Antipsychotic medication plays a critical role in the management of psychosis, particularly in individuals recovering from a first episode of psychosis (FEP). Reference Keepers, Fochtmann, Anzia, Benjamin, Lyness and Mojtabai1 Standard care typically involves maintaining antipsychotic treatment for 1–5 years following remission from FEP, to reduce the risk of relapse. Reference Hui, Chen, Verma, Tagata, Mizuno and Liu2 However, a key challenge for both individuals with FEP and clinicians arises after remission: the decision to continue or discontinue antipsychotic medication. Reference Thomas, Suarez, Lucksted, Siminoff, Hurford and Dixon3,Reference Kaar, Gobjila, Butler, Henderson and Howes4 This decision involves balancing the risks of relapse against the side-effects and long-term consequences of continued medication use, such as weight gain, metabolic disturbances and tardive dyskinesia. Reference Hui, Lam, Lee, Chan, Chang and Suen5,Reference Béchard, Corbeil, Malenfant, Lehoux, Stip and Roy6 Moreover, how prolonged medication use affects quality of life and social functioning in the long term remains uncertain. Reference Béchard, Desmeules, Bachand, Huot-Lavoie, Corbeil and Anderson7 Increasingly, psychosis management is recognised as a multidimensional process, where decisions are made not only to control symptoms, but also to support recovery goals, such as social reintegration, autonomy and the pursuit of life aspirations. Reference Warner8
In this context, shared decision-making has emerged as a key principle in modern mental healthcare. Reference Slade9 Shared decision-making is a collaborative process in which individuals and clinicians jointly explore treatment options, considering clinical evidence, individual preferences, values, and life circumstances. Reference Coulter and Collins10 It is especially relevant in situations where multiple treatment options exist, each with their own risks and benefits – such as the decision to stop or continue antipsychotic medication after remission from FEP. Shared decision-making empowers patients, Reference Stovell, Morrison, Panayiotou and Hutton11 fosters a stronger therapeutic alliance Reference Matthias, Fukui, Kukla, Eliacin, Bonfils and Firmin12 and has been associated with improved treatment adherence and satisfaction. Reference Renes, Metz, Nolen, Hoogendoorn, Kupka and Regeer13,Reference Pérez-Revuelta, González-Sáiz, Pascual-Paño, Mongil-San Juan, Rodríguez-Gómez and Muñoz-Manchado14 Despite these benefits, shared decision-making is underutilised in mental healthcare. Reference Hamann, Mendel, Cohen, Heres, Ziegler and Bühner15,Reference Coulter16
Patient decision aids (PDAs) offer a potential solution to this challenge. PDAs are tools designed to facilitate shared decision-making by providing people with clear, accessible information about their treatment options, helping them weigh the pros and cons of each option, and clarifying their values and preferences. Reference Stacey, Légaré, Lewis, Barry, Bennett and Eden17 They have been shown to reduce decisional conflict, improve patient knowledge and enhance participation in healthcare decisions. Reference Stacey, Légaré, Lewis, Barry, Bennett and Eden17 Currently, three PDAs support decisions about antipsychotic treatment in stable non-affective psychosis. Reference Kaar, Gobjila, Butler, Henderson and Howes4,Reference Zisman-Ilani, Shern, Deegan, Kreyenbuhl, Dixon and Drake18–Reference Dixon, Holoshitz and Nossel20 One helps individuals changing an antipsychotic medication by reviewing past experiences, prioritising symptom management, evaluating side-effects and considering administration preferences. Reference Kaar, Gobjila, Butler, Henderson and Howes4 The second PDA helps individuals with stable schizophrenia decide whether to reduce or maintain their antipsychotic treatment, specifically comparing the risks and benefits of dose reduction, monotherapy or continued polypharmacy. Reference Aoki, Takaesu, Matsui, Tokumasu, Tani and Takekita19,Reference Dixon, Holoshitz and Nossel20 A third PDA guides decisions about continuing, adjusting or stopping antipsychotics, focusing on relapse risk, side-effects and daily functioning. Reference Zisman-Ilani, Shern, Deegan, Kreyenbuhl, Dixon and Drake18 Although these PDAs are valuable resources, they do not fully address the broader recovery-oriented needs of individuals in remission from FEP. Considering recovery goals – such as autonomy, social reintegration and life aspirations – is essential because these goals reflect what individuals in remission from FEP value most in their lives. Treatment decisions that align with these priorities can enhance engagement, foster a stronger sense of control and support long-term well-being. Reference Leamy, Bird, Le Boutillier, Williams and Slade21 For many, decisions concerning antipsychotic medication go beyond relapse prevention; it is about reclaiming a meaningful and self-directed life. Reference Witteman, Maki, Vaisson, Finderup, Lewis and Dahl Steffensen22
The purpose of this article is to describe the development of a PDA designed for individuals in remission from FEP to support decisions about continuing, reducing or stopping antipsychotic medication. Developed through a collaborative process, the PDA uniquely emphasises personal recovery, addressing autonomy, life goals and functional well-being alongside clinical outcomes. By focusing on the individual’s perspective, it aims to promote shared decision-making that respects both evidence-based guidance and each person’s recovery journey.
