Crohn’s disease (CD) is a type of inflammatory bowel disease that can affect any part of the gastrointestinal tract, particularly the terminal ileum and proximal colon(Reference Dolinger, Torres and Vermeire1). CD is characterised by a segmental distribution of lesions within the gastrointestinal tract, with chronic, progressive and recurrent transmural inflammation of the bowel wall that leads to wall thickening and luminal narrowing(Reference Roda, Chien Ng and Kotze2). Current strategies for treating CD focus on achieving deep and long-term remission to improve quality of life while preventing complications and halting disease progression(Reference Dolinger, Torres and Vermeire1). While anti-inflammation therapy is the first-line treatment for CD, approximately 80 % of CD patients still need to undergo surgery during their lifespan(Reference Lowe, Sauk and Limketkai3), whereas 35 % needed a second or more operation(Reference Ma, Moran and Benchimol4). Nearly one-third of post-surgical patients develop complications, highlighting the importance of post-surgical management(Reference Ma, Moran and Benchimol4).
In post-surgical CD patients, dietary intervention offers a promising approach for disease management during the initial 4–6 weeks(Reference Levine, Sigall Boneh and Wine5–Reference Logan, Clark and Ijaz7). Importantly, dietary management is crucial for preventing post-surgical CD recurrence. Exclusive enteral nutrition (EEN), which involves using a complete, formulated liquid diet while excluding normal food components, can provide adequate nutrition and induce remission in up to 80 % of paediatric CD cases(Reference Day and Lopez8,Reference Ashton, Gavin and Beattie9) . EEN also has minimal essential side effects and is linked to high rates of mucosal healing(Reference Day and Lopez8). However, EEN is recommended only when drug therapy is declined or as an adjunct to standard treatments(Reference Wall, Day and Gearry10). A major limitation of EEN for adults is poor adherence due to the unpleasant taste of the formulations, which often leads to treatment discontinuation and reduced efficacy. Moreover, by eliminating the social and hedonic aspects of food intake, EEN can negatively affect psychosocial well-being and overall quality of life.
Recent studies have introduced the Crohn’s Disease Exclusion Diet (CDED), a progressive whole-food diet that incorporates partial enteral nutrition (PEN). CDED is characterised by its high protein content and low levels of animal fat, heme, gluten and additives, while also promoting fibre intake(Reference Yanai, Levine and Hirsch11). This diet has demonstrated significant benefits for adult patients with CD in randomised trials(Reference Yanai, Levine and Hirsch11–Reference Ghiboub, Penny and Verburgt13) and was initially recommended for managing mild-to-moderate active CD(Reference Bischoff, Bager and Escher14). Additionally, the American Gastroenterological Association advises that all patients with inflammatory bowel disease, unless contraindicated, follow a Mediterranean diet. This diet emphasises fresh fruits, vegetables, monounsaturated fats, complex carbohydrates and lean proteins, while minimising consumption of ultra-processed foods, added sugars and salt to support overall health and well-being(Reference Hashash, Elkins and Lewis15). However, the taste and flavours of both the CDED and the Mediterranean diet have not been fully embraced by Chinese inflammatory bowel disease patients due to regional and dietary differences. Traditional Chinese eating habits are often based on rice or wheat as staple foods, complemented by a wide variety of vegetables, soy products, freshwater fish and light cooking methods(Reference Wang, Lin and Bloomgarden16), whereas Mediterranean diets frequently incorporate olive oil, cheese and whole grains that may be less acceptable to Chinese patients. To address this challenge, we developed a Modified Mediterranean Diet (MMD) that aligns with the nutritional principles of the Mediterranean diet while incorporating culturally familiar Chinese ingredients and flavours.
Specifically, the MMD emphasises the use of rapeseed or soybean oil as primary sources of monounsaturated fats in place of olive oil and introduces soy products, tofu and freshwater fish as lean protein sources alongside moderate amounts of poultry. Whole grains are adapted to include brown rice, millet and oats, which are more acceptable in Chinese cuisine. The diet also integrates abundant seasonal vegetables and fruits, while maintaining reduced levels of red and processed meats, animal fats and refined sugars. In terms of culinary practices, the MMD encourages steaming, boiling and stir-frying with minimal oil to ensure palatability for Chinese patients.
