Introduction
Maternal nutrition plays an important role in the health of both the mother and her offspring. In particular, the adequate consumption of nutrient-dense food during pregnancy is necessary to support the growth of vital foetal organs, including the brain.(Reference Na, Mackean and Cape1,Reference Cortes-Albornoz, Garcia-Guaqueta and Velez-van-Meerbeke2) The brain develops through a multi-step process that begins approximately three weeks after fertilisation with the initiation of neurogenesis, where neuroepithelial cells differentiate into radial glial cells that produce intermediate progenitor cells and neurons.(Reference Cortes-Albornoz, Garcia-Guaqueta and Velez-van-Meerbeke2) These neurons migrate radially along the radial glial cell scaffolds to form the cortical plate in an ‘inside-out’ manner, while inhibitory interneurons migrate tangentially from the ganglionic eminence.(Reference Delgado, Allen and Keefe3,Reference Nowakowski, Pollen and Sandoval-Espinosa4) As neurons migrate, they differentiate and establish regional identities and functional roles, after which synaptogenesis occurs, whereby connections are formed between neurons to enable communication, and synaptic assembly and dendritic spine formation is refined through activity-dependent pruning.(Reference Delgado, Allen and Keefe3,Reference Yee, Xiao and Chen5) Concurrently, apoptosis eliminates excess neurons to ensure that the neural network is properly organised. These processes occur over a prolonged developmental period in humans and contribute to the complexity and functionality of the mature brain.(Reference Zhou, Song and Ming6)
The impact of an imbalanced maternal macronutrient intake in neurodevelopment is well documented. High-fat intake during pregnancy stimulates the proliferation of neuroepithelial and neuronal precursor cells and changes the migration of neurons, which alters the energy metabolism of the offspring.(Reference Cortes-Albornoz, Garcia-Guaqueta and Velez-van-Meerbeke2) In addition, excessive sugar intake during pregnancy has been linked to impaired hippocampal neurogenesis and altered synaptic plasticity in the offspring, which can negatively affect learning and memory.(Reference Yamazaki, Yamada and Munetsuna7) In contrast, the maternal intake of fruits can improve cognitive behaviour in humans and animals,(Reference Bolduc, Lau and Rosenfelt8–Reference Yonezawa, Ueno and Obara10) while fruit and vegetable consumption during pregnancy is inversely associated with hyperactivity and emotional problems among children, suggesting the neurodevelopmental properties of phytochemicals.(Reference Miyake, Tanaka and Okubo11)
Polyphenols are aromatic alcohol compounds that possess two or more phenolic groups as the functional components. They are widely distributed across various plant species and are found in fruits, vegetables, and plant-derived consumables such as tea and wine. Polyphenols are broadly divided into flavonoids and non-flavonoids. Flavonoids are a diverse group of phytonutrients present in various fruits and vegetables and belong to a family of polyphenols known for their antioxidant properties. Structurally, they consist of two phenolic benzene rings that are connected by three carbon bridges to form a heterocyclic ring.(Reference Tsao12) Flavonoids are abundant in apples, berries, citrus fruits, onions, spinach, tea, red wine, and soy products, and are grouped into several subclasses, which include flavonols, flavones, isoflavones, flavanones, flavan-3-ol, and anthocyanins. Examples of flavonoids are quercetin, fisetin, genistein, naringin, and epigallocatechin gallate (EGCG). Non-flavonoids are a diverse group of polyphenolic compounds that are not part of the flavonoid family. They are found in coffee, grapes, berries, whole grains, seeds, nuts, and olive oil and include stilbenes such as resveratrol and curcuminoids, and phenolic acids such as ferulic acid.
Recent polyphenol research has focused on the effects of polyphenols on brain health. Polyphenol-rich fruits and vegetables decrease oxidative stress, regulate signalling pathways, and promote the expression of genes involved in brain function.(Reference Carrillo, Arcusa and Xandri-Martínez13,Reference Witucki, Kurpik and Jakubowski14) Therefore, the study of these compounds could identify their potential to prevent and treat neurodegenerative diseases such as Alzheimer’s disease, Parkinson’s disease, as well as cerebral ischaemia.(Reference Calderaro, Patanè and Tellone15–Reference Pontifex, Malik and Connell17) Despite extensive research on individual polyphenols in the context of brain health, few studies have comprehensively investigated the effects of multiple types of polyphenols during the prenatal period, especially regarding foetal brain development through maternal dietary intake.(Reference Heland, Fields and Ellery18,Reference Nacka-Aleksić, Pirković and Vilotić19) Emerging evidence suggests that maternal intake of polyphenols may positively influence these developmental events by modulating redox balance, inflammatory pathways, and vascular function. For instance, maternal supplementation with resveratrol or pomegranate juice, polyphenol-rich sources, have been associated with improved offspring brain outcomes in both animal and clinical studies.(Reference Ross, Cherkerzian and Mikulis20–Reference Tain and Hsu22)
However, most available studies have examined individual polyphenols, often within specific maternal risk conditions such as intrauterine growth restriction or metabolic dysregulation.(Reference Ros, Argente and Chowen21) These findings limit broader mechanistic understanding and translational potential. Moreover, human studies remain constrained by ethical and practical challenges, restricting the scope of invasive mechanistic investigations during pregnancy. Nevertheless, a comprehensive understanding of how diverse polyphenols modulate foetal neurodevelopment across different experimental settings remains limited. In this context, rodent models provide a valuable experimental framework due to their controlled environments, genetic consistency, and well-defined developmental timelines.(Reference Bale23) This narrative review aims to synthesise recent findings from rodent studies examining the effects of maternal polyphenol intake on foetal brain development. By evaluating various polyphenol compounds and experimental models, this review seeks to identify common mechanisms, address current gaps in the literature, and offer insights for future research directions.
This narrative review was conducted to synthesise current evidence on the effects of maternal polyphenol intake on foetal brain development in rodent models. Studies were included in this review if they investigated the administration of polyphenols to pregnant rodents (Table 1) and reported experimental outcomes related to offspring brain development (Table 2), such as neurogenesis, oxidative stress, inflammation, or behavioural changes. Only in vivo rodent studies were considered. Although this review primarily focused on studies published within the last ten years, older studies were also included for polyphenols such as resveratrol, quercetin, naringenin/naringin and genistein due to the limited availability of recent research addressing their effects on foetal brain development. These earlier studies were selected based on their scientific relevance and contribution to mechanistic understanding of how maternal polyphenol intake influences foetal brain development. Exclusion criteria are clinical studies involving human participants, in vitro studies, non-rodent models, and studies not focused on brain-related outcomes were excluded from the review. Our search strategy is a literature search was conducted using PubMed for articles published from January 2015 to February 2025. Search terms included combinations of the following keywords: ‘polyphenol’ OR ‘resveratrol’ OR ‘curcumin’ OR ‘quercetin’ OR ‘naringenin’ OR ‘ferulic acid’ OR ‘genistein’ OR ‘fisetin’ OR ‘EGCG’, AND ‘maternal’ OR ‘pregnancy’, AND ‘brain’ OR ‘neuron’ OR ‘neurodevelopment’, AND ‘rodent’ OR ‘rat’ OR ‘mouse’. Relevant articles were also identified by manually screening the reference lists of included studies. Study selection was conducted in three stages: title screening, abstract screening, and full-text screening by the author to ensure eligibility based on predefined inclusion and exclusion criteria. Data were organised thematically based on the type of polyphenol used and the primary outcomes measured. Organising the findings according to specific themes enabled clear recognition of recurring mechanisms and research gaps across different experimental systems. Here, we summarised (Table 2) and discussed the role of maternal polyphenols in foetal brain development using recently published findings.
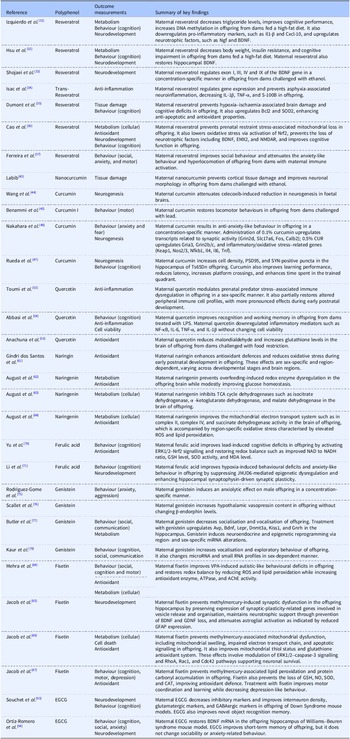
Summary of study design investigating the effects of maternal polyphenol supplementation on foetal and offspring brain development in rodent models. The table summarises the experimental model, rodent strain, stress model, route of administration (e.g. oral, intraperitoneal, subcutaneous), dose and duration of exposure, and timing of outcomes assessed. Polyphenols include resveratrol, curcumin, quercetin, naringin/naringenin, ferulic acid, genistein, fisetin, and EGCG

Summary of the effect of maternal polyphenol on offspring brain in rodent models. The table summarises evidence from rodent studies examining maternal supplementation of different polyphenols (resveratrol, curcumin, quercetin, naringin, ferulic acid, genistein, fisetin, and EGCG). Reported results are categorised into six major outcome measurements: antioxidant, anti-inflammation, metabolism, neurogenesis and neurodevelopment, cell death and tissue damage, and behavioural analysis. Reference numbers correspond to the studies cited in the text

Resveratrol
Resveratrol (3,5,4’-trihydroxy-trans-stilbene) is a natural polyphenolic compound (Figure 1) that consists of two phenolic rings connected by ethylene bridges and three hydroxyl groups. Each benzene ring contains a hydroxyl group, contributing to its strong antioxidant properties. Resveratrol is naturally present in a variety of foods, including berries such as blueberries and cranberries, as well as peanuts, dark chocolate, and red wines, which have a high concentration of resveratrol from the skins and seeds of red grapes.(Reference Teka, Zhang and Ge24,Reference Tian and Liu25) Resveratrol exhibits neuroprotective effects through the AMP-activated protein kinase (AMPK)/Sirtuin 1(SIRT1) pathway of Parkinson’s disease and alcohol-induced neurodegeneration.(Reference Wu, Li and Zhu26,Reference Gu, Cai and Cai27) The activation of AMPK by resveratrol stimulates SIRT1, leading to reduced inflammation by suppressing nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and promoting autophagy, thereby contributing to neuroprotection.(Reference Qi, Shang and Liao28,Reference Wang, Amato and Gilbert29) Resveratrol mitigates mitochondrial dysfunction associated with chronic stress by modulating the SIRT1/PGC1α/SIRT3 pathway, thereby restoring oxidative balance and enhancing neuroprotection.(Reference Tabassum, Misrani and Huang30) Through this mechanism, resveratrol counteracts cellular damage and alleviates anxiety- and depression-like behaviours induced by chronic unpredictable mild stress. These findings indicate that resveratrol exerts its neuroprotective effects through multiple pathways and targets inflammation and cellular resilience to oxidative stress, thereby promoting neuronal survival and cognitive function.
