Introduction
Wheat (Triticum aestivum L.) is a staple food for approximately 40% of the global population (Mao et al. Reference Mao, Jiang, Tang, Nie, Du, Liu, Cheng, Wu, Liu, Kang and Wang2023). In China, it covers an area of 23.6 million ha, accounting for 19.8% of total sown area of grain crops, and yielded 140.1 million tons in 2024 (NBSC 2024). The yield and quality of wheat are directly related to food security. Nevertheless, weed infestation is a major cause of yield losses in wheat (Minhas et al. Reference Minhas, Mumtaz, Ur-Rehman, Farooq, Farooq, Ali and Hussain2023). Weed competition can reduce the production of wheat by approximately 25% (Oerke Reference Oerke2006).
Italian ryegrass [Lolium perenne L. ssp. multiflorum (Lam.) Husnot] is one of the most troublesome weeds in wheat production systems worldwide (Norsworthy et al. Reference Norsworthy, Noe, Avent, Butts and Roberts2025; Thorne and Lyon Reference Thorne and Lyon2025). It is native to central and southern Europe, northwest Africa, and southwest Asia (Altop et al. Reference Altop, Meral, Zandstra and Mennan2022). Lolium perenne ssp. multiflorum was introduced into China as a pasture plant in 1980, but it has now spread into wheat fields as a persistent weed (Zhang et al. Reference Zhang, Zhang, Chen and Dong2021). This species exhibits high genetic diversity, resulting in considerable phenotypic plasticity in both plant and seed traits that facilitates broad environmental adaptability (Maity et al. Reference Maity, Singh, Jessup and Bagavathiannan2021a, Reference Maity, Singh, Martins, Ferreira, Smith and Bagavathiannan2021b). Lolium perenne ssp. multiflorum possesses extremely strong tillering and reproductive capacities, with a single plant producing up to 86 effective tillers and 15,000 seeds (Wu et al. Reference Wu, Xu, Sun, Su, Wei, Xue and Lu2022). The strong competitive ability of L. perenne ssp. multiflorum leads to its rapid proliferation in the wheat field and significant yield reductions in wheat.
Over the past two decades, weed management relied on chemical herbicides (Zhu et al. Reference Zhu, Wang, Gao, Liu, Li, Feng and Dong2023). However, the widespread use of herbicides has led to the rapid spread of herbicide resistance. To date, 77 herbicide-resistance records for L. perenne ssp. multiflorum have been documented in 15 countries (Argentina, Brazil, Canada, Chile, China, Denmark, France, Italy, Japan, New Zealand, Portugal, Spain, Switzerland, the United Kingdom and the United States), covering 8 different mechanisms of action (Heap Reference Heap2025). With the continued evolution of herbicide resistance in global populations, controlling this weed has become increasingly challenging.
Seed germination, a critical stage in the life cycle of plants, is significantly affected by various environmental factors (Fukuda et al. Reference Fukuda, Imaizumi and Koarai2023; Rezvani et al. Reference Rezvani, Nadimi, Zaefarian and Chauhan2021; Saffariha et al. Reference Saffariha, Jahani and Potter2020). Understanding the germination ecology of L. perenne ssp. multiflorum is therefore important to optimize and maximize the efficiency of management strategies (Wang et al. Reference Wang, Zhao, Li, Chen, Liu and Wang2020). Previous studies on related species provide initial insights into the germination responses of Lolium to environmental factors. For instance, studies on Wimmera ryegrass (Lolium rigidum Gaudin) indicate that elevated soil temperature and humidity promote seed dormancy release, and light exposure also modulates germination (Steadman et al. Reference Steadman, Bignell and Ellery2003a, Reference Steadman, Crawford and Gallagher2003b). Darnel ryegrass (Lolium temulentum L.) exhibits a temperature-dependent germination pattern, with optimal germination (91.6%) occurring at 10 and 20 C, no germination at 35 to 40 C, and a strict requirement for light (32.5% germination under 12-h photoperiod, 0% in continuous darkness) (Faraj et al. Reference Faraj, Colak and Isik2023). Its germination was not significantly affected by a broad pH range (4 to 10) or salinity levels up to 250 mM (Faraj et al. Reference Faraj, Colak and Isik2023). For L. perenne ssp. multiflorum, research shows that germination is influenced by temperature–salinity interactions, with germination reaching 79.3% at alternating temperatures of 20/30 C under 400 mM salinity but decreasing significantly to 46% at 10/20 C under the same salinity level (Lin et al. Reference Lin, Hua, Peng, Dong and Yan2018). Furthermore, under 90% shade, L. perenne ssp. multiflorum exhibits a germination rate of 98.7%, significantly higher than the 63.4% observed in perennial ryegrass (Lolium perenne L.) (Rossini et al. Reference Rossini, Ruggeri, Celli, Rogai, Kuzmanović and Richardson2019). However, a systematic assessment of the individual effects of key environmental factors (including osmotic potential, pH, burial depth, temperature, light, and salinity) on the seed germination and seedling emergence of L. perenne ssp. multiflorum under a consistent experimental framework is lacking. Elucidating its germination ecology is therefore essential not only for predicting potential distribution and invasion risk but also for designing tillage practices and other nonchemical strategies to suppress its soil seedbank and reduce its impact on wheat.
