Introduction
Obesity is a chronic, multifaceted condition(Reference Coral, Smit and Farzaneh1) that affects millions worldwide, with an increasing global prevalence, and may be accompanied by over 200 known complications(Reference Bray, Heisel and Afshin2). Traditional treatments for obesity, including lifestyle modifications, have yielded limited long-term success for many patients(Reference Kheniser, Saxon and Kashyap3), leading to the rise of more targeted interventions such as metabolic/bariatric surgery and pharmacotherapy. Metabolic/bariatric surgery has long been considered the gold standard for significant and sustained weight loss, as it induces anatomical changes and profound hormonal, neural and metabolic shifts that impact the disease of obesity(Reference Xie, Johnston and Waggoner4,Reference Faramia, Ostinelli and Drolet-Labelle5) . However, advancements in obesity medications are shifting the landscape, presenting patients with scalable and less invasive alternatives for controlling the disease of obesity(Reference Papamargaritis, Le Roux and Holst6). This review aims to evaluate whether pharmacotherapy can be an effective substitute for bariatric surgery, examining both approaches regarding mechanisms of action, weight loss outcomes and risk profiles.
Mechanisms of action
Both metabolic/bariatric surgery and pharmacotherapy treat the disease of obesity by targeting the subcortical areas of the brain to change adipocyte mass while having direct organ effects, such as the pancreas, liver, kidneys and heart, that impact metabolism (see Table 1). Furthermore, emerging evidence highlights the importance of individual biological variability in obesity, including genetic predisposition, which may influence treatment response and support the move towards more personalised therapeutic strategies(Reference Ko, McGillicuddy and Al-Humadi7). Metabolic/bariatric surgery alters the anatomy of the gastrointestinal tract and induces profound hormonal, neural and metabolic changes that persist long-term, supporting durable weight loss. In contrast, current pharmacotherapy primarily influences neurohormonal pathways and requires continuous use to maintain its effects, whereby cessation of medication leads to loss of treatment-mediated weight control(Reference Bray, Heisel and Afshin2,Reference Kheniser, Saxon and Kashyap3,Reference Wilding, Batterham and Davies8,Reference Rubino, Abrahamsson and Davies9) . These distinct mechanisms of action result in varied outcomes across patients, as individual responses and preferences for treatment can differ based on each patient. Understanding these divergences provides a nuanced framework for assessing each treatment’s advantages and limitations, particularly in managing obesity as a chronic disease. Different from medications, metabolic/bariatric surgery is usually a single procedure that leads to long-lasting physiological responses.
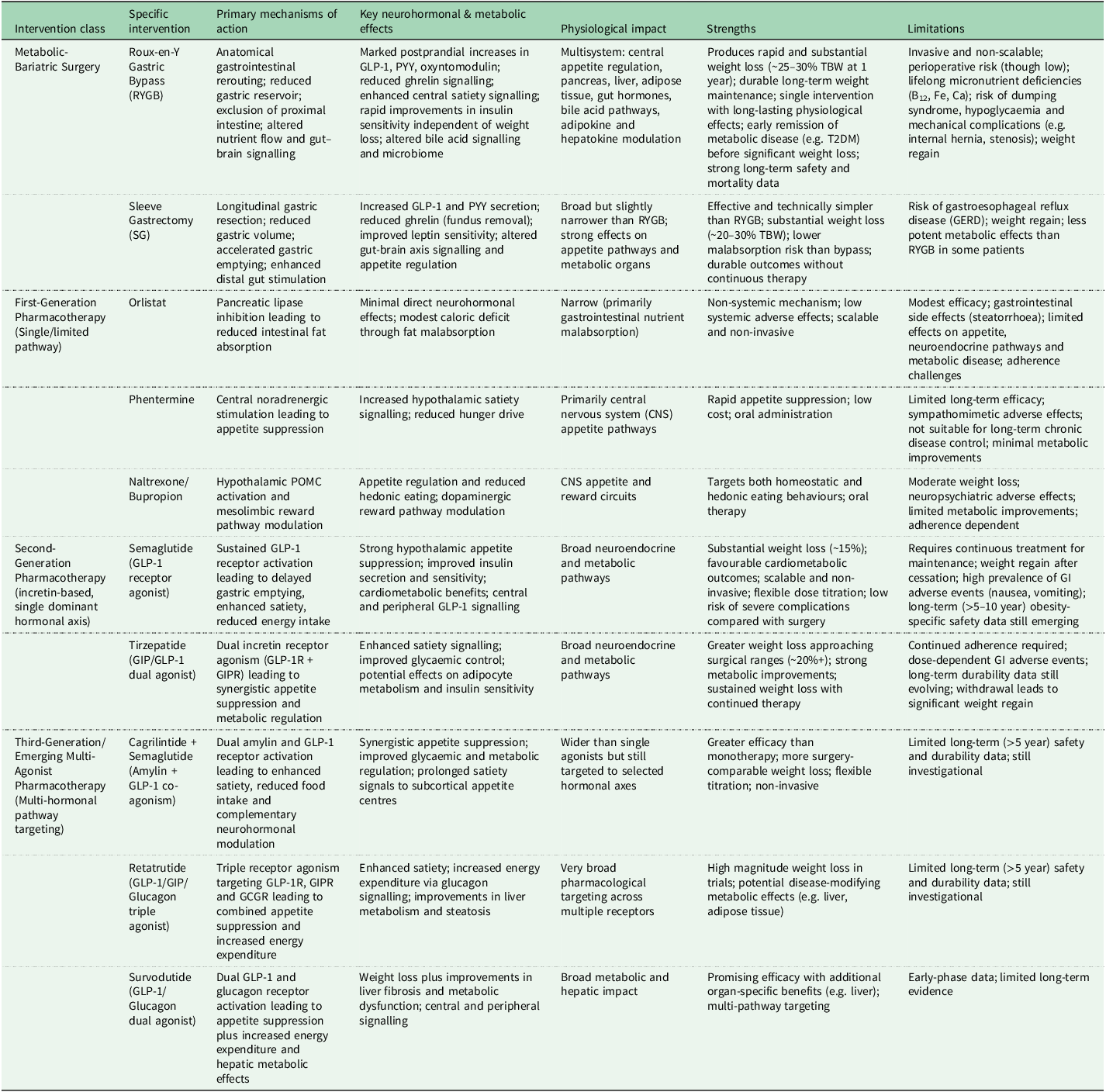
Comparative mechanisms of action, physiological effects and strengths/limitations of metabolic/bariatric surgery versus obesity pharmacotherapies

TBW, total body weight; GLP-1, glucagon-like peptide-1; GIP, glucose-dependent insulinotropic polypeptide; GCGR, glucagon receptor; PYY, peptide YY; CNS, central nervous system; T2DM, type 2 diabetes mellitus.
