Pregnancy is an important time physiologically whereby changes in body weight and adiposity occur.(Reference Chandra and Paray1) The total change in body weight from the time of conception to the onset of labour can be defined as total gestational weight gain (GWG).(2) Adequate GWG is required for healthy fetal growth. However, excessive GWG has been linked with several serious adverse effects, including increased risk of cesarean section, large for gestational age baby(Reference Kominiarek and Peaceman3) and gestational diabetes mellitus (GDM).(Reference Langley-Evans, Pearce and Ellis4) In addition, long-term effects of excessive GWG include, postpartum weight retention(Reference Meyer, Gjika and Raab5) and increased risk of the offspring experiencing childhood obesity.(Reference Kominiarek and Peaceman3) Inadequate GWG is also associated with an increased risk of negative outcomes, including an increased incidence of preterm birth(Reference Goldstein, Abell and Ranasinha6) and small for gestational age baby.(Reference Santos, Voerman and Amiano7) Despite the known adverse effects, there are currently no GWG guidelines used within practice in the United Kingdom (UK) or Ireland, as the most recent National Institute for Health and Care Excellence (NICE) guidelines(8) stated there was lack of evidence about what the optimal total weight change in pregnancy should be. This gap in evidence reflects the multifaceted challenges inherent in assessing GWG, encompassing its guidelines and the complexities of its assessment. The aim of this review is to consider the evidence on weight management during pregnancy, with a focus on the key challenges surrounding GWG and the practical considerations related to its assessment.
Challenges to monitoring weight during pregnancy
Prior to the 1990s, routine weighing during pregnancy had been part of clinical practice in the UK but was phased out due to concerns that it caused anxiety to pregnant women.(Reference Dawes and Grudzinskas9) However, as obesity rates continue to rise, it is now time to consider whether there is a need to reintroduce regular weighing into routine practice throughout pregnancy. This would enable the collection of evidence on changes in GWG and its potential link to obesity rates in women of childbearing age. The need for re-introducing routine weighing has been widely debated, with arguments for and against the practice being cited.(Reference Allen-Walker, Woodside and Holmes10,Reference Fealy, Davis and Foureur11) Recent reports no longer support the view that routine weighing causes anxiety to pregnant women,(Reference Brownfoot, Davey and Kornman12) finding women expected to be weighed during pregnancy.(Reference Allen-Walker, Mullaney and Turner13) Several studies(Reference Daley, Jolly and Jebb14–Reference Allen-Walker, Hunter and Holmes16) have also been carried out which involved regular weighing of women during pregnancy which reported no barriers from the women that would inhibit re-introduction of the practice into standard antenatal care. Still, the most recent UK guidelines(8) reaffirm previous guidance(17) that women should not be routinely weighed throughout pregnancy, unless there is a clinical reason to do so, e.g., GDM. Regardless of these guidelines, practice within UK healthcare varies between NHS trusts, with a study(Reference Goddard, Astbury and McManus18) of twenty-eight trusts in England finding eleven recommended routine weighing throughout pregnancy, however of these only two protocols clearly contained what the purpose of weighing was. Australian Clinical Practice Guidelines were updated in 2018,(19) and this included a recommendation that all women be routinely weighed at every appointment throughout pregnancy. Despite this change to policy, inconsistent weight management practices in Australia have continued to be reported,(Reference Hollis, Deroover and Licata20,Reference Fitzpatrick, de Jersey and Wilkinson21) meaning the effectiveness of this policy change is difficult to evaluate due to women receiving inconsistent care. A recent meta-synthesis of seventeen studies(Reference Escañuela Sánchez, Meaney and O’Connor22) from high income countries, including the UK, Ireland, US and Australia, identified facilitators and barriers reported by women which influenced weight management behaviours during pregnancy. One of the reported themes was healthcare professionals’ attitudes, which was identified in five of the seventeen studies, all of which were UK and Ireland based studies. These studies reported insensitive communication from health care professionals resulting in women feeling judged and dismissed, which influenced women’s ability to engage in positive weight management behaviours. For potential routine weighing and further education on weight management to be both positive and successful, the health care professionals carrying out these measurements need to be provided with and aptly trained on guidelines discussing weight and weight management. Given the lack of focus on GWG within UK guidelines it is perhaps not surprising that midwives have reported a lack of resources and expertise within the area of weight management, as well as a lack of clarity on when to refer to further weight management services.(Reference McCann, Newson and Burden23,Reference Atkinson, French and Ménage24) Barriers including lack of information and no set GWG guidelines within the UK, taken together with these factors mean health professionals are unlikely to raise the topic of weight.(Reference Langley-Evans, Pearce and Ellis4) Nevertheless, recent focus groups carried out with women post-pregnancy from the UK/Ireland identified that at an institutional and policy level, the system is failing due to lack of overall clear guidelines and lack of information surrounding weight management.(Reference Nolan, Gallagher and Hill25) Financial considerations of access to weight management support were also raised within the focus groups(Reference Nolan, Gallagher and Hill25) with those of low socioeconomic status often associated with increased risk of adverse outcomes,(Reference Thomson, Moffat and Arisa26) highlighting a need for strategies to ensure all women are supported with regards to weight management. It is clear that further changes need to be made within the UK; however, the debate on routine weighing is complex and further research on guidelines is needed.
