Introduction
Considering a lunar base construction, its running and effective performance, cultivation of plants in such a base is a key component because of (1) CO2 fixation resulting in biomass production to be utilised as food source for a base crew, (2) oxygen production and functioning as (3) biological life supporting systems (BLSS). The first short-term lunar test of BLSS was done just recently using the plants cultivated on lunar surface regolith (Xie et al., Reference Xie, Ding, Zhang, Ren, Qiu and Wang2024). The lunar experiment lasted 15 days and focused on germination rate and initial plant growth under intense radiation, low gravity and prolonged intense light. A substrate used in this study was vermiculite – a silicate mineral with chemical formula (Mg, Fe++, Al)3(Al, Si)4O10(OH)2.4(H2O) combined with a nutrient solution. However, for future lunar experiments, as well as plant cultivation in lunar base facilities, lunar regolith should be used in order to promote in-situ resource utilisation (ISRU) which make a base maintenance, lunar research and exploration and general crew activities less costly and more effective. Recently, an effort aimed to ISRU led to a comprehensive review of the ways to improve lunar regolith for successful plant cultivation (Ding et al., Reference Ding, Xu, Tan, Zhang, Xiong, Mei, Li and Xie2024).
To use lunar regolith as a substrate for plant cultivation during the first missions to the Moon has been suggested in the studies focusing on the way of living of future crews inhabiting a lunar base (Lytvynenko et al., Reference Lytvynenko, Zaetz, Voznyuk, Kovalchuk, Rogutskyy, Mytrokhyn, Lukashov, Estrella-Liopis, Borodinova, Mashkovska, Foing, Kordyum and Kozyrovska2006). Study of Paul et al. (Reference Paul, Elardo and Ferl2022) that used regolith samples from Apollo 11, 12 and 17 showed that Arabidopsis thaliana germinated and grew in these lunar regoliths. The study identified several problems, such as slow growth rate, malformations in plant morphology and molecular biology markers. Moreover, transcriptomic study was done as a part of the experiments in the regolith-germinating plants that identified differently-expressed genes associated with salt – and metal-induced stress resulting in reactive oxygen species (ROS) formation. Therefore, the study of Paul et al. (Reference Paul, Elardo and Ferl2022) stimulated follow up studies using lunar regoliths’ simulants. The studies focused on several aspects of biofertilisation of Lunar and/or Martian regolith (Duri et al., Reference Duri, Caporale, Rouphael, Vingiani, Palladino, De Pascale and Adamo2022) or their simulants (Zhao et al., Reference Zhou, Xu, Li, Yin, Xue, Li and Sun2024) with organic matter. Biofertilisation represents a prospective way to increase the agricultural potential of these substrates. Such systems utilising regolith and regolith simulants might be used in future space farming (Caporale et al., Reference Caporale, Palladino, De Pascale, Duri, Rouphael and Adamo2023). Moreover, the number of plant species tested in such growth systems has been increasing in the last decade (e.g., Eichler et al., Reference Eichler, Hadland, Pickett, Masaitis, Handy, Perez, Batcheldor, Wheeler and Palmer2020; Fackrell et al., Reference Fackrell, Schroeder, Thompson, Stockstill-Cahill and Hibbitts2020; Gonçalves et al., Reference Gonçalves, Wamelink, van der Putten and Evers2024; Rickard et al., Reference Rickard and Bode2021).
