Highlights
-
• There was no relationship between ALC and disease activity in the DMF-DMT interval.
-
• Lymphopenia alone should not deter initiation of an alternate DMT in RRMS patients.
-
• Timely commencement of an alternate DMT is associated with decreased risk of disease activity and should be started within 6 months of DMF cessation.
Introduction
Dimethyl fumarate (DMF), commercially known as Tecfidera, is an oral immunomodulatory agent that was approved for the treatment of relapsing-remitting multiple sclerosis (RRMS) in 2013. Reference Bomprezzi1 Though the exact mechanism of DMF’s disease-modifying activity in RRMS is under investigation, current literature suggests that it exerts its effects through nuclear factor erythroid-2-related factor (Nrf2)-dependent and Nrf2-independent intracellular signaling pathways, as well as by influencing the composition and phenotypes of various immune cell populations. Reference Mills, Ogrodnik, Plave and Mao-Draayer2,Reference Yadav, Soin, Ito and Dhib-Jalbut3 Notably, DMF has been shown to significantly decrease the populations of CD4+ and especially CD8+ T cell populations and, through various mechanisms, to promote an anti-inflammatory immune response. Reference Yadav, Soin, Ito and Dhib-Jalbut3,Reference Jordan, Yang, Fisher, Racke and Mao-Draayer4
While many of its immune-modulating actions contribute to the therapeutic benefit of DMF in RRMS, one significant side effect is lymphopenia. Reference Gold, Arnold and Bar-Or5,Reference Liang, Chai, Ng and Tremlett6 In a relatively recent meta-analysis investigating the safety of DMF in multiple sclerosis (MS), 19.4% of DMF-exposed patients developed grade I/II lymphopenia, and 4.1% experienced grade III/IV lymphopenia. Reference Liang, Chai, Ng and Tremlett6 Lymphopenia, especially when severe and prolonged, is a risk factor for the development of progressive multifocal leukoencephalopathy (PML), a potentially fatal demyelinating disorder of the central nervous system triggered by the John Cunningham virus. Reference Jordan, Yang, Fisher, Racke and Mao-Draayer4 Though the rate of PML in DMF-exposed MS patients is less than that of those undergoing treatment with fingolimod or natalizumab, Reference Sriwastava, Kataria and Srivastava7 to date, the estimated incidence of PML in DMF-treated MS patients is 0.02 per 1000. Reference Jordan, Yang, Fisher, Racke and Mao-Draayer4 Most MS patients who acquired PML had a variable degree of concomitant lymphopenia. Reference Jordan, Yang, Fisher, Racke and Mao-Draayer4 For this reason, it is recommended by Health Canada, 8 the US Food and Drug Administration 9 and the European Medicines Agency 10 that DMF be discontinued in patients whose absolute lymphocyte count (ALC) falls below 0.5 × 109/L for more than 6 months to minimize the risks of prolonged lymphopenia and PML. 8
Following DMF cessation, it is common to instigate a washout period prior to initiating an alternate disease-modifying therapy (DMT) to permit lymphocyte counts to recover and to reduce the risk of overlapping drug effects between sequential medications. During this interlude between treatments, healthcare providers in our multidisciplinary MS clinic observed MS disease activity, as shown by new brain and spinal cord MRI lesions and clinical relapses, in patients with lymphopenia, raising concern that low ALC may represent a risk factor. Indeed, several case reports have been published describing severe MS reactivation following the discontinuation of DMF in patients with persistent lymphopenia. Reference Harmel, Schlunk and Harms11,Reference Zecca, Antozzi and Torri Clerici12 Additionally, the phenomena of “rebound syndrome” and disease reactivation have been relatively well characterized both clinically and radiographically in subsets of patients following the discontinuation of fingolimod and natalizumab. Reference Barry, Erwin, Stevens and Tornatore13–Reference Prosperini, Kinkel, Miravalle, Iaffaldano and Fantaccini15 To our knowledge, however, similar larger-scale analyses have not been conducted in the context of DMF cessation.
