Highlights
-
• Patients with one or more features of SOD triad have a wide spectrum of neuropathological findings, resulting mainly from destructive processes in specific brain regions (optic nerves/chiasm, pituitary gland and nearby midline structures).
-
• SOD is not a single disease entity.
-
• The most frequent cause of death is a respiratory illness.
Introduction
In 1956, Georges de Morsier described “la dysplasie septo-optique” (septo-optic dysplasia), a condition characterized mainly by agenesis of the septum pellucidum (SP) and malformation of the optic tract, based on 11 prior publications and his own detailed clinical and neuropathological examination of 1 case. Reference De Morsier1 He concluded that there were often other malformations including neural tube defects and periventricular heterotopia. He did not offer a pathological explanation. In the 1970s, the clinical syndrome was linked to pituitary and hypothalamic dysfunction. Reference Hoyt, Kaplan, Grumbach and Glaser2,Reference LaFranchi3 In one autopsy report, a 19-month-old infant with small optic discs on funduscopy, diabetes insipidus, growth and adrenocortical hormones insufficiency and absent SP was found to have an absent posterior pituitary gland. Reference Benoit Gonin, David and Feit4 Another autopsy showed microcephaly, destruction of the anterior corpus callosum (CC), heterotopic nodules, absent olfactory tracts and atrophic optic nerves. Reference Patel, Tze, Crichton, McCormick, Robinson and Dolman5 The phenotype was quite variable, and the pathogenesis was unknown. With the advent of CT scanning, large clinical series of septo-optic-pituitary dysplasia (SOD) were published in the 1980s. Owing to the presence of cerebral atrophy and focal lesions, a time-specific insult was suspected to occur in utero, Reference Kuban, Teele and Wallman6 and vascular or infectious insults were proposed. Reference Margalith, Jan, McCormick, Tze and Lapointe7–Reference Brodsky9 Lubinsky in particular emphasized that SOD is part of a vascular disruption sequence. Reference Lubinsky10–Reference Brodsky12
It is not clear if isolated optic nerve hypoplasia (ONH) or isolated absence of the SP represents part of a spectrum. Reference Acers13–Reference Shuman and Leech15 Partial hypothalamic dysfunction, manifesting with pubertal or body weight disorders without hypopituitarism, has been suspected in some cases with isolated ONH, and such cases may represent one end of the SOD spectrum. Reference Cerbone, Güemes, Wade, Improda and Dattani16 Other autopsy cases showed destructive lesions in the hypothalamic region, Reference Kewitz, Girard and Probst17–Reference Brodsky, Conte, Taylor, Hoyt and Mrak19 while some showed SOD in association with more complex malformations. Reference Michaud, Mizrahi and Urich20 Some authors in the late 1980s recommended against the conflation of SOD and SOD that is associated with destructive lesions (e.g., schizencephaly or porencephaly), suggesting that the two conditions are separate entities and that ONH in the latter cases occurs secondary to schizencephaly. Reference Osborn and Byrd21
Rarely, the possibility of inheritability has been considered because some forms of neuronal migration disorders and SOD may occur in more than one family member. Reference Ahn, Ahn and Han22 Genetic abnormalities have been identified only in a small minority of cases. The first was a mutation in the HESX1 homeodomain, which is identified in <1% of SOD cases, Reference McNay, Turton and Kelberman23 with SOX3, SOX2 and OTX2 subsequently identified even less often. Reference Pasca, Politano and Morelli24
The aims of this study were to describe the spectrum of neuropathological abnormalities in patients with isolated, partial or all classic features of the SOD triad, speculate on the etiology of their findings and ascertain the causes of death.
Method
Study design
A retrospective review of autopsy reports concerning patients with isolated, partial or all classic features of the SOD triad. Ethics approval was granted by the Health Research Ethics Board, University of Manitoba.
Setting
Autopsies of patients with one or more SOD features were performed at the Health Sciences Center or Winnipeg Children’s Hospital, the only tertiary pediatric hospital in the province of Manitoba, Canada.
