Introduction
Several studies have indicated that the breakdown in immunity which results in the peri-parturient increase in nematode faecal egg count (FEC) in sheep can be moderated by dietary protein supply (Donaldson et al., Reference Donaldson, Van Houtert and Sykes1998 and Reference Donaldson, Van Houtert and Sykes2001; Houdijk et al., Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2000, Reference Houdijk, Kyriazakis, Coop and Jackson2001a and Reference Houdijk, Kyriazakis, Jackson and Coopb and Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2003). Other studies have suggested that degree of exposure to larvae and antigenic stimulation during mid and late pregnancy may influence immune responsiveness and therefore the rise in FEC on exposure to larvae in the peri-partum period (Leyva et al., Reference Leyva, Henderson and Sykes1982; Stankiewicz and Hadas, Reference Stankiewicz and Hadas1999). The question then arises as to whether the enhancement of immunity by protein supplementation represents a generally enhanced immune capability or an enhanced ability to respond immunologically to the antigenic stimulus of incoming larvae. This issue is important because sheep production systems in several countries are managed in such a way that ewes may be removed from exposure to larvae for several months during pregnancy, for example as a consequence of winter housing in the northern hemisphere or of grazing of spelled reseeded pastures with low levels of larval contamination in all-grass systems such as employed in New Zealand. In both cases, ewes can be re-exposed to larvae at the time of vulnerability to breakdown of immunity in the peri-partum period and during early lactation on return to contaminated pastures.
This paper describes a study designed to test the hypothesis that larval intake enhances the effect of dietary protein supply in limiting the relaxation of protection to nematode parasites in the peri-parturient ewe. Cells of the immune armoury in the gastro-intestinal tract and humoral immune responses were measured to investigate possible mechanisms of any responses.
Material and methods
Experimental design
Eighteen-month-old Coopworth ewes were used. They had been reared on ryegrass-white clover pastures under commercial conditions and would have received anthelmintic treatment at approximately monthly intervals until 8 months of age and then at 12 and 15 and 18 months. Following mating 80 scanned as carrying two lambs were treated with two anthelmintics (Ivomec, 200 μg ivermectin per kg live weight (LW); Merial New Zealand Ltd and Levben (4 mg ricobendzole +7.5 mg levamisole per kg LW; Novartis, New Zealand Ltd) and brought indoors into individual pens approximately 9 weeks before parturition.
They were allocated to four groups (no. = 20) in a 2 × 2 factorial design incorporating two levels of dietary protein concentration and the presence or absence of a vaccinating nematode infection during weeks − 8 to − 3 relative to anticipated mean date of parturition. The vaccinating infection comprised an average of 5000 T. circumcincta and 3000 T. colubriformis infective third stage larvae per day, given in three doses each week administered on filter paper by balling gun. All sheep were then treated with an anthelmintic (Systamex, 5 mg Oxfendazole per kg LW; Schering-Plough Animal Health Ltd, New Zealand) during week − 3 relative to parturition to remove worms associated with the vaccinating infection to enable measurement of the comparative response of vaccinated and unvaccinated sheep to a subsequent challenge infection. Previous work (Donaldson et al., Reference Donaldson, Van Houtert and Sykes1998 and Reference Donaldson, Van Houtert and Sykes2001) suggested that relaxation of immunity would have begun to occur at this stage of pregnancy. The challenge infection was imposed from week − 2 relative to parturition until 6 weeks of lactation and comprised an average daily infection with 10 000 and 7 000 third stage larvae per day of T. circumcincta and T. colubriformis, respectively, given in three doses each week. These rates of infection (vaccination and challenge infection) are considered to represent relatively large challenges, the former to ensure maximum antigenic stimulation. Comparable rates of infection are, however, possible during winter rationing of ewes in all-grass wintering systems in New Zealand (Familton and McAnulty, Reference Familton, McAnulty and Barrell1997). Five animals from each group, selected at parturition to balance groups for day of parturition and number of lambs reared, were necropsied for determination of worm burdens and sampling of abomasal and intestinal mucosal tissues at weeks +1 and +3 after lambing. These times were chosen as previous work had suggested that restoration of the ability of the ewe to limit development of incoming larvae could be occurring during the first few weeks of lactation (McAnulty et al., Reference McAnulty, Familton, Sedcole and Sykes2001). The remaining sheep in each group were maintained until week 6 for measurement of FEC. The experimental design is given in Table 1.
