Psoriasis is a chronic inflammatory skin disease with systemic complications that significantly impact on patients’ quality of life and mental health(Reference Greb, Goldminz and Elder1,Reference Armstrong and Read2) . Globally, psoriasis affects about 2 % of the population, with chronic plaque psoriasis accounting for 90 % of cases(Reference Griffiths and Barker3,Reference Christophers4) . While skin manifestations are usually the initial symptoms of psoriasis, increasing clinical and epidemiological evidence suggests a link between psoriasis and cardiovascular diseases such as stroke, coronary artery disease and peripheral vascular disease(Reference Miller, Ellervik and Yazdanyar5). Given the systemic nature of psoriasis and its links to other conditions, various biological factors have been explored. Among these, folate has garnered particular attention due to its essential role in cellular function and potential impact on the disease.
Folate is an essential water-soluble vitamin that plays a crucial role in maintaining one-carbon metabolism, and deficiency in folate can negatively affect development and the nervous system(Reference Lyon, Strippoli and Fang6). In the context of psoriasis treatment, methotrexate—a commonly used medication(Reference Coates, Merola and Grieb7,Reference Lopez-Olivo, Siddhanamatha and Shea8) —can inhibit folate metabolism, potentially worsening folate deficiency and leading to adverse effects(Reference Mazaud and Fardet9). To mitigate these effects, folate supplementation is commonly recommended during methotrexate therapy(Reference Baran, Batycka-Baran and Zychowska10,Reference Al-Dabagh, Davis and Kinney11) , underscoring the critical role of folate in psoriasis management.
Despite folate’s recognised importance, studies examining serum folate levels in psoriasis patients have yielded inconsistent results. Some studies have reported lower serum folate levels in psoriasis patients compared with healthy controls(Reference Malerba, Gisondi and Radaeli12,Reference Fry, Macdonald and Almeyda13) , while others have found no significant difference(Reference Weger, Hofer and Stanger14–Reference Cakmak, Gül and Kiliç16). These inconsistencies may be due to variations in study design, sample selection and the matching of control groups based on factors such as gender, age, race, comorbidities and medication use. Additionally, the lower folate levels in psoriasis patients could be attributed to reverse causality, as methotrexate treatment inhibits folate metabolism(Reference Lambie and Johnson17). A meta-analysis found that folate levels in psoriasis patients were lower than those in non-psoriasis patients(Reference Tsai, Yen and Huang18). However, even after conducting subgroup analyses based on psoriasis type, the presence of a healthy control group and exposure to methotrexate, some potential confounding biases could not be entirely avoided.
Given these inconsistencies and the limitations of traditional observational study, Mendelian randomisation (MR) provides a robust approach to address the gaps in understanding the causal relationship between folate levels and psoriasis risk. MR uses genetic variations, particularly single nucleotide polymorphisms (SNPs), as instrumental variables (IVs) to infer potential causal relationships between exposures and outcomes(Reference Emdin, Khera and Kathiresan19). This method offers more reliable causal inferences than traditional epidemiological studies, especially in the presence of confounding factors and reverse causality(Reference Sheehan, Didelez and Burton20).
Building on the above background, we designed this study to systematically explore the relationship between serum folate levels and psoriasis. First, we conducted an observational analysis using data from the National Health and Nutrition Examination Survey (NHANES) to examine the association between serum folate levels and psoriasis risk. Subsequently, we conducted a two-sample MR analysis to investigate whether genetically predicted serum folate levels have a causal effect on psoriasis onset.
Methods
Observational study
Data selection and study design
The study was based on the NHANES database. All NHANES protocols were approved by the NCHS Research Ethics Review Board, and informed consent was obtained from all participants. The detailed design and data from the NHANES study are publicly available at the following website: (https://www.cdc.gov/nchs/nhanes/). We extracted data from five NHANES cycles (2003–2004, 2005–2006, 2009–2010, 2011–2012 and 2013–2014). The exclusion criteria were as follows: (1) missing psoriasis diagnosis data, (2) age under 20 years, (3) missing serum folate data, (4) pregnant women and (5) missing values for other covariates. A total of 16 773 participants were included in the final analysis (online Supplementary Figure 1). We selected the Mobile Examination Center weights, calculated as 1/5 × WTMEC2YR.
