Introduction
Rocky intertidal areas are diverse and productive ecosystems, providing valuable habitat for both adult and juvenile fish populations (Compaire et al., Reference Compaire, Cabrera, Gómez-Cama and Soriguer2016; Dias et al., Reference Dias, Silva, Cabral and Vinagre2014). Historically, most attention has been given to sandy beaches and mudflats in tropical and temperate areas as nursery grounds for fish (Gibson, Reference Gibson1973; Marley et al., Reference Marley, Deacon, Phillip and Lawrence2020; Tse et al., Reference Tse, Nip and Wong2008). However, there is growing evidence of the importance of the rocky intertidal zone as a fish nursery (Dias et al., Reference Dias, Roma, Fonseca, Pinto, Cabral, Silva and Vinagre2016, Reference Dias, Silva, Cabral and Vinagre2014; Studebaker, Reference Studebaker2006; Studebaker et al., Reference Studebaker, Cox and Mulligan2009). Intertidal fish communities have been studied extensively worldwide, particularly within rocky estuaries (Carminatto et al., Reference Carminatto, Rotundo, Butturi-Gomes, Barrella and Petrere Junior2020) and tidepools (Bezerra et al., Reference Bezerra, Padial, Mariano, Garcez and Sánchez-Botero2017; White et al., Reference White, Hose and Brown2015), but much of this work has been in lower latitudes (Choat, Reference Choat1982; Dias et al., Reference Dias, Silva, Cabral and Vinagre2014). In contrast, our understanding of fish assemblages at higher latitudes remains limited (Gibson, Reference Gibson1972; Henriques and Almada, Reference Henriques and Almada1998; Pihl and Wennhage, Reference Pihl and Wennhage2002). These little studied systems are subjected to several anthropogenic stressors such as pollution (Crowe et al., Reference Crowe, Thompson, Bray and Hawkins2000), introduced alien species (Magnússon et al., Reference Magnússon, Hawkins, Gunnarsdóttir, Svavarsson and Gunnarsson2024), and extensive exploitation of living resources (Boaden and Dring, Reference Boaden and Dring1980), all of which are exacerbated by overarching effects of global change (Hawkins et al., Reference Hawkins, O’Shaughnessy, Branch, Airoldi, Bray, Brooks, Burrow, Castilla, Crow, Davies, Firth, Hiscock, Jenkins, Knights, Langmead, Leung, Mieszkowska, Moschella, Steyl, Tidau, Whittington and Thompsonin press; Thompson et al., Reference Thompson, Crowe and Hawkins2002).
Rocky shores, particularly sheltered rocky shores, provide abundant refuges, such as crevices, rockpools, and spaces beneath boulders and algae, hosting both transient juvenile marine fish species that use these habitats as nurseries (Henriques and Almada, Reference Henriques and Almada1998) and resident intertidal fish species (Horn et al., Reference Horn, Martin and Chotkowski1998). Amongst the transient species are cod (Gadus morhua Linnaeus, 1758) and saithe (Pollachius virens [Linnaeus, 1758]), two common species of boreal waters of the North Atlantic (Dutil and Brander, Reference Dutil and Brander2003; ICES, 2020; Saha et al., Reference Saha, Hauser, Kent, Planque, Neat, Kirubakaran, Huse, Homrum, Fevolden, Lien and Johansen2015) of commercial importance (Astthorsson et al., Reference Astthorsson, Gislason and Jonsson2007). Various aspects of the ecology of these species have been well studied, such as distribution (Armannsson et al., Reference Armannsson, Jonsson, Neilson and Marteinsdottir2007; Begg and Marteinsdottir, Reference Begg and Marteinsdottir2002; Rose et al., Reference Rose, deYoung, Kulka, Goddard and Fletcher2000), abundance (McCain et al., Reference McCain, Rangeley, Schneider and Lotze2016), feeding ecology (Jaworski and Ragnarsson, Reference Jaworski and Ragnarsson2006; Jónsson, Reference Jónsson1997; Nedreaas, Reference Nedreaas1987; Pálsson, Reference Pálsson1997), or foraging behaviour (Pálsson, Reference Pálsson1983), butvery few studies have focused on their use of the rocky intertidal zone (Elliott et al., Reference Elliott, Ahti, Heath, Turrell and Bailey2016; Pihl and Wennhage, Reference Pihl and Wennhage2002; Rangeley and Kramer, Reference Rangeley and Kramer1995a, Reference Rangeley and Kramer1995b). In contrast, more is known about resident species such as the shorthorn sculpin (Myoxocephalus scorpius [Linnaeus, 1758]), a common inhabitant of the Icelandic rocky intertidal zone (Jónsson and Pálsson, Reference Jónsson and Pálsson2006). It has various adaptations to deal with tide-out emersion and hypoxia in rock pools (Nonnotte and Kirsch, Reference Nonnotte and Kirsch1978; Sundell et al., Reference Sundell, Morgenroth, Ekström, Brijs, Axelsson, Gräns and Sandblom2021) and is a sit-and-wait predator that feeds on seaweed associated prey such as small crustaceans (Sawyer, Reference Sawyer1967; Shorty and Gannon, Reference Shorty and Gannon2013) and fish (Cardinale, Reference Cardinale2000; Dick et al., Reference Dick, Chambers and Gallagher2009; Hauksson, Reference Hauksson1999; Landry et al., Reference Landry, Fisk, Yurkowski, Hussey, Dick, Crawford and Kessel2018).
Ascophyllum nodosum (L.) Le Jolis is a large canopy-forming alga found on both sides of the North Atlantic (Jenkins et al., Reference Jenkins, Moore, Burrows, Garbary, Hawkins, Ingólfsson, Sebens, Snelgrove, Wethey and Woodin2008). It provides critical shelter and habitat for a diverse array of associated sessile and mobile species (Magnússon et al., Reference Magnússon, Hawkins, Gunnarsdóttir, Svavarsson and Gunnarsson2024; Pereira et al., Reference Pereira, Morrison, Shukla and Critchley2020; Phillippi et al., Reference Phillippi, Tran and Perna2014; Pocklington et al., Reference Pocklington, Jenkins, Bellgrove, Keough, O’Hara, Masterson-Algar and Hawkins2018). Increases in demand and access to large quantities of A. nodosum in Canada, Ireland, Scotland, Norway, and Iceland have led to commercial exploitation for various uses such as alginates, food, or fertilisers (Gunnarsson et al., Reference Gunnarsson, Burgos, Gunnarsdóttir, Egilsdóttir, Georgsdóttir and Pajuelo2019; Mac Monagail et al., Reference Mac Monagail, Cornish, Morrison, Araújo and Critchley2017; Pereira et al., Reference Pereira, Morrison, Shukla and Critchley2020). Ascophyllum nodosum has been commercially harvested in Breiðafjörður for several decades, with current extraction estimates ranging between 13,000 and 15,000 tonnes per annum (Icelandic Directorate of Fisheries, 2025). Mechanical harvesting in the area typically removes approximately 30–66% of the standing biomass, cutting the fronds above the holdfast to allow regrowth (Gunnarsdóttir, unpublished data). Intensive harvesting can exert profound effects on the targeted alga and associated biota (Fegley, Reference Fegley2001; Pereira et al., Reference Pereira, Morrison, Shukla and Critchley2020; Phillippi et al., Reference Phillippi, Tran and Perna2014; Seeley and Schlesinger, Reference Seeley and Schlesinger2012). Recovery rates vary depending on the extent and type of biomass removal. After complete removal, including holdfast complexes, regeneration can be particularly slow taking up to 20 years (Gendron et al., Reference Gendron, Merzouk, Bergeron and Johnson2018; Ingólfsson and Hawkins, Reference Ingólfsson and Hawkins2008; Jenkins et al., Reference Jenkins, Norton and Hawkins2004). In contrast, when a lower proportion of the biomass is harvested by cutting fronds above the holdfast, recovery is faster (Johnston et al., Reference Johnston, Mittelstaedt, Braun, Muhlin, Olsen, Webber and Klemmer2023; Lauzon-Guay et al., Reference Lauzon-Guay, Ugarte, Morse and Robertson2021; Ugarte et al., Reference Ugarte, Sharp, Moore, Anderson, Brodie, Onsøyen and Critchley2007).