Crop yield reduction is attributed to weed–crop competition for critical resources, including nutrients, water, and light, with weed density constituting a key factor influencing such competitive dynamics (Zimdahl Reference Zimdahl2008). Wu et al. (Reference Wu, Xu, Sun, Su, Wei, Xue and Lu2022) reported that L. perenne ssp. multiflorum generally causing a wheat yield reduction of approximately 30%, reaching 95% in severe cases. Even at low densities, it can reduce cereal and soybean [Glycine max (L.) Merr.] yields by 50% to 92% (Altop et al. Reference Altop, Meral, Zandstra and Mennan2022). S Yang et al. (Reference Yang, Jia, Yang, Li, Duan, Hu and Cui2023) reported that 50 plants m−2 of eared redstem (Ammannia auriculata Willd.) reduced rice (Oryza sativa L.) yield by 70%. Tian et al. (Reference Tian, Shen, Yuan, Song, Lu and Da2020) observed that even 4 plants m−2 of barnyardgrass [Echinochloa crus-galli (L.) P. Beauv.] and 8 plants m−2 of variable flatsedge (Cyperus difformis L.) led to rice yield losses of 23% and 11%, respectively. Increasing crop density can enhance competitive suppression of weeds (Ali et al. Reference Ali, Shabbir, Mahmood and Weiner2022). At low seeding rates, weeds have a favorable environment, providing them with the opportunity to germinate easily and grow quickly. However, at high crop seeding rates, crops have a competitive advantage over weeds due to earlier interception of resources and canopy closure (Ali et al. Reference Ali, Shabbir, Mahmood and Weiner2022; Anwar et al. Reference Anwar, Juraimi, Man, Puteh, Selamat, Begum, Anwar, Juraimi, Man, Puteh, Selamat and Begum2010). For example, increasing wheat planting density to 200 plants m−2 reduced L. rigidum biomass by about 50% and decreased weed-induced yield loss from 23% to 17%, without affecting grain size (Lemerle et al. Reference Lemerle, Cousens, Gill, Peltzer, Moerkerk, Murphy, Collins and Cullis2004). Ali et al. (Reference Ali, Shabbir, Mahmood and Weiner2022) found that increasing wheat density from 130 to 390 plants m−2 reduced weed biomass and seed production by 68% and 85%, respectively. Therefore, understanding the competitive interaction between L. perenne ssp. multiflorum, and wheat is critical for quantifying yield impacts and guiding integrated weed management (IWM). However, most competition studies have investigated the effect of varying either weed density or crop density in isolation. The interactive effect on yield components of simultaneously manipulating both L. perenne ssp. multiflorum density and wheat sowing density remains unquantified. This interaction is central to forecasting economic injury levels and refining threshold-based management strategies for this weed.
Therefore, the present study aimed to: (1) assess the effects of key environmental factors on seed germination and the seedling emergence of L. perenne ssp. multiflorum, and (2) quantify the relationship between wheat yield components and the interactive densities of L. perenne ssp. multiflorum and wheat. The findings contribute to understanding the adaptation strategies of L. perenne ssp. multiflorum to different management and tillage practices and to establishing critical competition thresholds for IWM.
Materials and Methods
Seed Materials
Seeds of L. perenne ssp. multiflorum used in this experiment were collected manually from a wheat field in Jinan City, Shandong Province, China (36.666°N, and 117.119°E), in 2022. The field was under a long-term maize (Zea mays L.)–wheat rotation system, with a tillage practice of maize straw incorporation followed by shallow rotary tillage. The collected seeds were air-dried at summer room temperature (25 to 35 C) and subsequently preserved in paper packets in refrigerator at 4 C until experimental use. After this cold storage treatment, seed dormancy was effectively broken.
General Germination Protocol in Laboratory Experiments
All laboratory germination experiments were conducted using 9-cm petri dishes lined with two layers of Whatman No. 1 filter paper. Fifty seeds were placed in each dish, and the filter paper was initially moistened with 5 ml of distilled water or the corresponding test solution. Additional solution was added as needed to maintain adequate moisture. Then, the petri dishes were placed in growth chambers at a diurnal temperature regime of 15/20 C (12-h dark/12-h light). Germination progress was recorded daily. Recording was terminated when no new seed germination was observed for 3 consecutive days. Germination was defined as the emergence of a visible radicle (≥2 mm), and germination rates were calculated accordingly. Each treatment was replicated four times, and each experiment was repeated twice.
Effects of Temperature on Seed Germination
To assess the effect of temperature on L. perenne ssp. multiflorum seed germination, seeds were incubated in growth chambers under seven constant temperature regimes: 5, 10, 15, 20, 25, 30, and 35 C. Temperature was regulated by the growth chamber’s control system. To ensure accuracy, the actual temperature inside the chamber was monitored daily using a calibrated mercury thermometer and maintained within ±0.5 C of each target throughout the experiment. The temperatures were selected to cover the typical seasonal temperatures encountered in the region. All other experimental conditions were consistent with those outlined in the “General Germination Protocol” section.
Effects of Light on Seed Germination
To evaluate the effect of light on the germination of L. perenne ssp. multiflorum seeds, petri dishes were incubated in growth chambers under diverse light regimes: 24-h darkness, 24-h light, and alternating light/dark cycles (16/8, 14/10, 12/12, 10/14, and 8/16 h). All other experimental conditions were consistent with those outlined in the “General Germination Protocol” section.
Effects of pH on Seed Germination
To evaluate the germination response of L. perenne ssp. multiflorum to pH, seeds were incubated in buffer solutions with pH values of 3, 4, 5, 6, 7, 8, 9, and 10. This range covers the typical pH variation of surface soils in China (5 to 9) (F Yang et al. Reference Yang, Jia, Yang, Li, Duan, Hu and Cui2023). Buffers for pH 3 to 6 were prepared with 2 mM MES [2-(N-morpholino) ethanesulphonic acid] and adjusted to the target pH using 0.1 M HCl (hydrogen chloride) or NaOH (sodium hydroxide). For pH 7 and 8, 2 mM HEPES [N-(2-hydroxymethyl) piperazine-N-(2-ethanesulphonic acid)] was used and adjusted with 0.1 M NaOH. Buffers for pH 9 and 10 were prepared with 2 mM Tricine [N-Tris (hydroxymethyl) methylglycine] and adjusted with 0.1 M NaOH (Chachalis and Reddy Reference Chachalis and Reddy2000). Subsequently, 5 ml of the respective buffer solution was added to each petri dish. All other experimental conditions were consistent with those outlined in the “General Germination Protocol” section.
Effects of Salinity on Seed Germination
To assess the effect of salt stress, seeds were incubated in solutions with different NaCl concentrations: 0, 50, 100, 150, 200, 250, and 300 mM. The selected levels of salinity were determined based on regional soil conditions. All other experimental conditions were consistent with those outlined in the “General Germination Protocol” section.
Effects of Osmotic Potential on Seed Germination
To determine how osmotic stress influenced seed germination for each study population, solutions with osmotic potentials of 0 (distilled water), −0.1, −0.3, −0.5, −0.7, −0.9, −1.1, and −1.3 MPa were prepared by dissolving polyethylene glycol (PEG 6000) in distilled water (Michel and Kaufmann Reference Michel and Kaufmann1973). These solutions were then added to the petri dishes. The selected levels of osmotic potential were determined based on regional soil conditions. All other experimental conditions were consistent with those outlined in the “General Germination Protocol” section.
