Ageing represents an irreversible and intricate process, a primary risk factor for many significant human diseases(Reference van Deursen1). As the population ages, healthcare costs increase(Reference Atella, Piano Mortari and Kopinska2). However, individuals with the same chronological age (CA) exhibit variations in the rate of ageing and various susceptibilities to many age-related diseases, indicating that CA is not a perfect measurement(Reference Lowsky, Olshansky and Bhattacharya3). Therefore, there is a need for a better understanding of the ageing process and the identification of the determinants of biological ageing.
The dysbiosis of the microbiota related to ageing contributes to the reshaping of immune responses during the ageing process(Reference Ling, Liu and Cheng4). It is accompanied by numerous age-related diseases both within and outside the gastrointestinal tract. The modifiable regulation of the gut microbiota suggests that it is a potential target for interventions in ageing(Reference Ling, Liu and Cheng4). Meanwhile, nutrition is considered an effective regulatory factor influencing health and ageing, and a new discipline has been established: ‘Nutrigerontology’, which combines insights from biogerontology, nutrition and medicine to understand the impact of diet and nutrition on the ageing process and age-related diseases(Reference Verburgh5). Diet is also a key modifiable factor influencing gut microbiota composition(Reference David, Maurice and Carmody6).
Researchers utilised dietary data from the Nurses’ Health Study II (NHSII) to construct a gut microbiota-derived dietary pattern: the sulphur microbial dietary (SMD) pattern, which is characterised by a low intake of fruits, vegetables, legumes, whole grains and nuts, along with a high intake of red meat, processed meat, high-fat dairy products, sugary beverages and coffee. Among them, processed meat, liquor and low-energy drinks were found to be positively associated with the enrichment of sulphur-metabolising microbes, while the remaining five components showed an inverse association(Reference Nguyen, Cao and Hur7). Sulphur-metabolising microbiota can convert dietary sulphur into genetically toxic hydrogen sulfide (H2S), which can cause DNA damage(Reference Attene-Ramos, Wagner and Gaskins8) and promote alterations in immune cells associated with inflammation and cancer(Reference Ramasamy, Singh and Taniere9). The association between SMD and various diseases such as non-alcoholic fatty liver disease(Reference Liu, Huang and Ruan10) and obesity(Reference Liu, Wan and Zhang11) has been studied in the UK Biobank. However, little is known about its association with ageing.
‘Biological age (BA)’(Reference López-Otín, Blasco and Partridge12) is crucial in understanding ageing, providing a comprehensive evaluation of ageing across multiple bodily systems(Reference Ferrucci, Gonzalez-Freire and Fabbri13) and measuring the accumulation of damage over time in individuals(Reference Levine14). Markers of BA exhibit significant predictive power for mortality, age-related ailments and declines in bodily function(Reference Li, Ploner and Wang15). Various BA methodologies such as the Klemera–Doubal (KDM) and the PhenoAge methods were devised to delineate the heterogeneity of ageing based on respiratory, metabolic, renal, immune and cardiovascular functions(Reference Gao, Geng and Jiang16). Numerous studies on BA have emerged, including its association with dietary inflammatory index(Reference Hu, Wang and Huan17), macronutrients(Reference Zhu, Xue and Maimaitituerxun18) and dietary oxidative balance(Reference Wang, Sarker and Cheng19). However, no research has yet explored the association between SMD and BA.
The liver plays an important role in the ageing process through metabolism(Reference Trenell and Marchesini20). Abnormal levels of two common enzymes in the liver, aspartate aminotransferase (AST) and alanine aminotransferase (ALT) may induce hepatocellular damage and consequently lead to DNA damage, which is considered to be one of the important factors in cellular ageing(Reference Kim, Flamm and Di Bisceglie21). Additionally, evidence suggests a positive association between obesity-related indicators – BMI and SMD(Reference Liu, Wan and Zhang11), with obesity also being regarded as a disease that accelerates biological ageing(Reference Etzel, Hastings and Hall22). Therefore, BMI and AST/ALT were considered as possible mediators of the association between SMD and biological age in our study.
To fill this gap, we performed a cross-sectional investigation in a well-established cohort of 71 579 adults in the UK to examine the association of SMD with two forms of AgeAccel, including KDMAccel and PhenoAgeAccel.
Methods and materials
Study population
Data from 500K individuals in the UK Biobank were available in our dataset. The UK Biobank, a large-scale biomedical cohort study, collected comprehensive health information from nearly 0·5 million participants aged 37–73 years across the UK(Reference Sudlow, Gallacher and Allen23). From 2006 to 2010, participants completed a face-to-face interview with professionals, a touch screen questionnaire and a whole-body physical examination and donated biological samples, including blood, urine and saliva at the nearest one of twenty-two assessment centres. All participants provided written informed consent. Participants who met certain criteria were excluded from this study. These criteria included missing dietary information (n 293 373), missing BA information (n 65 784), non-white and genetically related participants (n 31 615) and missing values of covariates (n 40 188). After exclusion, the final analyses included 71 579 individuals (online Supplementary Figure S1). The UK Biobank study had been approved by the North West Multi-Centre Research Ethics Committee (reference number 06/MRE09/65).