Experimental design and ewe reproductive performance
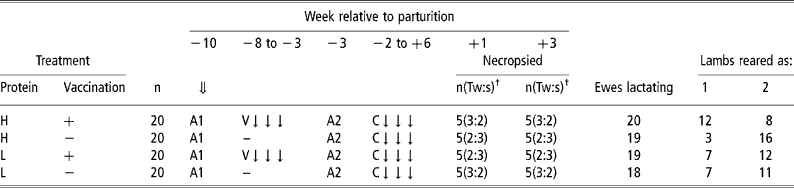
⇓ Ewes housed indoors.
A1 Treated with two anthelmintics (Ivomec+Lev/BZ combination).
V ↓ ↓ ↓ Trickle vaccinating infection with 5000 T. circumcincta and 3000 T. colubriformis per day.
A2 Treated with anthelmintic (Oxfendazole).
C ↓ ↓ ↓ Challenge infection with 10 000 T. circumcincta and 7000 T. colubriformis per day.
† Number of ewes and (proportion of twin:single suckling ewes).
Management and sampling
Animals were offered one of two complete feeds which differed in crude protein concentration, the composition and analysis of which are given in Table 2. The feed was offered in a ratio 2:1 (concentrate: chaffed hay) at rates designed to meet the metabolisable energy (ME) requirements of a twin-bearing ewe of 66.5 kg LW, the mean live weight of the ewes at housing, based on the recommendations of Agricultural and Food Research Council (AFRC, 1993). The crude protein concentrations were designed to provide 1.0 (low protein; LP) or 1.5 (high protein; HP) of the theoretical metabolisable protein (MP) requirement during pregnancy and about 0.8 (LP) or 1.3 (HP) of MP requirement during lactation according to AFRC (1993). These ratios were chosen in the expectation that AFRC (1993) may underestimate the protein requirement for maintenance of immunity to nematodes in the late pregnant and lactating ewe (Donaldson et al., Reference Donaldson, Van Houtert and Sykes2001). The amount offered was adjusted each week to take account of the increasing demand for pregnancy. After lambing and throughout lactation the sheep were offered 3 kg dry matter (DM) per head per day.
Composition (g/kg fresh weight) and analysis of feeds
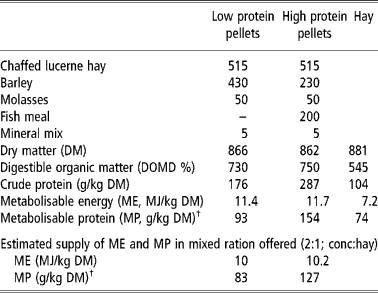
† Estimated from AFRC (1993) assuming rumen outflow rate (r) = 0.08.
Body weight was recorded immediately before feeding at 0830 h at weekly intervals until parturition and at less frequent intervals after lambing to minimise disturbance of the lambs. Faeces samples were taken from the rectum each week and nematode eggs counted immediately by the methods described by Xie et al. (Reference Xie, Stankiewicz, Huntley, Sedcole, McAnulty, Green and Sykes2004). Blood samples were collected into plain vacutainers (Becton Dickinson, USA) from the jugular vein prior to feeding every 2 weeks during weeks − 9 to − 3 relative to parturition, then weekly until week +3 and then during week +6.
After overnight storage at 14°C sera was separated by centrifugation at 2000 g for 20 min and stored at − 20°C. Samples of blood were analysed for total specific antibody and IgA specific antibody against the L3 stages of both T. circumcincta and T. colubriformis, by the methods of Xie et al. (Reference Xie, Stankiewicz, Huntley, Sedcole, McAnulty, Green and Sykes2004) and for specific IgE by the method described by Huntley et al. (Reference Huntley, Schallig, Kooyman, McKellar, Millership and Smith1998). In addition, interleukin 5 (IL5) was measured by indirect ELISA using the method developed and described by Else and Grencis (Reference Else and Grencis1991) and recommended for use in sheep by Doligalska et al. (Reference Doligalska, Moskwa and Stear1999).
Necropsy
The animals were stunned by captive bolt and the spinal cord and carotid artery and jugular veins severed immediately. The abdomen was opened along the central line and ligatures applied at the oesophageal junction with the abomasum, at the pyloris and in the small intestine 5 m distal to the pyloris and these tissues removed. Three samples (2 cm2) of abomasal wall from along the greater curvature and three samples of intestine taken 1 m from the pylorus were removed. One sample from each site was placed in each of liquid nitrogen, 5 ml paraformaldehyde or 5 ml modified Bouin-picric acid solution. The former was transferred to − 20°C after 30 to 60 min and the latter two into 70% alcohol (1.5 ml) after 6 h and then maintained at 4°C.
