Pregnancy is a period of rapidly increasing physiological demands, where optimal nutrition is crucial to support both maternal and fetal growth(Reference Stephenson, Heslehurst and Hall1). Substantial evidence suggests that the nutritional status of the mother and, therefore, the intra-uterine environment is a critical determinant of developmental plasticity, having significant implications for the development of chronic disease in later life(Reference Barker2,Reference Almond and Currie3) . Birth weight serves as an indicator of infant intra-uterine growth with epidemiological data demonstrating associations between abnormal birth weights and the development of several chronic diseases throughout the lifespan(Reference Min, Wang and Sheng4–Reference Raisi-Estabragh, Cooper and Bethell6). Postnatally, the increase in the number of adipocytes is highest during early life; however, adipose tissue accretion begins in utero, and thus pregnancy may be a sensitive period for influencing the trajectory of weight throughout childhood(Reference Orsso, Colin-Ramirez, Field and Madsen7,Reference Palacios-Marin, Serra, Jimenez-Chillarion and Herrero8) .
Dietary components that have gained considerable attention for their roles in pregnancy and fetal development are the long-chain n-3 PUFA, EPA (20:5n-3) and DHA (22:6n-3). The n-3 PUFA are traditionally known for their anti-inflammatory properties and role in neurodevelopment. However, they have also been shown to be associated with greater birth weight, lower risk of pregnancy complications and a lower risk of preterm delivery(Reference Meher, Randhir, Mehendale and Wagh9–Reference Abdelraham, Osama and Saeed11). Additionally, it has been proposed that the n-3 PUFA may have the ability to influence child weight throughout early life(Reference Bergmann, Bergmann and Haschke-Becher12,Reference Firouzabadi, Shab-Bidar and Jayedi13) . Evidence from animal models appears to suggest that prenatal exposure to arachidonic acid (AA; 20:4n-6), an n-6 PUFA, promotes adipogenesis via the production of biologically active lipid metabolites, also known as eicosanoids. These act in a pro-inflammatory manner stimulating the proliferation of adipocytes. In utero exposure to n-3 PUFA appears to counteract this effect through the production of more anti-adipogenic lipid mediators(Reference Fleckenstein-Elsern, Dinnies and Jelenik14,Reference Hao, Chen and Nie15) .
Despite the postulated biological mechanism for the prenatal exposure of n-3 and n-6 PUFA in influencing child weight, data from human studies have shown conflicting results. Findings from the Growing Up in Singapore Towards healthy Outcomes (GUSTO) cohort showed positive associations between maternal plasma linoleic acid (LA; 18:2n-6) concentrations and birth weight and abdominal adipose tissue volume within the child(Reference Bernard, Tint and Aris16). In contrast, a Chinese birth cohort reported higher maternal erythrocyte AA and total n-6 PUFA to be associated with a lower risk of overweight and obesity at 2 years(Reference Wu, Zhao and He17). Studies investigating the n-3 PUFA have also yielded inconsistent findings, with some reporting negative associations with weight throughout childhood(Reference Al-Hinai, Baylin and Tellez-Rojo18) and others observing positive or null associations(Reference Donahue, Rifas-Shiman and Gold19,Reference Pereira-da-Silva, Cabo and Moreira20) .
During pregnancy, the developing fetus is entirely dependent on the maternal supply of PUFA, with circulating concentrations known to fluctuate throughout gestation(Reference Larque, Pagan and Prieto21). Fish is one of the richest sources of n-3 PUFA within the diet and is known to contain several other nutrients essential for fetal growth, including vitamin D, Se and protein(Reference Mendivil22). Generally, regulatory agencies advise the consumption of 2–3 portions of fish (4 oz) per week during pregnancy on the basis that fish high in methylmercury, an environmental neurotoxicant, are avoided(23,24) . Studies examining the associations between maternal fish consumption and birth weight remain inconsistent with some reporting favourable fetal growth amongst women who consume seafood during pregnancy, whilst others have reported negative or null associations(Reference Leventakou, Roumeliotaki and Martinez25,Reference Wei, Li and Lei26) . Notably, fewer studies have assessed the associations between maternal fish consumption during pregnancy and offspring weight throughout childhood. Van den Berg et al. (2016) analysed mother–child data from the Dutch Prevention and Incidence of Asthma and Mite Allergy (PIAMA) cohort and found no evidence of an association between maternal fish consumption and BMI throughout childhood(27); however, this was a cohort of relatively low fish consumers, and further research in large population-based cohorts with a wide range in maternal fish consumption is warranted.
Given the ambiguity surrounding the roles of maternal PUFA status on child weight and the paucity of data on the effects of maternal fish consumption, the primary aims of this study were to examine the associations between maternal fish consumption and prenatal PUFA status (n-3 and n-6), including maternal and umbilical cord blood concentrations, with child weight at birth and throughout childhood (20 months, 7 and 13 years) in the high fish-eating Seychelles Child Development Study (SCDS) Nutrition Cohort 2 (NC2). It was hypothesised that higher intakes of fish, and thus higher concentrations of the n-3 PUFA, would be associated with a lower risk of overweight/obesity throughout childhood, whilst higher prenatal exposures to the n-6 PUFA would be associated with increased child adiposity.
Methods
Study population
The SCDS is an ongoing multicohort observational study with the overall aim of investigating the risks and benefits of prenatal fish consumption on child neurodevelopment. Mothers were enrolled onto NC2 during their first antenatal visit (from 14 weeks of gestation) between 2008 and 2011. Recruitment took place at eight health centres on Mahè, the main island of the Republic of Seychelles. Inclusion criteria included being native Seychellois, ≥ 16 years of age, having a singleton pregnancy and having no obvious health concerns(Reference Strain, Yeates and van Wijngaarden28).
