LEARNING OBJECTIVES
After reading this article, you will be able to:
-
recognise how fluctuations in ovarian hormones influence motivation, mood and sexual desire in women across the menstrual cycle, postpartum and menopause
-
evaluate the potential impact of hormonal contraceptives on mood and reward-related functioning to inform patient-centred prescribing and counselling
-
understand the clinical relevance of interactions between ovarian hormones and reward function in relation to depression, premenstrual dysphoric disorder and substance use disorders in women.
The survival of organisms depends on the ability to seek out and obtain resources. From food, mating and social attachment to more uniquely human things such as money, we have evolved specific functions that maximise our capacity for goal pursuit and attainment. These functions fall under the umbrella of reward processing, and consist of the ability to anticipate and expend effort towards reward, learn from previous reward experience and feel pleasure and/or satiety from rewards. Disruptions in reward processing are a hallmark of various psychiatric disorders, including depressive disorders, substance use disorders and psychotic disorders (Novick Reference Novick, Levandowski and Laumann2018). But separate from psychopathology, reward processing facilitates much of what makes life meaningful: engagement in work and hobbies; interpersonal relationships; and sexual intimacy.
The two major ovarian steroid hormones, oestradiol and progesterone, have significant interactions with the neurobiology of reward processing. Given the role of oestradiol and progesterone in reproduction, it is not surprising that they influence reward processing related to sex (Marcinkowska Reference Marcinkowska, Shirazi and Mijas2023), social attachment (Young Reference Young, Wang and Insel1998) and parental care (Numan Reference Numan and Insel2003). However, research has also found a more ubiquitous role in reward processing, making the interaction between ovarian hormones and reward a key component of women’s mental health. This article provides a brief overview of work related to ovarian hormones and reward processing, starting with preclinical data and working up to human neuroimaging studies. Studies were selected based on relevance to reward-related neural circuits or behaviours, direct measurement or manipulation of ovarian hormones, and use of established paradigms in both human studies and animal models. Special attention is given to research emerging on the synthetic derivatives of oestradiol and progesterone used in hormonal contraceptives and their effects on reward processing. Finally, the clinical implications of current knowledge are discussed in relation to both psychiatric disorders and quality of life.
Neurobiology of reward processing: a very brief overview
Reward processing relies on a variety of connected brain regions and neurobiological substrates. Perhaps the most well-known and studied is the mesocorticolimbic dopamine system, which consists of dopaminergic cells in the ventral tegmental area that project to the ventral striatum (and specifically, the nucleus accumbens) and areas of the prefrontal cortex. Dopaminergic activity in the ventral striatum and nucleus accumbens is most frequently associated with approach motivation for rewards, which includes the ability to anticipate rewards based on cues as well as energising the response to pursue rewards (Berridge Reference Berridge and Kringelbach2015). Opioid and cannabinoid activity within the nucleus accumbens, ventral pallidum and other regions facilitates the actual pleasure response on reward attainment (Berridge Reference Berridge and Kringelbach2015). Finally, neural activity within the prefrontal cortex assists with reward learning via the encoding of reward value to help guide future behaviour (Rushworth Reference Rushworth, Noonan and Boorman2011).