This culturally adapted approach is designed to preserve the anti-inflammatory and metabolic benefits of the Mediterranean dietary pattern while improving adherence among Chinese CD patients through greater familiarity and acceptability. Thus, the MMD represents a feasible dietary model that integrates evidence-based nutritional recommendations with local dietary traditions. Given its potential to improve both adherence and clinical outcomes, this study was designed to evaluate the effects of MMD supplemented with PEN in post-surgical CD patients compared with EEN. The primary objective was to assess its impact on quality of life, while secondary objectives included evaluating disease-related characteristics and nutritional status.
Methods
Study design and participants
This randomised, controlled, pilot study was conducted at The Second Affiliated Hospital of Zhejiang University School of Medicine between September 2022 and June 2024. Eligible patients were individuals aged 14 years or older who had a confirmed diagnosis of CD through endoscopy, histology and imaging. The enrolled patients were in the perioperative period, defined as within 2–7 d after surgery, having undergone terminal ileal resection, partial colectomy, ileocecal resection or small bowel resection due to CD. Participants were required to be able to orally consume nutritional formulations or food, own a smartphone with the WeChat application installed and demonstrate the ability to send and receive messages via WeChat. Additionally, participants needed to be willing and able to attend scheduled postoperative follow-up visits at the study centre. Exclusion criteria included any of the following: a planned surgery within 5 weeks postoperatively; ileostomy or colostomy; complete intestinal obstruction or fibrostenosis with pre-stenotic dilatation; pregnancy or breastfeeding; planned use of biologics within 5 weeks postoperatively; planned use of corticosteroids or prednisone greater than 20 mg per day or the equivalent within 5 weeks postoperatively to maintain remission; planned use of probiotics or prebiotics for more than 1 week postoperatively; planned use of antibiotics for more than 2 weeks postoperatively; allergies to known components of enteral nutrition; a BMI of less than 14 or greater than 28 kg/m2; celiac disease; coexisting diabetes or other autoimmune diseases (e.g. rheumatic disease, autoimmune liver disease, psoriasis, etc.); mental illness or malignant tumours. The BMI cut-off values were chosen because a BMI below fourteen indicates severe malnutrition, requiring more intensive nutritional interventions such as EEN, rather than participation in a randomised trial(Reference Bischoff, Bager and Escher14). Conversely, a BMI above twenty-eight is classified as obesity, and continuing EEN may exacerbate this condition(Reference Zhou17). Additionally, participants were excluded if they were involved in other clinical trials or had any condition that the investigator deemed made them unsuitable for participation in this study. The study was performed in accordance with the ethical principles of the Declaration of Helsinki. All participants provided written informed consent prior to the study. The study design was reviewed and approved by the Ethics Committee of Second Affiliated Hospital of Zhejiang University School of Medicine.
Randomisation and masking
All enrolled patients were randomly assigned in a 1:1 ratio to either the MMD supplemented with PEN group or the EEN group using a computer-generated random numbers table for simple randomisation. A dietitian provided nutritional guidance to the patients based on their assigned diet 2–7 d post-surgery. Due to the nature of the intervention, blinding was not feasible; therefore, this study was conducted as an open-label trial.
Interventions
All participants were not given any medication for treating CD, including aminosalicylates, immunosuppressants, corticosteroids, and biologics, during the 4–5 weeks dietary intervention that started on the date of surgery. Each participant completed comprehensive training on either the MMD supplemented with PEN or the EEN according to their assigned group, with the goal of achieving target energy intake of 30–35 kcal/kg·bw and target protein intake of 1·2–1·5 g/kg·bw. EEN requires patients to consume a specialised formula exclusively, with the exclusion of all solid foods and any liquids other than water. This formula is designed to provide complete nutrition, delivering all essential nutrients and energy content necessary to support healing and growth. In the MMD supplemented with PEN group, participants obtain approximately half of their energy from a specialised formula and half from food. The details regarding the food ingredients and cooking methods can be found in the online Supplementary material.