Lunar regolith simulant overviewed recently by Zhang et al. (Reference Zhang, Chao, Yao, Xu, Wuxiang, Ding, Liu, Zhao, Yinghe, Wang, Yu, Wang and Chen2021) is a prospective alternative for the studies focused on plant growth. The cultivation in lunar simulants, however, brings some specific problems related to the fact that lunar regoliths and/or their simulant differ in chemical composition and nutrients bioavailability from natural Earth-based soils. In general, regolith simulants are generally extremely poor in both nutrients and organic matter needed for successful plant growth (Seiferlin et al., Reference Seiferlin, Ehrenfreund, Garry, Gunderson, Hutter, Kargl, Maturilli and Merrison2009). Therefore, especially under such extreme conditions and in the absence of external nutrient inputs, organic amendment is particularly important. Addition of organic matter is one of strategies applied in lunar (see e.g., Caporale et al., Reference Caporale, Amato, Duri, Bochicchio, De Pascale, Simeone, Palladino, Pannico, Rao, Rouphael and Adamo2022) or Martian regolith simulant, as shown e.g., by (Zhao et al., Reference Zhao, Luo, Zhang, Yuan, Fang, Tong, Qian, Zhou, Yang, Zeng, Li, Xu, Xie, Gong and Guo2025 for Arabidopsis. thaliana). Recently, the role of bacteria on the improvement of lunar regolith (simulant properties) have been studied. For example, the effect of phosphorus-solubilizing Bacillus mucilaginosus, Bacillus megaterium and Pseudomonas fluorescens on an increase in phosphorus bioavailability for plant growth was reported by Xia et al. (Reference Xia, Yuan, Li and Sun2023). Such effect is possible due to by dissociating insoluble phosphorus from the lunar regolith. Apart from the improvement of lunar regolith by microbial environment modification, several other approaches have been tested involving e.g. addition of organic waste with earthworms (Wang et al., Reference Wang, Hou, Liao, Yao, Zhu, Liu and Feng2025), addition of antioxidants into the regolith (Barcenilla et al., Reference Barcenilla, Kundel, Hall, Hilty, Ulianich, Cook, Turley, Yerram, Min, Castillo-González and Shippen2024).
Recently, a great number of germination experiments have tested plant capability to germinate and perform satisfactory growth in lunar regolith simulants. Wamelink et al. (Reference Wamelink, Frissel, Krijnen, Verwoert and Goedhart2014) tested germination and early plant growth (50 d) of 14 different species in lunar and Mars soil simulants and found satisfactory germination rate. Although several biometrical parameters, as well as biochemical and molecular biology markers have been monitored during the early growth, less attention has been paid to the evaluation of photosynthetic processes in plants cultivated in lunar regolith and/or analogues.
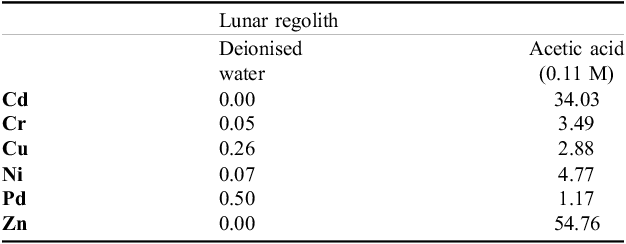
Heavy metals, like aluminium and chromium are also present in the extra-terrestrial regoliths. Aluminium is known to disturb plant growth (Kocjan et al., Reference Kocjan, Kwasniewska and Szurman-Zubrzycka2025) and even lead to cell/plant death caused by Al-induced oxidative stress (Chakraborty et al., Reference Chakraborty, Das, Pal, Roy, Sil, Adak and Hassanzamman2024). Previous study (Coufalík et al., Reference Coufalík, Stavrakakis and Argyrou2024) determined the mobile fraction of metals in lunar regolith simulant which was selected as model substrate for testing plant germination and early growth. Therefore, the aim of this study was to evaluate germination of Brassica oleracea var. botrytis italica in leachates form lunar regolith simulant. The leachates were extracts from the lunar regolith simulant (National Technical University of Athens) extracted either by acetic acid or deionized water, containing Cd, Cr, Zn, Ni, Pb and Cu (for details see Coufalík et al., Reference Coufalík, Stavrakakis and Argyrou2024). In our experiment, we tested hypothesis that water (aqueous) leachates would provide a better conditions for seed germination than acetic ones due to lower release of metal ions from the regolith particles. Since the release of the above-mentioned heavy metal ions is medium-dependent (different content of salts and acids), we used two contrasting options for dilutions, i.e. water (as a general reference) and acid (as most extreme).
