Introduction
Basidiomycete lichen fungi are well represented in the Americas, particularly in the Neotropics. This region is considered a biodiversity hotspot with high endemism rates for various genera, such as Acantholichen, Cora, Cyphellostereum and Dictyonema (Hygrophoraceae, Agaricales; Jørgensen Reference Jørgensen1998; Lücking et al. Reference Lücking, Barrie and Genney2014a, Reference Lücking, Thorn, Saar, Piercey-Normore, Moncada, Doering, Mann, Lebeuf, Voitk and Voitk2017; Dal Forno et al. Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016, Reference Dal Forno, Bungartz, Yánez-Ayabaca, Lücking and Lawrey2017; Moncada et al. Reference Moncada, Pérez-Pérez and Lücking2019; Marcano Reference Marcano2022), and Lepidostroma and Sulzbacheromyces (Lepidostromataceae, Lepidostromatales; Mägdefrau & Winkler Reference Mägdefrau and Winkler1967; Hodkinson et al. Reference Hodkinson, Moncada and Lücking2013; Sulzbacher et al. Reference Sulzbacher, Baseia, Lücking, Parnmen and Moncada2012; Coca et al. Reference Coca, Lücking and Moncada2018, Reference Coca, Gómez, Guzman-Guillermo, Trujillo, Clavijo, Zuluaga, Dal Forno and Lumbsch2023). In contrast, the Nearctic portion of North America, in the sense of Escalante et al. (Reference Escalante, Rodríguez-Tapia and Morrone2021), has distinctly fewer species. Some basidiolichens, such as Bryoclavula dryalisepiplutea T. S. Quedensley & T. J. Widhelm, Multiclavula (Cantharelalles) and Lichenomphalia (Agaricales), are well represented at higher latitudes of the Northern Hemisphere (Petersen Reference Petersen1967; Geml et al. Reference Geml, Kauff, Brochmann, Lutzoni, Laursen, Redhead and Taylor2012; Lücking et al. Reference Lücking, Thorn, Saar, Piercey-Normore, Moncada, Doering, Mann, Lebeuf, Voitk and Voitk2017; Quedensley et al. Reference Quedensley, Widhelm, Distefano and Wade2024; Malíček et al. Reference Malíček, Janošík, Mikulášková and Palice2025; van der Kolk & de Winkel Reference van der Kolk and de Winkel2025), while most other Nearctic species are restricted to warm temperate to subtropical regions of North America, such as Lepidostroma vilgalysii B. P. Hodk., L. winklerianum B. P. Hodk. & Lücking (Lepidostromatales; Hodkinson et al. Reference Hodkinson, Uehling and Smith2012, Reference Hodkinson, Moncada and Lücking2013) and Sulzbacheromyces leucodontium Coca et al. (Coca et al. Reference Coca, Gómez, Guzman-Guillermo, Trujillo, Clavijo, Zuluaga, Dal Forno and Lumbsch2023). In the case of the genera Cora, Cyphellostereum and Dictyonema, species distributions are centred around the south-eastern USA, with Cora records in northern Mexico (Lücking et al. Reference Lücking, Dal Forno, Lawrey, Bungartz, Rojas, Hernández, Marcelli, Moncada, Morales and Nelsen2013, Reference Lücking, Kaminsky, Perlmutter, Lawrey and Dal Forno2020; Dal Forno et al. Reference Dal Forno, Kaminsky, Rosentreter, McMullin, Aptroot and Lücking2019; Moncada et al. Reference Moncada, Pérez-Pérez and Lücking2019; A. Huereca, unpublished data). Of the genera mentioned above, Acantholichen is the only one to date not reported from North America.
Fuscopannaria frullaniae (Maass) E. Tripp & Lendemer (syn. Moelleropsis nebulosa subsp. frullaniae Maass; Pannariaceae, Lecanoromycetes) is a species of crustose lichen with a granular, dark, blue-green thallus overgrowing the epiphytic liverwort Frullania asagrayana, especially in epiphytic situations, in Atlantic Canada (Maass Reference Maass1986). Originally described in the genus Moelleropsis as a subspecies of M. nebulosa (Hoffm.) Gyeln. (now considered part of Fuscopannaria; Tripp & Lendemer Reference Tripp and Lendemer2019), the taxon was not reported again until populations were found in Europe on the Iberian Peninsula and in Macaronesia (Jørgensen Reference Jørgensen2000a; Carballal Durán & López de Silanes Vázquez Reference Carballal-Durán and López de Silanes Vázquez2006; Pérez-Vargas et al. Reference Pérez-Vargas, Hernández Padrón and Losada-Lima2014). Since its ecology differs from that of the species then known as Moelleropsis nebulosa s. str. (=Fuscopannaria nebulosa (Hoffm.) E. Tripp & Lendemer; Tripp & Lendemer Reference Tripp and Lendemer2019), Jørgensen (Reference Jørgensen2000a) suspected that the taxon probably belongs to a different genus, suggesting Parmeliella, and noted the need for fertile material or DNA. In North America, F. frullaniae was originally thought to be restricted to Canada; however, it was later reported from the eastern USA (North Carolina) by Tripp & Lendemer (Reference Tripp and Lendemer2019). Notably, all reported specimens appeared to be sterile.
Ekman et al. (Reference Ekman, Wedin, Lindblom and Jørgensen2014), in their phylogenetic revision of Pannariaceae, showed that the type species of Moelleropsis, M. nebulosa, was nested within Fuscopannaria (the only other species that had been placed in the genus, Moelleropsis humida, had already been found to belong elsewhere; Lumbsch et al. Reference Lumbsch, Del Prado and Kantvilas2005). However, Ekman et al. (Reference Ekman, Wedin, Lindblom and Jørgensen2014) did not undertake any formal changes pending a proposal to the nomenclature committee on the conservation of the widely used Fuscopannaria over Moelleropsis, which is the older name (Jørgensen et al. Reference Jørgensen, Ekman and Wedin2013). In anticipation of the decision by that committee to conserve Fuscopannaria, Lendemer & Tripp (Reference Tripp and Lendemer2019), citing the phylogenetic results of Ekman et al. (Reference Ekman, Wedin, Lindblom and Jørgensen2014), combined the two remaining specific or subspecific taxa in Moelleropsis (M. nebulosa and M. nebulosa subsp. frullaniae) into Fuscopannaria and elevated the latter to species level ‘because this taxon appears to be consistently sterile and is now known from three geographically disparate locations’. However, no DNA sequences of F. frullaniae were produced at the time.
Fuscopannaria frullaniae typically grows on bryophytes over trees or rocks, or rarely directly on bark, in perhumid temperate coastal swamps and wet forests of eastern North America but it is rare throughout its range. It is a species of conservation concern in Canada, considered a high-priority candidate for assessment by the Committee on the Status of Endangered Wildlife in Canada (COSEWIC 2025). This species co-occurs with several other rare and at-risk lichens, including Erioderma pedicellatum (Hue) P. M. Jørg., a red-listed lichen assessed as critically endangered worldwide (IUCN: CR; Scheidegger Reference Scheidegger2003), with the Newfoundland and Nova Scotia populations in Canada ranked as ‘Special Concern’ and ‘Endangered’, respectively (COSEWIC 2014). Threats to these rare lichens include habitat destruction, airborne pollution, overgrazing by exotic gastropods, and increasingly, climate change (COSEWIC 2014, 2019; Haughian et al. Reference Haughian, Gray and Harper2022; Rinas et al. Reference Rinas, Haughian and Harper2025).
As part of a broader assessment of several Pannariaceae in North America, we produced sequences from herbarium specimens of Fuscopannaria frullaniae from Nova Scotia, Canada, and North Carolina, USA. During preliminary attempts to generate sequence alignments, it became clear that they could not be easily aligned, not only with Pannariaceae but with any Ascomycota. BLASTn queries revealed a strong affinity with the neotropical basidiomycete lichen fungus Acantholichen (Hygrophoraceae, Basidiomycota). Incorporation of these sequences into a phylogenetic tree with members of the Dictyonema clade of Hygrophoraceae revealed that sequences obtained from multiple specimens of F. frullaniae were closely related to fungi from the basidiolichen genus Acantholichen. In this study, we discuss the phylogenetic position of Fuscopannaria frullaniae and introduce a new combination for it in the genus Acantholichen based on both molecular and morphological data.