Assessment of serum folate
From 2003 to 2006, serum folate was measured using the Bio-Rad Laboratories ‘Quantaphase II Folate’ radioassay kit. In 2009–2010, it was measured by microbiological assay and from 2011 to 2014 by isotope-dilution high-performance liquid chromatography coupled to tandem mass spectrometry. Since previous studies have suggested that the microbiologic assay is the most accurate among these methods, we used fractional polynomial regression to convert serum folate values from 2003 to 2006 to equivalent values from 2009 to 2014(Reference Liu, Geng and Wan21). Detailed specimen collection and processing procedures are outlined in the NHANES Laboratory Procedures Manual (https://www.cdc.gov/nchs/nhanes/). Vials were stored frozen (–20°C) until shipment to the National Center for Environmental Health for testing.
Assessment of psoriasis
Participants were defined as having psoriasis if they answered ‘Yes’ to either of the following questions: ‘Have you ever been told by a health care provider that you had psoriasis?’ or ‘Have you ever been told by a doctor or other health care professional that you had psoriasis (sore-eye-asis)?’ Participants who refused to answer or responded with ‘Don’t know’ were excluded(Reference Ma, Cui and Cai22).
Covariates
The covariates in our study that may affect the psoriasis include age (20–60, ≥ 60 years old), sex (male, female), education level (under high school, high school or equivalent, college graduate or above), race (White, Black, Mexican American, other), poverty income ratio (< 1·3, 1·3–3·5, ≥ 3·5), BMI (< 18·5, 18·5–25, 25–30, ≥ 30), hypertension (no, yes), diabetes (no, yes), smoking status (never, ever, current), alcohol use (no, yes) and dietary folate intake (insufficient intake, adequate intake, excessive intake).
Statistical analysis
Serum folate levels were analysed as a continuous variable or categorised into quartiles, with the first quartile (Q1) representing the lowest levels and the fourth quartile (Q4) representing the highest levels. Considering the complex multistage sampling design of NHANES, we strictly followed the NHANES analysis guidelines(Reference Johnson, Paulose-Ram and Ogden23). Continuous variables were presented as means with standard deviation (SDs), while categorical variables were presented as percentages. Comparisons between different groups were conducted using the Chi-square test for categorical variables and the t test for continuous variables. The association between psoriasis and serum folate levels was analysed using weighted multivariable logistic regression models. In addition, we considered serum folate levels exceeding 1·5 times the third quartile as outliers and excluded them for sensitivity analysis to verify the stability of the results. Four models were used: the crude model, which did not adjust for covariates; Model 1, which adjusted for age and sex; Model 2, which adjusted for age, sex, race, education level and poverty income ratio and Model 3, which adjusted for all covariates. The Odds ratios (ORs) and 95% confidence intervals (CIs) were used to estimate the strength of the association in the multivariable models. All analyses were conducted using R version 4.4.1.
Mendelian randomisation analysis
Selection of instrumental variables
Genetic data for serum folate were obtained from a genome-wide association study (GWAS) involving 37 341 individuals from Iceland and Denmark(Reference Grarup, Sulem and Sandholt24). Specifically, the sample size was 28 913 from Iceland and 8428 from Denmark. In the preliminary analysis, we selected three genome-wide significant SNPs (P < 5 × 10−8) as instrumental variables for serum folate (rs1801133, rs652197, rs17421511). Detailed information on these SNPs can be found in online Supplementary Table 1. Additionally, we excluded one SNP (rs17421511) from further analysis based on the linkage disequilibrium threshold (r2 > 0·001) used in the MR analysis. We then calculated the F-statistic to assess the strength of the instrumental variables, which helps detect weak instrument variable problems and improves the reliability and statistical power of the analysis. The F-statistics for serum folate met the MR analysis assumption of F > 10 (online Supplementary Table 1)(Reference Palmer, Lawlor and Harbord25).
