwent the mobility tests while wearing six Opal sensors (APDM Wearable Technologies, Portland, OR, USA), on their trunk, pelvis, wrists, and ankles. This validated clinical system (Mancini et al., Reference Mancini, King, Salarian, Holmstrom, McNames and Horak2011) extracted refined mobility metrics of gait speed, stride length, cadence, and double support time (Salarian et al., Reference Salarian, Horak, Zampieri, Carlson-Kuhta, Nutt and Aminian2010) among others, during the 10-meter walk test. Sit-to-stand and stand-to-sit transitions were assessed during the SPPB chair stand test using timing features, trunk angle, and right/left loading asymmetry (Salarian et al., Reference Salarian, Horak, Zampieri, Carlson-Kuhta, Nutt and Aminian2010). Quiet stance balance was assessed in the mini-BESTest stance test while the person is standing on a firm surface, feet together and eyes open, through an analysis of the estimated movement of the center of mass, as measured with sway area (Mancini et al., Reference Mancini, Salarian, Carlson-Kuhta, Zampieri, King, Chiari and Horak2012).
Real-life settings evaluations
Mobility was measured at home and in the community using two Series 6 Apple Watch® worn by the participants on their left wrist and right ankle. An application installed on the watch captured and logged the watch’s sensors’ (inertial and Bluetooth Low Energy (BLE)) signals. Measurement occurred at a stable DBI score during seven days preceding the laboratory visit, referred to as real-life assessment periods of seven days. A BLE beacon placed close to the front door informed on the position (inside/outside) of the participant. Step count was assessed as the total number of steps per day detected and segmented with inertial sensor signals using previously validated algorithms (Mueller et al., Reference Mueller, Hoefling, Muaremi, Praestgaard, Walsh, Bunte, Huber, Fürmetz, Keppler, Schieker, Böcker, Roubenoff, Brachat, Rooks and Clay2019). Detected walking bouts of more than 20 steps were analyzed to extract cadence (Trojaniello et al., Reference Trojaniello, Cereatti, Pelosin, Avanzino, Mirelman, Hausdorff and Della Croce2014).
Patient-reported outcomes
The Late-Life Function and Disability Instrument (LLFDI), a PROs questionnaire for community-dwelling older adults were used to assess both functional limitations (ability to perform discrete physical tasks) and disability (inability to participate in major life tasks and social roles) (Haley et al., Reference Haley, Jette, Coster, Kooyoomjian, Levenson, Heeren and Ashba2002; Jette et al., Reference Jette, Haley, Coster, Kooyoomjian, Levenson, Heeren and Ashba2002; Sakakibara et al., Reference Sakakibara, Routhier, Lavoie and Miller2013). In patients with a balance disorder, small and substantial meaningful changes have been shown by others for the global function scale (2 and 5), and for the basic lower-extremity (3 and 6), advanced lower-extremity (4 and 9), and upper-extremity (4 and 10) subscales (Beauchamp et al., Reference Beauchamp, Ward, Jette and Bean2019). Falls and consultations with a healthcare provider were self-reported by the participants in a calendar diary filled out on paper within the previous 30 days and since the most recent laboratory evaluation for the baseline and follow-up visits, respectively.
Potential confounders
During laboratory visits, the presence of comorbidities was evaluated using a combined Charlson/Elixhauser Comorbidity Index validated for the Quebec population (Simard et al., Reference Simard, Sirois and Candas2018) and frailty was assessed using the Clinical Frailty Scale (Rockwood et al., Reference Rockwood, Song, MacKnight, Bergman, Hogan, McDowell and Mitnitski2005). Participants were asked to report the use of a mobility assistive device, hearing and vision problems, and new physical activities.