Components of gestational weight gain
During pregnancy multiple physiological changes occur to support healthy growth of the fetus, these changes are what make up the components of GWG. A healthy BMI pregnancy would expect to gain between 11.5 and 16 kg, half of this is made up of the fetoplacental unit, including the fetus 3.2–3.6 kg (28%), placenta 0.7 kg (6%), uterus 0.9 kg (8%) and amniotic fluid 0.9 kg (8%). Further weight contributions include an increase in blood volume between 1.4 to 1.8 kg (7%), extravascular fluid between 0.9 and 1.4 kg (9%), breast tissue from 0.45 to 1.4 kg (8%) and an increase between 2.7 and 3.6 kg (26%) in fat stores.(Reference Dalfra’, Burlina and Lapolla27) All of these physiological processes require energy meaning during pregnancy energy demands increase in order to facilitate these changes.(Reference Ho, Flynn and Pasupathy28) As a result, during pregnancy the Scientific Advisory Committee on Nutrition(29) recommend an increase of 191 kcal per day, yet this increment is only recommended in the third trimester of pregnancy. This recommendation is considerably lower than other worldwide recommendations which advise increments in all three trimesters that increase as pregnancy progresses with a final additional caloric requirement of 475kcal per day in the third trimester(30). Scientific Advisory Committee on Nutrition’s recommendation(29) acknowledges a reduction in physical activity related energy expenditure during pregnancy meaning additional kilocalories are not required that could lead to additional gain of adipose tissue during pregnancy and therefore lead to excessive weight gain. This is the same recommendation from the previous Committee on Medical Aspects of Food and Nutrition Policy(31) report but is even more critical now due to increasing prevalence of overweight and obesity, as pregnancy has been identified as a time women are more open to health and lifestyle messages. All components of GWG are essential to ensure a healthy pregnancy however each component can vary in weight for each pregnancy, making it difficult to distinguish between essential weight gain and excessive weight gained as excess fat. For example, blood plasma volume has been shown to vary considerably throughout pregnancy effecting total weight, its status has also been linked with adverse pregnancy outcomes and fetal birthweight(Reference Pirani, Campbell and MacGillivray32), but monitoring can be expensive and invasive(Reference Aguree and Gernand33). The timings of these components growth also vary widely where maternal blood volume begins increasing in the first trimester, rises rapidly in the second, grows more slowly in the third and levels off in the final weeks of pregnancy(Reference Boron and Boulpaep34). All these factors make measuring excessive GWG more difficult to quantify. Weight gain is minimal during the first trimester assuming a 0.5–2 kg increase(2), which is mostly due to the increase in blood volume and early placental development, as fetal growth has a negligible contribution to GWG in the first trimester(Reference Gilmore, Klempel-Donchenko and Redman35). Meaning additional weight gained within this trimester can be attributed to an increase in maternal fat stores, and preliminary research has shown that excessive weight gained in the first trimester can predict excessive GWG throughout pregnancy(Reference Knabl, Riedel and Gmach36). Excessive weight gained in the first trimester has also been associated with an increased risk of developing GDM, highlighting the potential influence of the timing of weight gain(Reference MacDonald, Bodnar and Himes37), and the need for risks around excessive GWG to be discussed early in pregnancy or at pre-conception meetings.