Assessment of the sulphur microbial diet score
The Oxford WebQ utilised in the UK Biobank is an online 24-hour diet recall tool comprising questions on the consumption of nearly 200 foods and drinks. Its validity has been established through biomarker validation studies conducted elsewhere(Reference Greenwood, Hardie and Frost24). Participants were asked to complete the questionnaire on five separate occasions between 2009 and 2012, considering the seasonal variation in diet. Subsequently, an average measure was calculated for each participant across all five occasions to mitigate measurement error bias. These occasions included the baseline assessment (April 2009–September 2010, 70 684 participants), online cycle 1 (February 2011–April 2011, 100 574 participants), online cycle 2 (June 2011–September 2011, 83 239 participants), online cycle 3 (October 2011–December 2011, 103 761 participants) and online cycle 4 (April 2012–June 2012, 100 219 participants). In our study, 209 166 participants with complete dietary information were included after excluding those with missing data. For each participant, the average intake of each food item was calculated based on up to five dietary recall surveys.
Previous studies have specifically described the calculation methodology of SMD score(Reference Nguyen, Cao and Hur7). In brief, prior investigations identified forty-three sulphur-metabolising bacterial species carrying genes coding for at least two well-known sulphur-metabolising enzymes. A reduced rank regression was performed to link food intake with the log-transformed abundance of these microbes in the stool. The analysis identified eight food groups significantly associated with sulphur-metabolising bacteria. The finding was further validated in subsequent research and was found to explain 2 % of the variation in Bray-Curtis distances(Reference Wang, Nguyen and Mehta25,Reference Sikavi, Nguyen and Haruki26) . Specifically, certain food groups such as processed meats, liquor and low-energy drinks were positively associated with these bacteria, while others like beer, fruit juice, legumes, vegetables and sweets/desserts were negatively associated. Based on the intake of these foods, the SMD score was calculated by summing the product of beta-coefficients and corresponding serving sizes (online Supplementary Table S1). A higher SMD score reflects closer adherence to this dietary pattern and is considered ‘unhealthy’.
Assessment of the biological age
In this study, BA values were calculated using KDM and PhenoAge methods based on participants from the UK Biobank. The two methods had different purposes. KDM was calculated by performing a series of regressions on the biomarkers of CA, to quantify the decline in system integrity(Reference Gao, Huang and Guo27). PhenoAge was calculated based on biomarkers and mortality prediction scores and CA is used to predict the risk of death(Reference Gao, Huang and Guo27).
In brief, KDM was calculated from forced expiratory volume in one second (FEV1), systolic blood pressure and seven blood chemistry parameters (albumin, alkaline phosphatase, blood urea nitrogen, creatinine, C-reactive protein, glycated Hb and total cholesterol); PhenoAge was calculated based on nine blood chemistries (albumin, alkaline phosphatase, creatinine, C-reactive protein, glucose, mean cell volume, red cell distribution width, white blood cell count and lymphocyte proportion), four of which were the same as KDM. The BA values were calculated using the R package ‘BioAge’ for KDM and PhenoAge. The residual of the regression of BA on CA is used to reflect the age acceleration (AgeAccel), referred to as ‘KDMAccel’ and ‘PhenoAgeAccel’(Reference Kresovich, Garval and Martinez Lopez28). AgeAccel serves as the target outcome in our analysis. Participants with AgeAccel values greater than 0 were considered to have accelerated biological ageing(Reference Wang, Sarker and Cheng19). Based on whether AgeAccel was greater than 0, participants were classified into a binary variable, termed biological age indicators. More information was shown in the online Supplementary information.
Assessment of covariates
To control for potential confounding variables, we included covariates such as age, sex, BMI, Townsend deprivation index, education, income, smoke frequency per day, alcohol frequency per week, hypertension, diabetes and CHD, which may be associated with ageing, as well as dietary factors. At the baseline assessment centre visit, a trained nurse measured the participants’ height and weight, and BMI was calculated by dividing weight in kilograms by the square of height in meters. Townsend deprivation index was an indicator of material deprivation that was calculated based on non-home ownership, non-car ownership, unemployment and household overcrowding. A higher Townsend deprivation index score indicates higher levels of deprivation. Smoke frequency was defined as the maximum number of reported past or current cigarettes (or pipes/cigars) consumed per day. Alcohol frequency per week was determined as the average amount of various types of alcohol consumed per week. Education was defined by the UK Biobank and converted to the International Standard Classification of Education. Income was categorised into five groups (≤ £18 000, 18 000–30 999, 31 000–51 999, 52 000–100 000 and ≥ 100 000). Self-reported information and medical records were used to determine the history of hypertension, CHD and diabetes. Observations with missing values for any covariate were excluded from this study. Online Supplementary information provides detailed measurements of covariates.
Statistical analyses
In the present analysis, the SMD score was categorised into quartiles, and the lowest quartile was considered as the reference category. Values of baseline characteristics across the quartiles of the SMD score were indicated as the means (sd) for continuous variables or percentages (%) for categorical variables, respectively. We initially used generalised linear regression models to investigate the associations between the SMD score quartiles and two forms of AgeAccel and estimate β-coefficient and standard error (se), where AgeAccel served as the outcome variable. The regression analysis was conducted by three models: Model 1 adjusted for age and sex; Model 2 included additional adjustments for BMI, Townsend deprivation index, education, income, smoke frequency per day and alcohol frequency per week; Model 3 further incorporated hypertension, diabetes and CHD. Multiple stratified analyses were applied to evaluate the possible modifying effects of the following factors: age (≥ 60/< 60 years), sex (male/female), smoke status (yes/no) and BMI (≤ 24·9/24·9 to 29·9/≥ 30 kg/m2). Interaction analysis between the stratifying variables and SMD score was tested. The dose–response curves between the SMD score with AgeAccel and biological age indicators were analysed by restricted cubic spline regression. Mediation effect analyses were used to assess associations of SMD score with AgeAccel mediated by BMI and serum AST/ALT ratio.