Case ascertainment and study period
The full text of autopsy records held in the Department of Pathology at Health Sciences Center (Shared Health Manitoba) can be searched beginning January 1977 for pediatric cases and January 1996 for adult cases. From these start dates and until December 2024, a search was conducted for various terms related to ONH and SOD including “septo-optic,” “agenesis septum,” “pellucidum agenesis,” “optic atrophy,” “optic nerve hypoplasia,” “pituitary atrophy” and “hypopituitarism.” Seventy reports were screened, and 20 were reviewed in detail. An additional case not found in the initial search of the autopsy database was added later. Thus, a total of 21 cases were included in this study. Attempts to review the macroscopic findings were not always successful because photographs could not be located.
Case definition
We included cases with at least one or more of the following features: ONH, hypopituitarism/small pituitary gland and absent SP/abnormal CC. SOD was diagnosed clinically when patients with ONH had any one of: hypopituitarism/small pituitary gland, absent SP or abnormalities in the CC. ONH was diagnosed on clinical examination when a small optic disc on funduscopy was reported by one of three pediatric ophthalmologists who are familiar with this disorder. The optic disc in ONH is small in size and may be pale in appearance. The double ring sign may be seen. Cases typically presented with poor visual tracking, early-onset strabismus or nystagmus beyond the age of 4 months or as part of a workup for multiple congenital abnormalities that were noted clinically after birth or on a prenatal or postnatal brain MRI.
Study variables
The demographics included birth history, age at death, sex, clinical features and neuroimaging abnormalities. Pertinent findings on brain autopsy were extracted with emphasis on the following structures: optic nerves, optic chiasm, pituitary stalk and gland, hypothalamus, SP, CC, cerebral cortex, olfactory bulbs and tracts and cerebellum. Presumed pathogenesis was inferred from the complete neuropathological and autopsy examination findings. Cases were divided into six groups based on the presence of one or more of the classic SOD triad. The diagnosis of SOD required the presence of ONH and was the basis of this classification. The six groups were (A) SOD (ONH with hypopituitarism and absent SP or abnormal CC), (B) SOD (ONH and hypopituitarism/small pituitary gland with normal SP, (C) SOD (ONH and absent SP/or abnormal CC with normal pituitary gland), (D) ONH with normal pituitary and SP – not SOD, (E) abnormal pituitary gland and absent SP/or abnormal CC without ONH – not SOD and (F) absent SP without ONH and normal pituitary gland – not SOD.
Results
Of the 21 cases identified, 14 were females, and 7 were males. Median age at death was 2.3 years (25th–75th percentiles: 0.3–9.2 years, minimum: 5 mins, maximum: 50.0 years). One case was liveborn at 25 weeks’ gestation, and two cases were born at 36 weeks’ gestation. A summary of the clinical, neuroimaging findings and other characteristics of the cohort is displayed in Table 1.
Table 1. Demographic and autopsy details of patients with optic nerve hypoplasia and septo-optic-pituitary dysplasia spectrum

Note: CC = corpus callosum; ONH = optic nerve hypoplasia; SOD = septo-optic-pituitary dysplasia; SP = septum pellucidum; VSD = ventricular septal defect.
Ten cases had ONH with either (i) small pituitary gland/hypopituitarism (five cases) or (ii) absent SP/abnormal CC (two cases) or (iii) both (three cases). These 10 cases had SOD (Table 1, categories A, B and C). Eight of these 10 cases were considered a consequence of disruptions of brain development, while 2 were thought to be primary SOD (cases 3 and 4).
Three cases had ONH with a normal pituitary gland and SP. Three cases had hypopituitarism/small pituitary gland and absent SP/or abnormal CC without ONH, while five cases had an absent SP with neither ONH nor small pituitary gland or hypopituitarism.
Pregnancy: Infrequently, there was a maternal history of alcohol (n = 3) or cocaine use with maternal hypertension during pregnancy (n = 1) or a maternal history of insulin-dependent diabetes mellitus (n = 1). One fetus had amniotic bands and exencephaly with severe brain malformation (Table 1).
