Introduction
Cognitive disorders are a priority public health issue, considering their steadily increasing prevalence associated with an aging population throughout the world. There are over 50 million people worldwide living with dementia in 2020 and this number is expected to double every 20 years, reaching 139 million in 2050, with the fastest increase occurring in developing countries. 1 Their economic impact is very relevant, with nearly 820 billion dollars in 2015, requiring proportionate research investment in prevention, treatment, care and cure. Reference Prince, Wimo and Guerchet2
A better understanding of the diseases underlying cognitive impairment is one of the key elements to tackle this issue, namely establishing an early and correct diagnosis to implement medical and social care adapted to each individual patient. This may include personalized support, but also the introduction of treatments to slow the progression of the decline and reduce its symptoms. An improvement in diagnosis is also a prerequisite to properly test disease-modifying therapeutic strategies, some of which have recently shown promising results. Reference Mintun, Lo and Duggan Evans3–Reference van Dyck, Swanson and Aisen5
The clinical picture of cognitive disturbances alone does not allow identification of its underlying cause with adequate precision, and to differentiate, for example, a cognitive decline due to depression, from a neurodegenerative process. Among the main causes of dementia or cognitive impairment, the most common are Alzheimer’s disease (AD) – which alone accounts for two thirds of cases – vascular dementia, Lewy body dementia and frontotemporal dementia.
The role of imaging as biomarker in cognitive disorders
The initial diagnosis of a cognitive disorder is based on clinical and neuropsychological assessment, following which etiological hypotheses can be established, which are then confirmed or refuted through the use of different disease biomarkers. Among biomarkers, brain imaging plays a central role and includes morphological imaging with Magnetic Resonance Imaging (MRI) as well as functional and molecular imaging with Positron Emission Tomography (PET) and single photon emission computed tomography (SPECT). Thanks to recent technological advances, it is now possible to establish a precise etiological diagnosis in the majority of the cases. Reference Chetelat, Arbizu and Barthel6
The initial goal of anatomical imaging is to verify whether a treatable cause can explain the cognitive disorders (e.g.: ischemia/infarct, extra-/subdural hematoma, hydrocephalus, tumor). It can then be used to guide the diagnosis, in particular by evaluating the presence of regional brain atrophy that could suggest a primary degenerative dementia, as well as the load of vascular lesions or white matter lesions in general. Reference Dadar, Mahmoud and Zhernovaia7 The recommended first-line morphological imaging modality is MRI, which is more sensitive than CT. Reference Hanstock, Sun and Choi8
Molecular imaging by PET
Namely in the case of a clinically atypical or rapidly progressing form of cognitive impairment, in young patients (<65 years) and in all cases in which the combination of clinical, neuropsychological and morphological imaging assessment does not allow the physician to establish an etiological diagnosis with an adequate level of confidence, functional and molecular imaging examinations can be a valuable complementary tool. Reference Frisoni, Boccardi and Barkhof9 Several imaging modalities using different radiotracers have been tested. Reference Villemagne, Barkhof, Garibotto, Landau, Nordberg and van Berckel10 We will discuss here the most commonly used PET tracers, which are already established or have recently been introduced into clinical practice. These could be roughly divided in two main categories: tracers measuring the presence of neurodegeneration or tracers specific for pathological deposits.
PET markers of neurodegeneration
PET markers of neurodegeneration measure a functional parameter for a brain region or pathway which is typically affected by a neurodegenerative process and more or less tightly linked with clinical symptoms. Neurodegeneration markers per definition are nonspecific endpoints, as they do not distinguish between different etiologies affecting the same functional process; however, they can contribute to the process of differential diagnostic as the various etiologies usually have some pattern specificity.
Fluordeoxyglucose ([18F]FDG) PET
The most commonly used neurodegeneration PET marker is 2-[18F]fluoro-2-deoxy-D-glucose ([18F]FDG). Brain PET imaging with [18F]FDG allows to measure regional brain glucose metabolism, using a radiotracer consisting of a glucose molecule on which a hydroxyl group has been replaced by 18-fluorine. This radiotracer is delivered to the brain from blood vessels primarily via glucose transporter Glut-1, and then subject to intracellular phosphorylation. 18-Fluoro-2-deoxy-D-6-phosphate, however, is a poor substrate for subsequent metabolism in the glycolytic chain, thus remaining trapped and accumulating in the cells, allowing the study of regional cellular metabolism. The examination is usually performed with hybrid devices that combine PET acquisition with computed tomography imaging (PET/CT) and most recently with MRI (PET/MRI).