The most common types of bariatric surgery are Roux-en-Y Gastric Bypass (RYGB) and Sleeve Gastrectomy (SG). RYGB and SG reduce the size of the stomach, and the former reroutes the small intestine. This causes enhanced signals from the gut to the brain, which controls the disease of obesity. In the dynamic phase of weight reduction, patients feel less hungry and more full, have reduced appetitive behaviour and eat smaller portions, partly attributed to the postprandial gut hormone responses(Reference Chang, Stoll and Song10–Reference Chong, Ko and le Roux12). Some of these hormones, such as glucagon-like peptide 1 (GLP-1) and oxyntomodulin, can also directly impact receptors on organs such as the pancreas to improve type 2 diabetes mellitus (T2DM) or the liver to improve fibrosis, even before substantial weight loss occurs(Reference Courcoulas, Gallagher and Neiberg13).
To provide greater context and facilitate interpretation of therapeutic advancements, it is helpful to distinguish obesity pharmacotherapies by their primary mechanisms of action. First-generation medications, including orlistat, phentermine and naltrexone/bupropion act through relatively limited or single-pathway mechanisms(Reference Bray, Heisel and Afshin2,Reference Khera, Murad and Chander14) : orlistat reduces intestinal fat absorption via pancreatic lipase inhibition; phentermine primarily enhances central noradrenergic signalling to suppress appetite; and naltrexone/bupropion modulates hypothalamic appetite regulation and mesolimbic reward pathways. In contrast, second-generation agents such as semaglutide provide more potent and sustained GLP-1 receptor activation, leading to stronger effects on appetite regulation, energy intake and glycaemic control through enhanced signalling to subcortical appetite centres and peripheral metabolic organs(Reference Garvey, Batterham and Bhatta15,Reference Ryan, Lingvay and Deanfield16) . Emerging third-generation therapies, including Cagrilintide/Semaglutide (Cagri/Sema), retatrutide and survodutide, are designed as multi-agonist or combination therapies targeting multiple gut hormone pathways (e.g. GLP-1, amylin, GIP and/or glucagon receptors), thereby producing synergistic effects on satiety, energy expenditure and metabolic regulation(Reference Khera, Murad and Chander14,Reference le Roux, Steen and Lucas17–Reference Frias, Deenadayalan and Erichsen20) . This progressive shift from single-target to multi-hormonal pathway modulation helps explain the increasingly greater and more surgery-comparable weight loss observed with newer agents(Reference Papamargaritis, Le Roux and Holst6).
Dynamic phase of weight reduction
Weight reduction is a helpful metric in assessing the efficacy of obesity interventions, especially when considered alongside resolution of obesity complications. Short-term weight loss, typically assessed within 1–2 years post-intervention, provides insight into the potency of a treatment, fosters patient adherence and helps identify how well the patient responds to the intervention. When comparing metabolic/bariatric surgery to pharmacotherapy, surgery remains the most effective intervention for inducing rapid and significant short-term weight loss. However, third-generation pharmacotherapies may close this gap.
RYGB and SG are effective bariatric interventions, with approximately 25–30% reduction in total body weight within 1 year(Reference Kim, Kim and Park11,Reference Courcoulas, Gallagher and Neiberg13,Reference Peterli, Wölnerhanssen and Peters21–Reference Hofsø, Fatima and Borgeraas29) . The pace of weight loss with pharmacotherapy is slower than with surgery. Among the second-generation pharmacotherapies, tirzepatide (15 mg weekly) had a nadir weight loss of 21% (95% CI −21.8 to −19.9) at 72 weeks in a double-blind, randomised, controlled trial (n = 2539)(Reference Jastreboff, Aronne and Ahmad30), nearing the efficacy of sleeve gastrectomy. Subcutaneous semaglutide (2.4 mg, once a week) had a nadir weight loss of approximately 15% at 68 weeks (estimated treatment difference, −12.4 percentage points; 95% CI −13.4 to −11.5; P < 0.001) in a double-blind randomised controlled trial (n = 1961)(Reference Wilding, Batterham and Calanna31), while oral semaglutide (50mg, once per day) had a nadir weight loss of 15.1% after 68 weeks in a randomised, double-blind controlled trial (n = 667)(Reference Knop, Aroda and do Vale32). Cagri/Sema (administered as a fixed-dose of 2.4 mg each of carilintide and semaglutide subcutaneously once weekly) showed a 20.4% reduction in total body weight after week 68, compared to 3% with placebo in a randomised, double-blind controlled trial (n = 3417)(Reference Garvey, Blüher and Contreras33).