On day 2 or day 3 post-surgery, the trial protocol would commence, provided the surgeon deemed the participant suitable for oral intake or enteral nutrition. At this time, baseline data were collected. During the trial, all participants followed the EEN or MMD supplemented with PEN diets under the supervision of a dietitian based on their assigned groups (online Supplementary material). Each participant was instructed to eat their meals alone and to document the cooked weight of all foods ingested at each meal, along with the corresponding raw-to-cooked ratios. Dietitians utilised the recorded cooked weights and raw-to-cooked ratios to ascertain the raw weight of each food item, which was subsequently input into Nutritional Calculator software to estimate daily energy and nutrient intakes. The net weight of each food ingredient, both prior to and following cooking, was documented to facilitate the calculation of the raw-to-cooked ratio. The raw/cooked ratio was calculated as follows:
Dietary intake data were systematically recorded on a daily basis in an electronic food diary and subsequently transmitted to the dietitian for analysis via WeChat. The dietitian maintained continuous communication with participants between 17.00 and 22.00 each day to provide feedback and guidance, ensuring accurate monitoring and support of dietary adherence. Based on this information and nutrient analysis, the dietitian provided personalised dietary instructions for the following day, aiming to meet a daily energy target. Like the MMD supplemented with PEN group, participants in the EEN group also reported their daily consumption to the dietitian.
The programme lasted 4–5 weeks for all participants. This duration was selected for two main reasons: firstly, participants were scheduled to begin pharmacological treatments in the subsequent phase of care; secondly, considering that early postoperative dietary intervention may influence patients’ long-term food preferences, a 21-day period is generally sufficient for establishing dietary habits. If feasible in future studies, longer intervention periods may be explored.
Outcome measurement
The primary outcome of the study was quality of life, measured using the IBDQOL-22(Reference Ruan, Chen and Zhou18) at the end of the intervention. The IBDQOL-22 has twenty-two items, which assess the quality of life of patients with CD in four dimensions: bowel symptoms and their influences (six items), symptoms and discomfort (five items), emotional function (six items) and social functions (five items). The total score ranges from twenty-two to 110, and higher scores indicate a better quality of life. The IBDQOL-22 demonstrated good internal consistency in CD, with Cronbach’s α values ranging from 0·76 to 0·89, and excellent test–retest reliability, with intraclass correlation coefficients of 0·72 to 0·90(Reference Ruan, Chen and Zhou18).
Secondary outcomes included nutritional status and disease-related characteristics assessed at the end of the intervention. Nutritional status indicators included (g/l), erythrocyte (1012/l), creatinine (μmol/l) and albumin (g/l). Disease-related characteristics were comprehensively evaluated using clinical indices, symptom scales and laboratory markers. Clinical disease activity was assessed with the Harvey–Bradshaw Index (HBI)(Reference Harvey and Bradshaw19) and the Crohn’s Disease Activity Index(Reference Best, Becktel and Singleton20), both validated measures of disease severity. Gastrointestinal symptoms were captured using the patient-reported Gastrointestinal Symptom Rating Scale(Reference Kulich, Madisch and Pacini21). Laboratory markers included platelet count, C-reactive protein and erythrocyte sedimentation rate to reflect systemic inflammation, while faecal calprotectin was measured as a non-invasive indicator of intestinal mucosal inflammation and treatment response. Physician-assessed poor adherence was operationally defined as the fulfilment of at least one of the following criteria: (1) refusal to comply with the prescribed nutritional guidance provided at the time of study enrolment or (2) failure to submit the electronic food diary for more than 10 % of the total study duration (online Supplementary material).
Power calculation
A sample size estimation was not performed since the purpose of this study was to evaluate the feasibility and potential effects of the intervention to generate preliminary data for future large-scale randomised controlled trials. There was also no previous data available on the difference between IBDQOL-22 scores of the MMD supplemented with PEN and EEN groups. A total of forty-two to fifty-three participants were expected based on the average hospitalisation rate at the centre of investigation during the 21-month recruiting period, assuming 4–5 CD patients per month and an inclusion rate of 50 %.
Statistical analysis
Data analysis was conducted using SPSS software (Version 27, USA) on a modified intention-to-treat basis. Patients who experienced disease worsening, developed new complications or required drug treatment for CD were withdrawn from the trial, and their data were considered a failure of the intervention and excluded from the analysis. Categorical variables were analysed using the χ2 test. Continuous variables were reported as means (sd) or medians (interquartile range). Normally distributed data underwent analysis using Student’s t test, while skewed data were analysed using the Mann–Whitney U test or Kruskal–Wallis H test as appropriate. P-values less than 0·05 were deemed statistically significant.