As the physiological brain metabolism is glucose-dependent, the mapping of brain glucose metabolism generally is a reflection of neuronal and synaptic activity and thus allows topographical detection of hypo- or hyperfunctional areas, corresponding to a loss or a synaptic dysfunction at these levels. Since hypometabolism has a different regional topography for different neurodegenerative disorders, [18F]FDG PET has been established as a clinically validated tool useful to differentiate between them. Reference Nobili, Arbizu and Bouwman11–Reference Zahn, Buechert and Overmans13
In AD, a typical topographic pattern of metabolic changes involving mainly the posterior cortex is seen, with hypometabolism of the temporo-parietal neocortex and posterior cingulate gyrus, whereas the primary visual cortex, the cerebellum and the central gray nuclei are relatively spared. This pattern has been a stable and consisting finding through longitudinal as well as cross-sectional studies during the past 20 years, and [18F]FDG PET is likely the most thoroughly validated diagnostic tool in dementia, with a summarized evidence of a sensitivity of 80% and a specificity of 90% for AD in the dementia stage. Reference Garibotto, Herholz and Boccardi12 While there is no effective Lewy body PET ligand, [18F]FDG PET also is helpful in distinguishing Lewy Body Dementia from other dementia, with Lewy Body Dementia showing occipital hypometabolism with sparing of the posterior cingulate (cingulate island sign) and basal ganglia hypermetabolism. Reference Lim, Katsifis and Villemagne14 Dopamine imaging using SPECT ([123I]FP-CIT) or PET ([18F]F-Dopa) may be more specific indicating dopamine system denervation. Reference Miyagawa, Przybelski and Maltais15–Reference Hu, Okamura and Arai17 The development of cerebrospinal fluid (CSF) markers for synuclein holds the promise of introducing less invasive biomarkers, which are anticipated to significantly impact clinical practices once they become widely available. Reference Magalhaes and Lashuel18–Reference Shi, Tang and Toledo20 A caution will be the recognition that many cases of age-related neurodegeneration can show mixed pathology. Moreover, in early stages (i.e., MCI) PET in differential diagnosis may be more challenging.
Despite its limitations, [18F]FDG PET remains a mainstay of diagnostic criteria for various degenerative conditions, namely:
-
Alzheimer’s Dementia (AD) Reference McKhann, Knopman and Chertkow21
-
Mild Cognitive Impairment (MCI) Reference Albert, DeKosky and Dickson22
-
frontotemporal dementia (FTD) Reference Rascovsky, Hodges and Knopman23
-
corticobasal degeneration Reference Armstrong, Litvan and Lang24
-
progressive supranuclear palsy Reference Hoglinger, Respondek and Stamelou25
-
multiple system atrophy (MSA) Reference Gilman, Wenning and Low26 and
-
Lewy Body dementia (LBD) Reference Lim, Katsifis and Villemagne14
The identification of regional patterns of hypometabolism is based on visual interpretation of radiotracer distribution images, supported by a statistical voxelwise comparison with the normal distribution, as observed in healthy individuals. Multiple studies have consistently shown that the support of the semi-quantitative analysis improves the sensitivity and the specificity of [18F]FDG PET, even if this might differ across software. Reference Morbelli, Garibotto and Van De Giessen27,Reference Garibotto, Trombella and Antelmi28
The analysis of glucose metabolism is not only a valid diagnostic marker in various conditions, but also allows studying a number of pathophysiological processes, e.g. the specific networks associated with specific symptoms, Reference Morbelli, Chincarini and Brendel29–Reference Zahn, Juengling and Bubrowski31 or the reserve phenomenon, i.e. the ability of the brain to cope with neurodegeneration by using alternative functional pathways. Reference Bauckneht, Chincarini and Brendel32
Synaptic vesicle protein 2a (SV2A) PET
As a still experimental PET neurodegeneration marker, a number of tracers targeting the synaptic vesicle protein 2a receptor (SV2A) have been developed. Reference Bahri, Plenevaux and Aerts33–Reference Cai, Li and Zhang36 These have shown to represent synaptic density. Reference Finnema, Nabulsi and Eid37 The advantages of SV2A imaging in Alzheimer’s disease over [18F]FDG PET include a higher sensitivity for early detection of neurodegeneration in the hippocampus, potentially improving the differential diagnosis and prediction of disease progression even in the prodromal (MCI) stage of the disease, where [18F]FDG PET often fails to find significant reduction in regional cerebral glucose metabolism (rCGM), unless complex corrections for partial volume correction and spatial mapping are performed. Reference Bastin, Bahri and Meyer38–Reference Maldjian and Whitlow40 In previous trials, SV2A PET has shown reductions in synaptic density in vivo in the entorhinal cortex and hippocampus of individuals with amnestic MCI and mild AD dementia, Reference Chen, Mecca and Naganawa41 reflecting the pathological Braak stages I-III. Reference Braak, Alafuzoff, Arzberger, Kretzschmar and Del Tredici42 This marks the first demonstration of in vivo imaging correlates within a single imaging modality for a pathological staging system in AD.
SV2A-PET also has shown a distinct differential pattern of neurodegeneration for LBD, Reference Andersen, Hansen and Damholdt43,Reference Nicastro, Holland and Savulich44 offering an additional tool for the differential diagnostic between LBD and AD. While availability is currently the main limitation for a more widespread use, it has the potential to possibly replace the role of [18F]FDG PET in the future, provided it becomes more widely available. In Canada, SV2A-PET is currently available or in use for clinical trials at the Universities of Alberta and Toronto. 45
PET markers of pathology
Over the last two decades, a novel category of PET radiotracers has revolutionized the study of AD and other neurodegenerative disorders, i.e. PET tracers specific for amyloid and tau pathological deposits. As the gold standard for AD diagnosis is based on the postmortem immunohistological detection of amyloid and tau pathology, the availability of PET markers specific for these targets has opened the opportunity for an in vivo visualization of their presence and distribution, and ultimately for in vivo diagnosis. This has been reflected in the National Institute on Aging – Alzheimer’s Association (NIA-AA) Research Framework recommendation towards a biological definition of AD by presence of biological markers using the A/T/N classification, where A stands for Amyloid, T for Tau and N for Neurodegeneration. Reference Jack, Bennett and Blennow46
Other conditions characterized by specific pathological deposits, namely by the alpha-synuclein deposits, are Parkinson’s Disease (PD) and LBD; while FTD is characterized by TDP-43 and tau deposits. The development of PET markers for these conditions, however, is still in the earliest phases of development (see 6.1).