Aside from assessing average weight loss, evaluating the percentage of patients experiencing significant weight loss measures how many individuals achieve clinically meaningful outcomes from a treatment. Unlike average total weight loss, which outliers can skew, this metric highlights the consistency of the treatment’s effectiveness across the patient population. 86.2% of patients after metabolic/bariatric surgery achieved ≥20% total weight loss at the time of survey (reflecting outcomes across approximately 0–24 years post-surgery), with higher rates observed within 0–9 years post-surgery (89.3%) and sustained but slightly lower rates at 10–24 years (81.2%)(Reference Xie, Johnston and Waggoner4). Subcutaneous semaglutide (2.4 mg weekly) resulted in 32% of patients losing ≥20% at 68 weeks(Reference Wilding, Batterham and Calanna31). For tirzepatide (15 mg), 57% achieved ≥20% total body weight loss(Reference Jastreboff, Aronne and Ahmad30). This difference reflects surgery’s broader and more consistent metabolic impact compared to medications that target fewer signalling pathways in the subcortical brain areas involved in controlling adipocyte mass(Reference Akalestou, Miras and Rutter34).
CagriSema is one of the third-generation obesity medications in development(Reference Yao, Zhang and Li19), combining Cagrilintide, a long-acting acylated amylin analogue, with semaglutide, a GLP-1 agonist. A phase 2, multi-site double-blind randomised control trial, only extending into approximately half of the dynamic phase of weight reduction, showed 15.6% total weight loss at 26 weeks (2.4mg), compared to 5.1 and 8.1% with semaglutide (2.4 mg) and cagrilintide (2.4 mg) alone(Reference Frias, Deenadayalan and Erichsen20). Survodutide is another third-generation obesity medication binding the GLP-1 and glucagon receptors with weight reductions of up to 19% while still in the dynamic phase of weight reduction at 48 weeks(Reference Klein, Augustin and Hennige35), while also having additional benefits through glucagon receptors in the liver to reverse liver fibrosis(Reference Sanyal, Bedossa and Fraessdorf36). Retatrutide, which binds the glucagon receptor (GCGR), glucose-dependent insulinotropic polypeptide receptor (GIPR) and glucagon-like peptide-1 receptor (GLP-1R), resulted in an average weight loss of 17.5 and 24.4% at 24 and 48 weeks, respectively, while still in the dynamic phase of weight(Reference Kaur and Misra18), which is a substantially greater weight loss than previous drugs such as semaglutide, tirzepatide at 48 weeks of treatment. At 24 weeks of treatment, 86% of patients with established metabolic dysfunctional associated steatohepatitis (MASH) also achieve improved liver function, demonstrating health gains beyond rapid weight loss(Reference Sanyal, Kaplan and Frias37). However, despite these promising early results, the durability and long-term outcomes are yet to be established and these are still early clinical trials, with less established long-term data.
Pharmacotherapy is still less effective than surgery, but the ability to adjust the dose titration schedule produces weight loss more gradually and has fewer side effects during the dynamic phase of weight reduction. However, this also means a reduction in the rapidity of the metabolic shift, which may delay the resolution of obesity-related complications. However, for patients who prefer non-invasive treatments, the weight loss achieved with pharmacotherapy may be substantial enough(Reference Weintraub, D’Angelo and Tchang38).
Stable phase of weight maintenance
Long-term weight loss, typically evaluated beyond 2 years post-intervention, is crucial for sustained health benefits and reducing obesity-related complications, especially with the treatment of obesity as a chronic disease. Metabolic/bariatric surgery has demonstrated favourable long-term weight loss outcomes, with RYGB and SG patients maintaining 27.2 and 23.7% total body weight reduction at five years and beyond, respectively(Reference Ignat, Vix and Imad22,Reference Wölnerhanssen, Peterli and Hurme25) . RYGB, compared to SG, also has fewer patients who regain more than half of the weight they have lost after 5 years (5.4 vs 17.9%)(Reference Ignat, Vix and Imad22). Longer-term data after 10 years of RYGB is also reassuring with weight maintenance compared to nadir weight loss, albeit the variation of weight loss does get wider(Reference Nguyen, Kim and Vu39–Reference Sjöholm, Carlsson and Svensson41). Results from the Swedish Obese Subjects (SOS) study further suggests this weight maintenance is sustained after 20 years in surgical patients with significant reduction in mortality(Reference Sjöström42). The majority of surgical patients who do have recurrent weight gain never reach their preoperative weight(Reference Akalestou, Miras and Rutter34).
The long-term efficacy of obesity medication is only starting to emerge. Semaglutide in patients with established CVD in the SELECT study resulted in a mean weight nadir at the end of the dynamic phase of weight reduction of −10.2% at 208 weeks that was sustained until the end of the trial period of 4 years(Reference Ryan, Lingvay and Deanfield16). Semaglutide in patients without diabetes in the STEP 5 study resulted in a nadir at the end of the dynamic phase of weight reduction at 60 weeks of 15.6%. After 2 years and during the stable phase of weight maintenance, 15.2% weight loss was sustained(Reference Garvey, Batterham and Bhatta15). For tirzepatide, emerging long-term maintenance data similarly demonstrate a distinct dynamic phase followed by a stable phase of weight maintenance with continued therapy. In the SURMOUNT-4 randomised withdrawal trial, participants achieved a mean weight reduction of 20.9% during the initial 36-week lead-in (dynamic phase)(Reference Aronne, Sattar and Horn43). Those who continued tirzepatide for an additional 52 weeks experienced a further modest reduction (−5.5% from week 36) and maintained the majority of their prior weight loss, with 89.5% preserving at least 80% of the weight lost during the lead-in period(Reference Aronne, Sattar and Horn43). Overall weight reduction reached approximately 25.3% at 88 weeks, indicating sustained efficacy through the maintenance phase, whereas withdrawal led to significant regain(Reference Aronne, Sattar and Horn43).
The STEP 1 extension and the Surmount 4 trial showed that long-term use of pharmacotherapy is needed for weight loss maintenance, as in both studies, withdrawing medication incurred in recurrent weight gain(Reference Wilding, Batterham and Davies8,Reference Aronne, Sattar and Horn43) . In contrast, after metabolic/bariatric surgery, a significant number of patients maintain their weight loss with just a single intervention.