The primary aims of this study were to assess clinical and radiologic disease activity in RRMS patients following DMF discontinuation and to investigate how these outcomes correlate with patient variables, including ALC. We also explored whether DMF-exposed patients experience a rebound in disease activity after stopping the medication. Additionally, we examined the optimal duration of time without DMT following DMF cessation, with the goal of balancing the risk of disease activity and the potential risks of immunosuppression.
Methods
Study design and variables
This is a retrospective chart review that included DMF-exposed patients with RRMS. All study procedures were approved by the Biomedical Research Ethics Board (ethics ID no.: Bio 14-139) at the University of Saskatchewan. The main outcome of the study was MS disease activity, either clinical or radiographic, in the DMF-DMT interval. We defined the DMF-DMT interval as the time in years between the cessation of DMF and the initiation of a new DMT to treat MS. MS disease activity was defined as either clinical relapse or an MRI with activity including new or enlarged T2/FLAIR lesions and/or gadolinium-enhancing lesions in the brain and spinal cord.
Data sources
Information regarding patient demographics, disease activity and treatment course was attained from a combination of Saskatchewan electronic health records, the MS Clinic electronic charts (Accuro) and database and the Intellispace PACS application.
Eligibility criteria
The study included patients of the Saskatoon MS Clinic over the age of 18 with either RRMS or secondary progressive multiple sclerosis (SPMS), prior to disease progression, who had been treated with DMF. Patients were excluded if they had not started an alternate DMT within 5 years of DMF cessation.
Statistical analysis
All analyses were performed using SAS version 9.4 (SAS Institute, North Carolina). Descriptive statistics were generated for demographic and clinical variables. Group comparisons were conducted using Mann–Whitney U tests for continuous variables and chi-squared tests for categorical variables. Time-to-event analyses were performed using Kaplan–Meier survival curves to describe the timing of disease activity during the DMF-DMT interval. Cumulative incidence was calculated at fixed time points (3, 6, 9 and 12 months), and interval-specific absolute risks were derived from life-table methods among patients who remained at risk at the start of each interval. Univariable Cox regression analysis was used to evaluate the association between baseline covariates and time to disease activity. Paired comparisons of ALC before and after treatment with Tecfidera were performed using Wilcoxon signed-rank tests. An alpha level of 0.05 was used throughout the study as a threshold for determining statistical significance. No corrections were made for multiple comparisons.
Results
Sample demographics
The study cohort consisted of 109 patients from the Saskatoon MS clinic with relapsing-remitting MS who were exposed to DMF. Two subgroups based on the presence or absence of MS disease activity were examined, and basic demographic data were analyzed. Across both groups, the average age of patients was 45.5 years. Of the 109 patients included in the study, 32.1% (n = 35) experienced disease activity. Most of the patients in the study population were female (n = 73, 67.0%). Between the groups that did and did not experience MS disease activity in the DMF-DMT interval, there were no significant differences in the number of female versus male patients, age or age at onset of MS symptoms (Table 1).
Basic group demographics for the entire patient cohort, as well as subgroups defined by the main outcome (disease activity in the form of relapses or MRI activity during DMF-DMT interval)

Note: MRI activity included new or enlarged T2/FLAIR lesions and/or gadolinium-enhancing lesions in the brain and spinal cord. For continuous variables, the mean (SD) was reported, and Mann–Whitney U test was conducted. For categorical variables, denoted with an asterisk*, N(%) and chi-squared test or Fisher’s exact test was conducted.
Significant results are bolded and set at p < 0.05.
ALC = absolute lymphocyte count; DMF = dimethyl fumarate; DMT = disease-modifying therapy; ARR = annualized relapse rate.
The time in years from MS diagnosis to start of treatment with DMF was 5.52 (SD = 5.7) in the group who experienced MS disease activity in the DMF-DMT interval and 5.91 (SD = 7.2) in the group who did not (p = 0.67) (Table 1). The time that patients in each group spent on DMF treatment was not significantly different (mean 1.93, p = 0.19). In contrast, the duration of the DMF-DMT interval was 0.25 years (SD = 0.5) in the group that did not experience MS disease activity during this interval, compared to 1.23 years (SD = 0.9) in the group that did (p < 0.0001). Of the 35 patients with either clinical or radiological disease activity in the DMF-DMT interval, 24 (68.6%) patients had clinical relapses, 21 (60.0%) had MRI activity and 10 (28.6%) patients had both.