Endogenous ovarian hormones and reward neurobiology: oestradiol, progesterone and allopregnanolone
Endogenous ovarian hormones across the female lifecycle
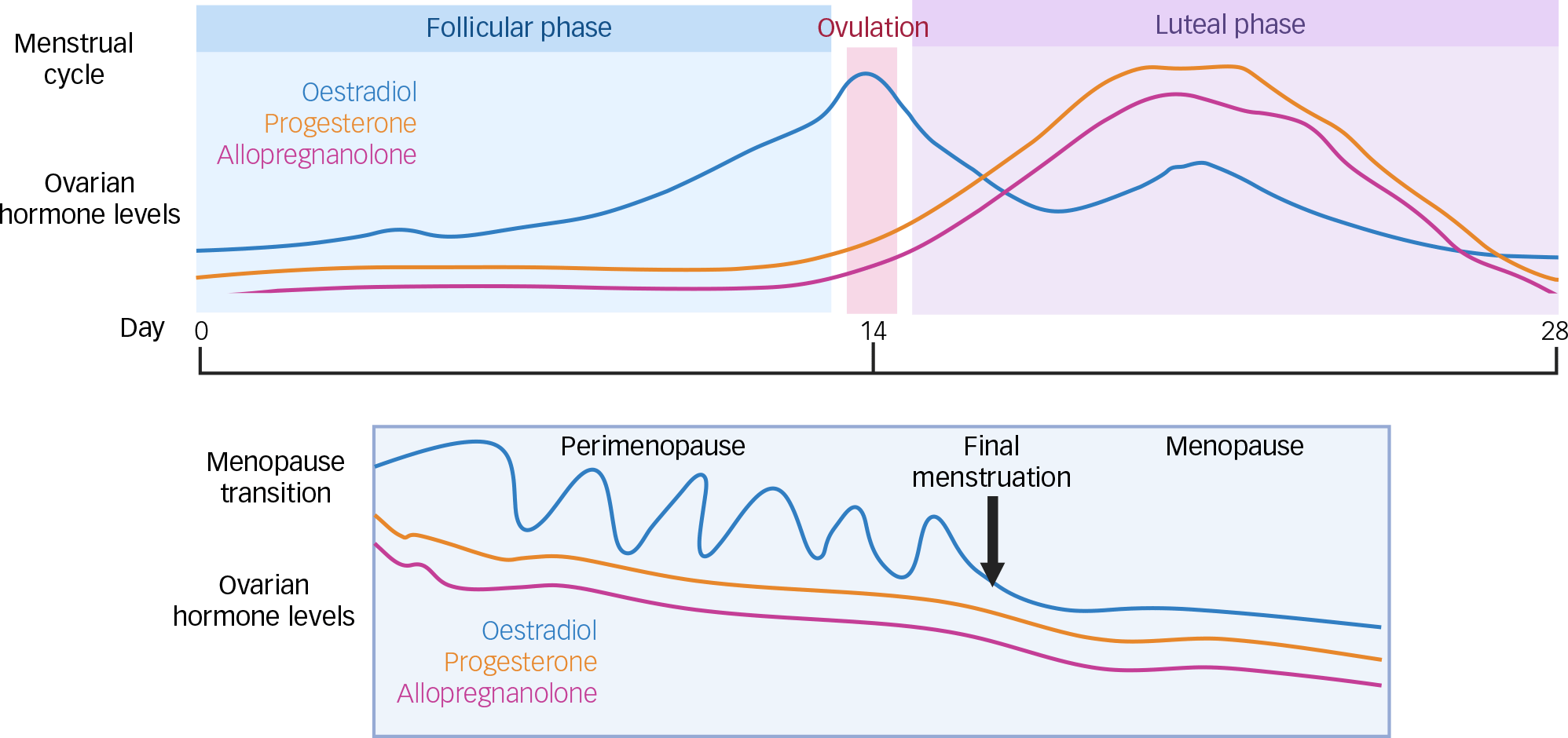
Prior to the menopausal transition (perimenopause), hormone levels in most adult females are dictated by the menstrual cycle, which lasts an average of 28 days and is commonly divided into two phases: the follicular phase and the luteal phase (Bull Reference Bull, Rowland and Scherwitzl2019). At the start of the follicular phase, both oestradiol and progesterone are low. Oestradiol levels subsequently begin to rise, such that during the mid-follicular phase, there are elevated oestradiol levels unopposed by progesterone. Oestradiol levels then peak around ovulation. Following ovulation, the luteal phase begins and is marked by rising levels of progesterone. At the end of the luteal phase, both progesterone and oestradiol rapidly decline to trigger menstruation and a repeat of the cycle (Gandara Reference Gandara, Leresche and Mancl2007). Because allopregnanolone is a metabolite of progesterone, levels will closely follow those of the parent hormone across the cycle (Kimball Reference Kimball, Dichtel and Nyer2020) (Fig. 1).
Relative changes in ovarian hormones across the menstrual cycle and during the menopausal transition. Image created with biorender.com.

Around the fourth to fifth decade of life, the menstrual cycle starts to become more irregular as women enter the menopausal transition. During this transition, levels of progesterone decrease, whereas oestradiol levels fluctuate in the context of an overall downward trend. Eventually, there is a full cessation of menstruation that corresponds to significantly decreased levels of oestradiol, progesterone and allopregnanolone compared with the premenopausal state (Harlow Reference Harlow, Gass and Hall2012; Kimball Reference Kimball, Dichtel and Nyer2020) (Fig. 1).
Oestradiol
Oestradiol represents the dominant and most potent endogenous steroid within the oestrogen family, and is thus the primary focus of this review. Receptors for oestradiol, including oestrogen receptors alpha (ERα) and beta (ERβ) and the G protein-coupled oestrogen receptor (GPER), are found within the ventral tegmental region and nucleus accumbens, where they can influence dopaminergic transmission (Almey Reference Almey, Milner and Brake2015). Overall, oestradiol has a facilitatory influence on dopamine activity, with acute treatment in rats enhancing dopamine release within the nucleus accumbens (Yoest Reference Yoest, Quigley and Becker2018). Oestradiol may also facilitate reward responsiveness/satiety via interactions with the opioid system. Specifically, oestradiol enhances release of the endogenous opioid β-endorphin, while also increasing levels of μ-opioid receptors, responsible for opioid-induced reward (Ethridge Reference Ethridge and Smith2023). Acutely, this allows for increased analgesia and reward; however, chronic treatment with oestradiol (at least in animals) eventually downregulates receptors and renders the brain less sensitive to opioid activation (Ethridge Reference Ethridge and Smith2023).