To avoid population source limitations, we also selected serum folate genetic data from a GWAS involving 38 000 individuals of South Asian descent for sensitivity analysis(Reference Jacobs, Stow and Hodgson26). We selected SNP with genome-wide significance (P < 5 × 10−8) and independence (r² = 0·001). A total of six SNPs were selected, with five new SNPs (rs116949171, rs61930974, rs74794636, rs1893007, rs12367973) in addition to rs1801133, which was previously mentioned. Detailed information on the serum folate SNPs can be found in online Supplementary Table 2.
Outcome data source
Genetic data for psoriasis were obtained from a GWAS involving 44 161 individuals of European ancestry, including 15 967 cases and 28 194 controls (https://gwas.mrcieu.ac.uk/) (ID: ebi-a-GCST90019016)(Reference Stuart, Tsoi and Nair27). The study also conducted a GWAS of psoriasis in South Asians, including 2590 cases and 1720 controls(Reference Stuart, Tsoi and Nair27).
Statistical analysis
Inverse variance weighting (IVW) based on fixed-effect and random-effect models was used as the primary analysis method(Reference Burgess, Foley and Zuber28). Cochran’s Q statistic was employed to assess heterogeneity among individual SNPs(Reference Hemani, Bowden and Davey Smith29). We also performed sensitivity analyses using the weighted median method and the MR-Egger method to verify the robustness of our results(Reference Bowden, Davey Smith and Burgess30,Reference Bowden, Davey Smith and Haycock31) . The intercept obtained from MR-Egger regression was used to measure directional pleiotropy(Reference Zhu, Zhang and Hu32). To address potential