Introduction
Globally, 47 million children under 5 years of age were affected by wasting in 2019, out of which 14·3 million were severely wasted. Most of these children are from lower-middle-income countries, with more than two-thirds residing only in Asia and more than one-fourth in Africa. Severe or moderate wasting or acute malnutrition places children at a higher risk of death(1). However, in the case of severe acute malnutrition (SAM), not all children present medical complications needing hospitalisation.
The World Health Organization (WHO) has outlined different dietary management guidelines for severely and moderately acute malnourished children. Ready-to-use therapeutic food (RUTF) is recommended for children with severe acute malnutrition, with or without nutritional oedema, who are enrolled in outpatient care. Children with moderate acute malnutrition (MAM) without oedema also need to be treated with a nutrient-dense diet. However, WHO recommends that the use of specially formulated foods (SFF), combined with counselling, should be considered over counselling alone for MAM children who meet specific criteria. These criteria include narrower anthropometric measures such as a mid-upper arm circumference (MUAC) of 11·5–11·9 cm or a weight-for-age z-score (WAZ) below −3 standard deviations (SD), failure to recover after other interventions, relapse into MAM, a history of SAM, presence of co-morbidities requiring follow-up care, or severe social circumstances(2). Among SFFs, lipid-based nutrient supplements (LNS) are preferred, and in the case of the unavailability of SFFs, fortified blended foods (FBFs) with added sugar, oil and/or milk are suggested. An in-depth examination of the composition, including nutritional profiles of the supplementary foods, is necessary to determine their effectiveness in aiding recovery from SAM and MAM.
Currently, various supplementary food formulations with differing compositions are being used in research settings. The standard RUTF used for treating SAM in outpatient care is a fortified formulation that contains peanuts, 25% milk, sugar and vegetable oil. However, since including a high quantity of milk powder substantially increases the cost of treatment, alternative formulations of RUTF have also been developed with low or no milk components, along with plant-based protein sources(Reference Kohlmann, Callaghan-Gillespie and Gauglitz3,Reference Bahwere, Akomo and Mwale4) . Specially formulated foods used in treating MAM include fortified dairy or non-dairy ready-to-use foods (primarily ready-to-use supplementary foods) that usually contain soy, peanuts, vegetable oil and sugar. Cereal-based blended food products used in treating MAM are typically fortified with micronutrients and include legumes such as soy; however, recent formulations include milk(Reference Stobaugh, Ryan and Kennedy5–Reference Nikiema, Huybregts and Kolsteren8). Few studies have examined the impact of including dairy in different formulations on key nutritional outcomes during the rehabilitation of SAM and MAM. A review by Potani et al.(Reference Potani, Spiegel-Feld and Brixi9) observed lower weight gain and recovery rates from SAM with low and non-dairy RUTF than standard milk-based RUTF. The study, however, also included trials with a notable human immunodeficiency virus (HIV)-positive sample. A review by Suri et al.(Reference Suri, Moorthy and Rosenberg10) observed that using blended foods containing dairy was beneficial for treating MAM; however, treatment with ready-to-use foods was beneficial for recovery and weight gain, irrespective of dairy content. However, neither of these studies reviewed the impact of nutrient quality of formulations on treatment outcomes. Although Cichon et al.(Reference Cichon, Das and Salam11) included trials with significant malaria and HIV-positive samples, the review found that recovery from MAM using fortified blended foods with improved micronutrient and milk content was comparable to lipid-based nutrient supplements. This review highlighted the importance of enhancing the composition of formulations to improve nutritional outcomes, although it does not specifically detail which nutrient content enhancements contribute to better recovery. Therefore, there is a need to comprehensively review the composition of food products to identify the food formulations that have the highest potential in effectively managing SAM and MAM.
Evidence suggests that optimal recovery from wasting requires the rapid restoration of both fat and lean tissues; hence, dietary regimes cannot address protein and energy deficits alone. Instead, supplementary foods must provide a balanced supply of essential amino acids from high-quality proteins and growth-promoting and functional micronutrients(Reference Golden12,Reference Golden13) . Along with protein quality, the density of essential micronutrients becomes an important consideration also because the nutrient content of formulated food products becomes compromised due to high quantities of added sugar, as it has no nutrients and serves only as a concentrated source of energy(Reference Louie, Moshtaghian and Boylan14). Hence, this narrative review was conceptualised to examine the efficacy of different food formulations based on their nutritional profiles, focusing on dairy protein and density of essential nutrients (type I and type II) in treating uncomplicated SAM and MAM in the community.
Methods
Eligibility criteria
We selected efficacy studies of food products used to treat SAM and MAM among children aged 6–60 months of age. We selected articles published between 1 January 2010 and 1 August 2020, a period that would not be influenced by the COVID-19 pandemic. Articles were selected if the full article text was freely available. We included randomised controlled trials (RCT), clinical trials, observational studies and meta-analysis/systematic reviews. Individual efficacy trials were excluded if the study sample size was less than thirty, the study involved a hospital/facility-based setting or the trial involved adults or children without malnutrition. Efficacy trials that administered the food products to SAM and MAM children with severe illnesses such as malaria, HIV or other illnesses (notable sample i.e. ≥2%) were also excluded. Unpublished trials, books, reports and studies published as protocols and reviews (non-systematic) were also excluded.
Search strategy
The articles were identified through databases using a two-stage screening process. The electronic search (first stage) was conducted on two databases: PubMed and Cochrane Library. The searches were conducted using keywords: ‘RUTF’, ‘Community-based management of acute malnutrition’, ‘alternative foods for acute malnutrition’, ‘comparison to ready-to-use therapeutic food’, ‘local foods for acute malnutrition’ and ‘supplementary foods for acute malnutrition’. The backward snowball search (second stage) was also used to identify additional efficacy studies (Fig. 1).
Flow diagram for search strategy to identify studies.

Data extraction and synthesis
Data related to general study characteristics, outcome results, ingredient composition and nutrient value were extracted from the eligible studies. The included randomized controlled trials (RCT) were also assessed for two domains of risk of bias related to the randomisation process using the Cochrane risk of bias 2 (RoB 2) tool: random sequence generation and concealment of allocation sequence(Reference Higgins, Savovic and Page15,Reference Eldridge, Campbell and Campbell16) . For the first part of the review, we undertook a qualitative synthesis of the selected studies to examine and compare the efficacy of dairy and non-dairy food models for treating SAM and MAM. Hence, food models were accordingly categorised as RUTF with dairy, RUTF without dairy, RUSF with dairy, RUSF without dairy, FBF with dairy and FBF without dairy. The primary outcomes that were extracted include recovery rate, weight and length gain.