Behaviourally, oestradiol can potentiate the dopaminergic effects of psychostimulants. In women with regular menstrual cycles administered amphetamine, higher levels of oestradiol were associated with enhanced subjective effects, particularly on measures of euphoria and energy (Justice Reference Justice and de Wit1999). The same group found that administration of exogenous oestradiol to women during the early follicular phase prior to amphetamine administration had more limited impact, but did increase subjective ratings of pleasant stimulation (Justice Reference Justice and de Wit2000).
Functional magnetic resonance imaging (fMRI) has provided further insight into oestradiol’s role in reward. In premenopausal women, 58 regularly menstruating women underwent fMRI brain scans while completing a monetary gambling task first during the mid-follicular phase and then after receiving a gonadotropin-release hormone agonist (GnRHa) to decrease levels of sex steroids. Following GnRHa administration, there was a significant decrease in reactivity of the amygdala to monetary reward (Macoveanu Reference Macoveanu, Henningsson and Pinborg2016), emphasising the importance of oestradiol in reward responsiveness. Further support for oestradiol’s facilitation of reward processing comes from comparing 13 women during the mid-follicular phase and then again during the luteal phase when progesterone levels are high. During the mid-follicular phase, women demonstrated greater brain activation of reward regions during both anticipation and receipt of monetary reward compared with the luteal phase (Dreher Reference Dreher, Schmidt and Kohn2007).
Not all human imaging studies on premenopausal women support a role for oestradiol in engaging the reward system. For example, one fMRI study of 28 women that utilised a monetary reward task during the mid-follicular phase and then again during the premenstrual phase (when levels of oestradiol are at their lowest) found that activation of the ventral striatum in anticipation of monetary reward was highest during the premenstrual phase (Ossewaarde Reference Ossewaarde, van Wingen and Kooijman2011). This would appear to be the opposite of previous results.
Additional research on the role of oestradiol in reward comes from work studying women in various phases of the menopause transition, in which oestradiol is decreased compared with levels in premenopausal women. In an fMRI pilot study comparing premenopausal women (n = 5) with women who had undergone removal of ovaries and uterus (n = 6), those in the surgical menopause group demonstrated decreased activation of various limbic regions in response to erotic stimuli (Archer Reference Archer, Love-Geffen and Herbst-Damm2006). Administration of oestradiol to the surgical menopause group increased limbic activation, with a combination of oestradiol and testosterone resulting in almost a brain-activation profile similar to that of the premenopausal group. In another placebo-controlled fMRI study of 15 perimenopausal women, administration of a hormone-replacement regimen of oestradiol plus progesterone increased activation of dopaminergic reward regions during both the anticipation and receipt of monetary reward (caudate, putamen, ventromedial prefrontal cortex) relative to placebo, and these increases correlated with blood levels of oestradiol (Thomas Reference Thomas, Météreau and Déchaud2014).
Despite variation in human fMRI findings with oestradiol, many studies – especially those using pharmacological manipulation rather than menstrual phase comparisons – are consistent with a role of oestradiol in facilitating multiple domains of reward processing across different stimuli types (e.g. erotica, money). There have been several in-depth reviews on ovarian hormones and the use of neuroimaging, to which the reader is referred (Comasco Reference Comasco, Frokjaer and Sundström-Poromaa2014; Pletzer Reference Pletzer, Winkler-Crepaz and Maria Hillerer2023).
Progesterone
Progesterone’s reputation as an inhibitor of neural mechanisms governing reward comes from animal and human data showing a decrease in both self-administration of, and positive subjective effects from, various drugs of misuse, leading investigators to consider progesterone as a potential intervention for substance use disorders (Peltier Reference Peltier and Sofuoglu2018). In addition, higher endogenous progesterone has a negative effect on sexual desire and activity (Marcinkowska Reference Marcinkowska, Shirazi and Mijas2023). Interestingly, in animal studies, administration of progesterone does appear to enhance dopaminergic activity in striatal regions, which would suggest a facilitation of reward-seeking function (Dluzen Reference Dluzen and Ramirez1984). However, this might be a time-dependent action, with initial increases in striatal dopamine release after progesterone administration, followed by decreases 24 h later (Dluzen Reference Dluzen and Ramirez1984). One possibility is that progesterone’s eventual inhibitory effects on dopaminergic activity stem from its metabolism to allopregnanolone, which has been found to inhibit dopamine release from reward regions in rats through enhancement of gamma-aminobutyric acid A (GABAA) receptor modulation (Scheggi Reference Scheggi, Concas and Corsi2024), and this is discussed further in the section below on allopregnanolone.