Amyloid PET
Over the last two decades, a number of tracers have been developed targeting extracellular beta-amyloid plaques with high sensitivity and specificity. Since the first report of successfully targeting beta-amyloid in 2004 using the PET tracer named Pittsburg compound B (PiB), labeled with carbon-11, Reference Klunk, Engler and Nordberg47 various fluorinated tracers have been developed, allowing tracer commercialization and distribution. These tracers, namely [18F]-Florbetapir, [18F]-Flutemetamol and [18F]-Florbetaben, have been validated for their accuracy in predicting the presence of neuropathological changes and are or have been authorized and marketed in the US and in Europe. [18F]-Florbetaben has been approved for marketing in Canada in 2017, Reference Chiotis, Saint-Aubert and Boccardi48 Table 1.
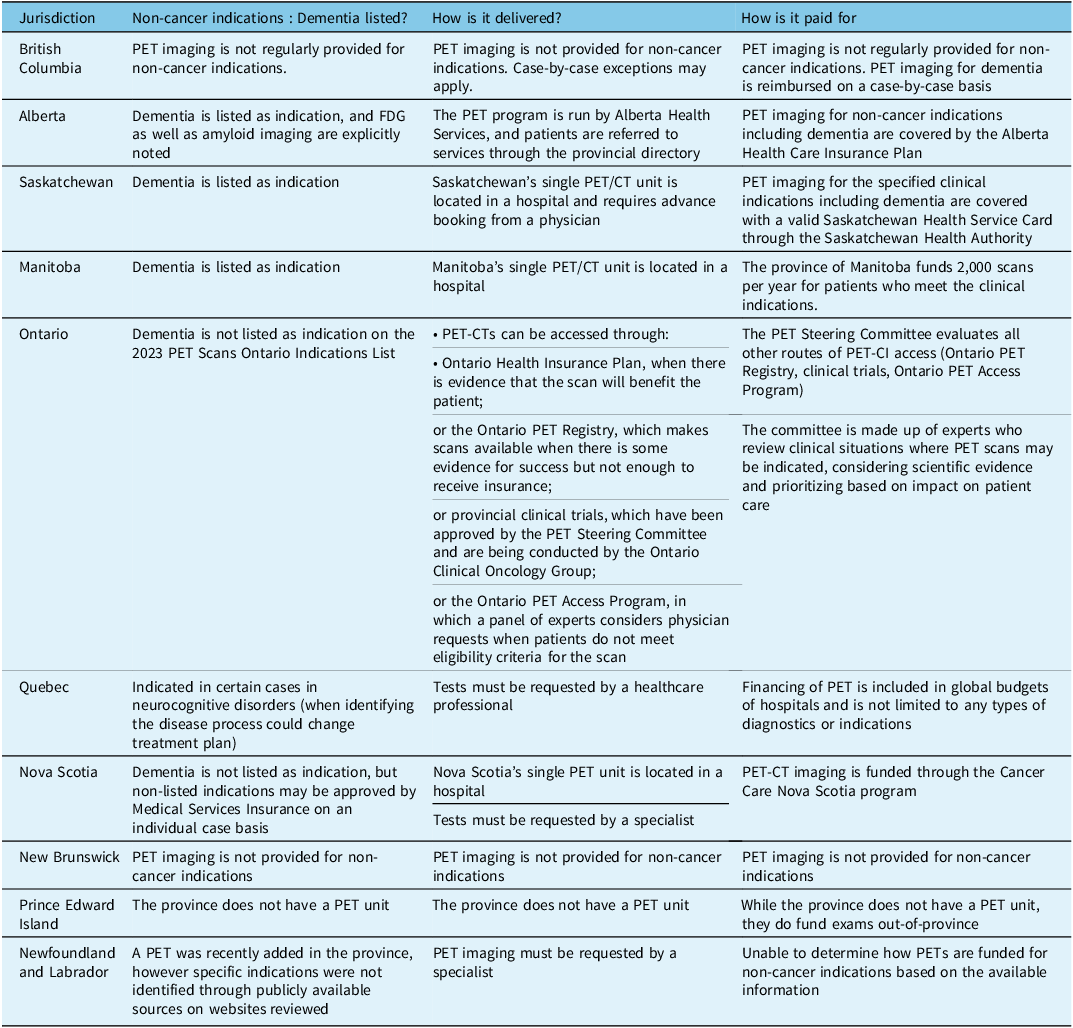
Reimbursement of PET for dementia in Canada (modified and updated from Reference Ismail, Black and Camicioli78 )

Image analysis shows a physiological fixation of the tracer within white matter in a negative case, and conversely, an increased uptake in cortical areas will be considered positive for cortical deposition of amyloid plaques. Specific reading guidelines have been developed for each tracer.
Negative imaging can exclude a diagnosis of AD, whereas positive imaging, especially in association with an imaging marker of neurodegeneration, such as temporomesial atrophy or posterior hypometabolism, allows the identification of prodromal AD among subjects with mild cognitive impairment (MCI) (cf. Figure 1).
