Venous thromboembolism (VTE), encompassing deep vein thrombosis and pulmonary embolism (PE), is a prevalent cardiovascular disorder with significant morbidity and mortality(Reference Khan, Tritschler and Kahn1). It presents with symptoms ranging from painful leg swelling to life-threatening PE and contributes to long-term complications such as recurrent VTE, post-PE syndrome and chronic thromboembolic pulmonary hypertension(Reference Bartholomew2). VTE affects approximately 10 million individuals globally, ranking as the third leading vascular diagnosis(Reference Di Nisio, van Es and Büller3). The economic burden of VTE is substantial with annual healthcare costs estimated between €1·5 and €3·3 billion in Europe and between $7 and $10 billion in the USA(Reference Grosse, Nelson and Nyarko4,Reference Barco, Woersching and Spyropoulos5) . VTE is responsible for significant mortality rates differing markedly between genders and age groups in Europe(Reference Barco, Woersching and Spyropoulos5). Although the prevalence in Asia is lower than in Western countries, the incidence is increasing annually(Reference Lee, Gallus and Jindal6,Reference Yamashita, Morimoto and Kimura7) .
The multifactorial nature of VTE risk encompasses acute triggers like surgery and sub-acute conditions such as inflammation, alongside demographic and behavioural factors such as obesity, highlighting the complexity of its epidemiology(Reference Gregson, Kaptoge and Bolton8). Recent prevention strategies have evolved to focus not only on hospital-associated risk factors but also on broader, lifestyle-related risks(Reference Lutsey and Zakai9). Therefore, appropriate control of risk factors is important in the management of VTE.
Malnutrition, defined as any nutritional imbalance, is a critical yet often overlooked condition that impacts health outcomes(Reference White, Guenter and Jensen10). While traditionally associated with undernutrition, malnutrition due to overnutrition also poses a significant risk, particularly in the context of severe acute illness or major trauma(Reference White, Guenter and Jensen10). Observational studies have shown that malnutrition is associated with increased risks of hospitalisation, surgery and VTE among patients with inflammatory conditions like inflammatory bowel disease(Reference Fine, Zhu and Shirazi11). Early screening for malnutrition and prompt nutrition intervention if indicated has been shown to prevent or mitigate many of these outlined risk factors(Reference Chiu, Oleynick and Raman12). However, despite the evidences in observational studies, a definitive causal relationship still lacks in high-quality research evidence between malnutrition and VTE.
Mendelian randomisation (MR) offers a robust approach to infer causal relationships between modifiable exposures and health outcomes, employing genetic variants as instrumental variables (IV)(Reference Emdin, Khera and Kathiresan13,Reference Sanderson, Glymour and Holmes14) . This method helps overcome the limitations of traditional observational studies, such as confounding and reverse causation, and provides a more reliable estimation of the effects of long-term exposure(Reference Larsson, Butterworth and Burgess15).
Therefore, the primary objective of this study is to explore the causal relationship between malnutrition, overweight and VTE using a two-sample MR design. By employing genetic data to approximate a randomised control setting, this study aims to clarify whether nutritional status significantly influences the risk of developing VTE, addressing a gap in current medical understanding and potentially guiding future nutritional and preventive strategies.
Experimental methods
Study design
This study utilised a two-sample MR approach to investigate the causal relationship between malnutrition and VTE, as illustrated in online Supplementary Figure S1. The conceptual framework was anchored in three fundamental principles essential to MR methodology. First, the relevance criterion requires a strong and demonstrable association between the IV and the exposure of interest. Second, the independence criterion demands that the IV are not associated with any confounders that might simultaneously affect the exposure and the outcome. Third, the exclusion–restriction criterion mandates that the IV influence the outcome exclusively through their impact on the exposure, precluding any alternative causal pathways(Reference Sanderson, Glymour and Holmes16).
This article is an MR study. The data for this study were obtained from publicly available databases and published literature data and do not require ethical approval and written informed consent.
