Introduction
Italian ryegrass [Lolium perenne ssp. multiflorum (Lam.) Husnot] (referred to as L. multiflorum hereafter) was originally introduced from Europe into the United States as a cool-season forage crop (Thorogood Reference Thorogood, Casler and Duncan2003). It has high genetic diversity, allowing it to adapt to various soil and environmental conditions (Jung et al. Reference Jung, Van Wijk, Hunt, Watson, Moser, Buxton and Casler1996). Lolium multiflorum emerges early in the fall and infests small grain crops, where it can cause yield losses >80% across the United States (Stone et al., Reference Stone, Cralle, Chandler, Miller, Bovey and Carson1998). It can grow vigorously under cool, wet conditions, with prolonged growth in autumn and rapid regrowth in spring. Lower seed cost and rapid growth makes it an ideal cover crop in the U.S. Midwest and Northeast (Johnson et al. Reference Johnson, Kaspar, Kohler, Corak and Logsdon1998; Kuo et al. Reference Kuo, Sainju and Jellum1997; Teasdale and Mohler Reference Teasdale and Mohler1993; Weston Reference Weston1990). Lolium multiflorum is currently grown as a winter annual forage or cover crop on approximately 1.62 million ha in the United States, including New York (DiTomaso and Healy Reference DiTomaso and Healy2007; Herbstritt et al. Reference Herbstritt, Richard, Lence, Wu, O’Brien, Emmett, Kaspar, Karlen, Kohler and Malone2022). Rapid growth, high fecundity, and profuse seed-shattering potential facilitate its persistence and evolution of weedy characteristics (Maity et al. Reference Maity, Singh, Jessup and Bagavathiannan2021). Currently, it is ranked as one of the most troublesome winter annual grass weed species in wheat (Triticum aestivum L.) and other agronomic and specialty crops throughout the United States (Van Wychen Reference Van Wychen2023; Webster and MacDonald Reference Webster and MacDonald2001; Yadav et al. Reference Yadav, Russell, Ganie, Patel, Price and Maity2025), including New York. Uncontrolled L. multiflorum at various densities can cause severe yield losses in different agronomic crops: up to 65% in corn (Zea mays L.), 85% in cotton (Gossypium hirsutum L.), 92% in wheat, and 37% in soybean [Glycine max (L.) Merr.] (Bayer Crop Science 2023; Hashem et al., Reference Hashem, Radosevich and Roush1998; Nandula Reference Nandula2014).
The widespread use of L. multiflorum as a cover crop or forage species has contributed to its introduction and subsequent establishment as a problematic weed. Biological traits such as obligate outcrossing and wind pollination make this species highly conducive to the evolution and rapid spread of herbicide resistance. As a result of continuous and repeated reliance on glyphosate for control, glyphosate resistance is now common among L. multiflorum populations in multiple U.S. states. For example, this species is particularly difficult to manage in grass seed production systems in the Willamette Valley of Oregon, where resistance to glyphosate, acetyl-CoA-carboxylase (ACCase) inhibitors, and acetolactate synthase (ALS)-inhibiting herbicides has been documented. Herbicide-resistant populations have been reported from 19 states of the United States. For instance, L. multiflorum populations resistant to inhibitors of ACCase (categorized by the Weed Science Society of America and Herbicide Resistance Action Committee [WSSA/HRAC] as Group 1), ALS (WSSA/HRAC Group 2), photosystem II (WSSA/HRAC Group 5), 5-enolpyruvyl shikimate phosphate synthase (EPSPS, WSSA/HRAC Group 9), glutamine synthetase (WSSA/HRAC Group 10), carotenoid biosynthesis (WSSA/HRAC Group 12), protoporphyrinogen oxidase (WSSA/HRAC Group 14), very-long-chain fatty-acid (WSSA/HRAC Group 15), and photosystem I (PSI) electron diversion (WSSA/HRAC Group 22) have been documented (Heap Reference Heap2025). Resistance to glyphosate has been widely reported among L. multiflorum populations in the United States (Heap Reference Heap2025). Furthermore, populations with multiple resistance to up to four different herbicides belonging to ACCase inhibitors, ALS inhibitors, EPSPS inhibitors, and PSI electron diversion have also been reported from California and North Carolina (Heap Reference Heap2025). Increasing cases of herbicide-resistant L. multiflorum populations limit herbicide options for effective control in agronomic crops.
Several mechanisms conferring glyphosate resistance have been reported in weed species. For instance, both target site–based (e.g., altered EPSPS gene via point mutation or EPSPS gene amplification) and non–target site based resistance (e.g., altered glyphosate absorption or translocation, glyphosate sequestration in the vacuole, and enhanced metabolism) are commonly reported mechanisms in glyphosate-resistant (GR) weeds (Aulakh et al. Reference Aulakh, Kumar, Brunharo, Veron and Price2024, Reference Aulakh, Kumar, Westrick, Price and Jhala2025; Gaines et al. Reference Gaines, Zhang, Wang, Bukun, Chrisholm, Shaner, Nissen, Patzoldt, Tranel, Culpepper, Grey, Webster, Vencill, Sammons and Jiang2010; Koger and Reddy Reference Koger and Reddy2005; Kumar et al. Reference Kumar, Jha, Giacomini, Westra and Westra2015; Lorraine-Colwill et al., Reference Lorraine-Colwill, Powles, Hawkes, Hollinshead, Warner and Preston2002; Perez-Jones et al. Reference Perez-Jones, Park, Polge, Colquhoun and Mallory-Smith2007; Powles and Preston Reference Powles and Preston2006; Riar et al. Reference Riar, Norsworthy, Johnson, Scott and Bagavathiannan2011; Salas et al. Reference Salas, Dayan, Pan, Watson, Dickson, Scott and Burgos2012; Yu et al. Reference Yu, Abdallah, Han, Owen and Powles2009). The EPSPS gene amplification, point mutations in the EPSPS gene, and reduced translocation have been reported among GR L. multiflorum populations (Pagnoncelli et al. Reference Pagnoncelli, Losada, Alvear, Gonzalez-Andujar, Trezzi, Bittencourt and Salomão2024; Perez-Jones et al. Reference Perez-Jones, Park, Polge, Colquhoun and Mallory-Smith2007; Salas et al. Reference Salas, Dayan, Pan, Watson, Dickson, Scott and Burgos2012, Reference Salas, Scott, Dayan and Burgos2015).