71 year old male patient with mild cognitive impairment, Mini Mental State Exam (MMSE) score 27. 18FDG-PET in representative, axial, coronal and sagittal orientation demonstrates temporoparietal and cingulate hypometabolism in a pattern typically seen in early AD (upper panel), while 18F-florbetaben-PET demonstrates significant cortical amyloid load, predominantly in the hypometabolic areas. PET images are fused with the individual patient’s morphological T1-weighted MR images (PET/MRI, isotropic MPRAGE images for coregistration and image display).

Visual interpretation of images is usefully supported by semi-quantitative indices, usually based on a ratio between cortical uptake and uptake in a reference region (often the cerebellum or white matter structures, such as supratentorial white matter or the pons). Importantly, a common scale, called “Centiloid,” has been proposed and is increasingly adopted in order to standardize the semi-quantitative measures obtained with different tracers.
The Centiloid scale is a method used to standardize and compare quantitative measurements of amyloid burden obtained from positron emission tomography (PET) scans. It converts the raw PET data into a common scale, making it easier to interpret and compare results from different studies and imaging centers. The Centiloid value (CL) is calculated using a three-level process, which involves scaling the data to a common reference and then applying a non-negative matrix factorization model to improve its robustness in longitudinal studies. For initial scaling, 0- and 100-anchor points of the Centiloid scale have been defined, with the 0-anchor point to represent a definitively amyloid-negative brain scan, and the 100-anchor to represent the amount of global amyloid deposition found in a typical mild-moderate AD subject. The Centiloid scale, being a mean value, may result in slightly negative values for some amyloid-negative scans. Similarly, there will be a range of “typical AD values” around 100 CL, and approximately half of all AD subjects will have values above 100 CL, sometimes significantly so. The benefits of the Centiloid scale include the ability to draw intuitive conclusions, reduce variability in measurements, improve sensitivity and make results more easily comparable across different analysis techniques and large datasets. and has been adapted to all of the currently available amyloid PET tracers. Reference Klunk, Koeppe and Price49–Reference Doré, Bullich and Rowe51
The largest body of the literature so far is, however, based on a binary classification of amyloid PET scans, as positive or negative, but recent data suggest that visual interpretation can also be sensitive for the earliest phases of amyloid accumulation, possibly providing relevant staging and prognostic information, even if this still needs to be confirmed in prospective studies. Reference Rowe, Jones and Doré52
As a most recent advancement, “early frame” acquisitions of amyloid PET tracers, i.e. acquisitions performed immediately after injection of the radiotracer, usually during the first 5 to 10 minutes, can provide an estimate of relative regional brain perfusion. Given that metabolic coupling of perfusion and glucose metabolism has been reported to be grossly undisturbed in neurodegenerative conditions, “early frame” acquisitions are closely correlated to [18F]FDG PET images, thus simultaneously allowing for obtaining information on neurodegeneration, combined with the information on amyloid status, using a multiphase acquisition. Reference Collij, Salvado and Shekari53,Reference Daerr, Brendel and Zach54 An example of “early frame” 18F-flutemetamol imaging obtained from 1 to 11 minutes post injection, in comparison with the result of [18F]FDG PET imaging, is provided in Figure 2. If a patient is scheduled for an amyloid PET scan as part of a clinical workup and has not undergone a previous [18F]FDG PET scan, obtaining an early frame of the amyloid PET scan may thus eliminate the need for an additional [18F]FDG PET scan for clinical purposes.
Patient with probable AD and cognitive deficits including aphasia. 18FDG-PET demonstrates a significant, predominantly left temporoparietal hypometabolism ( a ). Voxel-based comparison to a reference database ( b ) depicts brain regions with metabolism exceeding two standard deviations below norm (red color scale overlaid on a standard MR anatomical template, statistical parametric mapping, in neurological convention, left on the left side of the image). The early phase of 18F-flutemetamol-PET demonstrates hypoperfusion concordant to 18FDG-PET ( c ) and its deviation from norm ( d ). Results of amyloid-PET and tau-PET of the same patient are shown in Figure. 3 and Figure. 4.

18F -flutemetamol-PET in patient with probable AD and cognitive deficits (same patient as in Fig. 2, images acquired at recommended time interval). The study demonstrates a typical case of increased cortical amyloid in frontal cortex, posterior cingulate and temporolateral cortex, with the occipital cortex being spared. PET images are fused to the patient’s T2-FLAIR-MR images.

18F-flortaucipir-PET in patient with probable AD and cognitive deficits (same patient as in Fig. 2 and Fig. 3). PET demonstrates increased, predominantly left-sided cortical tau deposits in temporooccipital, parietal and frontal cortex, fused to coregistered T1-3D-MRI. Regions with increased tau deposits correlate well with hypometabolic areas seen in 18FDG-PET (Fig. 2) but are significantly more extensive than the metabolic changes.