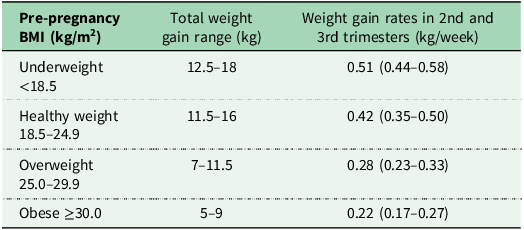
Current guidelines and recommendations for gestational weight gain
Currently there are no GWG guidelines within the UK, as reiterated in the most recent 2025 NICE guidelines.(8) These guidelines do not specify a recommended amount of weight change and as a result, advice states women are not to be routinely weighed throughout pregnancy unless there is a clinical reason to do so. However, the NICE guidelines(8) also stipulate if women are monitoring their own weight, healthcare professionals can refer them to the US guidelines published by the National Academy of Medicine’s (NAM) (previously known as the Institute of Medicine guidelines).(2) These guidelines were published in 2009 following an update from the previous 1990 guidelines,(38) in order to utilise pre-pregnancy BMI categories, as defined by the WHO (underweight <18.5 kg/m2, healthy weight 18.5–24.9 kg/m2, overweight 25.0–29.9 kg/m2 and obese ≥30 kg/m2), as the determinant for how much weight a woman should gain during pregnancy, with lower weight gain recommended for those within higher BMI categories (see Table 1). The NAM guidelines are widely used and referred to, with large cohort studies reporting associations between excessive GWG, defined by the NAM guidelines and an increased risk of adverse maternal and neonatal outcomes.(Reference Truong, Yee and Caughey39) A systematic review and meta-analysis of over 1 million pregnant women(Reference Goldstein, Abell and Ranasinha6) from several countries worldwide found that nearly 50% of women gained excessive weight whilst 23% gained less than recommended when comparing to the NAM guidelines. Further to that this same systematic review and meta-analysis reaffirmed that when compared with those with adequate weight gain, GWG greater than or less than NAM guidelines were associated with a higher risk of adverse maternal and infant outcomes. A UK based study of over 2 thousand women(Reference Hollis, Crozier and Inskip40) supported these results that again nearly 50% of women gained excessive gestational weight and 23% gained less than the guidelines, showing that the majority of women both worldwide and in the UK are gaining excessive weight during pregnancy, and therefore changes need to be made. Nevertheless, the NAM guidelines were developed for a US population, based on the prevention of both large for gestational age and small for gestational age babies, prevention of c-section and postpartum weight retention. Whilst GDM and preeclampsia were acknowledged as being important maternal health outcomes, the NAM guidelines were not developed with these outcomes in mind, indicating clear limitations as GDM is one of the most common complications of pregnancy. These guidelines are expected to be generalisable to other highly developed countries, yet they may not translate in a global context particularly to low-income countries. A further limitation is that these guidelines are not tailored to women with pre-existing health conditions, and despite there being three different classifications to describe the severity of obesity, all women living with obesity are recommended to gain within the same GWG range.
NAM US Guidelines for weight gain in pregnancy according to pre-pregnancy BMI

National Academy of Medicine (IOM, 2009) Note these guidelines are for a US population and are currently not adopted in the UK. BMI, Body mass index.
The Lifecycle Project(Reference Voerman, Santos and Inskip41) proposed independent updated GWG guidelines based on the range of GWG that was associated with lower risk of adverse outcomes from data on 197,670 pregnancies across 25 cohort studies from Europe and North America. Like the NAM guidelines recommendations were stratified by pre-pregnancy BMI, but the optimal ranges suggested varied substantially and overall showed limited predictive value, and only short-term maternal and neonatal outcomes were considered. Women categorised as overweight were advised to gain between 2 kg and<16 kg, more than 3 times the range recommended by NAM. In contrast, those in the underweight category were recommended to gain between 14 kg and <16 kg, representing just over half the range proposed by NAM. The stark difference in these ranges may be due to substantial differences in sample sizes of the pre-pregnancy BMI groups with n= 7809 women classified as underweight whilst n=38,828 were classified as overweight potentially resulting in increased heterogeneity. The project did also provide recommendations for each obesity class with optimal ranges suggesting those within; class 1 obesity (30.0–34.9 kg/m2) gain between 2 kg to less than 6 kg, class 2 obesity (35.0–39.9 kg/m²) were advised to lose weight or gain less than 4 kg, class 3 obesity (≥40·0 kg/m²) weight gain between 0 kg to less than 6 kg was suggested. Weight loss during pregnancy is currently not recommended due to the potential adverse effects for the baby.(8) However, due to the continued rising levels of obesity along with high prevalence of GDM and postpartum weight retention within this group, studies including the Lifecyle Project have questioned the need to lower recommendations, especially for severe classes of obesity. This was further investigated in a recent study of over 15 thousand pregnancies within the obese category from the Stockholm–Gotland Perinatal Cohort.(Reference Johansson, Bodnar and Stephansson42) Findings align with existing calls to reduce or remove the lower GWG threshold in the NAM recommendations for women with obesity. Furthermore, the results highlight the potential need for distinct guidelines tailored to women with class 3 obesity as within that class, weight gain below 5 kg was associated with lower risk of adverse outcomes.