Ethical approval
This study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human participants were approved by the Seychelles Ethics Board and the Research Subjects Review Board at the University of Rochester. Written informed consent was obtained from all participants.
Fish consumption
Habitual fish consumption throughout pregnancy was assessed retrospectively through a Fish Use Questionnaire (FUQ) administered at 28 weeks’ gestation. The FUQ was designed specifically for the Seychellois population and focused on the fish species available in the Seychelles. Participants were asked to report the number of times per week during pregnancy they consumed specific types of fish with frequency of consumption reported as meals per week. The reported weekly consumption of fishmeals was then converted to grams per d using the average portion size for each fish species and multiplying it by the frequency of consumption per week and dividing by 7 to estimate daily intake of fish in g/d(Reference Wesolowska, Yeates and McSorley29). Fish consumption data were further categorised into oily fish, lean fish and total fish intakes(Reference Conway, Mulhern and McSorley30). The FUQ was also completed at the 7- and 13-year time points to assess habitual fish consumption throughout childhood and adolescence. The FUQ has been compared against a 4-d semi-quantitative food diary using Bland–Altman plots and cross-validation to compare mean differences in fish consumption reported from each assessment tool. This analysis showed good concurrence between the questionnaire and diary with most data well within the 95 % limits of agreement, which indicates that the FUQ is a valid tool for measuring fish consumption in this population (data unpublished).
Blood sampling and PUFA analysis
Maternal non-fasting blood samples were collected at 28 weeks’ gestation, and cord blood samples were taken from the umbilical cord vein at delivery, from which serum was obtained. Maternal and cord samples were shipped at −80 °C to Ulster University for serum total PUFA analysis according to an adaptation of the Folch et al.(Reference Folch, Lees and Sloane Stanley31) method as previously described(Reference Strain, Yeates and van Wijngaarden28). Briefly, fatty acid methyl esters were quantified using GC tandem MS (7890A–5975C; Agilent), with reference to heptadecanoic acid (C17:0) as an internal standard. Individual PUFA measured within the analysis included LA (18:2n-6), α-linolenic acid (ALA; 18:3n-3), AA (20:4n-6), EPA (20:5n-3) and DHA (22:6n-3) with results presented as milligrams per millilitre. ALA, EPA and DHA were summed to calculate the total n-3 PUFA, and total n-6 PUFA were calculated by the addition of LA and AA concentrations.
Child weight
Birth weight (kilograms) was assessed at delivery by trained nurses and recorded to the nearest 2 decimal places using routine clinical procedures and standardised scales. Percentiles of birth weight according to gestational age were calculated based on the WHO Child Growth Standards(32). Appropriate fetal growth for gestational age (AGA) was defined as a birth weight between the 10th and 90th percentiles. Children born with a birth weight <10th percentile for gestational age were classified as small for gestational age (SGA) and those born with a birth weight >90th percentile were deemed large for gestational age (LGA).
Child length/height (metres) and weight (kg) measurements were recorded by trained nurses at the 20-month, 7-year and 13-year follow-ups. Child z-scores were calculated using the WHO AnthroPlus software, and children were classified according to BMI z-scores. Children with a BMI z-score < –2·0 were classified as underweight and those with a BMI z-score >1·1 were deemed as overweight/obese. Normal weight throughout childhood was defined as a BMI z-score between −2·1 and 1·0(Reference De Onis and Lobstein33). All anthropometric measurements were recorded using standardised procedures and equipment calibrated by the Seychelles Bureau of Standards prior to and throughout the duration of the study.
Covariates
Covariates were selected a priori based on the current literature and included maternal age, maternal BMI, socio-economic status, gestational age, child sex, parity (number of children) and alcohol use during pregnancy(Reference Donahue, Rifas-Shiman and Gold19,Reference Van Wijngaarden, Harrington and Kobrosly34–Reference Stratakis, Roumeliotaki and Oken36) . Child fish intakes were also considered as a covariate at 7 and 13 years. Owing to the low prevalence of smoking within the cohort (n 9), this was not adjusted for within the analysis. A questionnaire administered by trained nurses upon enrolment to the study was used to collect information on maternal age, parity and alcohol use throughout pregnancy (yes/no). Gestational age (weeks) and child sex were recorded at birth. At the 20-month follow-up, maternal socio-economic status was measured using the Hollingshead Social Status Index, modified for use within the Republic of Seychelles(Reference Davidson, Myers and Cox37). Data on prenatal BMI were unavailable within this cohort, and therefore, based on findings from a previous cohort that found a high correlation between early pregnancy BMI and postnatal BMI(Reference Davidson, Strain and Myers38); postnatal BMI was used as a surrogate for pre-pregnancy BMI. At the infants’ 20-month examination, maternal height and weight were recorded from which postnatal BMI was calculated (BMI = weight (kg)/height (m)2).
Statistical analysis
A total of 1535 mothers were initially recruited onto the NC2 study. Mothers with extreme gestational age (preterm birth defined as <34 weeks gestation) and children with very low birth weight (<1500 g) were excluded from the current analysis (n 17). A total of 1185 mothers had PUFA concentrations measured at 28 weeks’ gestation and had available data on all covariates (Figure 1). Of the 1185 mothers included within the current analysis, 1124 had complete data on maternal fish intakes and 932 had complete PUFA concentrations measured within cord blood.

Figure 1. Description of exclusions and missing data for the current analysis of the Seychelles Child Development Study Nutrition Cohort 2.
