Introduction
Contrast-induced encephalopathy (CIE) is an adverse event associated with contrast administration during diagnostic and therapeutic endovascular procedures. The spectrum of clinical presentation includes altered level of consciousness (LOC), hemiparesis, cortical blindness, neglect, speech dysfunction, seizures and coma. Reference Yu and Dangas1–Reference May, Ling, Geyer and Jabbari4 Symptomatology reflects brain regions affected by the contrast extravasation. Reference Quintas-Neves, Araújo and Xavier5 Characteristic radiographic findings include cortical contrast enhancement and vasogenic edema. Reference Yu and Dangas1,Reference Sharp, Stone and Beach6 CIE often develops within hours; clinical and radiographic resolution varies from hours to days. Among patients undergoing neuroendovascular procedures, CIE has been associated with poor disability scores at 3 months compared to patients who do not develop CIE. Reference Chu, Lee and Chen7
CIE was first reported in the 1970s. Reference Fischer-Williams, Gottschalk and Browell8 The overall incidence of CIE is estimated to be 2-3% in the context of cerebral angiography. Reference Quintas-Neves, Araújo and Xavier5,Reference Niimi, Kupersmith, Ahmad, Song and Berenstein9 A review identified 48 cases of CIE associated with neuroendovascular procedures, the majority with diagnostic procedures. Reference Quintas-Neves, Araújo and Xavier5 Evidence for CIE following endovascular treatment of cerebrovascular disease is sparse and limited to case reports; examples include the endovascular treatment of intracranial aneurysms Reference Guimaraens, Vivas and Fonnegra3,Reference Niimi, Kupersmith, Ahmad, Song and Berenstein9–Reference Leong and Fanning14 carotid stenosis. Reference Dangas, Monsein and Laureno2,Reference Fang, Kuo and Wu15 To our knowledge, only 9 prior cases of CIE have been reported following endovascular thrombectomy (EVT) for acute ischemic stroke (AIS). Reference Chu, Lee and Chen7,Reference Bakradze, Pasquale and Kirchoff-Torres16,Reference Aguirre, Trillo, Ximénez-Carrillo and Vivancos17
The blood-brain barrier (BBB) typically prevents iodinated contrast medium in circulating blood from crossing into both the cerebrospinal fluid and extracellular fluid of the central nervous system. Animal and human data support a mechanistic role for blood-brain barrier dysfunction (BBBd) in CIE. Reference Uchiyama, Abe and Hirohata13,Reference Wilson, Evill and Sage18–Reference Wang, Li and Zhao20 Preexisting microvascular dysfunction is a risk factor for both CIE, Reference Quintas-Neves, Araújo and Xavier5,Reference Chu, Lee and Chen7,Reference Bakradze, Pasquale and Kirchoff-Torres16,Reference Aguirre, Trillo, Ximénez-Carrillo and Vivancos17 and BBBd; Reference Bowman, Kaye and Quinn21–Reference Kassner and Merali27 examples include advanced age, hypertension (HTN), smoking history, diabetes mellitus (DM), chronic kidney disease (CKD) and prior stroke. Intuitively, these factors often reflect the underlying vascular pathology that prompted the endovascular intervention. Reference Spina, Simon, Markus, Muller and Kathir28 Procedural factors, including intraoperative HTN, contrast osmolality and volume have been investigated for their role in both BBBd and CIE. Reference Yu and Dangas1,Reference Uchiyama, Abe and Hirohata13,Reference Wilson, Evill and Sage18,Reference Wang, Li and Zhao20,Reference Sakaki, Tsujimoto, Nishitani, Ishida and Morimoto22,Reference Bassett, Rogers, Cherry and Cruzhit29
The demand for endovascular therapy in patients with cerebrovascular disease has grown in light of expanding indications, technological advancements and an aging demographic. Reference Zaidat, Lazzaro and McGinley30,Reference Riina31 Further research should clarify risk factors, improve symptom recognition and optimize treatment for CIE. Given this, we sought to: (1) describe our recent institutional experience with CIE following treatment of cerebrovascular disease; and (2) discuss animal and human literature supporting a mechanistic role for BBBd.