Although there is a significant amount of literature in the field, the reimbursement of amyloid PET has been met with reluctance in most countries. Reference Frisoni, Boccardi and Barkhof9 This is mainly linked to the lack of studies showing, beyond its analytical and clinical validity, its impact on patients’ management and care. With the first disease-modifying therapies using amyloid-antibodies having been FDA-approved Reference Boccalini, Peretti and Ribaldi55,56 , reimbursement status is gradually changing, as the proof of target expression may be required prior to initiation of an expensive – and not risk-free – antibody therapy. Currently, amyloid PET is reimbursed in Switzerland since August 2020, Reference Burge, Bieri and Bruhlmeier84 in France since August 2022, Reference Juengling, Allenbach and Bruehlmeier58 in the U.S since October 2023 Reference Garnier-Crussard and Flaus59 and in Japan since November 2023. 60 In Germany, amyloid PET is currently available to patients under an evidence development study protocol. 61
Large prospective studies focusing on the impact of amyloid PET images on patient management, Reference Teipel, Spottke and Boecker62,Reference Rabinovici, Gatsonis and Apgar63 were able to demonstrate that amyloid PET changes the care plan and medication prescription in over 60% of cases, both in MCI and in patients with dementia. Reference Teipel, Spottke and Boecker62
Tau PET
Finally, among the latest pathophysiological radiotracers developed for the diagnosis of AD are the tau radiotracers. Hyperphosphorylation and abnormal intracellular aggregation of tau proteins constitutes the second molecular marker of AD, in association with extracellular amyloid plaques. Tau targeting PET tracers have been developed mainly to selectively bind tau isoforms typical of AD pathology, i.e. neurofibrillary tangles. Tau PET images allow the visualization of the progressive accumulation of the radiotracer in different brain regions, in agreement with topographic patterns of Braak stages, as described from neuropathological studies.
This is a rapidly evolving field and one of the first-generation tracers tested, [18F]-Flortaucipir, has obtained approval for clinical use from the Food and Drug Administration in 2020. Reference Frisoni, Barkhof and Altomare64 These tracers have a great potential for staging and monitoring AD, but also for distinguishing AD versus non-AD pathologies, since they allow accurate measurement of the advanced neuropathological stages of AD that are almost invariably associated with the presence of amyloid plaques Reference Pascoal, Benedet and Tudorascu65 – an example of results of amyloid-PET and tau PET of the same patient are shown in Figure 3 and Figure 4. A strong clinical added value is expected based on the observation that tau PET combines the specificity for the disease process with the ability to stage it and thus provide relevant prognostic information, with the potential to usefully complement or even replace amyloid PET and [18F]FDG PET in specific indications. Reference Wolters, Dodich and Boccardi66
Many other first-generation and second-generation tracers have been or are being tested, all with a different degree of affinity for AD and non-AD tau pathology as well as different off-target binding profiles. Reference Villemagne, Barkhof, Garibotto, Landau, Nordberg and van Berckel10,Reference Fleisher, Pontecorvo and Devous67 While first-generation tracers demonstrated off-target binding in up to 10% of cognitively normal elderly, the second generation and third generation tracers have further improved on their specificity to diverse Tau fibril strains.
Several tau tracers are currently being used in clinical research studies at various Canadian universities, including the Universities of British Columbia, London, Montréal and Toronto. Reference Boccalini, Ribaldi and Hristovska68 However, none of these tracers has been authorized by Health Canada for routine clinical use as of yet.
In summary, while tau PET imaging holds promise for the diagnostic assessment of patients with cognitive impairment, its limitations in differentiating various neurodegenerative disorders and its utility in early disease stages should be taken into consideration. Potential off-target binding should always be kept in mind for clinical interpretation, which also will include assessment of further biomarkers, as negative results do not exclude other coexistent neurodegenerative disorders. Data to define their utility in differentiating non-AD tauopathies at an individual-patient level also is still limited.
PET imaging in diagnostic algorithms
As discussed above, multiple PET tracers can be proposed to refine the diagnosis of patients with cognitive impairment of a neurodegenerative etiology, and they all contribute with different and possibly complementary information. However, it is important to emphasize that diagnostic tools in clinical practice should be limited to those that are required to reach a diagnosis and favorably impact patient management.