Reasons for DMF discontinuation
The most common reason for patients stopping DMF was lack of efficacy (38.5%), followed by side effects/adverse events (29.4%) and lymphopenia (22.0%). Other reasons for discontinuation included family planning (5.5%), and in 4.6% of patients, the reason was undocumented.
Annualized relapse rates during the DMF-DMT interval
To determine whether there was a rebound phenomenon in the patients who experienced disease activity after stopping DMF, we compared annualized relapse rates (ARR) before DMF initiation from the date of their MS diagnosis to rates after discontinuation during the DMF-DMT interval. Though there is no consensus definition for what constitutes “rebound syndrome,” it is generally accepted to refer to recurrent disease activity after the withdrawal of a DMT that surpasses the patient’s baseline prior to starting the therapy. Reference Barry, Erwin, Stevens and Tornatore13,Reference Hatcher, Waubant, Nourbakhsh, Crabtree-Hartman and Graves14 The mean ARR for all patients (n = 109) from the date of MS diagnosis prior to starting DMF was 1.86 (SD = 4.9) and 0.46 (SD = 1.5) during the DMF-DMT interval. The difference between these values revealed a statistically significant reduction in ARR after DMF discontinuation (mean −1.40(5.2), p < 0.0001).
In patients with disease activity during the DMF-DMT interval (n = 35), the mean ARR per year from date of diagnosis up until starting Tecfidera was 0.92 (SD = 2.0). The mean ARR during the DMF-DMT interval was 1.42 (SD = 2.5). This corresponded to a p-value of 0.065, revealing no significant difference in clinical disease activity after DMF cessation compared to their pre-DMF baseline. Ultimately, the difference between pre-DMF and post-DMF disease activity in the overall study cohort and the subgroup of patients who did experience disease activity indicates that there was no rebound effect following DMF discontinuation.
Absolute lymphocyte count and MS disease activity
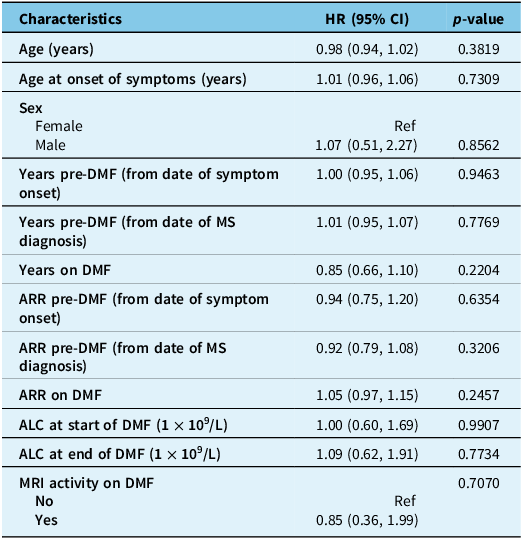
Next, we examined whether ALC at the time of DMF cessation portended a greater risk of MS disease activity. Among all 109 patients, ALC (cells x109/L) at the end of DMF treatment was significantly lower than at treatment initiation (Wilcoxon signed-rank test, mean −0.66(0.8), p < 0.0001). Of the 35 patients with clinical or radiographic disease activity in the DMF-DMT interval, 10 (28.6%) had lymphopenia (ALC < 1.0 × 109L) either at the time of DMF cessation or during the DMF-DMT interval. We compared the ALC in patients with disease activity in the DMF-DMT interval both before and after treatment with DMF. There was a significant reduction in ALC at the end of DMF therapy compared to prior to starting treatment (Wilcoxon signed-rank test, mean −0.56(0.9), p < 0.0014). A univariable Cox regression analysis did not show a statistical association between ALC at time of DMF discontinuation and disease activity in the DMF-DMT interval (HR 1.09 [0.62, 1.91], p = 0.77; Table 2). Therefore, though treatment with Tecfidera did result in a significant decrease in ALC for patients with disease activity in the DMF-DMT interval, this did not appear to be related to their risk of having clinical relapses or developing radiologic disease activity.