For each study, details on the type of food supplement and ingredient composition per 100 g, including milk protein, energy (kcal), added sugar and select type I and type II nutrients, were extracted and collated in Microsoft Excel (2019). For studies that did not cite nutrient data, the ingredient composition was used to estimate nutrient content further using databases such as FoodData Central(17). The content of ingredients that were not available on this database was estimated using other databases, such as the Indian Food Composition Tables (IFCT)(Reference Longvah, An_antan_ and Bhaskarachary18). The composition of formulations used for SAM and MAM rehabilitation was also compared with nutrient content recommendations(Reference Golden12,19) .
Results
Study characteristics
The database search and backward snowball search included a total of twenty-six studies (Fig. 1). All studies screened through the inclusion and exclusion criteria were found to be published in English. Table 1 presents the characteristics of the twenty-six studies identified through the search. Most studies involved children aged between 6 and 59 months, although a few had non-inferiority analyses of subgroups. Sample sizes of selected individual trials ranged from 71 to 4078 children diagnosed as having either SAM or MAM. However, two trials evaluated the efficacy of a food-based approach for both SAM and MAM children, and such studies had a larger sample size (Sample >1000 children). Both individual efficacy studies and systematic reviews collected or synthesised data from populations in low-income and lower- and middle-income countries based in Africa and Asia. Only two of the twenty included individual efficacy studies involved study designs other than a RCT. As per the criteria suggested by the RoB 2 tool, only twelve of the included RCT used an adequate random method to generate the allocation sequence and undertook adequate concealment of the allocation(Reference Higgins, Savovic and Page15,Reference Eldridge, Campbell and Campbell16) . In the included individual efficacy studies, a MAM child was either treated for or assigned a non-recovery status after 2–4 months. Similarly, in the case of SAM, the treatment duration or period after which non-responsive status was assigned ranged between 0·5 and 4 months.
Studies on food-based approaches to treat MAM and SAM identified from the database search

MAM, moderate acute malnutrition; SAM, severe acute malnutrition; RCT, randomised controlled trial; MUAC, mid-upper arm circumference; ITT, intention-to-treat; PP, per-protocol; RUTF, ready-to-use therapeutic food; PM, peanut milk; WHZ, weight for height z-score; WLZ, weight for length z-score; CSB, maize–soy blend; RUSF, ready-to-use supplementary food; IMCI, Integrated Management of Childhood Illness; CNS, central nervous system; HIV, human immunodeficiency virus; SD, standard deviation; SR, systematic review; NCHS, National Center for Health Statistics; WHO, World Health Organization; FBF, fortified blended foods; LNS, lipid-based nutrient supplements; HB, haemoglobin; TB, tuberculosis; LMIC, low and middle-income countries; m, month.
Studies largely identified children as SAM or MAM as per anthropometric cutoffs of mid-upper arm circumference or weight for height z-scores as per WHO reference with no complications, including chronic illnesses such as HIV. However, four of the eighteen efficacy studies clarified that HIV and/or tuberculosis were not exclusion criteria. No study, including those conducted in malaria-endemic countries in Sub-Saharan Africa, screened for malaria. Seven of the twelve studies that assessed the efficacy of ready-to-use therapeutic foods (RUTF) for SAM rehabilitation primarily involved a comparison between a standard peanut-milk RUTF and an alternative RUTF that had either milk content (lesser than 50% milk protein requirement) or no milk and relied on plant protein sources. The standard formulation used skimmed milk powder, whereas alternative formulations also used other dairy sources such as whey-based protein. Four of six systematic review studies included meta-analyses that assessed the efficacy of standard RUTF against non-milk and low-milk alternative RUTF. The ten trials that examined the efficacy of supplementary food products for MAM rehabilitation primarily involved two food models: ready-to-use supplementary foods (RUSF) and blended foods that were usually fortified (FBF). However, among the ten trials, three trials used RUTF to treat MAM. There was no standard RUSF formulation, with RUSF being plant-based (soy/sorghum) or dairy-based (milk/whey), including peanuts. Five efficacy trials included a maize–soy blend (non-dairy/dairy) as a control or intervention food product to treat MAM.
Efficacy of dairy and non-dairy formulations in treating MAM and SAM
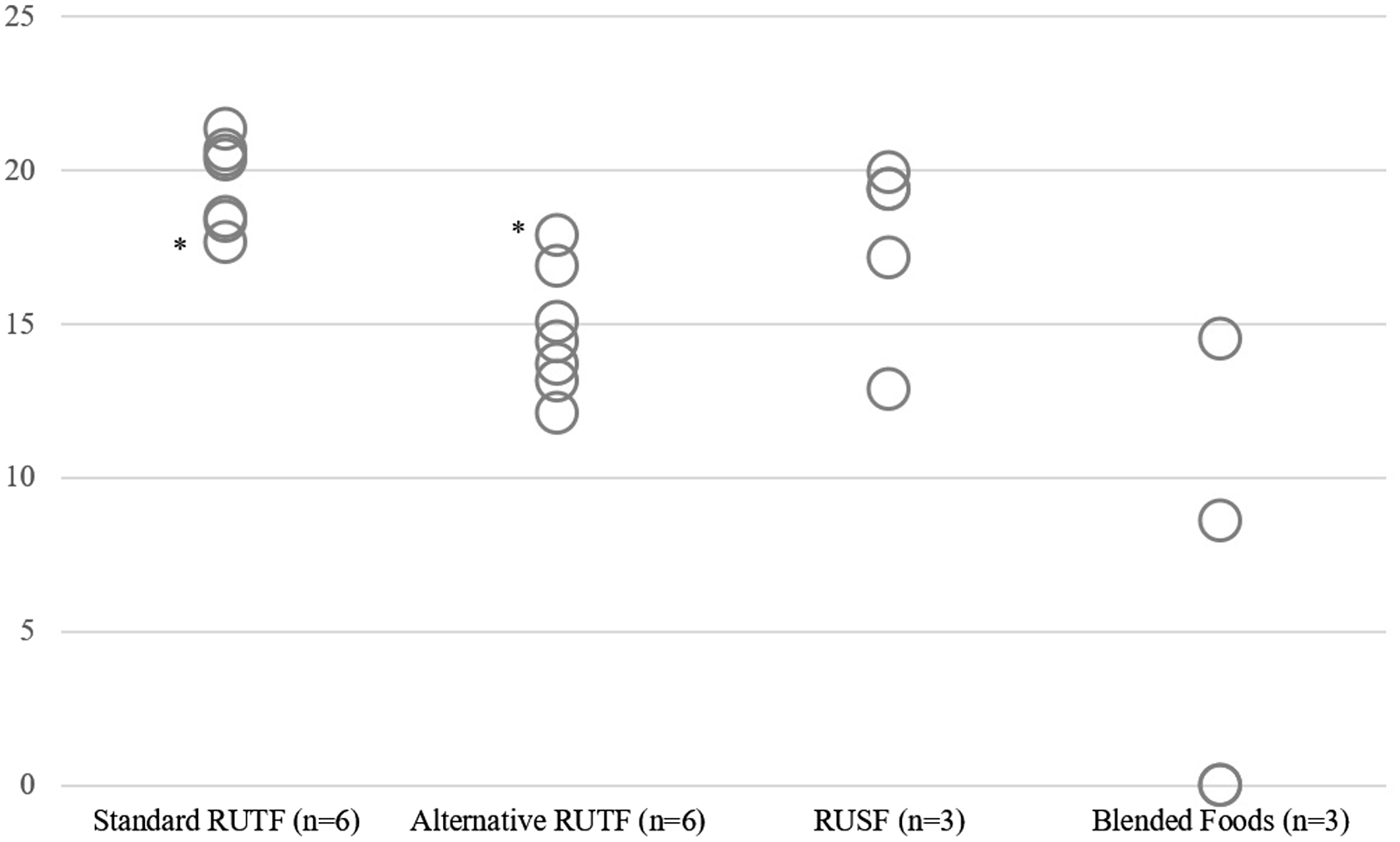