Overall, there is currently a lack of global consensus with regards to optimal GWG, as whilst the NAM guidelines are widely used, many countries opt for variations of these guidelines, which are still dependent on pre-pregnancy BMI, but advise slightly different ranges for optimal weight gain.(Reference Scott, Andersen and Valdez43) However, many Latin American countries follow their own guidelines as the average height of a Latin American woman is 20 cm lower than that of US women,(Reference Mardones, Rosso and Erazo44) making guidelines based on their own population a more appropriate choice. The Rosso-Mardones chart(Reference Rosso, Krasovec and Anderson45) was based on a study of Chilean women who had uncomplicated, full-term pregnancies in the 1980’s. This chart provides personalised weight gain targets based on each mother’s height and weight starting from 10 weeks gestation; but, it does use region-specific adjusted BMI thresholds.(Reference Mardones and Rosso46) In subsequent years, the chart was adapted and refined, resulting in the development of the Atalah curve.(Reference Atalah, Castillo and Castro47) Which unlike the Rosso-Mardones chart uses BMI thresholds categorised by the WHO to plot expected weight gain relative to gestational age. These charts track weight gain throughout pregnancy for each BMI category making it a useful tool for clinicians to monitor weight at any gestational week, and women can be classified as having inadequate, adequate or excessive weight gain for their BMI category.(Reference Morais, Nascimento and Godoy-Miranda48) Differing GWG guidelines and a lack of global consensus can make the comparison of cohorts difficult. Therefore, the WHO has assembled a Technical Advisory Group who are currently in the process of developing global GWG standards(49) to reduce the risk of maternal and infant adverse outcomes at a global level. These forthcoming standards may represent a shift away from NAM guidelines which were never intended for worldwide use and potentially adopt a chart-based system, similar to the Atalah curve described. This approach could enhance clinical utility for monitoring GWG during antenatal care, particularly if percentile-based cut offs are incorporated which could identify women at risk of adverse outcomes. This focus on monitoring GWG could also establish routine weighing practices with optimal timing of when weighing should take place. Overall, these developments could provide a more uniform approach to global guidelines making worldwide comparison easier in future research.
Interventions targeting weight management pre-pregnancy and during pregnancy
Maternal weight status is a key determinant of pregnancy outcomes, with overweight, obesity and excessive GWG all associated with increased risk of adverse effects.(Reference Langley-Evans, Pearce and Ellis4) Optimal GWG is influenced by pre-pregnancy weight status, and interventions implemented both before and during pregnancy aim to support healthy weight trajectories and improve maternal and neonatal outcomes.
Pre-pregnancy
In the UK, it is estimated that 23% of women enter pregnancy with a BMI in the obese category (≥30 kg/m²), with around 54% entering with a BMI ≥ 25.0 kg/m² (NMPA, 2022),(49) meaning over half of all pregnancies in the UK are affected by overweight or obesity. Adverse pregnancy outcomes associated with maternal obesity are well recognised, including an increased risk of pre-eclampsia,(Reference He, Dai and Hu51) GDM,(Reference McIntyre, Catalano and Zhang52) gestational hypertension,(Reference Athukorala, Rumbold and Willson53) macrosomia and caesarean section(Reference Marchi, Berg and Dencker54,Reference Vats, Saxena and Sachdeva55) . NICE guidelines(8) do advise women to enter pregnancy at a healthy weight in order to avoid these adverse effects.
Intervention studies targeting weight loss prior to pregnancy are difficult to conduct, due to difficulties in identifying women who are planning a pregnancy and recruiting women who do become pregnant within the time frame of the study.(Reference Lim, Harrison and Callander56) This means there is a lack of good quality intervention evidence on the effectiveness of weight loss interventions prior to pregnancy and their resultant impact on maternal and neonatal outcomes.(Reference Barker, Dombrowski and Colbourn57) Obesity has also been linked with infertility and is the motivation for many women to lose weight prior to pregnancy. These women are more likely to be open to intervention, with preconception weight loss interventions being shown to have a positive effect on pregnancy rates.(Reference Caldwell, Gorczyca and Bradford58) Prior to pregnancy women with obesity have utilised methods such as bariatric surgery in order to lose weight prior to conception and to maintain weight loss. Bariatric surgery has been seen to have positive effects on fertility helping women to conceive following weight loss. However, the guidelines on timing of conception following bariatric surgery are conflicting depending on factors such as the type of bariatric procedure (gastric bypass, gastric banding and sleeve gastrectomy) and age of the woman.(Reference Huluță, Apostol and Botezatu59) A recent cross-sectional, study of 1282 women who had undergone pre-pregnancy bariatric surgery,(Reference Eccles-Smith, Griffin and McIntyre60) were matched in a 1:10 ratio for age, parity, smoking status and BMI to women without bariatric surgery. It was found within this study that pre-pregnancy bariatric surgery was associated with a reduction in several obesity related pregnancy complications, such as GDM and gestational hypertension. Nonetheless, pre-pregnancy bariatric surgery was also associated with altered offspring growth, including an increased risk of small for gestational age and preterm birth. The use of glucagon-like peptide 1 receptor agonists (GLP-1 RAs), such as semaglutide and liraglutide, have also become more commonly used to promote weight loss, especially prior to pregnancy due to their indirect effects on fertility, primarily through improved insulin sensitivity and reduced body weight.(Reference Dib, Jones and Vounzoulaki61) Due to safety concerns, GLP-1 RA are not currently recommended during pregnancy, and contraception is advised to those taking these medications due to the unknown effects of exposure to GLP-1 RA in early pregnancy.(Reference Varughese, O’Mahony and Varadhan62) More research is required on the effects on fetal development and growth as well as impaired maternal glycaemic control.(Reference Varughese, O’Mahony and Varadhan62)
Despite the current lack of data regarding weight loss pre-pregnancy interventions, the association between overweight and obesity during pregnancy and adverse pregnancy outcomes remains clear.(Reference He, Dai and Hu51,Reference McIntyre, Catalano and Zhang52,Reference Marchi, Berg and Dencker54) Therefore, more work is required within this area to promote the public health message of entering pregnancy at a healthy weight, and relevant interventions on the most effective and safest way to lose weight prior to pregnancy need to be further explored.