Analysis
The implementation of our pilot study was assessed using a 4-category (resources, management, process, and scientific) framework for pilot studies (Thabane et al., Reference Thabane, Ma, Chu, Cheng, Ismaila, Rios, Robson, Thabane, Giangregorio and Goldsmith2010). The process evaluation (primary objective) included: 1) the number of laboratory visits during which the participants completed the 10-meter walk test, SPPB, and mini-BESTest with corresponding refined mobility metrics as well as the LLFDI questionnaire, 2) the number of days out of seven preceding each laboratory visit, for which at least eight hours of wearable devices data were available. The scientific exploratory efficacy analysis (secondary objective) was conducted on the 10-meter walk test, SPPB, mini-BESTest, LLFDI, and real-life mobility, between the baseline visit and either follow-up visit 1 or 2, whichever visit (designated evaluation visit) showed the greatest negative DBI difference from baseline. A safety analysis evaluated the re-prescribing of a stopped medication or the dose increase of a medication with a previous dose decrease. The participant’s characteristics were described using the median, minimal, and maximal values for continuous variables and percentages for categorical variables. The physical function and LLFDI evaluations were presented as mean and standard deviation. The exploratory objective and limited sample size precluded the use of statistical testing. The resources evaluation included the time required to complete the laboratory evaluations while wearable devices’ data capture was assessed for management.
Results
Process. The challenging recruitment, which started in July 2021 (1 patient), improved after the relaxing of pandemic restrictions in the spring of 2022, allowing the recruitment of four participants in 2 months in one PCC.
Scientific. The five participants had a median age of 67 (min: 64, max: 81) at baseline, four were female sex (gender not collected) and were taking a median of 12 (5, 21) medications; had a median DBI score of 1.7 (1.3, 5.4), a frailty score ranging from two to four and four participants had a Charlson-Elixhauser score of 0 (Table 1). The frailty score and Charlson–Elixhauser score did not alter between visits, indicating that the participants’ clinical status remained stable.
Baseline participants’ characteristics (n = 5)

a Unless otherwise stated.
b Excluding natural health products.
Deprescribing
Process. The baseline and two follow-up visits were completed by two individuals. After the first follow-up visit and completion of the deprescribing plan, the other three participants concluded their participation.
Scientific. The median change in DBI score of the five participants between the baseline and evaluation visit was −0.30 (−0.17, −0.85). Represcribing occurred in one participant, with the reintroduction of gabapentin for recurrent pain. The deprescription plan involved a single medication in two participants, two medications in two participants, and six medications for one participant. Of the 12 medications, three were completely stopped and the dose was decreased for the nine others. Apart from cyclobenzaprine (ATC = M03) the adjusted medications were acting on the nervous system (ATC = N) with medications from the N02-analgesics (morphine, gabapentin, pregabalin X2), N03-antiepileptics (topiramate), N05-psycholeptics (quetiapine, bromazepam), and N06A-psychoanaleptics (desipramine, amitriptyline, nortriptyline, trazodone) classes.
Laboratory evaluations (capacity)
Resources. Laboratory visits took approximately two hours to complete. The mean time between baseline and follow-up 1 was 152 ± 96 days. Opportunities to decrease this interval by improving communications between PCC pharmacists and the research team were identified. The baseline visits were conducted in the summer for all participants while the evaluation visits took place in the winter (four participants) and summer (one).
Process. The 10-meter walk test and SPPB were fully completed during the 12 visits. The mini-BESTest was interrupted in one patient during the baseline visit because of fatigue after completing 8 of 14 items. Refined mobility metrics were assessed in all 12 visits during the above three tests.
Scientific. The exploratory analyses of the impact of deprescribing on physical function between the baseline (n = 5) and evaluation visits (n = 5) are presented in Table 2. In the 10-meter walk test, a mean increase in gait speed was seen both at normal (+0.18 m/s) and fast (+0.14 m/s) pace. Three out of five participants showed improvements over the 0.14 m/s major clinical change threshold (Perera et al., Reference Perera, Mody, Woodman and Studenski2006), with +0.35 m/s being the largest difference. Refined mobility metrics for the 10-meter walk test at a normal pace revealed mean improvements in stride length (+0.15 m), cadence (+5.6 steps/min) and double support time (−2.0%). Similar mean improvements were seen at a fast pace (stride length: +0.12 m; cadence: +1.9 steps/min; double support: −1.6%).