Data sources
The exposures of this study included malnutrition and overweight. Malnutrition is defined as inadequate intake of protein and energies. Overweight is defined as BMI ≥ 25 kg/m2(Reference Berndt, Gustafsson and Mägi17). The following datasets were selected as outcome data: (1) malnutrition (IEU GWAS Catalog GCST90435738, https://www.ebi.ac.UK/gwas/studies/GCST90435738) with 1057 British ancestry cases and 406 492 British ancestry controls; (2) overweight (IEU GWAS Catalog ieu-a-93) with 93 015 European ancestry cases and 65 840 controls(Reference Berndt, Gustafsson and Mägi17). For the examination of the association with VTE as an outcome, GWAS summary-level data pertinent to VTE were derived from the IEU’s pooled datasets with 23 367 European ancestry cases and 430 366 controls. The outcome and exposure data were carefully selected to ensure the robustness and relevance of the genetic tools used in this study. Online Supplementary Table S1 systematically presents more details of the exposure factor and outcome datasets.
Instrumental variable selection
The selection of IV for this study was guided by a systematic process: (1) SNP exhibiting significant associations with the entire genome of malnutrition or VTE were identified. A stringent significance threshold of P < 5 × 10–8 was applied for associations with overweight, and P < 5 × 10–6 for malnutrition, with a minimum allele frequency criterion set at 0·01(Reference Li, Miao and Zhang18); (2) To evaluate and reduce the potential for linkage disequilibrium among the chosen IV, a clumping procedure was employed, setting a threshold of R 2 < 0·001 and a clumping distance of 10 000 kb(Reference Larsson, Butterworth and Burgess15,Reference Palmer, Lawlor and Harbord19) ; (3) In cases where an identified IV SNP was unavailable in the outcome summary data, substitute SNP with high linkage disequilibrium (R2 > 0·8) were considered as substitutes(Reference Larsson, Butterworth and Burgess15,Reference Palmer, Lawlor and Harbord19) ; (4) To ensure the robustness of each SNP within the IV and minimise the risk of weak instrument bias, the F statistic for each SNP was calculated using the formula: F = R 2 × (N − 2)/(1 – R 2). R 2 refers to the proportion of variance in exposure explained by the SNP in IV, and the requirement for the F value is > 10(Reference Burgess and Thompson20). Detailed information regarding the selected IV is provided in online Supplementary Table S2.
Mendelian randomisation analysis
The primary analytical method employed in this study was the inverse variance weighted (IVW) approach, which computes a weighted average of the effect sizes and uses the inverse variances of each SNP as weights(Reference Burgess, Butterworth and Thompson21). The IVW analysis adopted the fixed effects estimation method. This approach allows us to assess the causal relationship between exposure and risk of outcomes, producing estimates of the OR and 95 % CI. To ensure the robustness of the results, additional analyses were conducted using the MR-Egger, weighted median and weighted mode methods. The MR-Egger approach, accounting for the potential presence of pleiotropy through an intercept term, offers an unbiased estimate of causal effects even in its presence(Reference Burgess and Thompson22). The weighted median method, predicated on the validity of at least half of the IV, was applied to examine the causal link between exposure and outcome(Reference Bowden, Davey Smith and Haycock23). To further ensure the robustness of our findings and address potential reverse causality, we applied the Steiger filtering method from the R package. This method helps to identify and exclude SNP that may have a stronger association with the outcome than the exposure, thereby reducing the risk of reverse causality.