As described in the section above on oestradiol, human fMRI studies comparing menstrual cycle phases have found that during the luteal phase (when progesterone is high), there is decreased activation of reward regions in response to monetary reward compared with the mid-follicular phase, when oestradiol is unopposed by progesterone (Dreher Reference Dreher, Schmidt and Kohn2007). A separate study comparing female smokers in the follicular and luteal phases found decreased activation to smoking cues in the luteal phase when progesterone levels were high (Franklin Reference Franklin, Jagannathan and Wetherill2015). However, when females were administered progesterone or placebo during the early follicular phase or during the pill-free week for those using oral contraceptives (when progesterone levels were low), no differences in brain activation to smoking cues were found (Novick Reference Novick, Duffy and Johnson2022a). However, this study used a lower dose of progesterone compared with other studies of progesterone for smoking intervention (200 v. 400 mg), and it is possible that higher doses might have been associated with more significant neural effects.
Allopregnanolone
Allopregnanolone is one of the neuroactive steroid metabolites of progesterone that acts as a positive allosteric modulator of the GABAA receptor. Accumulating evidence supports allopregnanolone’s role in affective processes and psychiatric disorders such as depression, premenstrual dysphoric disorder (PMDD) and post-traumatic stress disorder (Deligiannidis Reference Deligiannidis2025).
Allopregnanolone has long been known to have dose-dependent effects on affect that follow either U or inverted-U patterns. This was elegantly demonstrated by Andréen et al, who administered various doses of progesterone to post-menopausal women and measured their subsequent mood and allopregnanolone levels (Andréen Reference Andréen, Sundström-Poromaa and Bixo2006). Low and high allopregnanolone concentrations were associated with favourable changes in mood, whereas concentrations in the middle had a paradoxical effect, increasing negative and decreasing positive aspects of mood.
When looking specifically at reward neurobiology and behaviour, allopregnanolone has similar dose-dependent effects, as reviewed recently by Scheggi et al (Reference Scheggi, Concas and Corsi2024). At physiological levels in rodents, allopregnanolone enhances dopamine release in the nucleus accumbens and promotes reward-related behaviour related to social interaction and sexual receptivity (Scheggi Reference Scheggi, Concas and Corsi2024). However, either blocking the formation of allopregnanolone or administering supraphysiological doses decreases dopamine activity as well as reward behaviour (Scheggi Reference Scheggi, Concas and Corsi2024). At first glance, these data in animals do not necessarily seem to fit with the human work of Andréen et al, given that on one hand, ‘middle’ physiological concentrations of allopregnanolone facilitate reward neurobiology and behaviour, yet they are also associated with the highest levels of negative mood. To reconcile this, more human studies are needed that simultaneously measure allopregnanolone, reward behaviour, and positive and negative mood. Allopregnanolone can, of course, influence mood in ways beyond reward via its effects on anxiety and its influence on modulating the stress response (Deligiannidis Reference Deligiannidis2025). Further complicating the picture is that progesterone has several neuroactive steroid metabolites beyond allopregnanolone, some of which are also positive allosteric modulators at the GABAA receptor, and some of which are negative allosteric modulators. It is now recognised that the concentration and ratio of these other neuroactive steroids likely determines the overall effect on the brain, rather than just the levels of allopregnanolone (Deligiannidis Reference Deligiannidis2025). Furthermore, different psychiatric phenotypes may result in differing sensitivity to allopregnanolone. For example, women with PMDD have evidence of decreased sensitivity to allopregnanolone (Hantsoo Reference Hantsoo and Epperson2020), which might shift the dose–response relationship as regards its influence on reward variables.
Despite the above-mentioned important nuance of allopregnanolone’s effects, there is some evidence that the positive effects of progesterone administration for substance use disorders might be mediated by allopregnanolone. For example, progesterone can prevent stress-induced reinstatement of cocaine self-administration in rats and this effect is blocked with inhibition of progesterone metabolism to allopregnanolone (Anker Reference Anker, Holtz and Zlebnik2009); and in cocaine-dependent humans administered progesterone, those with higher allopregnanolone levels had decreased cravings (Peltier Reference Peltier and Sofuoglu2018). However, in studies of smokers, higher allopregnanolone after progesterone administration was associated with increased positive effects of nicotine, which could be counterproductive to quitting – although it was also found that higher allopregnanolone levels following progesterone administration decreased withdrawal severity in female smokers (Novick Reference Novick, Duffy and Johnson2022b). This highlights that there are many aspects to treating substance use disorders, including potential sex differences, and effects of allopregnanolone on modulating withdrawal phenomena as well as the stress response might be just as important as any direct effects on the reward system.
Summary: oestradiol, progesterone, allopregnanolone and reward processing
Overall, a preponderance of animal research supports the view that oestradiol tends to enhance neural mechanisms promoting approach and consummatory aspects of reward, whereas progesterone may exert opposing effects. With allopregnanolone, the animal research suggests that physiological concentrations of allopregnanolone can facilitate reward function, although both very low and supraphysiological levels impair reward function.