Abomasal and intestinal contents and washings were collected and the tissues digested in acidified pepsin solution (Herlich, Reference Herlich1956) to recover mature worms and immature stages (Robertson and Elliott, Reference Robertson and Elliott1966). Worms were differentiated into developmental stages (third (L3), fourth (L4) and adult (L5)) based on the information of Threkeld (Reference Threkeld1934).
Histology
Excised abomasal tissues and fixed tissues were processed as described by Huntley et al. (Reference Huntley, Jackson, Coop, Macaldowie, Houdijk, Familton, Xie, Stankiewicz and Sykes2004). For the histochemical counting of mast cells, Bouins fixed sections were stained with toluidine blue at pH 0.5 (Enerback, Reference Enerback1966). Counts of globule leucocytes and eosinophils were made after staining 4% paraformaldehyde-fixed tissue sections with carbol chromotrope (Lendrum, Reference Lendrum1944), and were differentiated on morphological criteria. Stained cells were counted under a × 10 eye piece containing a calibrated graticule and a × 25 objective lens viewing an area of 0.08 mm2 and counts were made from 10 graticule fields made systematically from the lamina muscularis to the mucosal surface. The counts were expressed as mean numbers per 0.2 mm2.
Statistical analysis
Due to missing data resulting from scanning inaccuracies and loss of lambs at parturition (see Table 1) number of lambs reared was introduced as a treatment effect. Transformations were used on the data on the basis of residual plots. The actual transformations used are given in the appropriate captions of the tables and figures. The generalised linear model in Genstat (Lawes Agricultural Trust, 2002) was used for analysis of worm burdens and the residual maximum likelihood (REML) in Genstat for parameters with repeated observations in time and the covariance structure of these data accommodated. In the case of worm burdens analyses were carried out across the total population, using species and developmental stages of the nematodes as fixed effects, and separately for each nematode species. The ratio of L3+L4 to total worm burden was also analysed separately for each species.
The experiment was carried out under the authority of the Committee of Ethics of Animal Experimentation of Lincoln University, New Zealand (certificate no. 803).
Results
At parturition inaccuracies in scanning of foetuses was detected and there were some losses of lambs, particularly twin lambs. The number of ewes that successfully reared lambs and the distributions of twin and single reared lambs are given in Table 1. Vaccinated sheep on the high protein diet reared a disproportionately small and their unvaccinated contemporaries a disproportionately large number of twin lambs.
Animals generally consumed all the feed offered. As a consequence estimated ME intake increased from about 11.3 MJME per day during week − 8 relative to parturition through 14.5 MJME per day during week − 5 and 20.4 MJME per day during week − 2. These were within 5% of the theoretical requirement for twin-bearing ewes of this body weight. MP intakes, calculated according to AFRC (1993), see Table 2, were 82 and 127 g MP per day during week − 8 and 146 and 224 g MP per day during week − 2 relative to parturition for LP and HP groups, respectively. During lactation LP and HP sheep generally consumed all the feed offered which was estimated to provide 30–35 MJME per day for both groups and 260 and 380 g MP per day for those on the LP and HP diets, respectively.
Ewe live weight
The mean LWs of the ewes are given in Figure 1. There were significant interactions between time and litter size and effects of time (P < 0.01 in both cases) reflecting increase in LW before lambing and losses during parturition and subsequently during lactation, particularly in ewes rearing more than one lamb. There were no effects of dietary protein supply or vaccinating infection.
Live-weight changes in periparturient ewes infected with the nematodes T. circumcincta and T. colubriformis; effect of offering feed providing an estimated 130% (●) or 80% (○) of metabolisable protein requirement; effect of presence (■) or absence (□) of a vaccinating infection during mid-late pregnancy and effect of bearing and rearing multiple (▾) or single (▿) lambs. Note: H/D, housing and drench of all animals; V, vaccinating infection; D, drench of all animals; C, challenge of all animals.

Ewe faecal egg count
The changes in mean faecal egg count are given in Figure 2. There was a significant dietary protein × litter size × time interaction (P < 0.01) and significant protein × time and litter size × time interactions (P < 0.001 in both cases) due to the rise in FEC in all groups after parturition and larger increases in animals on the low protein diet and in multiple-rearing ewes. In particular the rise in FEC in single-rearing ewes on the high protein diet was delayed, compared with the other groups. There was also a highly significant time × vaccination treatment interaction (P < 0.001) as a consequence of lower FEC immediately after parturition in those ewes that received a vaccinating infection during late pregnancy.