Methods
Patient selection
A recent, severe case of CIE prompted us to search our institutional neurovascular database for instances of CIE following treatment of cerebrovascular disease during the period spanning August 1st, 2020, to January 31, 2021. This study was conducted in accordance with institutional guidelines and regulations and ethics approval was not required given the following: (1) informed consent was directly obtained from all subjects and/or their legal guardian(s); (2) no prospective experimentation was performed; (3) all procedures were completed as part of routine standard of clinical care; and (4) reporting adheres to the CARE guidelines for reporting of case series. Reference Gagnier, Kienle, Altman, Moher, Sox and Riley32
Diagnosis of CIE
We defined CIE in keeping with prior literature: Reference Quintas-Neves, Araújo and Xavier5,Reference Chu, Lee and Chen7 (1) diagnostic or therapeutic endovascular procedure performed ≤ 24 hours before clinical presentation; (2) unequivocal neurological deterioration or delayed improvement after therapeutic endovascular procedure that could not be explained by preexisting ischemic area in the CT perfusion study, recurrent stroke or hemorrhagic transformation; and (3) cranial radiographic imaging performed ≤ 7 days after onset of clinical presentation, demonstrating edematous change extending beyond the infarct core, with contrast staining.
Literature search
A narrative literature review was conducted by searching MEDLINE, Embase, CINAHL and Cochrane Central Register of Controlled Trials from inception to January 31st, 2022. Keywords searched were relevant to contrast encephalopathy and the BBB (Supplemental Information A). Animal and human studies of any methodological design were included if they examined the role of BBBd in the context of either CIE or endovascular administration of iodinated contrast. Reference lists of included articles were reviewed. Literature search results were reviewed and two authors (MM and JM) independently screened abstracts for study inclusion. We extracted article data pertaining to citation, publication year, type of study (animal, human), number of patients (if applicable), study methodology and study findings.
Data extraction and study outcome measures
Institutional case series data extracted pertained to demographics, relevant medical comorbidities, history of prior cranial or endovascular surgery, procedural variables (e.g. operative duration, contrast media type and volume, vessel(s) affected, ASPECTS and NIHSS scores), pre and postoperative symptoms, pre and postoperative radiographic findings (including brain region and laterality, presence of cerebral edema or contrast staining and time to resolution) and CIE-related treatments.
Statistical methods
Descriptive analysis was performed using extracted patient data. Both the Mann-Whitney U test, as well as Fisher’s exact test were performed using SPSS Version 27 (IBM Corp, Armonk, New York, USA). Significance was observed at p < 0.05.
Institutional series results
Baseline patient characteristics
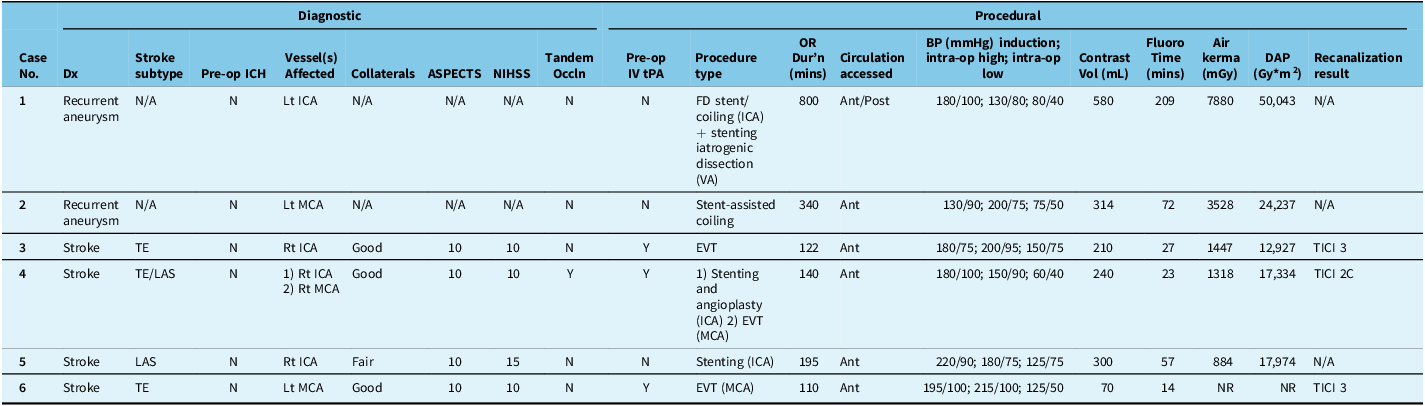
Six of 52 patients (11.5%) developed CIE following endovascular surgery for cerebrovascular disease (Table 1). Median patient age was 65.6 years (range 41-90). Half were female. Median BMI was 28.5 kg/m2 (range 22–41). All patients had risk factors for microvascular disease, including HTN (n = 6), dyslipidemia (n = 5), CKD (n = 5), prior ischemic stroke (n = 3), connective tissue disorders (n = 2; polycystic kidney disease and fibromuscular dysplasia, respectively) and prior open cranial or neuroendovascular surgery (n = 2). Both patients presenting with recurrent intracranial aneurysms had a history of aneurysmal subarachnoid hemorrhage (n = 2). Patients’ median preoperative serum creatinine was 76 μmol/L (range 70–130). Estimated glomerular filtration rate ranged between 39 and 104 (mL/min/1.73m2), corresponding to CKD stages 1–3b (Table 1). No statistical differences were observed for either demographic or clinical tests (Supplemental Material B).