Newer agents, including retatrutide, survodutide and cagrisema, have demonstrated more substantial short-term weight loss in clinical trials(Reference Papamargaritis, Le Roux and Holst6,Reference Kaur and Misra18,Reference Frias, Deenadayalan and Erichsen20,Reference Jastreboff, Aronne and Ahmad30,Reference Klein, Augustin and Hennige35) . The association of these drugs with more significant weight loss outcomes is promising, suggesting the potential for more profound long-term impacts. However, long-term data is currently lacking for these agents, making it difficult to ascertain their sustained efficacy and compare it to the established long-term efficacy of metabolic/bariatric surgery. Based on the longer-term data from semaglutide and tirzepatide, the expectation is that these third-generation medications will maintain the initial weight loss and have shown promising results thus far(Reference Garvey, Batterham and Bhatta15,Reference Jastreboff, le Roux and Stefanski44) . Metabolic/bariatric surgery remains the intervention with the most robust evidence for achieving and maintaining substantial weight loss over decades(Reference Elmaleh-Sachs, Schwartz and Bramante45,Reference Carlsson, Carlsson and Jacobson46) . While emerging longer-term evidence from semaglutide and tirzepatide suggests sustained weight loss during continued treatment, the absence of multi-decade follow-up for newer medications makes direct comparisons with the established long-term outcomes of metabolic/bariatric surgery premature.
This may be explained by the multifaceted mechanisms that impact both the physical structure of the gastrointestinal (GI) tract and a broad spectrum of metabolic, hormonal and cellular processes that are implicated in surgery, which extend beyond the effects of pharmacotherapy that are conducive to the maintenance of lower body weight(Reference Akalestou, Miras and Rutter34). Furthermore, the third generation medications (i.e. retatrutide, survodutide, and cagrisema) affect more than one but still selected hormonal pathways. While substantial weight loss (following both surgical and non-surgical intervention) facilitates a reduction in pro-inflammatory adipokines (e.g. TNF-α and IL-6) and an increase in anti-inflammatory adipokines (such as adiponectin and omentin), specific alterations in adipokine dynamics suggest a more complex, surgery-induced physiological response(Reference Faramia, Ostinelli and Drolet-Labelle5). Notably, immediate post-surgical shifts in adipokine (e.g. omentin), myokine (e.g. myostatin) and hepatokine (e.g. SHBG, adropin) levels can be observed well before significant weight reduction(Reference Faramia, Ostinelli and Drolet-Labelle5), likely in response to acute metabolic adjustments and nutrient signalling alterations. This early shift points to a direct effect of surgery on these protein expressions(Reference Faramia, Ostinelli and Drolet-Labelle5). Moreover, reported enhanced leptin sensitivity post-surgery, as suggested by increased leptin receptor expression in subcutaneous adipose tissue, underscores the potential role of bariatric surgery in modulating hypothalamic signalling pathways(Reference Khosravi-Largani, Nojomi and Aghili47). Whether obesity medications may improve leptin sensitivity or how they control the disease of obesity in the subcortical areas of the brain remains unknown(Reference Coutinho and Halpern48).
Recurrent weight gain and treatment adherence
Recurrent weight gain remains a critical challenge in the long-term management of obesity, impacting the sustainability of the benefits accrued by either metabolic/bariatric surgery and pharmacotherapy. Metabolic/bariatric surgery has the advantage that the anatomy does not revert to the presurgical state; therefore, recurrent weight gain is more likely related to a loss of weight control associated with the underlying chronic disease of obesity. For RYGB, all patients’ mean recurrent weight gain is 23.4% of maximum weight loss(Reference Cooper, Simmons and Webb49). For SG, up to 75.6% of SG patients experience some recurrent weight gain for 6 years. However, outcomes varied significantly between patients(Reference Lauti, Kularatna and Hill50). Therefore, while metabolic/bariatric surgery offers a more stable solution to weight loss than pharmacotherapy, ongoing support and behavioural modification remain essential to mitigate recurrent weight gain risks.
Recurrent weight gain may also be a significant challenge with pharmacotherapy. Not enough data exists to understand the impact of the disease of obesity on recurrent weight gain after pharmacotherapy, but recurrent weight gain has especially been linked with medications being discontinued. Thus far, no study has demonstrated sustained weight loss after discontinuation of pharmacotherapy(Reference Bray, Heisel and Afshin2). This is unsurprising because other chronic diseases, such as hypertension, dyslipidaemia and type 2 diabetes, also lose treatment-mediated control when therapy is withdrawn(Reference Burnier51). Discontinuation of obesity medications leads to substantial weight gain, emphasising sustained adherence for continued efficacy(Reference Kheniser, Saxon and Kashyap3,Reference Wilding, Batterham and Davies8,Reference Rubino, Abrahamsson and Davies9) . Within 68 weeks of semaglutide cessation, patients regain an average of 67% of their nadir weight loss, leading to a net weight loss of only 5.6% from baseline, albeit more recurrent weight gain can be expected given that the slopes of the recurrent weight gain curves did not suggest a stable phase of weight maintenance(Reference Wilding, Batterham and Davies8). The body weight reached after 68 weeks of stopping semaglutide is comparable to the body weight typically achieved through lifestyle interventions alone if participants had not commenced semaglutide(Reference Kheniser, Saxon and Kashyap3). In another study, after a mean weight loss of 10.6% undergoing 20 weeks of semaglutide (2.4 mg), the mean recurrent weight gain at week 68 was 65.1% of the nadir weight at week 20, demonstrating cessation of drug administration reverses weight loss effects(Reference Rubino, Abrahamsson and Davies9). In SURMOUNT-4, participants achieved a mean weight loss of 20.9% during 36 weeks of tirzepatide treatment(Reference Aronne, Sattar and Horn43). After randomisation, those who discontinued therapy regained approximately 14% of body weight over the following 52 weeks, whereas those who continued tirzepatide lost an additional 5.5%. At week 88, only 16.6% of participants who stopped tirzepatide maintained ≥80% of their initial weight loss compared with 89.5% in the continuation group, demonstrating substantial weight regain after cessation and sustained efficacy with ongoing treatment(Reference Aronne, Sattar and Horn43).