Mean faecal egg count (log10(count+1)) of periparturient ewes infected with the nematodes T. circumcincta and T. colubriformis: (a) effect of offering feed providing an estimated 130% (●) or 80% (○) of metabolisable protein requirement; (b) effect of presence (●) or absence (○) of a vaccinating infection; (c) effect of rearing single- (▿) or twin- (▾) lambs; (d) interaction of rearing status and MP supply-(●,○ HP; ▾,▿,LP), single- and twin-rearing represented in open and closed symbols, respectively. Note: H/D, housing and drench of all animals; V, vaccinating infection; D, drench of all animals; C, challenge of all animals.

Worm burdens
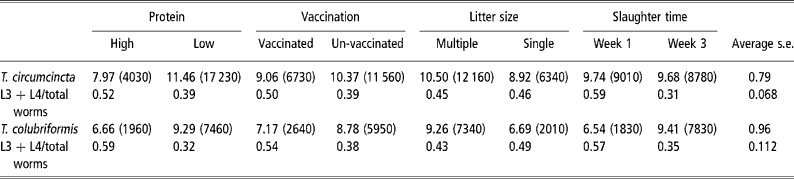
The mean numbers of T. circumcincta and T. colubriformis recovered from the groups slaughtered 1 and 3 weeks after parturition are given in Table 3. When analysed across nematode species there were interactions between species total and necropsy time (P < 0.001) and between numbers of nematodes in the different development stages with protein supply (P < 0.001), vaccination status (P < 0.01) and slaughter time (P < 0.001) and significant main effects of protein supply (P < 0.001), vaccinating infection (P < 0.01), number of lambs reared (P < 0.001) and nematode species (P < 0.01). Analyses within nematode species showed significant effects of dietary protein supply on the numbers of worms of T. circumcincta (P < 0.01) and of T. colubriformis (P < 0.001) and a significant effect of litter size on numbers of the latter (P < 0.01) but not the former nematode. Worm numbers were reduced by the vaccinating infection (P < 0.05 for both nematodes). Worm numbers of T. colubriformis increased between the first and second necropsies (P < 0.05) but those of T. circumcincta were stable. There was, however, a highly significant (P < 0.001) reduction in the proportion of the population of T. circumcincta in the developing stages (L3+L4) at the second necropsy, which reflected a reduction (proportionate 0.51) in L3+L4 numbers and an increase (proportionate 1.6) in adult numbers at the second necropsy. By comparison the trend for reduction (P>0.05) in the L3+L4 contribution to total worm burden of T colubriformis at the second necropsy was the resultant of large increases in both L3+L4 (proportionate 2.1) and adult (proportionate 6.4) worms. Only the effects of protein supply and vaccinating infection on the proportion of developing stages of T. circumcincta were statistically significant (P < 0.04 and 0.06, respectively).
Main effects of dietary protein, vaccination, number of lambs reared and time of slaughter after parturition on fourth root transformed total worm burdens and ratio of 3rd and 4th stage larvae to total worm numbers of T. circumcincta and T. colubriformis in periparturient ewes (back-transformed means of total burdens are given in parentheses) (number of ewes in each group=20)
Serum total antibody
The changes in serum total antibody against T. circumcincta and T. colubriformis are given in Figure 3. For antibodies to both parasites there was a significant vaccinating challenge × time interaction (P < 0.001 in both cases) reflecting a decline in antibody titres during pregnancy in unvaccinated sheep. Significant time effects for antibodies to both nematodes (P < 0.001 in both cases) were a reflection of values that peaked at parturition followed by an immediate decline and subsequent recovery.
Changes in serum total antibody against (a) T. circumcincta and (b) T. colubriformis in periparturient ewes; effect of offering feed providing an estimated 130% (●) or 80% (○) of MP requirement; effect of presence (■) or absence (□) of a vaccinating infection during mid-late pregnancy and effect of bearing and rearing multiple (▾) or single (▿) lambs. Note: H/D, housing and drench of all animals; V, vaccinating infection; D, drench of all animals; C, challenge of all animals.

Serum IgA
The parasite L3-specific serum IgA titres are given in Figure 4. For serum IgA against T. circumcincta there were vaccination × time (P < 0.001) and litter size × time (P < 0.05) interactions reflecting lower levels in unvaccinated sheep during pregnancy and after parturition and lower levels in single- than in multiple-bearing and rearing sheep.
Changes in serum specific IgA against (a) T. circumcincta and (b) T. colubriformis in periparturient ewes; main effect of offering feed with estimated 130% (●) or 80% (○) of MP requirement; effect of presence (■) or absence (□) of a vaccinating infection during mid-late pregnancy and effect of bearing and rearing multiple (▾) or single (▿) lambs. Note: H/D, housing and drench of all animals; V, vaccinating infection; D, drench of all animals; C, challenge of all animals.