Baseline patient characteristics

AFib = atrial fibrillation; BMI = body mass index in mg/k2; CKD = chronic kidney disease; CTD = connective tissue disorder; DLP = dyslipidemia; Endovasc = endovascular; EGFR = estimated glomerular filtration rate in mL/min/1.73m2; HTN = hypertension; F = female; FMD = fibromuscular dysplasia; IHD = ischemic heart disease; Lt = left; M = male; PCKD = polycystic kidney disease; PComm = posterior communicating artery; RF = risk factor; Rt = right; SAH = subarachnoid hemorrhage; sCr = serum creatinine in micromole/L.
Perioperative diagnostic and procedural data
Patients received elective neuroendovascular surgery for recurrent unruptured intracranial aneurysms (n = 2), and urgent intervention for AIS (n = 4) (Table 2; Supplemental Material C-F). All patients receiving EVT for stroke received intravenous tissue plasminogen activator (tPA) preoperatively. Among the 4 patients treated for AIS, collateral circulation was good (n = 3) or fair (n = 1), ASPECTS scores were 10 (n = 4), and NIHSS scores were 10 (n = 3) or 15 (n = 1). AIS diagnoses and treatment included: (1) thromboembolic stroke with complete occlusion of the proximal internal carotid artery (ICA), treated with stent retrieval and aspiration; (2) thromboembolism of the M2 segment of the left middle cerebral artery (MCA) treated with stent retrieval and aspiration; (3) ICA thromboembolism accompanied by large artery atherosclerosis, treated with stenting alone; and (4) tandem occlusion of right ICA and M1 segment of the right MCA (n = 1) treated with stenting and angioplasty.
Perioperative diagnostic and procedural data for patients undergoing endovascular therapy for cerebrovascular disease

Ant = anterior; Dur’n = duration; DAP = dose area product; EVT = endovascular thrombectomy; FD = flow diversion; Fluoro = fluoroscopy; ICA = internal carotid artery; LAS = large artery atherosclerosis; MCA = middle cerebral artery; mins = minutes; N = no; N/A = not applicable; NIHSS = National Institutes of Health Stroke Scale; Occln = occlusion; OR = operative; P = posterior; TE = thromboembolic; TICI = thrombolysis in cerebral infarction; tPa = tissue plasminogen activator; VA = vertebral artery; Vol = volume; Y = yes.
Procedural details are described in Table 2. All patients underwent anterior circulation procedures; vessels affected included internal carotid artery (ICA) (n = 3), middle cerebral artery (MCA) (n = 3) or both (n = 1). Temporary stents placed in intracranial vessels during AIS procedures were retrieved (n = 2). Extracranial stents placed were permanent (n = 2). Median operative duration and fluoroscopy time were 167.5 minutes (range 110–800) and 41.6 minutes (range 14.2–208.9), respectively. All patients received nonionic, iodinated contrast (Iohexol, Omnipaque). The median volume of contrast delivered was 270 mL (range 70–580). All 6 patients experienced severe arterial HTN (peak systolic pressure >180 mmHg) intraprocedurally (Table 2). Thrombolysis in Cerebral Infarction (TICI) scores for patients who underwent EVT were TICI 2c (n = 1) and TICI 3 (n = 2).