Univariable Cox regression analysis to examine factors associated with disease activity in the DMF-DMT interval (interval between DMF cessation and next disease-modifying therapy)

Note: None of the baseline covariates examined were significantly associated with risk of relapse or MRI activity during the DMF-DMT interval.
An alpha level of 0.05 was used to determine significance.
ALC = absolute lymphocyte count; CI = confidence interval; DMF = dimethyl fumarate; DMT = disease-modifying therapy; HR = hazard ratio; MS = multiple sclerosis; Ref = reference category.
Timing of disease activity after DMF cessation
A Kaplan–Meier survival analysis was performed to further characterize the risk and timing of disease activity following cessation of Tecfidera. Seventy-four patients (67.9%) were censored as they did not experience either clinical or radiographic disease activity during their individual DMF-DMT intervals. As can be seen in Figure 1, most censoring occurred within the first 6 months after DMF discontinuation, reflecting early initiation of an alternate DMT. Specifically, only 7 (9.5%) of the 74 censored patients remained off-treatment at 6 months and only 2 (2.7%) at 1 year. This corresponds with the data in Table 1, in which the mean duration of the DMF-DMT interval for those with no disease activity was only 0.25 years (SD = 0.5), which was significantly shorter than the group that did experience disease activity (mean 1.23 years, SD = 0.9; p < 0.0001). The Kaplan–Meier curve demonstrates that the probability of disease activity was highest within the first year after DMF cessation, as indicated by the steepest decline in this period (Figure 1). Of the patients who had clinical relapses in the DMF-DMT interval (n = 24), the median time interval to first relapse after stopping DMF was 0.5 years (IQR 0.13, 1.00), with a mean of 0.71 years (SD = 0.7).
Kaplan–Meier survival curve illustrating the time to first disease activity (clinical or radiologic) following the cessation of dimethyl fumarate (DMF). At baseline, 109 patients were at risk, out of which 35 patients experienced disease activity in the DMF-DMT interval (interval between DMF cessation and next disease-modifying therapy [DMT]). 74 patients were censored due to the fact that they did not experience disease activity prior to starting an alternate DMT for treatment of their relapsing-remitting multiple sclerosis. The slope of the curve illustrates that the probability of disease activity is highest shortly after stopping DMF (during the first 12 months) and decreases over time during the untreated period.

To further examine timing, cumulative incidence was calculated at fixed time points after stopping Tecfidera. The cumulative incidence of disease activity was 13.1% at 3 months, 25.0% at 6 months, 33.8% at 9 months and 52.2% at 12 months. Interval-specific absolute risks among patients still at risk of disease activity entering each period were 10.1% (0–3 months), 11.6% (3–6 months) and 11.5% (6–9 months), with increasing variability thereafter due to small sample sizes in later intervals. These analyses support the observation that a meaningful proportion of disease activity occurred within the first year after DMF cessation, though precision decreased beyond that as few patients remained off therapy.
Discussion
This study aimed to investigate RRMS disease activity in the post-DMF period as a means of determining the optimal time to reinitiate DMT following DMF discontinuation, as well as to elucidate patient factors that may increase the risk of relapse or MRI lesions. Among the baseline covariates included in our Cox regression analysis, none were significantly associated with disease activity after stopping DMF (Table 2).
As we were interested in the timing of disease activity after DMF discontinuation, a Kaplan–Meier survival curve was generated to illustrate the cumulative probability of relapse or MRI activity during the DMF-DMT interval. The Kaplan–Meier survival analysis (Figure 1) revealed that the probability of experiencing disease activity was highest shortly after stopping Tecfidera, with most clinical relapses or new MRI activity occurring early during the first year of the DMF-DMT interval. Half of the patients in our study who experienced relapses after stopping DMF did so within the first 6 months off therapy. Furthermore, the mean time to starting a new DMT in the group without disease activity was 0.25 years (3 months), significantly shorter than the 1.23 years (14.7 months) in the group with disease activity. Of the 74 patients with no disease activity during the DMF-DMT interval and who were censored from the Kaplan–Meier curve, 67 (90.5%) had already begun an alternate DMT by 6 months.
