Introduction
A 35-year-old female was referred in June 2023 to a regional emergency department by a community ophthalmologist, with bilateral papilledema. She complained of subacute-onset, position-dependent neck and low-back pain with transient visual obscurations. Symptoms began 2 years prior, after a mechanical fall from standing height. Weeks after the fall, she developed mechanical neck and low-back pain with burning and paresthesias to the right buttock and posterior thigh. Over months, her pain progressed such that she could not tolerate lying flat and slept in a semi-recumbent position. One month before presenting, she developed new painless transient visual obscurations, provoked by postural changes. She also experienced intermittent neck and back paresthesias during neck flexion and poorly localized numbness and paresthesias in both hands. She denied pulsatile tinnitus, fixed visual field defect or headache.
Review of systems was unremarkable. She was not pregnant and had no significant past medical history. She was of normal body habitus, denied hormonal contraception use and never used tetracycline or isotretinoin. She used no other medications, supplements or topical treatments. She was self-employed as a tattoo artist and in a long-standing monogamous relationship. She used cannabis daily, consumed minimal alcohol and did not use tobacco or illicit drugs. Family history was noncontributory.
On examination, she appeared uncomfortable and frequently changed positions. She had reduced neck and spine range of motion, accompanied by pain and difficulty tolerating supine and seated positions. There was bilateral papilledema on direct fundoscopy, but no visual field defect, color vision loss or impaired visual acuity. No focal neurological deficits were elicited. Considering the patient’s transient visual obscurations provoked by changes in intracranial pressure, absent fixed visual defect and presence of papilledema, we were concerned about increased intracranial pressure. Primary differential diagnoses were idiopathic intracranial hypertension and cerebral venous sinus thrombosis. Additional considerations were neurosarcoidosis, chronic arachnoiditis, obstructive mass lesion (e.g., glioma, metastases, lymphomatosis, disc herniation) and infection (e.g., tuberculosis).
An enhanced CT head in the emergency department revealed expanded Meckel’s caves, pituitary flattening and prominent optic nerve sheaths with flattening of the posterior globes and minimal protrusion of the optic nerve heads – all suggestive of intracranial hypertension. There was no evidence of cerebral venous sinus thrombosis. A diagnostic lumbar puncture was performed at the level of the iliac crest (L4–L5 space) with the patient in left lateral recumbent position. CSF opening pressure was 11.5 cm·H2O, and due to CSF hypercoagulability, we could obtain only 1 mL of viscous, clear yellow CSF (Figure 1). CSF analysis showed erythrocytosis (1663 × 106 cells/L; reference 0 × 106 cells/L) and hyperproteinemia (33.35 g/L; reference 0.15–0.40 g/L), with normal leukocytes (3 × 106 cells/L) and glucose (4.4 mmol/L). Cytology was not completed due to a normal leukocyte count. Findings of Froin syndrome – CSF hypercoagulability, erythrocytosis and hyperproteinemia – raised concern for a neuraxial obstructive lesion.