During pregnancy
Weight management interventions during pregnancy aimed at preventing excessive GWG usually focus on diet and/or physical activity interventions but have shown conflicting results. A recent systematic review and meta-analysis,(Reference Teede, Bailey and Moran63) of 117 randomised controlled trials including over 34,000 pregnancies aimed to evaluate the effect of diet and physical activity lifestyle interventions on GWG and maternal and neonatal outcomes. Overall findings concluded that interventions were associated with reduced GWG (−1.15 kg; 95% CI, −1.40 to −0.91), risk of GDM (odds ratio [OR], 0.79; 95% CI, 0.70–0.89) and total adverse maternal outcomes (OR, 0.89; 95% CI, 0.84–0.94). Dietary interventions appeared to have a greater impact on GWG than physical activity alone or combined interventions; however, the effects of diet alone versus diet combined with physical activity could not be distinguished. Diet interventions were associated with improved maternal and neonatal outcomes, whilst physical activity was associated with improving only maternal outcomes. Interventions with a nutritional component have previously been investigated and shown to be effective at reducing GWG and improved obstetric outcomes.(Reference Thangaratinam, Rogozińska and Jolly64) A further systematic review and meta-analysis(Reference Vincze, Rollo and Hutchesson65) on the effect of diet intervention on GWG found, out of 20 studies included during pregnancy, 11 had a significant effect on reducing GWG. Though, many studies were of moderate quality due to the lack of clarity of study methods. This demonstrates the importance of how these interventions are carried out as they can vary widely in their characteristics, including when they begin during pregnancy, the type of information provided and the intensity, duration and delivery methods, which can cause inconsistencies during analysis. Many of the interventions aiming to reduce excess GWG target those only within the overweight and obese categories, and whilst many of these interventions have been successful in reducing GWG, the resultant effect on maternal and neonatal outcomes has not always been evident.(Reference Wu, Jin and Hu66) The Lifestyle Interventions for Expectant Moms (LIFE-Moms) consortium(Reference Peaceman, Clifton and Phelan67) recruited a total of 1150 women with overweight or obesity across seven randomized clinical trials in the US. Pooled results found intervention to have a significant effect on reducing GWG in comparison to control groups, however most women in the intervention still exceeded US NAM guidelines. In addition to this, there was no resultant significant difference in maternal outcomes including macrosomia, caesarean section or GDM between intervention and control groups. The UPBEAT trial(Reference Poston, Bell and Croker68) intervention, carried out in the UK (2202 participants), which primarily aimed to reduce GDM risk in women with obesity, resulted in a modest reduction in GWG (0.55 kg), it did not significantly affect GDM incidence or other pregnancy complications. The International Weight Management in Pregnancy group conducted an individual participant data meta-analysis from 36 randomised trials including over 12,000 pregnancies.(69) This study aimed to assess the effects of diet and physical activity-based interventions, on GWG and maternal and offspring outcomes in women who were of a healthy weight or had overweight or obesity. Results confirmed that diet and physical activity-based interventions carried out during pregnancy significantly reduced GWG when compared with control groups (summary mean difference −0.70 kg, 95% CI −0.92 to −0.48 kg, I2 = 14.1%). This beneficial effect was observed across all maternal BMI categories underscoring the importance of providing weight management intervention to all due to the potential risk of postpartum weight retention,(Reference Rong, Yu and Han70) which could lead to entering further pregnancies at a heavier weight.(Reference Knight-Agarwal, Williams and Davis71) Despite the reduction in GWG, interventions did not significantly reduce the risk of negative maternal and offspring outcomes, with the exception of caesarean sections. Currently within the UK only women within obesity class 3 (≥40·0 kg/m²) are offered referral to a specialist obesity service for tailored advice on weight management during pregnancy. Meaning the majority of women with overweight and obesity (≥25·0 kg/m²) are not receiving specialist advice on weight management. It has also been shown that interventions that include delivery from a health care professional or those with an individual in person delivery format have been more successful in reducing GWG.(Reference Harrison, Bahri Khomami and Enticott72,Reference Yu, Li and Qian73) This could potentially be achieved through enhancing existing antenatal care models, which could be the most cost-effective solution.(Reference Walker, Bennett and Blumfield74) However, the success of using mHealth online and app-based interventions is yet to be determined during pregnancy, due to the vast heterogeneity of this new and emerging field.(Reference Raab, Geyer and Zagar75) More research into the most effective weight management strategies are still needed, and whilst some interventions are successful in reducing GWG, the resultant link, or lack of, to maternal and neonatal outcomes in both the short and long-term also need to be considered.