Results
Patient enrolment and baseline information
A total of 115 patients were recruited and assessed for eligibility for the study, of whom forty-six were randomly assigned to the EEN group (n 24) and MMD supplemented with PEN group (n 22) (Figure 1). One patient in the EEN group did not receive allocated intervention due to cholecystitis and was thus excluded from the analysis. A total of twenty-three patients in the EEN group and twenty-two patients in the MMD supplemented with PEN group adhered to the study protocol, received complete intervention and were included in the analysis. The mean age of the EEN and MMD supplemented with PEN groups was 31·91 (sd 8·26) years and 35·73 (sd 9·68), respectively. The proportion of males was 54·5 % and 45·5 % (P = 0·445). Mean BMI was 19·9 (sd 3·2) in the EEN group and 19·6 (sd 2·4) in the MMD supplemented with PEN group (P = 0·697). Median disease duration was 3·0 and 2·5 years, respectively (P = 0·471). The median baseline IBDQOL scores of the EEN and MMD supplemented with PEN groups were comparable (77·00 (sd 12·61) v. 72·27 (sd 13·42), P = 0·230). A higher proportion of patients in the EEN group had perianal disease compared with the MMD supplemented with PEN group (47·8 % v. 18·2 %, P = 0·035). This imbalance in baseline perianal involvement may potentially influence clinical outcomes, as perianal disease is often associated with more severe disease course and may affect nutritional status, response to dietary interventions and quality-of-life measures. Therefore, caution is warranted when interpreting intervention effects, and this factor should be considered (Table 1).
Study enrolment and allocation.EEN, Exclusive enteral nutrition; MMD, Modified Mediterranean diet; PEN, Partial enteral nutrition.

Baseline characteristics

EEN, Exclusive enteral nutrition; MMD, Modified Mediterranean diet; PEN, Partial enteral nutrition; CD, Crohn’s disease; IQR, Interquartile range; IBDQOL, Inflammatory bowel disease quality-of-life questionnaire; HBI, Harvey–Bradshaw Index score; CDAI, Crohn’s Disease Activity Index; GSRS, Gastrointestinal Symptom Rating Scale; PLT, platelet count; CRP, C-reactive protein; FC, Faecal calprotectin; ESR, Erythrocyte sedimentation rate; ALB, albumin; Cr, Creatinine.
* Data missing for one patient.
Primary and secondary outcomes
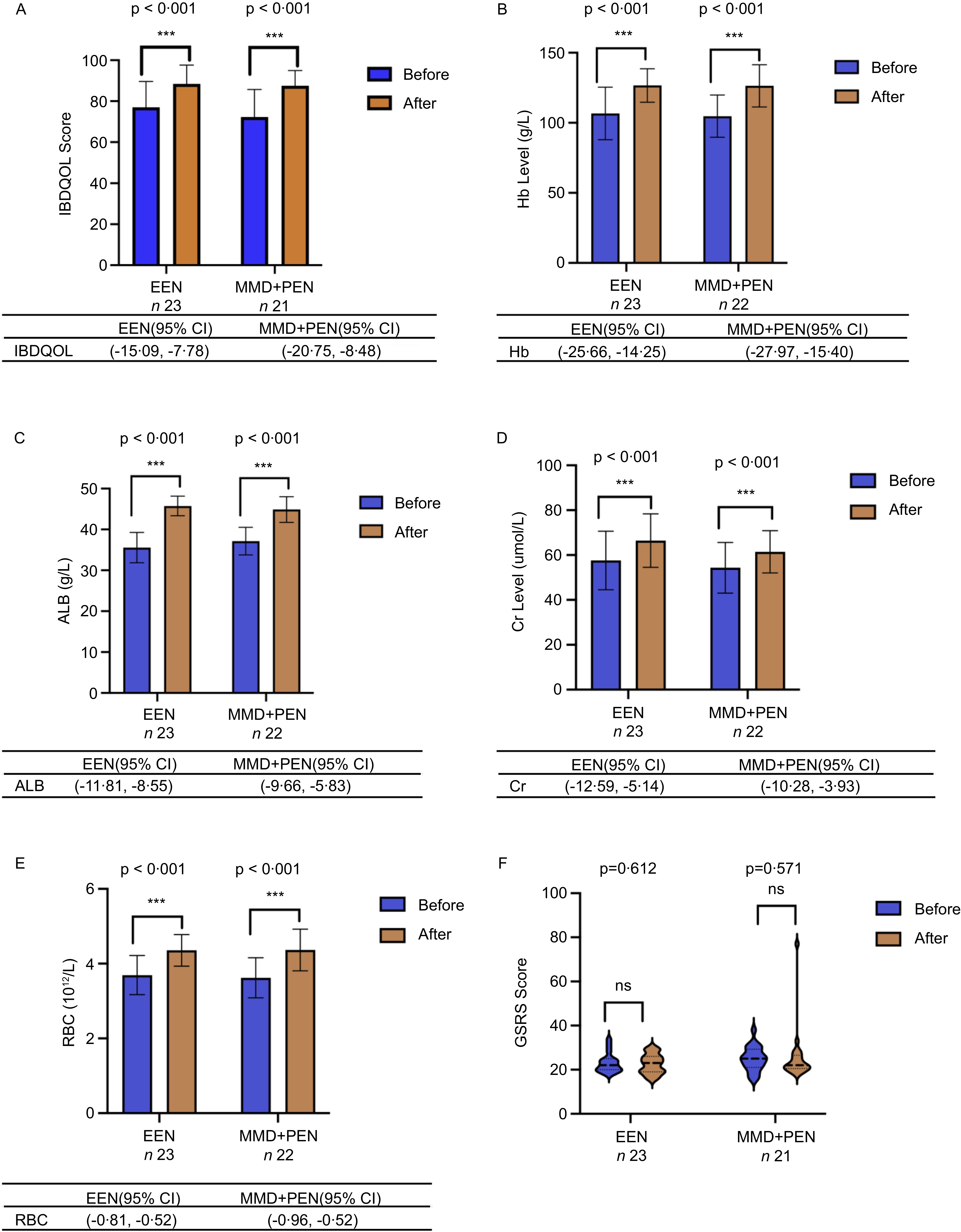
At the end of the intervention, the EEN and MMD supplemented with PEN groups achieved similar IBDQOL scores (88·43 (sd 9·17) v. 87·57 (sd 7·38), P = 0·734) (Table 2). Patients in both groups showed significant improvement in IBDQOL scores after intervention (P < 0·001) (Figure 2). There was also no significant difference in all four aspects of the IBDQOL between the EEN and MMD supplemented with PEN groups (P > 0·05) (Table 3).