The interpretation of weight loss data from these drugs is further complicated by high dropout rates of obesity pharmacotherapy within clinical trials. In an analysis of 28 randomised controlled trials of various weight-loss medications, attrition rates were 30–45%(Reference Khera, Murad and Chander14). A significant portion of discontinuation in clinical trials is due to adverse events(Reference Khera, Murad and Chander14), however reported side effects of pharmacotherapy and placebo are similar(Reference Rubino, Greenway and Khalid52). However, this may also relate to protocol-driven titration schedules used in clinical trials, which can increase adverse events in patients who may not tolerate target doses but could otherwise be maintained on a lower, maximum tolerable dose(Reference Caffrey and Borrelli53,Reference Pledger54) . Greater flexibility in dose titration may improve tolerability and treatment persistence. However, while flexible titration may improve tolerability and adherence(Reference Caffrey and Borrelli53,Reference Pledger54) , the magnitude of weight loss with anti-obesity pharmacotherapy is partly influenced by the dose tolerated and sustained as discussed earlier in this paper, meaning patients maintained on lower doses may experience smaller weight reductions than in trials employing fixed escalation to higher target doses.
Unlike metabolic/bariatric surgery, which modifies the gastrointestinal anatomy to produce long-lasting control of the disease of obesity, pharmacotherapy relies on the continuation of the medication to control the disease. However, pharmacotherapy offers the advantage of titration, allowing for precise adjustments in dosing to target specific receptors and neuroendocrine pathways in response to changes in weight. This enables clinicians to influence metabolic processes in a controlled manner directly. In contrast, while highly effective, metabolic/bariatric surgery provides less control over its impacts on these neuroendocrine pathways. Over time, the initial alterations in key hormonal mediators of appetite and metabolism, such as leptin and gut hormones induced by metabolic/bariatric surgery, tend to diminish(Reference El Ansari and Elhag55). This attenuation can partly lead to the re-emergence of weight gain, highlighting a limitation in the long-term efficacy of surgical interventions for weight management(Reference El Ansari and Elhag55). Thus, despite the superior weight loss effects of surgery, the adaptability of pharmacotherapy through dose titration provides a distinct advantage in addressing long-term metabolic challenges and mitigating recurrent weight gain.
Importantly, this discussion does not contradict the earlier observation that metabolic/bariatric surgery produces greater and more durable average documented weight loss than pharmacotherapy in current literature at the population level. Rather, the distinction lies in therapeutic flexibility. While surgery induces substantial and sustained anatomical and hormonal changes that underpin superior long-term weight loss as currently documented, these effects are not readily adjustable once performed. In contrast, pharmacotherapy allows dose titration and ongoing modulation of neuroendocrine pathways, which may offer a practical advantage in managing weight regain over time. Thus, although surgical intervention generally achieves larger and more sustained weight reductions, pharmacotherapy provides a dynamic, adjustable approach to long-term disease control, particularly in the context of recurrent weight gain.
Adverse effects and side effects
Both surgery and pharmacotherapy carry distinct profiles of adverse effects, side effects and discontinuation rates that underscore the need for both options, as each may be more appropriate for different patients depending on their health conditions, treatment goals and tolerance for potential risks. The adverse effect profiles of bariatric surgery procedures vary based on the type and extent of anatomical alteration. Mortality within 30 days post-surgery is approximately 1 in 1000(Reference Chang, Stoll and Song10). Mortality because of surgical complications within 90 days and beyond is also low, about 4 in 1000(Reference Chang, Stoll and Song10). Severe adverse events were rare and occurred in 5–8%, and often no reoperations were required(Reference Verrastro, Panunzi and Castagneto-Gissey23). Early complications within 30 days post-surgery are also rare, at 0.9 and 4.5% in SG and RYGB respectively(Reference Peterli, Wölnerhanssen and Peters21). This rises to 14.9 and 17.3% for late complications for SG and RYGB, respectively, but still remains low(Reference Peterli, Wölnerhanssen and Peters21). The most common adverse effects post-surgery are vomiting and constipation(Reference Verrastro, Panunzi and Castagneto-Gissey23,Reference Schiavon, Bersch-Ferreira and Santucci24) . Given this excellent safety profile and the benefits of surgery, patients may consider it more risky not to have surgery than to have surgery(Reference Carlsson, Carlsson and Jacobson46).
Pharmacotherapy is less invasive and does not carry the same 30- and 90-day mortality risks as surgery(Reference Iannone, Natale and Palmer56). This makes it an attractive option for many patients, especially when weight loss outcomes with newer drugs are approaching closer to those previously only seen with surgical intervention. However, while the rates of reported side effects for patients undergoing pharmacotherapy are high – 95.2% on semaglutide and 96.1% on liraglutide – it is worth noting that this is not substantially higher than the 95.3% of participants in the placebo group who also reported adverse events(Reference Rubino, Greenway and Khalid52). After 4 years of semaglutide in older patients with established CVD, serious adverse events were reported by 33.4%, which was lower than the 36.4% in the placebo group(Reference Lincoff, Brown-Frandsen and Colhoun57). This also suggests that taking medications may be safer than not taking medications. Serious adverse events (SAEs), defined as events resulting in death, life-threatening outcomes, hospitalisation or prolongation of hospitalisation, persistent or significant disability, congenital anomaly, or other medically important events, were noted across all drug groups, though with varying incidences(Reference Papamargaritis, Le Roux and Holst6,Reference Yao, Zhang and Li19,Reference Wilding, Batterham and Calanna31,Reference Rubino, Greenway and Khalid52,Reference Iannone, Natale and Palmer56) . For semaglutide, 9.8% of participants experienced SAEs, compared to 6.4% in the placebo group, with the higher rate attributed mainly to GI disorders (1.4% in semaglutide vs. 0% in placebo) and hepatobiliary disorders (1.3% in semaglutide vs. 0.2% in placebo)(Reference Wilding, Batterham and Calanna31). For tirzepatide, discontinuation rates due to adverse events were dose-dependent: 4.3% in the 5 mg group, 7.1% in the 10 mg group and 6.2% in the 15 mg group, compared to 2.6% in the placebo group(Reference Jastreboff, Aronne and Ahmad30). These discontinuation rates are critical indicators of the severity of side effects and reflect patient tolerability, as continuation is critical for intervention success. Furthermore, a more gentle dose titration schedule may have attenuated many of these adverse events.