In this study, we focused on a Lunar regolith simulant (Argyrou et al., Reference Argyrou, Stavrakakis and Chatzitheodoridis2024; Coufalík et al., Reference Coufalík, Stavrakakis and Argyrou2024; Stavrakakis et al., Reference Stavrakakis, Argyrou and Chatzitheodoridis2022) developed by the experts from the National Technical University of Athens (NTUA), Greece (see also Material and Methods) which showed high degree of similarity to lunar regolith and was, therefore, proposed as a substrate for plant cultivation in space analogue habitat greenhouses. The study aimed to germinate and grow broccoli (Brassica oleracea var. botrytis italica) in Murashige-Skoog (MS) liquid medium with addition of aqueous and/or acidic leachate. We hypothesised that experimental plant growth and primary photosynthetic processes will be negatively affected by the leachates. However, a degree of leachate dilution done before the addition to the MS medium would alleviate negative effects and stimulate plant growth and development in comparison to control (pure MS medium). Therefore, we decided to monitor several photosynthetic parameters, particularly those related to evaluation of photosystem II, during a germination study in order to point out crucial point in optimising dilution of leachates from lunar regolith simulants (in this study) and watering and/or nutrient solution composition to optimise growth in regoliths in general (a follow up study mimicking a lunar greenhouse set-up).
Material and methods
Lunar regoliths characteristics
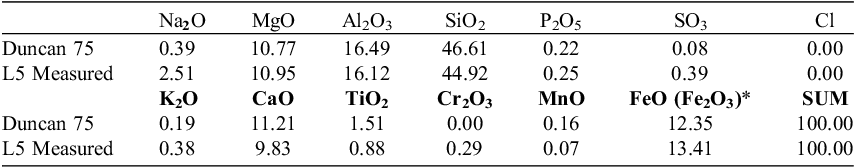
The lunar regolith simulant utilised in our study was developed at the National Technical University of Athens (NTUA), Greece, and is a part of the fifth batch (L5) produced by the team. The lunar regolith simulant was developed based on the Apollo 15 samples (Sample No. 15260 – Duncan et al., Reference Duncan, Sher, Abraham, Erlank, Willis and Ahrens1975). Multi mineral-rock approach during the simulant development process was used (Argyrou et al., Reference Argyrou, Stavrakakis and Chatzitheodoridis2024; Stavrakakis et al., Reference Stavrakakis, Argyrou and Chatzitheodoridis2022). The simulant evaluation has proven that it highly resembles the lunar surface (Coufalik et al., Reference Coufalík, Stavrakakis and Argyrou2024). The similarity was in mineralogy, grain size and chemical match, which was also evaluated based on the method of Schrader et al. (Reference Schrader, Rickman, McLemore, Fikes, Stoeser, Wentworth and McKay2009). The method showed a 95.83 chemical similarity (Figure of Merir, FOM) to the lunar regolith. Detailed chemistry of the simulant and its correlation with the Lunar surface data of the Sample No 15260 can be found in Table 1.
Chemical composition of L5 (Lunar regolith simulant) compared to the Apollo 15260 sample analysis (Duncan et al., Reference Duncan, Sher, Abraham, Erlank, Willis and Ahrens1975) adopted from Coufalík et al. (Reference Coufalík, Stavrakakis and Argyrou2024)

Note: *All iron measured as FeO.
Lunar regolith simulant leachate
Leachates were prepared form the lunar regolith analogue using deionised water and 0.11 M acetic acid. The extraction of a leachate was done on a shaker (GFL 3018, Germany) in 0.11 mol/L CH3COOH (Suprapur, 96%, Merck, Germany) in a ratio of 0.5 g of sample per 20 mL of solution at laboratory temperature for 16 h. Extraction in deionised water was carried out under the same conditions. Then, the extracts were centrifuged (Centrifuge 5804R, Eppendorf AG, Germany) at 9000 g for 10 min, and collected into HDPE scintillation vials (Kartell, Italy).