Postoperative clinical, treatment and radiological data
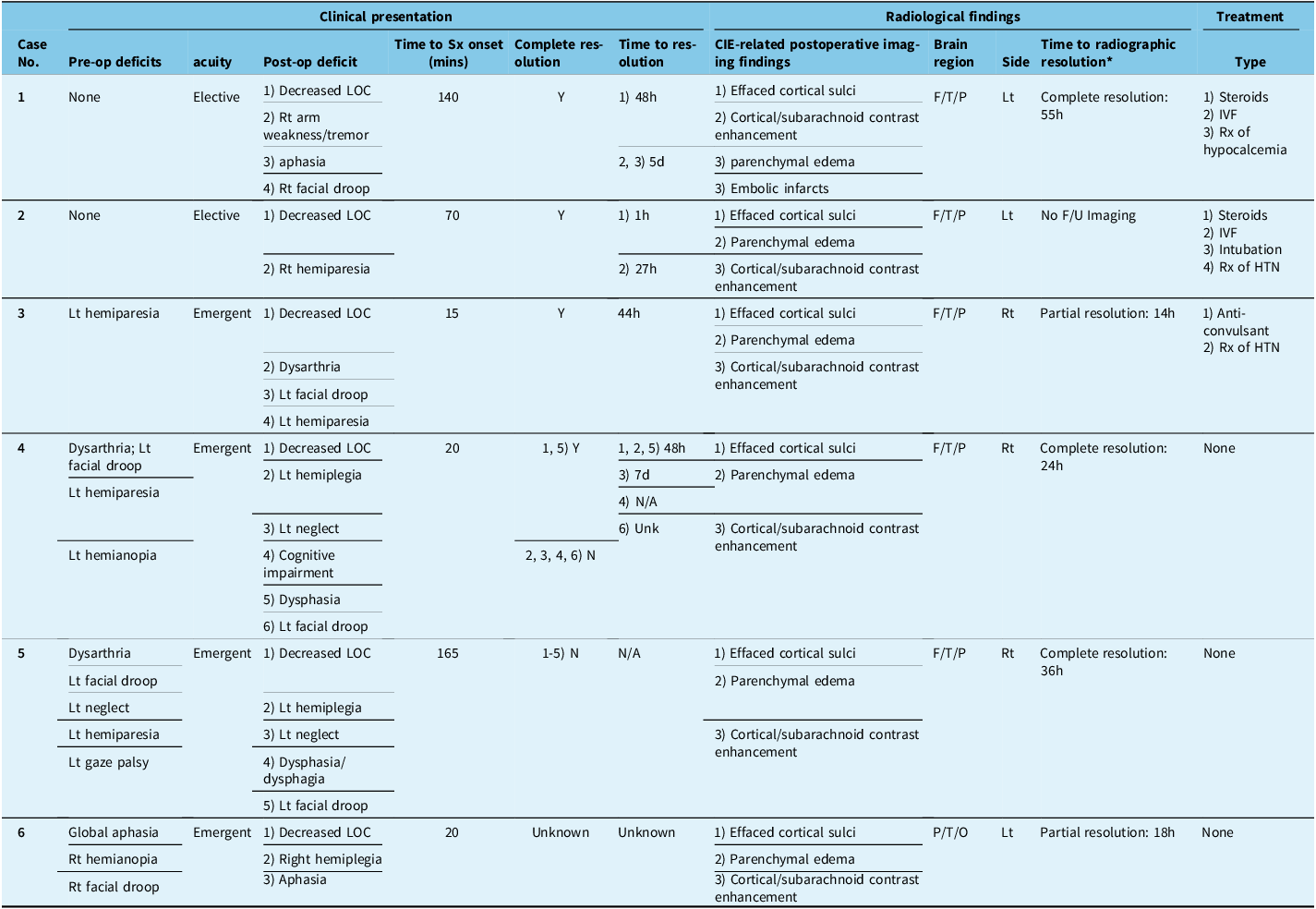
Clinical symptoms and signs associated with CIE are detailed in Table 3. Patients experienced postoperative decreased LOC (n = 6), lateralizing motor deficit (n = 6), dysphasia or aphasia (n = 5), hemiparesis progressing to hemiplegia (n = 4), ipsilateral facial droop (n = 4), neglect (n = 2), dysphagia (n = 1) and tremor (n = 1). Median time from procedure to onset of CIE-associated symptoms was 45 minutes (range 15–165). Both patients treated for intracranial aneurysms were neurologically intact preoperatively and made a complete clinical recovery. Patients (n = 2 of 3 for whom data were available) treated for AIS had preoperative neurological deficits which progressed postoperatively and did not fully recover. Two of 3 patients for whom data were available had an elevated serum osmolality postoperatively. Time to resolution of symptoms ranged from 1 hour to 7 days. CIE-directed treatments are detailed in Table 3.
Postoperative clinical, treatment and radiological data for patients with contrast-induced encephalopathy following endovascular therapy for cerebrovascular disease

d = day; F = frontal; F/U = follow-up; h = hour; HTN = hypertension; IVF = intravenous fluids; LOC = level of consciousness; Lt = left; MCI = mild cognitive impairment; N = no; N/A = not applicable; Op = operative; P = parietal; Rt = right; Sx = symptom; T = temporal; Unk = unknown; Y = yes.
* Time to radiographic resolution: Time to resolution was recorded from available postoperative imaging. Timing of postoperative imaging was not standardized. In cases of partial resolution, no further imaging was ordered to confirm complete resolution.
Radiological findings associated with CIE are described in Table 3. Effacement of the cortical sulci, cortical or subarachnoid contrast enhancement and parenchymal edema were observed in all patients (Figs. 1 and 2; Supplemental Material C–F). In patients (n = 5) who had follow-up imaging, complete (n = 3) or partial (n = 2) radiographic resolution of contrast extravasation was observed.