For T. colubriformis there were significant vaccination × time interactions and effect of litter size (P < 0.001 in both cases) but no litter size × time effects. For IgA antibodies to both nematodes significant effects of time (P < 0.01 in both cases) was due to increasing titres until the week before parturition and subsequent stabilisation. There was no effect of dietary protein supply on IgA titres to L3 larvae of either nematode.
Serum IgE
The changes in serum IgE against T. circumcincta and T. colubriformis are given in Figure 5. There were vaccination × time interactions (P < 0.001 and P < 0.01 for T. circumcincta and T. colubriformis, respectively), the result of lower values in unvaccinated sheep, particularly during pregnancy and significant time effects (P < 0.001 in both cases). The latter reflected a trend for increase in titres during pregnancy and decline during lactating, commencing just before parturition. The time effect was more marked for IgE against T. circumcincta. There was a protein × time interaction for T. colubriformis (P < 0.01) but not for T. circumcincta, though in both cases titres tended to be lower in high protein groups during lactation.
Changes in serum specific IgE against (a) T. circumcincta and (b) T. colubriformis in periparturient ewes; main effect of offering feed with estimated 130% (●) or 80% (○) of MP requirement; effect of presence (■) or absence (□) of a vaccinating infection during mid-late pregnancy and effect of bearing and rearing multiple (▾) or single (▿) lambs. Note: H/D, housing and drench of all animals; V, vaccinating infection; D, drench of all animals; C, challenge of all animals.

Serum interleukin 5
There was a significant effect of time (P < 0.001) due to the depression of concentrations around parturition. Concentrations (log 10 pg/ml) tended upwards during pregnancy from 1.9 to 2.1, 2 weeks before parturition, but fell to 1.7 in the 3 weeks around parturition and then rose during lactation. A protein × time interaction (P < 0.05) reflected lower concentrations during lactation in animals offered the high protein supply.
Mucosal inflammatory cells
The numbers of inflammatory cells in abomasal and intestinal lamina propria tissue at the two slaughter times are given in Table 4. The major finding was of significant between animal variation. Vaccination resulted in greater numbers of mucosal mast cells (MMC) in both tissues and a highly significant increase in globule leucocyte (GL) numbers in abomasal tissue but not in intestinal tissue. In the latter tissue there was a trend (P>0.05) for increase in eosinophil (EOS) numbers but in abomasal tissue a significant reduction though at much lower cell numbers. Increased MP supply was associated with an increase in MMC in both tissues, significantly so in intestinal tissue, with no change in EOS or GL in abomasal tissue, but with a trend for reduction in EOS (P>0.05) and increase (P < 0.05) in GL cell numbers in intestinal tissue. In abomasal tissue GL numbers increased between weeks 1 and 3 after parturition whereas in intestinal tissue GL cell numbers decreased during this time. Interactions between treatments were not observed for EOS numbers or for numbers of MMC in intestinal tissue. In abomasal tissue there was a trend for a vaccination × necropsy time interaction (P < 0.06) reflecting greater difference in numbers in vaccinated sheep at the first than at the second necropsy. For GL in both tissues there were several weak interactions (P < 0.06 to 0.1); (a) a trend for the difference in cell numbers between vaccinated and unvaccinated sheep to be greater at the first than at the second necropsy; (b) in intestinal tissue for lack of vaccinating infection to have greater effect on cell numbers at the lower than at the high protein supply; (c) in abomasal tissue, for cell numbers to be reduced by the low protein supply or by lack of vaccinating challenge to a greater degree in twin-rearing than in single-rearing ewes.
Main effects of dietary protein, vaccination, litter size status and time of slaughter after parturition on numbers of local inflammatory cells (log10(n+1) transformed data with back-transformed means in parentheses in abomasal and intestinal mucosa from periparturient ewes†

† EOS, eosinophil; GL, globule leukocytes; MMC, mucosal mast cell. *P < 0.05; **P < 0.01; ***P < 0.001.
Negative correlations were observed between numbers of T. colubriformis and density of MMC and GL in intestinal tissue ( − 0.65 and − 0.61; P < 0.001 in both cases) and between T. circumcincta worm burdens and MMC in abomasal tissue ( − 0.43; P < 0.01) when data from the treatment groups were combined. A positive correlation was also observed between abomasal worm burdens and EOS numbers in abomasal mucosa (0.37; P < 0.02). Relationships were not observed between eosinophil numbers in intestinal tissue and burdens of T. colubriformis or between worm burdens and numbers of GL in the abomasums.