The usage and role of fishes within canopy-forming fucoid assemblages such as A. nodosum beds in the rocky intertidal zone is also understudied (Rangeley, Reference Rangeley1994). Moreover, the impacts of harvesting large fucoids on mobile fish assemblages and populations have received little attention (Pereira et al., Reference Pereira, Morrison, Shukla and Critchley2020). Fish widely forage over the intertidal zone, thereby influencing community structure (Choat, Reference Choat1982), linking algae-dominated areas with other marine habitats, through migration (Fulton et al., Reference Fulton, Berkström, Wilson, Abesamis, Bradley, Åkerlund, Barrett, Bucol, Chacin, Chong‐Seng, Coker, Depczynski, Eggertsen, Eggertsen, Ellis, Evans, Graham, Hoey, Holmes, Kulbicki, Leung, Lam, Lier, Matis, Noble, Pérez‐Matus, Piggott, Radford, Tano and Tinkler2020; Kang et al., Reference Kang, Park, Choy, Choi, Hwang and Kim2015). Various studies have explored the role of fish in intertidal food webs through stomach content analysis (Da Silveira et al., Reference Da Silveira, Semmar, Cartes, Tuset, Lombarte, Ballester and Vaz-dos-santos2020), from which most current understanding of trophic ecology is derived (Braga et al., Reference Braga, Bornatowski and Vitule2012). These have yielded valuable insights into fish diet and food-webs within intertidal communities (Hadwen et al., Reference Hadwen, Russell and Arthington2007; Mendonça et al., Reference Mendonça, Flores, Silva and Vinagre2019).
Our overall aim was to investigate the fish assemblages across seasons in the Ascophyllum nodosum covered intertidal rocky shores in Breiðafjörður, Iceland, and experimentally assess impacts of commercial harvesting. We compared harvested areas with adjacent control areas using a multi-mesh netting approach, to characterise fish assemblage composition, population abundance, size/age structure and feeding ecology of the most caught resident species (M. scorpius) plus those using the intertidal as feeding or nursery grounds (G. morhua and P. virens). Thus, we explored whether current harvesting practices impact fish assemblages and the provision of ecosystem services such as feeding and nursery grounds.
Materials and methods
Study area and experimental design
The study was made between June 2019 and June 2020 at Borg, in the inner reaches of Breiðafjörður bay, a large semi-enclosed bay in North-West Iceland with a mean tidal amplitude at spring tide of 4.3 m.
Four experimental plots were randomly interspersed at the study site: two control plots in areas without A. nodosum harvesting (labelled C1 65°28.52 N, 22°00.91ʹ W and C2 65°28.48ʹ N, 22°00.88ʹ W), and two harvested plots (labelled H1 65°28.58ʹ N, 22° 00.91ʹ W and H2 65°28.46ʹ N, 22°00.91ʹ W) (Figure 1, Supp. Fig.1) using an Aquamarine™ floating aquatic plant harvesters operating in the area. Prior to harvesting, A. nodosum cover was visually assessed to be 100% in all plots. The study area had no prior history of commercial cutting, ensuring that any observed effects resulted from the experimental treatment. Harvesting was done once on the 1st of July 2019, following standard commercial practices. Commercial harvesting operations run from spring to autumn, using a rotation system in which areas are left untouched for 4–6 years between harvesting events. Harvesters will also typically leave large patches of untouched A. nodosum next to harvested areas. Both harvested plots were treated and cut equally, leaving the holdfasts intact, matching harvesting practices in the region (Gunnarsdóttir, unpublished data).
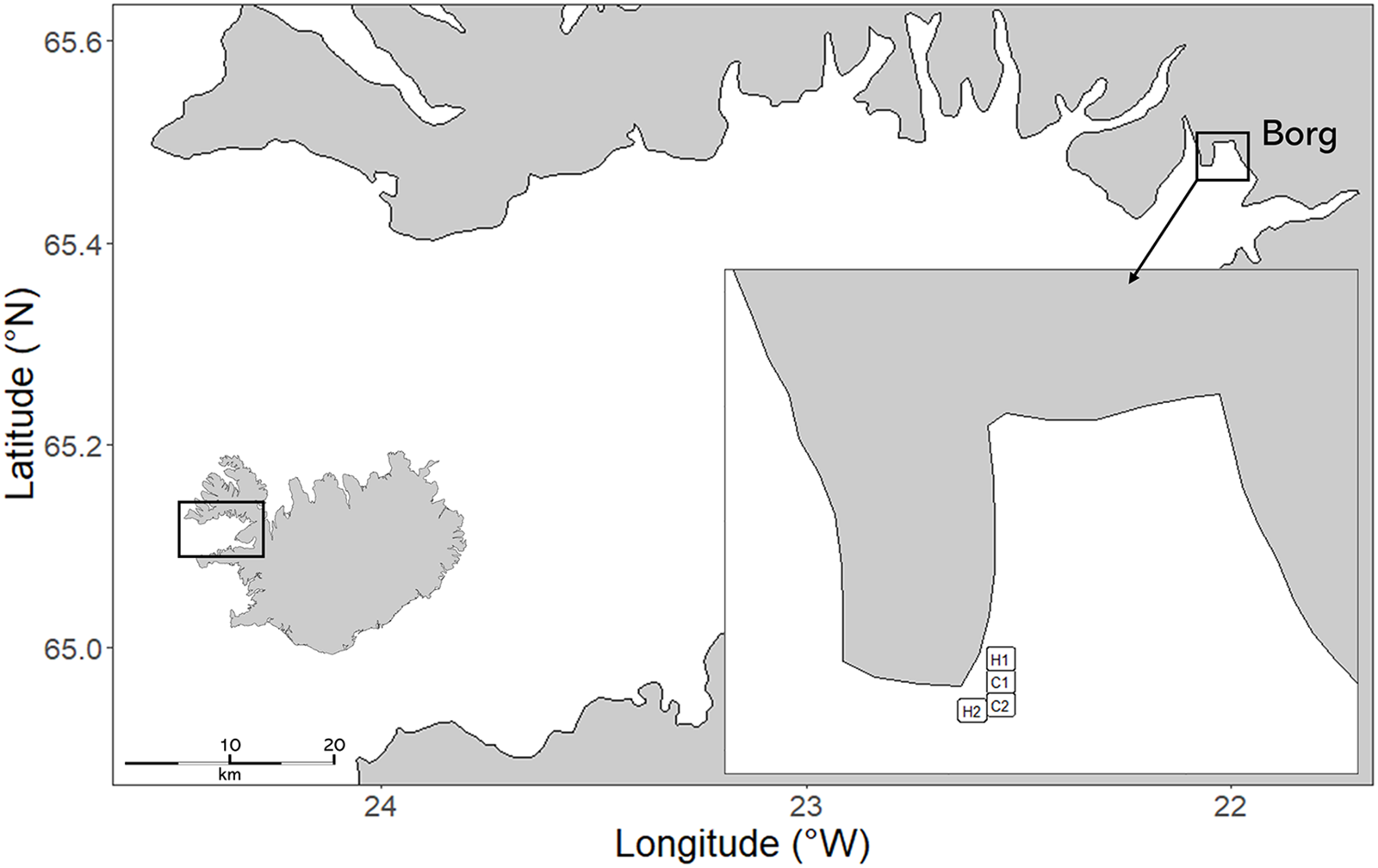
Figure 1. Breiðafjörður bay with insert showing the sampling area at Borg and all plots. Sampling plots included two harvested sites (H1 and H2) and two control sites (C1 and C2), all located within the same intertidal zone.