Case 1. (A) Unenhanced axial computed tomography (CT) demonstrates blood filling predominantly the anterior basal cisterns and bilateral sylvian fissures. (B) Oblique view cerebral angiogram, left internal carotid artery (ICA) injection demonstrating blister aneurysm of the supraclinoid segment (red arrow). (C) CT angiography (CTA) reconstructions demonstrate post-treatment blister aneurysm recurrence. (D) Unenhanced CT head following treatment of the aneurysm recurrence shows left hemispheric effacement of the cortical sulci and contrast staining. (E) Axial magnetic resonance imaging (MRI) re-demonstrates the contrast extravasation, focused on the hand knob region of the primary motor cortex. Diffusion-weighted imaging demonstrated embolism related infarction in the same region (not shown). (F) Follow-up MRI at 55 hours post-treatment demonstrates near resolution of contrast extravasation and sulcal effacement.

Case 6. (A) CTA 3-dimensional reconstruction demonstrates occlusion of the left MCA (white arrow). (B) CT perfusion demonstrated increased TTP in the left MCA territory. Anterior view of the left ICA injection digital subtraction angiogram pre- (C) and post- (D) EVT, demonstrating TICI 3 result. (E) Unenhanced CT head demonstrates contrast extravasation over the left temporal, parietal and occipital regions with effaced cortical sulci and parenchymal edema. (F) Repeat CT Head 18 hours later demonstrates resolution. CT = computed tomography; CTA = CT angiography; EVT = endovascular thrombectomy; ICA = internal carotid artery; MCA = middle cerebral artery; TICI = Thrombolysis in Cerebral Infarction; TTP = time to peak.

Discussion and review of literature
Here, we describe our experience with CIE following neuroendovascular treatment of cerebrovascular disease. All patients had multiple preexisting risk factors for microvascular disease, underwent long procedures (median 167.5 minutes), received large volumes of contrast (median 270 mL) and experienced severe intraoperative arterial HTN (peak systolic arterial pressure > 180 mmHg). All cases involved use of mechanical devices for stenting or clot extraction, representing a potential risk factor for iatrogenic endothelial injury. Clinical symptoms, radiological findings, CIE-directed treatments and clinical outcomes were similar to those previously reported. Reference Quintas-Neves, Araújo and Xavier5,Reference Chu, Lee and Chen7 Below, we discuss animal and human studies supporting a mechanistic role for BBBd, including: (1) preexisting microvascular dysfunction; (2) neuroinflammation; (3) iodinated contrast agent volume and concentration; (4) severe procedural HTN; and (5) cerebral blood flow (CBF) dynamics. We hypothesize that these risk factors for BBBd may also increase the risk of CIE.
Clinical risk factors for CIE
We identified demographic similarities between patients in our series (Table 1) and prior reports. Reference Quintas-Neves, Araújo and Xavier5 These include risk factors for preexisting microvascular disease, such as advanced age, uncontrolled preoperative arterial HTN, DM and CKD. Reference Quintas-Neves, Araújo and Xavier5 Five patients in our series had prior stroke and 2 patients had connective tissue disorders which may predispose patients to pathological neurovascular manifestations. Reference Kim, Brinjikji, Lanzino and Kallmes33 Previously reported risk factors for CIE include CKD, prior stroke, anticoagulant use and heart failure. The association between microvascular disease, ischemic stroke and BBBd is established. Reference Teng, Pannell and Rennert26,Reference Kassner and Merali27,Reference Serlin, Ofer and Ben-Arie34–Reference Amantea, Russo and Gliozzi38 As such, we hypothesize that the aforementioned variables are also risk factors for CIE.
Clinical and radiographic findings associated with CIE
Clinical manifestations of CIE may include encephalopathy (39.6%), cortical blindness (39.6%), motor weakness (37.5%)and decreased LOC (20.8%). Reference Quintas-Neves, Araújo and Xavier5 Aphasia, headache, seizures and neglect have also been reported. We identified a similar spectrum of symptoms (Table 3). Decreased LOC was observed in all cases. Symptom onset time was similar to prior reports. Reference Quintas-Neves, Araújo and Xavier5
CIE-related radiological findings typically include effacement of cortical sulci (37.5%), parenchymal edema (29.2%), cortical (54.8%) or subarachnoid (22.9%) contrast enhancement. Reference Quintas-Neves, Araújo and Xavier5 These findings (Table 3) were also similar to those previously reported. Reference Quintas-Neves, Araújo and Xavier5,Reference Chu, Lee and Chen7
Prognosis following CIE
Clinical outcomes following CIE vary. Reference Guimaraens, Vivas and Fonnegra3,Reference Quintas-Neves, Araújo and Xavier5,Reference Niimi, Kupersmith, Ahmad, Song and Berenstein9–Reference Leong and Fanning14,Reference Lantos39,Reference Nakao, Joshi and Hirose40 Following diagnostic procedures, most patients recover clinically and radiographically within days. Reference Quintas-Neves, Araújo and Xavier5 While data is sparse, outcomes following intervention for AIS may be less favorable; preoperative neurological deficits were associated with postoperative progression and incomplete clinical recovery. Similarly, Chu et al. reported unfavorable clinical outcomes in 6 of 7 CIE patients treated with EVT for AIS. Reference Chu, Lee and Chen7 No long term data is available for patients with CIE.