Discussion
The importance of dietary protein supply for the ability of the ewe to maintain protection against nematode parasites in the period around parturition is now well established as is the influence of numbers of lambs reared (Donaldson et al. (Reference Donaldson, Van Houtert and Sykes1998 and Reference Donaldson, Van Houtert and Sykes2001), Houdijk et al. (Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2000, Reference Houdijk, Kyriazakis, Coop and Jackson2001a and Reference Houdijk, Kyriazakis, Jackson and Coopb and Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2003) and Kahn et al. (Reference Kahn, Knox, Walkden-Brown and Lea2003)). The present data add further weight to the concept that nutrient scarcity per se, reflecting competing demands of lactation and the immune system for nutrients, is critical. Effects of vaccinating infection, though measurable in a halving of worm burdens of both nematode species 7 days after lambing, were small and transitory in their effect on FEC in comparison with the effect of protein supply or numbers of lambs reared. In this context the observations of Houdijk et al. (Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2003) of greater sensitivity of the immune system than milk synthesis to marginal protein supply is telling.
FEC, however, is the resultant of numbers of mature worms and their fecundity each of which may be the subject of limitation by the host immune response. While the experimental design did not allow analysis of these components the serial necropsy approach provided evidence that the direction of response to the two nematode populations to increased host MP supply, vaccinating infection and lactational demand were similar. In a temporal sense, however, there were differences in worm population dynamics (Table 3). Whereas the population of T. circumcincta was stable between weeks 1 and 3 after lambing, and there was evidence for limitation of development of incoming larvae, that of T. colubriformis was increasing. This inability to control the population of the intestinal nematode T. colubriformis may have been responsible for the trend for FEC to continue to increase and for differences in FEC between treatment groups to disappear as lactation progressed. Other comparable studies but using single-species infections with the abomasal nematode T. circumcincta have generally observed a decrease in FEC after about 3 to 4 weeks of lactation especially in protein supplemented groups (Houdijk et al., Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2003 and Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2005), consistent with observations of restoration of ability limit development of larvae of T. circumcincta within 2 to 4 weeks of parturition in well fed sheep (McAnulty et al., Reference McAnulty, Familton, Sedcole and Sykes2001).
The pattern of changes in cells of the immune armoury in the gastro-intestinal mucosa was consistent with the general direction of nematode population responses to treatments but there was evidence to suggest that the precise association between the host immune system and the nematode populations for the two nematodes may differ. The typical pattern of response of the immune armoury under larval challenge has been described by Balic et al. (Reference Balic, Bowles and Meeusen2000). In animals with no previous exposure a non-specific recruitment in mucosal tissues of eosinophils and production of IgA antibody is followed by increases in MMC and their de-granulated cell remnant, GL (Huntley et al., Reference Huntley, Newlands and Miller1984; Pfeffer et al., Reference Pfeffer, Douch, Shaw, Gatehouse, Rabel, Green, Shirer, Jonas and Bisset1996). Subsequently, EOS recruitment to tissue and antibody production diminish, a pattern which has been observed in peripheral EOS numbers (Kimambo et al., Reference Kimambo, MacRae, Walker, Watt and Coop1988) and IgA antibodies (Cripps and Steel, Reference Cripps and Steel1978; Henderson and Stear, Reference Henderson and Stear2006). Balic et al. (Reference Balic, Bowles and Meeusen2000) interpreted changes in EOS numbers as indicating recruitment to mucosal tissue until the components of the mature protective immune armoury are able to exclude larvae from mucosal tissue. Circulating concentrations of antibodies have been shown to be highly correlated with rates of antibody production (Cripps and Steel, Reference Cripps and Steel1978) though the sensitivity of peripheral IgA to small changes in production in mucosal tissue is unclear (Sinski et al., Reference Sinski, Bairden, Duncan, Eisler, Holmes, McKellar, Muurray and Stear1995; Henderson and Stear, Reference Henderson and Stear2006). The ubiquitous increase in IgA in peripheral circulation during the acquisition of immunity in naïve animals and its subsequent decline (Cripps and Steel, Reference Cripps and Steel1978; Henderson and Stear, Reference Henderson and Stear2006) and the lack of peripheral response in animals with established immunity (Greer et al., Reference Greer, Stankiewicz, Jay, McAnulty and Sykes2005) suggests that, like eosinophils, its rate of production in mucosal tissues, and therefore appearance in circulation, may reflect the extent of larval penetration of the tissue determined by the effectiveness of the mature mucosal-cell based immune response. The present sheep would have been expected to have had an established immunity to the nematode species used as a consequence of an inevitable exposure to larvae of these nematodes which are endemic in the farm environment in which they had been reared. Had withdrawal from the antigenic stimulation of the non-vaccinated sheep led to a reduction in MMC/GL (Huntley et al., Reference Huntley, Newlands, Jackson and Miller1992) we could have anticipated a greater recruitment of eosinophils and IgA production. Conversely, we could have expected a response dominated by higher numbers of MMC/GL and fewer eosinophils in vaccinated sheep, particularly on the high protein diet and a stronger shift towards greater numbers of MMC/GLs in the latter sheep between weeks 1 and 3 with more rapid re-establishment of immunity. The findings of positive correlation between EOS numbers and worm burdens in abomasal tissue, negative correlations between MMC and worm burdens in both abomasal and intestinal tissues and negative correlations between GL and worm burdens in intestinal tissue are consistent with this interpretation.