Plant cultivation
Individuals of broccoli (Brassica oleracea var. botrytis italica) were cultivated from seed in a Murashige-Skoog (MS) liquid medium (for medium composition, see Duchefa Biochemie, M0231.0050). The seeds were put into individual holes of microbiological plate, supplied with particularly diluted leachates (see Table 3) combined with a powdered MS medium (for medium specification, see Table 6). The seeds were provided equal volume of medium (1.0 ml per hole) at the beginning of germination. Control treatment was represented by exclusive MS medium. During germination and the early growth period, the microbiological plates were covered by a 12.6× 8.2 × 11.2 cm (l × w × h) transparent minibox and sealed by a Teflon (PTFE) lab tape around the margins, to prevent evaporation of the medium. Then, cultivation was carried out in a growth chamber (T = 21oC, PAR = 120 µmol m−2 s−1) for 14 d. During cultivation, germinating plants took medium in a slow rate reducing its volume in a hole. Therefore, due to uptake of medium and variety of directions of initial root growth including the root growth above water/medium upper surface, the germinating roots did not grow under permanent immersion. However, the medium was refilled regularly (typically once a week) to maintain volume of 1.0 ml to provide sufficient amount for successful plant germination and growth.
Plant growth and primary photosynthesis
The effects of acidic and water leachate on B. oleracea growth and primary photosynthetic performance were monitored during 2 weeks after germination. Functioning of photosystem II of the emerging plants was evaluated by chlorophyll fluorescence imaging system (Open FluorCam fluorometer, Photon Systems Instruments, Drásov, Czech Republic) by slow Kautsky kinetics supplemented with quenching analysis using the method described earlier (Barták et al., Reference Barták, Hájek, Orekhova, Villagra, Marín, Palfner and Casanova-Katny2021). Before experiments, B. oleracea plants were dark-adapted for 15 min (the time was tested before the experiment and found sufficiently long to open photosynthetic PSII centres). A single measurement started with an exposition of the experimental plants to low light for 30 s in order to induce the basal chlorophyll fluorescence F0, which is emitted from chlorophyll molecules located in light harvesting complexes. In this state, reaction centres of PSII (RC PSII) remained open. Then, a 2s-lasting saturation pulse was applied to close the RCs PSII and, therefore, induce the maximum chlorophyll fluorescence level (FM). After the saturation pulse, a 30 s dark period followed to allow dark relaxation of chlorophyll fluorescence signal. After this period, the plants were exposed to a 300s-lasting actinic light (AL) period. During the light period, polyphasic rise and decline of chlorophyll fluorescence (slow Kautsky kinetics) was monitored. First, a rapid increase to a peak value (F P) was achieved followed by a slow polyphasic decline in chlorophyll fluorescence to a steady-state chlorophyll fluorescence (F S). The FS level was measured at the end of the AL period. After another saturation pulse applied at the end of AL period, maximum chlorophyll fluorescence in the light-adapted state (F M′) was evaluated). Whenever the AL was switched off, the background chlorophyll fluorescence (F′0) was recorded. A final saturation pulse was given then to induce FM″ level after a 40 sec dark period. From the above specified chlorophyll fluorescence data, the chlorophyll fluorescence parameters (for definitions see Roháček, Reference Roháček2002) were finally calculated using the formulae specified in Table 4.
Results
Germination and growth rate
Water leachates when added to a MS medium in the two experimental dilutions (W10D, and W100D treatments) did not cause full inhibition of seed germination as recorded for some acidic leachate treatments = Figure 1 (see below). The early growth of B. oleracea was found dilution-dependent, lower growth rate was recorded for W100D than W10D treatment the rate of growth, however, was found lower for the W10D and W100D treatments than for a MS medium.
Germination of B. oleracea plants in water leachates recorded at the beginning (a), middle (b), and the end (c) of cultivation period. Left column: heterogeneity of chlorophyll fluorescence in germinating plants, the images show F V /F M values (pixel colour from low to high: blue-green-yellow-orange-red). Right: photographs of respective germination plants. Treatments: W10D, W100D. Particular treatments are divided by a blue line indicated in the middle of a microbiological plate.

Acidic leachate prepared from the lunar regolith simulant caused 100% suppression of B. oleracea germination (Figure 2) due to toxic elements effects that limited germination (for the underlying physiological mechanisms, see Discussion). Diluted leachates added to the MS growth medium showed concentration-dependent response. While the suppression of germination was still apparent in the little diluted leachates (i.e. A10D, A25D treatments), the greater dilution treatments (A50D, A75D and A100D) led to seed germination. The early growth showed fastest rate and most pronounced development of cotlyedons area in control (i.e. plants germinated exclusively in a MS medium – see pixel area growth in Figure 3). Germination and growth in the acidic leachate treatments showed an increase in time. However, the response was dilution-dependent. Higher dilution of acidic leachates led to a decrease in growth. The final pixel area ‘occupied’ by cotyledons, hypo- and epicotyl, recorded on the last day of experiment (day 18), decreased with the dilution as follows (related to control = 100%): A50D (96.6%), A75D (55.5%) and A100D (28.2%).