Evidence-based recommendations in this sense are still lacking. Recently completed or ongoing prospective large scale studies, mainly in the field of amyloid imaging, will provide pivotal data to answer these questions. Reference Rabinovici, Gatsonis and Apgar63 The majority of the studies conducted so far focused on the diagnostic performance of individual tests, and only a few studies have focused on the incremental diagnostic value of additional tests, when the result of other investigations already is available. Reference Lemoine, Leuzy, Chiotis, Rodriguez-Vieitez and Nordberg69–71
The diagnostic algorithms currently available are mainly based on expert consensus, and overall converge in proposing a stepwise approach, in which morphological imaging, MRI when possible, represents the first-line investigation, and the test to propose as second line investigation depends on the clinical and neuropsychological profile and thus the prevalent etiological hypothesis. Reference Chetelat, Arbizu and Barthel6,Reference Altomare, Caprioglio and Assal72,Reference Ramusino, Garibotto and Bacchin73 The level of diagnostic confidence reached on the basis of the results of the first test will help in deciding if the investigations should stop or continue. In Canada, four Canadian Consensus Conferences on the Diagnosis and Treatment of Dementia have provided guidelines in general since 2012, Reference Massa, Farotti and Eusebi74,Reference Perini, Rodriguez-Vieitez, Kadir, Sala, Savitcheva and Nordberg75 with a separate consensus guidelines on the use of amyloid imaging in Canada issued 2016. Reference Boccardi, Nicolosi and Festari76 These guidelines also include dopaminergic imaging, an important tool to evaluate the integrity of the nigrostriatal system, typically involved in LBD. A detailed review on the respective dopaminergic imaging methods is given in the joint practice guideline by the European Association of Nuclear Medicine and the Society of Nuclear Medicine and Molecular Imaging issued in 2020. Reference Montero-Odasso, Pieruccini-Faria and Ismail77
PET imaging for dementia: availability and reimbursement conditions in Canada
Public provision and funding of PET imaging for non-cancer indications varies widely within provinces of Canada. A survey conducted by the McMaster Health Forum in 2018 Reference Ismail, Black and Camicioli78 has shown that comprehensive information about reimbursement conditions were difficult to access, not all data for all provinces were publicly available, or the publicly available information were out-of-date. In this survey, PET imaging for dementia was only listed in 5 out of 10 Canadian provinces among the procedures reimbursed by the health insurances in Canada (Table 1, modified from Reference Ismail, Black and Camicioli78 )
[18F]FDG as a radiopharmaceutical for PET imaging is widely available throughout the Canadian provinces, has received Health Canada approval, and is generally reimbursed for oncologic imaging, but not paid for neurological indications in a minority of provinces. The amyloid-tracer Florbetaben (NeuraCeq®) has received marketing authorization from Health Canada for the commercial production and market supply in March 2017, but is currently produced only in a single production site in Hamilton (ON), which reduces its availability due to the short physical half-life of its radioligand 18F of less than 2 hours, even when directly shipped by air courier. Availability could be improved if production were out-licensed to one or more adequately equipped radiopharmacies, as established in many PET-Centers countrywide.
In many countries with comparable Health Systems, including the United States, France, Germany, Switzerland and Japan, [18F]FDG PET as well as amyloid PET are available to be used for diagnostic algorithms in dementia as proposed 57,Reference Brisson, Brodeur and Letourneau-Guillon79–Reference Morbelli, Esposito and Arbizu81 . The current practice in Canada regarding availability and reimbursement allows for the use of [18F]FDG PET for the differential diagnosis of dementia in the majority of provinces. However, due to cost and availability, amyloid-PET is yet rarely used for this purpose.
Once better availability is provided, similar to the practice in the U.S., Europe and Japan, the use of amyloid-PET is expected to significantly increase in Canada, given its role in determining patient candidates for therapy modifying treatment. Currently, albeit being invasive in nature, cerebrospinal fluid markers including amyloid-beta, tau and p-tau are more used in tertiary care settings to rule in Alzheimer pathology. Novel blood markers, particularly those related to amyloid and tau are not currently in clinical practice in Canada.
Trends and future perspectives for PET molecular imaging in dementia
Three main current trends will impact the role of PET molecular imaging for cognitive disturbances in the near future:
-
the development of novel tracers,
-
the advent of blood plasma markers of pathology and
-
the advent of disease-modifying drugs for neurodegenerative diseases.
Development of novel PET tracers
The evolution of molecular imaging is fundamentally linked to the availability and validation of novel markers, allowing to extend the spectrum of imageable targets and to improve the sensitivity and specificity of the measure.
In the field of neurodegenerative disorders, the validation of novel tracers is of the utmost importance to image: (i) pathological deposits, such as alpha-synuclein, TDP-43 or tau isoforms typical for non-AD tauopathies; (ii) pathophysiological processes linked to the brain response to pathology, such as neuroinflammation, or synapses loss.
Alpha-synuclein PET imaging
There have been a number of efforts to develop a PET tracer specific for α-synuclein, Reference Mattison, Gauvin and Wilson82 with first-in-human scans just recently presented. Reference Boccardi, Nicolosi and Festari83 None of these are currently available in Canada. The relevance of α-synuclein PET imaging lies in its potential to serve as a key biomarker for the evaluation of Parkinson’s disease (PD) and related disorders, including dementia with Lewy bodies (DLB) and MSA. Alpha-synuclein is a critical protein associated with the pathogenesis of PD, and its aggregation is a hallmark of these neurodegenerative conditions. Additionally, α-synuclein PET imaging may offer insights into the progression of α-synuclein pathology in the brain, which is essential for monitoring disease advancement and assessing treatment efficacy.