measurement error in the SNP-exposure effect, the maximum likelihood method was applied to provide more reliable estimates(Reference Hemani, Zheng and Elsworth33). Additionally, single-SNP analysis was conducted to evaluate the independent contribution of each SNP to the exposure–outcome relationship. The leave-one-out method was implemented by sequentially excluding each SNP to determine whether the estimates were driven by any single SNP. All analyses were performed using R version 4.4.1 and the ‘TwoSampleMR’ package.
Results
Observational analysis results
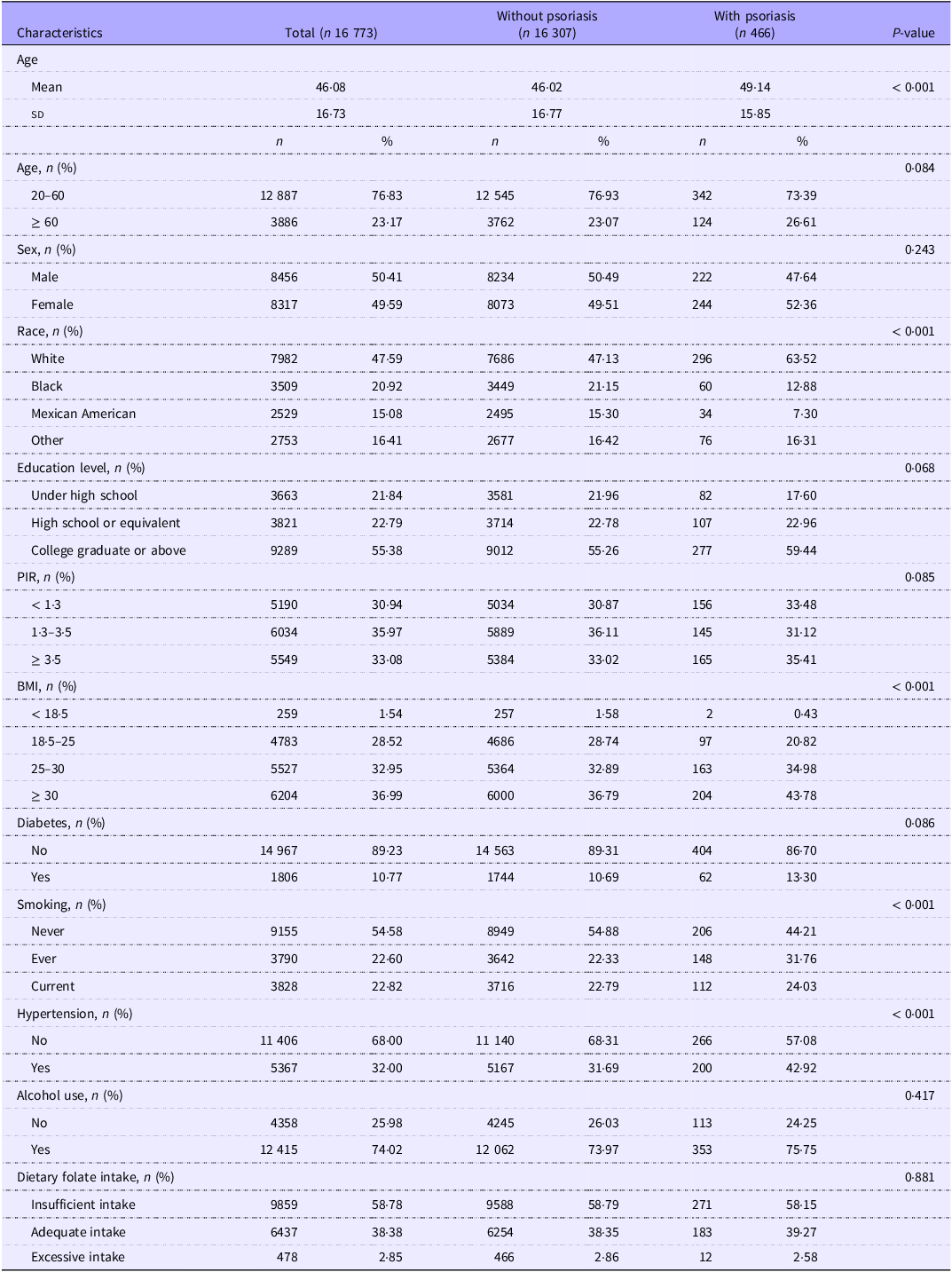
In the definition of psoriasis, 27 participants refused to answer the questions or responded with ‘don’t know.’ A total of 16 773 participants were included in this study, with 50·41 % being male and 49·59 % female. Among them, 466 participants (2·78 %) were diagnosed with psoriasis. There were significant differences in age, race, BMI, smoking and hypertension prevalence between psoriasis and non-psoriasis patients. Detailed demographic characteristics are presented in Table 1.
Baseline demographic characteristics of NHANES 2003–2006 and 2009–2014

NHANES, National Health and Nutrition Examination Survey; PIR, poverty income ratio.
Alcohol use, ‘Had at least 12 alcohol drinks in 1 year?’ If the answer is ‘No,’ then alcohol use is classified as ‘NO’; if the answer is ‘Yes,’ then alcohol use is classified as ‘YES.’
Dietary folate intake, Insufficient intake: Less than 400 micrograms; Adequate intake: Greater than or equal to 400 micrograms but less than 1000 micrograms; Excessive intake: Greater than 1000 micrograms.