Materials and Methods
Material used
We studied herbarium material deposited at the Nova Scotia Museum of Natural History (NSPM), the New York Botanical Garden (NY), and the University of Colorado Museum of Natural History (COLO).
DNA extraction and sequencing
Genomic DNA was isolated from lichen fragments c. 1 mm2, then freeze-dried at −80 °C in an ultra-low temperature freezer and pulverized using a TissueLyser II (Qiagen, Germany). Extractions were performed using the DNeasy Investigator Extraction Kit (Qiagen, Germany) according to the manufacturer’s protocol. For polymerase chain reactions (PCR), 2 μl of genomic DNA was used in a total of 22 μl master mix using KAPA3G Plant Kit (Millipore, Sigma Aldrich, Munich, Germany) following the manufacturer’s protocol. PCR reactions were performed using standard fungal primers for the internal transcribed spacer region (ITS, using primers ITS1F and ITS4; White et al. Reference White, Bruns, Lee, Taylor, Innis, Gelfand, Sninsky and White1990), the large (28S) nuc rRNA subunit (LR0R and LR7; Vilgalys & Hester Reference Vilgalys and Hester1990), and the RPB2 protein-coding gene (primers fRPB2-7cF and fRPB2-11aR; Liu et al. Reference Liu, Whelen and Hall1999; Liu & Hall Reference Liu and Hall2004). We also targeted the associated cyanobacterial photobiont and amplified the partial 16S region of ribosomal rRNA, using the primers 27F and 1492R (Weisburg et al. Reference Weisburg, Barns, Pelletier and Lane1991). To confirm successful amplification, 2 μl of the PCR reaction was visualized on a 1% agarose gel stained with GelRed. Positive reactions were subsequently cleaned up with Exonuclease I and Shrimp Alkaline Phosphatase enzymes (New England BioLabs, Inc., Ipswich, Massachusetts, USA). Sanger sequencing was carried out by Psomagen (Jamaica, New York, USA). Sequence quality was assessed by inspecting the electropherogram, trimming the primer ends, and checking for ambiguities.
Phylogenetic analysis
Our initial screening of the Nova Scotia material involved the ITS region. Since our initial BLASTn results indicated a close relationship to lichen fungi in the Hygrophoraceae, we attempted to produce sequences for the only other widely sampled loci in this group to date, 28S and RPB2. We then pooled sequence data from multiple previous studies (Table 1) into a new analysis. This dataset included a broader sampling of genera, including all five belonging to the subtribe Dictyonematinae in Hygrophoraceae (Acantholichen, Cora, Corella, Cyphellostereum and Dictyonema), with sequences produced in Dal Forno et al. (Reference Dal Forno, Lawrey, Sikaroodi, Bhattarai, Gillevet, Sulzbacher and Lücking2013, Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016, Reference Dal Forno, Bungartz, Yánez-Ayabaca, Lücking and Lawrey2017, Reference Dal Forno, Lawrey, Moncada, Bungartz, Grube, Schuettpelz and Lücking2022), Lücking et al. (Reference Lücking, Dal Forno, Lawrey, Bungartz, Rojas, Hernández, Marcelli, Moncada, Morales and Nelsen2013, Reference Lücking, Dal Forno, Sikaroodi, Gillevet, Bungartz, Moncada, Yánez-Ayabaca, Chaves, Coca and Lawrey2014b) and Lawrey et al. (Reference Lawrey, Lücking, Sipman, Chaves, Redhead, Bungartz, Sikaroodi and Gillevet2009), using Eonema pyriforme as an outgroup. For the Rhizonema phylogeny, we assembled a data matrix based on a slightly reduced 16S rRNA dataset employed by Dal Forno et al. (Reference Dal Forno, Lawrey, Sikaroodi, Gillevet, Schuettpelz and Lücking2021).
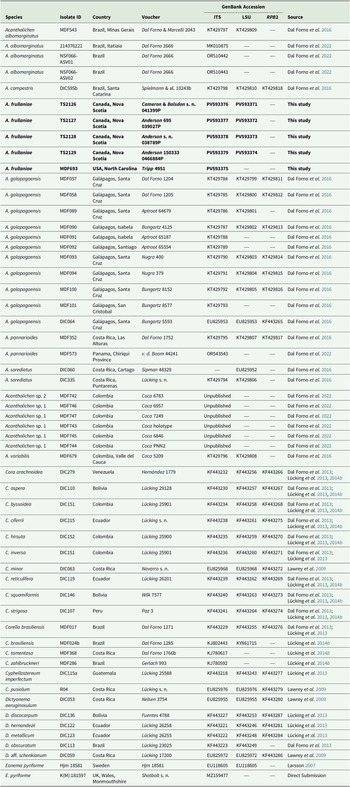
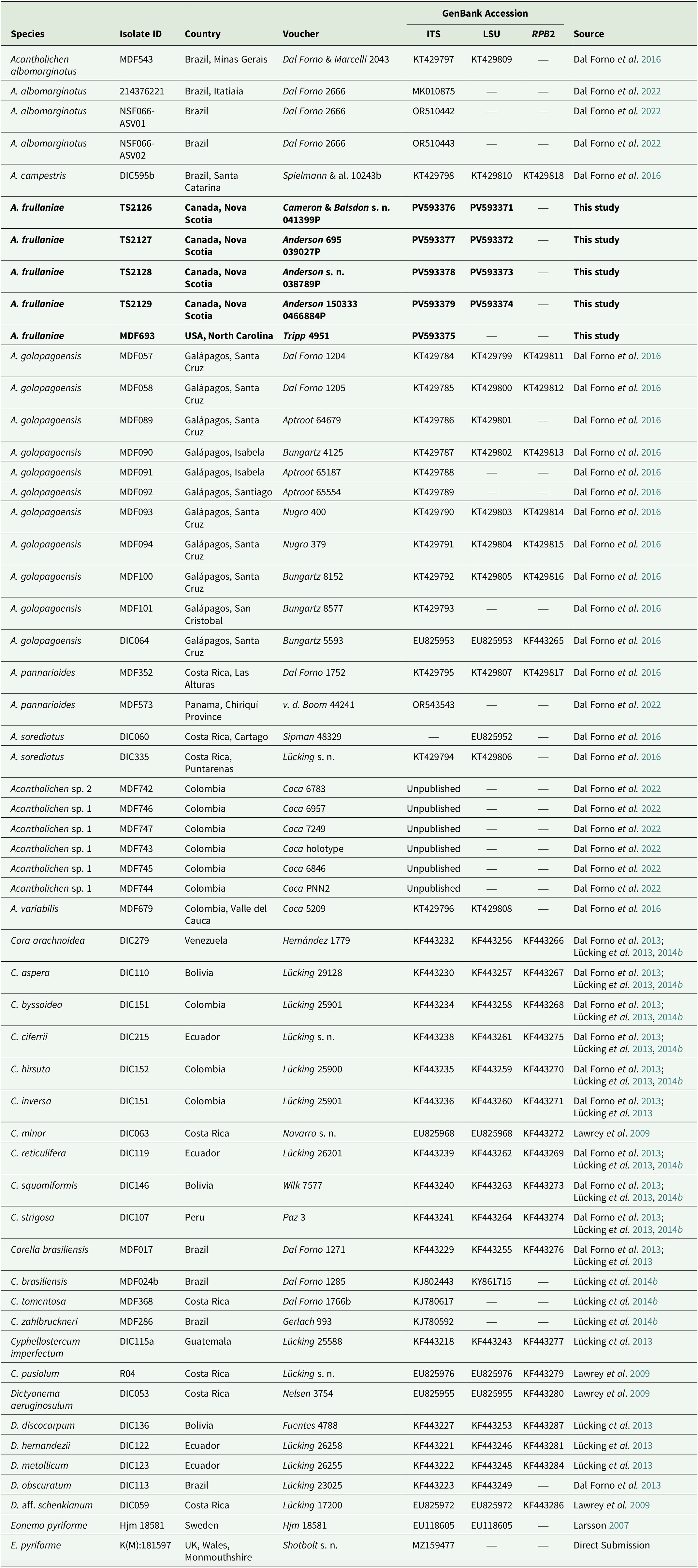
Voucher information of species and GenBank Accession numbers for sequences used in the multilocus phylogenetic reconstruction of the genera in the Dictyonema clade in Hygrophoraceae using Eonema pyriforme as an outgroup (Fig. 1). New sequences are highlighted in bold.