Physical function tests – laboratory

a Maximum number of points.
Abbreviations: SD, standard deviation; SPPB, Short Physical Performance Battery.
Scientific. Improvements in the mean global SPPB score (+2.2 points) exceeded the upper substantial clinical change limit of 0.99 to 1.34 points (Perera et al., Reference Perera, Mody, Woodman and Studenski2006). The participants either improved their global SPPB score by 1 point (2 participants), 2 points (2) or 5 points (1). Among the three SPPB tests, the greatest mean improvement was recorded for the chair stand test (+1.4 points). Total duration for the repeated chair stand test improved for all five participants (−7.9 seconds).
Scientific. The global mini-BESTest score changed by a mean of +2.4 points, which was less than the 3.5-point minimum detectable change (Godi et al., Reference Godi, Franchignoni, Caligari, Giordano, Turcato and Nardone2013). The global mini-BESTest score decreased in two participants (−2 and −3), remained stable in one patient, and increased in two participants (+3 and +14). Among the four mini-BESTest dimensions, sensory orientation (+16.7%) showed the most improvement. The sway area improved for all participants (mean sway area change: −0.08 m/s2).
Real-life settings evaluations (performance)
Process. In the 7 days preceding the 12 laboratory visits, ≥ 8 hours of wearable devices data per real-life assessment periods of seven days was available for 7 days (7 visits), 6 days (1 visit), 5 days (2 visits), and 2 days (2 visits). Participants did not voluntarily report problems with wearing a smartwatch on their wrist and ankle for 7 days, although we did not systematically probe this. The mean daily recording times were 790 ± 27 and 788 ± 124 minutes for the baseline and evaluation visits respectively.
Management. The analysis of indoor versus outdoor activities was not carried out since it was not possible to distinguish the time spent inside versus outside for two real-life assessment periods of seven days due to BLE beacon dysfunction.
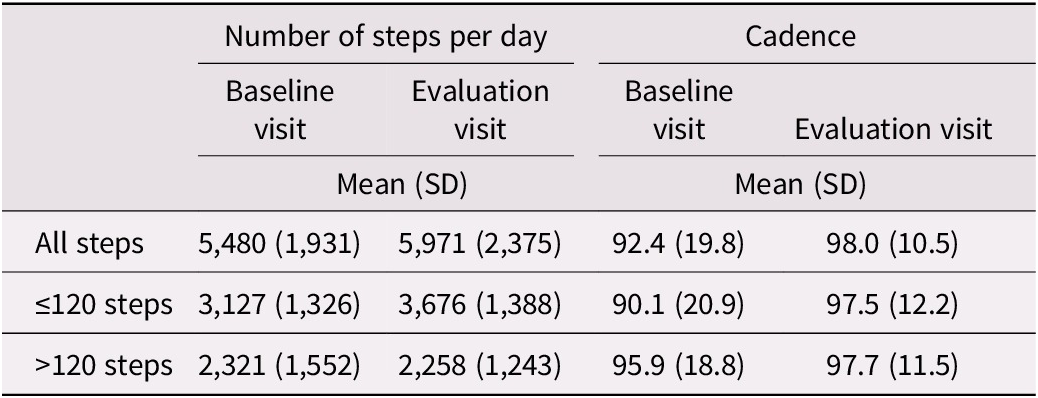
Scientific. The real-life settings evaluations showed an increase in the mean daily number of steps (+491) and mean cadence (+5.6 steps/min) (Table 3).
Real-life settings evaluations

Abbreviation: SD, standard deviation.
Patient-reported outcomes
Process. The entire LLFDI questionnaire was completed during 11 laboratory visits and partially in the other.