Effects of Burial Depth on Seedling Emergence
The experiment was conducted twice in a greenhouse at Shandong Academy of Agricultural Sciences, Jinan City, Shandong Province (36.708°N, 117.077°E). The temperature was maintained at 10 to 25 C under natural light. Seeds of L. perenne ssp. multiflorum with a high germination rate (>85%), plump maturity, and relatively uniform size were selected and sown in plastic pots (27.5-cm diameter, 24-cm height). The soil was sieved loam collected from the upper 20-cm layer of farmland in Licheng District, Jinan City, Shandong Province (36.716°N, 117.133°E). The site had no history of herbicide application. Soil was autoclaved before use. Pots were filled with soil in layers according to the preset depth. Fifty seeds were sown per pot. The seed burial depths were set at 0, 0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 5, 6, 8, 10, 12, 14, 16, 18, and 20 cm, with four replicates for each depth. The selected seed burial depths were designed to reflect a range of realistic soil conditions influenced by different tillage practices (no-tillage, shallow rotary tillage, deep plowing). After sowing, water was supplied via sub-irrigation until the soil surface was moist. Seedling emergence was defined as the visible appearance of the coleoptile at the soil surface, with daily recordings maintained until 35 d after sowing.
Effects of Different Densities of Lolium perenne ssp. multiflorum on Wheat Yield
The field experiment was conducted from 2022 to 2024 in a 2.5-ha wheat field in Jinan City, Shandong Province (36.666°N, 117.116°E). The experimental field, which suffered from severe infestation of L. perenne ssp. multiflorum, was managed under a maize–wheat rotation. The tillage practice before wheat sowing involved incorporating maize straw into the soil followed by shallow rotary tillage. Wheat (‘Jimai 22’) was manually sown on October 3 in both 2022 and 2023, and harvested on June 2 in 2023 and 2024, respectively. No herbicides were applied in this experiment, and other weeds were removed manually.
Based on typical wheat planting densities in Shandong Province and the agronomic characteristics of Jimai 22 wheat, three wheat sowing densities were established by three seeding rates: 67.5, 135.0, and 202.5 kg ha−1. For each wheat sowing density, 13 L. perenne ssp. multiflorum density treatments (0, 10, 20, 40, 60, 80, 120, 160, 240, 280, 320, 380, and 420 plants m−2) were arranged in combination with artificial sowing. To maintain the desired L. perenne ssp. multiflorum densities, seedling thinning was conducted three times throughout the growing season: a preliminary thinning was conducted before winter to retain a seedling density slightly higher than the target, followed by two additional thinning events in early spring and the wheat regreening stage to precisely adjust the actual seedling density to the preset level. The experiment followed a randomized complete block design with four replications, and each plot measured 1 m2 (1 m by 1 m).
At harvest, wheat yield components such as wheat spike density, 1,000-grain weight, grains per spike, and plot yield were recorded on the whole 1-m2 plot.
Statistical Analysis
All statistical analyses were conducted using DPS software (v. 9.01, Zhejiang University, Hangzhou, China), with Microsoft Excel 2019 (Microsoft, Redmond, WA, USA) used for data organization. The germination experiment was arranged in a completely randomized design, and the seed burial experiment employed a randomized complete block design. Each treatment was replicated four times, and each experiment was repeated twice. No significant differences were found between the two runs of any treatment, so data from both runs were pooled for analysis. One-way ANOVA was performed, and the significant difference of mean values was evaluated using Tukey’s honest significant difference test at P < 0.05. For field experiments, two consecutive growing seasons of the experiment were conducted from October 2022 to June 2024. Data from both seasons exhibited consistent trends, and statistical analysis indicated no significant difference between the 2 yr. Therefore, the data from the two growing seasons were pooled for final analysis. The field competition trial employed a two-factor (wheat sowing density and L. perenne ssp. multiflorum density) randomized complete block design with four replications. Simple linear regression models were used to quantify the relationships between L. perenne ssp. multiflorum density (independent variable) and wheat yield components (dependent variables) under each sowing density. All figures were prepared using Microsoft Excel 2019 and GraphPad Prism (v. 8.0, GraphPad Software, Boston, MA, USA).
Results and Discussion
Effects of Temperature on Seed Germination
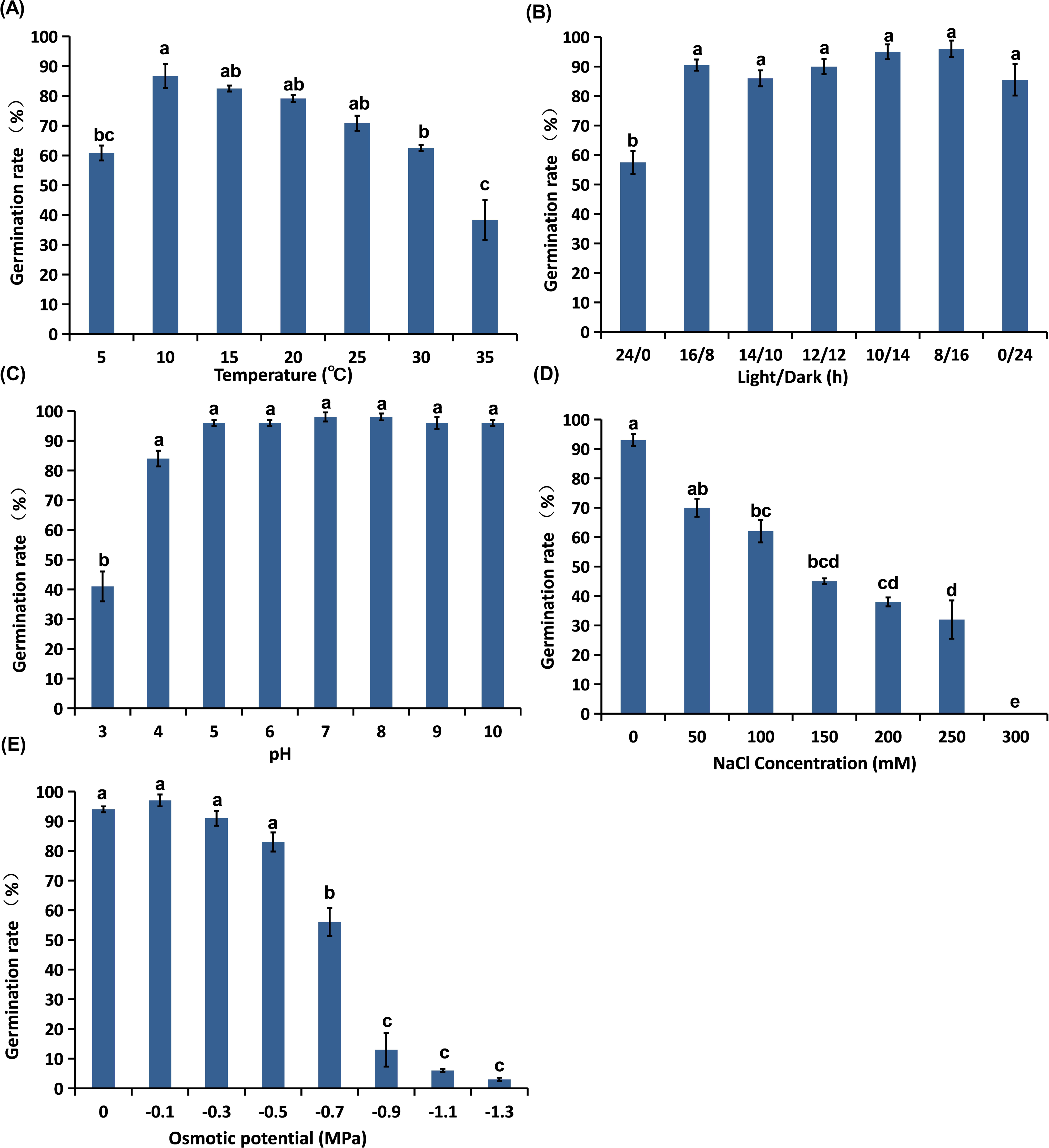