Method
Study design
We used a user-centred, multi-phase, mixed-methods design to develop a PDA for individuals in remission from FEP who are considering whether to continue, reduce or discontinue antipsychotic medication. The development process was guided by the 2021 International Patient Decision Aid Standards (IPDAS), including the minimum criteria for balanced, evidence-based and transparent information, Reference Joseph-Williams, Newcombe, Politi, Durand, Sivell and Stacey23,Reference Béchard, Anderson, Corbeil, Huot-Lavoie, Brodeur and Massé24 and the CHIME recovery framework, which emphasises Connectedness, Hope, Identity, Meaning and Empowerment to ensure the PDA supports patients’ broader recovery goals. Reference Witteman, Maki, Vaisson, Finderup, Lewis and Dahl Steffensen22 The process began with a systematic review and meta-analysis synthesising evidence on clinical outcomes associated with antipsychotic continuation or discontinuation, Reference Béchard, Desmeules, Bachand, Huot-Lavoie, Corbeil and Anderson7 followed by a qualitative needs assessment to explore the decision-making process and identify key motivators and support needs. Reference Witteman, Vaisson, Provencher, Chipenda Dansokho, Colquhoun and Dugas25 A multidisciplinary steering group and an external advisory group that included potential end users contributed throughout the development process, from conceptualisation to iterative refinement. Members of both groups were identified through purposive sampling of individuals with relevant expertise, facilitated through our professional and lived experience networks. Field testing in clinical practice has not yet been conducted, and will be the subject of future research.
Multidisciplinary steering group
To guide the PDA’s development, we assembled a multidisciplinary steering group, comprising two clinical pharmacist researchers (L.B., M.-F.D.), one psychiatrist researcher (M.-A.R.), one researcher in psychosocial dimensions of medication use (S.L.), one mental health services researcher with expertise in shared decision-making (M.M.) and one patient partner in research (J.M.). The group met regularly throughout the development process to conceptualise the PDA, define its objectives and ensure its alignment with both clinical priorities and personal recovery goals. Throughout the meetings, the perspectives of the member with lived experience (J.M.) were actively solicited and fully integrated into all decisions. After each iterative cycle, the group reviewed updated prototypes, analysed feedback from the external advisory group, and provided input to improve clarity, relevance and usefulness. The steering group also used the IPDAS 2.0 checklist to ensure that the PDA met international standards for balance, accessibility and evidence-based content.
External advisory group
The external advisory group was composed of three patient partners in research, one family caregiver and three healthcare professionals (two psychiatrists and one nurse practitioner specialising in psychiatry: O.R., S.B., E.B.). The group contributed end-user perspectives by reviewing each version of the PDA during its development (see Fig. 1). Individually, members provided written feedback and completed structured questionnaires assessing comprehension, relevance, ease of use and perceived usefulness. Their input – grounded in lived, caregiving and clinical experience specifically related to antipsychotic decisions after remission from FEP – helped refine the content and format to better meet the needs of its intended users.
Iterative development process of the patient decision aid (PDA) prototype. The PDA was refined through repeated cycles of drafting, review by a multidisciplinary steering group and feedback from an external advisory group, until a final version was reached.

Scope
The PDA was developed for adults in remission from non-affective or affective psychosis who are currently treated with antipsychotics, ideally after 1 year of remission from an FEP. However, it may also be used at other points in remission, with the expectation that clinicians adapt the information and discussion accordingly. The tool supports decision-making among individuals, caregivers and early intervention clinicians considering three options: discontinuing antipsychotics, continuing at full dose or reducing the dose.