Mechanisms of Resveratrol-Mediated Neurogenesis. (a), Chemical structure of resveratrol. (b) Mechanistic illustration of resveratrol-mediated neurodevelopmental pathways. Resveratrol activates AMPK, which increases NAD+ levels, subsequently activating SIRT1. SIRT1 activation leads to NF-κB suppression downregulating pro-inflammatory cytokines such as IL-1β and TNF-α. In addition, SIRT1 supports mitochondrial integrity by upregulating the anti-apoptotic proteins Bcl-2 and Bcl-xL, inhibiting cytochrome c release, and preventing apoptosis. p-AKT further enhances cell survival under stress. Resveratrol increases BDNF and synapsin-1, strengthening the synaptic plasticity that is critical for learning and memory. Sox2 and Olig1, regulated through SIRT1 signalling, support neurogenesis and myelination, respectively, contributing to neuronal resilience and efficient signal transmission. Illustration by BioRender. AMPK: AMP-activated protein kinase; BDNF: brain-derived neurotrophic factor, Bcl-2: B-cell leukaemia/lymphoma 2, Bcl-xL: B-cell lymphoma-extra large, IL: interleukin, NAD: nicotinamide adenine dinucleotide, NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells, Olig1: oligodendrocyte transcription factor 1, SIRT: Sirtuin 1, Sox2: SRY-Box transcription factor 2, TNF: tumour necrosis factor, p-AKT: phosphorylated RAC-alpha serine/threonine-protein kinase.

The role of resveratrol in foetal brain development has been tested in maternal models with imbalanced diets. Izquierdo et al. discovered that resveratrol prevented cognitive impairment associated with a maternal high-fat diet, increased deoxyribonucleic acid (DNA) methylation, and reduced pro-inflammatory gene expression.(Reference Izquierdo, Palomera-Ávalos and Pallàs31) That study used female senescence-accelerated mouse prone 8 mice that were fed a high-fat diet, 60% of total calories from fat alone, or a high-fat diet supplemented with resveratrol throughout pregnancy and lactation. The offspring were continued on the same respective diets. Resveratrol was administered at a dose of 1 g/kg w/w directly into the high-fat diet to ensure consistent daily intake. This supplementation prevented the cognitive impairment associated with a maternal high-fat diet and increased DNA methylation. In addition, treatment with resveratrol downregulated gene expression of pro-inflammatory cytokines such as interleukin (IL)-1β, tumour necrosis factor (TNF)-α, c-x-c motif chemokine ligand 10, and monocyte chemoattractant protein-1. The results showed that nerve growth factor and neurotrophin-3 expression and their respective receptor activations were significantly higher in the high-fat diet supplemented with resveratrol group compared to the high-fat diet-only group. A similar finding was reported in a rat model, in which supplementation with resveratrol improved the spatial memory of offspring of obese Sprague-Dawley rats fed a high-fat diet.(Reference Hsu, Sheen and Lin32) In that study, pregnant rats were given a regular diet or a high-fat diet with a composition of 58% fat from calories for 8 weeks. The high-fat diet group was supplemented with or without 50 mg/kg of resveratrol daily throughout the pregnancy and the male offspring were given the same diet as their mother. The maternal high-fat diet caused reduced levels of adiponectin, phosphorylated RAC-alpha serine/threonine-protein kinase (p-AKT), and brain-derived neurotrophic factor (BDNF) levels, while resveratrol prevented these alteration in the brains of offspring of high-fat diet treated mothers. Resveratrol also improved obesity-associated cardiometabolic factors including body weight, insulin resistance, blood pressure, aspartate aminotransferase, alanine aminotransferase, and total cholesterol.
In addition to the maternal high-fat diet, Shojaei et al. suggest that the effect of ethanol and resveratrol depends on the concentration of ethanol and the developmental stage of animals.(Reference Shojaei, Ghavami and Panjehshahin33) Those authors examined the effects of oral ethanol, alone or in combination with resveratrol, on BDNF transcript and pro-BDNF protein expression in the hippocampi of pregnant and embryonic Sprague-Dawley rats. Pregnant rats were orally gavaged with ethanol (0.25, 0.63, or 2.5 g/kg/day) and resveratrol (60 or 120 mg/kg/day) for 20 days. Ethanol altered messenger ribonucleic acid (mRNA) and protein levels of BDNF in various exons, while treatment with resveratrol increased BDNF expression during low-dose ethanol challenge.
Resveratrol is reported to exhibit protective roles in brains challenged with oxygen depletion during development. Isac et al. reported that the protective effects of a trans-resveratrol-enriched maternal diet on the immature hippocampi of neonatal Wistar rats exposed to perinatal asphyxia.(Reference Isac, Panaitescu and Spataru34) The rats were divided into three groups: control, perinatal asphyxia, and perinatal asphyxia with resveratrol. Perinatal asphyxia was induced by exposing 6-day-old pups to a hypoxic environment with 9% oxygen, 20% carbon dioxide, and 71% nitrogen for 90 minutes. The mother rats were given free access to water containing trans-resveratrol dissolved in 0.5% dimethyl sulphoxide at a dosage of 50 mg/kg/day from weaning until the birth of their pups and continuing until postnatal day 7. Maternal administration of trans-resveratrol significantly prevented asphyxia-induced upregulation of IL-1β, TNF-α, and S100 calcium-binding protein B (S100B) in the hippocampi of the offspring, indicating protection against neuroinflammation and nerve damage. In addition, resveratrol reduced the expression of MicroRNAs (miRs) 132 and 15a, suggesting an epigenetic effect. Dumont et al. evaluated maternal resveratrol supplementation in neonates with hypoxic-ischaemic brain injury.(Reference Dumont, Sanchez and Repond35) Hypoxia-ischaemia was induced at postnatal day 7 by permanent occlusion of the left common carotid artery, and maternal resveratrol (0.15 mg/kg) was orally administered over the following periods: final week of gestation and first week of lactation, last week of gestation only, first week of lactation only, and second week of lactation. Maternal resveratrol decreased hypoxia-ischaemia-induced brain lesions and protected the cognitive and sensorimotor function of the offspring. Furthermore, treatment with resveratrol upregulated SIRT1 and prevented the downregulation of genes associated with oxidative stress and metabolic pathways such as superoxide dismutase (SOD), monocarboxylate transporter 2, lactate dehydrogenase, and glutamate transporter 1.
Resveratrol may improve the brain development of offspring from mothers with stress or psychological disorders. Cao et al. administered resveratrol to Sprague-Dawley dams during restraint stress and examined the AMPK pathways in the brains of the offspring.(Reference Cao, Zheng and Xu36) Restraint stress was applied to the pregnant rats by placing them in transparent plastic tubes three times daily for 2 hours during gestational day (GD) 14 to 20. The stress-induced pregnant rats that received 100 mg/kg/day of resveratrol orally had offspring with increased AMPK activation that declined due to stress and increased phosphorylation levels. In addition, resveratrol prevented the loss of mitochondrial biogenesis proteins such as Complex I, III, IV, V, and PGC-1α against prenatal restraint stress. Moreover, treatment with resveratrol mitigated oxidative stress by increasing nuclear factor erythroid 2-related factor 2 (Nrf2) and its target gene expression, nicotinamide adenine dinucleotide (NADH), quinone dehydrogenase 1 (NQO1), and haem oxygenase-1 (HO-1). The carbonyl protein levels, which are a marker of oxidative stress, were decreased in the resveratrol-treated group, while the gene expression of neurotrophic factors such as BDNF and growth-associated protein (GAP) 43 were upregulated, suggesting a role in neurodevelopment regulation. Ferreira et al. explored the effects of resveratrol on behavioural and neurodevelopmental abnormalities associated with maternal immune activation, which is a risk factor for schizophrenia in offspring.(Reference Ferreira, de Moura and Hassib37) Resveratrol was administered at 40 mg/kg from GD 10.5 to 16.5, and maternal immune activation was induced in pregnant mice on GD 12.5 through an injection of polyriboinosinic-polyribocytidylic acid, a synthetic compound that mimics a viral infection. The offspring were assessed for social interaction, anxiety, and hyperlocomotion behaviours on postnatal days (PD) 45, and a molecular analysis focused on mRNA expression of synapsin I, oligodendrocyte transcription factor 1 (Olig1), and SRY-Box transcription factor 2 (Sox2) in the hippocampus and cortex, as these are markers associated with synaptic plasticity, oligodendrogenesis, and neurogenesis, respectively. Maternal resveratrol treatment improved social behaviours and reduced hyperlocomotion in the offspring, while the upregulated mRNA expression of Syn prevented the upregulation of Olig1 in the cortex and hippocampus of offspring against maternal immune activation.