Statistical analysis was conducted using the Statistical Package for Social Sciences (SPSS, version 29.0, IBM). Data for all variables were tested for normality. Descriptive statistics were used to summarise the distribution of maternal and child characteristics. Associations between maternal fish consumption and maternal and cord PUFA concentrations and birth weight defined as SGA, AGA and LGA were assessed using multinominal regression analysis. Owing to the small number of underweight children with this cohort, BMI classifications throughout childhood were dichotomised (BMI z-score <2·0 or BMI z-score >2·0), and associations between the primary exposures and overweight/obesity (BMI z-score >2·0) at 20 months, 7 years and 13 years were examined using binary logistic regression models. Separate models were fitted using maternal fish consumption and maternal and cord PUFA concentrations as categorical variables with exposures grouped into quartiles based on sample distribution to aid in interpretability. All models were adjusted for maternal age, maternal BMI, gestational age, socio-economic status, child sex, alcohol use during pregnancy and parity. Models at 20 months, 7 years and 13 years were additionally adjusted for child fish intake at that time point. A p-value <0·05 was considered statistically significant throughout.
Results
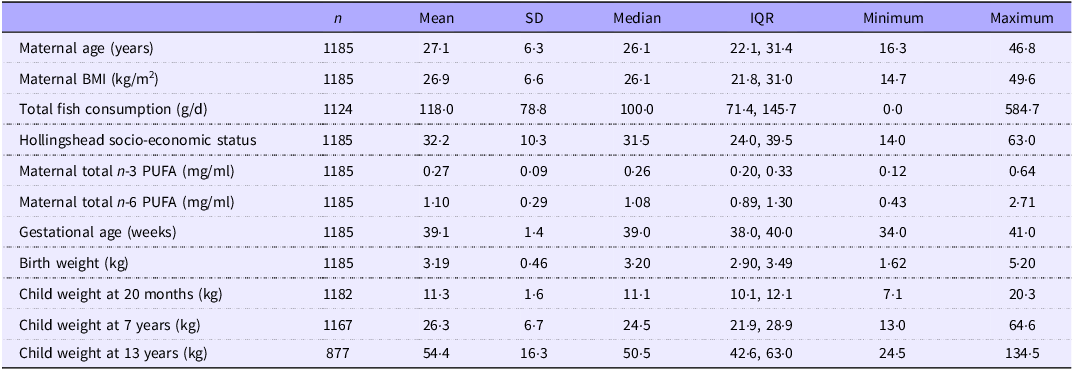
Table 1 displays the descriptive characteristics of the 1185 mother–child dyads included within the current analysis. Mothers had a mean age of 27·1 ± 6·3 years and were consuming on average 118·0 ± 78·8 g/d of fish throughout pregnancy (range: 0·0–584·7 g/d). Children comprised of 617 males and 568 females and had an average birth weight of 3·19 ± 0·46 kg. There were 209 (17·6 %) children classed as SGA, and only 38 (3·2 %) children were considered LGA. The mean birth weight of those classed as SGA and LGA was 2·62 ± 0·39 kg and 4·08 ± 0·42 kg, respectively. By 13 years, 305 (25·7 %) children were considered as overweight/obese. Maternal and cord PUFA concentrations are shown in Supplementary Table 1.
Table 1. Maternal and child characteristics of study population (n = 1185)

.IQR, interquartile range..
Maternal fish consumption
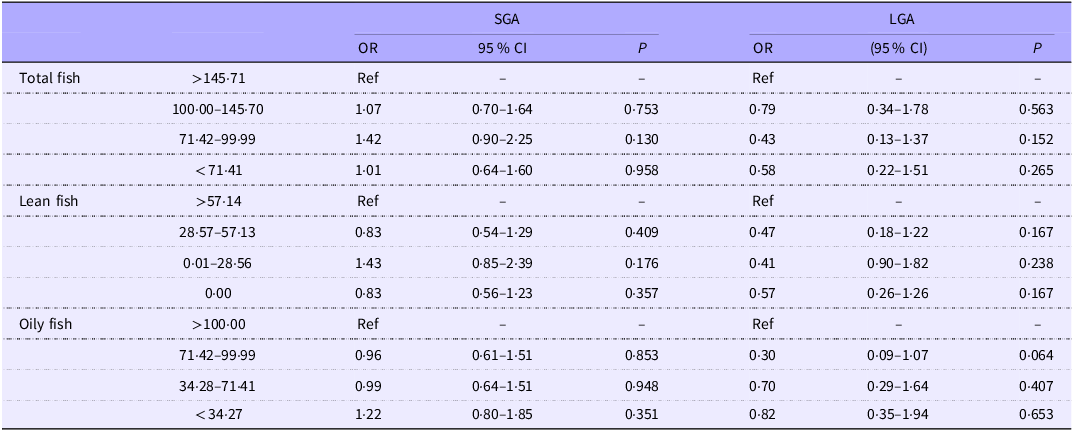
Covariate-adjusted associations between maternal fish intakes and birth weight are presented in Table 2. There were no significant associations between maternal total fish, oily fish or lean fish intakes and SGA or LGA within the child.
Table 2. Associations between maternal fish intakes and birth weight

SGA, small for gestational age; LGA, large for gestational age.
Adjusted for maternal age, maternal BMI, Hollingshead socio-economic status, gestational age, child sex, parity and alcohol use.