Figure 2 collates the recovery rates of reviewed study product arms, that is, dairy and non-dairy used in treating SAM and MAM(Reference Kohlmann, Callaghan-Gillespie and Gauglitz3–Reference Nikiema, Huybregts and Kolsteren8,Reference Amegovu, Ochola and Ogwok22–Reference Hsieh, Liu and Zeilani27,Reference Kajjura, Veldman and Kassier29–Reference Scherbaum, Purwestri and Stuetz32) . The recovery rates ranged from 47·5% to 92·4% for SAM children treated with standard RUTF. Recovery of SAM children treated with an alternative formulation of RUTF (i.e. low/no milk/other dairy or animal sources) ranged from 54·3% to 93·3%. The recovery rates for SAM were similar for standard milk, alternative milk and alternative non-milk RUTF. Home diets reported the lowest recovery rates among all study arms used in treating SAM, whereas a non-dairy alternative RUTF reported the highest recovery rate.
Recovery rates of different products used for treating SAM and MAM in the selected studies. SAM, severe acute malnutrition; MAM, moderate acute malnutrition; PM, peanut milk; RUTF, ready-to-use therapeutic food; D, dairy; ND, non-dairy; RUSF, ready-to-use supplementary food; BF, blended foods (fortified and non-fortified).
The symbols denote that the RUTF product was used for only MAM treatment (*) or in a combined SAM and MAM protocol (†). For non-inferiority analyses, if both intention-to-treat and per-protocol results were reported, then only per-protocol results were cited (‡).

In the case of rehabilitation of MAM children, the reviewed evidence suggested that recovery from MAM children with non-dairy food products was comparable to that with dairy products. The recovery rates of MAM children treated with dairy-ready-to-use food products (RUSF and RUTF) ranged between 62% and 93·4%. The recovery rates of non-dairy ready-to-use foods ranged from 74·2% to 85% and those of non-dairy blended foods from 67% to 90·4%.
Some studies have assessed the effect of including dairy on treatment outcomes (Table 2)(Reference Bahwere, Akomo and Mwale4,Reference Stobaugh, Ryan and Kennedy5,Reference Karakochuk, van den Briel and Stephens7,Reference Nikiema, Huybregts and Kolsteren8,Reference Bahwere, Balaluka and Wells23,Reference Scherbaum, Purwestri and Stuetz32,Reference Sigh, Roos and Chamnan33) . Two studies(Reference Bahwere, Akomo and Mwale4,Reference Bahwere, Balaluka and Wells23) assessed the efficacy of the standard milk RUTF against non-dairy plant-based RUTF. Both studies found outcomes of the non-milk plant-based RUTF were not inferior to the standard RUTF, although the weight gain in the non-milk plant RUTF was lower in one study. Another study(Reference Sigh, Roos and Chamnan33) observed no significant difference in weight gain between the milk–plant RUTF and the fish-based RUTF groups. For the plant-based products, 9·3% milk containing RUTF was not found to be inferior to 25% milk standard RUTF in terms of recovery for children aged 6–59 months but was confirmed to be inferior in terms of weight gain for 24–59-month-old children(Reference Bahwere, Akomo and Mwale4). In another study, the 10% milk RUTF had lower weight gain, height gain and recovery than the standard 25% milk RUTF(Reference Oakley, Reinking and Sandige30).
Efficacy studies where dairy products were compared with non-dairy products for SAM and MAM treatment (recovery rate, weight gain and height Gain)

SAM, severe acute malnutrition; MAM, moderate acute malnutrition; D, dairy; ND, non-dairy; NS, no significant difference; RUTF, ready-to-use therapeutic food; RUSF, ready-to-use supplementary food; FBF, fortified blended foods; m, month.
* For non-inferiority analyses, if both intention-to-treat and per-protocol results are reported, then only per-protocol results were cited in the table.
Two meta-analyses(Reference Das, Salam and Saeed34,Reference Schoonees, Lombard and Musekiwa38) found that, in terms of recovery, there was no difference (moderate quality evidence) between less or no milk against the standard milk RUTF in treating SAM. However, one meta-analysis(Reference Gera36) found better weight gain and recovery with the standard RUTF than with a non-milk–plant-based blend (grade 1-B evidence). In two meta-analyses(Reference Das, Salam and Saeed34,Reference Schoonees, Lombard and Musekiwa39) , no difference in height gain was observed between RUTF with less or no milk and the standard milk RUTF, with low-quality evidence reported in one. However, one study(Reference Schoonees, Lombard and Musekiwa39) found significant heterogeneity between the trials included in their analysis.
Few individual trials on therapeutic products for SAM treatment have also assessed the potential of alternative RUTF that partially replace skimmed milk with whey against standard RUTF that contain only skimmed milk. A recent study(Reference Hsieh, Liu and Zeilani27) found no significant difference between the standard RUTF and a high-oleic-acid RUTF containing sweet whey for recovery, weight gain and length gain. However, another study(Reference Kohlmann, Callaghan-Gillespie and Gauglitz3) found that recovery and weight gain from SAM were significantly higher in the standard milk RUTF group than in the group receiving the alternative RUTF containing 34% whey protein concentrate (WPC).