BBBd and CIE
The BBB is composed of microvascular endothelial cells, pericytes and astrocytic foot processes. Reference MacLean, Kamintsky, Leck and Friedman24 Tight junctions bridging endothelial cells and the basement membrane are comprised of transmembrane (e.g. Claudin and Occludin) and cytoplasmic proteins (e.g. ZO-1); both play an integral role in regulating integrity and permeability of the BBB. Reference Wang, Li and Zhao20 Several animal and human studies have implicated a mechanistic role for BBBd in CIE. BBBd may underly brain dysfunction and injury, either by direct disruption of brain homeostasis due to serum extravasation into brain neuropil, or indirectly via astroglia activation, endothelial dysfunction and pathological neuroinflammation (e.g. cytokine release). Below, we discuss the association between BBBd, microvascular disease, iodinated contrast medium, intraoperative HTN, neuroinflammation and iatrogenic endothelial injury as risk factors for CIE.
BBBd and iodinated contrast medium
Iodinated contrast agents do not typically cross the BBB. However, both ionic and nonionic iodine contrast media have been associated with CIE. Uchiyama et al. provided direct evidence of high iodinated contrast concentration in the cerebrospinal fluid of a patient with CIE, compared to controls. Reference Uchiyama, Abe and Hirohata13 The mechanism of contrast extravasation is likely multifactorial. Iodinated contrast agents have been shown to reduce expression of endothelial tight junction proteins. Reference Wang, Li and Zhao20 BBBd may also be related to a chemo-toxic effect of contrast on cerebral microvasculature, Reference Salvesen, Nilsen and Holtermann41–Reference Chase and Kricheff43 particularly in the setting of slow CBF with prolonged contact of contrast on the endothelium. Reference Uchiyama, Abe and Hirohata13,Reference Bassett, Rogers, Cherry and Cruzhit29,Reference Horwitz and Wener42 Alternatively, BBBd has been associated with iodinated contrast osmolality and volume. Reference Bassett, Rogers, Cherry and Cruzhit29 Contrast hyperosmolality may induce BBBd by drawing water out of endothelial cells, with resultant cell shrinkage and separation of tight junctions. Reference Uchiyama, Abe and Hirohata13 To this end, Wilson et al. compared mannitol to several nonionic, iodinated contrast agents varying in osmolality. They found that iodinated contrast media delivery resulted in BBBd in rabbits, whereas mannitol did not. Reference Wilson, Evill and Sage18 These findings suggest that BBBd is not related to contrast osmolality alone and other physical or chemical effects of iodinated contrast, such as viscosity, may alter BBB function. Reference Parker, Aboghazleh and Mumby52 Lastly, BBBd may also be related to contrast volume. Reference Bassett, Rogers, Cherry and Cruzhit29 The median lethal dose LD50 for commonly used iodinated contrast agents is 10–20 ml/kg. Reference Junck and Marshall44 In our study, high volumes of media delivered, ranging from 210 to 580 mL (i.e. up to 8 mL/kg; approaching the LD50), may have contributed to CIE.
BBBd in AIS: molecular mechanisms
An association between BBBd and cerebral microvascular disease has been demonstrated in animals, Reference Schoknecht, Prager and Vazana25,Reference Mazumder, Giri, Kumar and Borah45 and humans. Reference Bowman, Kaye and Quinn21,Reference Kassner and Merali27,Reference Serlin, Ofer and Ben-Arie34,Reference Lakhan, Kirchgessner, Tepper and Leonard36,Reference Sweeney, Sagare and Zlokovic46,Reference Jabbari and Vaziri47 In our series, and prior reports of CIE following EVT for AIS, Reference Quintas-Neves, Araújo and Xavier5,Reference Aguirre, Trillo, Ximénez-Carrillo and Vivancos17 multiple preexisting patient risk factors for microvascular disease were identified. As such, we hypothesize that preoperative microvascular disease is a risk factor for both BBBd and CIE during intervention.