To further characterize the timing of events, cumulative incidence and interval-specific absolute risks were calculated. Cumulative incidence increased progressively over time, reaching 13.1% at 3 months, 25.0% at 6 months, 33.8% at 9 months and 52.2% at 12 months. Interval-specific absolute risks among patients who remained untreated and event-free at the start of each interval demonstrated that the short-term risk of disease activity was already appreciable with the first 3 months (10.1%) and remained similar across subsequent early intervals (11.6% at 3–6 months and 11.5% at 6–9 months). Interpretation beyond 9 months is limited by the small number of remaining patients untreated. These findings indicate that disease activity does not emerge abruptly at a single time point but accrues steadily early after DMF discontinuation. While causality cannot be inferred, the finding that half of those with relapses experienced them within 6 months, and that most of those without disease activity had already started a new DMT, suggests that earlier initiation of an alternate agent may help mitigate the risk of disease activity. Although the decision to reinitiate DMT for RRMS should be individualized, considering factors like disease burden, side effects and lifestyle, these data support the practice of initiating a new DMT within 6 months of DMF discontinuation to reduce the time off therapy and potentially lower the risk of disease activity.
Though the number of case reports describing a rebound effect after DMF discontinuation is relatively rare, we were interested in investigating whether this phenomenon may be observed in our cohort of patients. One such report by Zecca and colleagues (2018) described a young female patient with lymphopenia from DMF exposure who, despite persistent lymphopenia, presented with a severe spinal cord relapse approximately 6 months following the discontinuation of DMF. Reference Zecca, Antozzi and Torri Clerici12 Similarly, Harmel and colleagues (2018) reported a 38-year-old male with relatively mild RRMS who was clinically stable on DMF for 2 years before the decision to stop therapy was made due to recurrent upper respiratory tract infections and mild lymphopenia. Reference Harmel, Schlunk and Harms11 Within 2 months of DMF withdrawal, the patient developed progressive quadriparesis, and his MRI revealed approximately 14 new supratentorial lesions and extensive contrast-enhancing and expanding lesions throughout the cervical and thoracic spinal cord. Reference Harmel, Schlunk and Harms11 Though the pathophysiology for such severe rebound in each of these patients with concomitant lymphopenia is not known, we sought to determine whether similar occurrences were evident in our cohort, as this would have implications for the duration of a potential “washout” period post-DMF to allow for lymphocyte recovery. Ultimately, we neither observed clinical rebound nor a relationship between ALC and the timing of disease activity in the DMF-DMT interval. This supports the concept that lymphopenia alone should not deter the timely commencement of a different disease-modifying agent in patients with RRMS.
Limitations
One of the main limitations of our study is the sample size. Although we included 109 patients in the study, only 35 of them experienced the main outcome (relapse or new MRI activity), leading to high censoring in the survival analysis, which undermines the statistical power of the study and the strength of the conclusions that can be made.
We elected to proceed with a Kaplan–Meier survival analysis and Cox regression rather than a logistic regression analysis as the latter ignores the timing of disease activity during the DMF-DMT interval for each patient. A limitation of this method is the informative censoring of patients who initiated a new DMT before experiencing relapse or radiographic activity. These patients were censored at the time of new DMT initiation, potentially underestimating the true risk of post-DMF disease activity. To mitigate this potential bias, future analyses could consider expanding the sample size or conducting a sensitivity analysis using a fixed follow-up period to assess the impact of censoring on our results.
As our provincial healthcare system is not well equipped to support frequent imaging for routine disease-monitoring purposes, many of the patients lacked MRI data during the DMF-DMT interval, especially if they (i) had a relatively short interval and (ii) if they were clinically stable during that time. Thus, it is possible that the current data as it pertains to the number of patients with new radiological disease activity is an underestimation. Finally, another limitation is that we did not account for prior exposure to other DMTs before starting DMF. Though some were treatment-naïve, others had received other therapies prior to DMF, which could have impacted their pre-DMF relapse rates.