Ready-to-use supplementary food (RUSF) and blended foods (fortified and non-fortified) are the primary food models used for treating MAM at the community level. Four studies(Reference Stobaugh, Ryan and Kennedy5,Reference Karakochuk, van den Briel and Stephens7,Reference Nikiema, Huybregts and Kolsteren8,Reference Scherbaum, Purwestri and Stuetz32) have examined the potential of the inclusion of dairy (skimmed milk/whey products) in supplementary foods used to treat MAM (Table 2). In one study(Reference Karakochuk, van den Briel and Stephens7) where MAM children were treated with either a milk RUSF or a non-milk fortified blended food, recovery in the milk RUSF group was found to be higher. On the other hand, no difference was found between non-milk plant-based RUSF and milk-fortified blended food in recovery, length gain and weight gain; however, the blended food included only 8% milk(Reference Nikiema, Huybregts and Kolsteren8). Comparing the potential of ready-to-use products (RUTF and RUSF) with dairy against those without dairy, one study(Reference Scherbaum, Purwestri and Stuetz32) did not observe a statistically significant higher recovery in the non-dairy RUTF arm against the standard milk RUTF. By contrast, the other study(Reference Stobaugh, Ryan and Kennedy5) found that MAM children treated with the RUSF containing whey permeate and WPC-80 had higher recovery and weight gain than the plant RUSF. Three systematic reviews(Reference Das, Salam and Saeed34,Reference Gera, Pena-Rosas and Boy-Mena35,Reference Lazzerini, Rubert and Pani37) included meta-analyses that evaluate the importance of dairy in formulations used for treating MAM. Two meta-analyses, one of which reported high-quality evidence, observed higher efficacy in terms of recovery with the dairy RUSF or lipid-based nutrient supplements (LNS) against supplements without dairy(Reference Das, Salam and Saeed34,Reference Gera, Pena-Rosas and Boy-Mena35) . A similar observation was made in another meta-analysis, which found that, even at complementary doses, milk-based RUSF had higher efficacy in terms of recovery than non-milk blended foods at full doses(Reference Lazzerini, Rubert and Pani37).
Efficacy of essential nutrients to manage acute malnutrition
Dietary supplements must contain minimum quantities of essential nutrients to ensure recovery from SAM or MAM(Reference Golden12,19,40) . The comparison between recommended requirements and nutritional content of food used for the treatment of SAM and MAM children is presented in Tables 3 and 5. The nutrient values are colour-coded to present the gap level (severe, moderate, low and marginal/no gap/ excess).
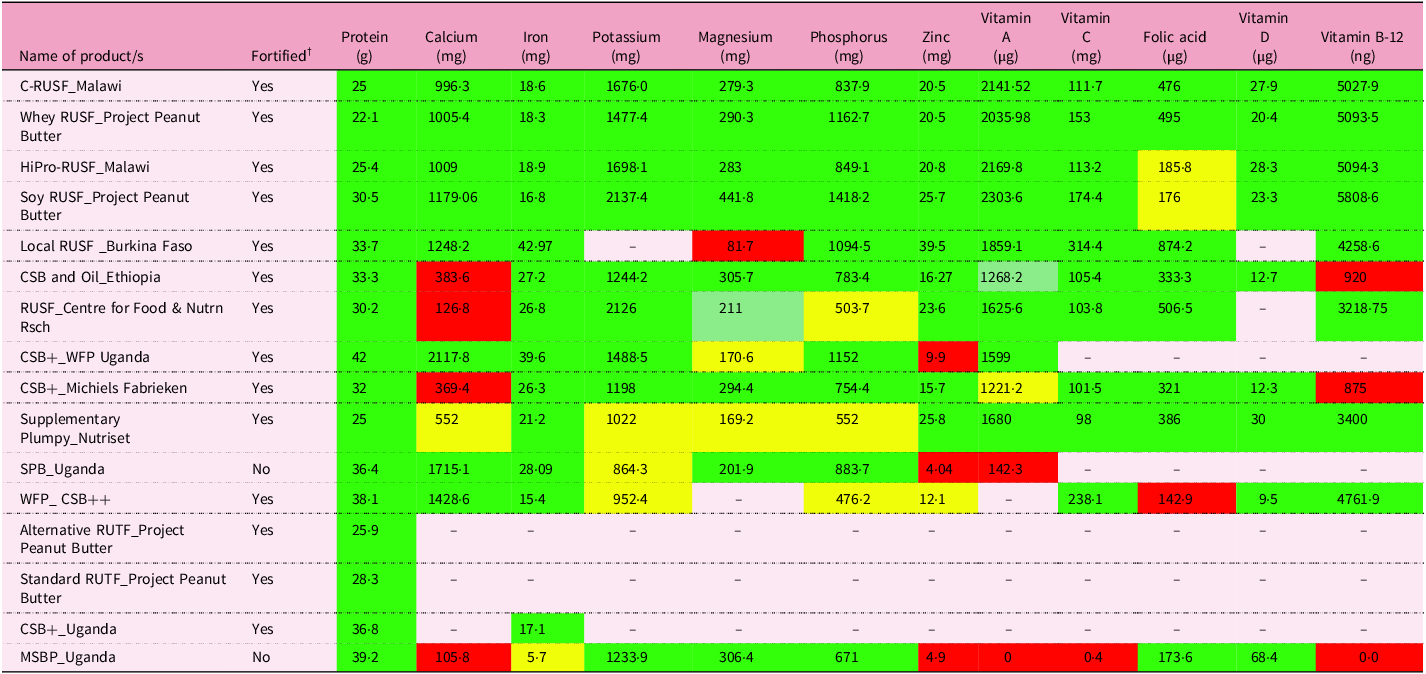
Nutrient densities (i.e. nutrient content per 1000 kcal) of type I and type II nutrients were examined in food products used for MAM treatment. Table 3 examines the nutrient content of sixteen products used in treating MAM (RUSF, RUTF and blended foods) listed in eight studies(Reference Kohlmann, Callaghan-Gillespie and Gauglitz3,Reference Stobaugh, Ryan and Kennedy5–Reference Nikiema, Huybregts and Kolsteren8,Reference Amegovu, Ochola and Ogwok22,Reference Kajjura, Veldman and Kassier29,Reference Roediger, Stein and Callaghan-Gillespie31) . The contents of most products for MAM rehabilitation (RUSF, RUTF, FBF and other food blends) were close to meeting the requirements (food or supplemental) for protein, iron, vitamin B-12 and vitamin D. RUSF products were more likely to meet the requirements for the twelve nutrients, especially phosphorus and magnesium (which are essential for skeletal tissue growth)(Reference Golden12). However, the content of calcium, zinc and folic acid in several products remained far below the recommended levels. The two sorghum-based dairy-free, non-fortified products could not meet the food-based requirements for crucial type I and type II nutrients. Products that replaced milk with maize or soybean reported severe gaps in calcium requirements. Plant-based (maize, soy or sorghum) blended food products were largely dairy-free and contained the linear growth promoter nutrient, zinc, in much lower amounts than the recommended requirements. One study(Reference Amegovu, Ochola and Ogwok22) found that MAM children receiving fortified maize–soy blended foods that contained higher zinc content exhibited faster recovery compared with the unfortified sorghum peanut blend arm. Fortification becomes essential for plant-based formulations to meet requirements of nutrients such as vitamin B-12 (i.e. cobalamin), which occurs exclusively in animal-source foods (Tables 3 and 4).
