Prenatal brain development is primarily guided by genetic mechanisms influenced by the mother’s biochemical environment, such as nutritional status and exposure to toxins during pregnancy(Reference Tierney and Nelson1). Though this dynamic process continues throughout a person’s life, the fetal brain’s heightened responsiveness to new experiences makes the prenatal period a time of great opportunity, but also vulnerability(Reference O’Sullivan and Monk2).
Early motor development may be a sensitive outcome for prenatal exposures and also a predictor of later cognitive functioning in children(Reference Murray, Jones and Kuh3,Reference Ghassabian, Sundaram and Bell4) . The Test of Infant Motor Performance (TIMP) is an assessment tool of early motor development that may be performed to identify motor delays in infants(Reference Campbell5–Reference Peyton, Schreiber and Msall7). In an overall evaluation of early motor assessment tools, the measurement qualities of the TIMP have been considered high, particularly in the youngest infants, with excellent predictive ability for future motor development(Reference Spittle, Doyle and Boyd8). We have previously found the tool to be reliable and feasible in a Nepalese context(Reference Kvestad, Silpakar and Hysing9).
Se is an important micronutrient for brain development and neurological function(Reference Behl, Mehta and Pandey10,Reference Rayman, Hatfield, Berry and Gladyshev11) . The demand for Se is increased in pregnant women, leading to a greater risk of Se deficiency(Reference Ambroziak, Hybsier and Shahnazaryan12). The transport of Se within the body is retained by Selenoprotein-P in the blood, which plays a dynamic role in the brain, specifically in the cerebral and cerebellar cortices(Reference Behl, Mehta and Pandey10,Reference Zhang, Zhou and Schweizer13) . Both are closely related to overall motor functions as the length of the cerebellum of infants has shown associations with maternal Se concentration(Reference Močenić, Kolić and Nišević14), and the cerebellar cortex is mainly responsible for overall motor development(Reference Manto, Bower and Conforto15). Vitamin B12 may be relevant in the context of Se, as deficiency can disrupt one-carbon metabolism, and potential interactions between Se and vitamin B12 in pathways related to oxidative stress and neurodevelopment have been suggested, although evidence remains limited(Reference Chen, Hedstrom and Whanger16–Reference Shahidin Wang and Wu19).
Previous studies have shown associations between maternal prenatal Se concentration and child neurodevelopment across a wide range of domains, including motor development(Reference Močenić, Kolić and Nišević14,Reference Skröder, Kippler and Tofail20–Reference Kobayashi, Itoh and Miyashita23) . Studies have demonstrated positive associations between maternal Se concentrations and motor development in offspring during infancy and early childhood(Reference Varsi, Bolann and Torsvik22–Reference Guo, Xu and Tian25). Contrasting with these studies, a study from Croatia showed no associations between prenatal maternal Se concentration and motor development using the Bayley Scales of Infant and Toddler Development, third edition(Reference Močenić, Kolić and Nišević14). To date, most studies examining these associations have focused on infants older than 6 months of age(Reference Močenić, Kolić and Nišević14,Reference Skröder, Hamadani and Tofail21–Reference Polanska, Krol and Sobala24) . One exception is a study from China showing an inverted U-shaped relationship between umbilical cord blood Se concentration and neonatal neurobehavioral development assessed when the infants were 3 d old by the Neonatal Behavioural Neurological Assessment(Reference Yang, Yu and Fu26). In this study, there was a negative association with higher Se concentration.