Scientific. A mean increase of 4.2 was observed for the LLFDI-function global scale with four participants improving over the +2 small meaningful change threshold (Table 4) (Beauchamp et al., Reference Beauchamp, Ward, Jette and Bean2019). The greatest mean change (+9.1) was noted for the basic lower extremities subscale with four participants improving beyond the +6 cut-off for substantial meaningful change (Beauchamp et al., Reference Beauchamp, Ward, Jette and Bean2019). The subscale’s most improved activities were ‘sitting down in and standing up from low soft couch’, ‘getting into and out of a car’ and ‘stepping on and off a bus’. A mean increase of 11.6 was observed for the disability–limitation global scale (Table 4). A mean change of +15.7 was noted for the instrumental role domain subscale with a greater increase for tasks of ‘visit friends and family in their homes’, ‘travel out of town’ and ‘prepare meals for self’. A similar change (+13.6) was observed for the management role domain subscale with greater improvement for the task of ‘invite people into your home’.
Late life function and disability instrument

a Scaled scores (0–100).
Abbreviations: LLFDI, Late-Life Function and Disability Instrument; SD, standard deviation.
The participants did not report any falls in the 30 days preceding the baseline laboratory evaluation. Two participants reported falling between the baseline and first follow-up, with one participant reporting two falls. One of the three falls occurred 30 days prior to the follow-up visit and led to a physician consultation. Four participants consulted their family physician during the study for issues unrelated to the deprescribing process.
Potential confounders
For all participants, no changes were noted for the comorbidity index, frailty scale, use of mobility assistive device, and hearing/vision problems. Two participants commenced new physical activities through a twice weekly fall reduction program at visit two.
Discussion
Our study showed the feasibility of implementing our comprehensive measurement approach to evaluate the impact of deprescribing anticholinergic and sedative medications on physical function in laboratory and in real-life settings using refined mobility metrics from wearable devices as well as PROs, in preparation for a future study.
In the feasibility evaluation of the processes, we showed a recruitment capacity of two patients per month per PCC after the relaxing of pandemic restrictions. Despite being mostly related to the pandemic, the challenging recruitment during the first 11 months of the study also suggests that numerous recruiting PCCs are necessary. The retention of all patients for the follow-up visits indicates that the evaluation process as a whole was accepted. The 10-meter walk and SPPB tests were fully completed during all laboratory visits and in 11 of 12 visits for the mini-BESTest. Our patient population could be considered healthy with a clinical frailty score of 2 to 4, and a Charlson-Elixhauser score of 0 for 4 participants. They had a median age of 67, were taking a median of 12 medications, and were not using any assistive mobility devices. Among the confounders that could have impacted mobility, frailty, and Charlson–Elixhauser scores remained constant throughout the study while the beginning of new physical activities was reported by two participants. Eleven of the 12 deprescribed drugs were acting on the nervous system (ATC = N), which differs from a study of outpatients of a geriatric day clinic in which 56% of the DBI drugs were acting on the central nervous system (Wouters et al., Reference Wouters, Van Campen, Kuitert, Kikkert, Hilmer, Taxis, Van der Meer and Lamoth2021).
In a scientific exploratory efficacy analysis, we showed that medication deprescribing was associated with clinically meaningful improvements in physical function measured in a laboratory setting with the 10-meter walk test and SPPB. Refined mobility metrics showed improvements in dynamic stability (cadence, stride length, double-support time, 10-meter walk test), anticipatory postural adjustments (repeated chair stand tests, SPPB), and sensory orientation (stance test, mini-BESTest). In real-life settings, we showed an increase in the mean daily number of steps and in mean cadence. Keeping in mind the small sample size, the value of our comprehensive approach is best illustrated by the improvement in the SPPB chair stand test (+1.4 point), broken down and analyzed by refined mobility metrics and corroborated by the change (+9.1) in the LLFDI basic lower extremities subscale with greater improvements for activities that involve sitting and standing. None of the participants reported a fall in the 30 days prior to the baseline visit while two participants reported three falls in the longer period between the baseline and first follow-up visit. This unexpected increase in falls warrants further evaluation. Our ongoing trial with more participants pursues this evaluation using a daily calendar diary.