Primary and key secondary outcomes

EEN, Exclusive enteral nutrition; MMD, Modified Mediterranean diet; PEN, Partial enteral nutrition; IQR, Interquartile range; IBDQOL, Inflammatory bowel disease quality-of-life questionnaire; HBI, Harvey–Bradshaw Index score; CDAI, Crohn’s Disease Activity Index; GSRS, Gastrointestinal Symptom Rating Scale; PLT, Platelet count; CRP, C-reactive protein; FC, Faecal calprotectin; ESR, Erythrocyte sedimentation rate; ALB, Albumin; Cr, Creatinine.
Quality of life and nutritional status in the EEN and MMD supplemented with PEN groups. CDAI, Crohn’s Disease Activity Index; HBI, Harvey–Bradshaw Index score; EEN, Exclusive enteral nutrition; MMD, Modified Mediterranean diet; PEN, Partial enteral nutrition; FC, Faecal calprotectin; PLT, platelet count; CRP, C-reactive protein; ESR, Erythrocyte sedimentation rate. Ns, non-significant; *P < 0·05, **P < 0·01, ***P < 0·001.

Inflammatory bowel disease quality-of-life questionnaire at baseline and after the intervention

EEN, Exclusive enteral nutrition; MMD, Modified Mediterranean diet; PEN, Partial enteral nutrition.
At the end of the intervention, there were no significant differences between the EEN and MMD + PEN groups in terms of HBI (1 (0, 3) v. 3 (0, 4), P = 0·463), Crohn’s Disease Activity Index (108·07 (sd 48·17) v. 115·47 (sd 51·61), P = 0·625) and Gastrointestinal Symptom Rating Scale (23 (19, 26) v. 22 (20·5, 26·5), P = 0·556) scores, or other disease-related characteristics, including platelet count, C-reactive protein, erythrocyte sedimentation rate and faecal calprotectin. Similarly, Hb, erythrocyte, creatinine and albumin levels were comparable between the two groups (Table 2).
Within-group comparisons showed that, although HBI and Gastrointestinal Symptom Rating Scale scores did not change significantly, the number of patients with HBI > 5 decreased in both groups after intervention (an HBI score greater than five generally indicates active CD, whereas a score below five is consistent with clinical remission). Both EEN and MMD + PEN groups exhibited increases in creatinine, Hb, erythrocyte and albumin levels (Figure 2), along with decreases in C-reactive protein and faecal calprotectin levels (Figure 3) relative to baseline. Additionally, the MMD + PEN group demonstrated a reduction in erythrocyte sedimentation rate levels at the end of the intervention (Figure 3).