We aligned, trimmed and concatenated the fungal dataset using a custom Python script developed by Resl (Reference Resl, Schneider, Westberg, Printzen, Palice, Thor, Fryday, Mayrhofer and Spribille2015), as implemented in Resl et al. (Reference Resl2015). The script aligns the dataset using MAFFT v. 7 (Katoh & Standley Reference Katoh and Standley2013), then trims using trimAl v. 1.2 (Capella-Gutiérrez et al. Reference Capella-Gutiérrez, Silla-Martínez and Gabaldón2009) using the ‘—gappyout’ flag based on gap distribution. A maximum likelihood analysis was performed for both data matrices in IQ-TREE v. 2.2.0 (Nguyen et al. Reference Nguyen, Schmidt, von Haeseler and Minh2015) with the partitioning flag ‘-p’ to evaluate evolutionary models for each gene. The tree was created using ultrafast bootstrapping of 1000 replicates to estimate branch support (Hoang et al. Reference Hoang, Chernomor, von Haeseler, Minh and Vinh2018), in addition to executing the Shimodaira-Hasegawa-like approximate likelihood-ratio test (SH-aLRT) with 1000 replicates (Guindon et al. Reference Guindon, Dufayard, Lefort, Anisimova, Hordijk and Gascuel2010). Only branches with ≥ 95% ultrafast bootstrap values and ≥ 80% SH-aLRT were considered well supported (Nguyen et al. Reference Nguyen, Schmidt, von Haeseler and Minh2015). The phylogeny was inferred based on the best-fitting model using ModelFinder (Kalyaanamoorthy et al. Reference Kalyaanamoorthy, Minh, Wong, von Haeseler and Jermiin2017) and the bootstrap algorithm UFBoot (Minh et al. Reference Minh, Nguyen and von Haeseler2013). The single most-likely phylogenetic tree resulting from the analysis was visualized using FigTree v. 1.4.4 (http://tree.bio.ed.ac.uk/software/figtree) and edited with Adobe Illustrator v. 27.5.
Microscopy
Morphology was studied using an Olympus SZX16 dissecting scope (Shinjuku, Japan), and macroscopic photographs were taken with an Olympus SC180 digital camera (Shinjuku, Japan). For microscopic examination, pieces of the thallus were mounted in water and squashed, and/or followed by application of 5% KOH, and/or stained with phloxine B, then studied using a Zeiss Axioscope A1 (Jena, Germany) compound microscope, equipped with an Olympus SC180 digital camera (Shinjuku, Japan).
Scanning electron microscopy (SEM) was used to enhance the visualization of cell-cell interactions in the thallus. For this, we selected sections of the thallus to include both young marginal areas and old granules near the centre. The samples were subjected to critical-point drying after fixing the material with an osmium tetroxide solution following the protocol described in Huereca et al. (Reference Huereca, Allen, McMullin, Simon, Belosokhov and Spribille2025). Specimens were then mounted on aluminium stubs with carbon adhesive tape and finally coated with gold or carbon. Examination of the samples was performed using a field-emission scanning electron microscope, model Zeiss Sigma 300 VP (Zeiss Group, Oberkochen, Germany), at the Department of Earth and Atmospheric Sciences of the University of Alberta.
Results and Discussion
A total of nine new fungal sequences were generated for this study (Table 1), comprising four 28S and five ITS sequences, all from Fuscopannaria frullaniae from eastern North America. We were unsuccessful in obtaining RPB2 for the newly extracted specimens. In the dataset of the Dictyonema clade in the Hygrophoraceae dataset, the concatenated alignment consisted of three loci of 56 taxa with 3121 characters after trimming (ITS: 773; LSU: 1347; RPB2: 1001), of which 2283 were invariable sites, 1010 were variable sites, and 577 were parsimony-informative. The best-fit models per gene were: Ime + I + G4 for ITS, TNe + I + I + R2 for LSU, and K2P + G4 for RPB2. The resulting maximum likelihood phylogenetic tree using the concatenated dataset of the Dictyonematinae subtribe (Fig. 1) had a log likelihood of −14975.14 and recovered Acantholichen as sister to Corella within the Hygrophoraceae, consistent with previous phylogenetic studies (Dal Forno et al. Reference Dal Forno, Lawrey, Sikaroodi, Bhattarai, Gillevet, Sulzbacher and Lücking2013, Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016, Reference Dal Forno, Bungartz, Yánez-Ayabaca, Lücking and Lawrey2017). As in earlier analyses, Dictyonema remains paraphyletic, while Acantholichen, Cora and Corella form a well-supported monophyletic group (Fig. 1). However, the sibling group relationship of Corella and Acantholichen is not supported given the current taxon and locus dataset.
Maximum likelihood phylogenetic tree inferred using three loci (ITS, 28S, RPB2), including all genera in the Dictyonema clade plus Eonema pyriformis as an outgroup (Table 1). Only values in branches with SH-aLRT support ≥ 80% and/or ultrafast bootstrap support ≥ 95% are shown. Labels in bold indicate sequences produced for this study. In colour online.
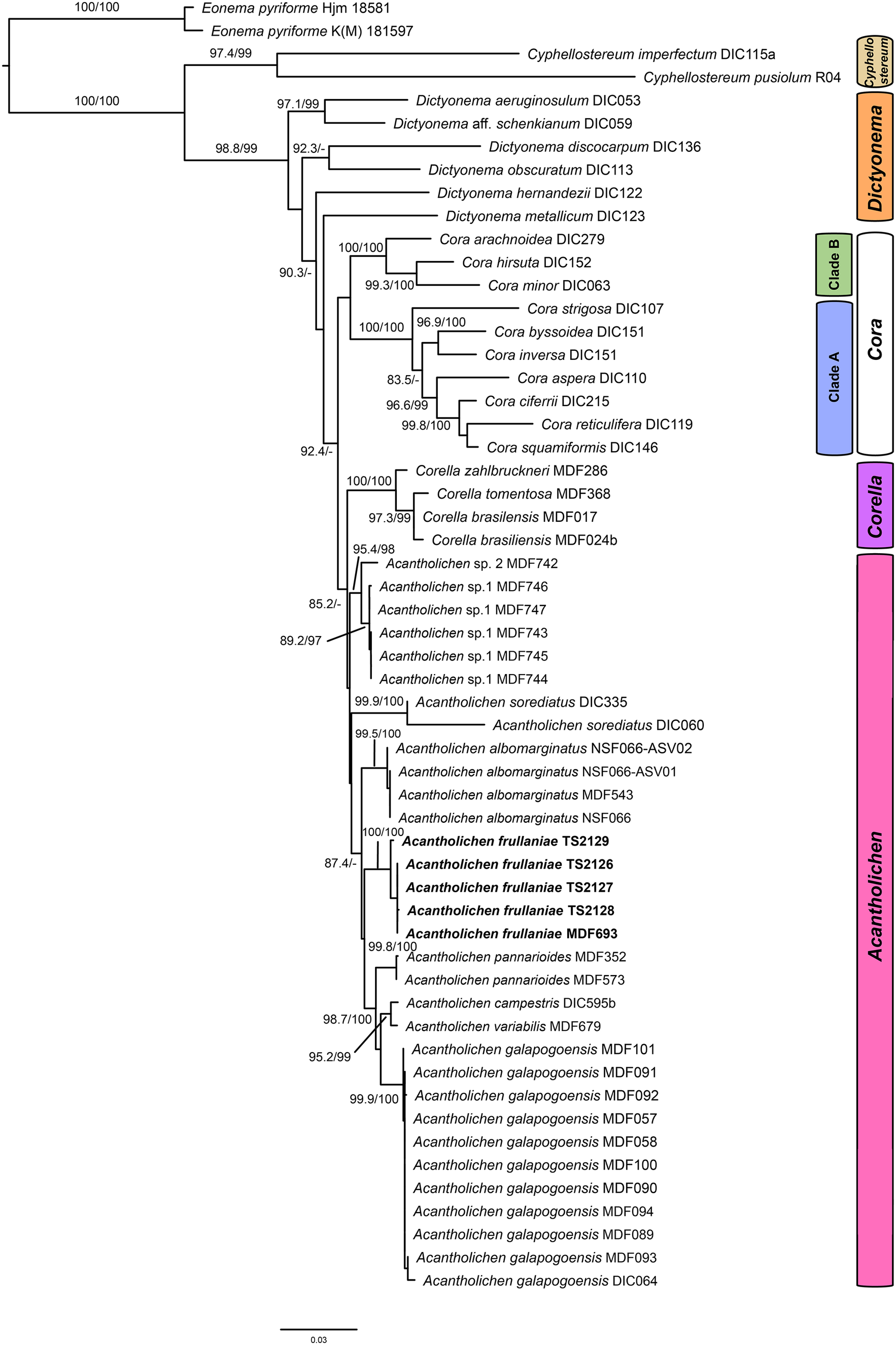
Our topology recovers Acantholichen sp. 1, which was treated by Dal Forno et al. (Reference Dal Forno, Lawrey, Moncada, Bungartz, Grube, Schuettpelz and Lücking2022), as sister to all other species in the genus, but with low branch support. This species is represented by ITS sequences only. This and the sparse locus sample for several other taxa could account for the low branch support at several nodes. The analysis recovers A. albomarginatus Dal Forno et al. as sibling to A. frullaniae and the remaining species of the genus, with 87.4% ultrafast bootstrap support. In the remainder of the topology, A. frullaniae is sibling to a clade that includes A. campestris Dal Forno et al., A. galapagoensis Dal Forno et al., A. pannarioides P. M. Jørg. and A. variabilis Dal Forno et al., with low UF bootstrap/aLRT support (74/95). Given the phylogenetic support in the phylogeny, we propose to combine Fuscopannaria frullaniae into the genus Acantholichen.