Comorbidities: Most patients had at least one comorbidity in addition to visual acuity impairment including hypopituitarism, developmental delay, seizures/epilepsy and cerebral palsy. Three cases had microcephaly. Three cases had shunted hydrocephalus (one with a large complex arachnoid cyst and two with congenital aqueduct stenosis presumed to be related to an infection in utero). Other congenital abnormalities included coloboma, microphthalmia, atrial or ventricular septal cardiac defect, vertebral anomalies and cleft lip and palate (Table 1).
Neuroimaging: Neuroimaging revealed small optic nerves/optic chiasm, small pituitary gland and absent SP in several cases, while abnormalities in the CC, large arachnoid cysts, focal ischemic lesions of the cerebrum (e.g., schizencephaly), neuronal heterotopia, simple gyral pattern, Rathke cleft cyst and cerebellar vermis hypoplasia were noted in only a few cases (Table 1).
Causes of death: The causes of death were pneumonia (8), cardiac arrest (3), seizures (2), sepsis and pulmonary hemorrhage (1), hypopituitarism and cardiac failure (1), gastrointestinal illness and dehydration (1), severe failure to thrive (1), severe brain malformation (1) and termination of pregnancy at 25 weeks’ gestation (1). Co-sleeping as a risk factor in sudden unexpected death was reported in three cases. Table 1 shows the details.
Neuropathology: There was a range of forebrain abnormalities, other brain anomalies and, in a few cases, extracranial abnormalities (Table 1). Some of these abnormalities were old and most likely occurred in utero in early gestation, while others occurred around the time of death (Table 1). Figures 1–4 show examples of the various abnormalities encountered in four of our cases.

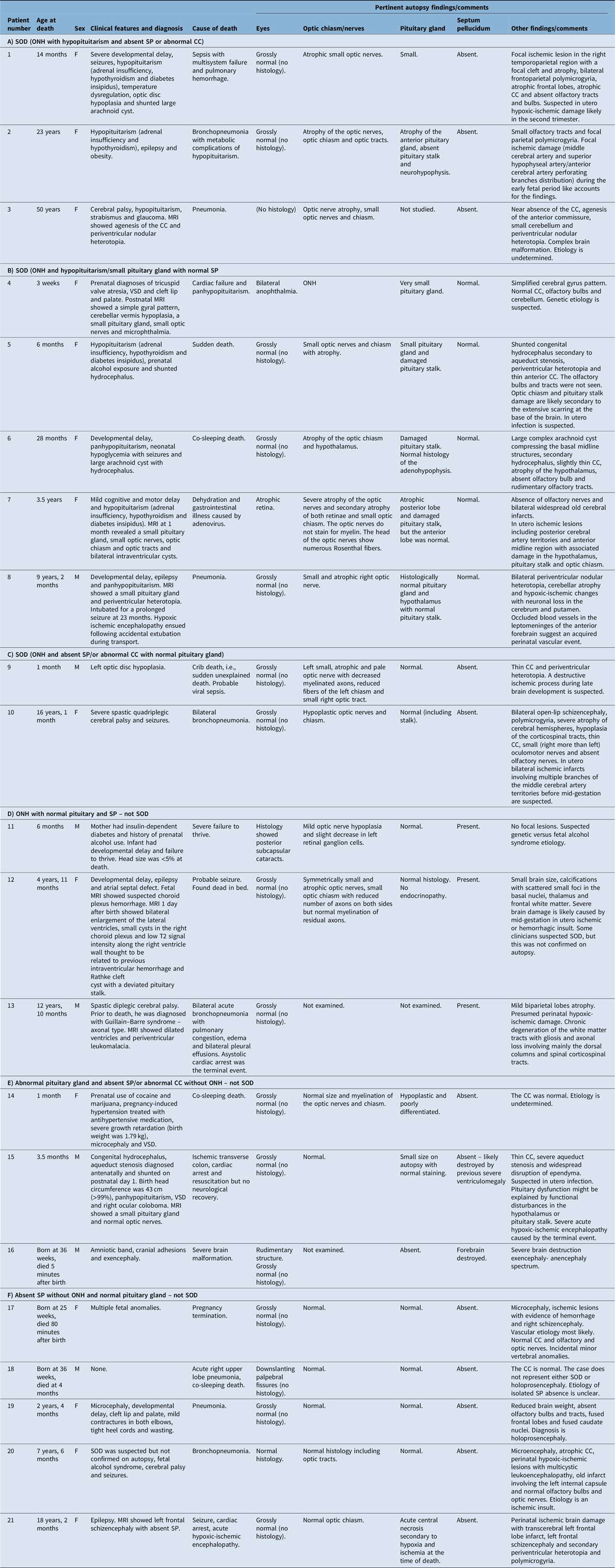
Figure 1. Case 2. (A) Photograph showing the base of the forebrain with severe atrophy of the optic chiasm (arrow) and normal olfactory tracts. (B) Photomicrograph showing atrophic prechiasmatic optic nerves (arrows; approximately one-third of the normal size). Eosin (pink) and solochrome cyanin (blue-myelin) stains; original magnification 12.5×. The postchiasmatic optic tracts were extremely atrophic (estimated one-tenth of normal size; not shown).