Gastrointestinal complications are the most commonly reported adverse event from surgery and pharmacotherapy. Dumping syndrome is a common adverse effect that affects 15.7% of patients for all procedures within six months and can increase up to 25% of patients 1–5 years after surgery(Reference D’Hoedt and Vanuytsel58). This is most prominent in RYGB due to the lack of a pyloric valve, which usually controls the release of stomach contents into the small intestine and allows hyperosmolar food to enter the jejunum rapidly. This shift leads to an osmotic gradient that draws fluid into the intestine, resulting in bloating, diarrhoea and abdominal cramping. The rapid glucose absorption also triggers a hyperinsulinemia response, causing postprandial hypoglycaemia in some patients(Reference Schiavon, Bersch-Ferreira and Santucci24). SG has also been linked with gastroesophageal reflux disease (GERD) in approximately 20% of patients due to increased intragastric pressure and altered gastric anatomy, compared to only 1% in RYGB(Reference Verrastro, Panunzi and Castagneto-Gissey23). The most common GI side effects across procedures include vomiting and constipation(Reference Verrastro, Panunzi and Castagneto-Gissey23,Reference Schiavon, Bersch-Ferreira and Santucci24) .
In comparison, vomiting and nausea are extremely common in GLP-1 receptor agonists in a dose-dependent relationship(Reference Bettge, Kahle and El Aziz59). Rates of nausea are 30–40% in tirzepatide and semaglutide (2.4 mg subcutaneously), respectively(Reference Elmaleh-Sachs, Schwartz and Bramante45). Vomiting rates were 12–25%, and constipation rates were 12–23% in the same drugs, respectively, suggesting higher rates than surgery(Reference Elmaleh-Sachs, Schwartz and Bramante45). This apparent difference may partly reflect methodological differences in adverse event ascertainment(Reference Sartor60). Pharmacotherapy trials typically employ systematic, prospective collection of patient-reported symptoms at predefined intervals(Reference Bettge, Kahle and El Aziz59), increasing the likelihood of capturing mild and transient events such as nausea, vomiting and diarrhoea. In contrast, surgical studies often prioritise perioperative complications and may rely on less standardised patient-reported outcome measures, potentially leading to underreporting of subjective gastrointestinal symptoms, particularly when they are anticipated as part of the immediate postoperative course(Reference Coulman, Abdelrahman and Owen-Smith61).
The incidence of GI symptoms such as nausea, diarrhoea, vomiting and constipation is high in patients treated with semaglutide (74.2%), compared to placebo (47.9%), with most cases being mild to moderate in severity and resolving without requiring discontinuation(Reference Wilding, Batterham and Calanna31). Similarly, tirzepatide participants reported GI symptoms primarily during forced dose escalation, with an adverse event incidence ranging from 78.9 to 81.8% across different doses, as opposed to 72.0% in the placebo group(Reference Jastreboff, Aronne and Ahmad30).
Patients after metabolic/bariatric surgery are at a higher risk of nutritional deficiencies(Reference Schiavon, Bersch-Ferreira and Santucci24). In RYGB, the food bypasses key segments of the intestine where intrinsic factor binds with vitamin B12 for absorption and where nutrients such as Fe and Ca are typically absorbed(Reference Mohapatra, Gangadharan and Pitchumoni62). This may exacerbate nutritional deficiencies(Reference Mohapatra, Gangadharan and Pitchumoni62). In RYGB patients, the risk of anaemia is 11%, secondary hyperparathyroidism is 14%, hypovitaminosis B12 is 28% and ferritin deficiency is 5%(Reference Schiavon, Bersch-Ferreira and Santucci24). In contrast, SG, which retains a larger portion of the small intestine in contact with food, has a lower risk of severe nutrient deficiencies. However, SG can still contribute to deficiencies indirectly, particularly in Fe and B12, due to reduced food intake and gastric acid production, which are critical for nutrient absorption(Reference Mohapatra, Gangadharan and Pitchumoni62). In comparison, the risk for malnutrition is much lower in patients undergoing pharmacotherapy, albeit longer-term data are still awaited.