Sensitivity analysis
Sensitivity analyses were conducted to explore potential heterogeneity and horizontal pleiotropy which could bias MR estimates. Heterogeneity among IV was assessed using Cochran’s Q test, where a P-value greater than 0·05 indicates low heterogeneity. This suggests that any variation among the IV is likely random and has minimal impact on the IVW results(Reference Greco, Minelli and Sheehan24). To evaluate the presence of horizontal pleiotropy, MR-Egger regression was employed. A non-significant intercept in this regression model suggests an absence of pleiotropy that could skew the MR estimates(Reference Bowden, Davey Smith and Burgess25). Additionally, the MR pleiotropy residual sum and outlier (MR-PRESSO) method was utilised to detect and adjust for outliers among the SNP. SNP with a P-value less than 0·05 were considered potential outliers and were excluded from the analysis to mitigate the effects of horizontal pleiotropy and enhance the accuracy of the causal estimates(Reference Verbanck, Chen and Neale26). Leave-one-out analysis was applied to identify the influence of outliers by systematically excluding each genetic variant one at a time from the analysis, ensuring that the findings are not driven by a single genetic instrument(Reference Larsson, Butterworth and Burgess15).
Statistical analysis
The analysis was conducted using R version 4.0.5, employing the ‘Two-sample MR’ package designed specifically for MR studies. Graphical representations such as forest plots, scatter plots and funnel plots were utilised to visually interpret the data.
Results