PDA content
Structure and frameworks
The PDA is designed for use during medical consultations, where members of the clinical team can support individuals in understanding the information provided when needed. To facilitate its use, a manual for clinicians was also developed and is available upon request. The PDA’s design and content were structured using the IPDAS minimal criteria (version 4.0), which define the minimum criteria for high-quality decision aids, including accuracy, balance, transparency and evidence-based content. Reference Béchard, Anderson, Corbeil, Huot-Lavoie, Brodeur and Massé24 The development was further informed by the User-Centered Design 11-item measure (UCD-11) checklist, which outlines best practices for user-centred design through iterative feedback, real-world relevance and involvement of potential users at multiple stages. Reference Speyer, Westergaard, Albert, Karlsen, Stürup and Nordentoft26 The qualitative study helped identify key decisional needs, common motivators and the overall decision-making process, informing what content to include, how to present options and who should be involved in the discussion. The CHIME framework further grounded the tool in personal recovery principles: it shaped the value clarification section around life priorities (Hope, Meaning); guided the inclusion of examples such as returning to work or regaining autonomy; supported the use of empowering, non-directive language (Empowerment); and emphasised the importance of involving trusted individuals in the process (Connectedness).
Evidence selection
Development of the PDA content on treatment options and outcomes relied on robust evidence from several studies, prioritising meta-analyses or clinical trials to ensure the highest level of scientific rigour. Our content development strategy drew on prior work from our team, including a published meta-analysis on the effects of antipsychotic discontinuation in FEP, Reference Béchard, Desmeules, Bachand, Huot-Lavoie, Corbeil and Anderson7 which documented key outcomes such as hospital admission rates, quality of life and recovery. Additional meta-analyses provided evidence on relapse risks, evaluated the reversibility of weight gain after antipsychotic discontinuation and assessed the risks of tardive dyskinesia and other adverse effects, thus addressing aspects of patients’ physical and mental well-being. Reference Hui, Chen, Verma, Tagata, Mizuno and Liu2,Reference Kishi, Ikuta, Matsui, Inada, Matsuda and Mishima27–Reference Shaffer, Brodney, Gavaruzzi, Zisman-Ilani, Munro and Smith35 When meta-analyses or systematic reviews were unavailable, we followed the evidence-based medicine hierarchy – prioritising randomised controlled trials, then non-randomised trials and observational studies – to select the most relevant available evidence, favouring studies conducted in FEP settings or, when necessary, among individuals with schizophrenia or antipsychotic users. The multidisciplinary steering group reviewed and discussed evidence to ensure the PDA content accurately reflected both patient and clinical priorities, with experts providing critical insights on the selected references. Relapse and hospital admission were treated as dichotomous outcomes, consistent with how these events are reported in the literature.
Narrative integration
To enhance relatability and support a values-based decision-making process, three narrative stories were incorporated into the PDA. These stories emphasise that there is no universally correct choice, only what is right for each individual. Reference Witteman, Vaisson, Provencher, Chipenda Dansokho, Colquhoun and Dugas25,36 They were inspired by real-life experiences from a qualitative study participant, a member of the external advisory group and a member of the multidisciplinary steering group with lived experience. All stories were co-written with the steering group member with lived experience, and reviewed by the external advisory group to ensure authenticity, diversity and person-centredness.
Language and readability
The French version of the PDA was professionally translated into English by a certified translator with lived experience of psychosis (N.P.), who also reviewed the tool to ensure clarity, relevance and inclusivity. She reported that engaging with the PDA prompted personal reflection and found it meaningful, offering informal validation of its usefulness from an end-user perspective.
To ensure adequate readability, we worked with the SHeLL Editor (web-based tool; Sydney Health Literacy Lab, University of Sydney, Sydney, Australia; https://shell.techlab.works/) to identify complex terms and replace them with simpler synonyms, resulting in a more accessible version. The readability of the English versions were assessed using the readability calculator Online-Utility.org (web-based tool; https://www.online-utility.org/english/readability_test_and_improve.jsp). Reference O’Connor and Cranney37 The grade reading level was estimated by averaging the SMOG and Gunning Fog scores, which indicate the number of years of formal education required to understand the text.