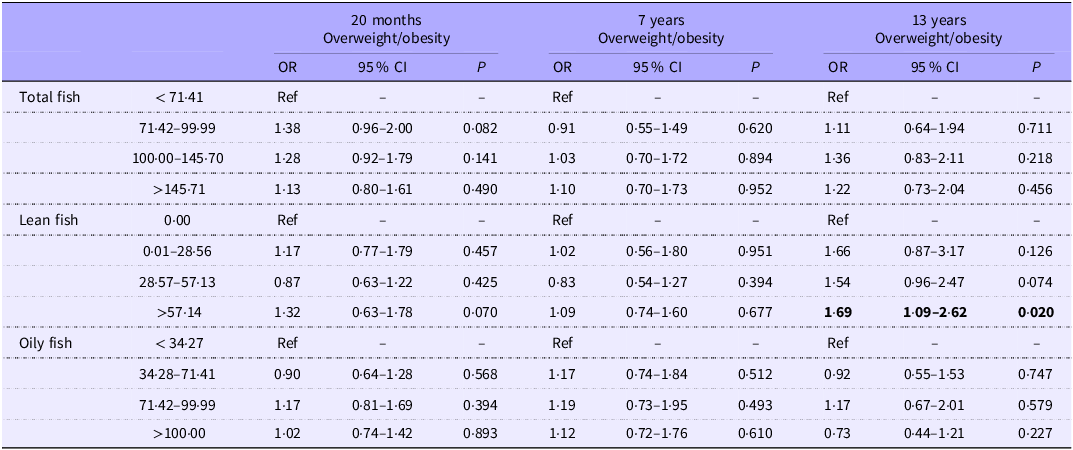
There were also no significant associations between maternal total or oily fish intakes and child weight at 20 months, 7 years or 13 years (Table 3). There was, however, a significantly higher risk of overweight/obesity at 13 years amongst those consuming >57·14 g/d lean fish when compared with those in the lowest quartile of lean fish consumption (0·00 g/d) (OR 1·69, 95 % CI 1·09–2·62, p = 0·020), albeit this did not follow a dose–response trend with the second and third quartiles of lean fish consumption showing no significant association with overweight/obesity at 13 years.
Table 3. Associations between maternal fish intakes (g/d) and risk of overweight/obesity throughout childhood

Adjusted for maternal age, maternal BMI, Hollingshead socio-economic status, gestational age, child sex, parity and alcohol. Models at 7 and 13 years additionally adjusted for child fish intakes.
Maternal PUFA
Multinomial logistic regression analyses for covariate-adjusted associations between maternal PUFA concentrations and birth weight are presented in Supplementary Table 2. Only maternal AA concentrations in the lowest quartile (<0·148 mg/ml) were associated with a significantly lower risk of LGA (OR 0·26, 95 % CI 0·07–0·94, p = 0·041) when compared with the highest quartile (>0·261 mg/ml). No significant associations were found between any of the other maternal PUFA and LGA. No significant associations were noted between maternal concentrations of n-3 or n-6 PUFA and SGA.
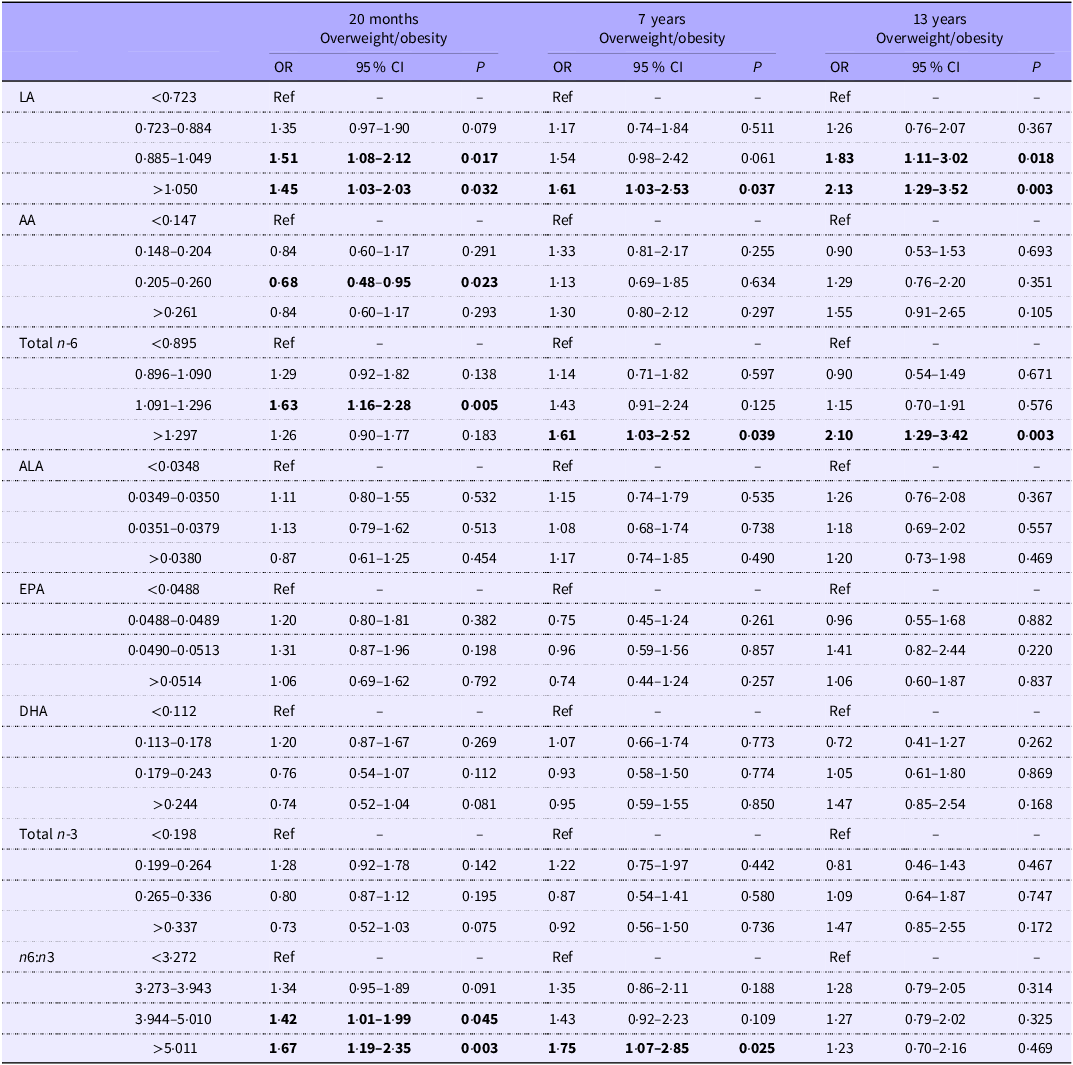
Table 4 displays associations between maternal PUFA status and overweight/obesity at 20 months, 7 years and 13 years. At 20 months, children in the highest quartile of maternal LA concentrations (>1·050 mg/ml) had a significantly higher risk of childhood overweight/obesity when compared with those in the lowest quartile of maternal LA concentrations (<0·723 mg/ml) (OR: 1·45, 95 % CI 1·03–2·03, p = 0·032). Similar