Lolium perenne ssp. multiflorum seeds can germinate across a broad temperature range. Germination was optimal at constant temperatures between 10 and 25 C, with rates ranging from 70% to 86%, and the highest germination rate was achieved at 10 C (86%). Even at a low temperature of 5 C, L. perenne ssp. multiflorum seed germinated at rate of 61% (Figure 1A). This contrasts sharply with winter wheat (‘Bainong Ak58’), for which germination plummets to only 8.8% at the same low temperature (5 C) (Su et al. Reference Su, Yang, Ma, Li, Xu, Xue, Sun, Lu and Wu2023). The high germination of L. perenne ssp. multiflorum at low temperatures aligns with its identity as a cool-season species (Rossini et al. Reference Rossini, Ruggeri, Celli, Rogai, Kuzmanović and Richardson2019) and represents a key adaptive trait. Although germination of L. perenne ssp. multiflorum was relatively low at 35 C (38%) and similar to that reported for wheat (∼25%) at this temperature (Su et al. Reference Su, Yang, Ma, Li, Xu, Xue, Sun, Lu and Wu2023), the critical advantage lies in its lower temperature threshold for germination. This capability enables it to initiate germination earlier in the season under cool field conditions, potentially securing a significant competitive advantage over wheat seedlings.
Effect of environmental factors on the germination (%) of Lolium perenne ssp. multiflorum: (A) temperature; (B) light; (C) pH; (D) salt stress; and (E) osmotic stress. Data are mean ± SE (n = 4). Different lowercase letters indicate significant difference (P < 0.05).

Previous studies have reported variable germination responses of L. perenne ssp. multiflorum to temperature across different populations. Butler et al. (Reference Butler, Celen, Webb, Krstic and Interrante2017) evaluated germination of L. perenne ssp. multiflorum seeds at seven constant temperature levels (5, 10, 15, 20, 25, 30, and 35 C) and found maximum germination (>95%) occurred at 10 to 30 C, with 61% germination at 5 C and a germination rate of 72% at 35 C, higher than in our study. Lin et al. (Reference Lin, Hua, Peng, Dong and Yan2018) tested germination of L. perenne ssp. multiflorum under non-stress conditions at four alternating temperature regimes (10/20, 15/25, 20/30, and 25/35 C), and observed optimal germination (>90%) across all tested regimes. Consistent with our findings, all aforementioned studies confirm a broad temperature tolerance for L. perenne ssp. multiflorum seed germination. Similar temperature adaptability has been observed in other Lolium weeds. Lolium temulentum exhibits optimal germination (91.6%) at 10 and 20 C and no germination at 35 to 40 C (Faraj et al. Reference Faraj, Colak and Isik2023). For L. perenne, 25 C is the optimum temperature for maximum germination (95%); however, a rapid decline was observed at 35 C (Javaid et al. Reference Javaid, Mahmood, Alshaya, AlKahtani, Waheed, Wasaya, Khan, Naqve, Haider, Shahid, Nadeem, Azmat, Khan, Balal, Attia and Fiaz2022). Lolium rigidum exhibited >90% germination across several alternating temperature regimes (20/10, 25/15, 30/20, and 35/25 C) (Thompson et al. Reference Thompson, Mahajan and Chauhan2021).
These results may partially explain why L. perenne ssp. multiflorum has such a wide geographic distribution, as this trait is an important determinant of species’ persistence under variable environmental conditions (Farooq et al. Reference Farooq, Onen, Tad, Ozaslan, Mahmoud, Brestic, Zivcak, Skalicky and El-Shehawi2021). This broad temperature tolerance strengthens its potential to establish and spread, presenting significant challenges for wheat production management.
Effects of Light on Seed Germination
Germination of L. perenne ssp. multiflorum did not differ significantly among alternating light/dark treatments or in complete darkness. In contrast, continuous light significantly inhibited germination compared with all other regimes, although the rate remained above 50%. Maximum germination (96%) was observed under an 8-h light/16-h dark photoperiod (Figure 1B).
Light is a key regulator of seed germination, but requirements vary widely among species. Some species prefer to germinate in light, others in complete darkness, and other species exhibit no preference (Baskin and Baskin Reference Baskin and Baskin1998). Our results indicate that light is not a limiting factor for L. perenne ssp. multiflorum, consistent with earlier reports. Rossini et al. (Reference Rossini, Ruggeri, Celli, Rogai, Kuzmanović and Richardson2019) evaluated germination of L. perenne ssp. multiflorum seeds under six different shade treatments (0%, 30%, 50%, 70%, 90%, and 100% shade) and found that under 0%, 30%, 50%, 70%, 90%, and 100% shade, L. perenne ssp. multiflorum exhibits germination rates of 87.3%, 85.7%, 90.6%, 87.5%, 98.7%, and 91.4%, respectively. Such light-insensitive germination is characteristic of many cool-season grasses (Rossini et al. Reference Rossini, Ruggeri, Celli, Rogai, Kuzmanović and Richardson2019). For example, Thompson et al. (Reference Thompson, Mahajan and Chauhan2021) found that the germination percentage of L. rigidum exceeded 90% for all populations when grown under light/dark conditions. The absence of a light requirement enables L. perenne ssp. multiflorum to germinate even when deeply buried, enhancing its survival and persistence under varying agricultural practices.