Unlike pharmacotherapy, metabolic/bariatric surgeries also pose risks of mechanical complications(Reference Elmaleh-Sachs, Schwartz and Bramante45). The rate of anastomotic leaks (leakage at the surgical connection between segments of the gastrointestinal tract) and stenosis (narrowing of the surgical connection causing obstruction) is relatively low but constitutes a severe complication when it occurs. Anastomotic leaks have an incidence rate of 1–7 and 0.6–4.4% for SG and RYGB, respectively, and stenosis has an incidence rate of 1–9% and 8–19% for SG and RYGB, respectively(Reference Elmaleh-Sachs, Schwartz and Bramante45). Incidences of intestinal obstruction (blockage of the bowel), internal hernias (protrusion of the intestine through mesenteric defects created during surgery) and intussusception (inward folding of one segment of intestine into another) are rare but still present (<1%, 1% & 1% respectively)(Reference Verrastro, Panunzi and Castagneto-Gissey23). The most common complication following RYGB is ulceration(Reference Nguyen, Kim and Vu39). SG has a similar risk profile but is associated with specific complications such as staple line leaks (1%), gastric stenosis (1%) and haemoperitoneum (bleeding into the abdominal cavity) (<1%)(Reference Verrastro, Panunzi and Castagneto-Gissey23). These complications, though infrequent, can increase morbidity and often require reoperation to prevent mortality. In a 10-year prospective randomised controlled trial, reoperation rates across surgical patients were 18.3%(Reference Nguyen, Kim and Vu39). However, Stenberg et al. reported internal hernias to be the leading cause of this higher reoperations rate, as surgeons did not close the Petersen and mesenteric defects, indicating the impact of this can be mitigated to lower reoperation rates further(Reference Carlsson, Carlsson and Jacobson46). Despite reoperation rates being generally low, these are not a feature of pharmacological treatments.
However, unlike metabolic/bariatric surgery, which has a well-established safety profile documented over decades, the newer obesity medications lack comparable data on long-term safety and efficacy. This absence of longitudinal evidence of safety does not mean there will be significant safety concerns, but it presents a significant gap in understanding these drugs’ potential risks and sustained benefits. As pharmacological agents achieve weight loss outcomes approaching those traditionally seen with surgery, this may change the profile of patients opting for surgical intervention. Thus, patients requiring surgery may be older, have higher rates of complications of obesity and be at higher risk for surgical complications.
Combining surgery and pharmacotherapy
The combination of bariatric surgery and pharmacotherapy offers a novel approach to managing obesity, capitalising on the strengths of each treatment modality to address the limitations observed when either is used alone. This integrated strategy may be particularly beneficial in maximising initial weight loss, sustaining long-term weight maintenance, reducing complications of obesity and mitigating weight regain – challenges often encountered in obesity management.
Pharmacotherapy has the potential to enhance surgical outcomes both preoperatively and postoperatively. When administered before surgery, obesity medications may facilitate initial weight loss, lower surgical risks and improve operative conditions. Although preoperative weight loss is not a prerequisite for metabolic/bariatric surgery(Reference Rubio-Herrera, Mera-Carreiro and Sánchez-Pernaute63), moderate evidence suggests that patients experience fewer perioperative complications with increased preoperative weight loss, which pharmacotherapy can facilitate(Reference Rubio-Herrera, Mera-Carreiro and Sánchez-Pernaute63,Reference Kushner and Eagon64) . Patients who achieved preoperative weight loss show modest reductions in surgical complications, such as anastomotic leaks and deep infections, and improved operative factors, including reduced liver size, less intraoperative blood loss and decreased operating time(Reference Kushner and Eagon64,Reference Tewksbury, Williams and Dumon65) . Greater preoperative weight loss is associated with a slightly reduced risk of perioperative complications (9.1 vs. 10.3%), though the overall impact remains modest(Reference Kushner and Eagon64,Reference Livhits, Mercado and Yermilov66) . This observation is not specific to the weight loss method; however, given pharmacotherapy’s increasing availability and efficacy, it is reasonable to hypothesise that incorporating pharmacological treatments before surgery is beneficial. In 2008, the American Association of Clinical Endocrinologists, The Obesity Society and American Society for Metabolic & Bariatric Surgery Medical Guidelines for Clinical Practice guidelines concluded that preoperative weight loss could improve the technical aspects of bariatric surgery based on moderate-quality evidence(Reference Mechanick, Kushner and Sugerman67). Subsequent studies and reviews, including a systematic review by Livhits et al., found that preoperative weight loss reduced operating time by an average of 23 minutes and recommended a 10% excess weight loss to optimise surgical outcomes(Reference Livhits, Mercado and Yermilov68). Livhits et al. conclude that there does not appear to be a significant perioperative risk for patients with obesity who lose 10% of their excess body weight prior to undergoing bariatric surgery(Reference Livhits, Mercado and Yermilov68). With the advent of new pharmacotherapies to achieve significant weight loss that surpasses lifestyle intervention alone, combining medical weight management with surgical preparation offers a promising approach to enhance obesity management outcomes further.
Postoperatively, pharmacotherapy can serve as an adjunct to mitigate recurrent weight gain by sustaining neurohormonal adaptations and appetite suppression, addressing one of the primary challenges in maintaining long-term weight loss after surgery(Reference El Ansari and Elhag55,Reference Cohen and Petry69) . The benefit of this is two-fold: to enhance weight loss in the remission of the disease of obesity and the dynamic phase of weight loss and to mitigate the impacts of weight regain. While bariatric surgery induces favourable metabolic shifts, the addition of pharmacotherapy extends these benefits, especially for patients with severe metabolic dysfunction or for non-responders to surgery(Reference Cohen and Petry69–Reference Mok, Adeleke and Brown71). When used alongside bariatric surgery, these agents help to stabilise metabolic improvements, providing a sustained remission of obesity-related complications(Reference Cohen and Petry69–Reference Mok, Adeleke and Brown71). In the long-term, post-surgical alterations in the levels of ghrelin, leptin and incretins diminish, resulting in recurrent weight gain due to physiological adaptions(Reference El Ansari and Elhag55). The addition of postoperative pharmacotherapy may help to counterbalance these adaptations, providing a sustained suppression of hunger and stabilisation of weight(Reference Miras, Pérez-Pevida and Aldhwayan70–Reference Jensen, Renström and Aczél72). Miras et al. found that adjunct therapy with liraglutide post-surgery was beneficial in increasing weight loss and improving glycaemic profile without serious adverse events related to treatment(Reference Miras, Pérez-Pevida and Aldhwayan70). A trial of 50 patients with recurrent weight gain showed that adjunct treatment with GLP-1 receptor agonists resulted in a loss of 67.4% of the recurrent weight gain over six months with no serious adverse events(Reference Jensen, Renström and Aczél72). Furthermore, the metabolic shift induced through surgery may not be significant enough in some surgical patients, resulting in insufficient weight loss post-surgery(Reference Mok, Adeleke and Brown71). For these patients with suboptimal post-surgical weight loss and GLP-1 response, a trial of 3.0 mg daily liraglutide adjunct treatment resulted in −7.67% adjusted mean difference in body weight post-surgery after 24 weeks, with significantly more patients achieving >5% weight loss than placebo (71.9 vs 8.8%), and no serious adverse events(Reference Mok, Adeleke and Brown71). Patients did not reach weight nadir by the end of the trial either, further suggesting that more weight loss and health benefits are achievable if continued(Reference Mok, Adeleke and Brown71). By reinforcing the effects of surgery through continuous neurohormonal modulation, the combination of surgery and pharmacotherapy addresses the limitations of each intervention, offering a more comprehensive treatment option.