All statistical analyses were completed through the software R 4·2·1, and statistical significance was determined by a two-sided P-value threshold of less than 0·05.
Results
Baseline characteristics
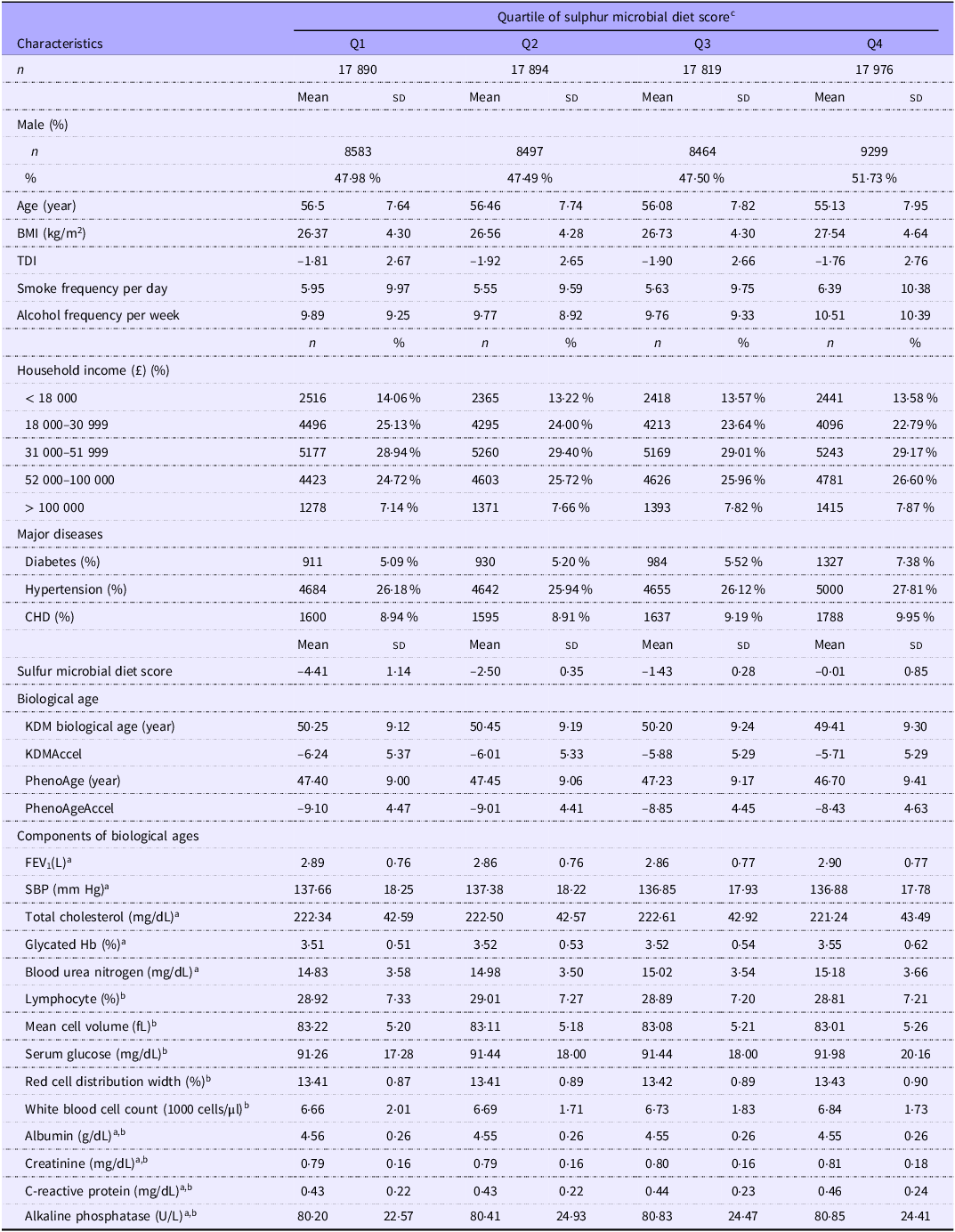
The descriptive statistics of participants are presented in Table 1. Among the 71 579 participants from the UK Biobank, the mean age was 56·04 (sd 7·81) years, with 51·32 % being women at baseline. Participants with a higher SMD score tended to be younger, have a higher household income and BMI, a higher frequency of smoke and alcohol consumption, a higher proportion of major diseases and a higher degree of AgeAccel compared with those with lower adherence.