associations were found at 7 and 13 years, with the highest quartile of maternal LA and maternal total n-6 PUFA being associated with the highest risk of childhood overweight/obesity. Although there were no significant associations between any of the maternal n-3 PUFA and being overweight/obese throughout childhood, a higher maternal n6:n3 ratio (>5·011) was associated with a higher risk of being overweight/obese at both 20 months (OR: 1·67, 95 % CI 1·19–2·35, p = 0·003) and 7 years (OR: 1·75, 95 % CI 1·07–2·85, p = 0·025). There were no significant associations between the maternal n6:n3 ratio and being overweight/obese at 13 years.
Table 4. Associations between maternal PUFA (mg/ml) and risk of overweight/obesity throughout childhood

LA, linoleic acid; AA, arachidonic acid; ALA, alpha-linolenic acid.
Adjusted for maternal age, maternal BMI, Hollingshead socio-economic status, gestational age, child sex, parity, alcohol. Models at 7 and 13 years additionally adjusted for child fish intakes….
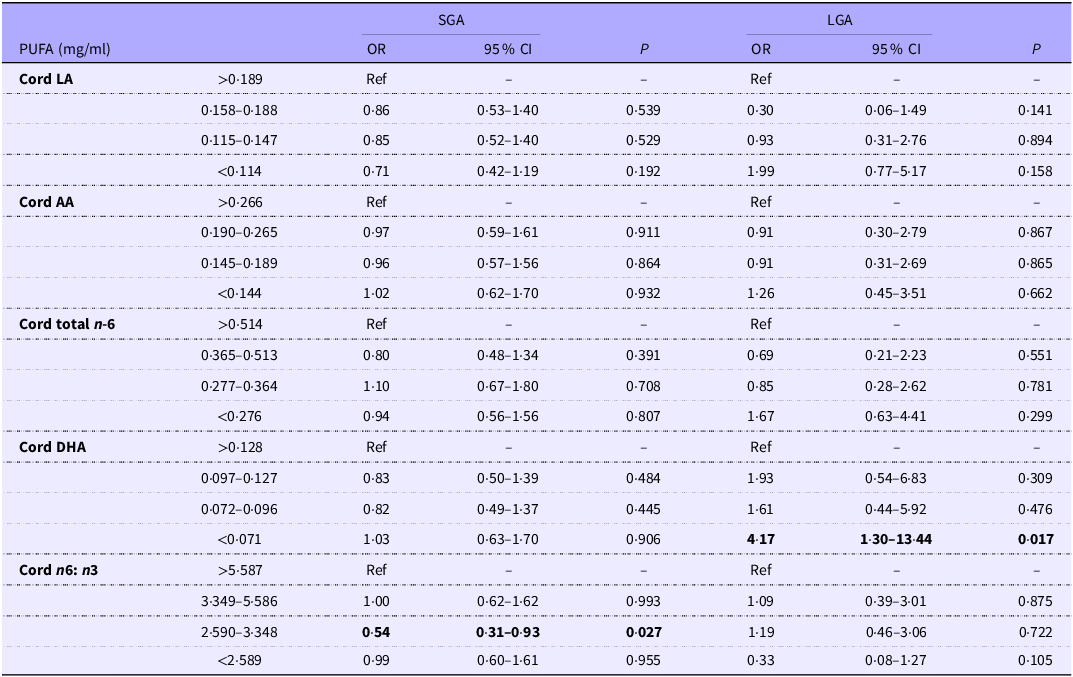
Cord PUFA
Table 5 outlines associations between cord PUFA concentrations and childbirth weight. When compared with the highest cord DHA quartile (>0·129 mg/ml), only cord DHA concentrations <0·071 mg/ml were found to be associated with a significantly higher risk of LGA (OR 4·17, 95 % CI 1·30–13·44, p = 0·017). No other significant associations between any of the cord n-6 PUFA or the cord n6:n3 ratio and LGA were found. No significant associations were observed between any of the cord PUFA and SGA, albeit the third quartile of the cord n6:n3 ratio was associated with a lower risk of SGA (OR 0·54, 95 % CI 0·31–0·93, p = 0·027) when compared with the highest quartile; however, this may be considered a spurious finding given the lack of any dose–response.
Table 5. Associations between cord PUFA (mg/ml) and birth weight

SGA, small for gestational age; LGA, large for gestational age; LA, linoleic acid; AA, arachidonic acid.