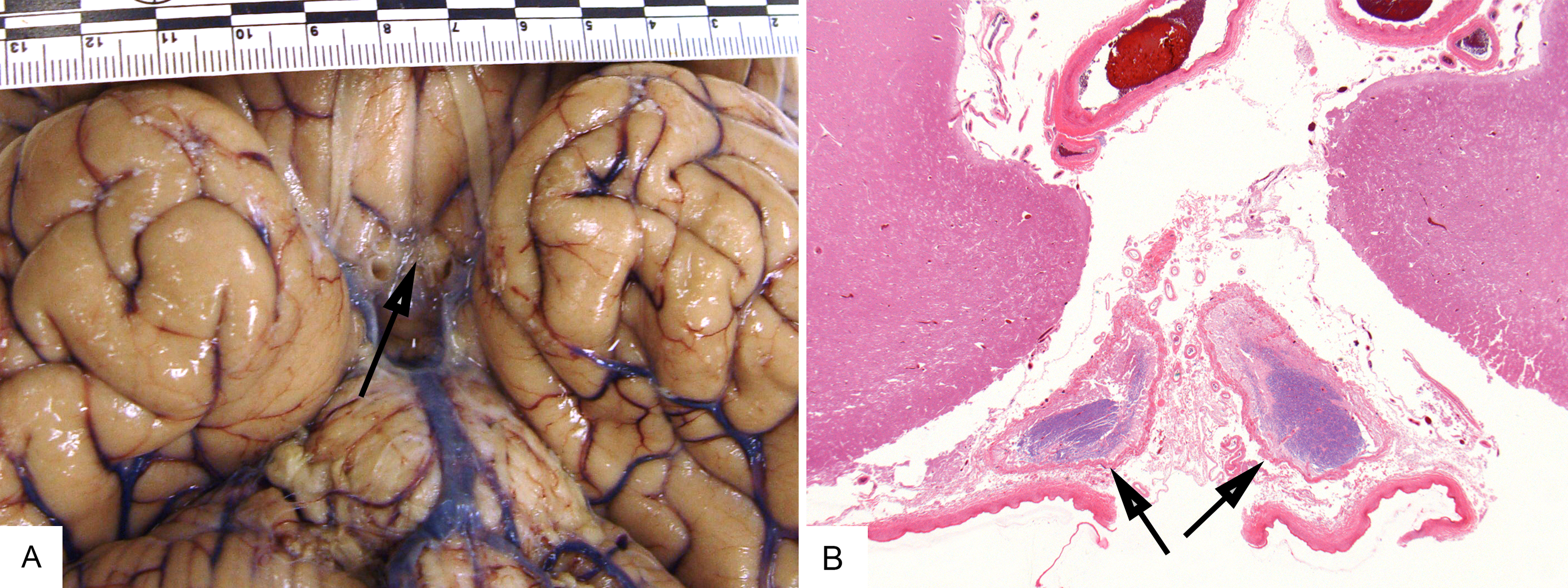
Figure 2. Case 5. Photograph showing the base of the forebrain with severe atrophy of the optic chiasm (red arrow), cavity in the hypothalamic region (black arrow) and absent olfactory tracts. These findings are all secondary to a focally destructive vascular lesion.