Basic characteristics of participants from UK Biobank according to the sulphur microbial diet score (Numbers and percentages; mean values and SD)

Data are either percentage or mean (sd) unless indicated otherwise. TDI, Townsend deprivation index; KDM, Klemera–Doubal method; KDMAccel, KDM biological age acceleration; PhenoAgeAccel, PhenoAge acceleration; SBP, systolic blood pressure.
aComponents of KDM biological age. b Components of PhenoAge. c The sulphur microbial diet score was calculated by summing the intake of foods (processed meats, liquor, low-energy drinks, beer, fruit juice, legumes, other vegetables, sweets and desserts) weighted by their regression coefficients (β).
Associations between sulphur microbial diet score quartiles and age acceleration
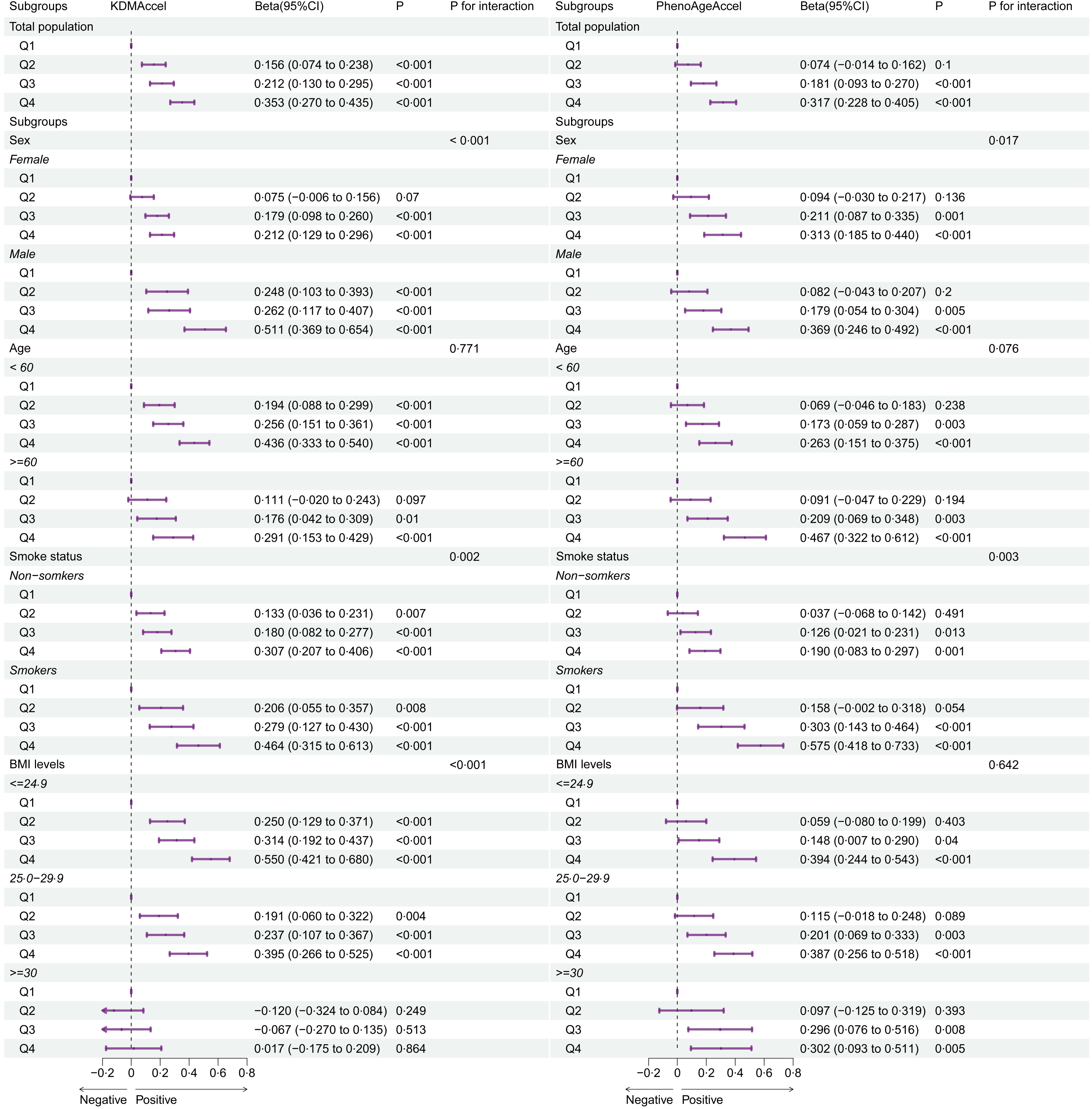
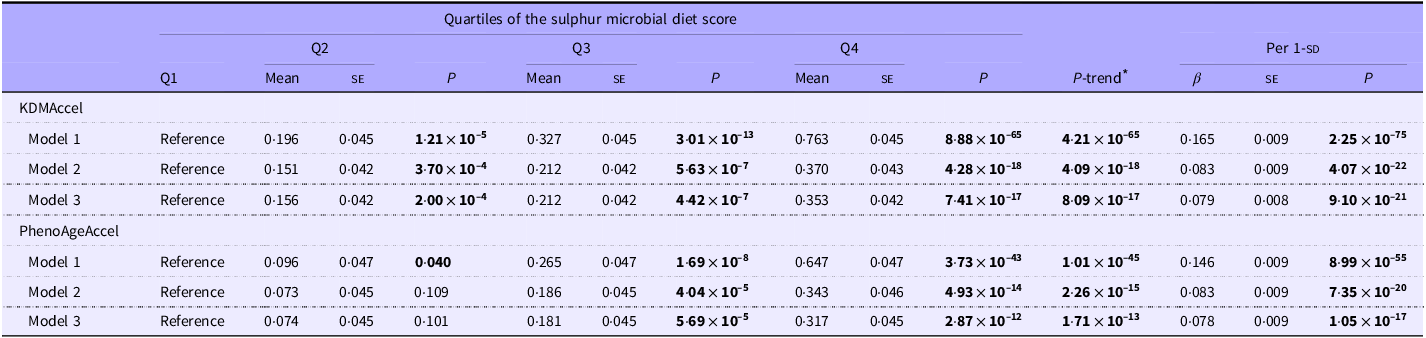
As shown in Figure 1 and Table 2, adherence to SMD was significantly associated with the two forms of AgeAccel (P -trend < 0·05). After adjusting for all covariates, compared with the first quartile of SMD score, β (95 % CI) of KDMAccel was 0·16 (0·07, 0·24; P < 0·001) for the second quartile, 0·21 (0·13, 0·29; P < 0·001) for the third quartile and 0·35 (0·27, 0·44; P < 0·001) for the highest quartile. Meanwhile, β (95 % CI) of PhenoAgeAccel was 0·07 (–0·01, 0·16; P = 0·100) for the second quartile, 0·18 (0·09, 0·27; P < 0·001) for the third quartile and 0·32 (0·23, 0·41; P < 0·001) for the highest quartile.