While the combined use of bariatric surgery and pharmacotherapy is promising, optimal combination strategies or a unified protocol about dosing, initiation and treatment is yet to be established. Additionally, the altered pharmacokinetics in post-bariatric patients, especially those undergoing significant gastrointestinal modifications, may influence drug absorption and efficacy, necessitating careful monitoring and potential dose adjustments(Reference Konstantinidou, Argyrakopoulo and Dalamaga73). In available trial results, patient responses to pharmacotherapy post-surgery vary(Reference Miras, Pérez-Pevida and Aldhwayan70,Reference Mok, Adeleke and Brown71) , with different bariatric procedures inducing distinct metabolic and hormonal changes that may influence the effectiveness of adjunct treatments. The studies focus on patients with specific characteristics, such as poorly controlled diabetes or those with insufficient GLP-1 response, implying the importance of tailoring treatments based on metabolic profiles. Until better predictors of response can be identified, a comprehensive and holistic approach remains essential for effective long-term management of obesity(Reference Ko, Miras and Pournaras74).
In this context, the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO) recently published a landmark guideline on the role of obesity medications in the context of metabolic/bariatric surgery(Reference Cohen, Busetto and Levinson75). Based on expert consensus and extensive evidence, this guideline provides a roadmap for healthcare providers worldwide, offering practical recommendations on when and how to incorporate pharmacotherapy into surgical care. These guidelines underscore the importance of a multimodal approach, integrating medications both preoperatively (neoadjuvant therapy) to reduce surgical risks and postoperatively (adjuvant therapy) to maintain weight loss and prevent regain.
Conclusion
In comparing metabolic/bariatric surgery and pharmacotherapy as interventions for obesity, it is evident that obesity medications, though increasingly effective, do not substitute the multifactorial impacts of surgery. Surgical procedures leverage anatomical changes that initiate profound and sustained alterations to control the disease of obesity. While newer medications such as retatrutide, Cagrisema and survodutide approach weight loss outcomes previously only achieved through surgery, their efficacy depends on continuous adherence, and cessation results in the loss of treatment-mediated control of the disease of obesity with subsequent weight regain. This limitation contrasts with the durability of weight loss seen post-surgery. Moreover, long-term safety and efficacy data for pharmacotherapy is limited, particularly beyond five years for the newest medications, albeit the more than 20 years of safety data of the GLP-1 class of medication for treating type 2 diabetes is very reassuring. The complication rates associated with bariatric surgery are likely at a nadir currently; however, these rates may increase as a greater proportion of patients presenting with severe obesity and more severe obesity-related complications are referred for surgical intervention. Challenges for pharmacotherapy include high rates of mild to moderate side effects and patient discontinuation, but these may be addressed by slowing down titration schedules and moving obesity management into a chronic disease model. At present, surgery remains a very effective intervention for achieving lasting control of the disease of obesity with substantial weight reduction and improvements in the complications of obesity, but surgery remains difficult to scale. However, pharmacotherapy also offers a viable, scalable, less invasive alternative that can yield meaningful control of the disease of obesity, weight loss and metabolic benefits. The combination of surgery and pharmacotherapy does hold promise to not only address recurrent weight gain after surgery but also to achieve treatment targets in patients with more severe forms of obesity and obesity complications. Together, these treatments represent complementary strategies in the comprehensive management of obesity, allowing for more personalised approaches based on patient needs and treatment targets. With fewer than 10% of all people with obesity currently receiving either medication or surgery(Reference Coulman, Margelyte and Jones76), it is less productive to debate which treatment is superior; instead, efforts should prioritise expanding access to both effective obesity interventions to benefit a greater number of patients.
Acknowledgements
None.
Author contributions
Louise Ko drafted the original manuscript. Carel le Roux supervised the work, provided conceptual guidance, and, along with Ricardo Cohen, reviewed and edited the manuscript. All authors reviewed and approved the final version of the manuscript.
Financial support
None.
Competing interests
LK None.
RVC has received research grants from Johnson & Johnson MedTech, Medtronic, Marlex Brasil, and has received payment for lectures from Johnson & Johnson MedTech, Medtronic, Merck and NovoNordisk.
ClR reports grants from the EU Innovative Medicine Initiative, Irish Research Council, Science Foundation Ireland, Anabio, and the Health Research Board. He serves on advisory boards and speakers panels of Novo Nordisk, Roche, Herbalife, GI Dynamics, Eli Lilly, Johnson & Johnson, Gila, Irish Life Health, Boehringer Ingelheim, Currax, Zealand Pharma, Keyron, AstraZeneca, Arrowhead Pharma, Amgen, and Rhythm Pharma. ClR is the Chair of the Irish Society for Nutrition and Metabolism. ClR provides obesity clinical care in the My Best Weight clinic and Beyond BMI clinic and is a co-owner of these clinics.