In each plot, two multi-mesh nets equipped with floats (one 4 m in length and the other 5 m) were deployed. The nets had 1 × 1 m panels with different mesh sizes in the following order: the 4 m net had 29, 35, 24, and 19.5 mm mesh-size panels; the 5 m net had 15.5, 10, 19.5, 12.5, and 8 mm mesh-size panels (mesh sizes recommended by Einarsson, Marine and Freshwater Research Institute of Iceland, personal communication). The nets were fixed to large boulders on the substratum, using bolted eye nuts and carabiners, floating upright on the flooding tide. To prevent algae from entangling the nets, a narrow 4 or 5 m long, 1 m wide strip of larger algae around each net was cleared at both control and harvested sites. Only the fronds immediately adjacent to the nets were trimmed, meaning that this clearing was minimal and localised, not comparable in scale to commercial harvesting. The nets were set during the evening low tide and then emptied on the next low tide at midday for three consecutive nights. Three sets of bolted nets were set in two control areas and two harvested areas on the following dates: 3–5 June 2019 (before first harvest), 3–5 July 2019, 3–5 September 2019, 23–24 February 2020, 5–8 May 2020, and 2–5 June 2020 (once per season post-harvest). In February 2020, most of the nets were damaged by a storm, consequently, data from that season were excluded from the analysis. The length of each fish captured was measured to the nearest 0.5 cm. Weight was recorded to the nearest gram using a digital hanging scale. Each specimen was photographed, and otoliths were extracted for subsequent age determination.
Fish ageing
The sagittal otolith pairs obtained from the fish caught were used to age the most abundant species in the catch (Pollachius virens, Gadus morhua, Myoxocephalus scorpius). P. virens and G. morhua were aged by embedding the otoliths in resin and cutting a 1 mm transverse section for subsequent examination under a dissecting microscope. The dark lines representing winter growth were counted in one of the pair of otoliths. M. scorpius ageing was based on the methods used in Ennis (Reference Ennis1970).
Stomach content
All prey present in the fish stomachs were sorted, counted, and identified to the most precise taxonomic level possible. Relative diet composition was investigated through frequency of occurrence (f i; number of individuals with the respective prey item, expressed as a frequency of the total number of non-empty stomachs) and relative prey abundance (p i; mean percentage of stomach contents of all predators that had each prey type) (Amundsen et al., Reference Amundsen, Gabler and Staldvik1996; Hyslop, Reference Hyslop1980). Where: f i = N i/N and p i = ∑S i/∑ST and N i is the number of predators with prey type i in their stomach; N is the total number of predators with stomach contents; S i is the amount of stomach contents composed by prey type i; and ST is the total amount of all prey types present in each stomach of the sample (Amundsen and Sánchez‐Hernández, Reference Amundsen and Sánchez‐Hernández2019). Partially digested prey that were still identifiable as fish were assigned to the category ‘Digested fish’. Other prey taxa were grouped into broad taxonomic categories. Prey that could not be identified to a chosen taxonomic group due to advanced stages of digestion were classified as ‘Unidentified remains’.
Statistical analysis
Biodiversity parameters (Shannon entropy, Shannon diversity, Simpson diversity, Pielou evenness, and Simpson evenness) were calculated with the ‘diversity’ function from the ‘vegan’ R package. Unconstrained ordination of treatment and date was performed by non-metric multidimensional scaling (NMDS) with the Bray–Curtis dissimilarity index using the vegan ‘metaMDS’ function (R Core Team, 2024).
A permutational multivariate analysis of variance (PERMANOVA) using Bray–Curtis dissimilarity with 999 permutations were performed to test for differences between species composition and stomach contents in the control and harvested areas, using the function ‘adonis’ in the ‘vegan’ R package (Oksanen, Reference Oksanen2015).
Cumulative frequency curves were used to visualise and compare fish size structure in harvested and control areas. As conditions for parametric tests were violated, non-parametric tests were used (Wilcoxon signed-rank test). Non-parametric Kruskal–Wallis test was also performed to test for differences of fish length between seasons. Finally, fish population size structures between harvested and non-harvested areas were compared with the Kolmogorov–Smirnov (K-S) test.
For stomach content analysis, a Bray–Curtis similarity matrix was constructed using fourth-root transformed data (Clarke, Reference Clarke1993). The similarity percentages (SIMPER) routine was used to identify the contributions of prey species to the differences detected between harvested and control areas. Food webs were constructed from the fish gut content analysis.
All tests were performed by using R and RStudio ‘Ghost Orchid’ (version 2021.9.2.382) (R Core Team, 2024; RStudio Team, 2024).
Results
Catch
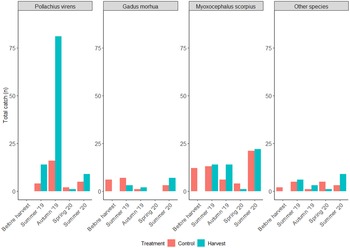
A total of 303 individuals (n control = 106, n harvest = 197) of nine fish species were caught using multi-mesh nets (Supp. Fig. 2). The number of fish caught across the mesh size spectrum was not significantly different between treatments (K-S test; n = 9, p= 0.7, and D = 0.3). However, the 12.5-mm mesh size captured the highest quantity of fish in harvested areas, driven by a large catch of P. virens in September 2019. Whereas in control areas, the 19.5-mm mesh size yielded the greatest catch.
The most common species were P. virens [saithe, n = 132, 43.6%], M. scorpius [sculpin, n = 107, 35.3%] and G. morhua [cod, n = 29, 9.6%], followed by Pleuronectes platessa Linnaeus, 1758 [European plaice, n = 13, 4.3%], P. gunnellus [butterfish, n = 11, 3.6%], Platichthys flesus (Linnaeus, 1758) [European flounder, n = 4, 1.3%], Salvelinus alpinus (Linnaeus, 1758) [Arctic charr, n = 3, 1%], Gasterosteus aculeatus Linnaeus, 1758 [three-spined stickleback, n = 2, 0.7%] and Limanda limanda (Linnaeus, 1758) [common dab, n = 2, 0.7%] (Supplementary Table S1). Fish diversity was higher in the control areas than in the harvested areas (Control: Shannon entropy = 1.4, Shannon diversity = 4.2, Simpson diversity = 3.2; Harvest: Shannon entropy = 1.3, Shannon diversity = 3.5, Simpson diversity = 2.6), driven by differences in evenness (Control: Pielou evenness = 0.6, Shannon evenness = 0.5 and Simpson evenness = 0.7; Harvest: Pielou evenness = 0.4, Shannon evenness = 0.3 and Simpson evenness = 0.6), as species richness was low and similar (7 in control, 8 in harvested). Higher numbers of total fish were caught in late summer and early autumn, with lower numbers recorded the following spring (Figure 2 & Supp. Fig. 3). Harvested areas yielded higher catches than control areas in July 2019, September 2019, and June 2020; whereas higher catches were made in June 2019 and May 2020. An increase in abundance was also observed during the summer months for the most caught species. Notably, September 2019 saw a high capture of P. virens.