In a large mother–child cohort from Nepal, we found 36 % of the pregnant women were Se-deficient, but there was no association between Se concentration and neurodevelopment including motor development measured when the children were 6–24 months by the Bayley Scales of Infant and Toddler Development-third edition(Reference Ranjitkar, Kvestad and Chandyo27). These pregnant women were originally included in a randomised controlled trial of the effect of vitamin B12 on neurodevelopment, where we detected no effect of B12 supplementation during pregnancy on the neurodevelopment at 6 and 12 months. There was, however, a negative effect of the intervention on early motor development as measured by TIMP when the children were 8–12 weeks(Reference Chandyo, Kvestad and Ulak28). These findings are interesting in the context of the association between maternal Se concentration and neurodevelopment, since there are previous studies that have described interaction effects of vitamin B12 in the association between maternal Se and neurodevelopment(Reference Lee, Ji and Raghavan29). Sex has also been shown to have interaction effects in the association between maternal Se concentrations and motor development(Reference Skröder, Hamadani and Tofail21). In a study from Bangladesh, higher Se concentrations were associated with improved psychomotor development in girls, but not in boys as measured by the Bayley Scales of Infant and Toddler Development-second edition when the children were 18 months old. Both maternal age and socioeconomic status may also potentially confound the association between maternal Se concentrations and infant neurodevelopment(Reference Phiri, Ander and Bailey30–Reference Rodrigues, Joshi and Rajadhyaksha36).
The main objective of the present study is to describe the association between maternal Se concentration measured during early pregnancy and infant motor development measured by the TIMP at 8–12 weeks. In addition, we seek to assess the potential moderation by sex of the child and vitamin B12 supplementation.
Method
Study design and setting
The included mothers and infants were originally participants in a double-blinded randomised controlled trial entitled ‘Supplementation of vitamin B12 in pregnancy and postpartum on growth and neurodevelopment in early childhood: A Randomized, Placebo Controlled Trial’(Reference Chandyo, Ulak and Kvestad37). The study was conducted in Bhaktapur, located 15 km from Kathmandu, the capital city of Nepal. According to the census in 2021, Bhaktapur district has a population of 432 132 with an annual growth rate of 3·32 %(Reference Mishra and Sijapati38). People from this area are primarily engaged in agriculture, small-scale business, service or daily wage labour work.