Germination of B. oleracea plants in acidic leachates recorded at the beginning (a), middle (b) and the end (c) of cultivation period. Left column: heterogeneity of chlorophyll fluorescence in germinating plants, the images show F V /F M values (pixel colour from low to high: blue-green-yellow-orange-red). Right: Photo of respective germination plants. The A50D and A75 treatments (addition of acidic leachates into MS medium) are presented.

Plant growth of B olearcea as affected by water leachates W10D and W100D (open bars) and in unaffected control (exclusive Murashige-Skoog medium, data points) in the cultivation period. Plant growth is expressed as number of pixels fitting into the area of cotyledons, epicotyl and hypocotyl visualised by chlorophyll fluorescence imaging. Means are presented ± standard deviations.

Primary photosynthetic processes
The effects of water and acidic leachates on the physiological processes in Brassica oleracea var. botrytis italica related to primary photosynthesis were monitored by several chlorophyll fluorescence parameters (F V/F M, ФPSII, qP and Rfd – for definition and meaning, see Table 4). Since all of them are associated with photosystem II and considered indicators of PSII functionality, the cultivation time dependent decrease in their values (Figures 5 and 6) is associated with gradual limitation of primary photosynthetic processes in PSII.
Capacity of photosynthetic processes in PSII (F V/F M) declined with the time of cultivation in W10D, and W100D treatments (Figure 5), however, the decline was similar to F V/F M time course in control plants. Moreover, no negative effect of the water dilutions on F V/F M were apparent, in spite of the fact that the final FV/FM value (day 15 in Figure 5a) were slightly higher (statistically unsignificant) for W10D than W100D. There was no effect of acidic leachates on FV/FM since the time courses of FV/FM recovered for A50D, A75D and A100D treatments were comparable to untreated control (MS solution) – see Figure 6a. Similarly, to water leachate, A100D treatment led to a slight decrease in FV/FM value (compared to control) at the end of cultivation time.
Effective quantum yield of photochemical photosynthetic processes in PSII (ФPSII) is considered an indicator of photosynthetic linear electron transport in between PSII and PSI as well as overall photosynthesis expressing an equilibrium between primary photochemical and secondary biochemical photosynthetic processes. The response of effective quantum yield (ΦPSII) to water and acidic leachates addition was similar to FV/FM. There were no statistically significant differences between the treatments and control. Time course of ΦPSII within the experimental cultivation time showed gradual decrease to about 60% of initial value. However, great dilution (W100D, and A100D) led to the lowest ΦPSII values found at the end of experimental cultivation.
No statistically significant decrease in photochemical quenching of chlorophyll fluorescence (qP) was found between untreated control and the treatments exploiting addition of water leachate (W10D, W100D). The same was true for the treatments with acidic leachates. There was no difference in qP when compared to control or even within the group of A50D, A75D and A100D treatments. The qP values, irrespective of the treatments showed either no change or declined to only a very limited extent with time of cultivation. Thus, the fraction of open PSII reaction centres remained more or less unchanged by the treatments exploiting the W10D, W100D, A50D, A75D and A100D treatments.
Vitality of germinating plants expressed as relative fluorescence decrease ratio (Rfd) showed a bi-phasic response (Figure 6d). Both in water and acidic leachates added into the MS medium, Rfd showed an increase apparent within the first few days of cultivation (day 2 to 6) followed by an approximately constant-rate decrease until the end of cultivation period, when minimum Rfd values were recorded for control (1.1) and water (0.25–0.3) and acidic leachates (0.8–0.1).