During the spring of 2023, a grower in Livingston County in western New York reported inadequate control of an L. multiflorum cover crop with two sequential applications of glyphosate (Roundup® PowerMax, Bayer CropScience, Saint Louis, MO, USA) at field use rates (1,260 g ha−1). Similarly, in the spring of 2024 and 2025, two separate field crop producers from Ontario and Genesee counties, respectively, reported control failure of L. multiflorum cover crops with glyphosate. It is speculated that GR populations in New York were most likely introduced through purchased cover crop seed of L. multiflorum. Many producers in the state rely on L. multiflorum seed labeled as “VNS” (variety not specified), which often contains mixtures of seed from different varieties, increasing the risk of introducing herbicide-resistant biotypes. In response to control failures, mature seeds of these L. multiflorum populations were collected from growers’ fields to evaluate suspected glyphosate resistance. Furthermore, limited information exists on the underlying mechanism(s) and the effectiveness of alternative postemergence herbicides to control these suspected GR L. multiflorum populations from New York. Therefore, the main objectives of this study were to (1) confirm and characterize the level of glyphosate resistance in three selected L. multiflorum populations, (2) determine the effectiveness of alternative postemergence herbicides to control these GR L. multiflorum populations, and (3) investigate whether the EPSPS gene amplification confers glyphosate resistance in these GR L. multiflorum populations.
Materials and Methods
Seed Source
Mature seeds from three L. multiflorum populations in Livingston, Ontario, and Genesee counties in New York were collected during the 2023, 2024, and 2025 growing seasons, respectively (Figure 1). Seeds from the Livingston County population were collected from a fallow field (42.9769°N, 77.70131°W), whereas those from the Ontario and Genesee counties were collected from soybean (42.9834°N, 77.2272°W) and cornfields (42.9691°N, 78.0530°W), respectively. Fifteen to 25 seed heads from each population (each seed head came from an individual plant within a population) were collected at maturity from the respective fields and were manually threshed and cleaned. Lolium multiflorum populations from Livingston, Ontario, and Genesee counties were designated as “NY_R1”, “NY_R2”, and “NY_R3”. In addition, seeds from a previously known glyphosate-susceptible (GS) L. multiflorum population (36.0974°N, 94.1665°W) from Arkansas (AR_S) were also included for comparison (provided by JKN).
Field sites in Livingston (A), Ontario (B), and Genesee (C) counties in New York where seeds of suspected glyphosate-resistant (GR) Lolium multiflorum populations were collected in 2023, 2024, and 2025 growing seasons, respectively.

Glyphosate Dose–Response Experiments
Greenhouse experiments were conducted at Cornell University’s Guterman Bioclimatic Facility in Ithaca, NY, during the spring and summer of 2024 (for seed collected from Livingston and Ontario counties during 2023 and 2024) and repeated in the summer and spring of 2025 (for seed collected from Genessee County in 2025) to confirm and characterize the glyphosate resistance levels in three suspected GR L. multiflorum populations (NY_R1, NY_R2, NY_R3). Seeds of all suspected GR L. multiflorum populations from New York, along with the AR_S population, were separately sown on the surface of germination trays (each tray measuring 54 by 34 by 6 cm) containing Cornell greenhouse potting mixture (a mixture of Canadian peat moss, vermiculite, perlite, dolomite lime, Jack’s 10-5-10 N-P-K media mix plus II, JR Peters Inc, Allentown, PA, USA and calcium sulfate). After emergence (1- to 2-leaf stage), seedlings from each population were separately transplanted into 10-cm-square plastic pots (Greenhouse Megastore, Danville, IL) containing the same Cornell greenhouse potting mixture. Experiments were set up in a randomized complete block design (blocked by population) with 15 replications (1 plant per pot = 1 replication) and repeated in space and time. Growing conditions in the greenhouse during experimental periods were set at 27/24 ± 3 C day/night temperatures with 16/8-h day/night photoperiods supplemented with metal-halide lamps (450 µmol m−2 s−1). Actively growing seedlings (4- to 6-leaf stage) from each population were sprayed with isopropylamine salt of glyphosate (Durango™, Corteva Agrisciences, Indianapolis, IN) at doses of 0, 319, 638, 1,276 (field use rate), 2,552, 5,104, 10,208, and 20,416 g ae ha−1 along with ammonium sulfate at 20 g L−1. Glyphosate doses were applied using a stationary cabinet spray chamber (Research Track Sprayer, De Vries Manufacturing, Hollandale, MN) equipped with a flat-fan 8002XR nozzle tip (TeeJet® Technologies, Glendale Heights, IL). The sprayer was calibrated to deliver 141 L ha−1 of spray solution at 276 kPa. After being sprayed, all treated plants were returned to the greenhouse and watered at 24 h after glyphosate application. Afterward, all L. multiflorum populations were watered on a daily basis to avoid moisture stress. Plants from each population were harvested at the soil level and dried at 65 C for 5 d to determine the aboveground shoot dry weight. The aboveground shoot dry weight reduction (%) was calculated as a percentage of the nontreated control using Equation 1.
 $$\;{\rm{Shoot}}\,{\rm{dry}}\,{\rm{biomass}}\,{\rm{reduction}}\,\left( {\rm{\% }} \right) = \left[ {{{{\rm{NTP}} - {\rm{TP}}} \over {{\rm{NTP}}}}} \right]{\rm{*}}100$$
$$\;{\rm{Shoot}}\,{\rm{dry}}\,{\rm{biomass}}\,{\rm{reduction}}\,\left( {\rm{\% }} \right) = \left[ {{{{\rm{NTP}} - {\rm{TP}}} \over {{\rm{NTP}}}}} \right]{\rm{*}}100$$
where NTP is the aboveground shoot dry weight from nontreated plants (average of 15 plants), and TP is the aboveground shoot dry weight of each treated plant.