Curcumin
Curcumin (diferuloylmethane) is a natural polyphenolic compound with two phenyl rings connected through an alkene bond, the centre of which features a di-keto structure that contributes bioactivity and other properties (Figure 2).(Reference Kotha and Luthria38) This polyphenol is abundant in turmeric, which is a member of the ginger family of plants. Curcumin neutralises reactive oxygen species (ROS) and protects cellular components such as DNA and lipids from oxidative stress through activation of the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt) and wingless-related integration site (Wnt) /β-catenin pathways.(Reference Lou, Gong and Yang39) In addition, curcumin regulates the cAMP Response Element-Binding Protein (CREB)/BDNF pathway, supporting synaptic plasticity and neuronal health(Reference Lou, Gong and Yang39); enhances the Nrf2 pathway, increasing HO-1 and NQO1 expression for antioxidant defence and autophagy via protein kinase C (PKC) δ-mediated phosphorylation of p62(Reference Park, Sohn and Koh40); and reduces cyclooxygenase-2 (COX-2) and increases BDNF, further supporting neuroprotection.(Reference Lou, Gong and Yang39–Reference Choi, Kim and Hwang42)
Mechanisms of Curcumin-Mediated Neurogenesis. (a) Chemical structure of curcumin. (b) Mechanistic illustration of the neurodevelopment pathways of curcumin. Curcumin activates the AMPK and PI3K/Akt pathways, promoting antioxidant and anti-inflammatory effects by reducing ROS and inhibiting the expression of pro-inflammatory cytokines such as IL-6 and TNF-α. Furthermore, curcumin modulates neurodevelopment by enhancing Wnt signalling and inhibiting GSK-3β, which stabilises β-catenin, leading to the upregulation of neurodevelopmental genes such as DBN1 and GAP-43. These processes influence transcription factors such as CREB and NF-κB, thereby supporting neuronal growth and synaptic plasticity essential for brain development. Illustration by BioRender. Akt: Protein kinase B, AMPK: AMP-activated protein kinase; CREB: cAMP Response Element-Binding Protein, DBN1: Drebrin 1, GAP: Growth-associated protein, GSK: Glycogen synthase kinase, IL: Interleukin, NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells, PI3K: Phosphatidylinositol 3-kinase, ROS: Reactive oxygen species, TNF: Tumour necrosis factor.

Maternal exposure to chemical or pharmacological compounds may negatively influence foetal brain development. Curcumin has been reported to protect the foetal brain from prenatal exposure to toxic compounds. Labib et al. studied the neuroprotective effects of nanocurcumin against the negative impacts of prenatal alcohol exposure.(Reference Labib43) Pregnant Wistar rats were assigned to four groups: control group receiving tap water, sham group with intraperitoneal nanocurcumin, ethanol-exposed group given 10% ethanol orally during gestation, and combination of ethanol and nanocurcumin group administered both treatments. Nanocurcumin (25 mg/kg) was administered twice daily, and at PD21, histological staining, electron microscopy, and molecular analyses were used to evaluate the prefrontal cortex changes of the offspring. Ethanol exposure significantly increased markers of oxidative stress and inflammation and decreased synaptic integrity and DNA stability in the prefrontal cortex of the offspring, as indicated by reduced drebrin 1 (DBN1) and altered DNA methyltransferase 1 expression. Histological analysis showed cellular shrinkage and vascular dilation, while ultrastructural examination revealed disrupted nuclear integrity and myelin sheath irregularities. Co-administration of nanocurcumin reduced oxidative stress, normalised synaptic and methylation markers, and improved cellular structure, demonstrating the neuroprotective effects of this compound against ethanol-induced damage. Wang et al. showed that curcumin supported neurogenesis in the foetal frontal cortex via the Wnt/β-catenin pathway in a mouse model.(Reference Wang, Tian and Yang44) Pregnant C57BL/6J mice were used to investigate the effects of maternal curcumin on celecoxib, a cyclooxygenase inhibitor commonly given to pregnant women. Mice were randomly assigned to three groups: control group receiving 10 mL/kg of saline vehicle from embryonic day 16.5 (E16.5) to E17.5, celecoxib group treated with celecoxib at 30 mg/kg in saline from E16.5 to E17.5 to inhibit neurogenesis, and combination of celecoxib and curcumin group, where 500 nmol/kg of curcumin was administered intraperitoneally daily from E13.5 to E16.5 prior to celecoxib treatment. After treatment, foetal brains were collected, fixed, and sectioned for immunofluorescence staining of neural progenitor markers such as BrdU, Pax6, and cyclin D1 to assess proliferation. For the in vitro study, primary neuronal progenitor cells were isolated from the foetal cortex at E16.5, and Wnt/β-catenin pathway components including glycogen synthase kinase (GSK)-3β and β-catenin were quantified using western blotting. The celecoxib treatment inhibited neurogenesis in the foetal frontal cortex, marked by suppression of the Wnt/β-catenin pathway and decreased cyclin D1 expression. In particular, the β-catenin levels were reduced and GSK-3β levels were increased in the neural progenitor cells, leading to impaired cell proliferation. In contrast, the group treated with both celecoxib and curcumin showed significant attenuation of the effects of celecoxib, with restored β-catenin and cyclin D1 expression and improved neural cell proliferation. Benammi et al. studied the effects of lead intoxication and curcumin treatment on sensorimotor and electrophysiological functions in Wistar rats.(Reference Benammi, Erazi and El Hiba45) Pregnant rats were divided into three groups: control group with standard drinking water, lead-treated group exposed to 3 g/L lead acetate in drinking water during gestation and lactation, and combination of lead and curcumin group that was additionally gavaged with 16 g/kg curcumin dissolved in olive oil. Offspring were evaluated at PD1 – PD2 and at a young age up to PD60 for gait analysis and spinal cord excitability through electrophysiological recordings of lumbar ventral roots to assess spontaneous and induced activity. Lead exposure significantly impaired body weight and motor coordination with affected pups showing delayed cliff-drop avoidance and forepaw grasping responses at PD1–PD2 and reduced walking velocity and increased stride length at PD60. Electrophysiological recordings revealed heightened spontaneous and rhythmic locomotor activity in lead-treated pups, indicating hyperexcitability in the spinal circuits. However, oral supplementation of curcumin attenuated the effects of lead intoxication.
In addition to alleviating the effects of maternal chemical exposure, curcumin attenuates the impacts of various brain disorders caused by mental, genetic, and hormonal factors. Nakahara et al. administered curcumin continuously from the gestational stage through adolescence to examine anxiety-like behaviour and fear extinction learning in offspring.(Reference Nakahara, Masubuchi and Takashima46) Pregnant Long-Evans rats were divided into three groups: control, 0.1 w/w% curcumin in diet, and 0.5 w/w% curcumin in diet. Immunohistochemical analysis and reverse transcription polymerase chain reaction was performed to assess changes in synaptic plasticity-related markers and genes related to neurogenesis, oxidative stress, and inflammation in brain regions such as the hippocampal dentate gyrus and medial prefrontal cortex. The rats treated with 0.1 w/w% curcumin showing reduced anxiety-like behaviour and improved fear extinction learning along with an increased expression of synaptic plasticity markers such as the Fos and activity-regulated cytoskeleton (Arc) proteins. Overall, low-dose curcumin promoted neurogenesis and synaptic plasticity, while a high dose could hinder these protective effects, highlighting the dose-dependent effects of curcumin on brain health. Rueda et al. utilised Trisomy 65 Down syndrome (Ts65Dn) mice, which are a model for Down syndrome, to examine the effects of prenatal curcumin on neurogenesis and cognitive function.(Reference Rueda, Vidal and García-Cerro47) Pregnant Ts65Dn females were administered 300 mg/kg of curcumin or vehicle subcutaneously from E10 to PD2. Offspring were assigned to control and treatment groups based on their prenatal exposure. Short-term assessments of neurogenesis, brain weight, and cellular density were conducted on PD2, and long-term effects on cognition, neurogenesis, and synaptic density were evaluated between PD30 and PD90. Additional postnatal treatments were administered from PD3 to PD15, with parallel analyses on PD15 and PD90 to analyse the immediate and extended effects. Standard immunohistochemical techniques were used to assess markers such as BrdU and Ki67 for neurogenesis, 4’,6-diamidino-2-phenylindole (DAPI) for cell density, and postsynaptic density protein 95 for synaptic density. Prenatal curcumin treatment significantly increased brain weight, BrdU and DAPI positive cell density, and granule cell layer volume in Ts65Dn offspring by PD2, suggesting enhanced neurogenesis and cellular resilience. However, no significant benefits in neurogenesis or cognition at short- or long-term intervals were noted with curcumin administration, indicating that prenatal exposure was critical for the observed neuroprotective effects.
Quercetin
Quercetin is a polyphenolic flavonoid compound with a 3-ring system consisting of two benzene rings connected by a heterocyclic pyrone ring (Figure 3). Quercetin is an antioxidant polyphenol that is predominantly found in grapes, broccoli, potatoes, onions, oranges, and berries, and its beneficial effects include the reduction of ROS and the resultant prevention of injuries induced by oxidative stress.(Reference Dagher, Mury and Thorin-Trescases48,Reference Babaei, Mirzababaei and Nassiri-Asl49) The antioxidant activity of quercetin is attributed to the 2,3-double bond in the carbon ring of the molecule and the 4-oxo group that captures ROS directly through a phenolic hydroxyl group.(Reference Li, Yao and Han50) Studies have shown that quercetin contributes to the prevention of neurodegeneration by ROS and relieving inflammatory responses by elevating the levels of antioxidant enzymes, catalase, SOD, and glutathione peroxidase while suppressing the activation of the PKC/mitogen-activated protein kinase (MAPK)/NADPH Oxidase pathway.(Reference Huang, Zhang and Zou51) Quercetin also activates the Nrf2 pathway, which is a key transcription factor in the regulation of antioxidant gene expression and an important component for the protection and survival of neurons.(Reference Xie, Zhao and Lowe52) These characteristics position quercetin as a potential therapeutic agent for the prevention and treatment of neurodegenerative diseases, providing a positive impact on neurodevelopment. Therefore, we focus on the neuroprotective and neurodevelopmental effects of quercetin.
Mechanisms of Quercetin-Mediated Neurogenesis. (a) Chemical structure of quercetin. (b) Mechanistic illustration of the neurodevelopment pathways of quercetin. Quercetin reduces oxidative stress both by directly scavenging ROS and activating the Kelch-like ECH-associated protein 1-Nrf2 pathway, which enhances the expression of antioxidant enzymes such as SOD, catalase, and GSH. This polyphenol also exhibits anti-inflammatory effects by inhibiting NF-κB activation, thereby decreasing the production of pro-inflammatory cytokines such as TNF-α and IL-6. In the mitochondria, quercetin modulates the Bax/Bcl-2 ratio, preventing cytochrome c release and inhibiting apoptosis. These combined effects contribute to the normalisation of neuronal migration, restoration of the external granular layer, and the promotion of anti-inflammatory and neuroprotective outcomes. Illustration by BioRender. Bcl-2: B-cell leukaemia/lymphoma 2, GSH: Glutathione; IL: Interleukin, NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells, Nrf2: Nuclear factor erythroid 2-related factor 2, ROS: Reactive oxygen species, SOD: Superoxide dismutase, TNF: Tumour necrosis factor.