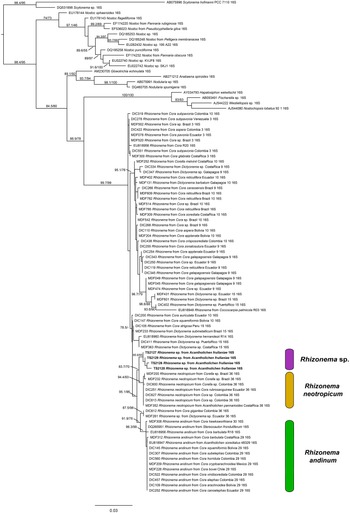
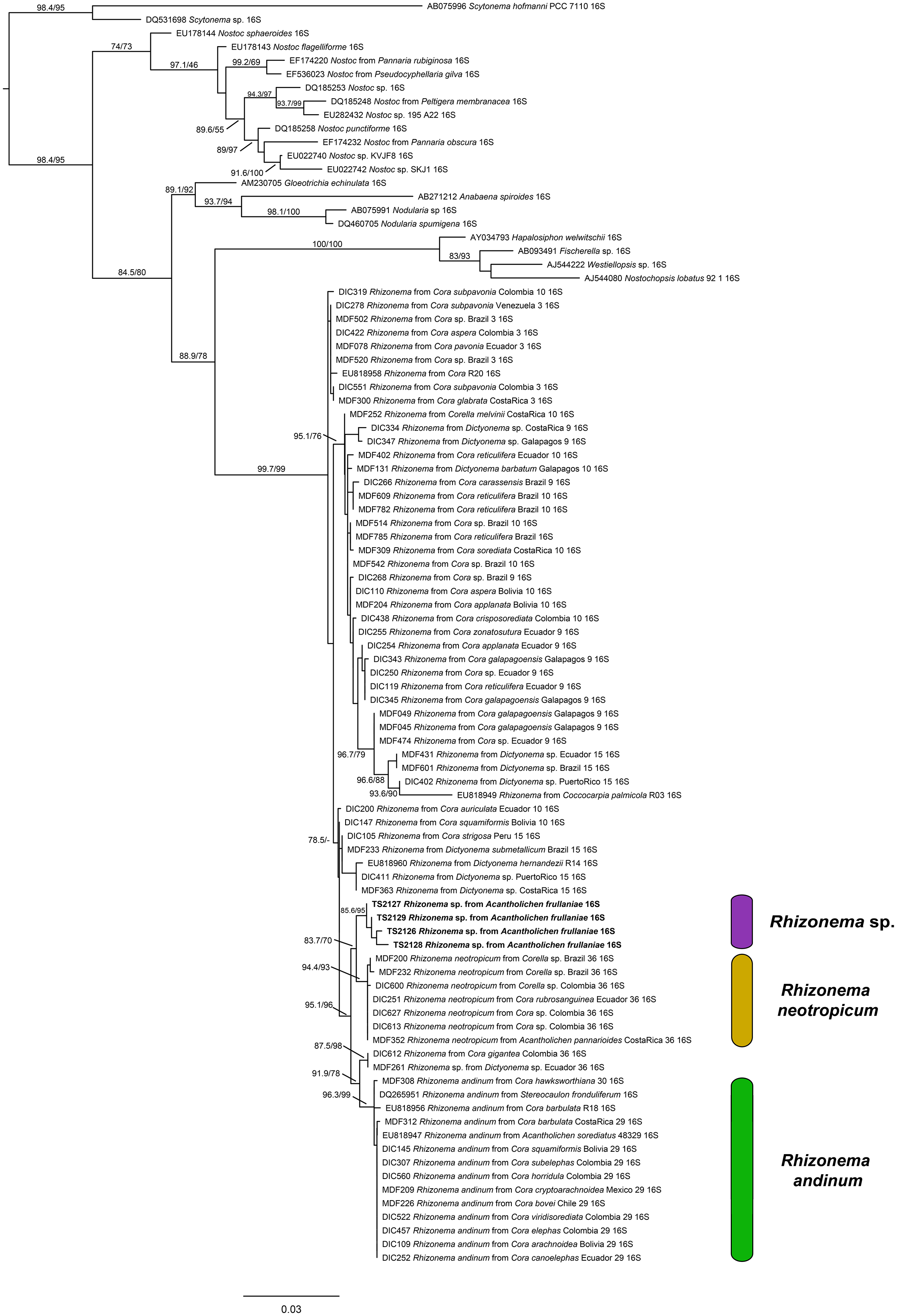
For the Rhizonema phylogeny, we generated four sequences for the bacterial 16S region, targeting the Rhizonema photobiont from the four Acantholichen frullaniae specimens from Atlantic Canada. The dataset of this alignment consisted of one locus of 93 taxa with 1396 characters, of which 1156 were invariable sites, 373 were variables, and 182 were parsimony-informative. The best-fit model for 16S was TIM3 + F + I + G4. The resulting maximum likelihood phylogenetic tree using the dataset of Rhizonema had a log likelihood of −5509.265 and recovered the Rhizonema associated with Acantholichen frullaniae as sister to Rhizonema neotropicum (Fig. 2). This clade was recovered with high support within the R. neotropicum-andinum clade, which represents species less common than those belonging to the Rhizonema interruptum complex.
(previous page) Maximum likelihood phylogenetic tree inferred using 16S rRNA gene sequences showing relationships among Rhizonema species based on a reduced dataset from Dal Forno et al. (Reference Dal Forno, Lawrey, Sikaroodi, Gillevet, Schuettpelz and Lücking2021), using Scytonema as outgroup. Only values in branches with SH-aLRT support ≥ 70% and/or ultrafast bootstrap support ≥ 95% are shown. Labels in bold indicate sequences produced for this study. In colour online.
Taxonomy
Acantholichen frullaniae (Maass) Huereca, Dal Forno & T. Sprib. comb. nov
MycoBank No.: MB 860347
Moelleropsis nebulosa subsp. frullaniae Maass, Proc. N. S. Inst. Sci. 37, 33 (1986); type: Canada, Newfoundland & Labrador, Newfoundland, Jipujijkuei Kuespem Provincial Park, Hwy 360, 1.8 km N of entrance to park, 29 June 1980, B. L. Hoisington & W. S. G. Maass et al. s. n. (CANL—holotype, not seen), MycoBank MB 133689.—Fuscopannaria frullaniae (Maass) E. Tripp & Lendemer, Syst. Bot. 44, 968 (2019), MycoBank MB 825250.