The relationship between pre-pregnancy BMI and gestational weight gain
Due to the clear association between overweight, obesity and adverse pregnancy outcomes, there has been a call for the focus to be moved away from GWG, instead placing focus on weight management prior to conception.(Reference Dodd, Deussen and Mitchell76) A recent study challenged the practice of managing GWG throughout pregnancy suggesting that excessive GWG does not have a direct effect on pregnancy outcomes whereas pre-pregnancy BMI has a clear direct effect on outcomes and therefore should be the primary focus.(Reference Dodd, Louise and Deussen77) Further to this, the Lifecycle project,(Reference Voerman, Santos and Inskip41) which aimed to estimate optimal GWG ranges in order to reduce adverse maternal and infant outcomes, did provide updated recommended ranges yet they did state these had limited predictive values on the adverse outcomes assessed. The project also suggested that pre-pregnancy weight would be a more important target for intervention instead of GWG. However, this study only focused on short term outcomes and did acknowledge that if the outcomes were to be measured over time, results may differ. The long-term effects of excess GWG are an important factor which also need to be considered as it has been shown that those who gain above the recommended NAM guidelines are more likely to have both a higher BMI(Reference Nolan, Hill and Gallagher78) and higher risk of obesity in the long-term after pregnancy.(Reference Mamun, Oken and McIntyre79) Excessive GWG has also been shown to be associated with childhood obesity(Reference Josey, McCullough and Hoyo80) a follow up study of 766 mother and child pairs from North Carolina USA, found excessive GWG was associated with a higher risk of childhood obesity, assessed via BMI z scores, at age three. These findings emphasise the impact of excessive GWG on the risk of childhood obesity, independent of mothers pre-pregnancy weight status, highlighting an important public health message for all mothers, supporting that GWG should be monitored for all women to avoid excessive weight gain. Nonetheless, establishing the specific threshold of GWG that should be classified as excessive and that significantly increases the risk of adverse outcomes, does require further clarification. A large population based retrospective cohort study of over 2 million pregnant women in the US found(Reference Truong, Yee and Caughey39) that women who exceeded guidelines by 20 lbs (9 kg) or more were particularly at risk of adverse outcomes, and this was found regardless of pre-pregnancy BMI. As women with a healthy pre-pregnancy BMI who gained more than 20 lb over the NAM guidelines had a sixfold higher risk of fetal macrosomia, compared to those who stayed within the guidelines. For women with overweight or obesity, gaining an extra 20 lb increased the risk by three to four times, again displaying the effect of excessive weight gain regardless of pre-pregnancy BMI. Whilst the effect of pre-pregnancy BMI is undeniable and an essential consideration, with an estimated 45% of pregnancies in the UK being unplanned(Reference Wellings, Jones and Mercer81) the advice to enter pregnancy at a healthy weight cannot always be implemented. GWG is therefore an important factor for women to consider, emphasising not only a focus on weight but also the promotion of healthy lifestyle behaviours that help prevent excessive GWG and support long-term health habits beyond pregnancy. Overall, supporting women to achieve and maintain a healthy weight both pre-pregnancy and during pregnancy are important public health priorities.(Reference Wellings, Jones and Mercer81)
Practical considerations when measuring weight during pregnancy
GWG is defined as the total change in body weight from the time of conception to the onset of labour; GWG is calculated by the final weight in pregnancy minus pre-pregnancy weight. However, several challenges arise when attempting to accurately capture these weight measurements. These challenges include obtaining a pre-pregnancy weight measurement, assessing total GWG and the use of self-reported body weight for GWG estimations, which are considered below, together with alternative methods for determining GWG.