Figure 3. Case 6. (A) Coronal T-2 weighted magnetic resonance image performed on day 1 of life showing a large fluid-filled cyst occupying the frontal and middle cranial fossae. After the cyst was shunted, the brain expanded but remained distorted and asymmetric. (B) Photograph showing the base of the forebrain (retracted away from the skull base) with atrophic optic nerves (arrows). (C) Photograph showing the base of the fixed brain. The brainstem and cerebellum are relatively normal. The forebrain is rotated with the interhemispheric fissure deflected to the right side. Pachygyria of the temporal lobes is evident (red arrows). The black arrow shows the location of the optic chiasm.

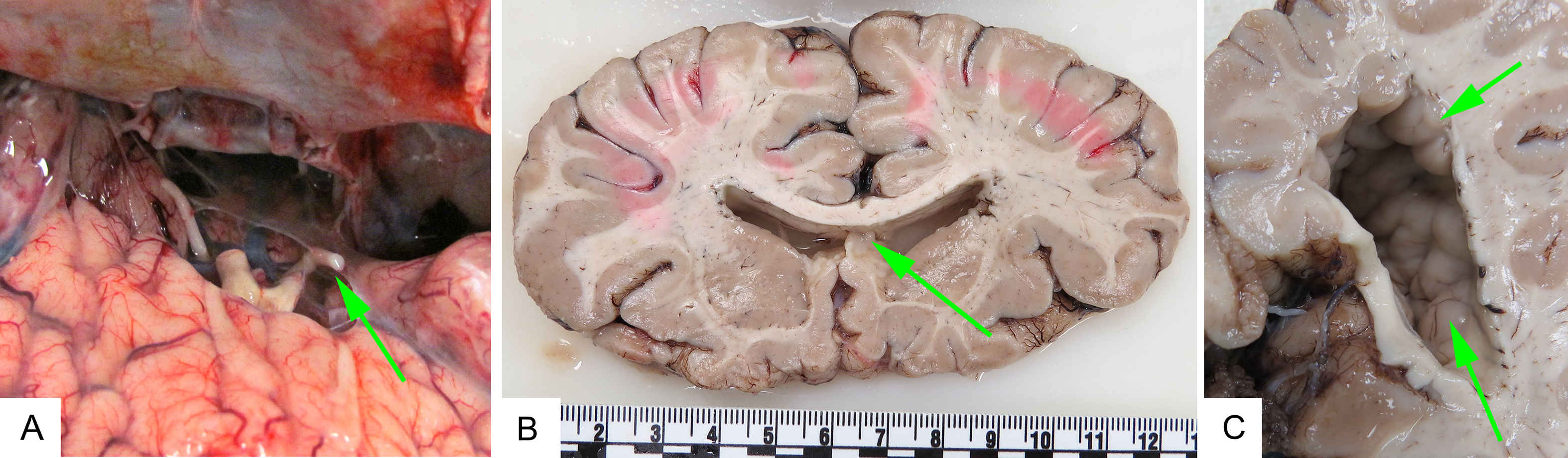
Figure 4. Case 8. (A) Photograph showing the base of the forebrain (retracted away from the skull base) with atrophic right optic nerve (green arrow). (B) Coronal slice through the frontal lobes showing absent septum pellucidum (green arrow). (C) Coronal slice through the right parietal lobe showing lumpy surface due to abundant periventricular heterotopia (green arrows).
Eyes – The eyes were grossly normal in most cases. Bilateral anophthalmia was noted in one case. Histology of the eyes was available in three cases. In two cases, the retina was atrophic with loss of retinal ganglion cells (bilateral in one and unilateral in another case), with posterior subcapsular cataracts noted in one of these two cases. In the third case, retinal histology was normal.
Optic nerves, chiasm and optic tracts – The optic nerve(s) and chiasm were atrophic in 12 of the 13 cases who had the clinical diagnosis of ONH or SOD (the optic nerves were not examined in 1 case). In 2 of 12 cases, the optic tracts were also atrophic.