Disruption of pulsatile flow across endothelial cells in the setting of ischemic stroke has numerous pathological implications, including: (a) increased BBBd via decreased expression of tight junction proteins; (b) thrombosis-induced endothelial cell release of proinflammatory cytokines (e.g. TNF-alpha, IL-1-beta); (c) glial cell activation; (d) leukocyte recruitment with local release of cytotoxic molecules; and (e) decreased nitric oxide production by injured endothelial cells, which may compromise autoregulation of the microcirculation, particularly in sensitive penumbra. Reference Teng, Pannell and Rennert26
In the context of ischemic stroke, BBBd has also been associated with cortical spreading depolarization (CSD), Reference Gursoy-Ozdemir, Qiu and Matsuoka48 seizures, Reference Vazana, Veksler and Pell49 elevated levels of glutamate, Reference Vazana, Veksler and Pell49 proinflammatory cytokines, Reference Kassner and Merali27,Reference Amantea, Russo and Gliozzi38 increased levels of matrix metalloproteinases (MMPs) and neuronal injury. Reference Wang, Li and Zhao20,Reference MacLean, Kamintsky, Leck and Friedman24,Reference Lakhan, Kirchgessner, Tepper and Leonard36,Reference Rempe, Hartz and Bauer37,Reference Gursoy-Ozdemir, Qiu and Matsuoka48 MMPs, including MMP-9, have an established role in modulating BBB function and structure; they degrade tight junction proteins and may induce paracellular leakage. Reference Rempe, Hartz and Bauer37 MMPs are upregulated in response to acquired brain injury and play a role in the neuroinflammatory response. Reference Cunningham, Wetzel and Rosenberg50 CSDs are common following AIS, Reference Dreier51 trigger the release of MMPs, Reference Parker, Aboghazleh and Mumby52 result in BBB breakdown, Reference Takizawa, Qin and Lopes de Morais53 upregulate proinflammatory cytokines, Reference Nakamura, Strong and Dohmen54 and contribute to ischemic lesion progression. Reference Park, Hwang, Song, Suh and Lee55 While these factors may contribute to CIE following AIS, the extent of subarachnoid contrast extravasation among our cases suggest other mechanisms may also be involved.
Blood-brain barrier dysfunction in acute ischemic stoke: cerebral blood flow dynamics and iatrogenic endothelial injury
Endothelial cell damage may result from iatrogenic mechanical trauma, altered CBF dynamics or reperfusion injury. Mechanical endovascular stenting or clot extraction may induce device-specific iatrogenic endothelial trauma following treatment of AIS in animals. Reference Gounis, Wakhloo and Chueh56,Reference Whisson, Wilson, Evill and Sage57 The use of such devices is increasing, particularly among the aging population, at longer time intervals since the onset of symptoms, with the goal of rapid and complete revascularization. Reference Zaidat, Lazzaro and McGinley30,Reference Riina31 These factors may potentially increase the likelihood of vessel perforation, contrast extravasation and subarchnoid hemorrhage. In vitro and in vivo studies provide valuable insight regarding the pattern and degree of endothelial injury inflicted, based on device size and shape. Reference Teng, Pannell and Rennert26 Furthermore, endothelial cell injury may vary based on vessel diameter, with greater endothelial cell injury in smaller vessels, emphasizing the important of appropriately matching device size based on vessel dimensions. Reference Teng, Pannell and Rennert26 These findings highlight the importance of laboratory studies in an effort to design novel technologies with the goal of minimizing endothelial trauma, thereby preventing BBBd, neuroinflammation and secondary neuronal injury. Reference Teng, Pannell and Rennert26
In our series, all patients experienced intraoperative arterial HTN (Table 2). We propose that severe intraoperative HTN, particularly in the setting of preexisting cerebrovascular disease, is a risk factor for BBBd and CIE. Aguirre et al., reported a case of CIE with severe intraoperative arterial HTN during EVT, in a patient with multiple risk factors for microvascular disease. Reference Aguirre, Trillo, Ximénez-Carrillo and Vivancos17 Whisson et. al., induced arterial HTN (190 mmHg peak systolic pressure) during carotid angiography in rodents and reported an association with increased BBBd. Reference Ivens, Gabriel, Greenberg, Friedman and Shelef58 While it is standard to address intra-procedural blood pressure optimization and be mindful of contrast volume administered, our cases highlight the occurrence of these variables among patients with CIE.