SNP selection
In this study, a total of twenty-three IV relevant to malnutrition and overweight were selected. When MR analyses were conducted with VTE as the outcome, all IV were aligned with corresponding information in the summary data sets. The detailed information of the included IV is summarised in online Supplementary Table S2. All selected IV exceeded the F-statistic threshold of 10.
Mendelian randomisation analysis
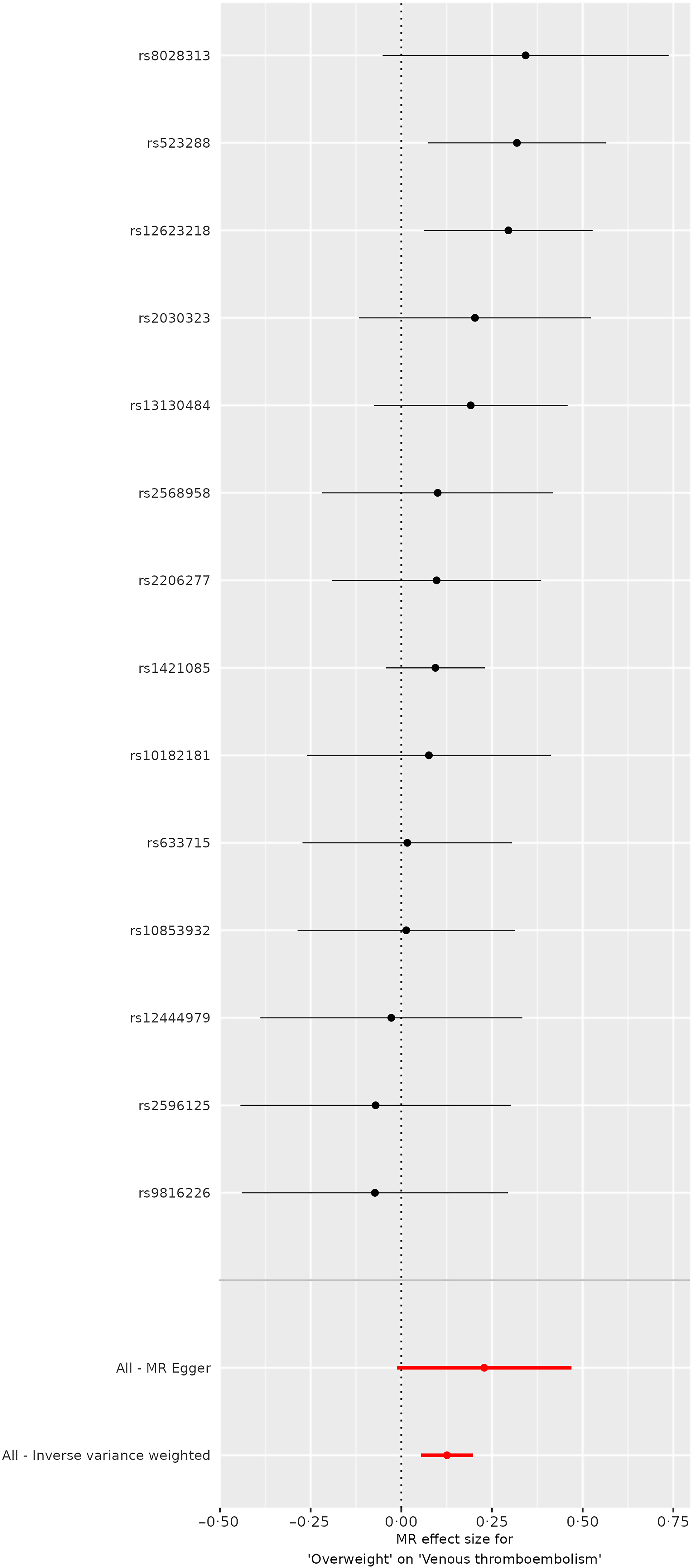
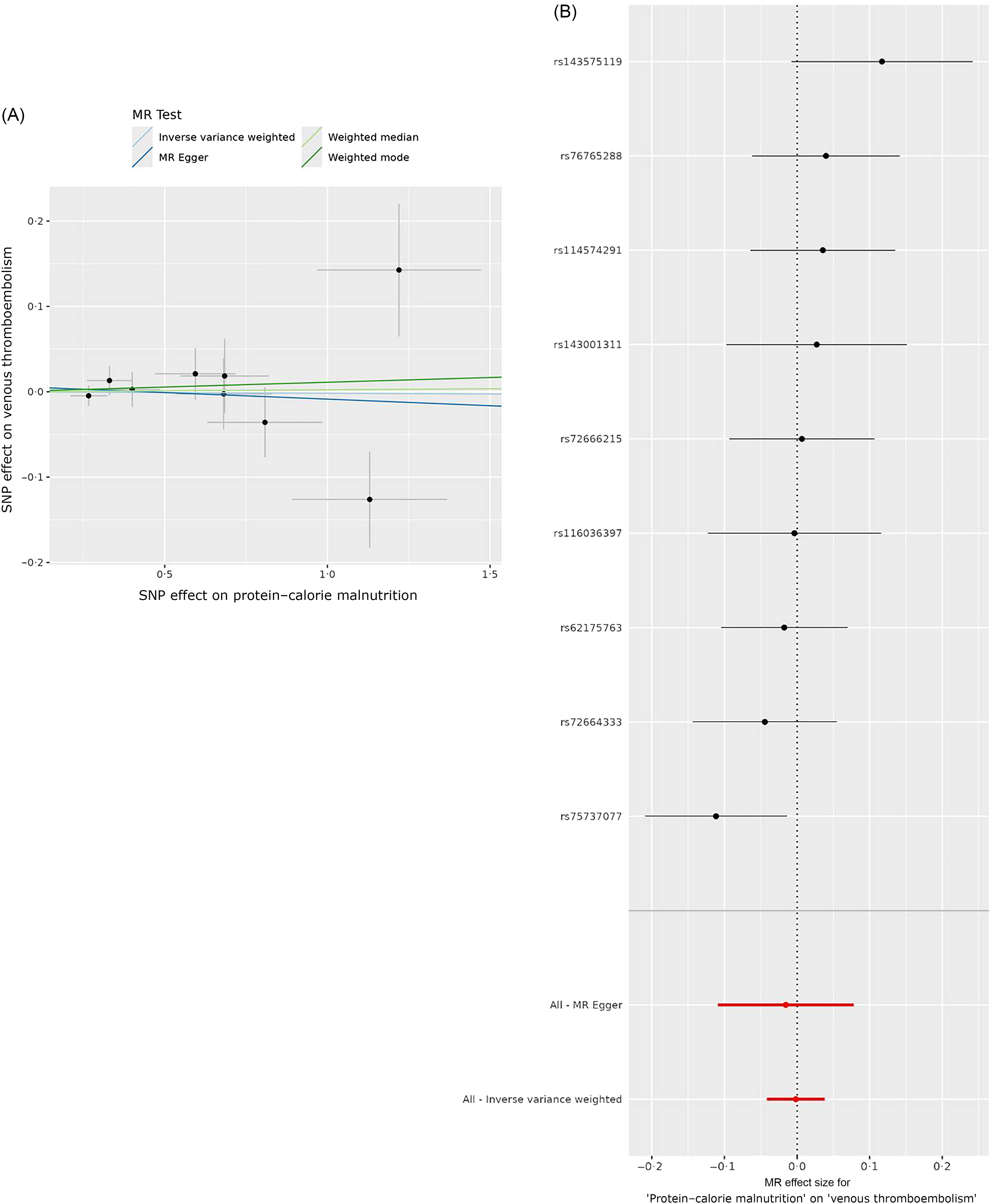
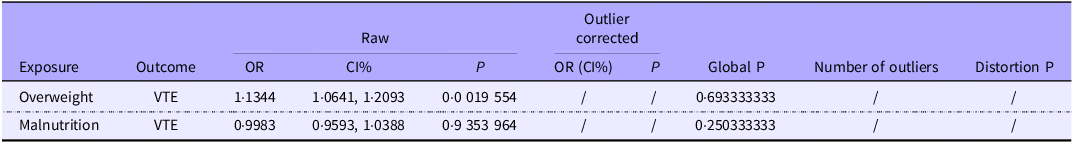
The inverse variance weighted (IVW) analysis suggested a positive genetic influence of being overweight on the risk of VTE, with an OR of 1·1344 and a statistically significant 95 % CI ranging from 1·056 to 1·2186 (P < 0·001). (Table 1 and Figures 1 and 2). However, the results of weighted median analysis, MR Egger and weighted mode analysis were not consistent with the IVW method (P > 0·05). IVW showed no genetic causal effect of protein–energy malnutrition (OR = 0·9983, 95 % CI = 0·9593, 1·0388, P = 0·9333) on VTE. The results of weighted median analysis, MR Egger and weighted mode analysis were consistent with the IVW method (Table 1 and Figure 3).
Genetic prediction of causal association between body weight and risk of VTE using IVW, MR-Egger, weighted mode and weighted median analysis (OR and 95 % CI)

MR, Mendelian randomization; IVW, inverse variance weighted; VTE, venous thromboembolism.
Scatter plots visualise the causal effects of overweight on venous thromboembolism (VTE).

Forest plots visualise the causal effects of overweight on VTE.