Effects of pH on Seed Germination
Lolium perenne ssp. multiflorum seeds exhibited broad pH tolerance for germination. Germination remained consistently high (84% to 98%) across a wide pH range from 4 to 10, with no significant differences observed within this interval (Figure 1C). Germination was only affected at pH 3, where the germination rate reached 41% (Figure 1C).
The typical pH range of most farmland soils in China is 5 to 9 (F Yang et al. Reference Yang, Jia, Yang, Li, Duan, Hu and Cui2023), which falls within the optimal scope for L. perenne ssp. multiflorum seed germination, indicating a high degree of adaptation of this weed to the soil acid–base conditions of local farmlands. The inhibition observed at pH 3 may be attributed to strong acid stress, ion imbalance, and disruption of normal physiological functions within the seed, resulting in hindered seed germination (Wang et al. Reference Wang, Yang, Wang, Li, Gao, Huang, Zhou, Wang and Zhao2025). Our findings are consistent with previous reports that document pH-insensitive germination in Lolium species. Cavallaro et al. (Reference Cavallaro, Maucieri and Barbera2014) demonstrated that pH (4.5 vs. 7.5) had no significant effect on L. perenne ssp. multiflorum germination. Similarly, L. perenne germination was unaffected by pH levels ranging from 4 to 10 (Javaid et al. Reference Javaid, Mahmood, Alshaya, AlKahtani, Waheed, Wasaya, Khan, Naqve, Haider, Shahid, Nadeem, Azmat, Khan, Balal, Attia and Fiaz2022; Wang et al. Reference Wang, Yang, Wang, Li, Gao, Huang, Zhou, Wang and Zhao2025). Faraj et al. (Reference Faraj, Colak and Isik2023) found that no significant effect of examined pH (4 to 10) on seed germination of L. temulentum. Comparable broad pH adaptability has been reported in other weeds. For example, Chauhan et al. (Reference Chauhan, Gill and Preston2006) reported that common sowthistle (Sonchus oleraceus L.) seeds achieved germination percentages exceeding 90% at pH 5 to 8. Nikolić et al. (Reference Nikolić, Šoštarčić, Pismarović, Šćepanović and Masin2022) found that jimsonweed (Datura stramonium L.) seeds maintained a germination rate of 70% to 80% across a pH range of 3 to 9, while germination was completely inhibited at pH 1.
Such wide pH tolerance suggests that soil acidity or alkalinity is unlikely to strongly restrict the establishment of L. perenne ssp. multiflorum in most agricultural ecosystems. This adaptive trait likely contributes to its capacity to invade and persist across diverse global environments where soil pH varies widely.
Effects of Salt Stress on Seed Germination
Seed germination of L. perenne ssp. multiflorum decreased with increasing NaCl concentration from 0 to 300 mM. The highest germination percentage (93%) was recorded under non–salt stress conditions (0 mM NaCl), and germination remained above 60% at NaCl concentrations up to 100 mM. Only 32% germination was observed at 250 mM NaCl, while germination was completely inhibited at 300 mM NaCl (Figure 1D).
The observed decrease in germination percentage under salt stress might result from osmotic effects (creating a low osmotic potential), ion toxicity (Na+, Cl−, etc.), or a combination of the two effects (Zhang et al. Reference Zhang, Irving, Tian and Zhou2012). Soils with NaCl concentrations above 100 mM are generally classified as saline (Chauhan Reference Chauhan2016). Although high soil salinity suppresses L. perenne ssp. multiflorum seed germination, our results indicate that L. perenne ssp. multiflorum can still establish in saline soils, a trait that may facilitate its colonization of saline areas. However, germination responses to salinity can vary among populations. Lin et al. (Reference Lin, Hua, Peng, Dong and Yan2018) reported that L. perenne ssp. multiflorum seeds could maintain a high germination rate (79.3%) under 400 mM NaCl at alternating temperatures of 20/30 C, while the population in our study exhibited complete germination inhibition at 300 mM. Such interpopulation differences in germination biology highlight the necessity of characterizing local L. perenne ssp. multiflorum populations for development of targeted management strategies. Similar salt tolerance during germination has been documented in other Lolium species. For instance, L. perenne maintained high germination (78.7%) even at 200 mM NaCl (Borawska-Jarmułowicz et al. Reference Borawska-Jarmułowicz, Mastalerczuk, Gozdowski, Małuszyńska and Szydłowska2017). Lolium rigidum exhibited 50% germination at 131 mM NaCl and complete inhibition at 250 mM NaCl (Thompson et al. Reference Thompson, Mahajan and Chauhan2021). These comparative findings highlight the ecological adaptability of the genus to saline stress.
Effects of Osmotic Stress on Seed Germination
Seed germination of L. perenne ssp. multiflorum declined as osmotic potential decreased from −0.2 to −1.3 MPa. The germination rate remained above 50% even at an osmotic potential of −0.7 MPa but was significantly inhibited under more severe drought stress (−0.9 to −1.3 MPa) (Figure 1E). This suppression of germination under severe osmotic stress is likely due to the impairment of water uptake, preventing the embryo from developing the radicle and plumule (Javaid et al. Reference Javaid, Mahmood, Alshaya, AlKahtani, Waheed, Wasaya, Khan, Naqve, Haider, Shahid, Nadeem, Azmat, Khan, Balal, Attia and Fiaz2022). Severe water deficit can disrupt enzymatic activity, cell division, and other physiological metabolic processes within the seeds (Wang et al. Reference Wang, Yang, Wang, Li, Gao, Huang, Zhou, Wang and Zhao2025). In addition, it may also be due to changes in hormones in the seeds (Kawaguchi et al. Reference Kawaguchi, Suriyasak, Matsumoto, Sawada, Sakai, Hamaoka, Sasaki, Yamane, Kato, Bailly and Ishibashi2023).