Forest plot of the association of the sulphur microbial diet score quartiles with two forms of age acceleration and its subgroup analyses. The adjustments involved the covariables selected in the full regression model. Q, quartiles.

Multivariable-adjusted associations between the sulphur microbial diet score and AgeAccel (Coefficient values with their standard errors)

For KDMAccel and PhenoAgeAccel, the effect was shown by a coefficient (β) and standard error (se).
Model 1 was adjusted for age and sex.
Model 2 was further adjusted for BMI, Townsend deprivation index, education, income, smoke frequency per day and alcohol frequency per week.
Model 3 was further adjusted for hypertension, diabetes and CHD.
AgeAccel, age acceleration.
* Tests for trends were conducted using the quartile category as a continuous variable.
The boldface results are statistically significant.
Association of sulphur microbial diet score quartiles with age acceleration stratified by baseline characteristics
Similar associations were detected while conducting extensive stratified analyses based on the variables of age, sex, smoke status and BMI levels. Notably, in Model 3, compared with females (KDMAccel: β Q4v.Q1 = 0·21, 95 % CI: 0·13, 0·30, P < 0·001; PhenoAgeAccel: β Q4v.Q1 = 0·31, 95 % CI: 0·19, 0·44, P < 0·001) and non-smokers (KDMAccel: β Q4v.Q1 = 0·31, 95 % CI: 0·21, 0·41, P < 0·001; PhenoAgeAccel: β Q4v.Q1 = 0·19, 95 % CI: 0·08, 0·30, P < 0·001), the association between SMD quartiles and AgeAccel exhibited greater strength among males (KDMAccel: β Q4v.Q1 = 0·51, 95 % CI: 0·37, 0·65, P < 0·001; PhenoAgeAccel: β Q4v.Q1 = 0·37, 95 % CI: 0·25, 0·49, P < 0·001) and smokers (KDMAccel: β Q4v.Q1 = 0·46, 95 % CI: 0·32, 0·61, P < 0·001; PhenoAgeAccel: β Q4v.Q1 = 0·58, 95 % CI: 0·42, 0·73, P < 0·001). More detailed information is shown in Figure 1.
Best-fitting dose–response curves of the associations of sulphur microbial diet score with age acceleration and biological age indicators
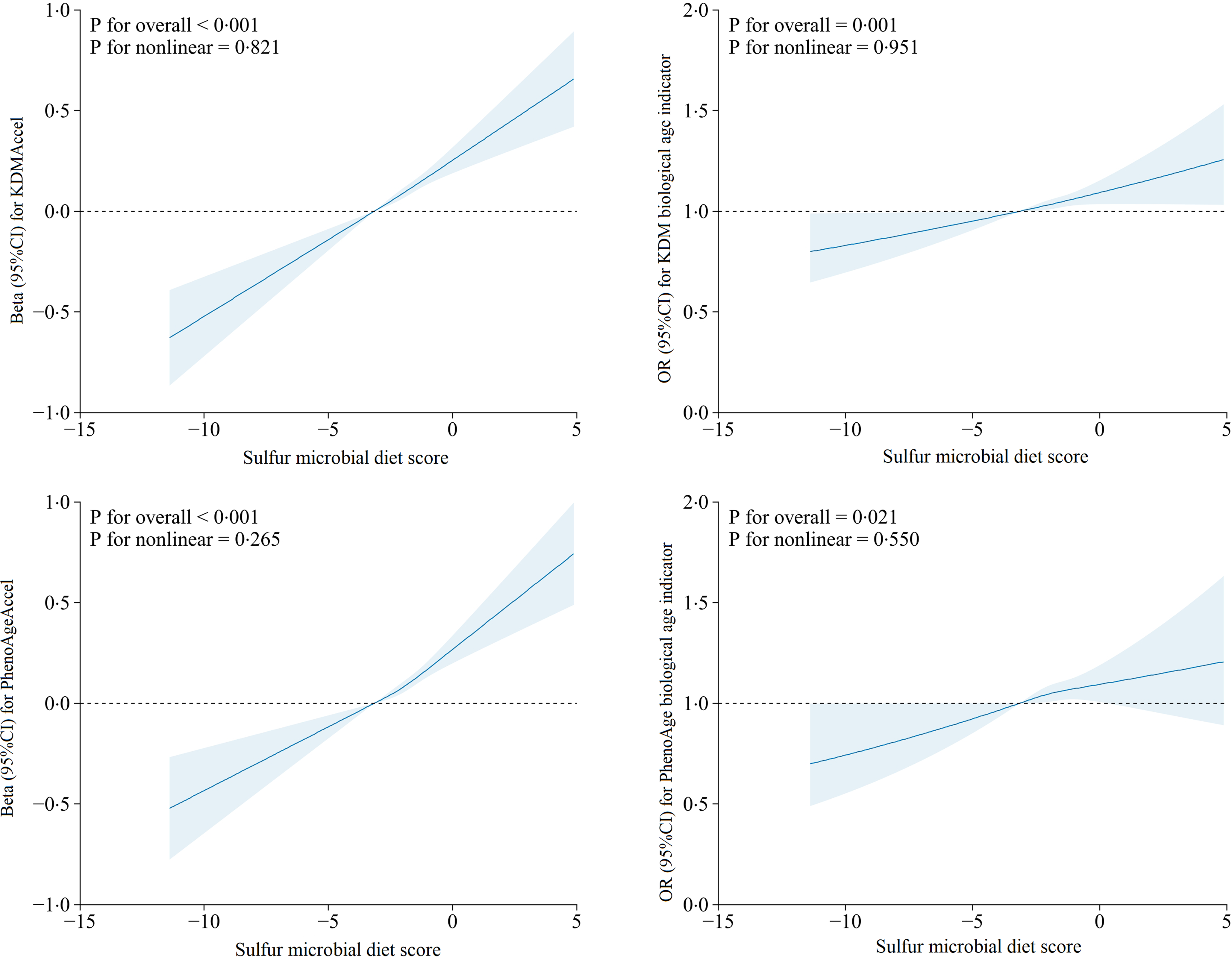