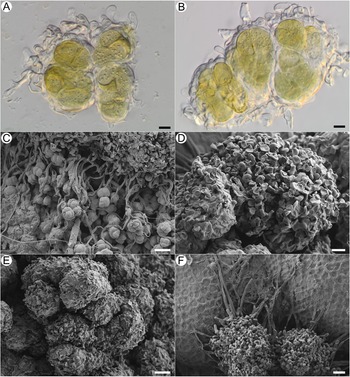
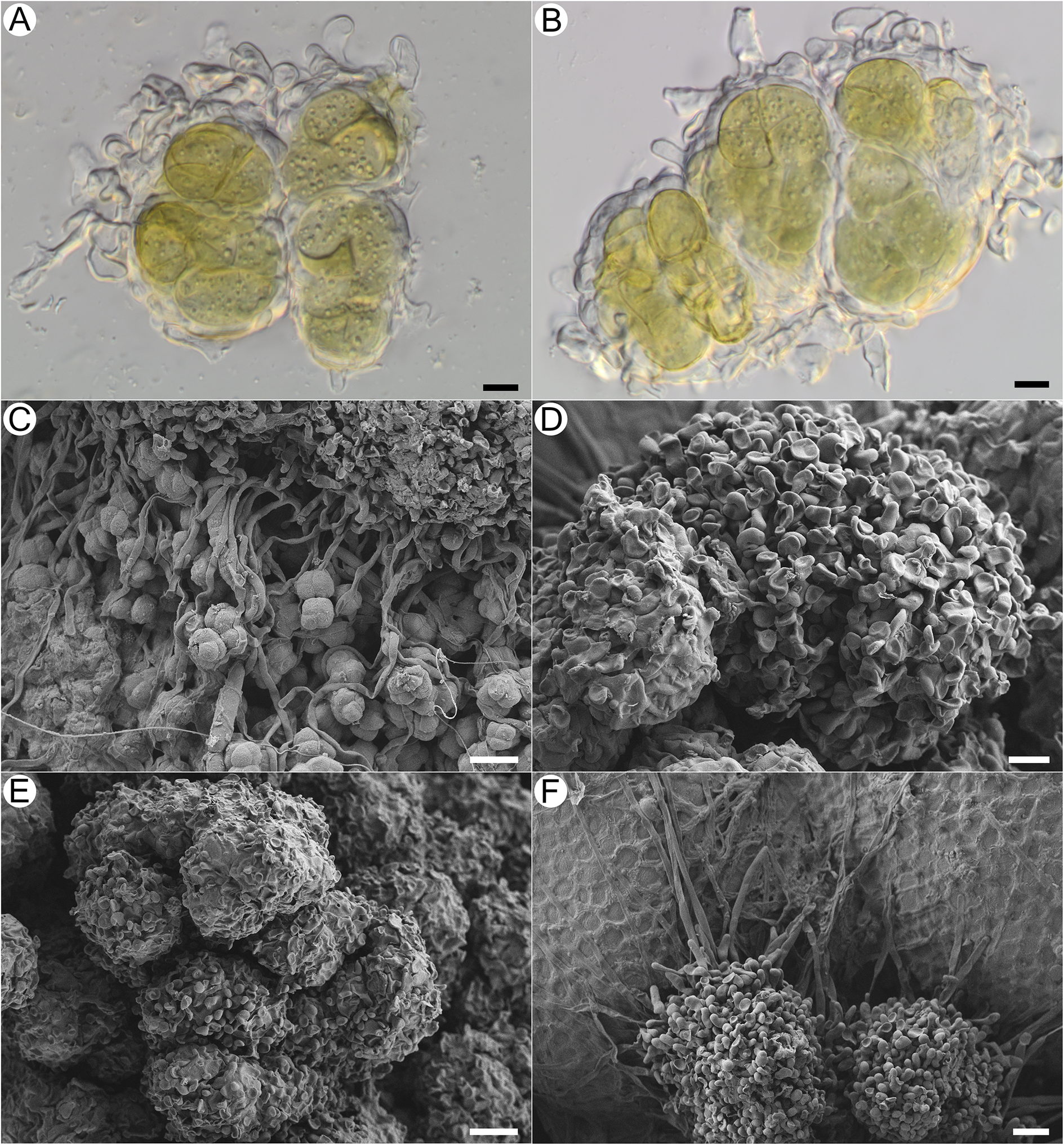
Habitus of Acantholichen frullaniae in different substrata. A & B, growing over Frullania asagrayana (041399P). C & D, growing on Abies balsamea bark with adjacent F. asagrayana (038789P); D, detail of the granules and hypothallus. E & F, growing on Hedwigia filiformis (Tripp 4951). Scales: A, C & E = 1 mm; B & F = 0.5 mm; D = 100 μm. In colour online.

Thallus crustose, dark blue with a frosted appearance when dry and blue-green when hydrated. Overall, thallus with a uniform white pruinose appearance; composed of conglutinated granules, 35–80 μm diam., forming clusters 110–160 μm diam., broadly adnate, not free and detaching (i.e. not becoming soredia). Granules consisting of wrapped, dense bundles of photobiont ensheathed in thin-walled, hyaline fungal cells that form jigsaw or jigsaw-like patterns (Fig. 5A & B; sensu Chaves et al. Reference Chaves, Lücking, Sipman, Umaña and Navarro2004; Lücking et al. Reference Lücking, Barrie and Genney2014a), with hyphae in these areas often slightly to strongly swollen and notably thin-walled, c. 8.2–11.1 × 5.4–6.5 μm, wrapping granules with knob-like thickenings at the tip (cystidia? Fig. 4A, B, D & E). Granules attached basally to the substratum with narrow hyaline hyphae, these septate, each cell 18.4–56.5 × 3–4.8 μm, with thicker walls than those that ensheathe the photobionts, and long, straight stretches of hyphae forming a kind of mat, the hyphae sometimes anastomosing (Fig. 4C & D). Clamp connections not observed. Photobiont Rhizonema sp., identified based on 16S rDNA from isolates TS2126 (PV683340), TS2127 (PV683341), TS2128 (PV683342) and TS2129 (PV683343); 3–9 cells visible per granule, often densely clustered, chain-like structures visible only in KOH, individual cells 11–22 μm diam. (Fig. 4A & B).