Adequacy of twelve nutrients in supplementary foods used in the treatment of MAM *

Legend. Red: <50% requirement met; yellow: 50–65% requirement met; green: 65–75% requirement met; dark green: >75% requirement met.
MAM, moderate acute malnutrition; RUSF, ready-to-use supplementary food; CSB, maize–soy blend; WFP, World Food Programme; SPB, sorghum–peanut blend; RUTF, ready-to-use therapeutic food; MSBP; malted sorghum-based porridge.
* Comparison of nutrients with recommended nutrient intakes per 1000 kcal recommended by Golden (2009)(Reference Golden12).
† Unfortified products compared with food RNI and fortified compared with supplementary RNI. ‡Nutrient value estimates using FoodData Central(17) and Longvah et al.(Reference Longvah, An_antan_ and Bhaskarachary18) do not provide processing qualities of extrusion or malting.
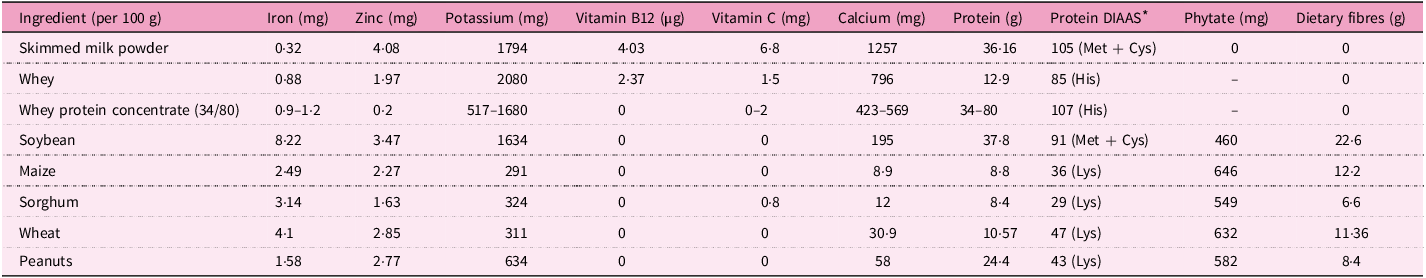
Nutrient and anti-nutrient content in principal ingredients of formulations (17,Reference Vincent, Grande and Compaoré41–47)

DIAAS, digestible indispensable amino acid score.
* First limiting amino acid in parentheses.
Anti-nutrient factors such as dietary fibre and phytate that are present in cereal grains (Table 4) inhibit mineral absorption and possibly impede recovery from acute malnutrition(Reference Gibson, Bailey and Gibbs48,Reference Adams, Sello and Qin49) . The same observation was made in an Ethiopian study(Reference Karakochuk, van den Briel and Stephens7) to account for the higher recovery from MAM with the milk-based RUSF than the milk-free maize–soy blend (CSB). Similarly, a study(Reference Stobaugh, Ryan and Kennedy5) that involved Malawian and Mozambican MAM children attributed the higher content of phytate in the soy RUSF as one of the possible factors to explain the lower recovery reported by the dairy-free soy RUSF compared with the whey RUSF. However, the nutrient content of plant-based formulations could be improved through processing(Reference De Pee and Bloem50). In a recent study in Uganda(Reference Kajjura, Veldman and Kassier29), while the haemoglobin levels of MAM children fed fortified CSB+ were higher at the end line, there was also an improvement in haemoglobin in both the CSB+ and the unfortified malted sorghum-based porridge groups. This improvement in the unfortified product group was attributed to the enzymes activated during the malting process.
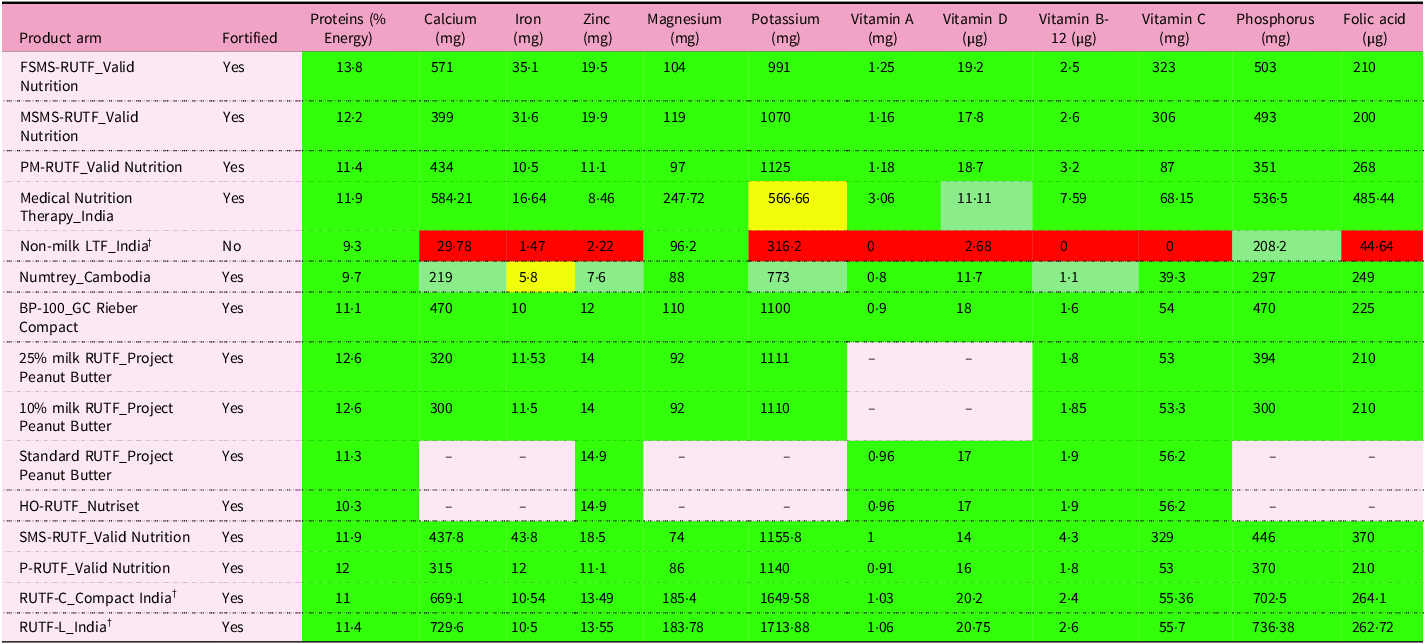
RUTF formulations used for the nutritional rehabilitation of SAM are required to meet the nutrient requirements recommended by WHO(19). Table 5 examines whether the fifteen products in eight trials(Reference Bahwere, Akomo and Mwale4,Reference Bahwere, Balaluka and Wells23,Reference Bhandari, Mohan and Bose25–Reference Jadhav, Karnik and Fernandes28,Reference Oakley, Reinking and Sandige30,Reference Sigh, Roos and Chamnan33) met the minimal requirements for key type I and type II nutrients set by WHO. Dairy and non-dairy RUTF met the minimum protein requirements. With the exception of the unfortified wheat peanut non-milk RUTF that largely met only protein and magnesium requirements, other standard milk and alternative milk and non-milk RUTF formulations were able to closely meet the nutrient requirements recommended by WHO. Alternative plant-based RUTF formulations that were low in dairy or dairy-free not only fully met the requirement of iron but were as much as three to four times the minimum requirement. However, these RUTF were better at correcting anaemia among SAM children than standard milk RUTF(Reference Bahwere, Akomo and Mwale4,Reference Akomo, Bahwere and Murakami21) . One study(Reference Bahwere, Akomo and Mwale4) that implemented three RUTF (i.e. two plant RUTF that were milk-free or low in milk and a standard milk RUTF) observed that increases in body iron stores among SAM children were inversely related to the milk content of the RUTF. However, there are concerns that excessive iron affects the absorption of other nutrients, causes foods to become rancid more rapidly and generates free radicals(Reference Golden12). An overload could also adversely affect gut microflora and growth in iron-replete children(Reference Lönnerdal51) though the observation was not consistent(Reference Akomo, Bahwere and Murakami21).