CSF from a bedside lumbar puncture. Opening pressure was 11.5 cm·H2O and yielded approximately 1 mL of clear yellow CSF with elevated erythrocytes (1663 × 106 cells/L) and total protein (33.35 g/L). CSF leukocyte count (3 × 106 cells/L; 82% lymphocytes) and glucose (4.4 mmol/L; plasma glucose 5.7 mmol/L) were within normal limits.

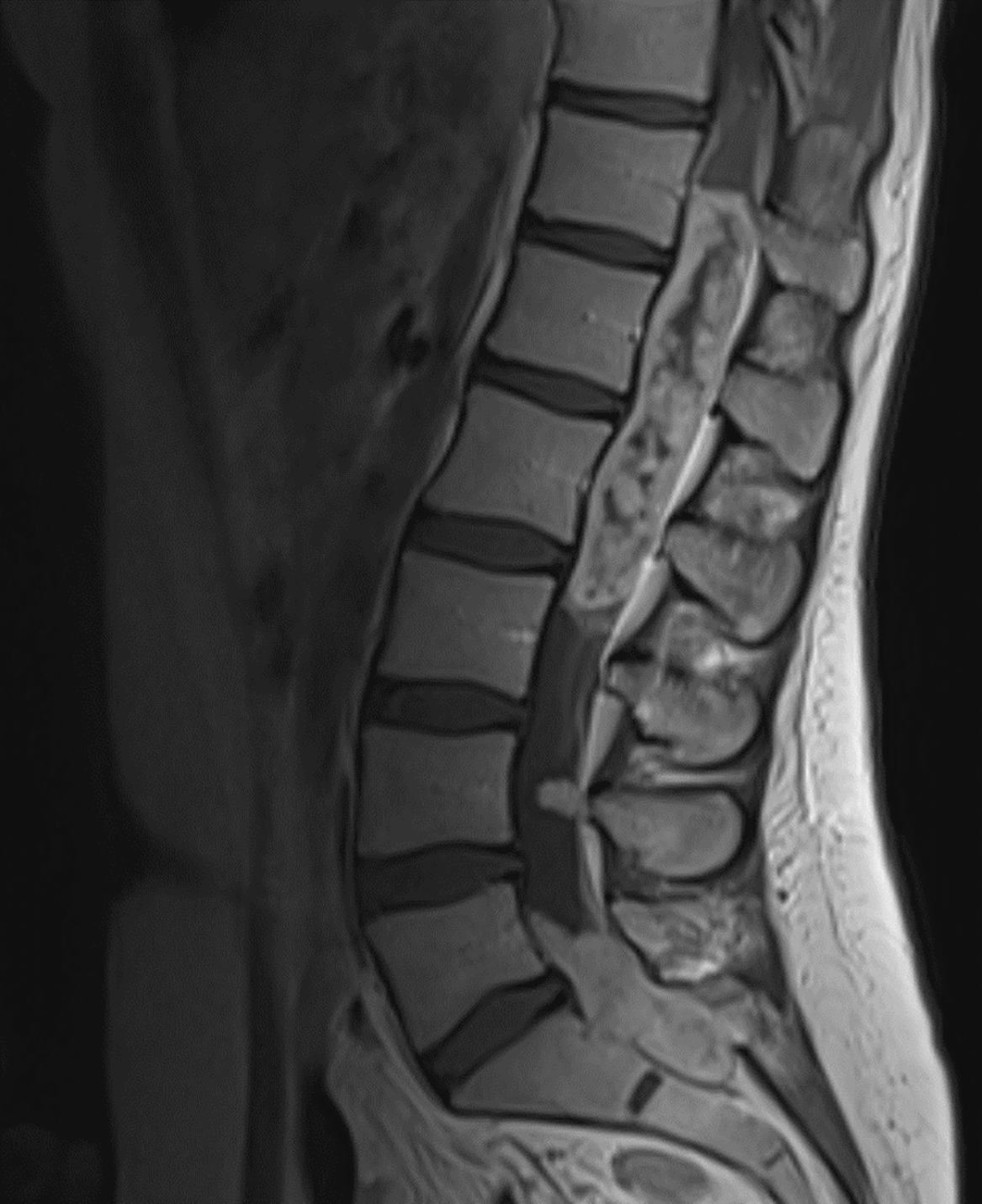
The patient was admitted for urgent neuraxial imaging and neurosurgical consultation. Enhanced spinal MRI demonstrated multiple heterogeneously enhancing intramedullary masses in the thoracolumbar and lumbosacral spine, with numerous enhancing nodules and linear enhancement in the cervical, thoracic and lumbar spinal canal (Figure 2). Imaging suggested myxopapillary ependymoma (ME) with spinal leptomeningeal dissemination. Brain MRI revealed bilateral optic nerve sheath prominence with increased T2 signal intensity, flattening of the posterior globes with bulging of the optic nerve heads and absent enhancement of the optic nerves or globes. The MR venogram exhibited minimal narrowing of the left transverse sinus. Findings were indicative of increased intracranial pressure without intracranial tumor dissemination or venous sinus thrombosis.
MRI of the lumbosacral spine. Post-gadolinium T1-weighted midsagittal image demonstrating a heterogeneously enhancing intramedullary mass centered on the conus medullaris and nerve rootlets of the cauda equina at T12–L3 levels within the spinal canal, as well as another heterogeneously enhancing mass within the spinal canal at L5–S2 levels. Imaging was suggestive of myxopapillary ependymoma. Additional images (not shown) demonstrated abnormal signal within the lower thoracic spinal cord reflecting edema and/or small syrinx and multiple other enhancing nodules and linear enhancement elsewhere within the cervical, thoracic and lumbar spinal canal with leptomeningeal tumor dissemination.

In July 2023, the patient underwent bilateral posterolateral T12–S2 laminectomy, decompression, partial tumor resection, duraplasty and dural repair. The procedure and postoperative period were uncomplicated, and there were marked improvements in visual symptoms, pain and mobility. Pathology was consistent with WHO Grade 2 ME. After reproductive counseling, she underwent spinal irradiation (5000 cGy in 25 fractions) in November 2023. Follow-up neurological assessments and surveillance imaging have revealed resolution of symptoms, papilledema and intracranial hypertension. There has been no tumor progression. She is receiving hormonal treatment for menopause and is functioning well.