Effectiveness of Postemergence Herbicides for GR Lolium multiflorum Control
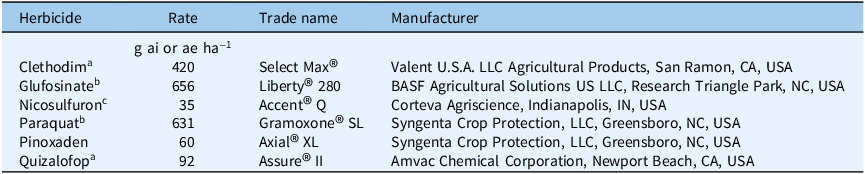
Greenhouse experiments were conducted at Cornell University’s Guterman Bioclimatic Facility in Ithaca, NY, during the summer of 2024 and repeated in the summer/fall of 2025 to evaluate the effectiveness of alternative postemergence herbicides for controlling GR L. multiflorum populations. Seedlings from all selected L. multiflorum populations (NY_R1, NY_R2, NY_R3, and AR_S) were grown in 10-cm-square plastic pots under the same growth conditions described in the glyphosate dose–response study. Experiments were set up in a randomized complete block (blocked by population) design with 9 replications (1 plant per pot = 1 replication). Information on tested postemergence herbicides, their rates, and adjuvants is summarized in Table 1. Actively growing seedlings (4- to 6-leaf stage) from each L. multiflorum population were treated with selected postemergence herbicides using a stationary cabinet spray chamber as previously described. At 21 d after treatment (DAT), the percent control of GR and GS L. multiflorum populations were visually assessed on a 0% to 100% scale (with 0% meaning no control and 100% meaning plant death). In addition, the aboveground shoot dry weights of all L. multiflorum populations were determined at 21 DAT and were expressed as a percentage of the nontreated control using Equation 1.
List of postemergence herbicides tested for controlling glyphosate-resistant and glyphosate-susceptible Lolium multiflorum populations in a greenhouse study at Cornell University, Ithaca, NY, USA.

a Nonionic surfactant (NIS) at 0.25 % v/v was included.
b Ammonium sulfate (AMS) at20 g L−1 was included.
c Methylated seed oil (MSO) at 0.5 % v/v was included.
Statistical Analysis
Due to nonsignificant treatment-by-experimental run interactions (P ≥ 0.05), data for glyphosate dose–response and alternative postemergence herbicide experiments were pooled across experimental runs before analysis. Data from both experiments were separately subjected to ANOVA using PROC MIXED in SAS software (SAS v. 9.3) to test the significance of fixed effects (L. multiflorum population, herbicide treatment, i.e., glyphosate dose or alternative postemergence herbicide, and their interactions). The random effects in the ANOVA model included replications and interactions involving replications with fixed effects. The ANOVA assumptions for data from the glyphosate dose–response study were checked using Shapiro-Wilk and Levene tests with PROC UNIVARIATE and GLM, and all data met the ANOVA assumptions. For the glyphosate dose–response study, the shoot dry weight reduction (% of nontreated) of each L. multiflorum population was fit against glyphosate doses using a four-parameter log-logistic model (Ritz et al. Reference Ritz, Baty, Streibig and Gerhard2015):
where y is the shoot dry weight reduction (% of nontreated), d is the maximum shoot dry weight reduction, c is the lower limit, e is the glyphosate dose required for 50% reduction in shoot dry weights (indicated as GR50 values, respectively), x is the glyphosate dose, and b represents the slope of each curve. The Akaike information criterion (AIC, value of 1,742) and a lack-of-fit test (P > 0.05) were used to select and confirm that the selected model described the shoot dry weight reduction of each L. multiflorum population (Ritz et al. Reference Ritz, Baty, Streibig and Gerhard2015). All estimated nonlinear regression parameters, standard errors, and GR90 values (glyphosate dose required for 90% reduction in shoot dry weights) were determined using the drc package in R software (v. 4.3.0; R Core Team 2023). The level of glyphosate resistance (indicated as R/S ratio) in each L. multiflorum population from New York was estimated by dividing the GR50 value for each GR L. multiflorum population from New York by the GR50 value of the AR_S population.
Data on percent visual control and shoot dry weight reduction (% of the nontreated) from the alternative postemergence herbicide experiment were arcsine square-root transformed before analysis to improve normality. Normal distribution was used for data analyses. We tried various data transformations (log, square root, Box-Cox, etc.) to improve the normality of the data. Among these transformations, only square-root transformation helped to improve the normality of data. Data were back-transformed for presentation, and means are reported in tables based on interpretations of the transformed values. Means were separated using Fisher’s protected LSD test at α = 0.05. We used the LSD after a significant ANOVA test to compare every pair of treatment means, because the overall F-test from ANOVA was significant. The LSD test also helps to calculate minimum differences needed to declare two statistically distinct means at a given alpha level (e.g., 0.05).
Determination of EPSPS Gene Copy Number
DNA Extraction
In the fall of 2025, seedlings from NY_R1, NY_R3 and AR_S L. multiflorum populations were grown in the greenhouse at Cornell University Guterman Bioclimatic Facility in Ithaca, NY, following the same procedure and growing conditions as mentioned in the glyphosate dose–response experiment. Actively growing seedlings (4- to 6-leaf stage) from NY_R1 and NY_R3 populations were treated with glyphosate (2,552 g ha−1) using the cabinet spray chamber as described earlier. Seedlings of the AR_S population were kept untreated. At 21 DAT, fresh leaf tissues were collected from three AR_S plants and three plants from NY_R1 and NY_R3 populations that survived the 2,552 g ha−1 dose of glyphosate. The collected leaf tissues from all three populations were shipped overnight to the University of Florida, Tropical Research and Education Center, Homestead, FL. Genomic DNA (gDNA) was extracted from leaf tissue of three L. multiflorum populations using the standard cationic detergent cetyltrimethylammonium bromide (CTAB) extraction method (Rieley et al. Reference Rieley, Kumar, Aulakh, Stanyard, Jangra, Singh and Price2025). The quality of the extracted DNA was assessed using 0.8% agarose gel electrophoresis and was quantified using a NanoDrop 1000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). The NY_R2 population was not included in the EPSPS gene copy number determination assay due to limited seed availability at the time of experiments.