Adjusted for maternal age, maternal BMI, Hollingshead socio-economic status, gestational age, child sex, parity and alcohol use.
Associations between cord PUFA and child weight at 20 months, 7 years and 13 years are displayed in Supplementary Table 3. There were no significant associations between any of the cord n-3 or n-6 PUFA and overweight/obesity throughout childhood.
Discussion
In this high fish-eating cohort from the SCDS, total fish consumption throughout pregnancy was not associated with repeated measures of child weight at birth, 20 months, 7 years or 13 years. There were, however, several associations between the n-3 and n-6 PUFA and child weight. The lowest quartile of maternal AA concentrations (<0·148 mg/ml) was associated with a lower risk of LGA. Furthermore, higher maternal total n-6 PUFA concentrations were associated with an increased risk of overweight/obesity at 7 years and 13 years. In cord blood, DHA concentrations <0·071 mg/ml were associated with a significantly higher risk of LGA within the child. None of the observed associations between the cord n-3 or n-6 PUFA and child weight were evident at 7 or 13 years of age. Overall, these findings appear to suggest differing influences of lower cord n-3 and higher maternal n-6 PUFA status on child adiposity at birth until adolescence.
In the current study, there was no clear evidence of any associations between maternal total fish consumption and child weight at birth or throughout childhood, despite the uniquely high fish intakes within this cohort. Observational studies investigating the associations between maternal fish consumption and measures of child growth are few and have been overall inconclusive. Findings from this study appear to be consistent with a number of other mother–child cohorts, albeit lower fish consumers, where no associations were found(27,Reference Guldner, Monfort and Rouget39,Reference Mohanty, Thompson and Burbacher40) . In contrast, one larger Norwegian Mother and Child Cohort Study with a much lower average fish intake of 36 g per d reported positive associations between maternal seafood consumption and birth weight(Reference Brantsaeter, Birgisdottir and Meltzer41). Furthermore, one pooled analysis of fifteen European and USA birth cohorts reported higher fish intakes (>3 times/week) to be associated with an increased risk of rapid infant growth and child overweight/obesity at 4 and 6 years which may be owing to contaminants found within fish(Reference Stratakis, Roumeliotaki and Oken36). We found that the consumption of >57·14 g/d lean fish was associated with an increased risk of overweight/obesity at 13 years; however, this association was not observed at any other time point, nor was it dose-responsive, and thus may be a chance finding. Studies which have reported a positive association between maternal fish intakes and child weight have noted that these associations appear to be dependent on the type of fish, with nutrients and contaminants within fish varying depending on the species and source which may also account for some variation between populations in addition to the differences in consumption patterns(Reference Stratakis, Roumeliotaki and Oken36,Reference Willer, Newton and Malcorps42) . Nevertheless, fish is an important source of protein and nutrients required for growth and development both in pregnancy and childhood(Reference Mendivil22), and uncertainty surrounding the benefits and/or risks of fish consumption during pregnancy on child body composition may lead to transitions towards more westernized-style diets which have been associated with metabolic complications including CVD(Reference Clemente-Suarewz, Beltran-Velasco and Redondo-Florez43). This transition is particularly relevant amongst the Global South, with developing nations such as the Seychelles undergoing a period of rapid dietary transition resulting in lower fish intakes(Reference Cardoso, Bovet, Viswanathan and Luke44). Given the limited and inconsistent data, alongside the public health implications, future longitudinal analyses using repeat measurements throughout childhood and which utilise direct measures of body composition are warranted to elucidate the relationships between maternal fish consumption during pregnancy and child adiposity.
Previous research has emphasised the importance of n-3 PUFA, primarily obtained in the diet from fish consumption, during pregnancy in relation to fetal growth, brain structure and neurodevelopment(Reference Makrides45,Reference Olsen, Halldorsson and Thorne-Lyman46) . There was no evidence of any associations between the maternal n-3 PUFA and child weight at birth, 20 months, 7 years or 13 years, which confirms results from our previous analysis from the SCDS, where we did not detect any associations between maternal n-3 PUFA and birth outcomes(Reference Van Wijngaarden, Harrington and Kobrosly34,Reference Yeates, Zavez and Thurston35) . The existing literature with regard to the associations between maternal n-3 PUFA status and child adiposity remains controversial and has primarily focused on early childhood which makes comparison to the current study challenging. Findings from the much larger Generation R Study noted higher maternal total n-3 PUFA concentrations within the second trimester were associated with a lower childhood body fat percentage and a lower android:gynoid fat mass ratio at 6 years(Reference Vidakovic, Gishti and Voortman47). Others have reported no associations between maternal n-3 PUFA concentrations and child fat mass at 6 years(Reference Moon, Harvey and Robinson48). It is noteworthy that the two latter studies measured PUFA concentrations in plasma phospholipids, whereas in the current study maternal PUFA were measured in serum which makes comparisons more difficult. Moreover, 13 years may be a period of rapid pubertal growth where changes in the hormonal milieu result in adiposity accretion(Reference Solorzano and McCartney49). In this cohort, maternal fish consumption was not correlated with maternal PUFA status, which is also likely reflective of other factors influencing biomarker status. Aside from the differences in n-3 PUFA intakes, genetic variations can also influence PUFA metabolism and may also explain some of the inconsistencies across studies(Reference Yeates, Love and Engström50). Future studies should consider the various genotypes which regulate PUFA metabolism when investigating the associations between maternal n-3 PUFA and child adiposity.