The current evidence suggests that α-synuclein PET imaging is not specific for the diagnosis of LBD. However, it shows potential in distinguishing MSA from other neurodegenerative diseases. Reference Burge, Bieri and Bruhlmeier84
TDP-43 PET imaging
Concerted efforts have been made to develop a TDP-43 specific tracer, all of which being still in the preclinical phase, and are not yet available in Canada. TDP-43, short for Transactive response DNA binding protein 43 kDa, is a protein primarily localized in the cell nucleus that binds to nucleic acids. It plays a crucial role in alternative splicing and is encoded by the TDP-43 gene on Chromosome 1. TDP-43 is ubiquitously expressed and consists of 414 amino acids. Mutations in the TDP-43 gene can lead to amyotrophic lateral sclerosis (ALS), frontotemporal lobar degeneration (FTLD) or LATE dementia. In many ALS cases, post mortem brain tissue samples show TDP-43 positive cytoplasmic inclusions. TDP-43 is considered a molecular signature of ALS and FTLD. In the context of Alzheimer’s disease (AD), TDP-43 pathology can impact the pattern of hypometabolism detected by [18F]FDG PET imaging and is a relevant driver of volume-uptake mismatch in old AD patients. The development of TDP-43 PET tracers and the understanding of TDP-43 pathology’s impact on neuroimaging markers hold promise for enhancing the diagnostic evaluation and characterization of neurodegenerative dementias, including AD and related conditions. Reference Chételat, Desgranges, De La Sayette, Viader, Eustache and Baron85,Reference Korat, Bidesi and Bonanno86
Further physiopathological processes linked to neuroinflammation and neurodegeneration
A number of PET-Tracers have been developed to depict and quantify neuroinflammation, the majority of which targeting the translocator protein 18kDa (TSPO), shown to be overexpressed in activated microglia and astrocytes. Reference Smith, Capotosti and Schain87 Main limitation of this family of PET-Tracers, despite some improvement on 3rd generation TSPO tracers, is the genetic variability of the TSPO gene, particularly the rs6971 polymorphism, which leads to different binding affinities for TSPO ligands. This results in individuals being classified as high-affinity binders (HABs), low-affinity binders (LABs) or mixed-affinity binders (MABs). This heterogeneity can significantly impact the interpretation of PET imaging results, with findings in LABs being of disputable relevance, significantly impairing clinical study design, which might have to exclude individuals based on their genetic profile, makes quantitation less reliable without a prior genetic testing. [11C]PK11195, the prototypical first-generation PET radioligand, is the best characterized TSPO ligand, but has severe limitations, including low sensitivity and poor amenability to quantification. The short half-life of 20.4 min of 11C severely limits its accessibility outside of specialized centers equipped with a cyclotron and an experienced radiopharmacy. The substitution of 11C with the more common, longer-lived 18F (half-life 109.7 min), significantly affects lipophilicity, stability, specific binding and metabolic degradation, affecting brain penetration, specific binding and reliability of quantitation. Reference Smith, Capotosti and Schain88 A low radiochemical yield also limits the number of exams per production run of the radiotracer. The “ideal” TSPO-tracer has yet to be developed, and currently available tracers each have their specific limitations, which only partly can be overcome by prior genetic testing to improve interpretation of individual findings. This has, so far, limited their widespread use, but nevertheless TSPO-PET has proven to be of significant value in clinical trials designed around the tracer specifics. In Canada, TSPO-PET is currently available or in use for clinical trials at the Universities of Alberta, British Columbia, Montréal and Toronto. Reference Steinacker, Barschke and Otto89
Alternatives to TSPO tracers for imaging of neuroinflammation are PET tracers targeting:
-
the purinergic P2X7 ionotropic receptor (P2X7R), which is expressed in astrocytes, microglia and oligodendrocytes, where they mediate inflammasome signaling Reference Knight, Morrone, Varlow, Yu, McQuade and Vasdev90
-
the cyclooxygenase-enzyme-2 (COX-2), Reference Corica, De Feo and Gorica91–93 which has been implicated in neuroinflammation preceding neurodegeneration, Reference Franceschini, Capece and Chiozzi94 and could provide a rationale for stage-dependent antioxidative treatment
-
the C-X-C motif chemokine receptors (CXCR)-4 and (CXCR)-3, which critically contribute to the disease process in systemic inflammation, Reference Bhardwaj, Kaur, Wuest and Wuest95–Reference Juengling, Wuest and Kalra97 opening another door to possible future therapeutic principles targeting the respective C-X-C motif chemokine receptor Reference Bhardwaj, Kaur, Wuest and Wuest95
-
the Glucose Transporter 5 (GLUT-5), which is exclusively expressed in microglial cells, Reference Yiangou, Facer and Durrenberger98 with 18-Fluoro-D-Fructose being the most extensively preclinically characterized compound. Reference Kircher, Herhaus and Schottelius99–Reference Chu, Shields, Zhang, Feng, Shields and Cai101 Further to providing information on neuroinflammation, the involvement of dietary fructose and cerebral fructose metabolism GLUT-5 in patients with concurrent diabetes has attracted interest. Reference Payne, Maher, Simpson, Mattice and Davies102
As a novel measure for neurodegeneration and neurorestoration, tyrosine kinase receptors as the binding targets of neurotrophins have attracted considerable interest regarding their diagnostic and therapeutic potential. Within this class of receptors, synthetic antibodies have recently been linked to PET-radioligands targeting the TrkB receptor, activated by brain derived neurotrophic factor (BDNF), which have passed the preclinical and clinical assessments. Reference Boyle, Murrell and Tong103–Reference Wuest, Trayner and Grant105 While this research still is in the experimental phase, the recent development of TrkB agonistic antibodies and BDNF-targeted gene therapies Reference Johnson, Gomez-Pinilla and Nagel106,Reference Bailey, Kaiser and Lindner107 could prove useful as an alternative line of therapy to amyloid modifying treatments, and changes in TrkB alterations as measured by PET/MRI during targeted therapy could potentially qualify as an imaging endpoint in clinical trials in AD. In Canada, TrkB PET using the novel tracer [18F]-TRACK is currently available or in use for clinical trials at the Universities of Alberta and Montréal. Reference Bernard-Gauthier, Mossine and Mahringer108
Markers of pathology in plasma
A major development of the last years has been the advent of highly sensitive assays able to accurately measure pathological markers in plasma. Reference Wuest, Bailey and Dufour109 The availability of these assays, representing a simple blood test versus the more invasive CSF test requiring a spinal tap, opens new perspectives for large scale population-level screening with a noninvasive and affordable test. Preliminary data suggests feasibility of diagnostic algorithms based on the results of plasmatic markers as first-line test, followed by further diagnostic modalities including molecular imaging, following an “integral diagnostic,” or precision medicine approach. Reference Tuszynski, Yang and Barba110 However, until more data is available, careful interpretation of these biomarkers is still required, as a recent study found that plasma-Tau abnormalities were rather associated with cerebral amyloid-beta deposition than with cerebral tau deposition measured by PET. Reference Osborne, Wang, Tassoni, Widdowson and Martin111
Disease-modifying drugs for neurodegenerative diseases
The research to develop pharmacological and non-pharmacological approaches to treat neurodegenerative conditions is actively testing a large panel of compounds and strategies to deal with these devastating conditions, namely targeting the most common condition, AD. 112
Encouraging although strongly debated results have been obtained by two antibodies targeting amyloid which have shown biological changes, namely using amyloid PET as treatment biomarker. Reference Kuller and Lopez4,Reference Simren, Leuzy and Karikari113,Reference Garibotto, Boccardi, Chiti and Frisoni114 Given the high costs for these treatments, and potential severe side-effects, Reference Therriault, Vermeiren and Servaes115,Reference Cummings, Lee, Ritter, Sabbagh and Zhong116 it seems straightforward to require a positive amyloid PET scan as proof of target engagement prior to therapy.