EPSPS Gene Copy Number
Using the DNA samples of NY_R1, NY_R3, and AR_S plants, the real-time quantitative polymerase chain reaction (qPCR) was performed to determine the EPSPS gene copy number using ALS as a reference (single-copy) gene. Primers described by Salas et al. (Reference Salas, Dayan, Pan, Watson, Dickson, Scott and Burgos2012), EPSPS_F2 (5′-CTGATGGCTGCTCCTTTAGCTC-3′) and EPSPS_R2 (5′-CCCAGCTATCAGAATGCTC TGC-3′), were used to amplify a 136-bp fragment of the EPSPS gene. Similarly, primers reported by Ribeiro et al. (Reference Ribeiro, Berry, Creed, Barroso, Mallory Smith and Gallagher2025), ALS_1568_F (5′-ATTGATGGGG ATGGTAGCTT-3′) and ALS_1707_R (5′-GCCTTGTAAAACCTGTCCTC-3′), were used to amplify a 140-bp fragment of the ALS gene. The qPCR assay used QuantStudio 3 (Thermo Fisher Scientific) real-time PCR. The 20-μl qPCR reaction mixture comprised: 10 μl of Luna® Universal qPCR Master Mix (2X, New England Biolabs, Inc., Ipswich, MA, USA), 0.5 µl each of forward and reverse primers (10 μM, Integrated DNA Technologies, Coralville, IA, USA), 2 μl of gDNA (20 ng μl−1), and 7 μl of nuclease-free water to make up the volume. Three biological and three technical replicates were performed for each L. multiflorum population. The following qPCR reaction conditions were used: initial denaturation at 95 C for 10 min, followed by 40 cycles of denaturing at 95 C for 30 s, and then annealing and extension at 57 C for 1 min. The relative EPSPS genomic copy numbers were calculated as ΔCt = CTALS – CTEPSPS, and the EPSPS copy number was shown as 2ΔCt. The 2−ΔΔCt method was used to quantify the EPSPS gene copy number relative to the ALS gene (Gaines et al. Reference Gaines, Zhang, Wang, Bukun, Chrisholm, Shaner, Nissen, Patzoldt, Tranel, Culpepper, Grey, Webster, Vencill, Sammons and Jiang2010). The EPSPS gene copy number in GR populations was represented as fold change relative to the susceptible population (AR_S).
Results and Discussion
Glyphosate Dose–Response
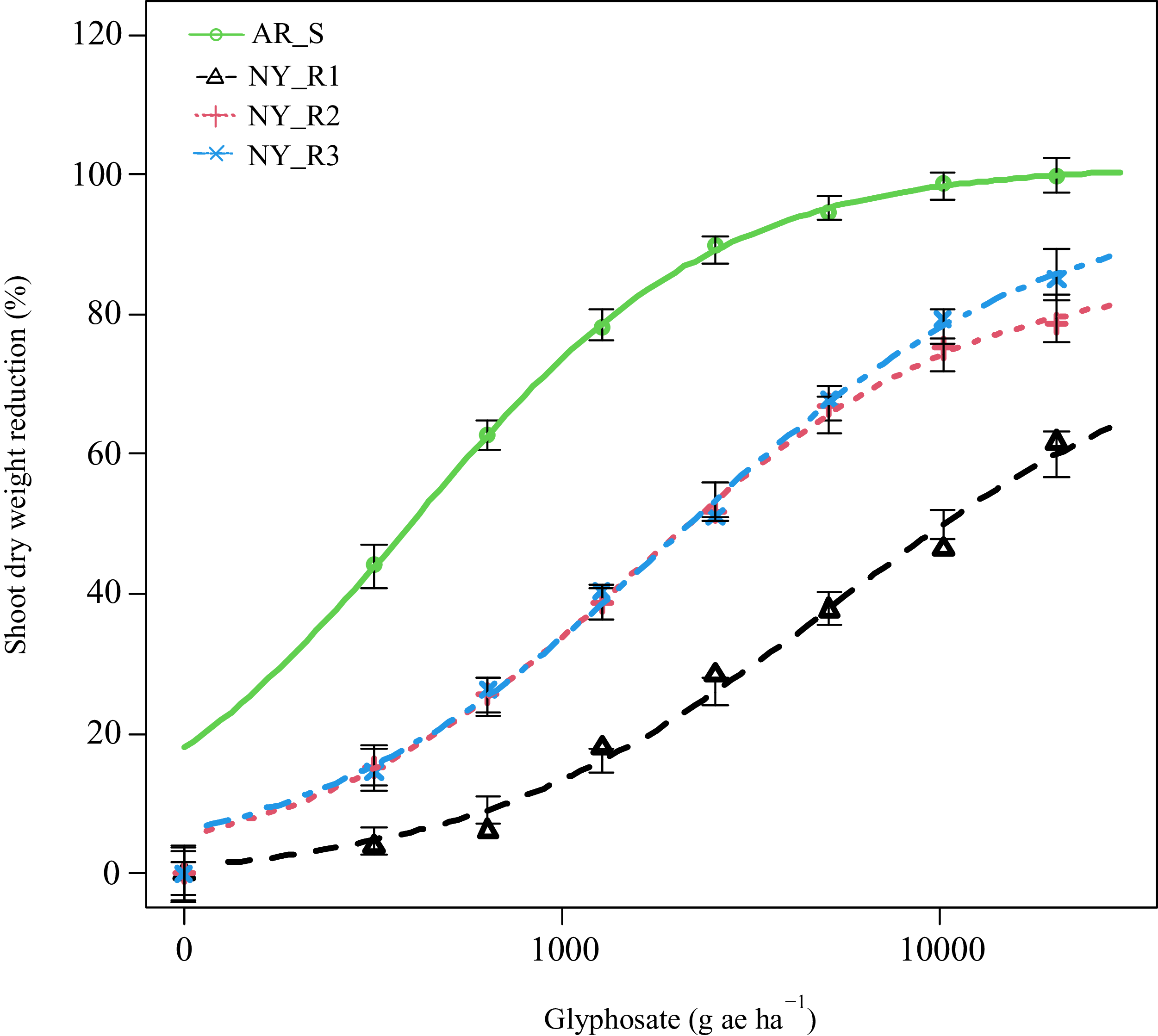
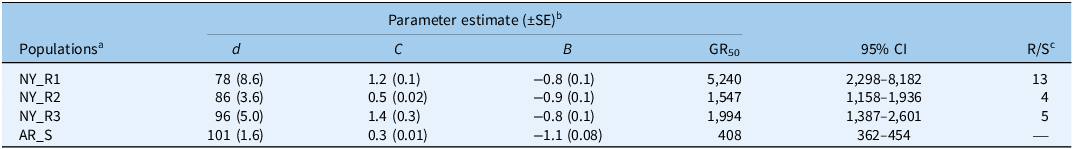
All three suspected L. multiflorum populations from New York survived glyphosate applied at the field use rate (1,276 g ha−1) and at higher rates at 21 DAT, confirming resistance to glyphosate (Figure 2; Table 2). Based on the fitted model, the estimated glyphosate doses (GR50 values) for 50% reduction in shoot dry weights of all three L. multiflorum populations ranged from 1,547 to 5,240 g ha−1, which were significantly higher than that of the AR_S population (408 g ha−1). Based on the GR50 values, the NY_R1 population exhibited a 13-fold (R/S ratio) resistance to glyphosate compared with the AR_S population (Table 2; Figure 2). Similarly, the NY_R2 and NY_R3 populations had 4- and 5-fold resistance to glyphosate, respectively, when compared with the AR_S population (Figure 2; Table 2). It is important to note that all three fields where L. multiflorum populations were collected were under continuous wheat–corn–soybean rotation (3-yr) for >5 yr, with L. multiflorum grown as cover crop and terminated with glyphosate every year. In addition, these growers were relying solely on glyphosate applications for weed control as preplant burndown, during the growing season in GR corn and soybean, and/or as postharvest cleanup. It is also speculated that resistance to glyphosate in these New York populations was most likely introduced through purchased cover crop seed (probably already resistant to glyphosate) of L. multiflorum. The combined effects of multiple factors, including the use of contaminated L. multiflorum seed with mixed varieties and the repeated, continuous use of glyphosate may have contributed to the occurrence of GR L. multiflorum populations. Supporting this hypothesis, we tested a seed source of L. multiflorum used by a farmer in Livingston County and found that seedlings grown from the seed bag survived a field use rate (1,260 g ha−1) of glyphosate under greenhouse conditions (VK, personal observations). These findings highlight the importance of routinely testing L. multiflorum cover crop seed for herbicide resistance before commercialization. Equally important is educating farmers to use only certified L. multiflorum seed varieties intended for cover crop or forage production.
Shoot dry weight reduction (% of nontreated) of selected Lolium multiflorum populations at 21 d after treatment (DAT) with various doses of glyphosate in greenhouse experiments conducted in 2024 and 2025. Symbols indicate actual values of shoot dry weights (% of nontreated), and lines indicate predicted values of shoot dry weights (% of nontreated) obtained from the four-parameter log-logistic model. Vertical bars indicate model-based SEs (±) of the predicted means. AR_S, glyphosate-susceptible Lolium multiflorum population from Arkansas; NY_R1, glyphosate-resistant Lolium multiflorum population from Livingston County, NY; NY_R2, glyphosate-resistant Lolium multiflorum population from Ontario County, NY; NY_R3, glyphosate-resistant Lolium multiflorum population from Genesse County, NY.