Although studies are limited, the effects of maternal quercetin on offspring brain development have been reported using mental and physical stress models of rats. A study performed by Toumi et al. investigated the effects of predator-induced stress during late gestation on maternal anxiety, brain oxidative stress, and corticosterone levels and offspring immune system parameters during early postnatal development.(Reference Toumi, Merzoug and Baudin53) The researchers administered quercetin orally at 50 mg/kg/day for six consecutive days prior to predator stress exposure. Pregnant Wistar rats were subjected to acute predator stress on GD 19 by placing them in a plexiglass chamber with a domestic cat for 15 minutes. Brain oxidative stress markers, including reduced glutathione (GSH) and malondialdehyde (MDA), as well as glutathione-S-transferase (GST) activity, were measured. Offspring immune cell counts, including white blood cell, lymphocyte, monocyte, and granulocyte count, were evaluated on PD 1, 7, 14, and 21. Predator stress significantly increased maternal corticosterone levels, induced anxiety-like behaviours and disrupted locomotion in dams, and caused brain oxidative stress, evidenced by reduced GSH levels, elevated MDA concentrations, and decreased GST activity. The offspring displayed leukopenia, lymphopenia, monocytosis, and granulocytosis in sex-dependent and time-specific manners, with male offspring being more affected. Quercetin pretreatment mitigated the effects of stress in dams by normalising corticosterone levels and improving the oxidative balance in the brain. In the offspring, quercetin effectively prevented early postnatal immune disturbances.
Abbasi et al. utilised a rat model of maternal immune activation to investigate the potential neuroprotective effects of quercetin supplementation.(Reference Abbasi, Ghavami-Kia and Davoodian54) Pregnant Wistar rats were supplemented with 50 mg/kg/day quercetin or a vehicle solution throughout pregnancy. On GD 15 and 16, 0.5 mg/kg of lipopolysaccharide (LPS) or saline was administered intraperitoneally to induce maternal immune activation. Hippocampal and prefrontal cortex tissues were collected, and histopathological examination, pro-inflammatory gene expression analysis, and immunostaining were performed to assess glial cell densities. The molecular analysis revealed decreased expression of pro-inflammatory genes, including NF-κB, IL-6, and TNF-α, in the prefrontal cortex and hippocampus of offspring from quercetin-supplemented dams. Immunohistochemistry showed that quercetin reduced the densities of activated microglia and astrocytes in these brain regions. In addition, maternal quercetin improved the recognition and working memory of offspring exposed to prenatal LPS.
Anachuna et al. evaluated the effects of quercetin on offspring subjected to prenatal and early postnatal food restrictions.(Reference Anachuna, Moke and Iyare55) The prenatal maternal food intake was reduced to 50% of the ad libitum diet during gestation of Wistar rats. Postnatal food restriction was implemented by increasing the litter size to 16 pups per mother from PD 2 to weaning, thereby restricting maternal milk availability. Animals in food-restricted groups were orally administrated with or without quercetin at doses of 50, 100, or 200 mg/kg daily. Maternal and pup body weights were measured at various times, while serum leptin and ghrelin levels were assessed using enzyme-linked immunosorbent assay (ELISA). Oxidative stress markers, including brain GSH and MDA, were quantified spectrophotometrically. The prenatal and postnatal food restrictions caused significant reductions in maternal and pup body weights, elevations in brain oxidative stress, and dysregulation of leptin and ghrelin levels. In pups exposed to combined presynaptic and postsynaptic nerve fibre responses, the effects were more pronounced, indicating cumulative stress. However, treatment with quercetin reversed these effects in a dose-dependent manner. Both antioxidants significantly increased maternal and pup body weights, improved hormonal balance including leptin and ghrelin levels, and mitigated oxidative stress by enhancing GSH and reducing MDA concentrations in the brain.
Naringin and naringenin
Naringin is a polyphenolic flavanone compound with a 3-ring system consisting of two benzene rings connected by a heterocyclic chromanone ring.(Reference Alam, Subhan and Rahman56) The flavonoid naringin and its glycoside form, naringin, which are found predominantly in citrus fruits, are considered antioxidant compounds due to their ability to scavenge free radicals.(Reference Salehi, Fokou and Sharifi-Rad57) Both compounds have demonstrated therapeutic potential in neurological diseases,(Reference Emran, Islam and Nath58) including neuroprotective effects on induced neurotoxicity or brain damage in animals. For example, naringin was reported to reduce oxidative stress and improve mitochondrial dysfunction via activation of the Nrf2/ARE signalling pathway in neurons isolated from rat brains.(Reference Wang, Chen and Huang59) In another study, naringin provided neuroprotection in rats with cognitive disorders caused by proinflammatory chemokines.(Reference Long, Chen and Liao60) Naringin treatment significantly decreased mRNA expression of IL-1β and IL-6, reduced the activities of SOD and glutathione peroxidase, and decreased the level of the oxidative stress marker MDA. These changes resulted in the protection of the structure and survival of the neurons in the cornu ammonis 1 zone of the hippocampus.
In addition to the protective effects on the mature brain, maternal supplementation with naringin or naringenin affects the brain during development of offspring. Maternal supplementation with 100 mg/kg/day of naringin during pregnancy changed the redox status in the brains of offspring in Wistar rats.(Reference Gindri Dos Santos, Peres Klein and Scortegagna Crestani61) The results showed naringin-mediated changes in dichlorofluorescein, GSH, and antioxidant enzymes such as glutathione peroxidase, catalase, and SOD in various regions of the offspring brain. However, the author noted that the mechanisms of underlying redox regulation induced by maternal naringin may differ by age, sex, and brain region of the offspring.
A study by August et al. investigated the effects of maternal naringin supplementation and exercise during pregnancy on offspring, with a particular focus on redox homeostasis in the brain affected by postnatal overnutrition.(Reference August, Maurmann and Saccomori62) Forty-eight female Wistar rats were divided into four groups: sedentary with no supplementation, sedentary with naringin supplementation, swimming exercise with no supplementation, and swimming exercise with naringin supplementation. Naringenin (50 mg/kg) was suspended in sunflower oil and administered orally through gavage after pregnancy began. The swimming exercise was initiated one week prior to pregnancy, with sessions 5 days per week, each lasting 30 minutes. The offspring of each group were divided into control and overfed subgroups, with overnutrition induced by reducing litter size to increase milk intake among pups. On PD21, the serum and specific brain regions, including the hippocampus, cerebellum, and hypothalamus, of the pups were assessed for redox balance and metabolic markers. Maternal exercise or naringin supplementation alone mitigated offspring weight gain and body fat accumulation and tended to reduce the blood glucose level. Maternal naringin supplementation prevented the reduction of thioredoxin reductase activity in the cerebellum of pups challenged with overnutrition. The activity of glyoxalase was increased in the hippocampus in the rats in the overnutrition group, whereas a combination of naringin and exercise nullified this effect. Furthermore, oral supplementation with 50 mg/kg naringenin during pregnancy changed the activity of tricarboxylic acid cycle dehydrogenases in the cerebellum of the offspring.(Reference August, Grings and Grunwald63) Female Wistar rats were treated with or without naringenin during pregnancy and the cerebella of the offspring were collected at PD 7. Offspring from naringenin-treated mothers showed lower levels of isocitrate dehydrogenase, α-ketoglutarate dehydrogenase, and malate dehydrogenase activity than those from the vehicle-fed controls. Naringenin potentially interacted with the binding sites of hydrogenases influencing their enzyme activities. Similarly, the offspring cerebella of mothers treated with 50 mg/kg naringin showed increased activity of the mitochondrial electron transport system,(Reference August, Klein and Grings64) suggesting naringenin may play a role in energy metabolism in the brains of offspring.
Ferulic acid
Ferulic acid is a naturally occurring polyphenol found predominantly in the cell walls of plants and is composed of a benzene ring with hydroxyl and methoxy substituents as well as an unsaturated carboxylic acid side chain.(Reference Srinivasan, Sudheer and Menon65) The chemical structure of ferulic acid contributes to its antioxidant activity in several mechanisms. First, the formation of extended π-conjugates from the phenolic nucleus and the unsaturated side chain support multiple forms of free radicals. Second, electron-donating hydroxyl and methoxy groups enhance the antioxidant capacity. Finally, the carboxylic acid group can anchor to lipid bilayers, providing additional protection against lipid peroxidation.(Reference Kanski, Aksenova and Stoyanova66) Similar to the antioxidants discussed earlier, ferulic acid has strong neuroprotective properties(Reference Di Giacomo, Percaccio and Gullì67) through the prevention of oxidative stress and inflammation.(Reference Moghimi-Khorasgani, Homayouni Moghadam and Nasr-Esfahani68) An analysis of 239 scientific articles suggested that the neuroprotective effects of ferulic acid is associated with the Nrf2/ARE signalling pathway.(Reference Singh, Arthur and Upadhayay69)
Ferulic acid has been indicated as a maternal supplement during pregnancy, whereby supplementation improved the cognitive function of cognitive-impaired mouse offspring.(Reference Yu, Pan and Zhang70) Pregnant mice were exposed to lead from GD 5 to PD 14 via drinking water to produce a cognitive deficit. Mice that were also administered 50 mg/kg of ferulic acid had offspring with significantly improved spatial learning and memory through the oxidative stress reduction that was indicated by changes in the NAD+/NADH ratio and GSH and MDA levels. Additionally, Nrf2 knockout mice showed that ferulic acid did not affect lead-induced memory deficits and oxidative stress, suggesting that Nrf2 was essential in mediating the enhanced cognition and antioxidant effects of ferulic acid.(Reference Yu, Pan and Zhang70) Furthermore, pregnant rats intraperitoneally injected with 8 mL/kg ferulic acid for 7 days produced pups with improved cognitive behaviours against maternal hypoxia.(Reference Li, Liu and Chen71) Offspring from dams exposed to a hypoxic chamber for 2 hours per day for 3 days showed impaired learning and memory; however, treatment with ferulic acid reversed the effect of intrauterine hypoxia. In addition, ferulic acid decreased the expression of Jumonji C domain-containing protein 6 and increased the expression of synaptophysin in offspring brains against intrauterine hypoxia during pregnancy.