Pituitary gland and stalk – Seven cases had a small pituitary gland, and three of seven had damaged pituitary stalks. One case had pituitary stalk damage and atrophy of the hypothalamus without pituitary gland abnormalities. In one case with exencephaly, the pituitary gland was absent. Acute pituitary necrosis was seen in one case who had hypoxic-ischemic encephalopathy following cardiac arrest before her death.
Septum pellucidum – The SP was absent in 12 cases. In 1 of 12 cases, severe ventriculomegaly was suspected to have destroyed the SP.
Corpus callosum – The CC was thin in seven cases (in two likely from severe ventriculomegaly) and was absent in one case.
Olfactory bulbs and tracts – The olfactory bulbs and tracts were thin or absent in seven cases.
In addition, one case (case 16) with severe brain malformation, the forebrain destruction encompassed the SP, CC and olfactory bulbs and tracts.
Cerebellum – A small cerebellum with an irregular left hemisphere was noted in one case.
Neuronal migration disorders – Neuronal migration abnormalities, some as a component of a primary malformative process and others secondary to destructive lesions, were seen. These included periventricular heterotopia in five, polymicrogyria in four and simplified cerebral gyrus pattern in one. Furthermore, one case (case 19) had holoprosencephaly with fused frontal lobes and caudate nuclei.
Ischemic lesions – In eight cases, evidence for focal or widespread ischemic lesions was noted with or without calcifications, including schizencephaly in three.
Hippocampus – Neuronal loss was reported in one case. No other abnormalities were reported.
Etiology: Autopsy findings suggestive of an acquired vascular event causing hemorrhagic or hypoxic-ischemic damage in utero were found in 12 (including one who had amniotic bands that caused head/brain damage). These involved the territories supplied by the middle cerebral artery or its branches, superior hypophyseal artery, anterior cerebral artery perforating branches and posterior cerebral arteries. In two cases that had aqueduct stenosis and severe hydrocephalus, the primary pathology was suspected to be an infection in utero. One case with severe hydrocephalus had a large complex arachnoid cyst that deformed the basal forebrain. One had holoprosencephaly, and two had features suggestive of an undefined genetic disorder. In three cases, the etiology is unknown (Table 1).
Discussion
We report a wide variety of neuropathological abnormalities in our autopsy series of mostly pediatric patients with isolated, partial or the classical triad of SOD. Blackburn et al. (2021) in their autopsy study of four SOD cases and their review of other published single-case autopsy studies also reported a host of additional neuropathological findings including malformations involving structures outside the forebrain and secondary changes beyond the triad of ONH, hypothalamic/pituitary gland dysfunction and midline SP or CC abnormalities. Reference Blackburn, Thomas, Hughes and Pierson25 However, their study did not describe retinal findings. In our study, the eyes were grossly normal in most cases; detailed retinal histology was only done in three cases, two of which had a paucity of retinal ganglion cells, a finding that has been reported in prior studies. Reference Kewitz, Girard and Probst17,Reference Roessmann, Velasco, Small and Hori18,Reference Jethwani, Mahadevan, Ramamurthy, Mangala and Shankar26
We found no specific disease entity that defined SOD pathologically on autopsy. A variety of different pathologies can affect the optic chiasm, septal and pituitary regions, as described in previous studies. Reference Patel, Tze, Crichton, McCormick, Robinson and Dolman5,Reference Roessmann, Velasco, Small and Hori18–Reference Michaud, Mizrahi and Urich20,Reference Blackburn, Thomas, Hughes and Pierson25–Reference Gilbert, Scott and Byard27 Most are destructive vascular lesions of uncertain origin. Rarely and as previously reported, an amniotic band can cause secondary vascular damage in some cases of SOD. Reference Stevens and Dobyns28
Isolated agenesis of the SP has no obvious pathogenesis. The SP develops early in the middle trimester as the medial walls of the enlarging lateral ventricle frontal horns fuse. Reference Rakic and Yakovlev29 The septum can be destroyed by severe ventriculomegaly as the CC elevates. From a functional standpoint, the absent SP itself has no clinical consequences. Reference Dahl, Kristoffersen Wiberg, Teär Fahnehjelm, Sävendahl and Wickström30 Its absence in SOD is simply an easily identifiable marker of anterior midline brain damage. Reference Brodsky9