Regression parameter estimates from the four-parameter log-logistic model fit to shoot dry weight reduction (% of nontreated) of suspected glyphosate-resistant (GR) Lolium multiflorum populations from New York State and a known glyphosate-susceptible (GS) population from Arkansas at 21 d after treatment (DAT) with various doses of glyphosate in greenhouse experiments conducted in 2024 and 2025 at Cornell University in Ithaca, NY, USA.

a Abbreviations: NY_R1, NY_R2, and NY_R3 are suspected GR L. multiflorum populations collected from Livingston, Ontario, and Genessee counties of New York State, respectively; AR_S is a known GS L. multiflorum population from Arkansas.
b Parameter d is the upper limit, c is the lower limit, b is the slope of each curve, and GR50 is the effective dose (g ha−1) of glyphosate required for 50% shoot dry biomass reduction (% of nontreated) for each L. multiflorum population. CI is the confidence interval.
c R/S is the ratio of the GR50 value of each suspected GR L multiflorum population from New York State relative to that of the GR50 value of a susceptible population from Arkansas.
The levels of glyphosate resistance observed in current study are comparable to those reported from other regions of the world. For instance, the 4- to 15-fold resistance observed in GR L. multiflorum populations from New York aligns with the first report of glyphosate resistance in four rigid ryegrass (Lolium rigidum Gaudin) populations in Australia (7- to 11-fold), where resistance was associated with reduced translocation of glyphosate to meristematic zones in plants (Powles et al. Reference Powles, Lorraine-Colwill, Dellow and Preston1998; Wakelin et al. Reference Wakelin, Lorraine-Colwill and Preston2004). Similarly, Jasieniuk et al. (Reference Jasieniuk, Ahmad, Sherwood, Firestone, Perez-Jones, Lanini, Mallory-Smith and Stednick2008) previously confirmed 2- to 15-fold resistance to glyphosate among four L. multiflorum populations collected from a diversity of crops (e.g., almonds [Prunus dulcis (Mill.) D. A. Webb], walnuts [Juglans spp.], prunes [Prunus domestica L.], wine grapes [Vitis vinifera L.], tomatoes [Solanum lycopersicum L.], wheat, cotton) and non-cropped areas (e.g., roadsides and fallow fields) in northern California. One of these GR populations from California revealed a Pro-106-Ser mutation, while plants from three other GR populations showed Pro-106-Ala mutations (Jasieniuk et al. Reference Jasieniuk, Ahmad, Sherwood, Firestone, Perez-Jones, Lanini, Mallory-Smith and Stednick2008). Similarly, Perez-Jones et al. (Reference Perez-Jones, Park, Colquhoun, Mallory-Smith and Shaner2005) reported the first U.S. case of GR L. multiflorum in Oregon hazelnut (Corylus avellana L.) orchards (where glyphosate was repeatedly used for weed control), demonstrating ∼5-fold resistance due to reduced glyphosate translocation. More recently, Ribeiro et al. (Reference Ribeiro, Berry, Creed, Barroso, Mallory Smith and Gallagher2025) documented a GR L. multiflorum population from a fallow field in Oregon (previously cropped with perennial ryegrass [Lolium perenne L.] where glyphosate was repeatedly used as preplant burndown) showing a 9-fold resistance due to EPSPS gene amplification (average of 30 EPSPS gene copies). In contrast to our findings, Nandula et al. (Reference Nandula, Poston, Eubank, Koger and Reddy2007) reported low-level (≈3-fold) glyphosate resistance in L. multiflorum populations from continuous GR cotton–soybean cropping systems in Mississippi, attributed primarily to reduced glyphosate translocation, with reduced absorption observed in one population. Furthermore, a L. multiflorum population from Desha County, AR, was found to be 23-fold resistant to glyphosate, primarily due to EPSPS gene amplification (average of 25 EPSPS gene copies) (Dickson et al. Reference Dickson, Scott, Burgos, Salas and Smith2011; Salas et al. Reference Salas, Dayan, Pan, Watson, Dickson, Scott and Burgos2012). GR L. multiflorum populations have also been documented from major agronomic crops (wheat, soybean, cotton) in the U.S. Midsouth and Southeast, including Louisiana, North Carolina, and Tennessee (Heap Reference Heap2025). More recently, Yadav et al. (Reference Yadav, Russell, Ganie, Patel, Price and Maity2025) reported 14% of a total of 44 tested L. multiflorum populations collected from wheat, soybean, peanut (Arachis hypogaea L.), cotton, and cornfields, as well as field borders, in Alabama were potentially resistant to glyphosate (21% to 79% injury). Glyphosate was frequently used in preplant burndown, in-season, or postharvest situations in these fields. Furthermore, a point mutation (Pro-106-Ser) in the EPSPS gene conferring up to 192-fold resistance was also observed in one GR L. multiflorum population (Yadav et al. Reference Yadav, Russell, Ganie, Patel, Price and Maity2025).