Genistein
Genistein is a natural isoflavone with a 3-phenylchromen-4-one skeleton that contains two aromatic rings, a heterocyclic ring with a carbonyl group, and hydroxyl groups at the 7th and 4’ positions, contributing to its antioxidant properties and its role as a phytoestrogen.(Reference Sharifi-Rad, Quispe and Imran72) Genistein is a plant-derived phytoestrogen commonly found in soybeans that can bind to oestrogen receptors and compete with natural oestrogen as an antioestrogen and perform activities similar to those of natural oestrogen.(Reference Sharifi-Rad, Quispe and Imran72) A recent study revealed that genistein-mediated autophagy via Forkhead Box O3-associated pathways played a significant role in protecting cells from neurodegenerative conditions such as Huntington’s disease.(Reference Pierzynowska, Podlacha and Gaffke73) In the other study indicated that genistein exerts neuroprotective effects in ischaemic stroke by modulating the Wnt/Ca2⁺ signalling pathway, thereby mitigating calcium overload, oxidative stress, and neuronal apoptosis.(Reference Li, Liu and Wang74)
Oral supplementation of maternal genistein improved offspring behaviour in rodent models. Rodrigues-Gomez et al. investigated the impact of maternal and early-life exposure to genistein on anxiety, aggression, and the nitric oxide system of offspring.(Reference Rodriguez-Gomez, Filice and Gotti75) Complete deletion (CD)-1 mice were given daily doses of genistein (5 μg/g or 100 μg/g body weight) during late pregnancy and early lactation. Male offspring underwent behavioural tests in adulthood to assess anxiety using elevated plus maze and open field tests and aggression using a neutral cage test. In addition, brain tissue was analysed for changes in neuronal nitric oxide synthase expression in key regions such as the amygdala and hypothalamus to determine the neurophysiological effects. Mice exposed to the higher dose of genistein (100 μg/g) displayed lower anxiety levels, as shown by an increase in the time spent and number of entries into the open arms of the elevated plus maze. This group showed a notable decrease in the number of neuronal nitric oxide synthase-positive neurons in the basolateral amygdala, while no changes were found in the hypothalamus or lateral amygdala. The lower dose (5 μg/g) of genistein caused heightened defensive responses but no significant changes in anxiety levels. Neither dose affected sexual behaviours or offensive aggression. These results demonstrate that genistein exposure during critical developmental periods can lead to dose-dependent, long-lasting effects on behaviour and brain structure. In addition, maternal genistein may regulate the hormonal and body fluid balance of the offspring. Scallet et al. investigated the effects of dietary exposure to genistein on hypothalamic concentrations of vasopressin and β-endorphin in Sprague-Dawley rats.(Reference Scallet, Wofford and Meredith76) Pregnant rats were fed diets containing varying genistein concentrations (25 ppm, 250 ppm, or 1250 ppm) beginning on GD 7. After birth, the offspring were provided with the same diets until PD 77. Brain tissues were dissected, and the hypothalamic contents of vasopressin and β-endorphin were measured using ELISA. The study found that hypothalamic β-endorphin levels were not significantly affected by genistein at any dietary concentration. However, the 1250 ppm genistein concentration significantly increased hypothalamic vasopressin content.
Butler et al. investigated the effects of prenatal and postnatal exposure to the endocrine-disrupting chemicals bisphenol A (BPA) and genistein on gene expression and miRNA profiles in the hippocampus and hypothalamus of California mice and assessed their correlation with behavioural and metabolic outcomes.(Reference Butler, Long and Kinkade77) California mice dams were fed diets containing 5 mg/kg of BPA, 50 mg/kg of BPA, 250 mg/kg of genistein, or a control diet free of phytoestrogens during mating, gestation, and lactation. Offspring were subjected to social and metabolic tests in adulthood. Hippocampal and hypothalamic tissues were analysed for candidate mRNA and miRNA expression to identify molecular changes induced by 1-ethyl-3-3-dimethylaminopropyl carbodiimide (EDC) exposure. The gene expression analysis revealed increased hippocampal arginine vasopressin, BDNF, Dnmt3a, Gnrh, Kiss 1, and Lepr in the genistein-treated group, while the miRNA analysis showed decreased miR-7a and miR-153 in genistein-exposed hippocampi. An integrative analysis linked these molecular changes to observed behavioural and metabolic disruptions, suggesting enduring neurobehavioral impacts of early-life EDC exposure. A similar study by Kaur et al. investigated the effects of developmental exposure to BPA and genistein on global miRNA profiles in the hypothalamus and their correlations with behavioural and metabolic changes in California mice.(Reference Kaur, Kinkade and Green78) Mice were fed one of five diets: control, 250 mg/kg genistein, 5 mg/kg BPA, 50 mg/kg of high-dose BPA, or combination of genistein and BPA. Offspring were assessed using metabolic measures, including indirect calorimetry and body composition analysis. Hypothalamic tissues were collected in adulthood for small RNA sequencing to identify differentially expressed miRNAs and potential mRNA targets linked to observed phenotypic changes. Developmental exposure to BPA and/or genistein led to significant alterations in the hypothalamic miRNA profiles. The integrative analysis demonstrated correlations between differentially expressed miRNAs and behavioural and metabolic traits, highlighting miR146 as a potential biomarker for EDC-induced neurobehavioral disruptions.
Fisetin
Fisetin is a flavonoid with a flavone backbone and hydroxyl groups, which contribute to its strong antioxidant properties.(Reference Jash and Mondal79) It contains two benzene rings connected by a heterocyclic pyran ring with a ketone group at the 4-position. Fisetin is derived from fruits and vegetables including strawberries, apples, onions, and kale and functions to regulate signalling pathways associated with cellular anti-inflammatory and antioxidant responses in various neurodegenerative disease models. Treatment with fisetin prevents phosphorylation of NF-κB and attenuates Aβ-associated neuroinflammation.(Reference Ahmad, Ali and Park80) Treatment with fisetin also attenuates the phosphorylation of AkT/mechanistic target of rapamycin (mTOR), thereby regulating downstream targets of inflammatory and apoptotic proteins in the hippocampus and cortex.(Reference Khatoon, Agarwal and Samim81) In addition, this flavonoid regulates the expression of toll-like receptor-4, IL-1β, IL-6, and TNF-α via NF-κB.(Reference Khatoon, Agarwal and Samim81) Similarly, fisetin decreased the expression of pro-apoptotic Bax and caspase-3 via regulation of the Nrf2/ARE pathway in a traumatic brain injury model.(Reference Zhang, Wang and Zhou82) An in vitro Parkinson’s model suggested that fisetin improves mitochondrial bioenergy via regulation of the Akt1-GSK3B signalling pathway.(Reference Ay83)
Mehra et al. investigated the neuroprotective effects of fisetin on neurodevelopmental damage caused by prenatal exposure to valproic acid in a rodent model of autism.(Reference Mehra, Ahsan and Sharma84) The research specifically targeted the mitochondria-regulated canonical Wnt signalling pathway, blood-brain barrier integrity, and apoptotic pathways. To induce autism-like symptoms, pregnant Wistar rats administered a single intraperitoneal dose of 400 mg/kg of valproic acid on GD 12.5. From GD 13 until delivery, fisetin was given orally at a dose of 10 mg/kg per day. Fisetin treatment significantly reversed developmental delays caused by prenatal valproic acid exposure. Fisetin also prevented valproic acid-induced loss of complexes I–IV activity suggesting mitochondrial protection. Additionally, treatment with fisetin helped protect blood-brain barrier integrity against valproic acid exposure. Furthermore, fisetin downregulated caspase 3 expression and prevented valproic acid-induced apoptosis in cerebrum and cerebellum. Histological analysis revealed that fisetin preserved the neuronal architecture, including the Purkinje cells in the cerebellum.
Oral administration of fisetin modulated the expression of genes and proteins involved in synaptic transmission and plasticity in the hippocampus of rat offspring exposed to prenatal methylmercury (MeHg).(Reference Jacob and Sumathi85) Pregnant rats were divided into four groups: control, 1.5 mg/kg of MeHg, MeHg + 30 mg/kg of fisetin, and fisetin-only. MeHg exposure significantly reduced the expression of key genes involved in synaptic plasticity and neurotransmitter secretion in the hippocampus, including neurogranin, dendrin, Stx1A, and complexin-2, whereas fisetin administration prevented MeHg-induced alteration in gene expression. Furthermore, treatment with fisetin prevented the loss of BDNF and glial cell line-derived neurotrophic factor against MeHg challenges suggesting its role supporting synaptic plasticity. In addition, fisetin inhibited reactive astrogliosis, evidenced by reduced glial fibrillary acidic protein (GFAP) expression, a marker of astrocyte activation. Similarly, oral administration of fisetin regulated apoptotic pathways and protected the hippocampus of rat offspring against prenatal MeHg.(Reference Jacob and Thangarajan86) Pregnant Wistar rats were divided into groups receiving 1.5 mg/kg body weight of MeHg, fisetin at 10–70 mg/kg doses, or a combination of MeHg and fisetin from GD 5 until delivery. MeHg exposure led to significant mitochondrial dysfunction, including increased oxidative stress, reduced ETC activity, and elevated apoptosis markers. Fisetin treatment effectively reversed these effects and restored mitochondrial antioxidant levels including those of SOD, catalase, and GPx. The treatment also inhibited cytochrome c release and caspase-3 activation, mitigating apoptotic pathways. This study concluded that fisetin provided neuroprotection against developmental MeHg toxicity by regulating mitochondrial health, apoptotic pathways, and signalling molecules in the hippocampus. Moreover, fisetin was shown to improve behavioural deficits associated with MeHg-induced neurotoxicity in offspring.(Reference Jacob and Thangarajan87) MeHg is a developmental neurotoxin that leads to cognitive and motor deficits. In this study, pregnant Wistar rats were treated with 1.5 mg/kg MeHg or MeHg combined with 10 mg/kg or 50 mg/kg fisetin from GD 5 to parturition. MeHg exposure caused significant neurobehavioral deficits, including impaired motor coordination, learning, and memory, as well as increased depression-like behaviours in offspring. MeHg increased lipid peroxidation and protein carbonyl levels, decreased antioxidant enzymes, and disrupted thiol content and membrane-bound ATPase activities in the brain. However, fisetin co-administration mitigated these effects, lowering oxidative stress, and preventing the loss of antioxidant enzyme activities and ATPase function. Histological analysis showed extensive neuronal damage, including gliosis and pyknotic nuclei in the hippocampus and cerebellum in MeHg treated groups. However, neuronal damage was significantly alleviated by fisetin treatment. The fisetin group improved behavioural outcomes with enhanced motor coordination, memory, and reduced depression-like behaviour.