For SMD score, the linearities and dose–response associations with AgeAccel and age indicators were flexibly modelled by conducting restricted cubic spline regression models (Figure 2). After multivariable adjustment, monotonic and linear associations were observed between SMD and KDMAccel (P overall < 0·001, P nonlinearity = 0·821), SMD and KDM biological age indicator (P overall = 0·001, P nonlinearity = 0·951), SMD and PhenoAgeAccel (P overall < 0·001, P nonlinearity = 0·265) and SMD and PhenoAge biological age indicator (P overall = 0·021, P nonlinearity = 0·550).
Association of the sulphur microbial diet score with age acceleration and biological age indicators evaluated by linear and binomial logistic regression models and RCS after adjusted for all covariates. The solid blue lines correspond to the central estimate, and the blue-shaded regions indicate the 95 % CI. RCS, restricted cubic spline.

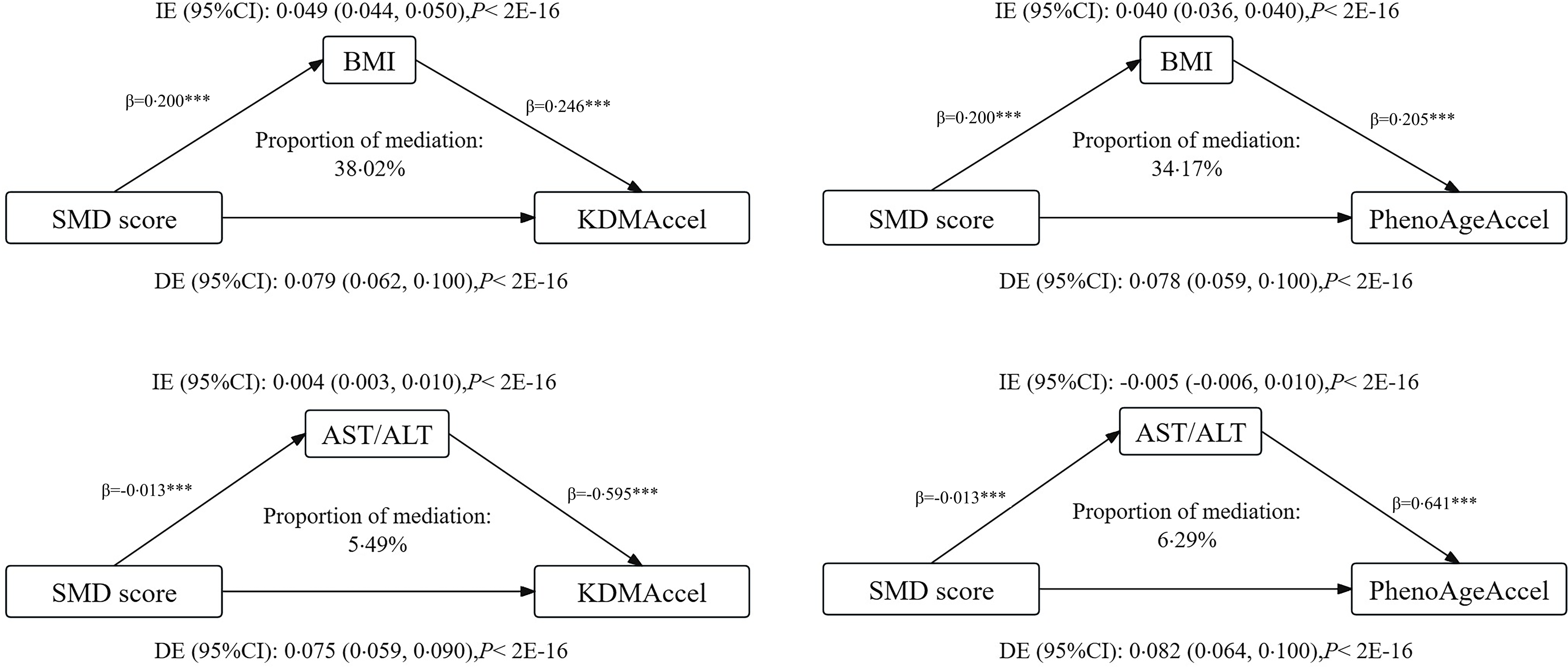
Mediation effects of BMI and aspartate aminotransferase/alanine aminotransferase ratio on the associations of sulphur microbial diet score with age acceleration
Mediation statistical models were performed to ascertain whether BMI and serum AST/ALT ratio had mediation effects on the associations. After adjustment for all the covariables in the linear regression model, the total effect of SMD score on KDMAccel and PhenoAgeAccel were 0·079 (P = 9·10 × 10–21) and 0·078 (P = 1·05 × 10–17) (Table 2). For the SMD score, the indirect effects on KDMAccel and PhenoAgeAccel were mediated by BMI measured at 0·049 (38·02 %) and 0·040 (34·17 %). The indirect effects on KDMAccel and PhenoAgeAccel mediated by AST/ALT were measured at 0·004 (5·49 %) and −0·005 (6·29 %). More information is shown in Figure 3.
Effects mediated by BMI and serum AST/ALT ratio on the associations of the sulphur microbial diet score with two forms of AgeAccel. ***P < 0·001. AST, aspartate aminotransferase; ALT, alanine aminotransferase; DE, direct effect; IE, indirect effect.

Discussion
In this cross-sectional study involving 71 579 participants, we observed a significant positive association between a dietary pattern linked to a higher SMD score and an elevation of AgeAccel, as assessed by multiple markers using two widely accepted algorithms. Furthermore, a higher score of SMD was associated with an increase in AgeAccel among males and smokers. Both BMI and serum AST/ALT ratio mediated the association between SMD and two forms of AgeAccel. These findings support the hypothesis that dietary variations with lower detrimental microbiome configurations related to sulphur metabolism may be associated with lower biological ageing.