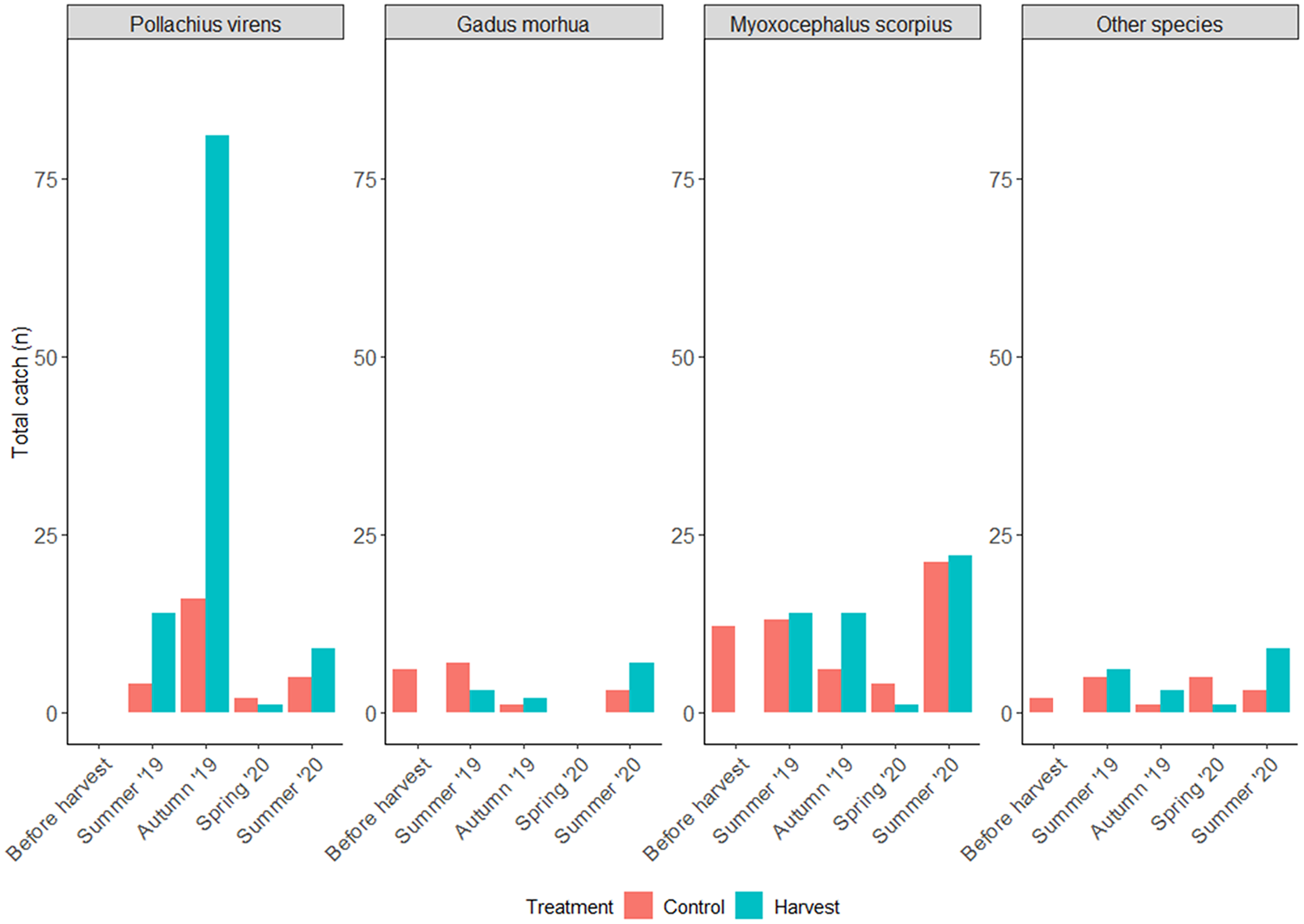
Figure 2. Total catch per season and treatment of the three most commonly caught species: Pollachius virens, Gadus morhua, and Myoxocephalus scorpius as well as the total of other species in harvested (H) and control (C) plots.
A PERMANOVA using distance matrices was used to compare fish communities in harvested and non-harvested areas, showing no significant differences (Pr(>F) = 0.6, F model = 0.8, df = 9). Nevertheless, the NMDS plot (Figure 3) shows that the assemblages are trending towards two clear treatment groups in the NMDS space, except for autumn 2019 (control) and summer 2019 (harvest), which fall within the ellipses of the opposite treatment group.

Figure 3. Non-metric multidimensional scaling (NMDS) of the fish assemblages at different seasons separated by treatment (all sampling occasions pooled). Stress = 0.07.
Size and age
Significant seasonal variations in median length over the study period can be observed for P. virens (Kruskal–Wallis; χ 2 = 71.7, df = 3, p < 0.01) (Figure 4). There was a sharp decrease in length from July 2019 to May 2020, followed by an increase in June 2020, consistent across treatments. Given the small sample sizes, no pattern was observable for G. morhua. M. scorpius maintained a relatively consistent length across both treatments and seasons.
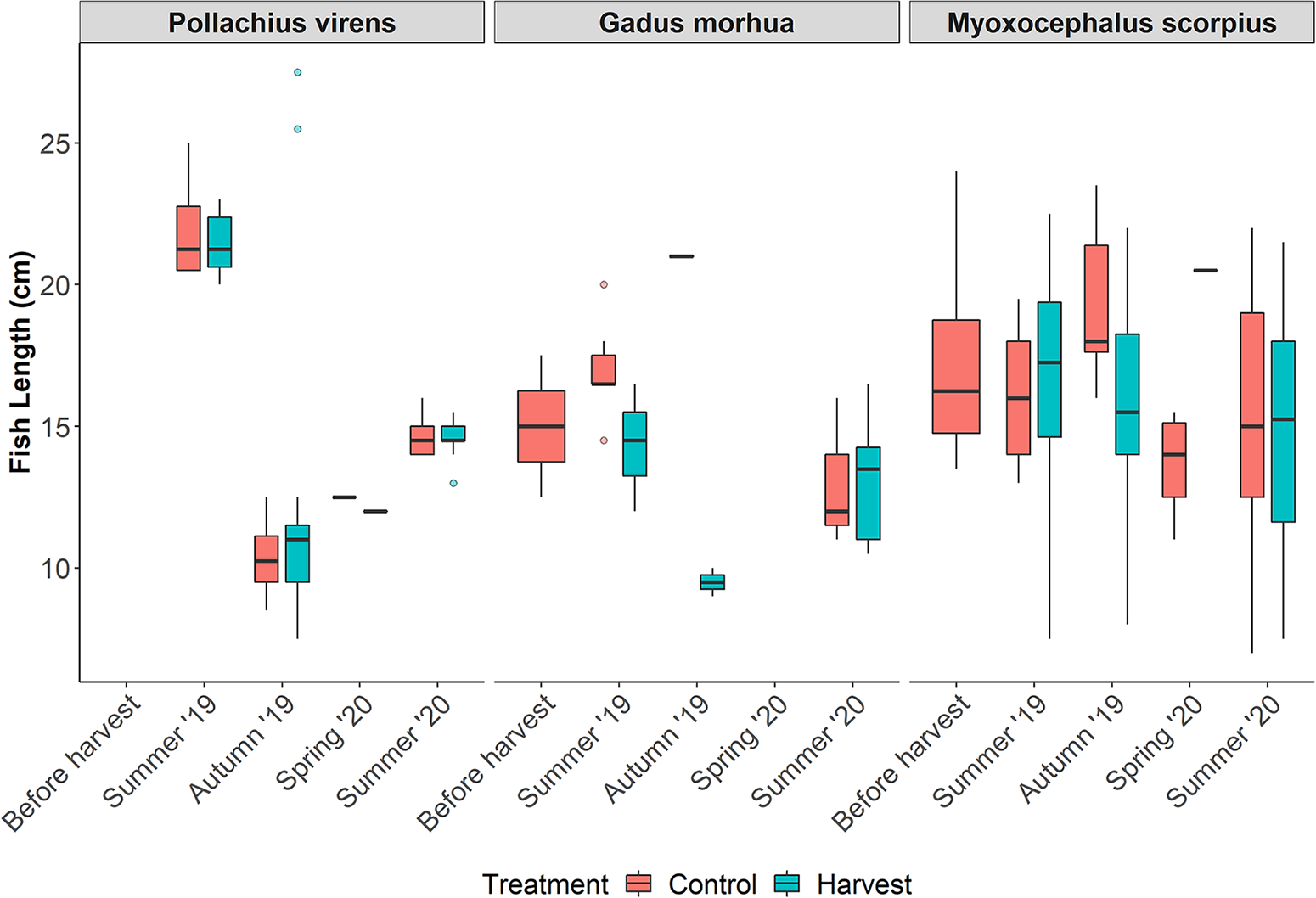
Figure 4. Size (length; cm) of the most represented fish species (Pollachius virens, Gadus morhua, and Myoxocephalus scorpius; bottom) separated by treatment and season. Each boxplot represents the interquartile range, the median (bold horizontal line) and two whiskers (1.5 times the interquartile range), with individual outliers shown as points.