Scatter plot and forest plot visualise the causal effects of malnutrition on VTE.

Sensitivity analysis
Cochran’s Q test for heterogeneity affirmed the consistency of MR analysis outcomes, revealing no significant heterogeneity in the relationship between malnutrition, overweight and VTE (all P-values surpassed 0·05) (Table 2). Furthermore, the MR PRESSO analysis identified no outliers in the data (Table 3). The MR-Egger intercept test, designed to detect potential directional pleiotropy, also did not yield compelling evidence of substantial horizontal pleiotropy (all P-values exceeding 0·05), thereby supporting the robustness of our findings (Table 2). Online Supplementary Figure S2 depicted the funnel charts of sensitivity analysis between all P-values and the results were not significant. To further explore the potential impact of outlying genetic variants, a leave-one-out analysis was performed, systematically removing each IV and recalculating the causal estimate. This analysis did not demonstrate any significant changes in the overall causal estimates (online Supplementary Figure S3), indicating that the null findings were not influenced by any individual influential IV.
Heterogeneity tests and multiple validity tests for instrumental variables

IVW, inverse variance weighted; MR, Mendelian randomization; VTE, venous thromboembolism.
Genetic prediction of causal association between body weight status and risk of VTE after removing outliners using MR-PRESSO analysis (OR and 95 % CI)

MR-PRESSO, MR pleiotropy residual sum and outlier;VTE, venous thromboembolism.