Pregnant women aged 20–40 years, within 15 weeks of gestation and planning to stay in the study area for the next 2 years were enrolled. Gestation weeks were calculated based on the last menstruation period and re-confirmed by ultrasonography at enrolment. Pregnant women who were taking or planning to take multivitamins containing vitamin B12, with severe illnesses, high-risk pregnancies or a BMI outside the range of 18·5–30·0 kg/m2 were excluded from the study. The 800 pregnant women were randomly assigned to daily oral vitamin B12 (50 μg) or placebo supplementation from early pregnancy until 6 months postpartum. As per national guidelines, they were also supplemented daily with 400 µg of folic acid during the first trimester and from the second trimester to 45 d postpartum, with 500–1000 mg of calcium and 60 mg of iron.
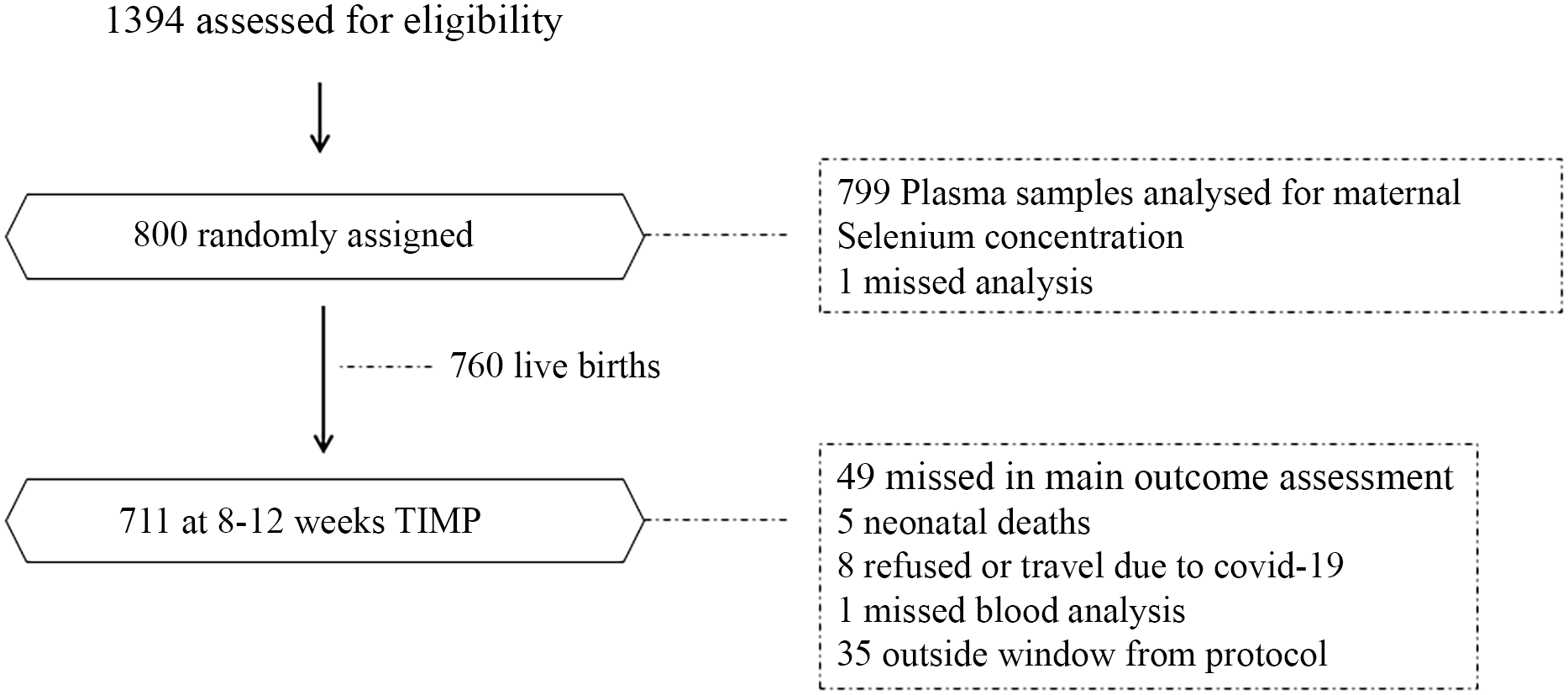
Pregnant women who met the eligibility criteria were enrolled from antenatal checkups from March 2017 to October 2020 and were followed every week until 6 months postpartum. When the infant was between 8–12 weeks of age, mother and child were requested to visit the study clinic for an assessment of motor performance. For the present study, we included mother–infant pairs for whom data were available on maternal Se concentration and TIMP scores within the specified assessment window period. Among 760 live births, five infants died, eight were not available due to refusal or travel during COVID-19 pandemic. A total of 740 infants were assessed using the TIMP, of which thirty-five were outside of the window period and were excluded, leaving 712 infants with valid TIMP assessments for the analysis. Among these, one participant lacked data on maternal Se concentration, resulting in a final sample size of 711. Details of the study flow are shown in Figure 1.

Figure 1. Study flow diagram for major activities from enrolment to 8–12 weeks in Nepalese mothers and children.
Demographic and clinical information
At enrolment, demographic and clinical information were collected. Socioeconomic status was expressed using the Water and sanitation, Asset ownership, Maternal education, and household Income index, with scores ranging from 0–1(Reference Psaki, Seidman and Miller39). The index was calculated based on baseline information provided by the participants on Water and sanitation, Asset ownership, Maternal education, and household Income(Reference Psaki, Seidman and Miller39). Caste/ethnicity was recorded as Newar, Brahmin/Chhetri, Indigenous groups (Gurung/Rai/Magar, Tamang/Lama) and others (Chaudhari/Madhesi/Muslim, Dalits and others). Years of parental schooling were dichotomised as completed secondary school level (> 10 grade) and above or not. Parental occupation was dichotomised as work (engaged in agriculture, business, carpet factory, daily wage earning, government employment, services in the private sector and foreign employment) or no formal work. Maternal weight and height were measured at enrolment following the standard procedure with standard scales by trained field workers at a clinic using a portable electronic scale (Seca) and a stadiometer (Prestige, Hardik Medi Tech), respectively. BMI of mothers was calculated as body weight in kilograms and height in metre squared (kg/m2).