Discussion
Acidic leachates had more negative effects on B. oleracea germination than water leachates. Acidic leaching of lunar regolith simulant obviously led to an increased content of the metal ions (compared to aqueous leaching) – see Table 2. The metal ions are present in higher amounts in acidic than water extracts (by the factor of 100–100). Therefore, no germination was found in the A10D, and A25D treatments might be attributed to too high concentration of metal ions in medium that caused full limitation of germination. Specifically for broccoli, concentration higher then 1mM of Cd and Ni in growth medium is reported to cause full inhibition of germination (di Salvatore, Reference Di Salvatore, Carafa and Carratù2008). Even in the plants that showed successful germination (A50D, A75D and A100D), if cultivated for a longer time (in terms of weeks) under the experimental conditions used in this study, may face negative effects on photosynthesis, vigour and growth rate. The reason is that excessive concentrations of heavy metals inhibit various biochemical pathways and cause activation of antioxidative response (both enzymatic, and non-enzymatic antioxidants, for broccoli see Dawood and Azooz, Reference Dawood and Azooz2019). Finally, heavy metal ions released from regolith and taken up by experimental plants may be the cause of heavy metabolic imbalance, consequent biomass reduction and plant death (Zornoza, Reference Zornoza, Vazquez, Esteban, Fernández-Pascual and Ca2002). In plants that have been tested in lunar regolith so far, significant interspecific differences are reported for germination rate (e.g. Rickard and Bode, Reference Rickard and Bode2021).
Extractable fractions (%) of metals in deionised water and 0.11 mol/L acetic acid in the leachates from lunar regolith simulant prepared by the National Technical University of Athens, Greece

Abbreviations and explanations of the treatments used in the germination experiment of B. oleracea. MS – Murashige-Skoog medium

Chlorophyll fluorescence parameters used in this study. For more details see Roháček (Reference Roháček2002)

Limitation of germination by heavy metal ions is a well-described phenomenon. Critical concentration of heavy metals that fully inhibits germination is species specific, however, the concentrations of Pb and Cu higher than 400 mg L–1 are reported to prevent seed germination (Vasilachi-Mitoseru et al., Reference Vasilachi-Mitoseru, Stoleru and Gavrilescu2023). In this study, suppression of seed germination in the A10D and A25D treatments might be attributed to heavy metal ions effect, especially Zn, Ni and Cr which were present in the leachate in high concentration. Substantial inhibition on seed gemination (below 25%) is reported for Zn when the concentration of 100 pp or higher is used (Bezini et al., Reference Bezini, Abdelguerfi, Nedjimi, Touati, Benziane and Yabrir2019). Similarly, Astuti et al. (Reference Astuti and Nugroho2022) reported almost zero germination caused by Cr (100 mg L−1). However, different species show different levels of tolerance to Cr2+. Germination experiment by Jun et al. (Reference Jun, Ling and Guanghua2009) showed that Lablab purpureus might be ranked among most sensitive species to Cr2+, while other tested species (Lathyrus ordoratus, Dumasia villosa) showed no reduction of germination.
Plant growth (see Figure 4) was much more vigorous in control than in the treatments exploiting water and acidic leachates from the lunar regolith simulant. The growth, however, declined with higher degree of the leachate dilution, both in water and acidic leachate. Reason for such reduced growth might be found in bio effectivity of particular concentrations of heavy metal ions in relation to macronutrients (N, P in particular), their uptake and effective utilisation in the emerging plants. This aspect will be addressed in follow up study of germinating plants cultivated in regolith and supplied either by demineralised water or Murashige-Skoog medium (unpublished data, Barták et al., MS in prep).
Plant growth of B. olearcea as affected by acidic leachates A50D, A75D and A100D in the cultivation period. The treatments affected by A10D, and A25D dilutions did not germinate. Plant growth is expressed as number of pixels fitting into the area of cotyledons, epicotyl and hypocotyl visualised by chlorophyll fluorescence imaging. Means are presented ± standard deviations.