Adequacy of twelve nutrients in formulations used in the treatment of SAM *

Legend. Red: <50% requirement met; yellow: 50–65% requirement met; green: 65–75% requirement met; dark green: >75% requirement met.
SAM, severe acute malnutrition; FSMS, milk-free soy–maize–sorghum; MSMS, low-milk soy–maize–sorghum; RUTF, ready-to-use therapeutic food; PM, peanut milk; HO, high-oleic acid; LTF, local therapeutic feed; SMS, soy–maize–sorghum.
* Comparison of nutrients with recommended nutrient intakes per 100 g recommended by WHO/UNICEF/WFP/UNSCN (2007)(19).
† Nutrient value estimated using FoodData Central(17) and Longvah et al.(Reference Longvah, An_antan_ and Bhaskarachary18)
Efficacy of added sugar in food products
Added sugar is a major component that alters the nutrient density of formulations. ‘Added sugar’ comprises sugars added to foods during processing, but it is not limited to refined sucrose and also includes ingredients containing monosaccharides and disaccharides, sugars from honey and molasses, sugars from single-ingredient syrups and maltodextrin(Reference Louie, Moshtaghian and Boylan14). Added sugar provides 4 kcal/g, similar to any other digestible carbohydrate(Reference Slavin52). In the current review, treatment formulations largely used sucrose to improve energy intake. Maltodextrin (MD), which is a low-sweet saccharide polymer(Reference Hofman, Van Buul and Brouns53) but contains no sugars(17), is also used by ready-to-use products (RUTF and RUSF) in addition to sugar (sucrose).
Figure 3 summarises the calorific contribution of added sugar in twenty-three food formulations in thirteen studies(Reference Kohlmann, Callaghan-Gillespie and Gauglitz3–Reference Stobaugh, Ryan and Kennedy5,Reference Nikiema, Huybregts and Kolsteren8,Reference Amegovu, Ochola and Ogwok22,Reference Bahwere, Balaluka and Wells23,Reference Bhandari, Mohan and Bose25–Reference Kajjura, Veldman and Kassier29,Reference Roediger, Stein and Callaghan-Gillespie31,Reference Sigh, Roos and Chamnan33) using steps 1–3 in Louie et al.(Reference Louie, Moshtaghian and Boylan14) RUTF formulations that are required to have high energy content to support recovery from SAM (WHO, 2007) contain high quantities of added sugar. The standard RUTF reviewed contained 24·6–29 g/100 g added sugar, contributing 17·6–21·4% of the total energy. Alternative RUTF contained 16·6–25 g/100 g of added sugar, contributing to 12·1–17·9% of energy. No alternative RUTF formulation derived more than 20% energy from added sugar. Standard RUTF, which include 25% or more milk powder, inherently have high total sugars because of the presence of milk sugars that contribute to product sweetness(17,Reference Scapin, Fernandes and Proença54) . Ready-to-use products (RUSF and RUTF) used for MAM treatment contained more added sugar (17–25·7 g/100 g or 12·8–19·4% energy) than food blends (0–15·6 g/100 g or 0–14·5% energy) that needed to be cooked before consumption. A high intake of added sugar can dilute the nutrient content of the diet and increase the total daily energy intake, resulting in weight gain(Reference Louie, Moshtaghian and Boylan14). It was observed that few studies that included products high in added sugar have also reported higher rates of weight gain or reported a high proportion of malnourished achieving the target weight gain(Reference Bahwere, Akomo and Mwale4,Reference Bhandari, Mohan and Bose25,Reference Jadhav, Karnik and Fernandes28) .
Added sugar in products used for treating SAM and MAM in the selected studies. SAM, severe acute malnutrition; MAM, moderate acute malnutrition; RUTF, ready-to-use therapeutic food; RUSF, ready-to-use supplementary food.
*Products used for both SAM and MAM in the study.

While therapeutic food guidelines do not mandate specific limits for sugar or carbohydrate intake in foods used to treat acute malnutrition(Reference Golden12,19,40) , WHO recommends that free sugars, which include added sugars, should not exceed 10% of the total energy intake in the diets of children with a further reduction to <5% energy to ensure optimal health(55). This shows that therapeutic food products that supplement the diet of SAM and MAM children are clearly exceeding dietary norms.