After delivery, information on gestational age and birth weight was obtained from hospital records. Those who were born < 37 weeks of the gestational period were defined as preterm, and birth weight < 2500 gm was considered low birth weight.
Lab procedure and plasma selenium
EDTA blood samples (S-Monovette EDTA K3E, Sarstedt) were collected by trained laboratory technicians, centrifuged at 2000 G for 10 min and aliquoted into cryotubes for further storage at −70°C in the biobank. The plasma samples were shipped to Norway on dry ice for Se analysis.
Se was measured using inductively coupled plasma MS (ICP-MS, Nexion 300D, PerkinElmer) at the Environmental Pollutant Laboratory, Department of Laboratory Medicine, University Hospital of North Norway. 200 µl plasma samples were diluted by a liquid handler (1:20) with an alkaline reagent with helium as cell gas at 4·8 ml flow in kinetic energy discrimination mode. Rhodium (Inorganic Ventures) acted as an internal standard(Reference Huber, Michel and Reijnen40). Matrix-matched calibration curves were prepared with clinical plasma samples (Recipe Chemicals and Instruments GmbH). All analyses were performed by qualified laboratory technicians according to the clinical accreditation standard NS-EN IS 15189:2012. The limit of detection for Se was 2·39 µg/l. Quality control samples from Recipe Chemicals, Germany, and Sero AS, Norway (ClinChek plasma and Seronorm serum, two levels each), were analysed together with each batch of 32 research samples and had coefficients of variation of 5 % and 6 %, respectively. All the quality controls were within the acceptance limits. The laboratory equipment was tested for background Se-contamination, and no significant contamination was found.
Motor development
TIMP is a standardised test designed to assess the infant’s motor performance from 34 weeks postmenstrual age to 4–5 months(Reference Campbell5). TIMP is considered to be both a clinically and psychometrically sound tool for assessing early motor performance of infants(Reference Noble and Boyd41) and is widely used by physical and occupational therapists(Reference Haffner and Sankovic42). The test consists of forty-two items, of which thirteen are observed (dichotomised to zero-not observed and one-observed) and twenty-nine are elicited items (scores range from 0–6). The elicited items assess the infant activities such as head control, turning of the head or holding a stable posture, turning the head at the sound direction in prone position with neck and trunk extended, and crawling. The observed items assess spontaneous activities such as control of fingers and ankles, orientation of head in midline and development of ballistic, oscillating and fidgety movements.
Prior to the study, two assessors (a paediatrician and a psychologist) were trained in test procedures and administration. Standardisation procedures were conducted on twenty infants, during which both examiners scored the test simultaneously, yielding excellent interrater agreement (intraclass correlation coefficient > 0·94). Throughout the study, 5 % of the infants were randomly selected for double-scoring by the second assessor to ensure quality and consistency in test administration, yielding an intraclass correlation coefficient greater than 0·93(Reference Kvestad, Silpakar and Hysing9).
The assessments were performed in a room that was well-lit and free from distractions. We ensured that the infants were well-fed, healthy and not sleepy before starting the test. The test took approximately 20–50 min to administer. No adjustments for testing were considered for the current setting. Brief feedback on the infant’s performance was given if the infant was found delayed after assessment.
The total possible score for the TIMP is 142 (observed: thirteen and elicited: 129), with higher scores indicating better motor performance. The TIMP also offers age-specific norms based on a USA normative sample classifying the motor performance into average range (± 1 sd of the normative mean), low average (–0·5 sd to –1 sd of the normative mean), below average (−1 to −2 sd below the normative mean) and far below average range (< −2 sd below the normative mean)(Reference Campbell5).