Iterative process
The PDA was developed through a three-cycle, user-centred iterative process involving both the multidisciplinary steering group and an external advisory group (see Fig. 1). Based on initial input from the multidisciplinary steering group – focused on structure, content accuracy, clarity and alignment with recovery-oriented and shared decision-making principles – version 1 of the PDA prototype was created. This version was then sent individually to each member of the external advisory group for review. They provided written feedback and completed structured questionnaires. All feedback was systematically documented in a tracking table (see Supplementary Material 1), where each comment was linked to either a corresponding modification in the next version or a justification when no change was made. All changes made in response to advisory group feedback were explicitly highlighted in the updated prototype to ensure transparency. The revised version was then returned to the steering group, discussed and refined in the next iteration. This cycle of feedback, documentation and revision was repeated three times until consensus was reached on the final version of the PDA.
Acceptability assessment
To evaluate acceptability, we administered two distinct questionnaires adapted from the Ottawa Hospital Research Institute Reference Zisman-Ilani, Parker, Thomas, Suarez, Hurford and Bowen38 to members of the external advisory group. Members with lived experience (n = 4) rated each section of the PDA (such as ‘What is a patient decision aid?’, ‘What are the pros and cons of each option?’, ‘How does this choice align with your priorities?’ and ‘Create an action plan’), using a five-point scale ranging from ‘poor’ to ‘excellent’. Members with clinical expertise (n = 3), on the other hand, completed a 14-item satisfaction questionnaire using a five-point Likert scale (1 = ‘strongly disagree’ to 5 = ‘strongly agree’) to assess the tool’s usability and perceived effectiveness (see Supplementary Material 2–5). Responses were summarised using descriptive statistics.
Additionally, an informal meeting was held with 31 clinicians from the Clinique Notre-Dame des Victoires, an early intervention clinic for FEP in Quebec City that follows approximately 250 individuals, to present the final version of the PDA and gather further feedback. Attendees included case managers from various professional backgrounds (such as nursing, occupational therapy and social work), one peer support worker, pharmacists, psychiatrists and psychologists.
Ethics statement
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2013. All procedures involving human subjects/patients were approved by the CÉR-S en neurosciences et santé mentale du CIUSSS-CN (approval number #MP-13-2021-2325). Formal consent was not required for this study, as determined by the institutional research ethics board. However, all participants involved in the steering group and external advisory group still provided written agreement to participate via email.
Results
IPDAS criteria and user-centredness
The prototype developed (see Supplementary Material) meets all IPDAS version 4.0 qualifying and certification criteria, Reference Béchard, Anderson, Corbeil, Huot-Lavoie, Brodeur and Massé24 and 21 out of 28 of the quality criteria. Similarly, 9 of the 11 UCD-11 items were met. Reference Joseph-Williams, Newcombe, Politi, Durand, Sivell and Stacey23,Reference Speyer, Westergaard, Albert, Karlsen, Stürup and Nordentoft26 The remaining criteria were not achieved solely because the prototype has not yet undergone field testing with patients and clinicians (see Supplementary Material 6 and 7).
Feedback and iterative revisions
Across three multidisciplinary steering group meetings, rigorous notes were taken and all suggestions were carefully reviewed and integrated. This iterative process led to substantial improvements in the PDA’s content, tone and format. Revisions focused on enhancing clarity, reducing stigma, improving personalisation and supporting patient engagement at different stages of the decision-making process. Drawing on themes from our qualitative study currently under review, discussions also explored how self-stigma – and related experiences, such as internalised negative views of psychosis, medication as a reminder of illness or a perceived shift in identity – might influence the desire to stop medication. Additional materials, such as a clinician guide, were also developed to support implementation.
Across the three versions of the PDA, a total of 143 comments were submitted by the external advisory group. For version 1, 101 comments were received, with 66 (65%) leading to revisions aimed at improving clarity, refining terminology (e.g. remission, treatment options) and better aligning examples with recovery goals. Suggestions not incorporated were typically beyond the tool’s scope, redundant or inconsistent with its purpose. In version 2, 19 comments were received, of which 14 (74%) prompted changes, including the addition of experiential knowledge, clarification of clinical concepts (e.g. insight, defined daily dose) and population details. Broader issues, such as comorbidities and layout, were noted, but not fully addressed. For version 3, 23 comments were submitted, resulting in 12 (52%) modifications focused on wording and clarifications, whereas broader suggestions like expanding to other diagnoses or including external guides were not adopted because of scope limitations.