Discussion
Malnutrition occurring among young children may lead to the loss of both lean and fat tissue in the body, and both types of tissue need to be replaced during recovery. Malnourished children are also likely to suffer from a deficiency of essential nutrients that constitute the building blocks of tissues and support growth. Correcting the deficiency and replacing essential nutrients in a balanced manner is vital(Reference Golden12). In CMAM, growth needs to occur at an accelerated rate within a short period of time. These principles that constitute the basis of treating acute malnutrition require implementing food products with high nutrient adequacy. Hence, examining the nutrient content of products used in nutritional rehabilitation is essential.
The importance of a food-based approach in treating acute malnutrition is well established(Reference Lelijveld, Beedle and Farhikhtah56,Reference Murray and Manary57) . However, the treatment of SAM or MAM in the community does not involve a single food model, and, despite the emphasis on using dairy protein to ensure growth, not all formulations of a given food model include dairy(58). Hence, this narrative review aimed to synthesise evidence on the nutrient density of different food models (animal and non-dairy/plant sources) in managing SAM and MAM cases. The evidence also suggests that the nutrient adequacy of different food products has significance in improving nutritional recovery from acute malnutrition.
Prevailing literature suggests that animal-source foods such as milk include constituents that improve the nutrient density of formulations and aid recovery from acute malnourishment. Milk is known to stimulate insulin-like growth factor-1 concentrations in children largely because of the casein fraction of milk protein, thereby also aiding linear growth, whereas the whey fraction of milk protein supports muscle protein accretion, increases absorption of essential minerals and improves the intestinal function and immune system(Reference Hoppe, Mølgaard and Juul59–Reference Layman, Lönnerdal and Fernstrom61). Milk sugars, that is, lactose, also improve the absorption of essential minerals such as calcium and have positive prebiotic effects(Reference Grenov, Briend and Sangild62). Lactose intolerance is a concern in the use of milk-based products for the management of acute malnutrition(63), but there is inadequate evidence to support the argument that the reduction of lactose content is necessarily advantageous(Reference Hoppe, Andersen and Jacobsen60). Although this narrative review has collated evidence favouring the inclusion of dairy in formulation for better recovery from malnutrition, ambiguity regarding a conclusive finding between dairy and non-dairy still exists. The varied percentage of dairy protein content in the formulations used in various studies is a limiting factor for the efficacy comparison.
Children recovering from acute malnutrition need to grow at an accelerated rate to catch up; hence, their nutrient requirements differ from those of normal children. Moreover, multiple deficiencies may be prevalent among acutely malnourished children, given that they have likely been consuming a diet deficient in important nutrients that are essential in maintaining health. To ensure recovery, formulations used in treating acute malnutrition need to contain adequate nutrients to ensure skeletal growth and deposit of lean tissue(Reference Golden12). Type I nutrients are a category of essential nutrients that largely have a ‘protective role’ as they support specific metabolic functions in the body, and their deficiency can impact growth if the deficiency results in a severe illness. However, type II or growth nutrients are crucial for the reversal of malnutrition as these nutrients are required by tissues for cellular division and growth(Reference Golden12,Reference Golden64) . In the current review, formulations that could not meet the recommended requirements of key type I and type II nutrients were unfortified, non-dairy, plant-based products.
Protein is an important type II nutrient essential to maintain and increase lean mass(Reference Moughan, Boland and Singh65). While the current study found that protein content requirements for SAM and MAM treatment were usually met by both dairy and non-dairy formulations, it is also important to consider that high-quality protein sources would contain an appropriate balance of indispensable amino acids that make new lean tissue(Reference Golden12). The importance of using high-quality protein sources, that is, digestible indispensable amino acid scores (DIAAS ≥75) in treating acute malnutrition, is reiterated in formulation guidelines(Reference Herreman, Nommensen and Pennings44). The WHO recommends that at least half of the proteins in RUTF used in treating SAM should come from milk products(19). Among the reviewed studies for SAM treatment in this study, this criterion is met mainly by standard RUTF formulations. Plant-based alternative RUTF formulations include a combination of low-quality plant protein sources (maize, maize or sorghum) as well as high-quality protein sources (soy)(Reference Herreman, Nommensen and Pennings44,Reference Bailey and Stein45) . WHO does not similarly stipulate milk content requirements in supplementary foods used for MAM rehabilitation but instead recommends protein to be derived from high-quality sources such as milk that have a high protein digestibility amino acid score(40).
Few studies that have examined the amino acid profile provide evidence of the importance of protein quality for recovery from SAM. A Malawi trial(Reference Sato, Furuta and Matsunaga20) found that treatment with the amino acid-enriched plant-based RUTF that were milk-free and low in milk could replenish plasma essential amino acids to levels comparable to those with the standard peanut-milk RUTF. Bahwere et al.(Reference Bahwere, Akomo and Mwale4) found that the recovery rates of the alternative RUTF were not inferior to the standard RUTF. In another study,(Reference Bahwere, Balaluka and Wells23) both the milk-free plant RUTF and the standard RUTF corrected amino acid deficiencies to a similar level, and neither RUTF was associated with excess fat deposition. However, the low content of certain amino acids was seen as a possible cause for the inferior response of the milk-free plant RUTF in terms of recovery among SAM children <2 years. Most plant-based foods are limited in one or more of the indispensable amino acids(Reference Herreman, Nommensen and Pennings44,Reference Mathai, Liu and Stein46) . Milk and milk-based products can complement and act as potential protein balancers for plant-based products as they supply the prominent limiting amino acid lysine and are also a good source of branch-chained amino acids (e.g. leucine) that regulate muscle protein synthesis(Reference Mathai, Liu and Stein46,Reference Moughan, Boland and Singh65) . Owing to its high protein quality and density in nutrients such as calcium, magnesium and available phosphorus, which have growth-promoting abilities, it may be suggested that the addition of milk to a fortified plant-based RUTF can further improve its nutrient content and aid in the recovery of SAM children(Reference Michaelsen, Hoppe and Roos66). One study in Malawi(Reference Oakley, Reinking and Sandige30) found that recovery rates were higher among SAM children who received the standard RUTF with 25% milk than the children who received the soy RUTF with 10% milk. However, the study observed that the plant-based formulation included soy that was not dehulled and contained antinutrients, which possibly resulted in its inferior response.