Disease-related characteristics in the EEN and MMD supplemented with PEN groups. IBDQOL, Inflammatory bowel disease quality-of-life questionnaire; EEN, Exclusive enteral nutrition; MMD, Modified Mediterranean diet; PEN, Partial enteral nutrition; ALB, albumin; Cr, Creatinine; GSRS, Gastrointestinal Symptom Rating Scale. Ns, non-significant; *P < 0·05, **P < 0·01, ***P < 0·001.

Safety
One patient in the EEN group (4·2 %) had cholecystitis and was withdrawn from the study. No other adverse events or serious adverse events were observed or reported during the study.
Patient adherence
Participant adherence to the study protocol was evaluated. Based on the predefined criteria, adherence in both study groups was determined to be 100 %, with no participants classified as exhibiting poor adherence. This favourable outcome is likely attributable to the comprehensive initial training and the regular communication with a dietitian to review daily food and nutritional formulation intake, which may have enhanced participant engagement and self-awareness in nutritional management.
Discussion
In this pilot study, we demonstrated that MMD supplemented with PEN achieved comparable improvements in overall quality of life to EEN in post-surgical CD patients. Both groups achieved similar IBDQOL scores. Furthermore, MMD supplemented with PEN and EEN yielded comparable outcomes in disease-related characteristics and nutritional status at the end of the study, which were significantly improved compared with baseline. Post-surgical CD patients often face substantial dietary restrictions, which can compromise adherence and limit the long-term utility of nutritional interventions. Remarkably, in this trial, all participants completed the study, suggesting that the real-world feasibility and cultural adaptability of the MMD may help mitigate adherence challenges typically encountered with restrictive regimens such as EEN. Notably, the anti-inflammatory efficacy of MMD with PEN was confirmed by significant reductions in C-reactive protein and faecal calprotectin levels, achieved without serious adverse events. These findings suggest that MMD supplemented with PEN may be a promising diet that delivers adequate nutrition and effectively reduces inflammation in post-surgical CD patients, with the potential for wider application in Chinese population.
Immunosuppression is still the primary treatment for CD, despite its potential side effects and high costs. In contrast, dietary therapy offers a promising alternative for managing CD, particularly for patients who are not suitable candidates for immunosuppressive therapy within 4–5 weeks after surgery, and it carries a relatively low risk of side effects. Due to the potential inflammatory factors present in normal diets, CD patients are often over-concerned about diet control, leading to strict and unnecessary dietary restrictions to maintain remission and prevent recurrence(Reference Norman and Hoque22). This may lead to malnutrition, additional stress, disordered eating, as well as diminished emotional and social well-being, ultimately reducing their quality of life. While EEN could provide sufficient nutrition with low or no side effects, it is non-sustainable(Reference Grover, Muir and Lewindon23) and has poor palatability that induces poor compliance and adherence(Reference Ashton, Gavin and Beattie9), limiting its efficacy. Moreover, the re-introduction of a habitual diet after EEN may induce a rebound increase in inflammatory indices in CD patients(Reference Levine, Wine and Assa6,Reference Logan, Clark and Ijaz7) . Alternative dietary approaches, such as CDED(Reference Levine, Wine and Assa6,Reference Yanai, Levine and Hirsch11,Reference Sigall-Boneh, Pfeffer-Gik and Segal24) , Mediterranean diet(Reference Lewis, Sandler and Brotherton25), solid food diet(Reference Zhang, Vootukuru and Niewiadomski26) and low fermentable oligosaccharides, disaccharides, monosaccharides and polyols diet(Reference Popa, Pop and Dumitrascu27), have shown efficacy in several studies in different populations. Importantly, CDED and similar diets may provide long-term protection by alternating the gut microbiota(Reference Correia, Oliveira and Antunes28), with high sustainability due to their relatively low cost and high rate of compliance. However, they may be less suitable for Chinese CD patients due to different dietary habits and limited ingredient accessibility. In this study, we introduced MMD supplemented with PEN to cater to Chinese CD patients’ dietary habits. MMD supplemented with PEN has a similar nutritional composition to CDED and the Mediterranean diet but is based on food ingredients that are easily accessible in China. MMD supplemented with PEN also emphasises mild cooking methods, including steaming, boiling and low-temperature frying with a limited amount of oil. These techniques may reduce inflammatory triggers in meals while promoting nutrient absorption, providing a culturally appropriate and effective dietary solution for Chinese CD patients. Indeed, MMD supplemented with PEN significantly improved the quality of life of post-surgical patients, reaching similar clinical outcomes compared with EEN. It should also be noted that patients receiving drug treatment were excluded from this study, eliminating the potential biases introduced by drug treatment in our study. The contribution of PEN should also be acknowledged in interpreting these results. As a controlled and nutrient-dense supplement, PEN may help compensate for nutritional gaps inherent in the MMD, which could have supported the improvements observed in both clinical and biochemical outcomes. Therefore, the comparable efficacy of MMD with PEN to EEN may be explained by the synergistic effect of an anti-inflammatory dietary pattern combined with the consistent nutrient delivery provided by PEN.