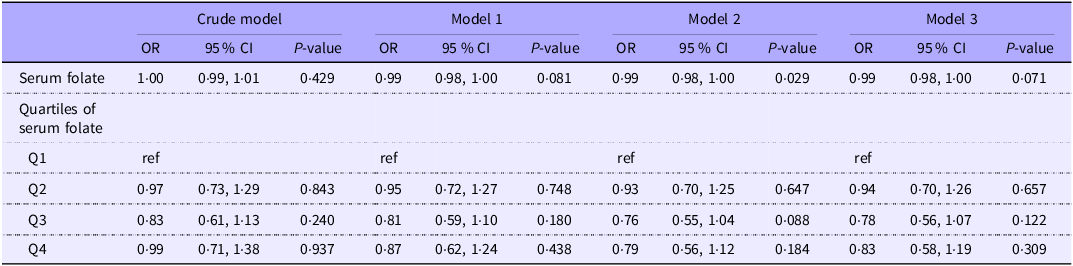
After performing weighted multivariable logistic regression analysis (Table 2), our results indicated limited evidence for an association between serum folate levels and psoriasis in our crude model (OR = 1·00, 95 % CI: 0·99, 1·01, P = 0·429), Model 1 (OR = 0·99, 95 % CI: 0·98, 1·00, P = 0·081), Model 2 (OR = 0·99, 95 % CI: 0·98, 1·00, P = 0·029) and Model 3 (OR = 0·99, 95 % CI: 0·98, 1·00, P = 0·071). The results were similar when serum folate was analysed as a categorical variable. Comparing the highest quartile (Q4) with the lowest quartile (Q1), the association remained non-significant across the crude model (OR = 0·99, 95 % CI: 0·71, 1·38, P = 0·937), Model 1 (OR = 0·87, 95 % CI: 0·62, 1·24, P = 0·438), Model 2 (OR = 0·79, 95 % CI: 0·56, 1·12, P = 0·184) and Model 3 (OR = 0·83, 95 % CI: 0·58, 1·19, P = 0·309).
Weighted multifactorial logistic analysis of serum folate levels and psoriasis

Model 1 was adjusted for age and sex. Model 2 was adjusted for age, sex, race, education level and poverty income ratio. Model 3 was adjusted for age, sex, race, education level, poverty income ratio, BMI, diabetes, smoking, hypertension, alcohol use and dietary folate intake.
We considered serum folate levels exceeding 34·65 ng/ml (1·5 times the third quartile value) as outliers. After excluding 1317 outlier samples, a sensitivity analysis was performed. The results showed no significant association between elevated serum folate levels and a reduced risk of psoriasis. In the fully adjusted model, neither serum folate level as a continuous variable (OR = 0·99, 95 % CI: 0·97, 1·01, P = 0·205) nor serum folate quartiles Q4 v. Q1 (OR = 0·81, 95 % CI: 0·57, 1·14, P = 0·219) showed a significant association. Detailed results are presented in online Supplementary Table 3.
Mendelian randomisation analysis results
The effect estimates from the MR analysis examining the association between serum folate levels and psoriasis are illustrated in Figure 1. For the MR analysis of serum folate (Iceland and Denmark) and psoriasis, Cochran’s Q test found no evidence of heterogeneity (P > 0·05), so the fixed-effects IVW model was chosen for the MR analysis. The IVW method indicated that higher genetically predicted serum folate levels were associated with a reduced risk of psoriasis (OR = 0·73, 95 % CI: 0·53, 1·00, P = 0·049). Sensitivity analyses confirmed similar results. The weighted median method showed an OR of 0·75 (95 % CI: 0·56, 1·00, P = 0·047), and the maximum likelihood method yielded an OR of 0·73 (95 % CI: 0·56, 0·95, P = 0·019). MR-Egger regression found no evidence of directional pleiotropy (P > 0·05).
Forest plot of the MR analysis. The MR shows that higher genetically predicted serum folate levels were associated with a reduced risk of psoriasis. LD, linkage disequilibrium; MR, Mendelian randomisation.

After excluding one SNP due to linkage disequilibrium, the association between serum folate levels and psoriasis remained consistent with the preliminary results. Higher genetically predicted serum folate levels were associated with a reduced risk of psoriasis, as shown by both IVW method (OR = 0·63, 95 % CI: 0·45, 0·88, P = 0·005) and the maximum likelihood method (OR = 0·63, 95 % CI: 0·45, 0·88, P = 0·006). Due to the requirement of at least three SNPs for MR-Egger regression, we were unable to assess horizontal pleiotropy using this method. Single SNP analysis revealed that both serum folate SNPs act as protective factors against psoriasis (online Supplementary Figure 2A).