Rate of plant germination and the early phases of shoot and root growth might be affected by liquid culture itself. In this concept, liquid culture may potentially affect plant seed germination. Root physiology, respiration in particular, might be limited by insufficient oxygen supply in fully submerged roots. It is well established that oxygen is a key factor controlling seed germination (Corbineau, Reference Corbineau2022). Prolonged root submersion may lead to asphyxia and disruption of root function and, finally, result in hypoxia and plant death. During a prolonged hypoxia (typically 3 weeks of fully submerged non-aerated roots), enzymatic activity is altered in order to stimulate alternative N assimilation pathway (Horchani and Aschi-Smiti, Reference Horchani and Aschi-Smiti2010). Recent studies focus on different aspects of species-specific tolerance to prolonged hypoxia (Fukao et al., Reference Fukao, Kennedy, Yamasue and Rumpho2003, Kretzschmar et al. Reference Kretzschmar, Pelayo, Trijatmiko, Gabunada, Alam, Jimenez, Mendioro, Slametloedin, Sreenivasulu and Baileyserres2015) mainly in relation to waterlogging (for review see Zhou et al. Reference Zhou, Chen, Meng, Chandrasekaran, Luo, Yang and Shu2020). In our experiment, however, root anoxia during the cultivation time was unlikely and physiological limitations caused by insufficient oxygen supply to roots might be excluded since the roots were not fully submerged during the cultivation period (see Material and Methods).
Germination of seeds in liquid in the holes of microbiological plates entails the aspect of missing support for the root and vertical shoot growth. It is very likely that, if provided some support by e.g. 1.0–3.0 mm sized glass/ceramic balls, the germinating B. oleracea would show more vigorous early growth and leaf area development. It is important to say that our experiments with plants cultivated in fine-grained regolith provided with water or MS proved vigorous growth (Ul Haq et al., Reference Ul Haq, Hájek and Barták2026). Another limitation of our germination system could be the fact that some leaves remained located on the liquid surface before starting to incline towards the vertical. This might be considered the cause of the relatively small growth inhibition since the leaves showed inability to extend the shoot vertically for a couple of days, however, later, they tended to grow normally (cf. Figure 1a,b).
Apart from the growth of germinating B. oleracea, time courses of chlorophyll fluorescence parameters showed dilution-dependent decrease in PSII functioning (cf. FV/FM and ΦPSII values for control and W100D, A100D in Figures 5a,b and 6a,b). Reason for such decrease is unclear, however, it may be caused by a complex interaction between light harvesting complexes and reaction centres of PSII, light energy absorption and transfer to photosystem II in particular and activity of oxygen-evolving complex (OEC) attached to the lumen side of the PSII complex, when affected by heavy metal ions. To point out a single reason for heavy metal-induced decline on chlorophyll fluorescence parameters related to PSII is difficult even for specialised biophysical techniques evaluating heavy metals-induced reactive oxygen species and their effect on energy transformation in PSII. General negative impact of heavy metals in PSII is associated with concentration-dependent inhibition of PSII effectivity. Typically, heavy metal effects in PSII lead to a decrease in FV/FM and ΦPSII values (Jocsák et al., Reference Jócsák, Csima and Somfalvi-Tóth2024 for Cd and Zn, Djukic et al., Reference Djukić, Djunisijević-Bojović, Pavlović, Mitrović, Grbić, Skočajić and Lukić2014 for Pb, Gong et al., Reference Gong, Wang, Dai, Zhou, Kang, Chen, Li and Li2019 for Cu, Chen et al., Reference Chen, Mao, Ma, Wu, Zhang, Su, Zhang, Yuan, Zhang, Zeng and Yuan2018 for Cr), similarly to other stressors, such as high light (Ul Haq et al., Reference Ul Haq, Benita and de Caralt2024), salt stress (Akhter et al., Reference Akhter, Noreen, Mahmood, Athar, Ashraf, Alsahli and Ahmad2021) or the two stressors in combination (Yan et al., Reference Yan, Bian, He, Han, Lv, Guo and Lu2018). However, the early fluxes of absorbed light energy through PSII studied by fast chlorophyll fluorescence transient (OJIPs and OJIP-derived parameters) respond sensitively to heavy metals in chloroplast (thylakoid membrane) of model algae (e.g., Perrault et al., Reference Perreault, Dionne, Didur, Juneau and Popovic2011) and vascular plants (Zhou et al., Reference Zhou, Xu, Li, Yin, Xue, Li and Sun2024).