Some of the behavioural and neuroimaging data in humans supports what is seen in animals on oestradiol, progesterone and allopregnanolone, but it is by no means uniform. Variability in findings may relate to small sample sizes and lack of controlled, interventional designs that can better infer causation. Human studies relying on menstrual cycle phase tended to vary in their definitions and determinations of these phases. Another source of divergent findings in the human literature could be differences in sensitivity to the effects of hormones on reward function within the population. For example, a proportion of women may be particularly sensitive to oestradiol–dopamine interactions such that surges of oestradiol trigger maladaptive risk-taking behaviour, whereas rapid drops in oestradiol trigger anhedonia and depression (Peters Reference Peters, Schmalenberger and Eng2025).
To better elucidate the effects of ovarian hormones on reward processing, future research will need to employ larger sample sizes with multimodal assessment (hormone levels, neuroimaging and behavioural measures). The use of controlled interventional designs that administer various doses of hormones along with placebo will be important to better infer causation and complement more naturalistic designs that study women according to menstrual cycle phase. Finally, ongoing research to better understand and identify phenotypes with differing hormonal sensitivity will be essential for understanding the role of ovarian hormones in reward processing in different populations.
The influence of hormonal contraceptives on reward processing
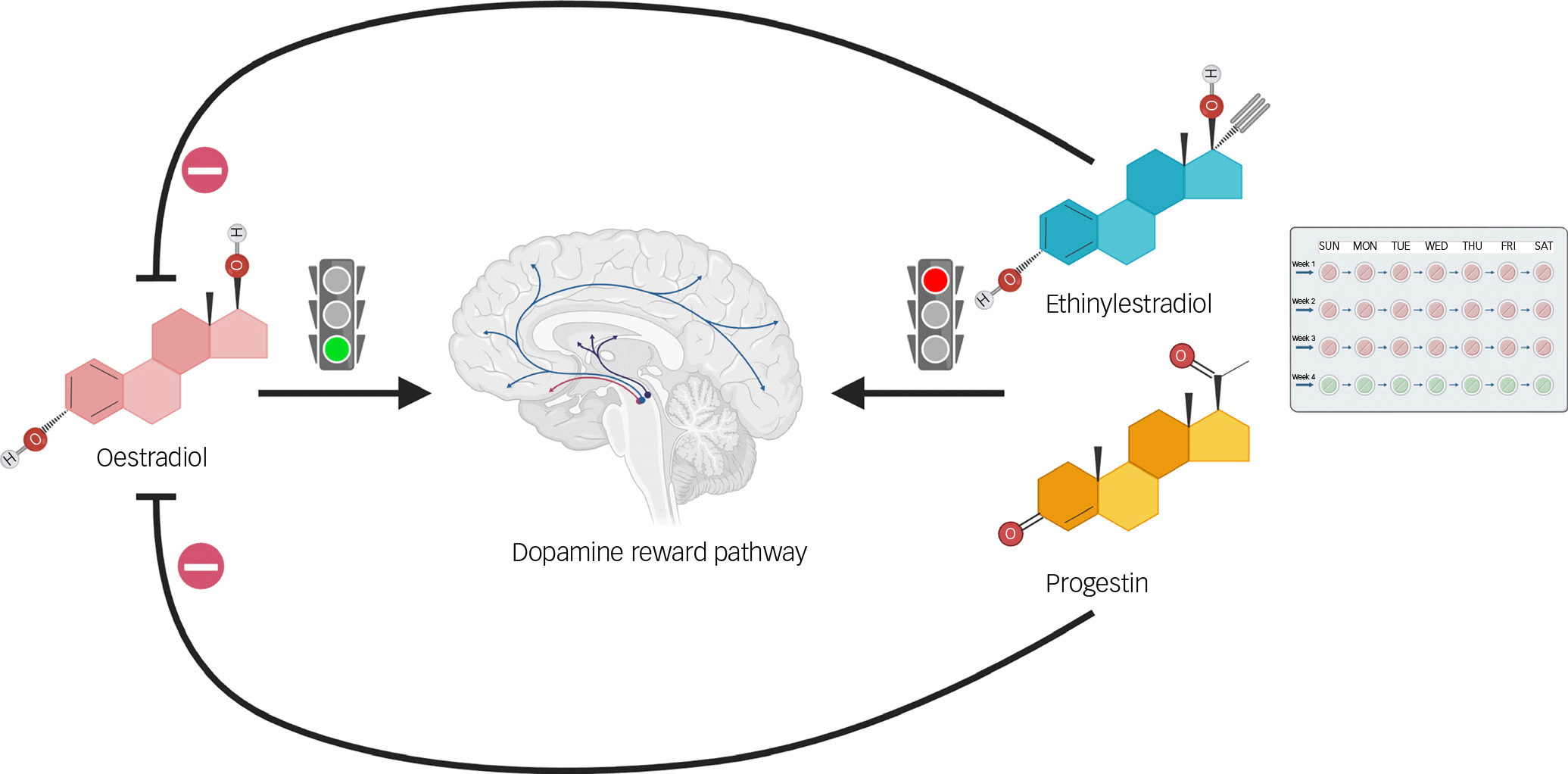
Almost all versions of hormonal contraception for women employ synthetic derivatives of progesterone (also known as progestins) with or without an oestrogen component. Depending on the formulation, hormonal contraceptives can result in suppression of endogenous ovarian hormones (Tronson Reference Tronson and Schuh2022). This raises the question as to whether the addition of these derivatives, with or without endogenous hormone suppression, has different effects on the brain compared with naturally cycling ovarian hormones. Numerous challenges exist when it comes to studying the brain effects of hormonal contraceptives, given the variety of formulations (in terms of both different hormone derivatives and delivery mechanisms) as well as a survivor effect, in which women prone to adverse effects are less likely to participate in research (Tronson Reference Tronson and Schuh2022; Larsen Reference Larsen, Frokjaer and Ozenne2025). Nonetheless, adverse changes in mood and decreases in sexual desire are commonly reported side-effects with hormonal contraceptives (Sanders Reference Sanders, Graham and Bass2001), suggesting that the distinct pharmacology of these derivatives may differentially affect brain functions such as reward processing (Fig. 2).
Potential mechanisms by which hormonal contraception influences reward processing. Endogenous oestradiol has an overall facilitatory effect on dopamine activity in reward pathways. Both the progestin and ethinylestradiol component of an oral contraceptive pill may directly decrease dopamine activity while simultaneously suppressing endogenous oestradiol. Image created with biorender.com.

Progestins