For the MR analysis of serum folate (South Asian) and psoriasis (European), Cochran’s Q test found no evidence of heterogeneity (P > 0·05). The IVW method indicated that higher genetically predicted serum folate levels were associated with a reduced risk of psoriasis (OR = 0·84, 95 % CI: 0·72, 0·98, P = 0·025). The weighted median method showed an OR of 0·82 (95 % CI: 0·68, 0·97, P = 0·024). The MR-Egger method showed an OR of 0·60 (95 % CI: 0·31, 1·14, P = 0·192), and the maximum likelihood method yielded an OR of 0·84 (95 % CI: 0·71, 0·98, P = 0·026). MR-Egger regression found no evidence of directional pleiotropy (P > 0·05). The results of the single SNP analysis are shown in online Supplementary Figure 2B. The leave-one-out analysis plot (online Supplementary Figure 3) showed that, except for SNP rs1801133, which had a certain impact on the results, no potentially influential SNPs affected the causal relationship.
Additionally, we explored available Asian psoriasis GWAS data (2590 cases/1720 controls). Analysis with this dataset showed similar trends to our main findings, though results were not statistically significant due to limited sample size and matched SNPs (only three). The effect estimates were obtained using the IVW method (OR = 0·59, 95 % CI: 0·24, 1·47, P = 0·258), weighted median (OR = 0·55, 95 % CI: 0·28, 1·08, P = 0·073) and maximum likelihood (OR = 0·59, 95 % CI: 0·33, 1·05, P = 0·074).
Discussion
Our observational study found no significant association between serum folate levels and psoriasis. This lack of association may be due to the relatively small sample size of psoriasis patients (2·78 % of participants) in our NHANES dataset. In contrast, the MR analysis indicated a significant causal relationship between serum folate and the risk of psoriasis, identifying serum folate levels as a protective factor. To our knowledge, this is the first study to investigate the causal relationship between serum folate and psoriasis.
Previous studies have mainly focused on whether there is a significant difference in serum folate levels between psoriasis and non-psoriasis populations, but the results have been inconsistent. Seven studies(Reference Malerba, Gisondi and Radaeli12,Reference Fry, Macdonald and Almeyda13,Reference Refsum, Helland and Ueland34–Reference Sharaf, Ghanem and Marzouk38) reported significantly lower serum folate levels in psoriasis patients, which differs from our observational study findings. Several factors may explain this discrepancy: first, differences in sample sizes—many previous studies were small case-control studies, while our observational study involved a larger sample; second, variations in sample quality control—four studies(Reference Malerba, Gisondi and Radaeli12,Reference Fry, Macdonald and Almeyda13,Reference Karabudak, Ulusoy and Erikci36,Reference Sharaf, Ghanem and Marzouk38) strictly controlled sample selection, including healthy controls and excluding patients who had been exposed to methotrexate, as both disease factors and methotrexate use could affect serum folate levels. Our study, due to its large sample size and inability to fully control for other diseases and medications, may have been influenced by these factors. On the other hand, four studies(Reference Weger, Hofer and Stanger14–Reference Cakmak, Gül and Kiliç16,Reference Juzeniene, Stokke and Thune39) found no significant difference in serum folate levels between the case and control groups, consistent with our observational study results. These studies also differed in sample size and drug control. Additionally, different age groups, ethnic populations and gender compositions may also have an impact on the results.
Our MR analysis suggests that serum folate levels is a protective factor against psoriasis, and this is consistent with the trend observed in our observational study. MR studies can significantly reduce the influence of confounding factors and reverse causation, effectively addressing the limitations of observational study. Although recent MR studies have explored the association between serum folate levels and other autoimmune diseases(Reference Yang, Song and Li40), the causal relationship between serum folate levels and psoriasis has not been thoroughly investigated. Our study fills this gap by providing the first evidence of this relationship, using rigorously selected instrumental variables and considering potential limitations such as ethnicity. These methods help ensure the accuracy of causal inference and reduce possible biases. This finding supports the hypothesis that higher serum folate levels may help prevent the onset of psoriasis. However, to confirm these findings and establish the optimal dosage and duration, further investigation through large-scale cohort studies or randomised controlled trials is required.