Wearable devices have recently been introduced to assess the impact of medication use on the mobility of older adults. In a cross-sectional analysis of outpatients of a geriatric day clinic, a DBI score >1 was associated with poorer function on the regularity, pace, and complexity dimensions derived from accelerometer-based dynamic gait parameters (Wouters et al., Reference Wouters, Van Campen, Kuitert, Kikkert, Hilmer, Taxis, Van der Meer and Lamoth2021). In a longitudinal cohort of community-dwelling older adults, anticholinergic use was associated, at 2 to 3 years of follow-up, with a greater decline in activity measured over seven days by accelerometer (Clarke et al., Reference Clarke, Sniehotta, Vadiveloo, Donnan and Witham2018). In residents of long-term care centers, increased sedative or anticholinergic burden was associated with an increase in sedentary time measured by accelerometer over a 24 hour period (Lim et al., Reference Lim, Dumuid, Parfitt, Stanford, Post, Bilton, Kalisch Ellett, Pratt and Roughead2023).
Strengths and limitations
Our study’s main strength is its comprehensive assessment of physical function using wearable devices in both laboratory and real-life settings as well as PROs. The use of a longitudinal design and the measurement of multiple potential confounders are essential elements when the clinical equipoise favors deprescribing and precludes random assignment to a control group. Compared to other anticholinergic scales, the DBI includes sedative medications and takes into account the dosages of medications (Hilmer et al., Reference Hilmer, Mager, Simonsick, Cao, Ling, Windham, Harris, Hanlon, Rubin, Shorr, Bauer and Abernethy2007). While pre-clinical studies had demonstrated improvements in function by deprescribing a polypharmacy regimen with high DBI (Mach et al., Reference Mach, Gemikonakli, Logan, Vander Wyk, Allore, Ekambareshwar, Kane, Howlett, de Cabo, Le Couteur and Hilmer2021), this is the first human interventional study to demonstrate an improvement in physical function when reducing DBI. While deprescribing has been shown to be safe in terms of mortality and hospitalization, there is a paucity of evidence on the effects of deprescribing on function (Wu et al., Reference Wu, Kouladjian O’Donnell, Fujita, Masnoon and Hilmer2021). The comprehensive methodology applied in this study provides a novel method to generate this evidence.
The main limitation of our study was the limited sample which consisted of 80% female participants, with well-controlled health conditions who did not require an assistive walking device. The recruitment over a 12-month period in a complex pandemic context precluded data collection on the number of screened patients and reasons for exclusion. For two real-life assessment periods of seven days, valid data was limited to two days and the breakdown of time spent indoor/outdoor could not be determined. Two participants started a fall reduction program which could lead to improved physical function beyond the reduction of DBI medications.
Future developments
This study showed trends in outcomes that suggested a larger study powered to detect statistically significant and clinically important changes was worthwhile. Our group’s subsequent, ongoing study (clinicaltrials.gov NCT05735379) has incorporated important learnings from this pilot study. The recruitment is conducted at six PCCs. A reduction of 0.3 in the DBI score is targeted considering the clinically significant improvements in physical function seen in this trial with a DBI decrease less than the target of 0.5. The Brief Pain Inventory is added to assess how deprescribing can impact pain, a potential intermediate variable between the DBI score and physical function. An increase in home wearable device use to 12 days with daily remote monitoring and patient feedback will increase the number of days with valid data.
Conclusion
Our study showed the feasibility of our comprehensive evaluation in the laboratory, in real-life settings, and by PROs of the impact of reducing anticholinergic and sedative medications on the physical function of community-dwelling older adults. The exploratory efficacy analysis conducted on this limited sample highlights the potential of our approach not only to measure but also to better understand how these medications affect the physical function of older adults. This novel methodology can provide critical evidence on the effects of deprescribing on function, which is important for research, practice, and policy.
Acknowledgements
The authors would like to thank the participants who gave their time for the study.
Funding support
This work was supported by the Research Center on Aging of the CIUSSS de l’Estrie-CHUS (108272).