Pre-pregnancy weight
Several challenges arise with the practicality of obtaining a measured pre-pregnancy weight from a preconceptual visit. To obtain an accurate measured pre-pregnancy weight close to conception, a woman would need to be planning to conceive in order to know a weight is required, however with 45% of pregnancies being unplanned(Reference Wellings, Jones and Mercer81) this is not feasible in nearly half of cases. The method of obtaining an accurate pre-pregnancy weight has been shown in large preconception cohort studies(Reference Boxem, Blaauwendraad and Mulders83) such as the Southampton Women’s Survey(Reference Hollis, Crozier and Inskip40) which recruited non pregnant women aged 20–34 years. At recruitment all women were weighed and those who subsequently became pregnant were then followed up. Of the 12,583 women recruited, 25% had live singleton births and due to missing data on weights, a final total of 2,559 were included in the sample. This demonstrates the large-scale studies that are required to accurately capture pre-pregnancy weight. Whilst capturing pre-pregnancy data is valuable it does still come with its limitations, within this study some pre-pregnancy weights were obtained several years prior to conception, with a reported median of 1.8 years and interquartile range of 0.8–3.2 years. Fluctuations in weight up to 3 years prior to pregnancy could be substantial, particularly if within those years, women decided to plan to conceive and therefore made changes to diet and lifestyle habits and as a result weight.(Reference Diouf, Charles and Thiebaugeorges84)
Assessment of total gestational weight gain
An early weight collected during the first trimester of pregnancy is often used as a substitute for a pre-pregnancy weight. This is due to the wide availability of this weight as NICE guidelines(8) do advise that all women be weighed at their first face-to face appointment in order for BMI to be calculated. Weight gain during the first trimester is also reported to be minimal, which assumes a first trimester weight gain between 0.5 and 2 kg(2) which is why many studies use an early weight as a substitute to pre-pregnancy weight.(Reference Fattah, Farah and Barry85) Further research reinforces this where 474 Chinese women were weighed both before pregnancy (median 17.1 weeks prior to pregnancy) and in early pregnancy (8–14 weeks gestation).(Reference Retnakaran, Ye and Wen86) Consistency was found between mean pre-pregnancy weight (49.8 kg ± 6.4) and early pregnancy weight (51.1 kg ± 7.0), showing an early pregnancy weight taken between 8–14 gestational weeks could be a reasonable substitute for pre-pregnancy weight. Nevertheless, there is variability on how studies define baseline weight. Many longitudinal studies(Reference Reynolds, Egan and O’Malley87) exclude women from analysis if their first pregnancy weight is after 15 weeks of gestation, as this is deemed as a cut-off point before substantial weight gain begins. The Lifestyle Interventions for Expectant Moms (LIFE-Moms),(Reference Clifton, Evans and Cahill88) used between 9 and 15 weeks as the baseline weight for their studies. For participants with a baseline weight within the first trimester of pregnancy no weight gain was assumed. However, within this study those with a baseline weight measurement at 14 or 15 weeks had a modified GWG with a subtraction of 0.45 kg for those at 14 weeks and 0.91 kg for those at 15 weeks to account for extra weight gained after the first trimester. These deductions were based on data from a previous study of over 10,000 women,(Reference Roberts, Myatt and Spong89) this method has also been used in recent studies(Reference Crivellenti, Carreira and de Lima90) as a standardised method to define baseline weight. Further studies have reported(Reference O’Higgins, Doolan and Mullaney91) weight gain does not begin until around 18 weeks gestation meaning any measurement before 18 weeks can be used as a baseline measurement.(Reference O’Higgins, Doolan and McCartan92) Similarly, this issue is also experienced with the final weight of pregnancy where ideally weight would be measured prior to the onset of labour but is more often measured somewhere within the third trimester which is anywhere from 28 to 40 weeks. Differing gestational week cut offs both at the beginning and the end of pregnancy can make comparing studies difficult. In order to address these issues many studies do calculate rate of weight gain per week (total weight gain divided by the number of gestational weeks available) which makes comparison between studies easier, however if gestational weeks are limited this may not provide an accurate estimation of total GWG. Gilmore and Redman (2015)(Reference Gilmore and Redman93) conducted a study on a small sample of 52 women using four commonly used methods to calculate weight gain. This included: Method 1, final weight in pregnancy – pre-pregnancy weight, Method 2, final weight in pregnancy (corrected for length of gestation) – pre-pregnancy weight, Method 3, final weight in pregnancy – early weight in pregnancy, Method 4 calculated incremental weight gain, (final weight in pregnancy– second trimester weight)/no. weeks (or days) between weights. Following calculation each result was then categorised as below, within or exceeding the NAM guidelines, symmetry and agreement were assessed between each method using Bowker’s Test and Kappa coefficient, respectively. There was medium classification agreement and symmetry between Methods 1 and 3, most likely due to the typically minimal GWG during the first trimester. Nonetheless, the level of agreement was low and symmetry was significantly different between other methods, demonstrating the essential need for a uniform approach in order to be able to easily compare results from different studies. Overall, this study recommended to use Method 2 of calculating GWG whilst accounting for the length of gestation, which is the most accurate method to analyse total GWG and would ensure consistency and alignment across the research field.
Self-reported weight
In the absence of a measured pre-pregnancy weight, a recalled pre-pregnancy weight is often used to calculate GWG. The accuracy of self-reported weight has been widely investigated, with reports finding women are more likely to underestimate weight which does introduce bias.(Reference Fayyaz, Bataineh and Ali94) Studies focused on self-recalled weight during pregnancy have demonstrated these same trends, often meaning self-recalled weight is deemed unreliable.(Reference Fattah, Farah and O’Toole95) Nonetheless, a study of 5092 women found 87% correctly self-reported their BMI category when compared to a measured pre-pregnancy weight, with a higher risk of misclassification found in women in the overweight and obese categories.(Reference Han, Abrams and Sridhar96) A similar study of over 3000 women found a similar result with 85% of women correctly identifying their pre-pregnancy BMI category.(Reference Bannon, Waring and Leung97) Overall, self-reported pre-pregnancy weight status categories have been shown to be generally reliable and acceptable for population-based research. It has also been suggested that self-reported weights should be assessed using mathematical models rather than simple correlation. For example, the Bland-Altman method is used for assessing the agreement between two measures for a true measure of agreement,(Reference Flegal, Graubard and Ioannidis98) in order to fairly validate self-recalled pre-pregnancy weight with a measured weight which has statistical backing.(Reference Thomas, Halawani and Phelan99) However, in planning future research self-reported data should be avoided,(100) and robust plans on appropriate methods for obtaining measured weights and height should be incorporated into the study design.