Another significant advantage of the MMD supplemented with PEN is its emphasis on enhanced patient monitoring and education. In this study, all patients in the MMD supplemented with PEN group received initial training in the first week and maintained regular communication with a dietitian to discuss their daily food consumption. These interactions may have strengthened their engagement and improved their awareness of dietary control. It has been shown that patient-centred care, active participation and shared decision-making are critical in long-term CD management(Reference McDermott, Healy and Mullen29). The European Crohn’s and Colitis Organisation consensus also agreed that ‘optimising quality of care in inflammatory bowel disease involves information and education’(Reference Elkjaer, Moser and Reinisch30). Such active involvement may also positively affect the patients’ psychological state and adherence by developing a sense of empowerment and control. Furthermore, patient education on diet preparation and nutrition may bring long-lasting benefits even beyond the intervention programme and contribute to long-term CD remission.
There are several limitations with this study. Firstly, this pilot, open-label, single-centred study only included a limited number of post-surgical CD patients, and the findings may be subject to bias and have limited generalisability. Since no prior data were available on the difference between the MMD + PEN and EEN groups, no a priori sample size estimation was feasible. Consequently, the small sample size may have reduced the statistical power to detect subtle between-group differences, which could partly explain the lack of statistically significant findings for quality-of-life outcomes. A well-powered study is required to validate the efficacy of MMD supplemented with PEN in a larger and more diverse patient population. Secondly, the intervention lasted only 4–5 weeks as participants were scheduled to initiate pharmacological treatments in the subsequent phase of care. This short duration may have restricted the potential to detect longer-term effects, including sustained improvements in quality of life and the ability of the MMD supplemented with PEN to induce or maintain remission. Thirdly, most participants were in remission or had mildly active disease at enrolment, which limits conclusions regarding the efficacy of MMD supplemented with PEN in patients with more severe disease activity. Finally, endoscopic evaluation was not performed at the end of the intervention, preventing confirmation of mucosal healing. Larger, adequately powered and longer-term studies are warranted to validate these findings and to further clarify the role of MMD supplemented with PEN in post-surgical CD management.
In summary, this study is the first prospective clinical trial to investigate the effects of MMD supplemented with PEN on post-surgical CD patients in China. Our findings suggest that MMD supplemented with PEN could improve the quality of life, nutritional status and disease-related characteristics in these patients, reaching outcomes comparable to those of EEN. These results suggest that MMD supplemented with PEN may serve as a promising dietary plan for the management of post-surgical CD patients. Future large-scale clinical trials are needed to validate the efficacy and safety of MMD supplemented with PEN.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114526106588
Acknowledgements
The authors gratefully acknowledge all study participants for their voluntary participation and contribution to this study.
This research was funded by the Zhejiang Provincial Health and Technology Plan (2022KY798) and the China Crohn’s & Colitis Foundation (CCCF; CCCF-QF-2022C07-15), both awarded to Xiaoxu Huang, as well as the Zhejiang Provincial Health Department Youth Innovation Project (2023RC168) and another grant from the China Crohn’s & Colitis Foundation (CCCF-QF-2022C21-16), both awarded to Qiao Yu. The funders had no role in the design of the study, data collection, data analysis, manuscript preparation or the decision to publish.
X. H. and Y. C. wrote the manuscript. Z. Z., S. L. and Y. T. were responsible for data collection. W. H., Y. H. and J. L. designed the research. Z. C. analysed the data. Y. Z., X. L., Q. Y. and P. Z. provided critical revisions for the manuscript’s content and presentation. All authors approved the manuscript’s content prior to submission.
The authors disclose no conflicts.
All data are available upon reasonable request.