Epigallocatechin gallate (EGCG)
Epigallocatechin gallate (EGCG) is a polyphenolic catechin compound with a 3-ring system consisting of two benzene rings connected by a heterocyclic pyran ring. It features multiple hydroxyl groups and a gallate ester, contributing to its strong antioxidant properties.(Reference Minnelli, Galeazzi and Laudadio88) EGCG is a branched polyphenol found abundantly in green tea and various natural produce.(Reference Mokra, Joskova and Mokry89) EGCG is one of many chemical compounds belonging to the catechins family, which are known for their antioxidant activities. Specifically, EGCG scavenges reactive oxygen and metal ion species and prevents cell damage-induced oxidation.(Reference Wnuk, Zwolak and Kochanowicz90,Reference An, Qi and Huang91) The neuroprotective mechanism involves effective suppression of inflammation and oxidative stress. The exposure of BV2 microglial cells to CoCl2 increases the expression of inflammatory mediators such as IL-6, iNOS, and COX-2, and promotes the expression of hypoxia-inducible factor (HIF)-1α and the production of ROS.(Reference Kim, Seong and Kim92) EGCG ameliorates the inflammatory responses by inhibiting the NF-κB pathway and suppresses HIF-1α and ROS production, thereby decreasing IL-6 production. Furthermore, EGCG activates the Nrf2/HO-1 pathway to enhance the cellular defence mechanisms against oxidative stress and inhibits the cleavage of PARP and caspase-3 to prevent hypoxia-induced cell death.
The effect of EGCG on genetic- and developmental brain disorders has been reported. Souchet et al. investigated the impact of prenatal treatment with EGCG-enriched green tea extract on Down syndrome mouse models.(Reference Souchet, Duchon and Gu93) Transgenic mBACtgDyrk1a and Dp(16)1Yey mice were used to investigate developmental and cognitive defects associated with an overexpression of DYRK1A, a kinase implicated in Down syndrome. Pregnant mice were administered food pellets containing 600 mg/kg of EGCG-enriched extract, equivalent to a daily dose of 50 mg/kg EGCG, throughout gestation, with some groups continuing treatment until adulthood. The control groups received standard food pellets. Offspring brain tissues were analysed for inhibitory and excitatory synaptic markers, including GAD67, vesicular GABA transporter 1, and vesicular glutamate transporter 1, using immunohistochemistry, immunoblotting, and stereological techniques. Prenatal EGCG treatment effectively normalised GAD67 expression and interneuron density in the hippocampi and cortexes of mBACtgDyrk1a and Dp(16)1Yey mice. These findings highlight the necessity of prenatal intervention for long-term correction of GABAergic pathway disruptions and associated cognitive deficits in Down syndrome models. The results support the potential of ECGC as a therapeutic agent targeting DYRK1A during early neurodevelopment. Additionally, the neuroprotective and cardioprotective effects of EGCG in a Williams-Beuren Syndrome (WBS) was reported in a mouse model.(Reference Ortiz-Romero, Borralleras and Bosch-Morató94) CD mice, representing the WBS model, were treated with EGCG (2.5–3.0 mg/day) dissolved in drinking water, at various developmental stages, including prenatal, postnatal, and adulthood. Behavioural tests assessed short-term memory, sociability, and anxiety-like behaviours, while histological and molecular analyses evaluated cardiac hypertrophy and hippocampal function. EGCG treatment ameliorated short-term memory deficits in the CD mice, evidenced by improved performance in the novel object recognition test. Cardioprotective effects were verified through a significant reduction in cardiac hypertrophy and normalisation of cardiomyocyte size. EGCG prevented alteration of Nrf2 associated with WBS. However, no impact was observed on sociability or anxiety-like behaviour and ECGC did not fully correct spatial working memory deficits or hippocampal structural abnormalities.
Discussion
Metabolism of polyphenols
Polyphenols undergo complex pharmacokinetic processes that determine their biological availability and potential neurodevelopmental effects. Following maternal ingestion, most polyphenols are absorbed in the small intestine, often requiring hydrolysis of glycosylated forms by intestinal enzymes or gut microbiota prior to uptake.(Reference Manach, Scalbert and Morand95,Reference Manach, Williamson and Morand96) Once absorbed, they are subject to extensive phase II metabolism in the intestinal epithelium and liver, resulting in conjugated metabolites with altered biological activity.(Reference Manach, Scalbert and Morand95) These metabolites circulate bound to plasma proteins and are distributed to maternal tissues, with distribution patterns influenced by molecular size, polarity, and lipophilicity.(Reference Manach, Scalbert and Morand95,Reference Stromsnes, Lagzdina and Olaso-Gonzalez97)
A critical factor in the maternal-foetal context is polyphenol bioavailability, which is often limited by poor absorption, rapid metabolism, and efficient excretion.(Reference Manach, Scalbert and Morand95,Reference Manach, Williamson and Morand96) Nonetheless, both in vitro placental perfusion studies and in vivo rodent experiments indicate that certain polyphenols and their metabolites can cross the placental barrier.(Reference Stromsnes, Lagzdina and Olaso-Gonzalez97) The extent of placental transfer is determined by physicochemical properties such as molecular weight, conjugation status, and interaction with specific placental transporters.(Reference Manach, Scalbert and Morand95) For example, maternal resveratrol supplementation in a murine model of uteroplacental insufficiency resulted in resveratrol detection in foetal plasma and placental tissue, improving foetal growth and reducing oxidative stress, thereby providing experimental evidence of direct foetal exposure to bioactive polyphenols.(Reference Poudel, Stanley and Rueda-Clausen98) Similarly, maternal exposure to quercetin throughout gestation led to quercetin accumulation in the foetus, affecting iron storage and reducing oxidative DNA damage, indicating foetal tissue exposure to bioactive conjugates.(Reference Vanhees, Godschalk and Sanders99) In addition, polyphenol-rich sources such as pomegranate juice, resulted in detectable levels of ellagic acid in neonatal circulation, providing experimental evidence of placental transfer of bioactive polyphenolic metabolites.(Reference Loren, Seeram and Schulman100)
Importantly, the pharmacokinetic profile of polyphenols differs substantially depending on the route of administration. Oral administration, which most closely reflects human dietary intake, is characterised by limited bioavailability due to extensive first-pass metabolism in the intestine and liver, as well as rapid conjugation to glucuronide and sulphate metabolites.(Reference Atkinson, Stanescu and Frampton101) In contrast, intraperitoneal administration bypasses initial hepatic metabolism, often resulting in higher systemic exposure and increased tissue concentrations in rodent models.(Reference Zhang, Shen and Huang102) These route-dependent differences likely contribute to variability in reported neurodevelopmental outcomes and complicate direct extrapolation of rodent dosing regimens to human pregnancy, where exposure occurs almost exclusively through oral intake.
These pharmacokinetic considerations are essential for interpreting maternal supplementation studies, as the efficacy of polyphenols depends not only on their intrinsic antioxidant, anti-inflammatory, or neurotrophic potential, but also on their ability to reach the foetal brain in biologically relevant forms. Understanding these dynamics underscores the importance of dosing strategies, timing of administration, and selection of specific polyphenol sources in both experimental designs and translational applications. While these preclinical findings encouraging, it is important to note that certain pharmacokinetic characteristics of polyphenols, low oral bioavailability due to limited intestinal absorption, extensive first-pass metabolism, and rapid excretion,(Reference Manach, Williamson and Morand96,Reference Ciupei, Colişar and Leopold103) limited the blood–brain barrier,(Reference Figueira, Garcia and Pimpão104) and individual variability in metabolism(Reference Di Lorenzo, Colombo and Biella105) can influence foetal exposure to active compounds. In addition, factors such as placental transport efficiency and the timing of supplementation can further impact the extent of active metabolite delivery to the foetus.(Reference Sánchez-Martínez, Valdés and Gallego106) These factors reflect intrinsic properties of polyphenols and underscore the importance of optimising formulation, dosage, and timing to maximise translational potential.
Energy metabolism
Maternal HFD impacts foetal energy metabolism.(Reference Wang, Yu and Tiao107) Maternal obesity alters mitochondrial metabolism in oocytes.(Reference Elias-Lopez, Vazquez-Mena and Sferruzzi-Perri108) The excessive oxidation of lipids generates ROS, which damages mitochondrial membranes. The loss of the mitochondrial membrane potential impairs ATP production, ultimately disrupting normal embryonic development.(Reference Elias-Lopez, Vazquez-Mena and Sferruzzi-Perri108) Oocytes from maternal HFD mice showed defects in mitochondrial morphology and chromosome alignment, and the foetal brain exhibited abnormalities in ventricles and the choroid plexus.(Reference Luzzo, Wang and Purcell109) Although the mechanism of maternal HFD on mitochondria in the foetal brain is less studied, HFD is known to lower mitochondrial density, impair ETC function, including producing changes in levels of Complex I, II, and III, and increase the ADP/ATP ratio in the myocardium.(Reference Chen, Li and Zhang110) Proteomic analyses indicate a widespread decline in mitochondrial protein expression, including that of ATP synthase subunits α and γ and ADP/ATP translocase 1, suggesting that these changes reflect broader mitochondrial dysfunction.(Reference Chen, Li and Zhang110)
Additionally, obesity decreases the translation of ATP synthase β-subunit (β-F1-ATPase) in human skeletal muscle(Reference Tran, Langlais and Hoffman111); the lack of change in β-F1-ATPase mRNA levels indicates that post-transcriptional regulatory mechanisms play a role in the inhibition of protein synthesis.(Reference Tran, Langlais and Hoffman111) Increased levels of non-esterified fatty acids in obesity further suppress β-F1-ATPase translation.(Reference Tran, Hanavan and Campbell112) Additionally, miR-127-5p has been identified as a key regulatory factor that inhibits β-F1-ATPase translation in obesity. Notably, HFD upregulation of uncoupling protein 3 expression in skeletal muscle(Reference Chou, Cha and Jung113,Reference Matsuda, Hosoda and Itoh114) suggests defence mechanisms against obesity. The c-subunit of ATP synthase has been shown to function as an uncoupling channel.(Reference Alavian, Beutner and Lazrove115) Therefore, evaluating the ratios of the c-subunit and other subunits of ATP synthase may be important.