Animal studies have reported that several progestins found in hormonal contraceptives, either with or without an oestrogen component, decrease dopamine levels in various brain regions, including the striatum, potentially owing to progestin increases in monoamine oxidase activity (Pletzer Reference Pletzer, Winkler-Crepaz and Maria Hillerer2023). Monoamine oxidase is one of the enzymes in the brain that metabolises neurotransmitters, such that increases in monoamine oxidase result in decreased availability of presynaptic dopamine for release.
Ethinylestradiol
Oral contraceptive pills most commonly utilise the synthetic oestrogen ethinylestradiol because it is highly resistant to first pass metabolism in the liver and thus has high bioavailability when taken orally. By comparison, 17β-oestradiol (the primary endogenous form of oestradiol) is extensively metabolised by the liver such that only 2–10% of it ends up being bioavailable when taken orally (O’Connell Reference O’Connell1995), making it less ideal for inclusion in oral contraceptive pills. There are likely differences between ethinylestradiol and endogenous oestradiol in relation to reward. For example, ethinylestradiol has decreased affinity for ERβ compared with endogenous oestradiol (Stanczyk Reference Stanczyk, Winer and Foidart2024). As this receptor has been found to regulate dopamine activity in rodents (Almey Reference Almey, Milner and Brake2015), one might speculate that ethinylestradiol lacks some of the pro-dopaminergic properties of endogenous oestradiol relevant to reward processing.
Limited evidence in humans supports a possible link between ethinylestradiol and decreased reward function. One study demonstrated improved sexual desire after switching patients on a hormonal contraceptive containing ethinylestradiol to one containing the endogenous 17β-oestradiol (Caruso Reference Caruso, Cianci and Cariola2017). Despite the known advantages of ethinylestradiol in terms of increased oral bioavailability compared with 17β-oestradiol, this suggests that utilising 17β-oestradiol may be feasible, and it provides the impetus for further studying the potential benefits for reward function when using 17β-oestradiol instead of the traditional ethinylestradiol in hormonal contraceptives. That said, it should be noted that owing to first pass metabolism of oral 17β-oestradiol, there are still significant differences between this and endogenously released oestradiol (or oestradiol administered transdermally), namely increased levels of the oestrone metabolite, such that oral 17β-oestradiol does not necessarily mimic the physiological milieu of endogenous 17β-oestradiol (O’Connell Reference O’Connell1995).
Influence of hormonal contraceptives on neuroactive steroids
Another possible way by which hormonal contraceptives might interfere with reward neurobiology is via decreases in various neuroactive steroids in the brain that facilitate reward function. Specifically, treatment with the common combined oral contraceptive ethinylestradiol and levonorgestrel decreases levels of pregnenolone, allopregnanolone and testosterone in the rodent brain (Porcu Reference Porcu, Serra and Concas2019). Similar decreases are found in plasma of women treated with combined oral contraceptives (Porcu Reference Porcu, Serra and Concas2019). Both pregnenolone and testosterone enhance dopamine release in the rat nucleus accumbens (Zheng Reference Zheng2009). As described in the section on allopregnanolone above, the effects of allopregnanolone on reward are nuanced; however, animal studies would suggest that hormonal contraceptive-induced decreases in allopregnanolone below usual physiological levels would impair dopaminergic activity (Scheggi Reference Scheggi, Concas and Corsi2024).
