Article contents
Biomimetic protein-harpooning surfaces
Published online by Cambridge University Press: 06 April 2018
Abstract
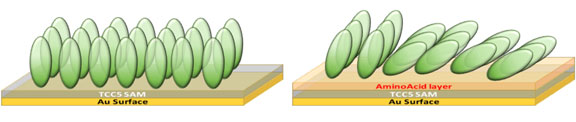
Properly driving protein interactions with solid surfaces play a very important role in many natural processes, stimulating a great interest for the design of new biomaterials and medical devices. Despite the progress in this field, many further upgrades have to be achieved to better exploit the protein driving, in terms of control of amounts and conformation of the adsorbing proteins. In this paper, new biocompatible amino acid–calix[4]crown-5 bilayers were built as nano-templating surfaces, hosting a controlled number of anchoring sites, able to immobilize proteins in well-defined quantity, and the evaluated footprint data support the idea of oriented protein on analyzed substrates. The efficiency of the setup was tested for the particular case of antibacterial lysozyme adsorption on biocompatible surfaces.
Information
- Type
- Research Letters
- Information
- Copyright
- Copyright © Materials Research Society 2018
References
A correction has been issued for this article:
- 2
- Cited by
Linked content
Please note a has been issued for this article.