Various polyphenols, including resveratrol, quercetin, curcumin, EGCG, and genistein, regulate ATP synthase activity.(Reference Gledhill, Montgomery and Leslie116,Reference Zheng and Ramirez117) Resveratrol binds to ATP synthase and prevents the rotation of the γ-subunit, potentially inhibiting both the synthetic and hydrolytic functions of the enzyme.(Reference Gledhill, Montgomery and Leslie116,Reference Zheng and Ramirez117) Quercetin and piceatannol, polyphenols that are structurally similar to resveratrol, bind to the hydrophobic pocket around the γ-subunit and inhibit the rotary mechanism. Although such inhibition may be viewed as detrimental under normal conditions, in the context of obesity or a high-fat diet, reduced ATP output may prevent further metabolic imbalance by mitigating excess energy availability. Additionally, by inducing a modest energy deficit in cells with surplus fuel, polyphenols may prevent the overproduction of ROS associated with nutrient overload, thus reducing oxidative stress and protecting mitochondrial integrity. Future research may elucidate whether this partial inhibition of ATP synthase acts as a hormetic stress that confers cellular benefits by strengthening adaptive responses.
Given these findings, further research is needed to explore whether ATP synthase inhibition by polyphenols represents a metabolic adaptation rather than solely a pathological consequence. Future studies should investigate whether the targeted modulation of ATP synthase can be therapeutically leveraged to regulate mitochondrial function in metabolic disorders. The dual effects of polyphenols — inhibition of ATP synthase and enhancement of mitochondrial efficiency — suggest a complex regulatory mechanism that requires deeper elucidation. Understanding this intricate balance may allow optimising the therapeutic potential of polyphenols to manage obesity and metabolic diseases and provide new insights into the role of polyphenols in mitochondrial function and energy metabolism.
Antioxidants
A central mechanism through which maternal polyphenol supplementation exerts neuroprotective effects is the enhancement of endogenous antioxidant defence systems.(Reference Cao, Zheng and Xu36,Reference Anachuna, Moke and Iyare55,Reference August, Klein and Grings64,Reference Yu, Pan and Zhang70) These polyphenols, especially resveratrol and ferulic acid, improve redox regulation by activating Nrf2 and upregulating downstream targets such as HO-1 and NQO1. The antioxidant effects of ferulic acid were abolished in Nrf2 knockout models, indicating that the protective effects of this polyphenol are dependent on this transcription factor.(Reference Yu, Pan and Zhang70) Curcumin’s modulation of oxidative pathways through its moderation of CYP2E1 expression and miR-335(Reference Labib43) may represent alternative regulatory mechanisms beyond the Nrf2 pathway.(Reference Xu, Yu and Sang118)
Building upon these Nrf2-related mechanisms, other polyphenols such as quercetin and fisetin have demonstrated antioxidant activity through the regulation of classical enzymes such as SOD, catalase, and glutathione peroxidase. Quercetin has been associated with improved GSH levels and lower MDA maternal immune activation models.(Reference Anachuna, Moke and Iyare55) In foetal brain models, fisetin enhanced GSH and NO, supporting its role in oxidative stress control.(Reference Jacob and Thangarajan87) Additionally, fisetin restored mitochondrial complex I–IV activity and inhibited ROS generation.(Reference Mehra, Ahsan and Sharma84) Naringin has demonstrated time- and region-specific redox effects in the brains of offspring, particularly in the cerebellum and striatum, where ROS levels, GSH content, and lipid peroxidation markers vary across developmental stages.(Reference August, Klein and Grings64) Although these effects were context-dependent, naringin supplementation was also associated with increased activity of thioredoxin reductase and antioxidant enzymes in the early postnatal period.(Reference August, Maurmann and Saccomori62)
Expanding on these enzymatic mechanisms, recent studies suggest an expanded mechanistic perspective that highlights mitochondrial biogenesis regulation via the PGC-1α pathway as a novel antioxidant mechanism. Polyphenols such as resveratrol and fisetin may stimulate PGC-1α, a transcriptional coactivator that regulates mitochondrial biogenesis and antioxidant defence. In a Parkinson’s disease model, activation of PGC-1α by resveratrol contributed to improved mitochondrial function and reduced oxidative damage.(Reference Chen, Yang and Lin119) Incorporating this mechanism into the broader Nrf2-based antioxidant network may provide a more comprehensive understanding of how polyphenols confer mitochondrial protection during prenatal development. Beyond transcriptional regulation, Nrf2 influences mitochondrial redox balance. Curcumin enhances mitochondrial antioxidant defences by upregulating Nrf2 and increasing mitochondrial SOD2 expression in rodent studies.(Reference Park, Sohn and Koh40) Similarly, naringenin enhanced mitochondrial ETC activity and reduced ROS in models of mercury-induced neurotoxicity.(Reference Krishna Chandran, Christina and Das120) These findings indicate that the antioxidant actions of specific polyphenols extend to the mitochondrial compartment and may contribute to neuroprotection during critical periods of development.
Apoptosis
Apoptosis plays a pivotal role in shaping neural architecture during development. Although necessary for normal remodelling, excessive apoptosis induced by prenatal stress can lead to long-term neurodevelopmental impairment. Several polyphenols, including resveratrol, curcumin, and fisetin, mitigate these effects by modulating apoptotic signalling. Resveratrol and fisetin increase anti-apoptotic B-cell leukaemia/lymphoma (Bcl)-2 protein while decreasing pro-apoptotic proteins such as cytochrome c and cleaved caspase-3, particularly under metabolic or hypoxic stress.(Reference Dumont, Sanchez and Repond35,Reference Mehra, Ahsan and Sharma84) These findings highlight polyphenols’ shared ability to protect mitochondrial membrane integrity, thus preventing the release of pro-apoptotic proteins. Although other proteins involved in apoptosis have not been thoroughly studied with polyphenols, caspase-3 is known to drive post-translational proteolytic cleavage of B-cell lymphoma-extra Large (Bcl-xL), which converts anti-apoptotic full-length Bcl-xL to pro-apoptotic fragmented Bcl-xL.(Reference Ofengeim, Chen and Miyawaki121,Reference Park and Jonas122) Thus, the prevention of caspase-3 activity or production by polyphenol may help maintain anti-apoptotic Bcl-xL.
Additionally, the mitochondrial unfolded protein response (UPRmt) plays a role in a stress-adaptive pathway that preserves mitochondrial proteostasis and prevents the release of apoptogenic factors. Activation of UPRmt enhances the expression of mitochondrial chaperones and proteases, mitigating protein misfolding and sustaining mitochondrial membrane potential.(Reference Torres, Fleischhart and Inestrosa123) Tetrahydrocurcumin, a metabolite of curcumin, has been shown to activate UPRmt via the PGC-1α/activating transcription factor 5 (ATF5) axis in cardiac tissue.(Reference Zhang, Tan and Zhang124) Notably, UPRmt is associated with the expression of Bax and Bcl-2 through heat shock protein regulation in a traumatic brain injury model.(Reference Xiaowei, Qian and Dingzhou125) These molecular changes coincide with improvements in mitochondrial membrane potential, emphasising the coordinated role of mitochondrial quality control in modulating neuronal apoptosis.(Reference Sun, Ding and Wang126) Fiorese et al. further support the importance of ATF5 in UPRmt activation by highlighting its potential role in maintaining mitochondrial integrity under stress.(Reference Fiorese, Schulz and Lin127) This mitochondrial protection may work in conjunction with antioxidant and neurotrophic signalling pathways, reinforcing the notion that anti-apoptotic actions are not isolated but embedded within the broader neuroprotective framework of maternal polyphenol intake during gestation.
Anti-inflammation
Inflammation plays a transient and tightly regulated role during normal neurodevelopment and contributes to synaptic pruning, neuronal differentiation, and tissue remodelling. However, under pathological conditions such as maternal stress, immune activation, or high-fat diet exposure, sustained inflammation can disrupt these critical developmental processes and cause long-term cognitive and behavioural impairment.(Reference Zilkha, Chuartzman and Fishman128,Reference Rincel, Lépinay and Janthakhin129) A growing body of evidence indicates that maternal dietary polyphenols, particularly resveratrol, curcumin, and quercetin regulate inflammatory responses in offspring. Resveratrol has demonstrated anti-inflammatory effects in several prenatal models. In a perinatal asphyxia model, maternal resveratrol administration significantly decreased the concentration of IL-1β and TNF-α and the expression of S100B in the foetal brain, indicating attenuation of astroglial activation.(Reference Isac, Panaitescu and Spataru34) Similarly, in a maternal high-fat diet model, resveratrol supplementation downregulated of MCP-1 and TGF-β1 in foetal brain tissue.(Reference Izquierdo, Palomera-Ávalos and Pallàs31) Quercetin also acts on glial activation pathways. In a maternal immune activation model, Iba-1 and GFAP immunoreactivity in the foetal cortex and hippocampus were decreased following quercetin administration.(Reference Abbasi, Ghavami-Kia and Davoodian54) These changes reflect the suppression of both microglial and astrocytic responses. In a stress-based model, Toumi et al. reported low maternal corticosterone, suggesting a secondary effect on inflammation via neuroendocrine signalling.(Reference Toumi, Merzoug and Baudin53) Curcumin exhibits anti-inflammatory effects in a dose-dependent manner. In the foetal hippocampus, a low-dose exposure decreased TNF-α mRNA expression, whereas high doses were ineffective.(Reference Nakahara, Masubuchi and Takashima46) Furthermore, in a prenatal ethanol exposure model, curcumin downregulated key transcriptional mediators of inflammation such as COX-2 and FBJ murine osteosarcoma viral oncogene homolog (c-Fos).(Reference Labib43)
Recent findings provide mechanistic insights into how polyphenols regulate inflammation. In particular, the SIRT1-mediated control of HMGB1 signalling has emerged as a promising pathway. Resveratrol is shown to activate SIRT1 and suppresses HMGB1 translocation, thereby disrupting the amplification of NF-κB signalling associated with persistent neuroinflammation.(Reference Lu, Jin and Lu130) This mechanism extends the known cytokine-suppressing effects of polyphenols because it influences the resolution phase of inflammation. Moreover, polyphenols exert anti-inflammatory effects by regulating inflammasome activity by activating AMPK, which in turn inhibits the NOD-like receptor family, pyrin domain containing 3 inflammasome complex through Unc-51-like kinase 1-dependent signalling.(Reference Xiong, Zhou and Tang131) This inhibition attenuates neuroinflammatory responses by suppressing the production of pro-inflammatory cytokines such as IL-1β.