Strengths of the study and future directions
This study addresses an important gap in the literature by investigating the potential risk of disease reactivation following cessation of DMF. As DMF is widely used for treating RRMS, understanding the consequences of discontinuing it, particularly in relation to disease activity and lymphopenia, is clinically valuable. This research is relevant for informing clinical decisions and guiding future therapeutic strategies for persons with RRMS. The use of a Kaplan–Meier survival analysis to better understand the timing of disease activity after DMF cessation as it relates to the duration of the treatment interval offers important insights into the optimal timing for initiating a new DMT after DMF has been discontinued. Furthermore, we used a wide range of variables in our analysis, including a comprehensive comparison of ARR across different time periods. Finally, the retrospective chart review design, though accompanied by its own set of limitations, offers a real-world perspective on the clinical outcomes and challenges faced by patients after stopping DMF.
Although we identified patients with disease activity in the DMF-DMT interval, it is unclear whether this translated to worsening neurological deficits or a significant decline in functional outcomes. Future analyses may therefore be conducted to correlate disease activity with Expanded Disability Status Scale scores and other markers of patient function.
Conclusions
Lymphopenia is a relatively common consequence of treatment with immunosuppressive agents, including DMF, a pharmaceutical used for the treatment of RRMS. Isolated case reports have described DMF-exposed patients experiencing significant increases in disease activity despite ongoing lymphopenia after discontinuing the drug, and we were interested to see if this applied to our patient cohort. Cox regression showed no association between ALC at the time of DMF cessation and subsequent disease activity. In contrast, Kaplan–Meier analysis demonstrated that relapses and new MRI lesions were most likely to occur early after DMF discontinuation, with the risk of disease activity steadily accruing with longer duration off-treatment as demonstrated by interval-specific absolute risk and cumulative incidence estimates. Taken together, these findings suggest that lymphopenia in isolation due to DMF should not prevent or delay the commencement of other DMTs for RRMS. Earlier initiation of therapy within 6 months may be warranted to lower the risk of relapse, lesion burden and potential accrual of disability in RRMS patients.
Acknowledgments
Thank you to the Saskatoon Multiple Sclerosis Clinic and Office of the Saskatchewan MS Clinical Research Chair for the support of this research.
Author contributions
MCL and KZ developed the study design and managed data collection. CK and MCL participated in and oversaw clinical patient encounters, and CK managed initial data entry. MK performed the statistical analysis. KZ wrote the manuscript under MCL’s guidance. All authors contributed to preparing and editing the final manuscript.
Funding statement
The authors did not receive funding for this project.
Competing interests
For Michael C. Levin, MD, FRCPC, PhD:
-
• Grants funded by: MS Canada, Tri Agency New Frontier Research Fund (Canada), Saskatchewan Health Research Foundation, Innovation Saskatchewan, University of Saskatchewan College of Medicine
-
• Received honoraria or funding from Biogen, Novartis, Roche, EMD Serono and, as Editor of the Neurology Section, The Merck Manual.
-
• Provisional Patent US 63/544,434. HNRNP A1 nucleocytoplasmic mislocalization inhibitors and uses thereof.
-
• Support for attending meetings and/or travel from ACTRIMS 2024 meeting organizers.
-
• On the Medical Advisory Committee for MS Canada.
For Chantal Kahovec, NP:
-
• Has participated in advisory boards with Roche, Amgen, EMD Serono and Horizon Therapeutics.
-
• Received honoraria or funding from Biogen and Novartis.
-
• Support for attending meetings and/or travel from Roche and Amgen.
Kasia Zubkow and Min Young Kim have no disclosures.
Prior presentation(s)
-
• This work was presented in a poster presentation at the Consortium of Multiple Sclerosis Centres, 38th Annual Meeting. Nashville, TN. 2024.




Target article
Disease Activity Following Cessation of Dimethyl Fumarate in Patients with Relapsing-Remitting Multiple Sclerosis
Related commentaries (1)
Reviewer Comment on Zubkow et al. “Disease Activity Following Cessation of Dimethyl Fumarate in Patients with Relapsing-Remitting Multiple Sclerosis”