The SMD is characterised by a low intake of fruits, vegetables, legumes, whole grains and nuts, along with a high intake of red meat, processed meat products, high-fat dairy products, sugary beverages and coffee(Reference Wang, Nguyen and Mehta25). However, Wang et al. indicated that the relative abundance of sulphur-metabolising bacteria may be determined by dietary sources of sulphur and specific sulphur-containing compounds, rather than total sulphur content(Reference Wang, Nguyen and Mehta25). As for food components, red and processed meats are rich in both sulphur-containing amino acids and inorganic sulphur from preservatives(Reference Wang, Nguyen and Mehta25) and also can significantly worsen age-related diseases, such as cardiovascular disease, sarcopenia, cognitive dysfunction and cancer(Reference Kouvari, Tyrovolas and Panagiotakos29). In addition, a study utilising plasma protein profiles to identify accelerated and decelerated ageing discovered that the consumption of sugar-sweetened beverages increased the predicted CA by 2–6 years(Reference Enroth, Enroth and Johansson30). On the contrary, a high intake of whole grains, vegetables, fruits and nuts is associated with a reduced risk for all-cause mortality(Reference Ekmekcioglu31). Cruciferous vegetables are rich in sulphur-containing glucosinolates, which can be hydrolysed to isothiocyanates by gut microbiota that expresses myrosinase. Isothiocyanates and their downstream products have anticarcinogenic effects(Reference Song, Garrett and Chan32). What’s more, healthy dietary patterns such as Mediterranean, Japanese, Okinawan and Nordic diets have been associated with long-term survival and a reduced incidence of non-communicable diseases(Reference Dominguez, Veronese and Baiamonte33). These dietary patterns share common features that may explain the mechanisms of healthy ageing. For example, common features include increased intake of vegetables and whole grain foods, consumption of unsaturated fatty acids, moderate intake of primarily plant-based protein, avoidance or limitation of alcohol consumption, avoidance of red and processed meats, and limitation of sugar intake(Reference Dominguez, Veronese and Baiamonte33).