Within this cohort, maternal AA concentrations <0·148 mg/ml were associated with a lower risk of LGA at birth when compared with higher maternal AA concentrations. Additionally, the maternal n-6 PUFA were consistently associated with an increased risk of overweight/obesity throughout childhood. The current results are broadly consistent with the limited previous human studies(Reference Vidakovic, Gishti and Voortman47,Reference Moon, Harvey and Robinson48,Reference De Vries, Gielen and Rizopoulos51) . The Southampton Women’s Survey reported positive associations between maternal plasma n-6 PUFA concentrations and child fat mass at 4 and 6 years(Reference Moon, Harvey and Robinson48). Similar findings were also reported in the Generation R Study where higher maternal plasma n-6 PUFA were found to be associated with a higher childhood total body fat percentage and abdominal preperitoneal fat mass area(Reference Vidakovic, Gishti and Voortman47). Taken together, these findings, along with the current study, appear to suggest that higher prenatal exposures to the n-6 PUFA may influence adipogenesis development in utero and may be associated with an increase in child adiposity until early adolescence.
There are several existing mechanisms which may help to explain these observations. The n-6 PUFA are known to elicit a more pro-inflammatory immune response, leading to the production of pro-inflammatory lipid mediators known as eicosanoids(Reference Calder52). These eicosanoids are highly biologically active, exerting effects on many cell types via cell membrane G-protein-coupled receptors(Reference Tang, Hou, Schwartz and Haeggstrom53). For example, the n-6 PUFA are known to act as a precursor for prostacyclin (PG I2) which has been shown to bind to PPARγ which regulates adipogenesis via the proliferation of preadipocytes into mature adipocytes(Reference Majed and Khalil54,Reference Rahaman55) . Moreover, there is also some evidence from murine models which suggests differing effects of fatty acids in the metabolic programming of the fetus in utero through epigenetic modification such as DNA methylation. Such epigenetic changes may influence growth in later life by influencing appetite control and energy balance(Reference Kabaran and Besler56) and, thereby, may offer some plausible biological basis as to why associations were not apparent until later in childhood, albeit further work is needed to confirm this. We also noted an increased risk of overweight/obesity at 20 months and 7 years with a higher maternal n6:n3 ratio, which is generally associated with a greater pro-inflammatory immune response. The observed associations between the n-6 PUFA and child adiposity during childhood likely reflect the influence of the n-6 PUFA on the inflammatory milieu throughout pregnancy, and future work may need to measure eicosanoid production to improve mechanistic understanding in this area.
Typically, LA is abundant within the diet and found within several sources, including vegetable oils, nuts and seeds(Reference Orsavova, Misurcova and Ambrozova57). Endogenously, LA can undergo the elongation and desaturation pathway to the more biologically active n-6 PUFA, although AA is also found in high amounts in red meat from grain-fed animals(Reference Mariamenatu and Abdu58). Westernised-style diets are high in processed foods, red meat and eggs, which are typically higher in n-6 PUFA(Reference Liput, Lepczyński and Ogłuszka59). Previous research from our group has shown that this style of dietary pattern is most typically followed within this cohort of pregnant Seychellois women, reflecting the transition from the traditional Seychellois diet to a more westernized-style diet within the Republic of Seychelles(Reference Conway, Mulhern and McSorley30). The westernised dietary pattern has also been shown to be associated with an enhanced pro-inflammatory immune response and may be involved in promoting intracellular inflammatory pathways in adipocytes(Reference Monaco-Brown and Lawrence60). These findings have significant public health implications in light of the transition towards westernized-style diets throughout pregnancy and thus higher intakes of n-6 PUFA, particularly within the Global South. Such dietary changes may have important consequences for adiposity accretion and subsequently obesity development in the child in later life.
Although maternal n-3 PUFA were not found to be associated with child weight, cord DHA concentrations <0·071 mg/ml were associated with an increased risk of LGA. Much of the existing evidence has focused on maternal n-3 PUFA exposure earlier in pregnancy and the risk of preterm birth and gestational duration(Reference Baker, Calder and Kermack61), whereas fewer have examined cord PUFA status at delivery and its potential influence on fetal growth at term. One study of 847 mother–child pairs reported a negative association between cord erythrocyte DHA concentrations and birth weight(Reference Cinelli, Fabrizi and Ravà62), whilst another, smaller observational study of 253 mother–child pairs also found a negative association between cord DHA concentrations and birth weight(Reference Kilari, Mehendale and Dangat63). Whilst preterm births were excluded in the current analyses, and therefore the results may not be generalisable to pregnancies ending preterm or to outcomes related to gestational duration, the findings of the current study suggest that later-stage n-3 PUFA exposure may be important in relation to birth size. The n-3 PUFA are essential in mediating the immune response, acting as precursors for eicosanoids and docosanoids which are considered to be more anti-inflammatory(Reference Calder64). The n-3 PUFA inhibit nuclear transcription factor kappa B, promoting a more anti-inflammatory immune response, which in turn may modulate adipokine secretion and thus lower adipogenesis and promote a more metabolically healthy phenotype(Reference Albracht-Schlulte, Kalupahana and Ramalingam65). Within this cohort, only thirty-eight (3·2 %) of children were considered LGA which is considerably lower than many other birth cohorts within Africa(Reference Sridhar, Ferrara and Ehrlich66,Reference Younes, Samara and Salama67) but is reflective of the Seychellois population which has an incidence of 4·4 %. The traditional Seychellois diet is rich in oily fish and fish dishes, which has been previously shown to be associated with a higher n-3 PUFA status(Reference Conway, Mulhern and McSorley30). Given both the rising incidence of LGA babies in many populations and the long-term metabolic complications that are often associated with LGA(Reference Hong and Lee68), the unique dietary patterns within the Seychelles paired with the low incidence of LGA within this population may provide promising evidence of potential dietary interventions in helping to lower the risk of LGA within the child through increasing n-3 PUFA intakes throughout pregnancy.