The trend for lower EOS numbers in the abomasal and intestinal mucosa of HP and vaccinated animals (Table 4) suggests a greater ability to exclude incoming larvae from tissues which is consistent with the lower worm numbers in these groups and the findings of Donaldson et al. (Reference Donaldson, Van Houtert and Sykes2001) that increased protein supply was associated with lower establishment of larvae of T. circumcincta. Houdijk et al. (Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2003) observed a similar sensitivity of abomasal eosinophil numbers to MP supply 3 weeks after lambing in Teladorsagia circumcincta-infected sheep but only at much lower MP supply (180 g/day) than the range (220 to 380 g/day) used in the present study. The trend for greater EOS numbers in abomasal tissue and similar numbers in intestinal tissue from single-rearing ewes is, however, inconsistent with their lower worm burdens than twin-rearing ewes.
MMC numbers, as expected (Balic et al., Reference Balic, Bowles and Meeusen2000), were lower in unvaccinated sheep and tended to be lower in sheep on the LP diet, especially in the intestine. The actual numbers were comparable with the 30 to 40 cells per 0.2 mm2 observed by Houdijk et al. (Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2005) in lactating sheep. These authors, however, observed a marked depression from the 80 to 120 cells per 0.2 mm2 in non-pregnant sheep to less than 10 cells per 0.1 mm2 in the period immediately around parturition followed by a rise during lactation. Thus it would appear that MMC numbers are inherently diminished by factors associated with the reproductive process. Our data suggest that the extent of the reduction is influenced by antigenic experience and protein nutrition. In this respect the observation, in the present work, of significant reduction in interleukin 5 in the period 2 weeks before to a week after parturition may be relevant. It is, we believe, the first evidence that signaling through cytokines in the sheep immune system may be disturbed around parturition and deserves further study in relation to migration and functionality of cells of the local immune response. The technique used in this study was the best available at the time of the experimentation; the conserved nature of IL-5 was considered sufficient to ensure a high level of cross reactivity as discussed by Doligalska et al. (Reference Doligalska, Moskwa and Stear1999). Clearly, these findings suggest it should be further investigated using specific monoclonal antibodies against ovine IL-5.
The increase in GL numbers in the intestine as a result of the HP diet and single-rearing suggests changes in MMC de-granulation also consistent with enhanced immune capability. Though these specific trends were not shown in abomasal tissue, vaccination was associated with much greater numbers of GL in this tissue. In fact, the total numbers of GL in abomasal and intestinal tissue were very different, averaging 13 and less than 2, respectively the former being much greater than the 2 to 4 cells per mm2 observed by Houdijk et al. (Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2003). If GL are de-granulated remnants of discharged MMC (Huntley et al., Reference Huntley, Newlands, Jackson and Miller1992) the present data suggest a more rapid turnover of MMC in abomasal than in intestinal tissue.
When viewed together and from a standpoint of immune system ontogony, the association of low EOS numbers with high GL numbers in abomasal tissue and of high EOS numbers with low GL in intestinal tissue does suggest that the host ability to regulate worm populations was more advanced in abomasal tissue, which is consistent with the worm population dynamics discussed earlier. This hypothesis involves an as yet unvalidated assumption that the pattern of involvement of cells of the immune response is identical in the two tissues and Balic et al. (Reference Balic, Bowles and Meeusen2000) cautioned that ‘it may well be that present methods for quantifying MMC/GL numbers may not accurately reflect their number or activation status.’ The present appears to be the first study in which cells have been measured in the two sites simultaneously and does suggest there may be differences in mechanism or timing of immune responses. The broadly similar direction of changes in EOS, MMC and GL as a consequence of vaccination and increased protein supply suggests that the effect of dietary protein in enhancing protection operates through enhancing retention or recruitment of cells associated with the local cellular development in line with the findings of Coop et al. (Reference Coop, Huntley and Smith1995) that the ability to generate mast cell derived protease activity during vaccinating infection of immunologically naïve animals is enhanced by dietary protein supply.