Statistical analysis
Statistical analyses were performed according to a predefined analysis plan registered at zenodo.org (https://zenodo.org/records/14523619). Demographic and clinical characteristics of the participant mothers are presented as means and sd and numbers (n) and percentages (%). We omitted any case that had missing data in any of the variables. With 290 participants in the Se-deficient group and 509 in the normal group, we had 80 % and 90 % power to detect standardised effect size differences of 0·21 and 0·24, respectively. These calculations were based on a two-sided α level of 0·05. Before the main analysis, we checked for curve linear associations between Se concentration and the TIMP scores through visual inspection (Stata command two-way fpfitci) and could not identify obvious curve linear relationships. The associations between the maternal Se concentration (continuous, dichotomised and tertiles) and the TIMP scores (total, observed and elicited) were estimated in multiple linear regression models adjusting for predefined potential confounders (i.e. maternal age and Water and sanitation, Asset ownership, Maternal education, and household Income index). Adjustment variables were set based on the literature that considered the variables as predictors both for maternal Se and motor development of infants(Reference Phiri, Ander and Bailey30–Reference Rodrigues, Joshi and Rajadhyaksha36). We also examined the associations between the Se concentration and the total TIMP score dichotomised as scores below the average range (< –1 sd of the average norm scores) and within the average range (± 1 sd of the average norm scores) using logistic regression. We also ran all models, linear and logistic, with interaction terms between Se and child sex and Se and vitamin B12 treatment in separate models. Significance level was set at p-value <0·05. We used Stata version 18·5 for all analyses.
Results
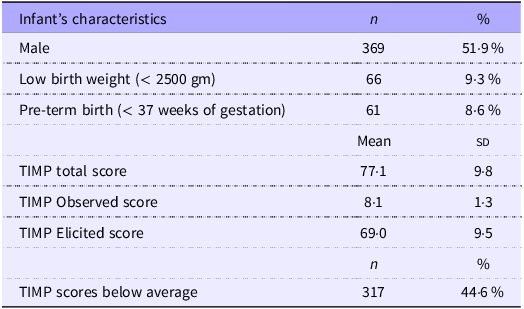
The average maternal age (sd) at enrolment was 27·6 (4·0) years. Of the 711 included women, around 68·2 % were working, and 79 % had completed secondary school or above. Altogether 66·1 % of the women were living in joint families, and 23·8 % were residing in rented houses and 26 % had kitchen and bed in the same room. Among the participants, 69·1 % had their own land, and 10·6 % received remittances from abroad (Table 1). Among the infants, 51·9 % were male, 9·3 % were born with low birth weight, and 8·6 % were born preterm (Table 2). The mean (sd) plasma maternal Se concentration was 74·8 µg/l (10·3 µg/l), and 35·7 % had Se deficiency using the cut-off of 71·1 µg/l. The mean (sd) TIMP total score when the infants were 8–12 weeks old was 77·1 (9·8), 317 of the infants (44·6 %) have TIMP scores below average.
Table 1. Demographic, socioeconomic and nutritional features of 711 pregnant mothers of Bhaktapur, Nepal

TIMP; Test of Infant Motor Performance.
All numbers are numbers (percentage) if not otherwise stated, †Nepalese Rupees, *Water and sanitation, household assets, maternal education and household income, range 0–1.
Table 2. Characteristics and motor development of 711 infants in Bhaktapur, Nepal

TIMP, Test of Infant Motor Performance.
Among infants with TIMP scores below (n 394) and within (n 317) the average mean (sd), maternal Se concentration was 74·9 µg/l (10·5 µg/l) and 74·8 µg/l (10·1 µg/l), respectively. Maternal Se concentrations at gestational week up to 12 weeks and after were 75·9 µg/l (10·3 µg/l) and 73·4 µg/l (10·2 µg/l), respectively (Table 1).