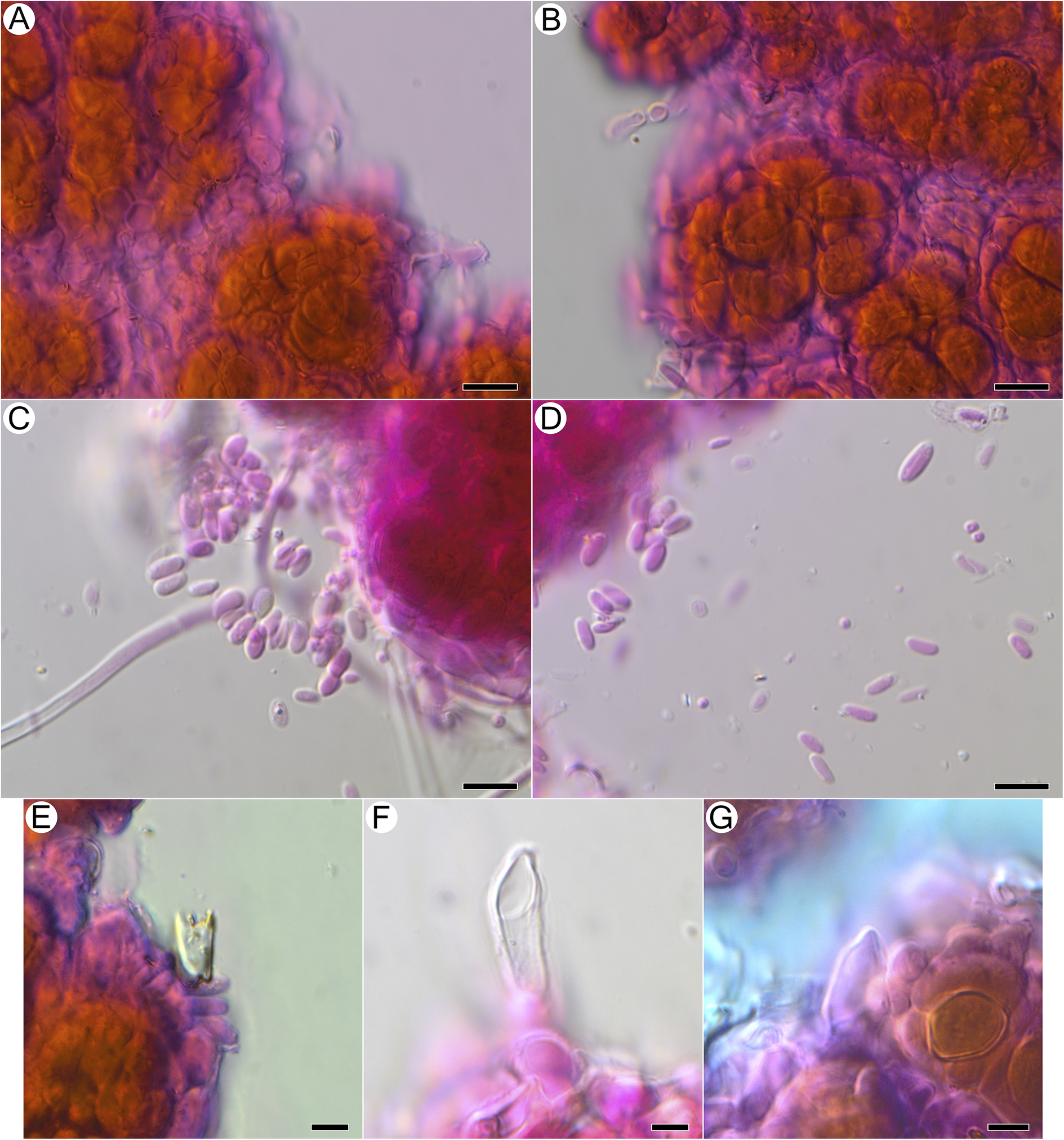
Microscopical features and granule morphology of Acantholichen frullaniae using light microscopy and SEM. A & B, granules; hyphae surround photobiont rounded cells in irregular clusters with terminal lobulate, knob-like hyphae present. C, hyphae starting to surround photobiont cells. D, granule with lobulate, knob-like hyphal tips. E, older granule at the centre of the thallus. F, young granule at the thallus margins, adhering to a Frullania leaf. Scales: A, B & D = 10 μm; C, E & F = 20 μm. In colour online.
Basidiomata not observed as differentiated hymenophores, but putative basidiospores and basidia observed, detected only after treatment with KOH and subsequent staining with phloxine, arising from the surface of granules in close proximity to photobiont cells, with attached basidiospores seen singly or in groups of two or four, or detached and aggregated between granules; putative basidia 10.5–20.0 × 4.2–6.3 μm (n = 6), balloon-shaped or pointed at tip, found in vicinity of swarms of putative basidiospores, no sterigmata observed with confidence (Fig. 5E–G); basidiospores ellipsoid, (5.3–)5.7–7.3(–8.6) × (2.4–)2.5–3.7(–4.6) μm (n = 26) (Fig. 5C & D).
Microscopical features of Acantholichen frullaniae after KOH and staining with phloxine B, using light microscopy. A & B, hyphae wrapping photobiont cells in jigsaw-like pattern. C & D, putative basidiospores released from granules after KOH treatment. E–G, putative basidia arising from granules after KOH treatment. Scales: A–D = 10 μm; E–G = 5 μm. In colour online.
Secondary metabolites
TLC not performed for this study, but Maass (Reference Maass1986), using both TLC and mass spectrometry, did not detect any secondary metabolites. European reports of Fuscopannaria frullaniae also did not yield any secondary metabolites (Jørgensen Reference Jørgensen2000a; Pérez-Vargas et al. Reference Pérez-Vargas, Hernández Padrón and Losada-Lima2014).
A complete description of a specimen from the eastern USA is provided by Tripp & Lendemer (Reference Tripp and Lendemer2019). Pérez-Vargas et al. (Reference Pérez-Vargas, Hernández Padrón and Losada-Lima2014) provided a description of a specimen from the Canary Islands.
Material examined
Canada: Nova Scotia: Guysborough County, near Forest Hill, 45.28602, −61.70455, on Frullania sp. on Abies balsamea in treed swamp, 19 xi 2009, Frances Anderson 150333 (NSPM 046884P), DNA isolate TS2129; Lunenburg County, Aspotogan Peninsula, below Deep Cove Mtn, 44.52821, −64.08743, on Frullania sp. on Abies balsamea next to brook, 13 xii 2006, Frances Anderson 695 (NSPM 039027P), DNA isolate TS2127; Halifax County, near Webber Lake, 44.82852, −63.00953, on Abies balsamea trunk, adjacent to Frullania sp. near brook crossing, 25 xi 2006, Frances Anderson s. n. (NSPM 038789P), DNA isolate TS2128; Moose River Gold Mines, 44.97689, −62.92232, on Frullania sp. on Abies balsamea in moist coniferous forest, 25 viii 2008, B. Cameron & J. Balsdon s. n. (NSPM 041399P), DNA isolate TS2126.—USA: North Carolina: Haywood Co., Great Smoky Mountains National Park, West side of Purchase Rd, c. 0.3 mi SE of Appalachian Highlands Science Learning Center (‘Purchase Knob’), 35.58081 −83.06332, 1265 m elev., on mosses over vertical face of rock in a dense deciduous forest and deeply shaded rock, 25 ix 2014, E. Tripp & J. Lendemer 4951 (COLO, NY), DNA isolate MDF693; Haywood Co., Great Smoky Mountains National Park, Appalachian Trail 0–1.1 mi E of Laurel Gap, 35.6631 −83.3242, 1788 m elev., on bryophytes over exposed rock outcrops in mature mixed montane forest, 02 viii 2013, J. Lendemer 37884 (NY).
Taxonomic remarks
Acantholichen frullaniae represents an enigma in the genus on multiple accounts. It differs from all other species in the absence of acanthohyphidia, previously thought to be characteristic for the genus (Jørgensen Reference Jørgensen1998; Dal Forno et al. Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016; Marcano Reference Marcano2022). However, A. frullaniae does possess some suspiciously similar cellular features in place of acanthohyphidia. When Maass (Reference Maass1986) described A. frullaniae, at the time as a subspecies of Moelleropsis nebulosa, he noted that it differed from the typical subspecies by the presence of ‘knob-like enlargements of the terminal ends of the hyphae’ (figs 8 & 10 in Maass (Reference Maass1986)). Maass (Reference Maass1986) interpreted smaller hyphal ends in F. nebulosa (fig. 9 in Maass (Reference Maass1986)) to be simply smaller ‘lobules’ than those found in what we are now calling A. frullaniae, but there is reason to suspect these structures are not, in fact, homologous. The structure of the granules of F. nebulosa (and the superficially similar Gregorella humida (Kullh.) Lumbsch) was also investigated by Vondrák et al. (Reference Vondrák, Palice, Mares and Kocourková2013), who found that the granules are formed by short hyphae, only partly covering mucilaginous envelopes of Nostoc. Notably, they did not mention any thickening of terminal hyphae, and their micrographs do not depict any lobulate hyphae (Vondrák et al. Reference Vondrák, Palice, Mares and Kocourková2013, fig. 3B & D).
The position of the enlarged cells in A. frullaniae takes on new significance when seen in the context of the discovery of sexual structures. We found putative basidiospores and basidia in four of four collections checked, but only following dissolution of thallus granules in KOH (Fig. 5). If confirmed as basidiospores, these would constitute the first sexual structures seen in the genus and could suggest that the photobiont-occupied granular mat represents a simple basidioma. This, in turn, raises the possibility that the inflated, balloon-like cell endings observed by Maass (Reference Maass1986) and us in SEM constitute differentiated hymenial hyphae, such as smooth cystidia, and that the structures called acanthohyphidia in other Acantholichen species are simply spiny cystidia. We considered the possibility that both we and Maass (Reference Maass1986) failed to observe basidiospores in SEM owing to the fixation and/or critical point drying treatment of samples, in our case using a multi-step osmium tetroxide fixation. We accordingly examined thallus granule patches from one specimen in variable pressure (environmental) mode without any fixation of sputtercoating and, like Maass (Reference Maass1986), observed only wrinkled, mucilage-rich cell surfaces with no obvious basidiospores. Based on our collective observations to date and the consistency with which we have been able to get KOH-treated samples to release numerous spores that are consistent in size and shape and bear what appears to be a small hilar appendix, we suspect that the samples are genuinely fertile but that basidiospores become trapped in surface mucilage. How their release is effected in nature is unknown.