Growth factors
Polyphenol supplementation during pregnancy enhances foetal brain development through the experimentally confirmed modulation of neurotrophic and synaptic signalling. Among the compounds studied, resveratrol, curcumin, fisetin, and EGCG have consistently demonstrated upregulation of BDNF and downstream effectors via well-defined intracellular pathways. Resveratrol administered under maternal high-fat diet or stress conditions upregulated BDNF and neurodevelopmental genes such as GAP43 and activated the AMPK pathway to promote synaptic plasticity and neuroprotection.(Reference Cao, Zheng and Xu36) Curcumin enhanced synaptic maturation by stimulating β-catenin signalling in the foetal hippocampus(Reference Zhang, Yang and Gong132) and modulating miR-335, indicating transcriptional and epigenetic regulation under prenatal ethanol exposure.(Reference Labib43) Fisetin increased BDNF expression and upregulated hippocampal genes associated with synaptic function in prenatal neurotoxicity models, indicating structural preservation under developmental stress.(Reference Jacob and Sumathi85) In a prenatal Down syndrome model, EGCG corrected early synaptic deficits by restoring BDNF levels and normalised glutamatergic synaptic protein expression.(Reference Ortiz-Romero, Borralleras and Bosch-Morató94)
A recent study utilising a Pb-induced cognitive impairment model demonstrated that resveratrol treatment ameliorated hippocampal BDNF deficiency and improved spatial memory performance through the activation of upstream metabolic regulators. Specifically, resveratrol promoted the nuclear localisation and phosphorylation of SIRT1 and partially restored the expression of AMPK and PGC-1α — both of which are implicated in mitochondrial biogenesis and antioxidant regulation.(Reference Zhang, Tu and Wang133) These effects coincided with significant upregulation of BDNF and tropomyosin receptor kinase B (TrkB) in the hippocampus. Curcumin has also been shown to activate the BDNF–TrkB–MAPK/PI3K–CREB signalling cascade in cultured rodent cortical neurons.(Reference Wang, Li and Xu134) In vitro experiments demonstrated that curcumin treatment increased BDNF expression and phosphorylation of TrkB, ERK, and Akt. These effects were abolished by TrkB-neutralising antibodies and specific inhibitors of MAPK and PI3K pathways. Additionally, CREB phosphorylation was suppressed by the same inhibitors, indicating that this intracellular cascade may mediate curcumin’s neurotrophic effects. Although these findings derive from an in vitro system, they provide strong molecular support for the proposed mechanism.
These findings suggest that polyphenols exert multi-layered effects involving transcriptional activation and regulation of developmental genes, ultimately targeting key molecular endpoints like BDNF, TrkB, CREB, and synaptic scaffold proteins. Such integrated actions support neural protection during critical periods of synaptogenesis and circuit formation.
Limitations
This review has several limitations. It remains uncertain whether maternal polyphenols exert their effects mainly through foetal programming during gestation or by promoting postnatal recovery. Although the general metabolic fate of polyphenols has been described, their specific delivery to the foetal brain, bioavailability, and regional distribution during pregnancy are poorly understood. Moreover, many studies describe antioxidant and neuroprotective effects only in general terms, without clearly describing the specific mechanisms involved. Information on the timing, duration, and route of supplementation was also inconsistently reported across studies, limiting the ability to draw more detailed conclusions. Furthermore, most included studies relied on purified compounds, whereas relatively few examined polyphenol-rich food sources, which are more relevant for translational dietary applications.
Although the focus of this review is on rodent studies, it is also important to consider the translational aspect for human pregnancy. At present, clinical evidence remains sparse and indirect. Small-scale observational and exploratory studies, such as pomegranate juice supplementation in pregnancies complicated by intrauterine growth restriction, have reported encouraging neonatal magnetic resonance imaging outcomes.(Reference Ross, Cherkerzian and Mikulis20,Reference Hamlbar, Mirghafourvand and Shaseb135) However, these studies were limited to high-risk populations and did not directly assess offspring neurodevelopment. Reports of maternal supplementation with compounds such as resveratrol, EGCG, or curcumin have primarily addressed maternal outcomes, such as blood pressure modulation, folate stability, or postpartum mood, rather than foetal brain development.(Reference Hinojosa-Nogueira, Romero-Molina and González-Alzaga136–Reference Johnson, Siblini and Al-Hendy138) Moreover, uncertainties remain regarding the appropriate human dose, duration of intake, and long-term safety of polyphenol supplementation during pregnancy as well as evidence of maternal toxicity or adverse side effects. Collectively, these limitations highlight the need for well-designed clinical trials in broader populations, including food-based interventions, to evaluate neurodevelopmental efficacy and translational feasibility.
In addition, dose selection across studies was often based on precedent rather than pharmacokinetic or dose–response justification. As a result, there is currently no consensus regarding an effective or optimal dose for individual polyphenols, and the absence of systematic dose–response designs limits the ability to define a therapeutic window. Furthermore, although multiple routes of administration were employed across studies (oral, intraperitoneal), no study directly compared different routes within the same experimental framework. This precludes conclusions regarding route-dependent efficacy, foetal exposure, or translational relevance to human dietary intake. Safety assessments were also limited. Only a small subset of studies reported biochemical markers, histological analyses, or maternal and foetal tissue evaluations, and few directly examined dose-dependent toxicity or long-term adverse outcomes in either dams or offspring. Finally, substantial variability in behavioural paradigms, outcome measures, and analytical approaches further restricts cross-study comparability and limits quantitative interpretation of neurobehavioral effects.
Future directions and research recommendations
Future research should include well-designed dose–response studies to define therapeutic dosage for individual polyphenols during pregnancy. Such studies should systematically evaluate multiple dose levels, incorporate gestational timing, and relate maternal dosing to foetal plasma and brain concentrations in order to distinguish effective, subtherapeutic, and potentially adverse exposure ranges. Comparative evaluation of routes of administration represents another research gap. Direct comparisons between oral and parenteral delivery within the same experimental models would clarify the impact of bioavailability, first-pass metabolism, and systemic exposure on foetal neurodevelopmental outcomes, and would substantially enhance translational relevance.
Delivery strategies such as encapsulation approaches, including nanoparticles, liposomes, polymer-based carriers, or prodrug formulations, may improve bioavailability and reduce the need for high maternal doses. Future studies should evaluate whether such delivery platforms modify dose–effect relationships or mitigate potential toxicity, particularly in the maternal–foetal context. Future investigations should routinely incorporate safety-focused assessments of maternal and offspring toxicity, including hepatic and renal biochemical markers, body weight trajectories, placental and foetal histopathology, and long-term neurobehavioral follow-up. These measures are critical for establishing both efficacy and safety profiles of maternal polyphenol supplementation as well as for clinical translation.
Finally, future studies should systematically incorporate sex as a biological variable, reporting the number of offspring per sex and, where applicable, estrous cycle stage in females. Standardisation of behavioural paradigms and inclusion of translational biomarkers would further strengthen comparability across studies and facilitate progression toward human clinical trials.
Conclusion
This review summarises intervention strategies in rodent models (Table 1), and highlights the multifaceted neuroprotective potential of maternal polyphenol supplementation on offspring (Table 2). By targeting key molecular pathways such as Nrf2/ARE, NF-κB, CREB/BDNF, and Akt/mTOR, polyphenols including resveratrol, curcumin, quercetin, and fisetin exert overlapping effects on redox regulation, mitochondrial protection, anti-inflammatory signalling, apoptosis regulation, and neurotrophic support. These mechanisms not only mitigate the detrimental effects of maternal stress such as high-fat diets, immune activation, hypoxia, and toxin exposure, but also enhance neurodevelopmental trajectories under normal conditions, suggesting both preventive and therapeutic roles.
The efficacy of polyphenols is influenced by dose, timing of exposure, and maternal–foetal metabolic variability. Although the current evidence from animal models is promising, translation into human health recommendations requires caution. Future studies should address long-term safety, optimal delivery strategies, and the potential for interaction with other dietary components. Investigating population-specific factors and developmental timing windows is essential for tailoring interventions. In summary, polyphenols represent a compelling, biologically plausible strategy to support foetal brain development through the nutritional modulation of molecular resilience mechanisms.
Author contribution
KW was involved in drafting the original manuscript, interpretation of findings, and editing of the manuscript. MG was involved in drafting the original manuscript, interpretation of findings. GK was involved in drafting the original manuscript, interpretation of findings. RB was involved in drafting the original manuscript, interpretation of findings. EP was involved in drafting the original manuscript, interpretation of findings. KR was involved in drafting the original manuscript, interpretation of findings. EAJ was involved in interpretation of findings and editing of the manuscript. YB was involved in interpretation of findings and editing of the manuscript. HP was involved in interpretation of findings and editing of the manuscript.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
The authors declare none.