Damage to the hypothalamus, pituitary stalk or gland and optic nerves/chiasm define the key clinical features of SOD. Reference Margalith, Jan, McCormick, Tze and Lapointe7,Reference Margalith, Tze and Jan8,Reference Cerbone, Güemes, Wade, Improda and Dattani16 In attempting to explain the full spectrum of the clinical and neuropathological findings in SOD, Lubinsky and Encha-Razavi in 2022 Reference Lubinsky and Encha-Razavi31 proposed that proximal anterior cerebral artery branch disruptions cause isolated ONH most often, with isolated SP abnormalities occurring less frequently. Small regional vascular disruptions can involve the pituitary, while larger disruptions may extend and cause cortical infarctions (and secondary cortical changes like polymicrogyria). The authors suggested that isolated pituitary abnormalities or cortical defects are separate from SOD and that micro- or anophthalmia with ONH is not part of SOD. They proposed that vascular disruptions produce toxic by-products that are transported in the CSF, thus affecting the developing brain at a distance from the primary location of the disruption. Reference Lubinsky and Encha-Razavi31,Reference Lubinsky32 These postulated toxic by-products might then affect development adversely, causing developmental, cognitive and evolving endocrine dysfunction reported in SOD cases Reference Margalith, Jan, McCormick, Tze and Lapointe7,Reference Margalith, Tze and Jan8,Reference Dahl, Kristoffersen Wiberg, Teär Fahnehjelm, Sävendahl and Wickström30,Reference Salman, Ruth, Yogendran and Lix33 and/or the structural abnormalities including thin CC, ventriculomegaly and olfactory bulbs/tracts absence documented in this investigation and in previous studies. Reference Patel, Tze, Crichton, McCormick, Robinson and Dolman5,Reference Roessmann, Velasco, Small and Hori18–Reference Michaud, Mizrahi and Urich20,Reference Blackburn, Thomas, Hughes and Pierson25,Reference Salman, Hossain and Rozovsky34
Studies documenting mortality rates in patients with ONH/SOD are sparse. The crude mortality rate in one study was 3.7 per 1000 person-years in patients with ONH/SOD during a median follow-up period of 12.0 years. Reference Salman, Ruth, Yogendran and Lix35 In our study, most deaths occurred in early childhood. The causes of death in SOD cases vary and are usually multifactorial. Most commonly, death was caused by pneumonia. Respiratory illness is the most common cause of acute hospital admission in SOD. Reference Salman, Ruth, Yogendran and Lix36 Patients who have a neurological disease such as developmental delay, intellectual disability, cerebral palsy and seizures, as many patients with SOD do, Reference Dahl, Kristoffersen Wiberg, Teär Fahnehjelm, Sävendahl and Wickström30,Reference Salman, Ruth, Yogendran and Lix33 are at a specially higher risk of death from respiratory compromise, for example, aspiration and pneumonia. Reference Reddihough, Baikie and Walstab37,Reference OLeary, Cooper and Hughes-McCormack38 Sudden death occurred in two of our SOD cases as reported by others in patients with SOD. Reference Kewitz, Girard and Probst17,Reference Brodsky, Conte, Taylor, Hoyt and Mrak19,Reference Gilbert, Scott and Byard27,Reference Bonasoni, Reyes, Cromwell, Halliday, Taylor and Chiasson39 Seizures may be implicated in causing death in patients with SOD; Reference Bonasoni, Reyes, Cromwell, Halliday, Taylor and Chiasson39 however, none of our SOD cases died from seizures. Technically, case 12 is the only sudden unexpected death in epilepsy (SUDEP). Although case 21 death is seizure-related, it is not strictly speaking SUDEP. Neither of these two cases had SOD. In the study by Bonasoni et al. (2014), there was no acute anatomic pathology that could explain the sudden death in six of their seven cases who died at home. They postulated that patients with acute symptoms of viral illness develop an acute adrenal crisis, which may cause sudden death. They also highlighted bed-sharing with an adult as a potential cause of overlaying and positional asphyxia. Reference Bonasoni, Reyes, Cromwell, Halliday, Taylor and Chiasson39 Co-sleeping was reported in three of our cases.