Cumulative frequency curves were used to compare fish size structure in harvested and control areas (Figure 5). For P. virens (Harvest average length: 12.6 cm, median length: 11.5 cm; Control average length: 13 cm, median length: 11.5 cm), there were no significant differences between treatments in terms of median length (Wilcoxon rank sum test; n = 142, W = 1528, p= 0.5) and size distribution (K-S test; p= 0.7 and D = 0.2). In G. morhua (Harvest average length: 13.1 cm, median length: 13.5 cm; Control average length: 16.12 cm, median length: 16.5 cm), there were significant differences in median length (Wilcoxon rank sum test; n = 29, W = 163, p< 0.01) but not for size distribution (K-S test; p= 0.08, D = 0.5) between harvested and control areas, with control areas tending to have slightly bigger fish. However, it is important to interpret these results cautiously, due to the relatively small sample size (n harvest = 12 and n control = 17). For M. scorpius (Harvest average length: 16 cm, median length: 16.5 cm; Control average length: 16.1 cm, median length: 16 cm) no differences in size distribution (K-S test; p= 0.6, D = 0.2) or median length (Wilcoxon rank sum test; n = 107, W = 1407 p= 0.9) were found between treatments.
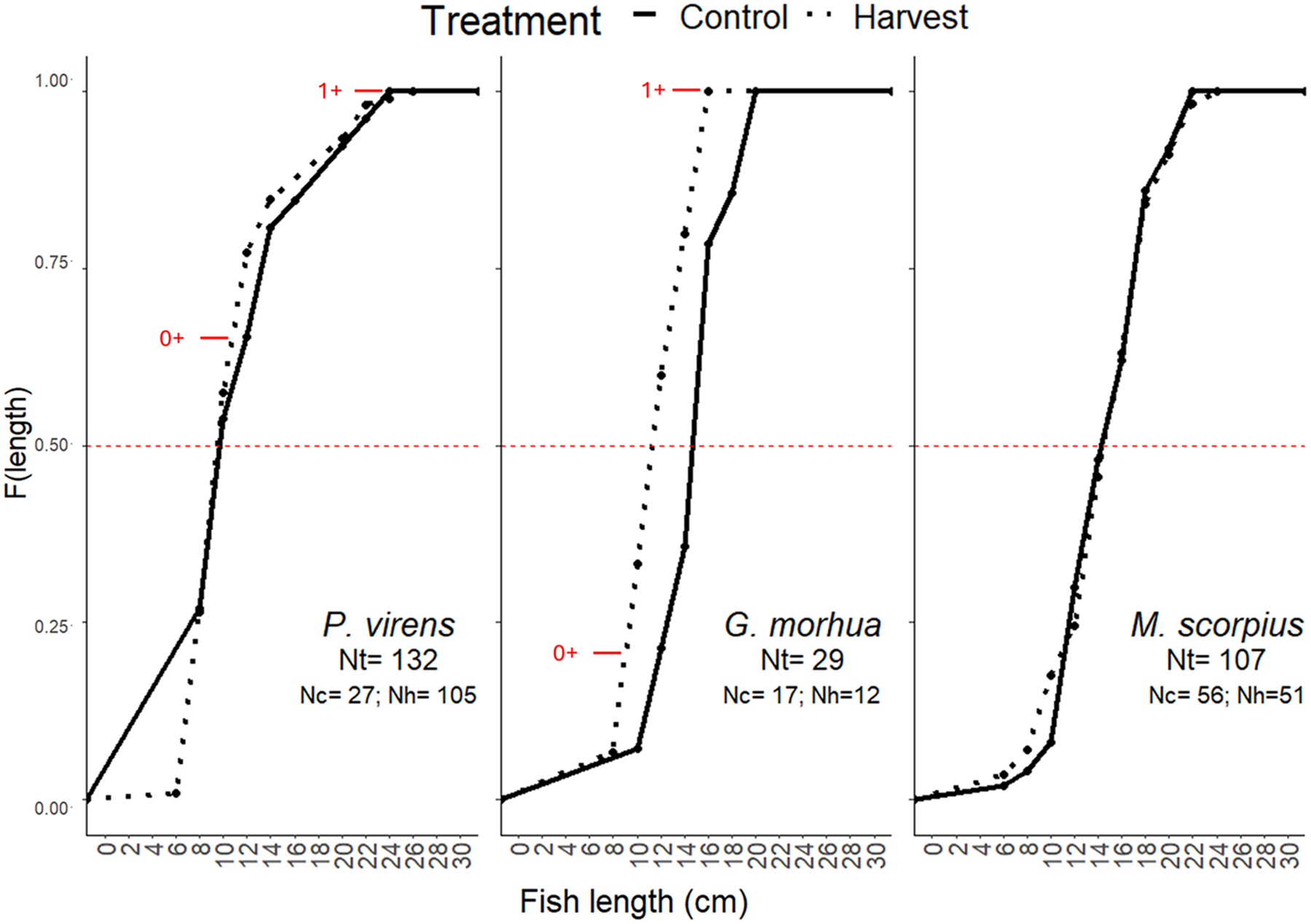
Figure 5. Cumulative frequency curves of length of the three species most represented in the catch separated by treatment and pooled across sampling occasions. nt = total number (n), nc = n for control areas and nh = n for harvested areas. Red lines marks the difference between 0+ and 1+ cohorts.
Resident M. scorpius had a much larger age range with up to 5 years, than the transient visitors P. virens (mostly 0+ and 1+) and G. morhua (mostly 0+) as they use the intertidal zone throughout their lifespan (Supplementary Table S2).