All our patients with AIS had restoration of CBF to the affected vascular territory. A mechanism analogous to cerebral hyperperfusion syndrome (CHS) may have facilitated CIE, as both ischemia and reperfusion injury are known to induce BBBd. BBBd has been documented in animal models of cerebral hyperperfusion, Reference Sakaki, Tsujimoto, Nishitani, Ishida and Morimoto22,Reference Amantea, Russo and Gliozzi38,Reference Chassidim, Veksler, Lublinsky, Pell, Friedman and Shelef59 as well as a clinical case. Reference Chassidim, Veksler, Lublinsky, Pell, Friedman and Shelef59 The perfusion pressure breakthrough threshold in chronically ischemic brains is decreased in the setting of new ischemic insults and HTN, both resulting in BBBd. Reference Sakaki, Tsujimoto, Nishitani, Ishida and Morimoto22 Reperfusion of ischemic brain regions may contribute to BBBd and CIE via mechanisms related to impaired autoregulation such as those observed with CHS.
Long procedures could potentially result in downstream ischemia, leading to BBBd and secondary CIE, particularly if a support platform is in place for a prolonged period and occlusive in a vessel. In our long cases, however, no contrast stasis was noticed during the injections, indicating a nonocclusive guiding catheter position in the neck vessels. Operative durations in the EVT cases were all under 200 minutes.
Future avenues for research: BBB imaging and CIE-directed therapies
A future cohort study would provide the opportunity for more comprehensive multi-variate analysis. The inclusion of a control group in future studies may allow for analyses assessing variables predictive of CIE. Dynamic-contrast enhanced MRI (DCE-MRI) has been used to describe the extent and localization of BBBd in patients with neurological conditions, including TIA and stroke. Reference Serlin, Ofer and Ben-Arie34,Reference Yamasaki, Matsuura, Shozuhara, Onodera, Itoyama and Kogure60 DCE-MRI may be useful for the diagnostic work-up of patients with suspected CIE and serve as a biomarker for measuring CIE-directed treatment efficacy.
Previously described CIE-directed therapies include corticosteroids, vigorous hydration and blood pressure optimization. Reference Quintas-Neves, Araújo and Xavier5 In the absence of standardized guidelines for the treatment of CIE, these options are reasonable supportive adjuncts. Airway protection, anticonvulsants and hyperosmolar therapy may be considered on an as needed basis. The optimal duration and intensity of such therapies warrant further study. The utility of such therapies likely depends on timely diagnosis and education around CIE, which may improve diagnostic recognition and facilitate early treatment.
Future studies may explore therapies for CIE that target pathological mechanisms underlying BBBd. These may include inhibition of proinflammatory cytokines, CSDs or MMP release. Inhibition using IL-1-receptor antagonists has been shown to attenuate brain damage induced by stroke, Reference MacLean, Muradov and Greene61 whereas inhibition of MMP-9 activity prevents cytokine maturation and reduces ischemic brain volume. Reference Amantea, Russo and Gliozzi38 Lastly, CSDs are associated with massive release of glutamate and increased BBBd. Reference Takizawa, Qin and Lopes de Morais53 NMDA-receptor antagonism has been shown to inhibit CSD, Reference Sakowitz, Kiening and Krajewski62,Reference Reinhart, Morton, Brennan, Carlson and Shuttleworth63 reduce BBBd, Reference Vazana, Veksler and Pell49 and improve neuronal recovery following CSD in peri-infarct brain tissue.64
Limitations
Given the small sample size, the analysis is limited to univariate, nonparametric comparisons. As described above, our future work includes conducting a cohort study with a control group to elicit pre- and peri-operative variables predictive of developing CIE. The time course for obtaining postoperative radiographic imaging was nonstandardized and driven by findings on neurological assessment in this retrospective series. This reflects lack of wide-spread consensus on the definition of CIE and absence of guidelines pertaining to the work-up and management.
Conclusion
Here, we describe our institutional experience with CIE following endovascular surgery for cerebrovascular disease. Prior animal and human data support a mechanistic role for BBBd in CIE. Risk factors for BBBd may increase the risk of CIE. These include preexisting microvascular pathology, neuroinflammation and altered CBF dynamics, and procedural variables such as intraoperative arterial HTN, contrast concentration and the use of endovascular devices for stenting or clot extraction that may induce mechanical endothelial injury. Aggressive control of risk factors for microvascular disease, including arterial blood pressure control, may reduce the risk of CIE.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/cjn.2024.38.
Data availability statement
All data utilized is provided herein and available on request from the corresponding author Dr. Mark A. MacLean (mark.maclean@dal.ca).
Acknowledgements
None.
Author contributions
All authors contributed to conceptualization; MM, GP, AW, PR, and DV participated in data collection; MM, JM, and RG performed the data analysis; MM and JM performed the literature review; all authors contributed to manuscript preparation and reviewed the manuscript for integrity and accuracy.
Funding statement
This study was not funded.
Competing interests
None.
Ethical approval
Consent for study inclusion was obtained directly from each patient and/or their substitute decision makers.
Consent
All authors consent to publication.