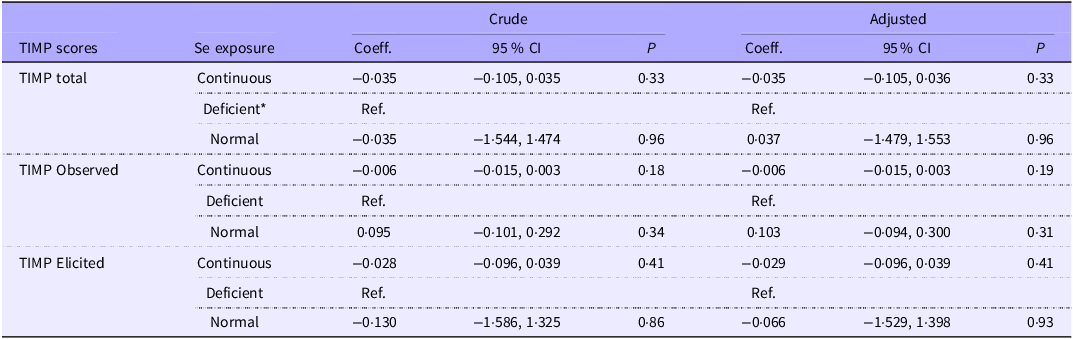
The association between maternal plasma Se concentration and TIMP scores is present in Tables 3–5. There were no linear associations between maternal Se concentration and the TIMP total and subscale scores (β for the total TIMP score: −0·035 (95 % CI: adjusted −0·105, 0·036) (Table 3), and no association between maternal Se concentration and TIMP scores dichotomised as below and within average range (OR = 0·999 (95 % CI: adjusted −0·986, 1·014) (Table 5). When dichotomising the maternal Se concentration into deficient v. normal (Table 3) and categorising in tertiles (Table 4), there were no associations between Se concentration and the TIMP scores. Our analyses did not identify significant effect modifications by sex of the child and vitamin B12 supplementation across any of the models including analyses with TIMP scores as continuous and dichotomised and Se with continuous, dichotomised and categorised in tertiles (all P-values were above 0·05).
Table 3. Associations between maternal plasma selenium concentration and TIMP scores in 711 infants from Bhaktapur, Nepal

TIMP, Test of Infant Motor Performance.
*Se concentration < 71·1 μg/l. Adjusted for maternal age and Water and sanitation, Asset ownership, Maternal education, and household Income. Analyses are based on linear regression models.
Table 4. Associations between maternal plasma selenium concentration (tertiles) and TIMP scores in 711 infants from Bhaktapur, Nepal

TIMP, Test of Infant Motor Performance.
Adjusted for maternal age and Water and sanitation, Asset ownership, Maternal education, and household Income, *Reference group.
Analyses are based on linear regression models.
Table 5. Associations between maternal plasma selenium concentration and below average and average TIMP scores in 711 infants from Bhaktapur, Nepal

TIMP, Test of Infant Motor Performance.
Adjusted for maternal age and Water and sanitation, Asset ownership, Maternal education, and household Income, Analyses are based on logistic regression models.
Discussion
In this sample of Nepalese mother–infant dyads, a substantial proportion of the women were Se-deficient as measured within 15 weeks of gestation. The current study aimed to investigate the association between maternal Se concentration and motor performance measured in early infancy. Although Se plays a vital role in motor functions(Reference Rederstorff, Krol and Lescure43,Reference Kelly, Coe and Shenkin44) and there is a described maternal deficiency in the population, we could not find associations between maternal Se concentration and infants’ motor performance measured at 8–12 weeks of age in these Nepalese infants.