Drought stress is a major factor limiting the development and distribution of L. perenne ssp. multiflorum (Pan et al. Reference Pan, Meng, Wang, Ma, Fan, Yang, Zhou and Zhang2018). Our results indicate that while the species can germinate under moderately dry conditions, germination is strongly limited at very low water potentials. In China, annual precipitation ranges from 500 to 800 mm in the north, while it exceeds 1,000 mm in the humid regions of the Yangtze Delta (Zhao et al. Reference Zhao, Li, Guo, Zhang, Ge and Wang2018). Therefore, the soil osmotic potential is suitable for L. perenne ssp. multiflorum germination in most crop areas in China, although extremely arid conditions are an inhibitory factor. Similar osmotic sensitivity has been reported in related Lolium species. For L. rigidum, the osmotic potential causing a 50% reduction in germination varied among three Australian populations, ranging from −0.39 to −0.46 MPa, with complete inhibition at −0.8 MPa (Thompson et al. Reference Thompson, Mahajan and Chauhan2021). Lolium perenne maintained high germination (87.3%) at −0.6 MPa but failed to germinate at −1.2 MPa (Borawska-Jarmułowicz et al. Reference Borawska-Jarmułowicz, Mastalerczuk, Gozdowski, Małuszyńska and Szydłowska2017). Similarly, germination of L. perenne was unaffected by osmotic potential ranges from 0 to −0.4 MPa and was completely inhibited at both −0.8 and −1.0 MPa (Javaid et al. Reference Javaid, Mahmood, Alshaya, AlKahtani, Waheed, Wasaya, Khan, Naqve, Haider, Shahid, Nadeem, Azmat, Khan, Balal, Attia and Fiaz2022).
Effects of Burial Depth on Seedling Emergence
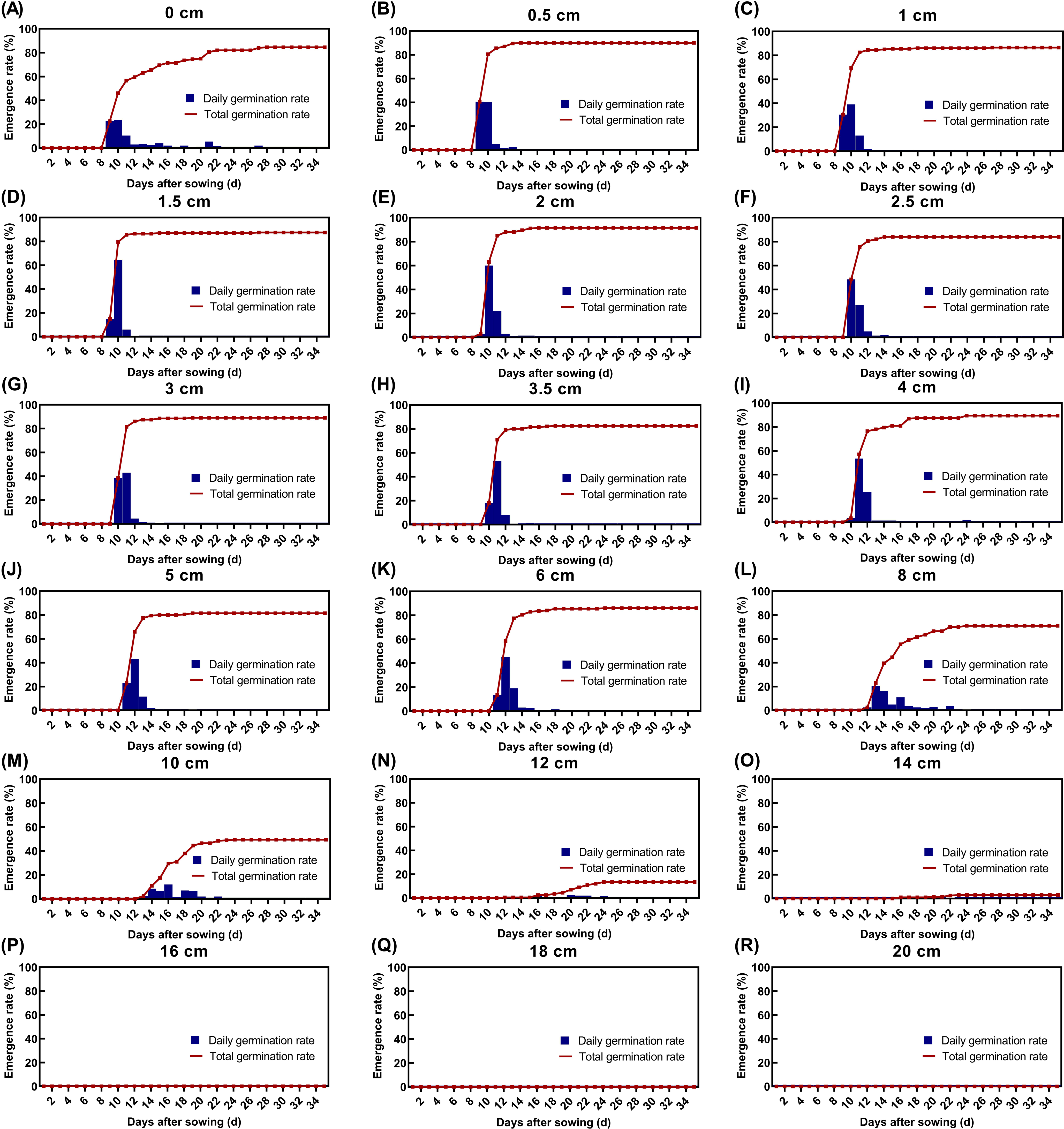
Seedling emergence of L. perenne ssp. multiflorum was significantly affected by burial depth. Maximum seedling emergence (92%) was observed for seeds buried at 2 cm. Surface-sown seeds (0 cm) exhibited a slight delay in emergence onset but still attained a high final emergence rate of 85%. For seeds buried at 0.5 to 6 cm, seedling emergence increased rapidly between 9 and 13 d after sowing, with final rates exceeding 80%. In contrast, seeds buried at 8 cm and 10 cm exhibited delayed emergence, beginning only after 13 d and reaching final rates of 71% and 50%, respectively. Emergence declined markedly to 14% at 12-cm depth, became negligible at 14 cm, and was completely inhibited at greater depths (Figure 2).
Effect of different seed burial depths on seedling emergence (%) of Lolium perenne ssp. multiflorum.