The final version of the PDA was presented to 31 clinicians from early intervention services during an informal meeting. Although no formal evaluation was conducted and no specific comments were received, the overall reception was broadly positive, reinforcing the tool’s perceived relevance and usability in clinical practice.
Description of the final PDA
The final prototype is a 15-page, paper-based tool designed to be introduced during clinical encounters. It includes a definition of patient decision aids, rationale for the decision, presentation of the three main options (continue, reduce or stop antipsychotics), pros and cons by domain (mental health, life impact, short- and long-term side-effects), values clarification, summary alignment with user priorities, action planning and three real-life narratives (see Supplementary Material). Although initially presented in a clinical setting, the tool is designed to support ongoing reflection afterward. It is recovery-oriented in that it helps users explore how each option aligns with their life goals, encourages hope through concrete examples (e.g. returning to work, regaining autonomy) and invites the involvement of trusted individuals in the decision-making process, among other features designed to support personal recovery. Reference Witteman, Maki, Vaisson, Finderup, Lewis and Dahl Steffensen22
Acceptability
The PDA received progressively higher ratings across iterations, with the final version scoring highest on nearly all acceptability items. Among respondents with clinical expertise, the largest improvements (0.7 to 1.1 points) were observed for items related to ease of use, clarity, perceived reliability, adaptability, alignment with clinical practice, anticipated benefit/harm ratio and support for informed decision-making (see Table 1). Other items showed minimal change (0 to 0.3 points), but were already rated positively, with average scores around 4, reflecting agreement on compatibility with usual practice, suitability for value-laden choices and partial use of the tool. In contrast, two items – time efficiency (mean: 3.3) and avoiding changes to clinical routines (mean: 3.7) – consistently received lower scores. These findings suggest that although the PDA was viewed as useful and relevant, clinicians did not anticipate time savings and perceived that its use might require adapting existing workflows.
Average acceptability ratings from healthcare providers in the external advisory group across iterative versions

Each value represents the mean score per statement for a given version (1 = strongly disagree; 5 = strongly agree).
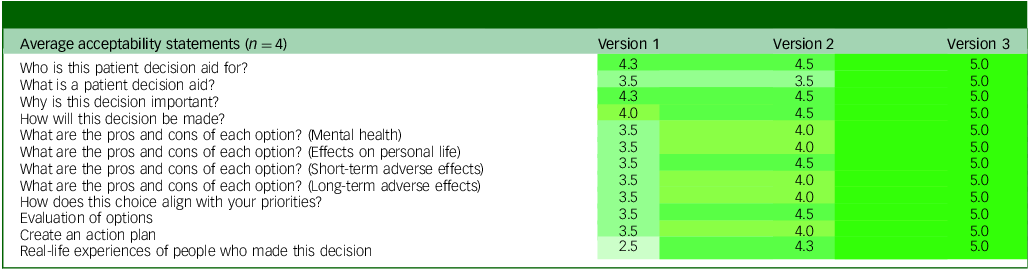
According to the acceptability questionnaire completed by participants with lived experience (see Table 2), the greatest improvements (1.5 to 2.5 points) were observed for sections on the pros and cons of each option, values clarification, action planning and real-life experiences. The narrative stories section showed the largest gain, increasing from a mean score of 2.5 to 5.0 after the addition of a third story and revisions to better reflect diverse experiences. Moderate improvements (0.7 to 1.0 points) were noted for sections such as ‘What is a patient decision aid?’, ‘Why is this decision important?’ and ‘How will this decision be made?’, all of which reached perfect scores in the final version.
Average acceptability ratings from members with lived experience in the external advisory group across iterative versions

Each value represents the mean score per section of the patient decision aid for a given version (1 = mediocre; 5 = excellent).
The final version of the PDA was also considered balanced, with most participants finding the length and amount of information appropriate (Table 3). Six out of seven respondents stated they would have found the tool very or extremely useful when facing the decision to continue or discontinue antipsychotics. Supporting its accessibility, the plain-language version of the PDA achieved an average readability level equivalent to grade 9 (Gunning Fog = 9.00; SMOG = 9.40).
Perceived acceptability of the final version of the decision aid among external advisory group members

Proportions reflect the number and percentage of members (n = 7) who agreed with each acceptability item regarding the final version of the patient decision aid.