Stomach contents
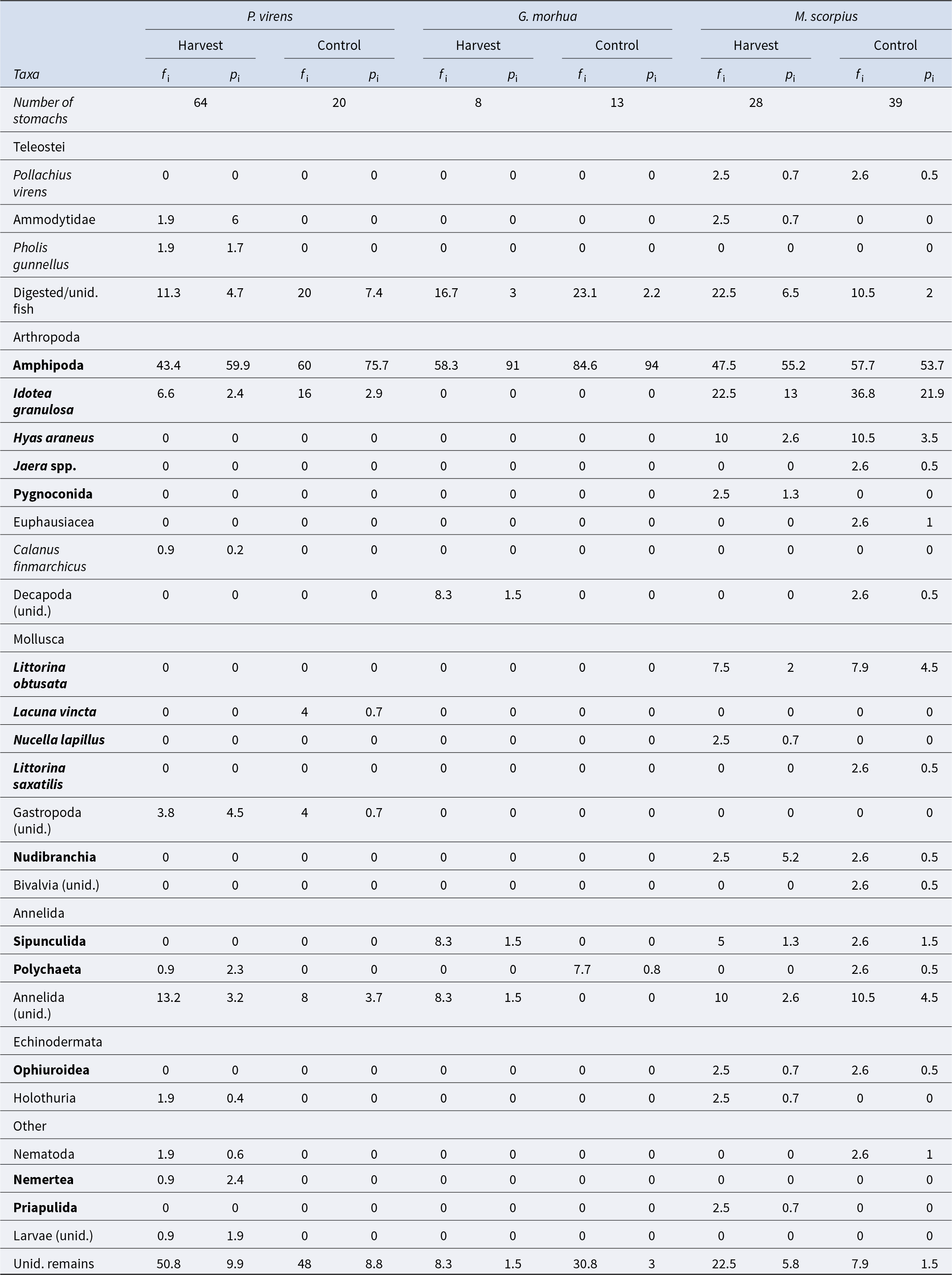
Of the 303 stomachs examined, 50 were empty (16.5%) and 253 contained prey (83.5%). Thirty-one different prey items were identified. Pooling across all species, the most common taxa eaten was Amphipoda, found in half of the stomachs examined (Table S3). This was followed by the broad category of ‘Unidentified remains’ (34.8%) and Idotea granulosa Rathke, 1843 (15.2%). Control areas had a 13% higher frequency of occurrence for amphipods and I. granulosa; otherwise, no notable differences were observed. Fish in harvested plots exhibited significantly greater prey richness (Wilcoxon rank sum test; n = 172, W = 4205, p< 0.05), but prey diversity did not differ significantly between treatments (Wilcoxon rank sum test; n = 172, W = 4201, p= 0.06).
The diet of P. virens stomachs was mainly composed of amphipods (63%), followed by unidentified prey, categorised as ‘Unidentified remains’ (9.8%), and partially digested fish (5.3%). The diet of G. morhua was dominated by amphipods (93%), with ‘Unidentified remains’ comprising a minor portion (2.4%). In the case of M. scorpius, amphipods also constituted the largest dietary component (53%), followed by I. granulosa (18%) (Table 1). Trophic diagrams were created for fish from both harvested and control areas based on these dietary data (Figure 6). The primary dietary components both for harvested and control areas were amphipods, followed by unidentified remains, and the isopod I. granulosa which was particularly important for M. scorpius and P. virens. Harvested areas had a broader variety of prey types, and unidentified gastropods and sand eels (Ammodytidae) appeared as important food for P. virens (Table 1).

Figure 6. Trophic linkages of fishes and their most represented prey from Breiðafjörður, in the control (top) and harvested (bottom) areas. The width of linking lines correspond to the contribution by percentage (pi; mean percentage of stomach contents that had each prey type) of each food item to fish diets, based on Table 1. Abbreviations for food types: Gastropoda unidentified (gastr.), Nudibranchia (nud.), Hyas araneus (H.A.), Polychaeta (poly.), Digested/unidentified fish (D.F.), Amphipoda (amp.), Idotea granulosa (I.G.), Unidentified remains (U.R.), Littorina obtusata (L.O.), Annelida unidentified (ann.), Ammodytidae (amm.), Nemertea (nemt.) And items in bold live amongst A. nodosum fronds or holdfast.