Compared with weeds such as cutleaf geranium (Geranium dissectum L.) (Atabaki et al. Reference Atabaki, Gherekhloo, Ghaderi-Far, Ansari, Hassanpour-bourkheili and Prado2023) and herb sophia [Descurainia sophia (L.) Webb ex Prantl] (Yang et al. Reference Yang, Liu, Gao, Li, Kong, Liu, Liu, Zeng, Fang and Li2025) that can only germinate at soil depths of 0 to 2 cm and 0 to 0.5 cm, respectively, L. perenne ssp. multiflorum exhibits a strong ability to germinate and emerge from deep soil. This ability can be attributed to two key factors. First, light was not an absolute requirement for the germination of the L. perenne ssp. multiflorum seeds. Second, the seeds are relatively large sized and possess enough endosperm reserves, providing the physiological basis for penetrating through thicker soil layers. As reported by Boccaccini et al. (Reference Boccaccini, Cimini, Kazmi, Lepri, Longo, Lorrai and Vittorioso2024), seed size and quality are important factors affecting germination, emergence from soil, and seedling growth. Under dark, buried conditions, seedling emergence depends largely on these internal reserves (Zhao et al. Reference Zhao, Li, Guo, Zhang, Ge and Wang2018). In addition, larger seeds have stronger stress resistance, thereby enabling seedlings to survive and grow better (Boccaccini et al. Reference Boccaccini, Cimini, Kazmi, Lepri, Longo, Lorrai and Vittorioso2024).
Our results indicate that L. perenne ssp. multiflorum seedling emergence is likely to be higher under reduced- or no-tillage systems, where seeds remain near the soil surface or at shallow depths conducive to emergence. In contrast, emergence was effectively suppressed when seeds were buried deeper than 14 cm, likely due to oxygen deficiency and limitation of gas diffusion in deep soil layers (Wang et al. Reference Wang, Yu and Gou2019). These findings suggest that in conventional tillage systems, deep ploughing (burying seeds below 14 cm) could serve as an effective cultural control strategy.
Effects of Different Densities of Lolium perenne ssp. multiflorum on Wheat Yield
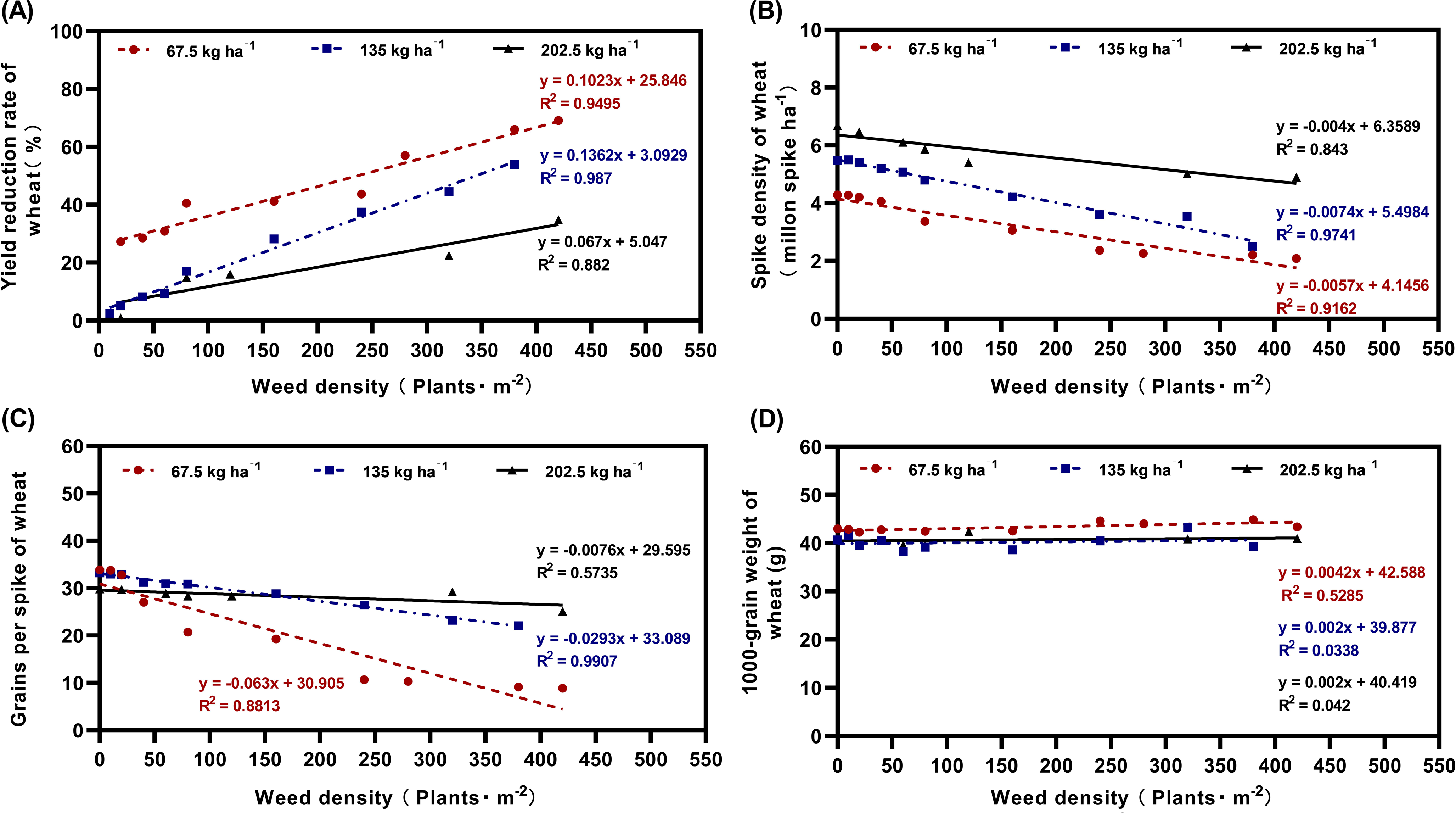
Field experiments quantified the competitive effects of L. perenne ssp. multiflorum density on wheat yield components, revealing clear competition thresholds and the mitigating role of increased wheat sowing density. Wheat yield loss increased linearly with rising weed density under all sowing densities (Figure 3A). At the lowest wheat sowing density (67.5 kg ha−1), it had a strong linear positive correlation (y = 0.1023x + 25.846, R2 = 0.9495) between the yield reduction rate and weed density, reaching 27% yield loss at the lowest L. perenne ssp. multiflorum density (20 plants m−2) and 69% at the highest weed density (420 plants m− 2). At the intermediate wheat sowing rate (135 kg ha−1), yield loss was only 5% at low weed density (20 plants m−2) but increased more steeply (y = 0.1362x + 3.0929, R2 = 0.987), attaining 54% yield loss at the highest weed density (380 plants m−2). The steeper slope at this density indicates that as weed pressure increases, the required level of weed control to maintain high yield becomes more stringent. The highest wheat sowing density (202.5 kg ha−1) further alleviated yield loss across the weed density gradient (y = 0.067x + 5.047, R2 = 0.882), with yield losses of 1% and 35% at low (20 plants m−2) and high (420 plants m−2) weed densities, respectively (Figure 3A).