Discussion
This study provides a comprehensive analysis of the genetic underpinnings linking malnutrition to VTE. Our results suggest that being overweight has a positive genetic causal effect on the risk of developing VTE. However, this positive result needs to be interpreted with caution as the weighted median and weighted mode methods do not support the results of the IVW method. Conversely, no significant genetic causal associations were observed for malnutrition in relation to VTE. This finding remains consistent across various analytical approaches. By employing a two-sample MR approach, the study minimises potential confounding and provides a clearer causal inference between genetic predispositions to certain nutritional statuses and the risk of VTE. These insights highlight the potential for targeted interventions in populations at risk due to their genetic profile, thus paving the way for more personalised approaches in managing and preventing VTE.
The WHO classifies overweight as a type of malnutrition, emphasising imbalances in nutrient and energy intake(Reference Kobylińska, Antosik and Decyk27). Our MR study substantiates the possible positive genetic causal effect of being overweight on the risk of developing VTE, providing novel insights into how an imbalance nutritional status influences VTE, particularly due to overweight. Several observational studies have demonstrated that obesity and components of metabolic syndrome are significant risk factors for VTE, with specific impacts on the recurrence and development of thrombotic events(Reference Stewart and Kline28,Reference Heit29) . Notably, the elevated risk associated with obesity is apparent across different age groups and is particularly pronounced in younger individuals(Reference Horvei, Brækkan and Hansen30,Reference Stokes, Breheny and Radulescu31) . However, it has also been noted that no evidence of an association between obesity and recurrent VTE has been observed in elderly groups(Reference Mueller, Limacher and Méan32). Inconsistency in observational studies reflects the need for further research. Our MR results align with most observational findings, which further elucidate the genetic basis of these observations and reinforce the role of overweight in VTE risk. Meanwhile, analyses by MR minimise the confounders and reverse causality, which are common limitations in observational studies. Notably, the effect of overweight on VTE appears to be extremely small in this study. Also, there is some heterogeneity among the different MR methods. While the effect may be detectable in a statistical sense due to the large sample size or the precision of measurement, its practical importance is minimal. We therefore believe that more careful consideration should be given to whether the effect of overweight on VTE is clinically significant.
Recent MR studies have demonstrated various aspects of body composition as significant contributors to VTE risk. Xiao et al. demonstrated that leptin is a risk factor for PE, suggesting that targeting leptin levels in obese individuals could reduce PE risk(Reference Xiao, Li and Feng33). Similarly, Wang et al. found significant causal relationships between both waist circumference and hip circumference and VTE, reinforcing the role of central obesity in VTE pathogenesis(Reference Wang, Tan and Hua34). Furthermore, the study by Li et al. expanded on this by showing that height and BMI have causal effects on varicose veins and deep vein thrombosis, further underscoring the systemic impact of body size on venous thrombotic risks(Reference Li, Chen and Gui35). Martin et al. provided insight into the broader spectrum of diseases associated with higher adiposity, identifying conditions where metabolic effects of adiposity primarily exacerbate disease risks, including VTE(Reference Martin, Tyrrell and Thomas36). Yuan et al. and a subsequent study emphasised the dose–response relationships between BMI, waist circumference and VTE, highlighting the significant population-attributable risks due to elevated BMI and reinforcing the causal role of abdominal obesity in VTE(Reference Yuan, Bruzelius and Xiong37). Our MR study further pointed out the possible positive genetic causal effect of overweight on the risk of VTE, suggesting that in addition to waist circumference and hip circumference, simple weight gain may also be involved in the development of VTE. These findings collectively suggest that interventions aimed at reducing obesity levels, particularly central adiposity, could possibly decrease the incidence of VTE. More research is still needed to further confirm this conclusion.