Folate exerts its protective effects against psoriasis may through several biological mechanisms. First, folate is a precursor to the methyl donor S-adenosylmethionine, which plays a critical role in DNA methylation. DNA methylation is a key epigenetic regulatory mechanism that regulates gene expression and can suppress the abnormal activation of genes associated with psoriasis(Reference Crider, Yang and Berry41,Reference Ly, Hoyt and Crowell42) . By maintaining proper DNA methylation patterns, folate may help prevent the excessive proliferation of skin cells characteristic of psoriasis. Second, folate deficiency can impair cell division and growth(Reference Choi and Mason43), leading to abnormal proliferation of skin cells, which is a hallmark of psoriasis. Furthermore, folate plays an important role in maintaining immune system balance; a lack of folate can damage dendritic cell function, affecting Th cell differentiation and response(Reference Wu, Huang and Lin44), potentially exacerbating the inflammatory processes involved in psoriasis. Lastly, antioxidant properties of folate may help alleviate psoriasis symptoms by reducing oxidative damage through decreased free radical production(Reference Garbicz, Całyniuk and Górski45). These biological mechanisms support that folate is a rational and consistent protective factor for psoriasis. Given that folate cannot be synthesised in the body and must be obtained through diet, folate supplementation may be significant in preventing or alleviating psoriasis. Future research should explore the long-term effects of folate supplementation on psoriasis outcomes.
This study has several strengths. We initially conducted an observational study using the NHANES database to investigate the association between serum folate levels and psoriasis incidence. This large cross-sectional study benefits from NHANES’ multi-stage complex sampling design, which plays a crucial role in ensuring the representativeness and accuracy of the survey results. Subsequently, we utilised MR analysis to explore the causal relationship between serum folate levels and psoriasis. By employing these methods, our study provides a comprehensive examination of the relationship between serum folate levels and psoriasis. The MR analysis helps mitigate biases related to confounding factors and reverse causation, addressing the limitations of the observational study.
However, our study also has several limitations. Firstly, the data on psoriasis patients in the observational study were collected via self-report, and the number of psoriasis patients included in the study was relatively small. Additionally, we were unable to determine whether the study participants were using medications or other factors that might influence serum folate levels, which could introduce bias into the analysis. Secondly, the MR study was constrained by a limited number of SNP related to serum folate levels, particularly for the European population, weakening the robustness of the causal inference. Caution is therefore advised when interpreting these MR results. Future studies, utilising larger datasets, are essential to further investigate and confirm the relationship.
Conclusions
In summary, our MR study suggests that serum folate levels are a protective factor against psoriasis, and higher serum folate levels may help prevent the onset of psoriasis. However, due to the limitations of the studies and other constraints, these associations warrant further investigation. Nonetheless, considering that folate supplementation may reduce the side effects of methotrexate treatment for psoriasis, we recommend that psoriasis patients consider appropriate folate supplementation.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114525000819
Acknowledgements
We are grateful to the researchers who shared their summary data, which significantly facilitated this research.
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
The authors declare that they have no conflict of interest.
C-J. D. and R-X. L. conceived, designed and planned the study. C-J. D., J-W.Y. and Y-C. Z. acquired and analysed the data. C-J. D., J-W. Y., D-S. D. and R-X. L. interpreted the results. C-J. D. drafted the manuscript. D-S. D. and R-X. L. contributed to the critical revision of the manuscript and supervised the study. All authors read and approved the final manuscript.
The datasets used in this study are publicly accessible and were obtained from sources that have received ethical approval from their respective ethics committees. Additionally, this study adhered to the principles outlined in the Declaration of Helsinki.
Consent for publication: Not applicable.