In line with the potential interference of hormonal contraception with reward neurobiology, combination treatment with ethinylestradiol and levonorgestrel in rats decreases social interaction, with conspecific as well as copulatory behaviour (Porcu Reference Porcu, Serra and Concas2019). Interestingly, the decrease in social and sexual motivation correlated with decreases in allopregnanolone, suggesting the importance of hormonal contraception-induced decreases in neuroactive steroids in reward function. Apart from this study, animal research on hormonal contraception and reward behaviour has been limited, with one recent study demonstrating lack of effects of ethinylestradiol or levonorgestrel on reinforcement responding to shut off of an overhead light (McNealy Reference McNealy, Oevermann and Knabel2024). However, given the tendency of rats to avoid illuminated areas, the relevance of this study to appetitive reward processing is unclear.
Human neuroimaging studies of hormonal contraceptives and reward processing
Human studies on hormonal contraceptives and reward processing have produced mixed results. In one study that administered oxytocin to women prior to viewing pictures of their partners, women using hormonal contraceptives (n = 21) demonstrated decreased activation of the nucleus accumbens and rated the attractiveness of their partners lower compared with women not on hormonal contraceptives (n = 19) (Scheele Reference Scheele, Plota and Stoffel-Wagner2016). Similarly, women on various forms of hormonal contraceptive (n = 12) demonstrated decreased activation of the insula and precentral gyrus during anticipation of erotic images compared with women not on hormonal contraceptives (n = 12) (Abler Reference Abler, Kumpfmüller and Grön2013). However, the same research group using the same cohort found that women taking hormonal contraceptives showed increased activation of the insula in anticipation of monetary reward (Bonenberger Reference Bonenberger, Groschwitz and Kumpfmueller2013). Although these studies were limited by studying a small number of women already on hormonal contraceptives and taking different formulations, they do suggest that hormonal contraceptives might predominantly influence reward processing related to social and sexual reward.
Hormonal contraceptives and reward: the potential role of adverse childhood experiences
Given frequent conflicting results within hormonal contraception research, it is likely that certain factors may make women more susceptible to negative side-effects. As regards hormonal contraceptive-induced deficits in reward processing, a history of adverse childhood experiences (ACEs) may play a prominent role. ACEs on their own are associated with deficits in reward processing (Novick Reference Novick, Levandowski and Laumann2018), leading to the hypothesis that hormonal contraceptives may exacerbate this vulnerability further (Novick Reference Novick, Johnson and Lazorwitz2022c, Reference Novick, Stoddard and Johnson2023). Women with a history of two or more ACEs were more likely to report previous discontinuation of hormonal contraception owing to decrease in sexual desire compared with women with one or no ACEs (Novick Reference Novick, Johnson and Lazorwitz2022c). A follow-up study evaluated anticipated and experienced reported pleasure when viewing erotic images in both current and former hormonal contraception users with both a high and low number of ACEs (Novick Reference Novick, Stoddard and Johnson2023). In current hormonal contraceptive users, among those reporting decreased sexual desire due to their hormonal contraceptive, the high-ACEs group gave lower anticipated pleasure ratings for erotic images compared with the low-ACEs group (Novick Reference Novick, Stoddard and Johnson2023). Importantly, among former hormonal contraceptive users who discontinued their use because of decreased sexual desire, there were no differences in anticipatory reward for erotic images between ACEs groups, suggesting that the presence of the hormonal contraceptive (as opposed to another variable) was affecting reward behaviour.
Summary: influence of hormonal contraceptives on reward processing
A significant amount of neurobiological and behavioural evidence from both animal and human studies suggests that hormonal contraceptives have the capacity to interfere with reward processing, particularly with sexual reward. Inconsistencies in this research stem from the use of convenience samples of women already taking hormonal contraceptives (increasing risk of a survivor effect) and the inclusion of multiple formulations. As with all research investigating the effects of hormonal contraceptives on the brain, there continues to be an urgent need for prospective, placebo-controlled studies, ideally that separate oestrogen and progestin components. Measuring the concentrations of these specific components is important to help determine whether dose–response relationships exist, as these can support causal inference. Finally, given that hormonal contraceptive formulations result in variable suppression of endogenous hormones, measurement of oestradiol, progesterone and other neuroactive steroids in future studies will assist in elucidating underlying mechanisms.