Effects of different densities of Lolium perenne ssp. multiflorum on wheat yield: (A) yield reduction rate of wheat (%); (B) spike density of wheat (million spike ha−1); (C) grains per spike of wheat; and (D) 1,000-grain weight of wheat (g).

Spike density declined linearly with increasing L. perenne ssp. multiflorum density (Figure 3B). At 67.5 kg ha−1 wheat sowing density, there was a significant linear negative correlation (y = −0.0057x + 4.1456, R2 = 0.9162), with spike density reduced by 2% and 51% at low (20 plants m−2) and high (420 plants m−2) weed densities, respectively. A similar trend was observed at 135 kg ha−1 wheat sowing density (y = −0.0074x + 5.4984, R2 = 0.9741), with spike density reduction of 2% and 54% at low and high weed densities, respectively. For 202.5 kg ha−1 wheat sowing density, the decline was less pronounced (y = −0.004x + 6.3589, R2 = 0.843), leading to only 3% and 27% reductions at the lowest and the highest weed densities, respectively (Figure 3B).
Grains per spike also decreased with increasing weed competition (Figure 3C). The strongest reduction occurred at the lowest sowing density (y = −0.063x + 30.905, R2 = 0.8813), with 3% and 74% losses at the lowest (20 plants m−2) and the highest (420 plants m−2) weed densities. At 135 kg ha−1 wheat sowing density, the decline was milder (y = −0.0293x + 33.089, R2 = 0.9907). The highest sowing density further attenuated the effect (y = −0.0076x + 29.595, R2 = 0.5735), limiting the loss to 16% even at the highest weed density (Figure 3C). The relatively low R2 at this density further suggests that under a high wheat sowing density, the grains per spike value was less determined by weed density.
In contrast, 1,000-grain weight remained stable across all weed and sowing density treatments (Figure 3D). Thus, 1,000-grain weight remained a relatively stable yield component, largely unaffected by the varying competitive pressures imposed by either L. perenne ssp. multiflorum density or wheat sowing density in this study.
Our findings confirm that increasing wheat sowing density can substantially mitigate weed-induced losses in yield, spike density, and grains per spike, although it did not affect 1,000-grain weight. This supports the view that denser wheat stands enhance resource preemption and suppress weed growth (Manalil et al. Reference Manalil, Coast, Werth and Chauhan2017). For instance, raising wheat planting density from 82 to 164 plants m−2 led to 78% and 77% decreases in the biomass of herbicide-resistant and herbicide-susceptible S. oleraceus biotypes, respectively, when compared with the non-competition control treatment (Mobli et al. Reference Mobli A, Yadav and Chauhan2020). D’Amico et al. (Reference D’Amico, Chantre, Calandrini and González-Andújar2021) documented that doubling the recommended sowing density of barley led to a 24% decline in the population density of L. rigidum, which was attributed to interspecific competitive interactions. Our research has found that at low weed density (20 plants m−2), yield loss was similarly low (<5%) under both intermediate and high sowing densities. This implies that once wheat stand density is sufficiently high to preempt resources and suppress weed establishment, further increases provide diminishing returns in terms of yield protection under low weed pressure. However, under high weed pressure, even the highest sowing density could not prevent considerable yield loss (35% at 420 plants m−2). This indicates that increasing crop density alone cannot fully offset yield losses under severe weed pressure. Therefore, elevated wheat sowing rates should be regarded as an important component within an IWM system, rather than a stand-alone solution. Our results emphasize that spike density and grains per spike are the primary yield components affected by L. perenne ssp. multiflorum competition, whereas 1,000-grain weight remains relatively stable. This suggests that higher wheat density can suppress L. perenne ssp. multiflorum growth during the tillering and booting stages, reducing competition for resources required for spike formation and grain set.
Overall, this study provides the first systematic quantification of key biological and competitive traits of L. perenne ssp. multiflorum within a Chinese winter wheat cropping system, offering novel and actionable insights for its sustainable management. We reveal that L. perenne ssp. multiflorum exhibits broad adaptability to environmental conditions, germinating effectively across a wide range of temperatures (10 to 25 C), pH levels (4 to10), and light conditions, and tolerating moderate salinity and osmotic stress, all of which contribute to its success as a dominant weed in wheat fields. Building on our findings, we propose two targeted, ecology-based management strategies against L. perenne ssp. multiflorum in winter wheat systems. First, strategic deep tillage (>14 cm) can be used periodically to bury seeds below their maximum emergence depth. To minimize the associated risk of soil erosion, this practice should be applied as an occasional intervention rather than a fixed component, ideally within a rotation that includes reduced- or no-tillage phases. Second, moderately increasing wheat sowing density can mitigate yield loss by enhancing crop competition. Although higher seeding rates raise input costs, they may lower long-term reliance on herbicides and slow the evolution of herbicide resistance (Datta et al. Reference Datta, Ullah, Tursun, Pornprom, Knezevic and Chauhan2017). We recommend a wheat sowing rate of 135 kg ha−1 for fields with moderate L. perenne ssp. multiflorum pressure, whereas in fields with severe or persistent infestations, sowing rates could be cautiously increased to 202.5 kg ha−1. It is important to note that wheat sowing density should remain within agronomic limits to avoid adverse effects such as lodging, reduced light penetration, and increased disease pressure (Weiner et al. Reference Weiner, Griepentrog and Kristensen2001). Crucially, the optimal seeding rate should be integrated with other management strategies. This integrated approach ensures that sowing density is not a stand-alone solution but a key component within an adaptive, ecology-based management system. These findings provide a basis for developing sustainable IWM strategies that reduce herbicide reliance and protect food security.
Funding statement
The present work was financially supported by the National Key R&D Program of China (no. 2023YFD1400500), National Natural Science Foundation of China (no. 32502523), and Shandong Postdoctoral Science Foundation (no. SDZZ-ZR-202501120).
Competing interests
The authors declare no conflicts of interest.