Effectiveness of Alternative Postemergence Herbicides
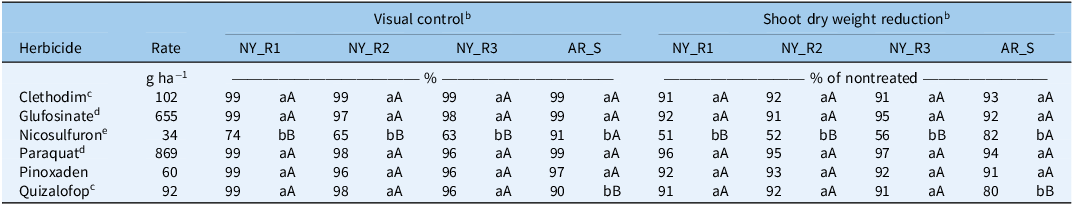
The GR L. multiflorum populations from New York responded similarly to alternative postemergence herbicides, as indicated by visual control ratings and shoot dry weight reductions (Table 3). Clethodim, glufosinate, paraquat, pinoxaden, and quizalofop were highly effective, with 96% to 99% control of tested GR L. multiflorum populations from New York at 21 DAT (Table 3). With nicosulfuron, visual control did not exceed 74% for the GR L. multiflorum populations; however, it was 91% for the AR_S population, indicating the latter was also susceptible to ALS-inhibiting herbicides. It is important to note that all alternative postemergence herbicides tested have L. multiflorum already listed on their labels. Our results are consistent with the findings of de Sanctis et al. (Reference de Sanctis, Cahoon, Everman, Gannon, Jennings and Taylor2025), who recently reported average control of 92%, 93%, 98%, and 73% at 28 DAT with paraquat, glufosinate, clethodim, and nicosulfuron, respectively, across 38 L. multiflorum populations from North Carolina. The inadequate control of all three GR L. multiflorum populations with nicosulfuron further suggests the potential resistance to ALS-inhibiting herbicides in these populations. The evolution of L. multiflorum populations with multiple resistance to glyphosate and ALS-inhibiting herbicides has already been reported in Alabama, California, and North Carolina (Heap Reference Heap2025; Jones et al. Reference Jones, Taylor and Everman2021; Yadav et al. Reference Yadav, Russell, Ganie, Patel, Price and Maity2025). In addition, L. multiflorum populations with confirmed resistance to clethodim, fluazifop-butyl, imazamox, mesosulfuron, paraquat, pinoxaden, and pyroxsulam have also been reported elsewhere (Brunharo and Hanson Reference Brunharo and Hanson2018; de Sanctis et al. Reference de Sanctis, Cahoon, Everman, Gannon, Jennings and Taylor2025; Heap Reference Heap2025; Salas et al. Reference Salas, Burgos, Mauromoustakos, Lassiter, Scott and Alcober2013).
Percent visual control and shoot dry weight reduction (% of nontreated) of Lolium multiflorum populations from New York State and Arkansas with various postemergence herbicides at 21 d after treatment (DAT) in greenhouse experiments conducted in 2024 and 2025 at Cornell University, Ithaca, NY, USA. a

a Abbreviations: AR_S, glyphosate-susceptible Lolium multiflorum population from Arkansas; NY_R1, glyphosate-resistant Lolium multiflorum population from Livingston County, NY; NY_R2, glyphosate-resistant Lolium multiflorum population from Ontario County, NY; NY_R3, glyphosate-resistant Lolium multiflorum population from Genesse County, NY.
b Means followed by the same lowercase letters within each Lolium multiflorum population indicate no statistical difference according to Fisher’s protected LSD test (P < 0.0001), whereas means followed by the same uppercase letters within each herbicide treatment indicate no statistical difference according to Fisher’s protected LSD test (P < 0.0001).
c Nonionic surfactant (NIS) at 0.25 % v/v was included.
d Ammonium sulfate (AMS) at 20 g L−1 was included.
e Methylated seed oil (MSO) at 0.5 % v/v was included.
Consistent with visual control, shoot dry weight reductions followed a similar pattern with all tested postemergence herbicides across all L. multiflorum populations (Table 3). The majority of tested postemergence herbicides significantly reduced shoot dry weights (91% to 97% of nontreated) of three tested GR L. multiflorum populations (Table 3). With nicosulfuron, the shoot dry weight reduction of three GR L. multiflorum populations was only 51% to 56%. Among all tested postemergence herbicides, clethodim, glufosinate, paraquat, and pinoxaden reduced shoot dry weights (% of nontreated) of the AR_S population by 91% to 94% (Table 3), which was higher than shoot dry weight reduction with nicosulfuron (82%) and quizalofop (80%).