Chlorophyll fluorescence parameters (F V /F M – A, Φ PSII – B, qP – C and Rfd – D, for definitions, see Table 4 ) measured in B. oleracea growing in a MS medium with addition of water leachate (W10, W100, i.e, 10 times, and 100 times diluted).

Chlorophyll fluorescence parameters (F V /F M – A, Φ PSII – B, qP – C and Rfd – D, for definitions, see Table 4 ) measured in B. oleracea growing in a MS medium with addition of acidic leachate from a lunar regolith simulant (A50, A75, A100, i.e. 50, 75 and 100 times diluted).

Gradual decrease in chlorophyll fluorescence parameters related to functioning of PSII in chloroplasts is clearly comparable to experimental evidence gained for B. oleracea in similarly-designed germination experiment that used Antarctic regolith from James Ross Island (Krejčí et al., Reference Krejčí, Stavrakakis, Barták, Sekerák and Argyrou2024). Gradual decline in PSII functioning, specifically ΦPSII might be attributed both to cultivation conditions (ΦPSII declined with time of cultivation even in control plants), and negative effects of heavy metal ions released from a lunar regolith simulant, taken up by emerging plants and allocated in chloroplasts. High content of heavy metal ions in chloroplasts and consequent reactive oxygen species formation are reported for plants grown in lunar (Barcellina et al., Reference Barcenilla, Kundel, Hall, Hilty, Ulianich, Cook, Turley, Yerram, Min, Castillo-González and Shippen2024) and Martian (Chinnannan, Reference Chinnannan, Somagattu, Yammanuru, Nimmakayala, Chakrabarti and Reddy2023) regoliths or their simulants among the reasons of decreased chlorophyll contents in leaves. Oxidative stress, and activation of antioxidative mechanisms, might be considered one of the most important physiological response of photosynthetic tissues in plants cultivated in lunar regolith and its analogues. It was shown by Paul et al. (Reference Paul, Elardo and Ferl2022) that the plants grown in lunar regolith do activate a series of stress response pathways, most notably the oxidative stress response. Moreover, heavy metals-induced alterations in nutrient uptake rate from MS cultivation medium might be considered another co-acting factors. In acetic leachate, Zn represented a major element. Therefore, Zn-induced interactions with the PSII photosynthetic apparatus might be taken into consideration because heavy metals-induced inhibition of PSII functioning is reported for a variety of plants (Qin et al., Reference Qin, Jiang, Qin, Zhao, Dai, Liu and Chen2023).
Concluding remarks
This study revealed that acidic leachates from lunar regolith simulant, when added to Murashige-Skoog cultivation medium, caused a more pronounced inhibition of Brassica oleracea germination compared to water leachates leading to complete suppression of seed germination. Even when diluted 10– or 25-fold, the acidic leachate continued to inhibit germination. In contrast, undiluted leachates from the simulant, both acidic and water, entirely suppressed the seed germination. On the other hand, water leachates did not show such detrimental effects. B. oleracea exhibited satisfactory germination when treated with a 10-fold diluted water leachate. This suggests that water leachates from lunar regolith or its simulants could be viable for future studies aimed at cultivating plant species in such media. In support of this, a recent study by our team (MS in prep.) on the growth of B. oleracea, Raphanus sativus and Capsicum annuum in Antarctic regolith without added organic matter (for regolith characteristics, see Coufalík et al. Reference Coufalík, Stavrakakis and Argyrou2024) showed successful plant growth and adequate photosynthetic performance, even when irrigated solely with demineralised water. However, the aboveground growth and development were less vigorous compared to plants watered with Murashige-Skoog medium.
Acknowledgements
The authors would like to express their thanks for the VAN2024 and VAN2025 (Czech Antarctic Research Infrastructure, the projects provided by the Czech Ministry of Education, Youth and Sports) for support (MB, JH) and the facilities of the Extreme Environment Life Laboratory of the Masaryk University, Brno. The support from junior team members (ŠK, JS) provided by the MUNI/A/1492/2023 project is also acknowledged. The work of PC was supported by the Czech Science Foundation under grant No. 24-10051S. Plant Sciences Core Facility of CEITEC Masaryk University is acknowledged for the technical support.