Table 1. Stomach content of three most common species, separated by treatments and pooled across sampling occasions
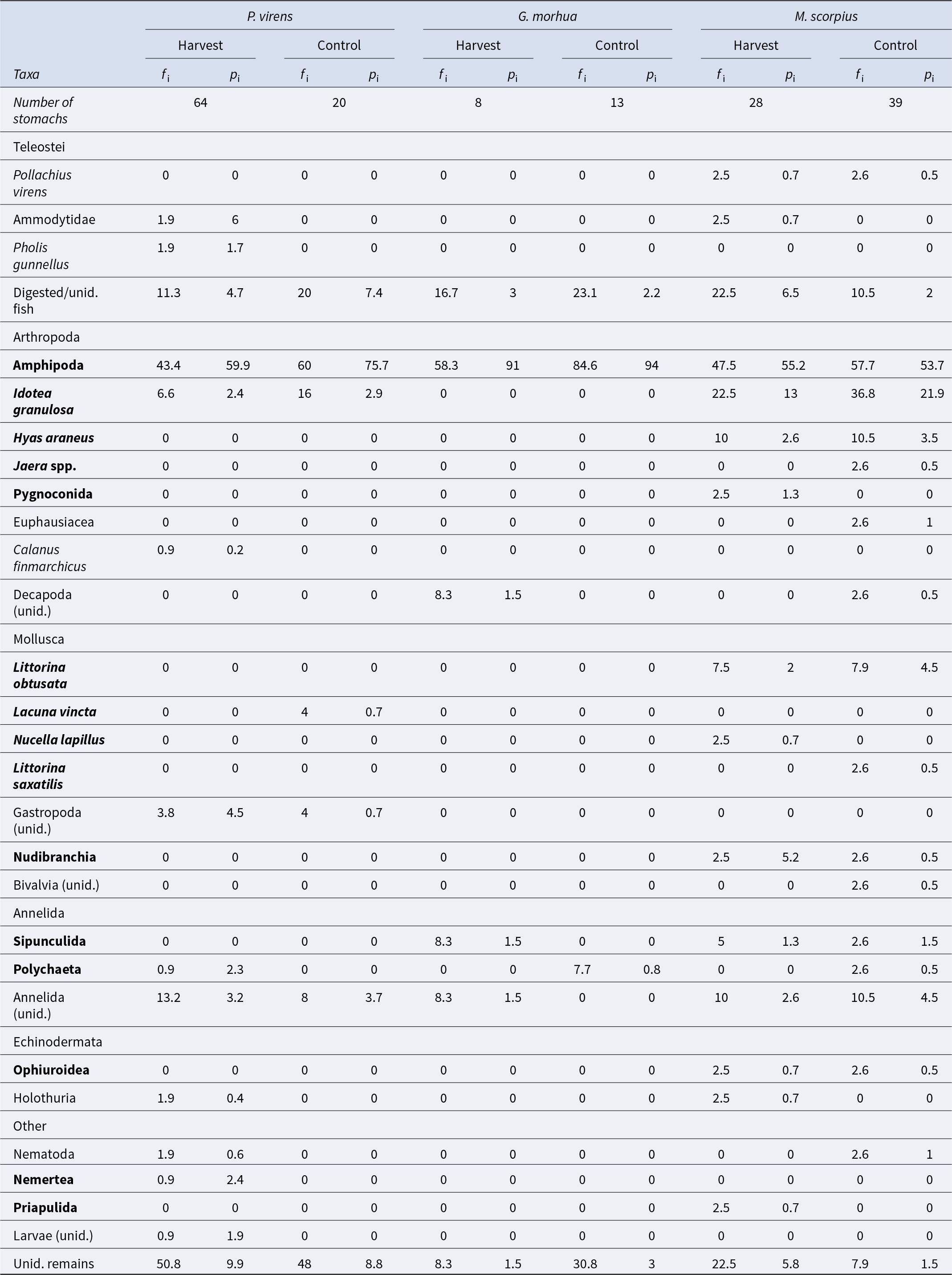
Frequency of occurrence (f i) mean percentage of stomach contents of all predators that had each prey type (p i). taxa in bold live amongst A. nodosum fronds or holdfast.
When comparing fish diet in harvested and control areas for each species with PERMANOVA, no differences were found (P. virens: Pr(>F) = 0.4, F model = 1.01, df = 83; G. morhua: Pr(>F) = 0.2, F model = 1.6, df = 20 and M. scorpius: Pr(>F) = 0.3, F model = 1.2, df = 66). Nevertheless, SIMPER analyses revealed dietary dissimilarities across species between harvested and control areas. For P. virens, average dissimilarity was 69.8% between treatments, with Amphipoda contributing most to this difference (42% contribution), followed by ‘Unidentified remains’ (20% contribution) and unidentified digested fish (13% contribution). Similarly, between treatment dissimilarity for G. morhua was 70.7%, primarily driven by Amphipoda (64% contribution) and, to a lesser extent, unidentified digested fish and ‘Unidentified remains’ (13% contribution each). M. scorpius exhibited the highest level of dissimilarity at 90.3%, with Amphipoda (32% contribution), I. granulosa (15% contribution), and ‘Unidentified remains’ (10% contribution) as the main contributors. Across all three species, Amphipoda consistently accounted for the greatest proportion of differences between treatments, being more abundant in fish stomachs in control areas (Supplementary Table S4).
Discussion
Our study provides an assessment of the transient and resident fish species found in the A. nodosum-dominated rocky intertidal zone of the sub-Arctic, focusing on the tide-in fish assemblages and their dietary preferences. The multi-mesh gill net proved to be an effective method in capturing a range of species and sizes over the course of the sampling period. The data revealed a varied assemblage of species using A. nodosum beds, either as place of permanent residence (M. scorpius, P. gunnellus, and G. aculeatus), or as juvenile nursery grounds, including commercially important gadoids P. virens and G. morhua and the flatfish L. limanda, P. flesus, and P. platessa. Our findings emphasise the role of A. nodosum beds in supporting fish communities, aligning with earlier studies that highlight the importance of nearshore macrophyte-dominated areas for fish in the Northern Atlantic Ocean (Schmidt et al., Reference Schmidt, Coll, Romanuk and Lotze2011; Vercaemer et al., Reference Vercaemer, Wong and Bravo2018).
The fish assemblage underwent seasonal changes in composition and abundance. Abundance peaked during the summer with lower numbers for spring, aligning with previous findings and likely driven by lower water temperatures and available food (Faria and Almada, Reference Faria and Almada2006; Gibson, Reference Gibson1972; Milton, Reference Milton1983; Pihl and Wennhage, Reference Pihl and Wennhage2002). A large shoaling event significantly increased the number of individuals caught, contributing to observed abundance patterns. These were predominantly small P. virens in the 0+ age group. The decline in size during late summer suggests that larger fish may have migrated to other areas. By the following year, evidence of growth in P. virens was observed, indicating cohort progression.
The use of the rocky intertidal by juvenile P. virens has been previously recorded in Canada (Rangeley and Kramer, Reference Rangeley and Kramer1995a, Reference Rangeley and Kramer1995b), where they were seen to move in large groups from the subtidal to the intertidal zone with the rising tides, using the intertidal zone as a refuge and as foraging areas (Rangeley and Kramer, Reference Rangeley and Kramer1998). Juvenile G. morhua have also been the focus of research both in the Eastern (Pihl, Reference Pihl1993) and Western (Hanson, Reference Hanson1996; Rangeley and Kramer, Reference Rangeley and Kramer1998) Atlantic Ocean, including Iceland (Jónasson et al., Reference Jónasson, Gunnarsson and Marteinsdóttir2009; Pálsson, Reference Pálsson1976, Reference Pálsson1980). Abundances in shallow waters are highest in the summer and then decrease as fish migrate to deeper waters in the winter (Methven and Bajdik, Reference Methven and Bajdik1994; Pálsson, Reference Pálsson1980). Intertidal use by these juvenile fishes usually follows tidal movements, with a migration to shallower waters most probably as feeding excursions (Keats, Reference Keats1990). In our study, both cod and saithe were in the +0- and +1-year classes, which again suggests the use of the rocky intertidal as nursery grounds. Juvenile use of the intertidal zone is supported by previous studies that have shown the importance of the rocky intertidal at high tide for juvenile fish (Faria and Almada, Reference Faria and Almada2006; Schmidt et al., Reference Schmidt, Coll, Romanuk and Lotze2011; Seitz et al., Reference Seitz, Wennhage, Bergström, Lipcius and Ysebaert2014; Studebaker, Reference Studebaker2006). Even when the adult stock declined, studies in Canada for saithe and cod confirmed the persistence of juvenile fish in coastal habitats, aiding stock recovery by supplying recruits to the adult stocks (McCain et al., Reference McCain, Rangeley, Schneider and Lotze2016), which in turn highlights the importance of proper management and monitoring of these areas.