It is important to recognise the differing associations between the maternal and cord PUFA observed within this study. For example, in this cohort, maternal n-6 PUFA were consistently associated with an increased risk of overweight/obesity at 7 and 13 years, whilst few associations were found with the cord PUFA. These differing associations are interesting, considering the developing fetus relies entirely on the maternal supply of PUFA owing to insufficient desaturase activity within the placenta or fetus to synthesise PUFA(Reference Hanebutt, Demmelmair and Schiessl69). The circulating concentrations of PUFA fluctuate throughout gestation with the placenta controlling the uptake and transfer of maternal PUFA to the fetus(Reference Duttaroy70). Previous evidence has suggested that placental uptake and transfer differ, with the transfer favouring DHA and AA, particularly during the last trimester of pregnancy. The differing associations apparent within the current study may be owing to differences in genetics influencing PUFA metabolism, in addition to the altered fatty acid metabolism throughout pregnancy; the maternal PUFA status reflects exposure throughout the entire pregnancy, whilst the cord PUFA status may more accurately depict exposure throughout the last trimester. The placenta has been shown to play an active role in fetal programming(Reference Kwon and Kim72), and given that PUFA transfer across the placenta differs throughout gestation, it may point towards specific periods during pregnancy where the developing fetus may be particularly sensitive to the effects of the n-3 and n-6 PUFA. In future, a focused examination using repeat measurements of PUFA status throughout pregnancy may help to elucidate the differing associations between the maternal and cord PUFA and child adiposity.
The current study has several strengths. First, the use of robust biological measures of both cord and maternal PUFA concentrations and the assessment of dietary intakes of fish provide a robust depiction of exposures throughout pregnancy. The use of numerous time points also helped to determine if associations persisted throughout childhood. The large cohort size and the high participant retainment throughout the follow-up appointments are also particular strengths of this research. Finally, this cohort was well characterised at numerous time points, allowing the comprehensive adjustment of confounders, including fish intakes throughout childhood. There are, however, several limitations. Like all observational epidemiological studies, no cause-and-effect relationships can be established and can only identify associations, which might be non-causal. Although models were adjusted for a range of key maternal and child characteristics, we cannot exclude the possibility of residual confounding from unmeasured factors. Variables such as gestational weight gain, paternal BMI, maternal physical activity, broader dietary patterns or other environmental exposures may have influenced child body weight and should be considered when interpreting these findings. These variables were not available as the present study was a secondary analysis of an existing cohort that was not originally designed to address these specific outcomes. The practical challenges of conducting data collection in a developing country also limited the feasibility of capturing all potential confounders. Maternal fish intakes were assessed using an FUQ administered at 28 weeks’ gestation which may not accurately capture fish consumption across the entire pregnancy. Moreover, the FUQ cannot capture other dietary factors which may have influenced results. Additionally, maternal PUFA status was measured at one time point only, and as mentioned, future research may wish to use repeated measures to determine any sensitive periods as to when the developing fetus may be differentially susceptible to the effects of the n-3 and n-6 PUFA. We were also unable to assess PUFA status throughout childhood. It is worth noting that in the present study, fatty acids were reported as absolute concentrations rather than as percentages of total fatty acids which may limit comparability to other studies. During pregnancy, however, absolute concentrations may more closely reflect maternal–fetal exposure, as placental transfer depends on circulating levels. In contrast, percentage values can be affected by changes in other lipid fractions, which fluctuate substantially in pregnancy(Reference Brenna, Plourde and Stark73). Finally, the methods used were to enable the detection of subtle associations between exposures and measures of child weight, and thus, the clinical significance of these findings is unclear.
Conclusions
In summary, in this high fish-eating cohort, maternal total fish consumption throughout pregnancy was not associated with child weight at birth or throughout childhood. This study, however, suggests that lower cord DHA, an n-3 PUFA, may be associated with higher risk of LGA at birth, whilst higher n-6 PUFA during pregnancy may be associated with adiposity development throughout childhood to early adolescence. This study has global relevance given the transition towards westernized-style diets, typically higher in n-6 PUFA and lower in n-3 PUFA, in many countries. Future research is needed to explore the potential long-term metabolic consequences of these associations.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/S0007114526106369
Acknowledgements
This study was supported by the National Institutes of Health (grants R01-ES010219, P30-ES01247,T32-ES007271 and R24-ES029466) and in-kind support from the government of Seychelles. The funders had no involvement or restrictions in relation to the publication of this manuscript.
James E McMullan: Conceptualisation, Methodology, Investigation and Writing – Original Draft. Alison J. Yeates: Writing – Review and Editing and Funding Acquisition. Philip J. Allsopp: Writing – Review and Editing. Maria S. Mulhern: Writing – Review and Editing and Funding Acquisition. Marie C. Conway: Writing – Review and Editing and Investigation. Toni Spence: Writing – Review and Editing and Investigation. J.J. Strain: Writing – Review and Editing, Funding Acquisition, Conceptualisation and Resources. Edwin van Wijngaarden: Writing – Review and Editing, Funding Acquisition, Resources and Conceptualisation. Gary J. Myers: Writing – Review and Editing, Funding Acquisition, Conceptualisation and Resources. Emelyn Shroff: Writing – Review and Editing, Resources and Funding Acquisition. Juliette Henderson: Resources. Conrad F. Shamlaye: Writing – Review and Editing, Funding Acquisition, Resources and Conceptualisation. Emeir M. McSorley: Supervision, Conceptualisation, Methodology, Writing – Review and Editing, Resources and Funding Acquisition.
All authors declare no conflicts of interest.