The external advisory group described the prototype as a useful, reassuring and accessible resource to support decision-making (see Supplementary Material 8). They highlighted its ability to foster open and collaborative dialogue among patients, families and clinicians. The following quotes are free translations from French: a caregiver shared, ‘This tool would have helped me better support my sons when this issue became significant for them’. A healthcare professional noted, ‘It makes things easier … it provides the necessary information to make an informed choice’. Users also appreciated the figures and tables and the way the tool encourages reflection on values: ‘It facilitates decision-making through its visual format and the decision balance exercise, which is well-supported by evidence and peer testimonies’. Another participant commented, ‘I would probably have felt more secure in my decision receiving this document, because being encouraged to make an informed choice shows that my opinion and point of view matter’.
Discussion
This article presents the systematic development of a novel, evidence-based PDA to support individuals in remission from an FEP as they consider whether to continue, reduce or discontinue antipsychotic medication. In addition to addressing relapse prevention, the tool emphasises personal recovery goals such as autonomy, hope and social reintegration – priorities frequently reported by individuals with lived experience. Reference Zisman-Ilani, Parker, Thomas, Suarez, Hurford and Bowen38 Its development followed a rigorous, iterative and user-centred process, aligned with IPDAS standards and guided by input from a multidisciplinary team and stakeholders with both clinical and lived experience.
Key contributions and innovations
Although several PDAs exist to support decisions about antipsychotic treatment, Reference Kaar, Gobjila, Butler, Henderson and Howes4,Reference Zisman-Ilani, Shern, Deegan, Kreyenbuhl, Dixon and Drake18–Reference Dixon, Holoshitz and Nossel20 ours is the first developed specifically for individuals in remission from an affective or non-affective FEP. Unlike tools focused on switching or reducing an antipsychotic medication, or designed for broader psychosis populations, our PDA addresses the full range of options (continuing, reducing or discontinuing) within the context of early recovery. This is particularly important, as some individuals express a desire to stop treatment – a need previously acknowledged, but not directly addressed in existing tools. Reference Kaar, Gobjila, Butler, Henderson and Howes4 In our PDA, discontinuation is stated explicitly because many individuals are unaware that it is even a possibility, whereas in practice, this option is operationalised through a gradual taper.
Our PDA places stronger emphasis on personal recovery goals, involves trusted others in the decision-making process, and encourages ongoing reflection beyond the clinical encounter. When disagreement occurs, the PDA helps make differences in preferences and concerns explicit so that the person and clinician can explore them together and work toward a mutually acceptable plan. It was co-developed with individuals with lived experience and guided by the CHIME framework, ensuring that recovery-oriented values are embedded throughout. Notably, our PDA is the only one to include narrative stories – developed in response to needs identified in our qualitative study – which help users relate to different recovery paths and support values-based deliberation. Reference Witteman, Vaisson, Provencher, Chipenda Dansokho, Colquhoun and Dugas25
Although our tool shares aims with the Antipsychotic Medication Decision Aid (APM-DA) Reference Zisman-Ilani, Shern, Deegan, Kreyenbuhl, Dixon and Drake18,Reference Zisman-Ilani, Parker, Thomas, Suarez, Hurford and Bowen38–Reference Simonsen, Åsbø, Slade, Wold, Widing and Flaaten40 – the only other PDA specifically addressing continuation, adjustment or discontinuation post-remission – there are key differences. The APM-DA was pilot-tested in an FEP setting with good results, Reference Zisman-Ilani, Parker, Thomas, Suarez, Hurford and Bowen38 but it was developed with a broader psychosis population. In contrast, our PDA was designed from the ground up for FEP, a population with typically better outcomes and for whom the evidence base is more limited. Reference Simonsen, Åsbø, Slade, Wold, Widing and Flaaten40 This makes the clear communication of uncertainty especially important, and highlights the need for tailored decision support in early intervention settings.
Strengths and limitations
This study has several strengths. The PDA was developed using a transparent, iterative and user-centred process grounded in established frameworks (UCD-11, IPDAS and CHIME), with meaningful input from individuals with lived experience, clinicians and researchers. It is one of the few tools specifically designed for individuals in remission from an FEP, integrating both clinical evidence and recovery-oriented perspectives. The PDA supports reflection beyond the clinical encounter and addresses a wide range of outcomes, from relapse prevention to personal goals such as autonomy and social reintegration. Additionally, the inclusion of narrative stories based on real experiences responds to needs identified in our qualitative research, and enhances the tool’s relatability and potential to reduce stigma. Finally, the development process is documented in detail, with successive versions and justifications for changes available in the Supplementary Material.