None of the serum antibody isotypes was affected by dietary protein supply which perhaps adds weight to the hypothesis that the effect of enhanced protein supply in the peri-partum period is primarily through effect on local mucosal cell populations and their activity.
Total antibody titres were only maintained by a current larval intake, titres declining in unvaccinated sheep during pregnancy but were very rapidly restored during the last 2 weeks of pregnancy on restoration of larval challenge. This pattern clearly refutes a putative immuno-suppressive effect of pregnancy on antibody production as previously suggested by Lloyd (Reference Lloyd1983). The subsequent reduction in titres 2 weeks after lambing could reflect suppression around parturition but, perhaps more plausibly, the active transfer into colostrum (Brambell, Reference Brambell1970; Butler et al., Reference Butler, Kiddy, Pierce and Rock1972; Sasaki et al., Reference Sasaki, Davis and Larson1976).
IgA levels increased significantly during pregnancy only in sheep experiencing vaccinating infections but, like total antibody, increased rapidly on commencement of challenge infections of unvaccinated sheep, suggesting increase in production of the antibody in mucosal tissue during late pregnancy. A similar rise in titres observed in ewes around parturition during infection with T. circumcincta (Jeffcoate et al., Reference Jeffcoate, Wedrychowicz, Fishwick, Dunlop, Duncan and Holmes1992) was attributed to either ‘a general increase in IgA in preparation for lactation… or to increased antigenic stimulation caused by the presence of greater numbers of establishing and, or, developing worms at this time’. The latter interpretation seems plausible in view of the fact that elevation of peripheral IgA titres, like eosinophilia, appears to be a feature only of the acquisition phase of the immune response Greer et al. (Reference Greer, Stankiewicz, Jay, McAnulty and Sykes2005). The fact that IgA levels increased in the vaccinated sheep as pregnancy advanced, and were greater in multiple- than in single-bearing and rearing ewes during late pregnancy and early lactation despite an anticipated greater transfer to milk, does perhaps support the hypothesis of increased antigenic stimulation as a consequence of relaxation of cell mediated immunity during pregnancy (Monterroso and Hansen, Reference Monterroso and Hansen1993; Roberts et al., Reference Roberts, Satoskar and Alexander1996; Raghupathy, Reference Raghupathy1997; Miyaura and Iwata, Reference Miyaura and Iwata2002) as the nutritional demands of reproduction increased. The occurrence of eggs in the faeces of LP sheep during the later stages of the vaccinating challenge (Figure 2) and the low numbers of MMC observed in peri-parturient compared with non-pregnant sheep by Houdijk et al. (Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2005) also support this conclusion.
Although IgE is considered to be one of the important components of the protective mechanism in the gastric mucosa (Miller, Reference Miller1984) concentrations in peripheral circulation were more related to the reproductive cycle than to larval challenge or nutritional status, a pattern observed by Houdijk et al. (Reference Houdijk, Kyriazakis, Jackson, Huntley and Coop2005) and observed previously in other species (Piccinni et al., Reference Piccinni, Guidizi, Biagiotti, Beloni, Giannarini, Sampognaro, Parronchi, Manetti, Annunziato, Livi, Romagnani and Maggi1995; Roberts et al., Reference Roberts, Satoskar and Alexander1996; Miyaura and Iwata, Reference Miyaura and Iwata2002).
Conclusions
These findings provide further evidence that the ability to limit nematode worm populations in the peri-partum period in the breeding ewe is susceptible to dietary protein supply, and that responses in worm establishment can be anticipated at intakes in the range 250–374 g MP/d. In addition, withdrawal from antigenic stimulation prior to lambing, as a result of husbandry practices, could have effect on ability to regulate worm populations but is probably of much less significance for FEC than protein nutrition or numbers of lambs suckled. Retention and recruitment of cells of the mucosal immune response – mucosal mast cells and globule leucocytes – are dependent on continuous larval challenge and dietary protein supply and may explain the mechanism of effect of dietary protein supply on resistance to nematode infection.
Acknowledgements
The skilled technical assistance of C.M. Logan, M. Ridgway and H. Howden. This project was funded by Meat NZ. J. Huntley and A. McKellar are supported by the Scottish Executive, Environment and Rural Affairs Department.