ME is a slow-growing, intramedullary tumor of the lumbosacral spinal cord, conus medullaris, cauda equina and/or filum terminale, arising from ependymal glia. Reference Bertero, Ricci, Tampieri, Cassoni and Modena1 ME is a rare adult tumor, with an incidence of 0.6–1.0 per million patient-years. Reference Bates, Choi and Milano2 Patients present in their third or fourth decade, often with radicular back pain. Reference Weber, Wang and Miller3 ME rarely presents as bilateral papilledema and symptoms of intracranial hypertension, with lumbar puncture yielding a “dry tap” and CSF hyperproteinemia. Reference Fan, Owji, Pakravan, Charoenkijkajorn and Lee4 Despite its relatively indolent growth characteristics, ME has potential for neuraxial seeding by drop metastasis and leptomeningeal dissemination. Reference Bertero, Ricci, Tampieri, Cassoni and Modena1 Local mass effect and neuraxial seeding can impair CSF flow and resorption, respectively, causing CSF hypertension above the lesion, CSF loculation below and impaired CSF resorption from drop metastases. Reference Fan, Owji, Pakravan, Charoenkijkajorn and Lee4 Consequently, CSF analysis and complete neuraxial imaging with MRI are recommended. Reference Bertero, Ricci, Tampieri, Cassoni and Modena1 We sampled CSF from the L4–L5 level, between two masses spanning T12–L3 and L5–S2 levels. A small volume of hyperproteinemic CSF with a falsely “normal” opening pressure suggested the region was not in communication with the intracranial compartment. Imaging was indicative of elevated CSF pressure above the masses and presence of leptomeningeal metastasis in the cervical, thoracic and lumbar spinal canal.
On spinal MRI, ME is characterized by a large and well-circumscribed ovoid or sausage-shaped mass, usually spanning multiple levels and compressing or encasing nearby nerve roots. Reference Kahan, Sklar, Post and Bruce5 The tumor is T1 isointense and T2 hyperintense, with multiple regions of signal heterogeneity owing to hemorrhage and calcification. Reference Hutuca, Egervari, Merkler and Vargas6 Heterogeneous gadolinium enhancement is a prominent feature, and drop metastasis or leptomeningeal seeding can be seen. Reference Hutuca, Egervari, Merkler and Vargas6 On gross pathology, ME appears pink or tan-gray, is often encapsulated and is soft and gelatinous, sometimes with areas of hemorrhage and cystic change. Reference Louis, Perry and Wesseling7 Histopathologically, there is a radial arrangement of glial tumor cells arranged in a papillary configuration around central hyalinized blood vessels, with myxoid material accumulated around vessels and in microcysts. Reference Louis, Perry and Wesseling7 Management involves laminectomy with subtotal or total resection, with or without adjuvant radiotherapy. Reference Weber, Wang and Miller3 Estimated 10-year overall and progression-free survival rates are approximately 92% and 61%, respectively, with tumor recurrence or progression occurring in approximately 32% of patients. Reference Weber, Wang and Miller3 Without prompt diagnosis and treatment, patients may develop progressive and irreversible lower limb sensorimotor impairments, loss of bowel and bladder function, sexual dysfunction or permanent vision loss. Reference Weber, Wang and Miller3,Reference Fan, Owji, Pakravan, Charoenkijkajorn and Lee4
Patients with papilledema and no structural intracranial lesion to explain their elevated intracranial pressure will often be diagnosed with idiopathic intracranial hypertension. Reference Wakerley, Mollan and Sinclair8 If such patients report symptoms of elevated intraspinal pressure, further evaluation with spinal neuroimaging is warranted. This patient presented with intracranial hypertension lacking features of obesity, hormonal contraception or tetracycline or isotretinoin use. Reference Wakerley, Mollan and Sinclair8 Progressive radicular low-back pain and intolerance to trunk flexion or supine positioning suggested elevated intraspinal pressure. CSF findings of Froin syndrome prompted us to adjust the differential diagnosis and line of investigation, Reference Dancel and Shaban9,Reference Koton and Bisharat10 leading to the correct diagnosis and appropriate management.
In cases with elevated intracranial and intraspinal pressures and Froin syndrome, neuraxial imaging is critical to evaluate for a spinal tumor. Reference Fan, Owji, Pakravan, Charoenkijkajorn and Lee4 In the absence of a spinal tumor, the differential diagnosis of Froin syndrome should be expanded to consider CNS inflammatory conditions like neurosarcoidosis, demyelinating polyradiculoneuropathies, hematologic malignancies or chronic meningeal infection or arachnoiditis, which likewise raise CSF protein concentrations. Reference Dancel and Shaban9,Reference Koton and Bisharat10
Acknowledgments
The authors thank the patient for their permission to present this case for educational purposes. This case report was approved by Newfoundland and Labrador Health Services and prepared in accordance with the CARE Guidelines for Case Reports (https://www.care-statement.org/). Memorial University of Newfoundland did not require ethics approval for this report.
Author contributions
NJS: management of the patient, design and conceptualization, literature review, drafting of the manuscript, final approval of the manuscript. SA: management of the patient, critical review of the manuscript, final approval of the manuscript. RE: management of the patient, critical review of the manuscript, final approval of the manuscript. HAE: management of the patient, critical review of the manuscript, final approval of the manuscript. AOO: management of the patient, design and conceptualization, critical review of the manuscript, final approval of the manuscript.
Funding statement
The authors report no external funding for this work.
Competing interests
The authors have no disclosures or competing interests to declare. This work has not been published, posted or submitted elsewhere in any other form.