Other uncommon causes of death in SOD in our study include complications caused by hypopituitarism, as documented previously. Reference Margalith, Jan, McCormick, Tze and Lapointe7,Reference Margalith, Tze and Jan8,Reference Dahl, Kristoffersen Wiberg, Teär Fahnehjelm, Sävendahl and Wickström30,Reference Rushworth, Torpy, Stratakis and Falhammar40,Reference Rushworth, Chrisp, Dean, Falhammar and Torpy41 Hypopituitarism in our cases may have been a contributing factor that increased vulnerability in the context of heart failure (case 4), pneumonia (cases 2 and 8) or sepsis (case 7) and possibly in case 5 since there was no acute anatomic explanation for the death.
Study limitations
Studies based on autopsy reports are inherently subject to some limitations. For example, missing information because some patients in this cohort were not followed or were only followed briefly or their medical chart was not available. Concordance between clinical diagnosis of SOD syndrome and pathophysiology seemed suboptimal, with some clinicians using the diagnostic label SOD incorrectly. Neuroimaging studies were not available in some cases; therefore, neuropathological findings could not be correlated with neuroimaging findings in all cases. Our study of 21 autopsy cases including 10 with SOD is small; however, to the best of our knowledge, this is the largest autopsy study of SOD.
Modern genetic investigations, for example, whole exome or genome sequencing, were not performed, and other genetic test results were not available to us. However, causative mutations are found in only a very small minority of SOD cases. Reference McNay, Turton and Kelberman23
Perhaps most importantly, autopsy cases by definition are biased toward the most severe spectrum of the disease. There was variation with regard to the extent of neuropathological details available in the autopsy reports because they were performed by more than one neuropathologist over a time period spanning a few decades. These limitations may affect the general implications of our data for all patients with one or more features of the SOD triad.
Conclusions
Based on autopsy reports of patients with one or more features of the SOD triad, our study revealed a wide spectrum of neuropathological features that appear to be the result of a destructive (usually vascular lesion) or, less commonly, impaired developmental processes. Our findings suggest that SOD is not a single disease entity but represents a syndrome with ONH and one or more of: hypothalamic-pituitary dysfunction and absent SP or abnormal CC. We consider ONH to be an essential part of SOD.
These lesions affect the structure and/or function of the optic nerves/chiasm and hypothalamus/pituitary gland or nearby midline structures including the SP, CC and olfactory bulbs and tracts. When the pathological process is more widespread, then damage to other cortical structures occurs, resulting in other clinical features, for example, hydrocephalus, developmental delay and cerebral palsy.
Data availability statement
The data used for this study were obtained from the autopsy reports and are available in Table 1.
Author contributions
MDB conceptualized and initiated the study. He extracted part of the data for Table 1, provided the figures and their legends and wrote parts of the manuscript. He aided in the interpretation of the study’s findings. He reviewed and edited subsequent drafts of the manuscript. MSS extracted further data from the autopsy reports and organized/categorized the data in Table 1. He aided in the interpretation of results, wrote the first draft and edited subsequent drafts of the manuscript. Both authors approved the final version of the manuscript.
Funding statement
None.
Competing interests
Michael Salman declares no competing interests or conflicts of interest. Marc del Bigio declares the following: Royalties for sales of the textbook – Perinatal Neuropathology (personal) from Cambridge University Press, fees for reporting of autopsy case from Directeur des poursuites criminelles et pènales (DPCP) Quebec as payment for expert testimony and waived registration fees for an invited speaker at the American Association of Neuropathologists 2025 annual meeting.
Ethical statement
Ethical approval was obtained from the University of Manitoba’s Health Research Ethics Board.




Target article
Septo-Optic-Pituitary Dysplasia Is a Clinical Syndrome, Not a Neuropathological Entity: An Autopsy-Based Study
Related commentaries (1)
Reviewer Comment on Salman et al. “Septo-Optic-Pituitary Dysplasia Is a Clinical Syndrome, Not a Neuropathological Entity: An Autopsy-Based Study”