Despite its unique characteristics, A. frullaniae shares some features with other Acantholichen species. Morphologically, the most similar species is A. albomarginatus, which can also develop a thallus that is granulose to flat microsquamulose, and acanthohyphidia are sometimes sparse or absent in parts of the lamina but always abundant along the margins (Dal Forno et al. Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016). Acantholichen sorediatus is also similar, especially due to the soredia in its borders, which detach and appear granular. In the case of A. frullaniae, the thallus is entirely granular, without the formation of squamules, and acanthohyphidia are absent (or, depending on how the cells are interpreted, it possesses smooth cystidia). Another shared feature is the colour of the thallus. Acantholichen frullaniae is dark blue with a frosted appearance when dry (Fig. 3), and blue-green when hydrated (see iNaturalist observation: https://www.inaturalist.org/observations/15356946; https://www.inaturalist.org/observations/119837586). Several Acantholichen species have similar blue-green shades (Dal Forno et al. Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016; Table 1). Acantholichen albomarginatus has a blue-green colour with white margins when fresh and dark bluish green to grey when dried; A. campestris is bluish grey when rehydrated and grey when dry; A. variabilis is dark blue when fresh and dark bluish grey when dry; A. pannarioides and A. sorediatus Dal Forno et al. both have a dark blue and dark bluish grey thallus, respectively, however when fresh, A. pannarioides is greyish blue-green and A. sorediatus is green.
One piece of evidence that might have served as an early red flag that A. frullaniae is not closely related to members of Fuscopannaria is the identity of the photobiont. When Maass (Reference Maass1986) described Moelleropsis nebulosa subsp. frullaniae, he characterized it as being associated with ‘very compact and rolled-up filaments of Nostoc with pale heterocysts and large bluegreen cells of 8–18 μm in size’. This distinction was subsequently carried over in future taxonomic treatments and geographical reports, such as those by Jørgensen (Reference Jørgensen2000a), Pérez-Vargas et al. (Reference Pérez-Vargas, Hernández Padrón and Losada-Lima2014) and Tripp & Lendemer (Reference Tripp and Lendemer2019). However, Cornejo & Scheidegger (Reference Cornejo, Nelson, Stepanchikova, Himelbrant, Jørgensen and Scheidegger2016) and Cornejo et al. (Reference Cornejo, Nelson, Stepanchikova, Himelbrant, Jørgensen and Scheidegger2016), who studied the photobionts of the populations of the endangered Erioderma pedicellatum and its ecological associates in eastern Canada, found that the photobiont of A. frullaniae was not Nostoc but Rhizonema (Rhizonemataceae, Nostocales). Rhizonema is widespread in basidiolichens in the subtribe Dictyonematinae (Hygrophoraceae) and several ascolichen genera such as Coccocarpia, Lichinodium, Leptogidium and Parmeliella (Lücking et al. Reference Lücking, Lawrey, Sikaroodi, Gillevet, Chaves, Sipman and Bungartz2009, Reference Lücking, Barrie and Genney2014a; Muggia et al. Reference Muggia, Nelson, Wheeler, Yakovchenko, Tønsberg and Spribille2011; Cornejo et al. Reference Cornejo, Nelson, Stepanchikova, Himelbrant, Jørgensen and Scheidegger2016), and it is the only cyanobacterial genus known to be associated with Acantholichen (Dal Forno et al. Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016, Reference Dal Forno, Lawrey, Sikaroodi, Gillevet, Schuettpelz and Lücking2021). Furthermore, Vondrák et al. (Reference Vondrák, Palice, Mares and Kocourková2013) confirmed via PCR that Fuscopannaria nebulosa (as Moelleropsis), like other species of Fuscopannaria (Jørgensen Reference Jørgensen2000b), is associated with Nostoc species grouped in the core cluster of Nostoc, which includes symbiotic species associated with plants and lichens. We reconstructed a new 16S rRNA phylogeny using a slightly reduced dataset employed by Dal Forno et al. (Reference Dal Forno, Lawrey, Sikaroodi, Gillevet, Schuettpelz and Lücking2021) (Fig. 2). The resulting tree revealed that these new sequences probably represent a previously undetected lineage within Rhizonema. It is possible that our sequences correspond to the same taxon reported previously by Cornejo et al. (Reference Cornejo, Nelson, Stepanchikova, Himelbrant, Jørgensen and Scheidegger2016) and Cornejo & Scheidegger (Reference Cornejo and Scheidegger2016), but this cannot be confirmed at this time, since their data are based solely on rbcLX, whereas ours are limited to 16S. Nonetheless, this lineage appears distinct, suggesting that our samples may belong to a novel cyanobacterial symbiont clade within Rhizonema, which appears sibling to Rhizonema neotropicum in our new 16S phylogenetic tree. This putative new species was recovered with high support within the R. neotropicum-andinum clade, which represents species less common than those belonging to the Rhizonema interruptum complex.
In North America, Acantholichen frullaniae could be mistaken for other crustose cyanolichens. Fuscopannaria cyanolepra (Tuck.) P. M. Jørg. has a granular crust-like thallus with lead blue soredia and overgrows mosses on cliff faces, as well as bare soil banks. It is found from California northwards to Washington, Idaho (Jørgensen Reference Jørgensen2000b), Montana (McCune et al. Reference McCune, Rosentreter, Spribille, Breuss and Wheeler2014) and British Columbia (West Kootenay region, Crawford Bay on Kootenay Lake, T. Spribille 51806, CANL). However, unlike A. frullaniae, Fuscopannaria cyanolepra can have brown corticate areas at the centre of the thallus and produces apothecia, albeit rarely. Fuscopannaria leprosa P. M. Jørg. has a leprose thallus, composed of granular plumbeous soredia, with unidentified terpenoids and fatty acids. The type is from trunks of Acer macrophyllum and it has also been reported from twigs of a Salix sp. (Jørgensen Reference Jørgensen2000b). Unlike A. frullaniae, F. leprosa produces secondary metabolites, including unidentified terpenoids and fatty acids. It could not be relocated at the type locality in western Washington, USA, in a recent survey (T. Spribille, personal observation). Fuscopannaria nebulosa has an effuse leprose thallus, with blue-grey, corticated granules, and occurs on soil as a short-lived pioneer species on recently disturbed soil. Jørgensen (Reference Jørgensen2000b) cites three specimens from North America, one from Shoshone County, Idaho, one from Massachusetts, and one from California. Even though the previously mentioned species share a granular thallus with similar colour, some characteristics separate them from A. frullaniae. Fuscopannaria nebulosa grows on soil, and some specimens produce apothecia with a granular exciple.
Distribution
Acantholichen frullaniae is known from eastern Canada, occurring in oceanic forests in Newfoundland and Nova Scotia (Maass Reference Maass1986), and from the eastern USA in high-elevation mixed hardwood-conifer forests in the southern portion of the Appalachian Mountains of North Carolina (Tripp & Lendemer Reference Tripp and Lendemer2019). The name Moelleropsis nebulosa subsp. frullaniae has been applied to European material, specifically in the Iberian Peninsula from western Portugal and northern Spain, as well as the Canary Islands (Jørgensen Reference Jørgensen2000a; Carballal-Durán & López de Silanes Vázquez Reference Carballal-Durán and López de Silanes Vázquez2006; Pérez-Vargas et al. Reference Pérez-Vargas, Hernández Padrón and Losada-Lima2014). We did not study any of these specimens, and no sequences have been produced. Based on our results, we cannot be certain that these specimens belong to A. frullaniae and not another member of the Pannariaceae with similar morphology occurring in the area, such as Fuscopannaria cyanolepra, F. nebulosa, Vahliella atlantica (P. M. Jørg. & P. James) P. M. Jørg., or V. isidioidea Pérez-Vargas et al. (Jørgensen Reference Jørgensen2005; Pérez-Vargas et al. Reference Pérez-Vargas, Hernández Padrón and Losada-Lima2014). However, all European specimens have also been reported to occur together with Frullania, similar to the populations from eastern North America. Molecular data would be required to confirm the presence of A. frullaniae in Europe.