To our knowledge, this is one of very few studies to investigate the association between maternal Se concentration and infants’ motor performance. We have been able to identify one previous study from China that also measured early development(Reference Yang, Yu and Fu26); however, findings from this study contrasted with our results showing an association between Se concentration and early development. The differences may be due to methodological issues. In the previous study, they used umbilical cord blood from neonates to measure Se concentration and motor development was measured by the Neonatal Behavioural Neurological Assessment scale. The Neonatal Behavioural Neurological Assessment is a broader scale not specifically designed to measure motor development(Reference Al-Haddad, Olson and Reardon45), and it was conducted when the children were only three days old(Reference Yang, Yu and Fu26). Another key difference is the use of cord blood Se plasma concentration as a biomarker which might differ from Se concentration measured during the prenatal period. There are also other studies that showed significant positive associations between Se and motor development(Reference Varsi, Bolann and Torsvik22–Reference Guo, Xu and Tian25). These assessed motor performance after 6 months of age; however, and in contrast to our study, most have measured the maternal Se concentrations in later pregnancy (i.e. during second and third trimester) or after delivery using umbilical cord blood(Reference Varsi, Bolann and Torsvik22,Reference Kobayashi, Itoh and Miyashita23,Reference Yang, Yu and Fu26) .
We could not find effect modifications of vitamin B12 in the association between maternal Se concentration and motor development of infants. Our findings also do not signify sex as a moderating factor in the association between maternal Se and motor development, that is discrepant from the findings from Bangladesh and China showing protective effects of Se in girls(Reference Skröder, Hamadani and Tofail21,Reference Wang, Wang and Yan46) . This could be because of relatively narrow safe range of Se concentrations required for optimum enzyme functioning among the participating mothers in our study(Reference Thomson47) showing neither very high nor very low maternal Se concentration, which may result in limited ability to detect sex-specific effect modifications in motor performance of the infants. Moreover, the null findings could be due to the lack of severely Se-compromised conditions in our sample, which might otherwise have led to neurodegeneration due to the competition between brain and testes for Se consumption in males(Reference Pitts, Kremer and Hashimoto48). Other reasons for the null findings could be attributed to the presence of multiple factors influencing neurodevelopment in high-risk groups, particularly in low-income and middle-income countries. These factors may overshadow or counteract the effects of Se, even though a significant proportion of individuals in the study was classified as Se-deficient based on the defined threshold (< 71·1 µg/l). Interaction between Se and other micronutrients has also been reported for various clinical outcomes. For example, higher folate status in combination with high Se has been associated with an increased risk of autism spectrum disorder(Reference Lee, Ji and Raghavan29). In addition, interactions between Se and iron status have been suggested, whereby adequate Se intake may protect against reactive oxygen species in conditions of iron overload, such as secondary haemochromatosis(Reference Bjørklund, Aaseth and Skalny49) and calcium has also shown interactions with Se in the improvement of skeletal muscle activity in mice(Reference Bodnár, Ruzsnavszky and Oláh50). In the current study, all mothers, regardless of study group, received folic acid, iron and calcium. We were, therefore, unable to examine the potential effect modification by these supplements. The questions on interactions between baseline maternal micronutrient biomarker concentration and maternal Se concentration in relation to neurodevelopmental outcomes should be raised in future studies.
About 45 % of the infants in the current study fall into the below average category of TIMP scores when using USA norms. This indicates that many of the children have a delay in motor development compared with the USA normative sample. According to our results, however, Se deficiency in pregnancy does not account for this developmental delay. In the current study population, there are many risk factors that may limit development in the children, such as low level of knowledge about child development among Nepalese mothers(Reference Shrestha, Ulak and Strand51), leading to a risk of limited exposure to learning materials and stimulation at home. Being born with low birth weight and length-for-age, preterm birth, anaemia and diet patterns are other prevalent risk factors that have been shown to be related to child development in this setting(Reference Ranjitkar, Hysing and Kvestad52,Reference Siegel, Stoltzfus and Kariger53) . Another potential factor could be the short stature of mothers which have shown negative associations with cognitive and motor development in low-income and middle-income countries(Reference Sania, Sudfeld and Danaei54). In other words, the null findings of our study could indicate that maternal Se concentration might not be a key factor of motor development in early infancy in the current population.