Our results were biologically possible via microbial H2S generation and extended previous findings demonstrating the role of diet-induced microbial changes in the ageing process. Current evidence indicates that persistent low-grade inflammation and oxidative stress accelerate cellular and tissue ageing, which has adverse effects on biological age(Reference Kanasi, Ayilavarapu and Jones34). Inflammation and oxidative stress are vital physiological processes influenced by various factors such as age, diet and lifestyle(Reference Wang, Sarker and Cheng19). H2S is produced within the gastrointestinal tract by resident gut bacteria metabolising sulfates, sulfites and various proteins(Reference Smith, Shorten and Altermann35). H2S exhibits genotoxicity, promoting inflammation and causing DNA damage in epithelial cells. Elevated concentrations of H2S disrupt intestinal permeability, facilitating the absorption of lipopolysaccharides into the bloodstream, further exacerbating inflammation(Reference Blachier, Andriamihaja and Larraufie36), which is associated with increased risks of obesity(Reference Liu, Wan and Zhang11), non-alcoholic fatty liver disease(Reference Liu, Huang and Ruan10) and gastrointestinal cancers(Reference Xiao, He and Xiang37), thereby increasing the risk of ageing.
In our study, we found that the association between SMD and AgeAccel is partially mediated by BMI and AST/ALT ratio, which are biomarkers of obesity status and liver function. Atypical elevation of AST/ALT is used to assess liver function and the severity of liver disease, suggesting the presence of underlying liver issues(Reference Kim, Joo and Sung38). Liver dysfunction can impact ageing, primarily manifested as declining liver function, malnutrition, hormonal imbalance and immune dysregulation(Reference Sheedfar, Di Biase and Koonen39). Obesity leads to many adverse health outcomes, including cardiovascular diseases, diabetes and cancer, which are considered age-related diseases(Reference Santos and Sinha40). Meanwhile, it is significantly positively associated with epigenetic AgeAccel(Reference Lundgren, Kuitunen and Pietiläinen41). Therefore, these two indicators are considered potential risk factors for the association. Moreover, the mediation results suggest that reducing body weight and supporting liver health by adherence to dietary patterns with lower SMD scores may help slow down the ageing process.
It is worth noting that a more significant association between SMD quartiles and AgeAccel in males and smoking populations was observed. There are several possible explanations for this observation. Firstly, adherence to the SMD diet is positively associated with the risk of obesity, with sex stratification revealing a more significant positive association in males than females(Reference Liu, Wan and Zhang11). Secondly, there is a higher proportion of males among smokers in our study. Smoking is associated with increased levels of inflammatory oxidative stress(Reference Wang, Sarker and Cheng19). Specifically, smoking damages intestinal barrier function promotes inflammatory responses in the process of intestinal disease occurrence and enhances carcinogenic MAPK/ERK signalling(Reference Bai, Wei and Liu42), as evidenced by a study finding a positive correlation between SMD and the risk of colorectal adenomas, particularly notable in males and smokers(Reference Xiao, He and Xiang37).
The present study contains some strengths. Firstly, our study includes a relatively large sample size and rich information on dietary patterns. Secondly, previous studies have rarely examined the effect of the sulphur-metabolism microbial diet on biological ageing. We were the first to assess the effect of BMI and AST/ALT ratio as a mediator on biological ageing. Nonetheless, limitations should be acknowledged. Firstly, a substantial number of participants were excluded due to missing dietary or BA information, which may introduce selection bias and limit generalisability. However, the large sample size and rigorous methodology help mitigate these concerns. Secondly, it is worth noting that the SMD was initially constructed based on a US cohort of older men. However, the UK Biobank does not provide information on the microbiomes of the participants, which prevents us from evaluating the effect estimates for specific food types. Thirdly, limited by the cross-sectional study design, we are unable to assume causality of the observed association. Fourthly, although the 24-hour diet was retrospectively assessed several times at baseline, this may have allowed participants to change their dietary patterns. Finally, as our study was conducted with a predominantly White sample of 37–73 years participants, caution is needed when generalising our findings to other populations. Future analyses comparing SMD with other dietary patterns in terms of shared components and health outcomes as well as exploring the potential interactions between systemic inflammation or metabolic pathways and sulphur-metabolising bacteria would be valuable.
In conclusion, this study is the first to discover the accelerated effects of a gut microbiota-derived dietary pattern, the SMD pattern, on biological ageing. The monotonic and linear association between them emphasised the change beyond the threshold in the sulphur bacterial diet score dramatically increasing the risk of AgeAccel. BMI and serum AST/ALT mediate the association between SMD and two AgeAccel. These findings support not only the role of diet in the ageing process but also the possibility of using a gut microbiota-targeted dietary modification to slow down the ageing process.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114525000534
Acknowledgements
This research has been conducted using the UK Biobank Resource under Application Number 46478.
This work was supported by the Natural Science Basic Research Program of Shaanxi Province [2021JCW-08].
Y. L. had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. F. Z. conceptualised and designed the study. All authors contributed to the data’s acquisition, analysis and interpretation. Y. L. drafted the manuscript. F. Z. helped with the critical revision of the manuscript for important intellectual content. Y. L. performed the statistical analysis. F. Z. provided administrative, technical or material support. F. Z. supervised the study. All authors have read and agreed to the published version of the manuscript.
All authors report no biomedical financial interests or potential conflicts of interest.
The North West Multi-Centre Research Ethics Committee (MREC) and Human Tissue Authority (HTA) have approved UK Biobank.
The datasets used and/or analysed during this study are available from the corresponding author upon reasonable request.
Informed consent was obtained from all subjects involved in the study.