Seeking objective methods for assessing gestational weight gain
Measuring gestational body composition change through only monitoring total weight change has been criticised due to the challenges and practical considerations highlighted in successfully capturing a direct measure of excessive fat, due to variations in muscle mass and fluid retention. This is even more critical during pregnancy due to the many components in GWG including not only maternal fat stores but also blood volume, placenta, etc., as previously mentioned within this review. Further to this, during pregnancy some commonly used methods to measure body composition have been deemed not safe, such as dual-energy X-ray absorptiometry(Reference Gilmore, Klempel-Donchenko and Redman35) due to radiation exposure. Therefore, other methods to measure specifically fat mass during pregnancy have been used. Bioelectrical impedance analysis (BIA) is a cost-effective non-invasive technique used to estimate total body water, fat mass, percentage body fat and fat free mass. This is achieved through sending a small electrical current through the hands and feet, which travels at different speeds through different tissues with additional patient information used to calculate results.(Reference Obuchowska, Standyło and Kimber-Trojnar101) BIA has been shown to be safe for use throughout pregnancy and to accurately predict fat mass and percentage body fat(Reference Berlit, Tuschy and Stojakowits102) especially at the beginning of pregnancy.(Reference Mullaney, Cawley and Kennedy103) Yet, as pregnancy develops care needs to be taken with methods and calculations used as accurate measures can be difficult to achieve due to the changes in fluid throughout pregnancy with interactions with extracellular fluid, intracellular fluid and total body water.(Reference Lof and Forsum104) BIA has also been used to detect adverse effects such as gestational hypertension(Reference Valensise, Andreoli and Lello105) and preeclampsia(Reference Sween, Althouse and Roberts106) through monitoring variation in body water compartments. Further methods include measuring total body water through hydrometry and measuring body density through hydrodensitometry. Whilst proven to be safe to use during pregnancy these methods can be time consuming and difficult for pregnant women to complete, especially at the end of pregnancy.(Reference Açar and Köksal107) Air Displacement Plethysmography is widely used to monitor body composition, using a BOD POD which is an expensive piece of equipment to purchase, there has also been reports of unsuccessful measurements particularly near the end of pregnancy and in those with overweight and obesity.(Reference Most, Marlatt and Altazan108) Nevertheless, the use of simpler and less time-consuming anthropometric measurements do remain the most commonly used both in research and in clinics including, weight and height, as well as skin fold thickness and middle upper arm circumference measurements.(Reference Siega-Riz, Faith and Nicholson109) These measurements are used commonly within clinics, and when used alongside prediction formulae by well-trained staff they can be used to estimate body composition, to provide further insight than simply reporting BMI.(Reference Açar and Köksal107)
Conclusion
Assessing GWG is complex and challenging due to lack of guidelines, difficulties in defining excessive GWG, a lack of agreement on routine weighing, and the practicalities of obtaining accurate and timely measurements during pregnancy. Furthermore, the practice of managing GWG itself has been challenged due to reports it does not have a direct effect on pregnancy outcomes but instead an indirect effect alongside maternal BMI. Whilst this is an important consideration, GWG may still represent a meaningful marker of risk; still, without a gold standard uniform approach for its assessment, and given the dependence on accurate measurement, its utility in research and clinical practice is currently being limited. However, the WHO is currently in the process of developing global GWG standards to reduce the risk of adverse maternal and infant outcomes. It is anticipated that these newly developed standards will help address many challenges and practical considerations highlighted in this review by establishing a consistent gold standard for assessing GWG. Furthermore, with increasing recommendation for new research to avoid using self-reported weights and emerging evidence no longer supporting the view that routine weighing causes anxiety to pregnant women, the implementation of routine weighing within the UK and Ireland could be supported. This would allow clinicians to monitor GWG, provided that appropriate training on weight management during pregnancy is ensured. Overall, researchers designing studies investigating GWG and/or its related adverse outcomes must carefully consider the methods used to assess GWG, in order to ensure accurate and reliable results are captured.
Acknowledgements
The authors would like to thank the Irish Section of The Nutrition Society for inviting the present review paper as part of the postgraduate review competition.
Author contributions
R.N drafted the manuscript. A.J.H. and A.M.G. critically revised the manuscript for important intellectual content. All authors have read and approved the final manuscript.
Financial support
This work was undertaken as part of a PhD scholarship funded by the Department for the Economy.
Competing interests
None.