The strengths of the present work include a relatively large sample of up to 800 mother–infant dyads from a general Nepalese population and the high-quality measurement of motor development(Reference Kvestad, Silpakar and Hysing9). Laboratory procedures were carried out by trained and qualified technicians. Blood samples were stored and shipped from the study site in Bhaktapur to the laboratory in Norway in accordance with established biobanking standards. Moreover, we used Se concentration both as a continuous and categorical (deficient v. normal and in tertiles) variables in the regression models. Likewise, we assessed the associations using the motor scores both on a continuous scale and dichotomised according to USA norms. There are some limitations of this study. Plasma Se concentration might not be the ideal biomarker for Se status leading to random misclassification, and thus, both under- and overestimations of the association between maternal Se and motor performance could occur. Yet, we see that previous studies have found associations using the same biomarker(Reference Varsi, Bolann and Torsvik22,Reference Polanska, Krol and Sobala24) . The current cohort is from a healthy population participating in a clinical trial on the effect of vitamin B12 with specific exclusion criteria such as the requirement of adequate nutritional status. Generalisability of the results should therefore be cautiously handled.
Conclusion
Despite Se deficiency being prevalent in the current study sample, we could not identify associations between maternal Se concentration and infant motor performance in a healthy cohort of Nepalese pregnant women. The findings could indicate that Se alone might not be a key factor of motor development in early infancy. Future research is suggested for an update of the nutrition recommendations for pregnant women and to better understand the specific contributing factor of early motor development.
Acknowledgments
We would like to thank all the participating mothers and their children who took part in the study. We would also like to thank all the study staff (Psychologists, Supervisors, Data managers, Lab technicians and Field workers) and all the Child Health Research Project administrative staff. We would also specially thank psychologist Jaya Shilpakar for the assessment of TIMP. We are also thankful to Prof. Laxman Shrestha and the founder president of Siddhi Memorial Foundation, Shyam Sunder Dhaubhadel, for being cooperative throughout the study period. We are also thankful to the laboratory technicians at the Environmental Pollutant Laboratory, University Hospital of North Norway, for conducting the Se analysis.
This study is funded by RCN (Research Council of Norway) project grant number 336566. RCN had no role in the design, analysis or writing of this article.
Conceptualisation and funding acquisition: S. R., R. K. C., T. A. S., M. A., M. H. and I. K.; Investigation: S. R., M. H., R. K. C., T. A. S., M. U., S. H., M. A., K. S. B. and I. K.; Formal analysis: S. R., M. H. and I. K.; Blood sample analysis: S. H. and M. A.; Writing – original draft: S. R., M. H., R. K. C., T. A. S., M. A. and I. K.; Writing – review and editing: S. R., M. H., R. K. C., T. A. S., M. U., S. H., M. A., K. S. B. and I. K.; Primary responsibility for final content: SR; All authors: read and approved the final manuscript.
None of the authors reported a conflict of interest related to the study.
Data will be available on request. For that purpose, the request should be first approved by the Nepal Health Research Council, Nepal, and then should be sent to the authors by sending an email to the Department of Paediatrics, Institute of Medicine, Tribhuvan University, Kathmandu, Nepal (chrp2015@gmail.com).
This study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human subjects/patients were approved by the Nepal Health Research Council in Nepal (NHRC, NHRC; 253/2016, follow up approval 423/2023) and from the Regional Committee for Medical and Health Research Ethics in Norway (REK vest; reference number 2016/1620). Written informed consent was obtained from all pregnant women at enrolment. The study was registered at ClinicalTrials.gov, NCT03071666.