Our MR study indicates that malnutrition due to inadequate protein or energy intake does not exhibit a genetic causal effect on the risk of developing VTE. This finding contrasts with observational studies that have suggested malnutrition associated with inflammatory conditions like inflammatory bowel disease may increase VTE risk. Fine et al. observed that high malnutrition risk significantly correlates with increased occurrences of VTE in Inflammatory Bowel Disease (IBD) patients(Reference Fine, Zhu and Shirazi11). Other studies have also shown that malnutrition is a risk factor for many complications in IBD, including VTE(Reference Chiu, Oleynick and Raman12). However, our MR findings do not support these associations strongly, indicating that while malnutrition correlates with VTE in clinical settings, its genetic basis in causing VTE may not be as pronounced.
Additionally, existing literature has shown that conditions like ulcerative colitis, a type of IBD, are genetically linked to higher VTE risk.(Reference Lv, Gao and Liu38) Research into other nutrients such as vitamin D and its metabolic pathways suggests that while traditional deficiency symptoms are not causally linked to VTE, the genetic expressions involved in vitamin D metabolism might influence VTE risks(Reference Zhang, Sun and Li39). These disparities highlight the complexity of nutritional impacts on VTE, suggesting that the relationship may be influenced by other factors or types of malnutrition. The MR approach encourages a nuanced examination of how different aspects of nutritional status may interact with genetic predispositions to influence VTE risk. Therefore, further studies are required to explore other forms of nutritional status and their potential causal relationships with VTE to fully understand the spectrum of nutritional impacts on venous thromboembolic diseases.
While this study provides valuable insights into the genetic association between malnutrition, overweight and VTE, several limitations must be acknowledged. The lack of statistical significance in the MR-Egger, weighted median and weight mode estimates for being overweight warrants careful interpretation of the results. The intercept term of the MR-Egger regression was close to zero in our analysis, indicating minimal horizontal pleiotropy and reinforcing the validity of the IVW results(Reference Burgess and Thompson22,Reference Bowden, Davey Smith and Burgess25,Reference Bowden, Del Greco and Minelli40) . Therefore, we assume that the result of the IVW method was valuable to some extent, but other replicated studies are needed to further argue for this result. Also, if these variants influence VTE risk through pathways unrelated to nutritional status, it could lead to biased estimates, thereby complicating the causal interpretation. Lastly, the generalisability of our findings may be constrained by the demographic characteristics of the study population, which primarily included individuals of broadly defined European ancestry. While two-sample MR assumes that the exposure and outcome datasets are ancestrally homogeneous and that the same causal processes operate in both datasets, subtle differences within European populations may still exist. Variations in genetic architecture, environmental exposures and lifestyle factors across subpopulations could influence the observed associations. These limitations highlight the need for caution when generalising these findings to more diverse populations or subgroups within European ancestry. Therefore, it is essential to replicate this study in diverse populations to verify the validity and applicability of the findings and ensure a broader understanding of the genetic relationships between weight status and VTE. In addition, trauma and surgery are recognised as major risk factors for VTE as noted before, which highlights the importance of understanding how these factors interact with other potential influences. Therefore, an interesting and potentially impactful direction for future research could be to examine whether overweight (or obesity) acts as an effect modifier in the relationship between trauma or surgery and VTE risk, which could potentially lead to more tailored prevention strategies for those at the highest risk of VTE.
Analysis using two-sample MR revealed a statistically significant genetic association where overweight correlates with an increased risk of VTE. No causal relationships were established between malnutrition and VTE, which could provide new insights into the aetiology of VTE.
Acknowledgements
None.
No financial support was provided.
Y. W. and J. S. carried out the studies, participated in collecting data and drafted the manuscript. Y. W. and J. S. performed the statistical analysis and participated in its design. Y. W. and J. S. participated in the acquisition, analysis or interpretation of data and drafted the manuscript. All authors read and approved the final manuscript.
The authors declare that they have no competing interests.
This article is a MR study. The data for this study were obtained from publicly available databases and published literature data and do not require ethical approval and written informed consent.
Consent for publication: not applicable
All data generated or analysed during this study are included in this article and supplementary information files.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114525000704