Clethodim, glufosinate, paraquat, and quizalofop are currently effective postemergence options for managing the tested GR L. multiflorum populations in New York cropping systems. To date, the absence of resistance to these sites of action in New York populations is largely attributable to limited use of these chemistries for L. multiflorum management. Our results show that soybean growers in New York confronting GR L. multiflorum can make a spring preplant burndown treatment of glufosinate or paraquat. Subsequently, clethodim, glufosinate, or quizalofop may be applied early to mid-postemergence in glufosinate-tolerant soybean varieties for managing GR L. multiflorum populations. Similarly, corn growers in New York may apply glufosinate or paraquat as a burndown treatment in early spring. Additionally, early to mid-postemergence application of glufosinate in glufosinate-tolerant corn or quizalofop in 2,4-D/glufosinate/glyphosate-resistant corn (Enlist corn®, Corteva™ Agriscience, Indianapolis, USA) can help control GR L. multiflorum. Wheat growers may use pinoxaden in conventional wheat varieties or quizalofop in quizalofop-tolerant wheat (CoAXium® Wheat Production System (a joint, non-GMO wheat production system developed by Albaugh, LLC, the Colorado Wheat Research Foundation (CWRF), and Limagrain Cereal Seeds)) for L. multiflorum control in New York. In addition, a fall tillage, where possible, can also help control first flush of small L. multiflorum seedlings and may be considered in combination with chemical control strategies in wheat.
EPSPS Gene Copy Numbers
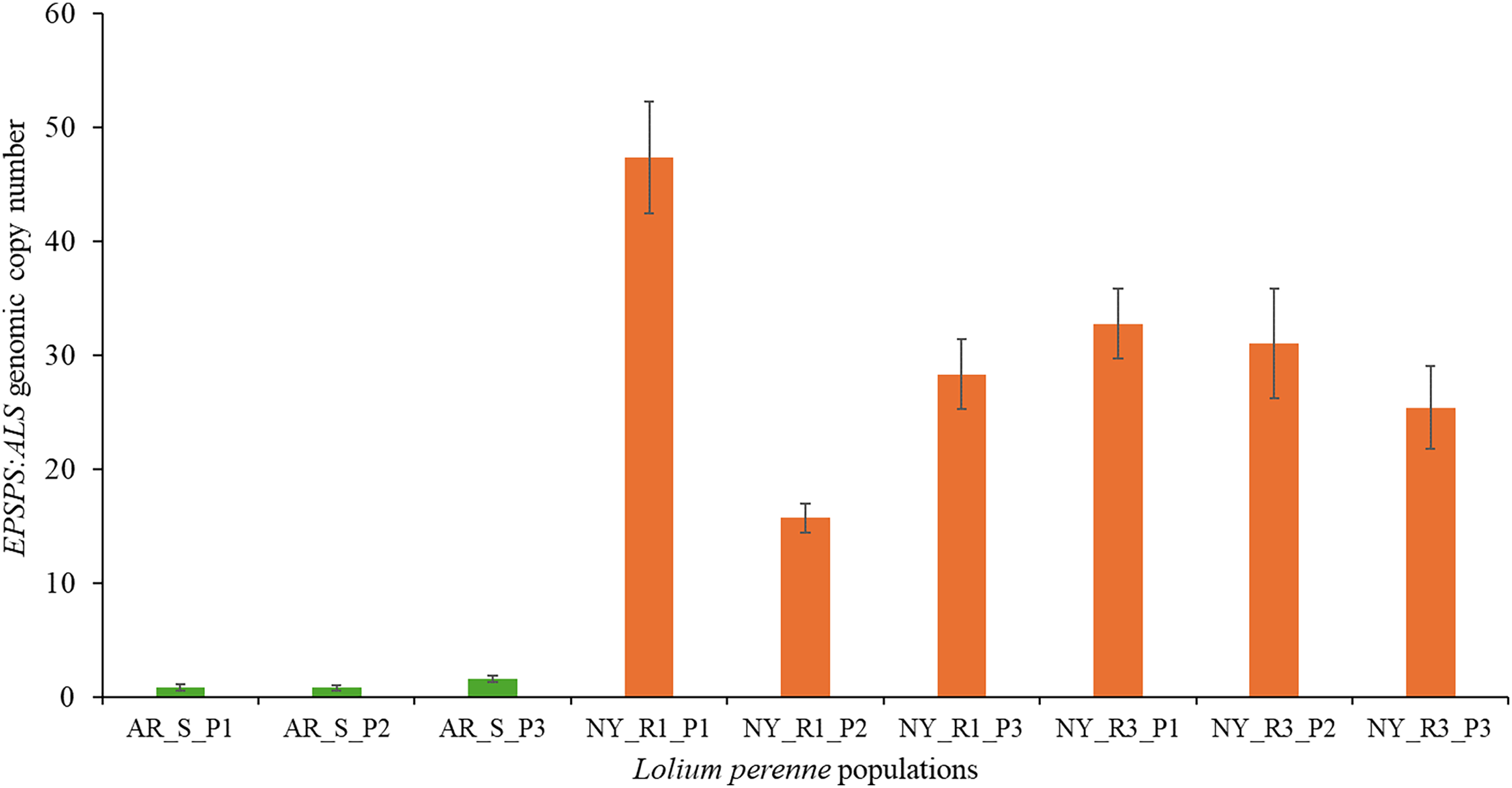
The qPCR assays revealed that three plants from the GS L. multiflorum population (AR_S) had EPSPS gene copy numbers ranging from 1 to 1.5 (Figure 3). In contrast, the NY_R1 population from Livingston County showed relatively higher EPSPS gene copy numbers, with individual plants exhibiting 16 to 47 copies (Figure 3). The NY_R3 population from Genesee County also exhibited higher EPSPS gene copy numbers (average of 30 copies), with individual plants showing 25 to 33 copies (Figure 3). Evidently, the average EPSPS gene copy number in the NY_R1 and NY_R3 populations (30 copies) was approximately 30-fold higher than that of the AR_S population.