Molecular imaging markers are surrogate markers now consistently integrated in clinical trials, and clearly the advent of current and future disease-modifying drugs will profoundly change the role and the implications of diagnostic processes in AD and related disorders. PET imaging biomarkers have proven to provide a framework for PET-based Braak staging of AD to monitor AD severity in living humans, Reference Mintun, Lo and Duggan Evans117 which will provide further rationale for a patient-adapted approach on therapeutic interventions, as a further example of precision medicine.
Conclusions
In conclusion, there is a clinical need for an accurate diagnosis of cognitive disorders using biologic criteria, Reference Knopman118 both for the adaptation of the management of these patients and for the implementation and development of new treatments. PET represents already a mainstay of the etiological diagnostic process, as a key element to approach an anatomo-pathological diagnosis in vivo, and is susceptible to play an increasingly important role. [18F]FDG PET still is the worldwide most frequently used PET imaging agent in dementia, and is broadly available and in clinical use in Canada. Amyloid PET has been extensively investigated in clinical trials in Europe, Switzerland, Japan and the U.S., and is increasingly reimbursed for the use in dementia as amyloid-targeted treatment continues to expand. A lack of reimbursement and limited availability limit the clinical use of amyloid PET in Canada, which consequently is rarely used clinically.
From the author’s perspective, in the light of extraordinarily high costs of beta-amyloid altering therapy, and its potentially life-threatening side-effects, beta-amyloid altering therapy should not be administered without prior proof of target engagement, which ideally would include PET imaging and/or other biomarkers relevant to the case, including CSF examination or blood biomarkers. Should Canada secure reimbursement for amyloid PET, aligning with the practice in the U.S., both the industry and local radiopharmacies in PET-centers with cyclotron availability, stand ready to scale up production and distribution. Read-out of amyloid PET has been highly standardized, and diagnostic scales, such as the Centiloid Scale can be adapted with little local effort or industry support, similar to prior practice in various fields of PET imaging.
Regarding current and future developments including novel tracers for dementia and neurodegenerative disease, Canada has been engaging for more than a decade in research initiatives at the highest, international level, with a number of unique tracers being created and evaluated in Canadian university labs and clinics. However, the country’s vast geography and uneven population distribution create significant challenges in making these advancements accessible to all Canadians in need. Overcoming this accessibility gap will require a collaborative effort that combines academic ingenuity, renowned logistical expertise and industry support. This approach has already demonstrated success with the introduction of [18F]FDG PET in oncologic imaging in Canada during the past decade.
Author contribution
Conception and manuscript writing: FJ, VG. Specific contributions: FW, RS, JA, JS, AT, RC. Correction, revising, final approval: FJ, FW, RS, JA, AT, JS, RC, VG. Revisions and proofreading: FJ.
Funding statement
None.
Competing interests
JA declares to have received a research grant from the Canadian Neuroendocrine Tumor Society. JS declares to have received payments for PET studies interpretation from Optina Diagnostics, as well as payment or honoraria from the Royal College of Physicians and Surgeons of Canada. RC is team lead of the CIHR – Canadian Consortium on Neurodegeneration of Aging, is site-PI/site investigator of the NIH SPARX3 clinical trial, CIHR Compass-ND, the Weston Foundation Synergjc2 clinical trial and has received funding through a subcontract with the University of Calgary from CIHR/Brain Canada for functional assessment of vascular reactivity. RC also is unpaid scientific advisor for the Weston Foundation, Western University on Ambroxol for Parkinson’s Disease Dementia, is unpaid member of the scientific board of the Parkinson Society of Canada, as well as an unpaid member of the Michael J Fox Gait Advisory Board. VC has received research funding to her institution from Siemens Healthineers and GE Healthcare, and has received speaker fees from GE Healthcare, Siemens Healthineers, Novo Nordisk and Janssen. All other authors declare that the review was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.