This work has some limitations. Although the PDA benefited from strong participation by individuals with lived experience, the composition of the advisory and steering groups lacked ethnocultural diversity, as all members were Quebec-born. This may have limited the range of perspectives integrated into the tool. Future development should aim to include more diverse voices to enhance cultural sensitivity and inclusivity. In addition, although the narrative stories were revised to improve relatability, they may not reflect the full spectrum of patient experiences. Some clinicians expressed theoretical concerns about time demands and workflow integration based solely on reviewing the prototype, highlighting the need for future implementation research once the PDA is field-tested. Finally, although the tool was developed for individuals in remission from FEP, its applicability to other diagnostic groups remains to be tested.
In conclusion, this recovery-oriented PDA provides an evidence-based tool to help individuals in remission from FEP make informed decisions about the continuation or discontinuation of antipsychotic treatment. Rather than promoting a single ‘right’ choice, it encourages reflection on personal values and goals, with clinicians providing support through empathy, clear communication and the best available evidence. Next steps include pilot-testing the PDA in early intervention settings through a randomised trial to evaluate its impact on decision quality, patient satisfaction, treatment engagement, clinical outcomes such as rates of relapse, and recovery outcomes. Future adaptations may include tailoring the tool for individuals with chronic psychosis and developing digital formats to improve accessibility.
Supplementary material
The supplementary material is available online at https://doi.org/10.1192/bjo.2026.11034. The supplementary materials for this paper are published under a CC-BY NC licence: © The Author(s), 2026. Published by Cambridge University Press on behalf of Royal College of Psychiatrists. This is an Open Access article, distributed under the terms of the Creative Commons Attribution-NonCommercial licence (https://creativecommons.org/licenses/by-nc/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited. The written permission of Cambridge University Press must be obtained for commercial re-use.
Data availability
The data that support the findings of this study are available from the corresponding author, L.B., upon reasonable request.
Acknowledgements
We would like to express our gratitude to Suzanne Peloquin, Luc Vigneault, Louis-David Gingras and Marie-Ève Côté for their insight and contribution to this work.
Author contributions
L.B., M.-F.D., M.-A.R., S.L. and M.M. conceptualised the study and guided the overall methodological approach. L.B. led the development of the PDA prototype, with input from the multidisciplinary steering group composed of L.B., M.-F.D., M.-A.R., S.L., M.M. and J.M. L.B., O.R., S.B., E.B., J.M., N.P., M.M., M.-A.R., S.L. and M.-F.D. contributed to interpreting the results and refining the PDA’s iterative versions. L.B. drafted the manuscript. O.C., M.H.-L., O.R., S.B., E.B., J.M., N.P., A.M.A., M.M., M.-A.R., S.L. and M.-F.D. reviewed and revised the manuscript. S.L., M.-F.D., M.-A.R. and M.M. provided administrative, methodological and technical support throughout the project. All authors have reviewed and approved the final version of the manuscript.
Funding
This research received no specific grant from any funding agency or commercial or not-for-profit sectors.
Declaration of interest
L.B. has received a grant from Fonds de recherche du Québec–Santé (FRQS) in partnership with Unité de Soutien SSA du Québec (grant number 331332). O.C. is currently in receipt of a fellowship award from the Canadian Institutes of Health Research (award number 202210MFE-491926-64860). M.H.-L. is supported by the Frederick Banting and Charles Best Canada Graduate Scholarship Doctoral Awards from the Canadian Institute of Health Research (award number FID-172598) and by a Mitacs grant (grant number IT34510). M.-A.R. has received unrestricted grants from Mylan Canada and the Otsuka-Lundbeck Alliance Canada. He has served on advisory boards for AbbVie, Janssen and the Otsuka-Lundbeck Alliance. He has also been a lecturer for AbbVie and Janssen Canada. M.-F.D. reports consulting fees from AbbVie. She has also served on advisory boards and been a lecturer for AbbVie and the Otsuka-Lundbeck Alliance Canada. S.L. is a research scholar with funding from the FRQS in partnership Unité de Soutien SSA du Québec (grant number 313085). S.L. has received unrestricted research grants from Eli Lilly Canada and Pfizer Canada for other studies not related to the one presented in this manuscript. All other authors have no competing interests to declare.





eLetters
No eLetters have been published for this article.