EPSPS gene copy numbers relative to the ALS reference gene in glyphosate-susceptible (AR_S) from Arkansas and glyphosate-resistant Lolium multiflorum populations (NY_R1, NY_R3) from New York.

These results are consistent with those of Salas et al. (Reference Salas, Dayan, Pan, Watson, Dickson, Scott and Burgos2012), who previously reported a GR L. multiflorum population from Desha County, AR, with up to 25 EPSPS gene copies conferring 12- to 13-fold resistance to glyphosate. Our results also align with those of Ribeiro et al. (Reference Ribeiro, Berry, Creed, Barroso, Mallory Smith and Gallagher2025), who recently reported a 30-fold increase in the EPSPS gene copies in a GR L. multiflorum population from Oregon conferring 9-fold resistance to glyphosate. In contrast to findings in the current study, Salas et al. (Reference Salas, Scott, Dayan and Burgos2015) reported 11 to >100 copies of the EPSPS gene causing 7- to 19-fold resistance to glyphosate among 6 GR L. multiflorum populations from Arkansas. We did not perform any correlation analysis between glyphosate resistance level and EPSPS gene copy numbers found in these GR L. multiflorum populations. However, inconsistency found between the glyphosate resistance levels and the EPSPS gene copy numbers was probably due to different individual seedlings of GR L. multiflorum used in the glyphosate dose–response study and qPCR assays. Previous studies have reported a positive correlation between glyphosate resistance level and EPSPS gene copy numbers in GR L. multiflorum populations (Ribeiro et al. Reference Ribeiro, Berry, Creed, Barroso, Mallory Smith and Gallagher2025; Salas et al. Reference Salas, Dayan, Pan, Watson, Dickson, Scott and Burgos2012). In these studies, the same seedlings were used in both glyphosate dose–response studies and the EPSPS gene copy number estimation. Alternative mechanisms of glyphosate resistance have been reported in GR L. multiflorum populations, including point mutations in the EPSPS gene (Pro-106-Ser or Pro-106-Ala) and alterations in glyphosate absorption and/or translocation (Jasieniuk et al. Reference Jasieniuk, Ahmad, Sherwood, Firestone, Perez-Jones, Lanini, Mallory-Smith and Stednick2008; Nandula et al. Reference Nandula, Poston, Eubank, Koger and Reddy2007; Perez-Jones et al. Reference Perez-Jones, Park, Colquhoun, Mallory-Smith and Shaner2005; Yadav et al. Reference Yadav, Russell, Ganie, Patel, Price and Maity2025). To our knowledge, there is currently no previous report of GR L. multiflorum populations with both target site–based mechanisms (point mutation and EPSPS gene amplification). However, there are populations of GR L. multiflorum being reported with both target site–based (point mutation in the EPSPS gene) and non–target site based (altered translocation) mechanisms in Australia (Preston et al. Reference Preston, Wakelin, Dolman, Bostamam and Boutsalis2009).
Results from the present research confirm the first case of glyphosate resistance among L. multiflorum populations identified from New York. In addition, these confirmed GR L. multiflorum populations also exhibited potential resistance to nicosulfuron, which requires further investigation. However, several postemergence herbicides such as clethodim, glufosinate, pinoxaden, and paraquat effectively controlled all three GR L. multiflorum populations. In addition to these effective postemergence herbicide options, growers should diversify their crop rotations by integrating perennial forage crops (such as alfalfa [Medicago sativa L.]) that can further help in depleting the seedbank of GR L. multiflorum populations through frequent cuttings of alfalfa. The use of novel mechanical weed control tactics, including harvest seed control (seed impact mill and chaff lining) in combination with effective herbicide programs can also help in managing the seedbank of GR L. multiflorum populations in wheat-based cropping systems (Maity et al., Reference Maity, Young and Schwartz-Lazaro2022). Quantitative PCR assays revealed approximately 30-fold higher EPSPS gene copy numbers in two GR L. multiflorum populations from New York compared with the AR_S population. However, it is important to note that this study did not assess other potential target site–based mechanisms (e.g., mutations in the EPSPS gene) or non–target site based mechanisms (e.g., altered glyphosate absorption or translocation) in these GR L. multiflorum populations. Investigation of these additional potential resistance mechanisms is warranted. The EPSPS gene amplification creates excess enzyme, often inherited as a largely dominant, single-gene trait. Similarly, resistance via altered translocation of glyphosate via sequestration or target site–based mutations can be inherited as a dominant single gene (Preston et al. Reference Preston, Wakelin, Dolman, Bostamam and Boutsalis2009). Some populations show complex inheritance, with resistance linked to reduced translocation appearing multigenic, not just the result of a single gene. Furthermore, both non–target site and target-site (point mutations) mechanisms of glyphosate resistance can render Lolium spp. at a competitive disadvantage. For instance, a significant fitness penalty (reduced growth and less seed production up to 48%) associated with the glyphosate resistance allele in some GR L. multiflorum populations has been reported (Fernández-Moreno et al., Reference Fernández-Moreno, Alcántara-de la Cruz, Smeda and De Prado2017). Nevertheless, the inheritance pattern and associated fitness cost (if any) of glyphosate resistance via EPSPS gene amplification in GR L. multiflorum populations from New York are unknown and warrant investigation. Future studies should also assess forage and cover crop seed sources of L. multiflorum for the presence of herbicide-resistance traits and emphasize the use of certified varieties among growers. We are also planning to collect and screen additional L. multiflorum populations across New York and other northeastern states to determine the distribution and frequency of herbicide resistance in the region. Multi-location field studies will also be conducted to investigate various integrated weed management tactics, including effective preemergence and postemergence herbicides, cereal rye (Secale cereale L.) cover crops, and harvest weed seed control for managing GR L. multiflorum populations and preventing its spread.
Acknowledgments
We thank Preetaman Bajwa, Henrique Scatena, and Anas Ashfaq for their technical assistance in greenhouse experiments.
Funding statement
We thank the National Institute of Food and Agriculture, U.S. Department of Agriculture, Hatch Project (2024-25-142) for providing financial support to conduct this research work.
Competing interests
The authors declare no conflicts of interest.