Ecology
In Atlantic Canada, Acantholichen frullaniae is associated with the liverwort Frullania asagrayana (syn. Frullania tamarisci subsp. asagrayana), which has a broad ecological amplitude, growing on trunks of Abies balsamea, Acer spp., Betula alleghaniensis, Fraxinus spp. and Thuja occidentalis (Maass Reference Maass1986; Haughian et al. Reference Haughian, Bagnell, Daley, Frego, Smith and Clayden2016). Acantholichen frullaniae is, however, more limited in its distribution than Frullania asagrayana, occurring primarily in close proximity to the Atlantic Coast, in wet mixed wood forests, as well as in swamps dominated by Acer rubrum and Abies balsamea, where Erioderma pedicellatum occurs (Maass Reference Maass1986; Cameron Reference Cameron2009). The ecology of the specimen from North Carolina, USA, is different from the occurrences in eastern Canada. Tripp & Lendemer (Reference Tripp and Lendemer2019) reported A. frullaniae as occurring on a vertical, granitic rock face overgrowing mosses (Atrichum and Aulacomnium) in mixed hardwood-conifer forests at 1700 m. One of the US specimens included here from North Carolina (Tripp & Lendemer 4951, COLO) is from the Tripp & Lendemer (Reference Tripp and Lendemer2019) study and grows on the moss Hedwigia filiformis (Fig. 3E & F). In western Europe, the specimens identified as Moelleropsis nebulosa subsp. frullaniae are found in moist microhabitats at elevations ranging from 500 to 1200 m. Jørgensen (Reference Jørgensen2000a) identified a specimen collected from western Portugal, growing on Frullania sp. on Arbutus unedo bark in a shaded, moist habitat at 500 m. In northern Spain, Carballal-Durán & López de Silanes Vázquez (Reference Carballal-Durán and López de Silanes Vázquez2006) reported it growing on a terricolous Frullania sp. on a vertical soil talus in a warm, moist canyon at 947 m. Lastly, Pérez-Vargas et al. (Reference Pérez-Vargas, Hernández Padrón and Losada-Lima2014) reported two specimens from the Canary Islands: one was collected on Frullania teneriffae over rocks at 850 m in an evergreen laurel forest, and the other on F. dilatata growing on soil at 1200 m in a Canary pine woodland.
Conservation and threats
As with several other rare and at-risk lichens that are known from near-coastal wet forests and swamps in Atlantic Canada, Acantholichen frullaniae appears to depend upon the long-term continuity and stability of its habitat, and may therefore be threatened by a combination of habitat loss and a warming climate. Habitat loss from industrial and residential development, forest harvesting, and the increasing frequency and intensity of windthrow events associated with tropical storms have been implicated in the continued declines of co-occurring species such as Erioderma mollissimum (Samp.) Du Rietz, E. pedicellatum, Fuscopannaria leucosticta (Tuck. ex E. Michener) P. M. Jørg., Pannaria lurida (Mont.) Nyl. and Pectenia plumbea (Lightf.) P. M. Jørg. et al. (COSEWIC 2010, 2014, 2016, 2019, 2021). Moreover, climatic variables have been consistently important in species distribution models (Cameron & Neily Reference Cameron and Neily2008; Cameron et al. Reference Cameron, Neily and Clayden2011; Cameron Reference Cameron2021; Haughian et al. Reference Haughian, Gray and Harper2022), and climate projections suggest that a warming climate could make much of these species’ current ranges unsuitable within the next 50 years (COSEWIC 2016; Haughian Reference Haughian2021; M. Frison & S. R. Haughian, unpublished data). As with Erioderma pedicellatum, a further limitation for Acantholichen frullaniae may be that its primary host tree, Abies balsamea, is relatively short-lived in these forested wetlands, rarely exceeding 80 years in age (Cameron & Neily Reference Cameron and Neily2008). Consequently, protecting the known occurrences of Acantholichen frullaniae, particularly along its current range limit in northern Nova Scotia and Newfoundland, will be necessary, but probably not sufficient for ensuring its persistence. Studies on inter-tree and inter-habitat dispersal, habitat regeneration, and the bioclimatic envelope of this species are needed to facilitate more effective conservation. Notably, many of the same conservation concerns in Atlantic Canada also apply in the US Appalachians. Allen & Lendemer (Reference Allen and Lendemer2016) have modelled drastic changes in lichens at high elevations in the southern Appalachians in areas where A. frullaniae has been found under likely climate change scenarios.
Another species in the genus, Acantholichen galapagoensis, has already been formally assessed and is currently listed as Vulnerable (VU) under the IUCN Red List (criteria B1ab[iii] + 2ab[iii], D2; Bungartz Reference Bungartz2018). This designation reflects its limited and fragmented distribution within the Galápagos Islands and its strong dependence on the presence of specific liverwort hosts, which are themselves sensitive to habitat alterations. While A. frullaniae may face increasing pressures from forest harvesting, storm-related windthrow and rising temperatures, A. galapagoensis is primarily threatened by invasive plant management efforts, especially the removal of Cinchona pubescens which inadvertently disturbs its delicate microhabitat. Despite their occurrence in ecologically distinct regions, both species exemplify the vulnerability of narrowly distributed cyanolichens to environmental change, underscoring the importance of habitat conservation and long-term ecological stability in preserving lichen biodiversity.
Conclusions
Acantholichen is only the latest addition to a series of biogeographical range extensions of neotropical genera that extend into Canada. In the case of the genus Erioderma, three species occur in boreal-temperate coastal forests, whereas the bulk of the species diversity occurs in tropical regions worldwide (Jørgensen Reference Jørgensen2001; Jørgensen & Arvidsson Reference Jørgensen and Arvidsson2001, Reference Jørgensen and Arvidsson2002; Jørgensen & Sipman Reference Jørgensen and Sipman2002). Another genus that follows the same trend is Coccocarpia, with two species occurring in temperate and Arctic latitudes, and the vast majority of species diversity found in the Neotropics and Paleotropics (Maass & Yetman Reference Maass and Yetman2002; Power et al. Reference Power, Cameron, Neily and Toms2018; Spribille et al. Reference Spribille, Fryday, Hampton-Miller, Ahti, Dillman, Thor, Tønsberg and Schirokauer2023; Coca et al. Reference Coca, Lumbsch, Mercado-Díaz, Widhelm, Goffinet, Kirika and Lücking2025). A notable example from the Canadian Pacific coast is Dictyonema moorei (Nyl.) Henssen, reported from Haida Gwaii (Brodo Reference Brodo1995), which may warrant re-examination using molecular approaches, since it could represent an undescribed taxon (I. M. Brodo, personal communication 1998, in Goward (Reference Goward1999)). The voucher specimen at CANL could not be located for study for this paper (I. M. Brodo, personal communication 2025).
Acantholichen frullaniae is the first Nearctic species of the genus. Once considered monotypic (Lawrey et al. Reference Lawrey, Lücking, Sipman, Chaves, Redhead, Bungartz, Sikaroodi and Gillevet2009), Dal Forno et al. (Reference Dal Forno, Lücking, Bungartz, Yánez-Ayabaca, Marcelli, Spielmann, Coca, Chaves, Aptroot and Sipman2016) revealed the existence of five additional species of Acantholichen, all distributed throughout Central and South America. Recently, another species, A. dendroideus V. Marcano, was described from Venezuela (Marcano Reference Marcano2022) but it lacks available DNA sequences. Each of these has a distinct distribution. With the addition of A. frullaniae, the genus is now composed of eight species and spans from the tropics to upper temperate regions. We hope that our new work will raise awareness and lead to new, targeted searches for this rare species.
Acknowledgements
Special thanks to Nathan Gerein (University of Alberta Earth and Atmospheric Sciences SEM unit) for help with scanning electron microscopy. John Brinda is thanked for his prompt identification of the moss Hedwigia. MDF is grateful to James Lendemer and Erin Manzitto-Tripp for sharing a duplicate of their collection and for being the first to recognize the North Carolina material as Acantholichen. This work was supported by a Discovery Grant to TS from the Natural Sciences and Engineering Research Council of Canada (RGPIN-2019-04892) and a Canada Research Chair in Symbiosis.
Author ORCIDs
Alejandro Huereca, 0000-0002-6460-2380; Toby Spribille, 0000-0002-9855-4591.