Studies suggest that a higher quantity of high-quality protein sources may be required for MAM rehabilitation. In a study in Malawi(Reference Roediger, Stein and Callaghan-Gillespie31), there was no difference in recovery rates of MAM between the treatment and control RUSF when the dairy protein quantity of the treatment RUSF was marginally higher, but the protein quality was inferior to the control RUSF. The two RUSF used whey permeate, but the treatment RUSF additionally used dry skimmed milk, whereas the control RUSF additionally used whey protein concentrate (WPC). Another study(Reference Stobaugh, Ryan and Kennedy5) showcased the importance of milk protein in treating MAM. The study found that a whey RUSF containing WPC-80 and whey permeate significantly improved nutritional recovery compared with the milk-free soy RUSF despite providing 33% less total protein and reporting a comparable DIAAS.
Evidence suggests that the nutritional composition of a formulation can be improved not only by including high-quality protein sources but also by fortification and processing. Fortification becomes essential, especially for plant-based formulations that rely less on animal-source foods such as milk to meet the recommended nutrient requirements. Maintaining a nutrient such as vitamin B-12 becomes crucial as it has a role in enterocyte turnover, protein formation from amino acids, transfer of methyl groups, and carbohydrate and fat metabolism(Reference Michaelsen, Hoppe and Roos66,Reference Friis, Cichon and Fabiansen67) . Another concern is that the plant-based formulations would also have low vitamin–mineral bioavailability. However, dehulling, extrusion and malting are standard processing methods that can be used to prepare the formulations for the treatment of both MAM and SAM(Reference Stobaugh, Ryan and Kennedy5,Reference Nikiema, Huybregts and Kolsteren8,Reference Bahwere, Balaluka and Wells23,Reference Kajjura, Veldman and Kassier29) . Although these processing methods may not necessarily improve the nutrient content of foods, they help to reduce the phytate content and thereby improve the bioavailability of nutrients(Reference De Pee and Bloem50,Reference Ghavidel and Prakash68) .
Sugar is generally added to formulations to improve the palatability of their products. This raises concerns about compromising nutrient quality as supplements provide ‘empty calories’ or calories with little or no related nutrients(Reference Louie, Moshtaghian and Boylan14). A study based on CSFII survey data observed that, when sugar exceeded 16% of the total energy intake, there was a significant reduction in the intake of key nutrients, including calcium and vitamin A, by preschool children(Reference Kranz, Smiciklas-Wright and Siega-Riz69). High consumption of empty calories that do not contain all nutrients in balance inhibits lean tissue synthesis and results in excess energy deposited as fat tissue, which contributes to the development of obesity. Such diets would not help malnourished children regain physiological normality even with rapid weight gain(Reference Golden12). Studies have found added sugar to be associated with adverse cardiometabolic health markers in children, such as elevated diastolic blood pressure, triacylglycerols, very low-density lipoprotein cholesterol and homeostasis model assessment of insulin resistance(Reference Kell, Cardel and Bohan Brown70,Reference Sharma, Roberts and Lustig71) . One study observed an increased risk of type 2 diabetes in adults with high added sugar intake at higher C-reactive protein levels(Reference Ramne, Drake and Ericson72). Lusting et al.(Reference Lustig73) observed that sugar causes chronic adverse long-term health effects similarly to alcohol, which also is why children get diseases such as type 2 diabetes that are related to alcohol without consuming alcohol. In a UK study(Reference Hong, Whelton and Douglas74), children who consumed drinks and food with added sugar more frequently were more likely to develop dental caries. Given the detrimental health impacts, public health agencies have recommended restrictions on added sugar intake(55). There is evidence that MD is not a superior source for improving the sweetness of a product. One rat-based model(Reference Kendig, Lin and Beilharz75) found that MD influences rapid weight gain similar to sucrose, but regular intake of MD is cautioned as it may likely result in impaired insulin sensitivity and increased blood cholesterol and blood lipids(Reference Hofman, Van Buul and Brouns53). The review also found that several products with reported effectiveness in increasing weight gain contain high amounts of added sugar, but sustenance of the increased gain, improvement in lean body mass and metabolic changes behind the rapid catch-up growth were not monitored.
The present narrative review has effectively provided a comprehensive summary of current evidence on the efficacy of dairy protein and nutrient adequacy in terms of density of type I and type II nutrients in the products used in managing SAM and MAM. The review has a few limitations. The search was limited to major databases and open-access documents before the pandemic, which may have omitted some relevant studies. Despite the limitations, this review goes beyond the scope of similar review studies that synthesise the effectiveness of products in managing SAM and MAM at the community level. It also assesses whether the different food product formulations are nutrient-dense and examines the significance of specific nutrient components in the formulation for its efficacy in correcting malnutrition among children in community settings.
Conclusion
Milk aids recovery, but recent evidence shows that even non-dairy food products can be potentially used to treat uncomplicated acute malnutrition (moderate or severe) at the community level. However, the comparison remains ambiguous due to the varying percentages of dairy protein used in different studies. Thus, more research with consistent dairy protein percentages is needed to draw a definitive conclusion. The review found that the nutrient content of non-dairy products can be improved by fortification and that these products are found to perform satisfactorily towards certain outcomes, such as correcting anaemia and improving haemoglobin, which could be secondary goals of nutritional interventions in low- and middle-income country settings.
The review also examines the importance of nutrient adequacy in ensuring product efficacy. The study highlights concerns about low essential nutrient adequacy and higher added sugar content, which also affect nutritional recovery from acute malnutrition. Products with high added sugar are likely to result in weight gain, possibly due to higher fat deposits instead of increases in lean body mass that may also have long-term health implications; hence, a reduction of added sugar content in formulation is necessary. More research examining the status of specific essential nutrients on recovery from uncomplicated malnutrition can aid the development of food products for CMAM. Given the use of different food models to manage uncomplicated SAM and MAM, there also needs to be a consensus on appropriate formulation combinations using various ingredients for each food model to meet prescribed norms. The findings of this review study potentially contribute to such an exercise.
Whereas the current study largely examines the efficacy of food products in terms of key nutritional outcomes such as recovery or weight gain, research recognises that comparing lean mass and fat mass increases would be a necessary indicator to appropriately assess body composition and also evaluate possible side effects of food products. However, given the limited number of studies in the current literature, high-quality research is needed to evaluate these clinical outcomes.
Acknowledgements
This work was initially conceptualised as part of a collaboration between IIT-Bombay and CFNS. The authors would like to thank Meeta Mathur for her input on the review plan and initial version of the synthesis and also for providing the nutrient composition of a few products. We also thank Dr. Rupal Dalal for her feedback on the initial version of the synthesis.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
None.
Authorship
Review planning: D.R.C. and M.A.; literature search, data extraction and synthesis, and original draft (writing): M.A.; review: D.R.C. and S.B.A.; editing: D.R.C. and M.A. All authors approved the final version of the manuscript.