A. nodosum harvesting and its impact on intertidal communities have been the subject of several studies in the North Atlantic (Black and Miller, Reference Black and Miller1991; Fegley, Reference Fegley2001; Rangeley, Reference Rangeley1994), yet the effects of harvest on transient and resident fishes and crustaceans remain understudied, particularly at high latitudes (Magnússon et al., Reference Magnússon, Hawkins, Gunnarsdóttir, Svavarsson and Gunnarsson2024). Diversity and evenness were higher in the control areas, while richness and density were higher in harvested areas. However, there were no statistically significant differences on fish community composition between treatments. This suggests that while abundance distribution might vary slightly, species identity remains consistent, possibly reflecting behavioural responses to habitat structure changes. When examining species individually, P. virens seemed to show a preference for harvested areas, possibly reflecting the species’ pelagic and shoaling behaviour. This could make it more susceptible to capture in structurally altered habitats, such as the shorter and less dense algal canopies after harvesting. M. scorpius length distribution did not show any effects of harvesting, probably because it is a resident benthic fish that feeds and stays close to the bottom (Landry et al., Reference Landry, Fisk, Yurkowski, Hussey, Dick, Crawford and Kessel2018), so a wider range of sizes and ages are expected to be present throughout the year and would be less affected by harvesting. Only median length of G. morhua was significantly less in the harvested plots, but as the total number was less than 30, and fish were infrequently caught, no strong conclusions can be made.
Stomach content analysis showed some differences in prey selection and in the frequency of occurrence of prey items between treatments. There were also differences amongst the three main species, indicating niche differentiation. The prevalence of amphipods, isopods, and annelids in rocky intertidal fish stomachs as a key dietary component aligns with previous studies that emphasise their importance in intertidal ecosystems (Compaire et al., Reference Compaire, Cabrera, Gómez-Cama and Soriguer2016; Norton and Cook, Reference Norton and Cook1999; Velasco et al., Reference Velasco, Gómez-Cama, Hernando and Soriguer2010) particularly in areas with dense macroalgal cover under, on and amongst which they live (Gollety et al., Reference Gollety, Thiebaut and Davoult2011; Larsen, Reference Larsen2012; Pavia et al., Reference Pavia, Carr and Åberg1999). Similarly, the importance of fish and fish remains in examined stomachs implies that it may be a significant component of the fishes diet (Norton and Cook, Reference Norton and Cook1999).
While some differences between treatments were found in the relative contribution of prey items (e.g. amphipods), dietary composition did not differ significantly between treatments. This suggests that, despite some dietary differences in frequency of occurrence or abundance, the overall trophic structure remains similar. Stomachs from fish caught in harvested plots exhibited significantly greater prey richness, although prey diversity did not differ significantly between treatments, which suggests that individuals in harvested areas are consuming a broader range of prey types, but not necessarily with greater dietary complexity. Gastropods for example, were more frequently recorded in fish diets in harvested areas, which could suggest that harvesting creates disturbed foraging environment and provide less refuge from predation than full canopy areas. The most common prey items found, amphipods, isopods (i.e. I. granulosa) along with L. obtusata and L. vincta live on and amongst A. nodosum canopy, in most cases feeding on it (Pavia et al., Reference Pavia, Carr and Åberg1999; Pavia and Toth, Reference Pavia and Toth2000), emphasising their key role in intertidal food webs. M. scorpius, as a resident species feed mostly on I. granulosa, H. araneus and L. obtusata. Both I. granulosa and L. obtusata feed on A. nodosum or its epiphytes (Naylor, Reference Naylor1955; Williams, Reference Williams1990); thus, M. scorpius is part of a tightly linked food web, directly utilising primary production from A. nodosum. Harvesting can alter diversity of prey available to fish visitors or resident in the intertidal zone through changes in plant biomass and complexity as well as habitat patchiness (Sharp et al., Reference Sharp, Ugarte and Semple2006) although previous studies have found small or temporary impact of harvesting on seaweed-associated communities (Lauzon-Guay et al., Reference Lauzon-Guay, Feibel, Morse and Ugarte2023; Trott and Larsen, Reference Trott and Larsen2010). Conversely, variety of available prey may increase due to fewer refuges; thus, harvesting could have subtle effects on trophic dynamics, including decreasing availability of some taxa that feed on A. nodosum to predators (e.g. L. obtusata is found in more stomachs in the control areas) or provide better access to benthic species. Amphipods on the other hand, which primarily live on or amongst A. nodosum and are clearly a crucial part of the food web, were not significantly different between treatments. Current harvesting practices may not entirely disrupt trophic interactions, but could be altering prey composition, warranting further study to explore long-term ecological impacts. The observed high variability in stomach content data suggests that larger sample sizes may be required to adequately describe fish diet composition in Breiðafjörður.
Our study did not detect strong effects of A. nodosum harvesting on intertidal fish communities, which aligns with previous studies in the North Atlantic (Lauzon-Guay et al., Reference Lauzon-Guay, Feibel, Morse and Ugarte2023; Phillippi et al., Reference Phillippi, Tran and Perna2014; Sharp and Pringle, Reference Sharp and Pringle1990; Trott and Larsen, Reference Trott and Larsen2010). Nonetheless, the relatively small sample size and sampling dates may have hindered our ability to detect subtle differences between treatments. Sites were selected to minimise variation of abiotic factors, but unmeasured differences in physicochemical parameters could have also influenced fish distributions. Nevertheless, given the proximity between plots (50–100 m) and the homogeneous nature of the study area, we assumed minimal variation in abiotic conditions between treatments. Additionally, differences in habitat complexity (i.e. substrate variation or canopy density and structure) could have also influenced fish distribution and behaviour.
While harvesting reduced A. nodosum cover in treatment sites, potentially altering prey accessibility, shelter availability and habitat complexity, our results suggest that these did not lead to major changes, likely due to the localised and small-scale nature of the disturbance and their resistance to perturbation (Phillippi et al., Reference Phillippi, Tran and Perna2014; Sharp and Pringle, Reference Sharp and Pringle1990). Finally, while our experimental harvesting was done following local practices, precise biomass measurements were not collected, limiting our ability to fully quantify the harvesting intensity. Different levels of biomass removal (e.g. 30% vs. 60%) could lead to different ecological effects. Despite these limitations, our study provides insights into high-latitude fish assemblages in A. nodosum-dominated intertidal habitats and their seasonality, highlighting the ecological significance of these areas as nursery grounds for commercially important species that rely on them before migrating offshore.
Conclusion
Our study demonstrated the occupancy and use of the rocky intertidal zone by several fish species for feeding, living, and as nursery grounds. We also highlighted the significance of A. nodosum beds as feeding grounds for transient fish and the subtle impacts of harvesting on food web linkages.
No significant effects of A. nodosum harvesting were detected on fish assemblages and populations of common species. Nevertheless, a precautionary approach should be implemented when harvesting, as juvenile fish are susceptible to nursery habitat degradation which could affect the adult stocks of commercially important gadoids. Future research should focus on long-term monitoring to assess long-term trends in recovery rates, harvesting impacts, and diet composition of fish populations in commercially harvested areas.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0025315425100301.
Acknowledgements
The authors thank Bylgja Sif Jónsdóttir, Svanhildur Egilsdóttir, Eygló Ólafsdóttir, Kristín Valsdóttir, and Einar Pétur Jónsson at MFRI for their assistance with data collection, and Haraldur Einarsson at MFRI for his advice on mesh sizes for the sampling nets. We would also like to thank the assistance from the harvesting crew at Thorverk Ltd. for cutting the experimental plots and lastly, Dr. A. Silva for her help with the SIMPER analysis. We would also like to extend our sincere gratitude to the anonymous reviewers for their insightful comments.
Author contributions
SJH, LG, and KG conceptualized and designed the study. KG and LG were responsible for funding acquisition and project administration. JTM, LG, and KG collected the data. JTM analysed the data, wrote the first draft of the manuscript and prepared tables and figures. SJH, KG, JS, and JTM revised and edited successive drafts of the manuscript. All authors contributed to manuscript revision and have read and agreed on the submitted version.
Funding
This work was partly funded by The Icelandic Research Fund, as part of the project grant 185529-51.
Competing interests
The authors declare none.