Clinical implications
The interactions between ovarian hormones, hormonal contraceptives and reward processing carry significant clinical implications, particularly in relation to management of menopause, hormonal contraceptive prescribing and psychiatric conditions such as PMDD, substance use disorders and major depressive disorder. It is important to note that certain women demonstrate significant sensitivities to changes in ovarian hormones across the lifespan, such that PMDD is associated with the incidence of postpartum depression and depression during the menopause transition (Eccles Reference Eccles and Sharma2023). Also, as discussed above, certain individuals may be particularly sensitive to hormonal contraception’s effects on reward. Therefore, clinically relevant influences of ovarian hormones on reward processing may occur in some but not all women.
Variations and eventual decreases in oestradiol during the menopause transition are already associated with new onset or exacerbations of major depressive disorder as well as other psychiatric disorders. It is reasonable to assume that loss of oestradiol’s facilitation of reward is partly to blame for this increase in vulnerability. As described in the section on oestradiol, treatment with this hormone helps to ameliorate deficits in reward processing in menopausal women in terms of both sexual and non-sexual reward. In a recent study of women with perimenopausal-onset major depressive disorder, transdermal oestradiol specifically decreased symptoms of anhedonia (Walsh Reference Walsh, Prim and Gibson2025). This emphasises one of the many ways by which hormone therapy with oestradiol for menopausal women can assist with brain health and quality of life.
Preliminary data suggest that there may be benefits for sexual function and mood with the use of formulations containing 17β-oestradiol versus ethinylestradiol, and that the higher doses of progestins in hormonal contraceptives such as levonorgestrel may increase the risk of depression (Larsen Reference Larsen, Mikkelsen and Ozenne2024). As discussed above, individuals with existing brain vulnerabilities to deficits in reward processing, such as those with ACEs, may be at increased risk for experiencing decreases in sexual desire with hormonal contraceptives (Novick Reference Novick, Johnson and Lazorwitz2022c, Reference Novick, Stoddard and Johnson2023). Ongoing research will ideally allow for more definitive guidance for physicians and their patients to select the types of hormonal contraceptive that allow for healthy reward processing and the least risk of psychiatric exacerbation. This would be especially important for women with PMDD and major depressive disorder, who already show deficits in reward processing.
In the context of substance use disorders, research on progesterone in both humans and animals has provided the intriguing possibility that natural forms of this hormone might be used therapeutically for alcohol, nicotine and stimulant use disorders (Peltier Reference Peltier and Sofuoglu2018). Some research suggests that therapeutic effects of progesterone in substance use disorders might be mediated through its neuroactive steroid metabolites, such as allopregnanolone (Peltier Reference Peltier and Sofuoglu2018): such direct administration of these neuroactive steroids, as opposed to their precursors, might help improve treatment efficacy, avoid side-effects and circumvent individual differences in steroid metabolism.
Conclusion
Endogenous ovarian hormones play a ubiquitous role in brain function, and their role in reward processing is no exception. Despite the limitations of human research to date, the facilitation of reward processes by oestradiol and inhibition by progesterone remains a general theme in the literature. This has particular relevance when it comes to hormonal contraceptives, which depending on formulation and delivery method, may suppress endogenous oestradiol. Even without suppression of ovarian hormones, the derivatives of oestradiol and progesterone used in hormonal contraceptives may have differential effects on reward neurobiology, although this requires further study.
Ultimately, the intersection of ovarian hormones, hormonal contraceptives and reward processing underscores the need for further research and tailored clinical strategies. Prospective, placebo-controlled studies focusing on the distinct roles of oestrogens and progestins are essential to unravel the nuances of these interactions and optimise outcomes for both healthy women and those with relevant clinical conditions.
MCQs
Select the single best option for each question stem
-
1 Which of the following best describes oestradiol’s influence on reward processing?
-
a It inhibits dopamine release in the nucleus accumbens
-
b It enhances dopaminergic activity and reward responsiveness
-
c It reduces opioid receptor density in reward regions
-
d It primarily suppresses GABAergic tone in the amygdala
-
e It increases cortisol release via the hypothalamic–pituitary–adrenal axis.
-
-
2 During which menstrual cycle phase are women most likely to show reduced activation of reward-related brain regions on fMRI?
-
a Mid-follicular phase
-
b Ovulatory phase
-
c Luteal phase
-
d Early follicular phase
-
e Perimenstrual phase.
-
-
3 In clinical practice, which scenario most warrants a discussion about possible effects of hormonal contraceptives on reward processing?
-
a A woman reports increased libido after starting a combined oral contraceptive
-
b A woman experiences persistent anhedonia and loss of sexual desire after beginning a new hormonal contraceptive
-
c A woman with premenstrual dysphoric disorder seeks to reduce cyclical mood symptoms
-
d A woman using a copper intrauterine device (IUD) presents with irritability
-
e A woman reports mood improvement after discontinuing a selective serotonin reuptake inhibitor.
-
-
4 Allopregnanolone is best described as:
-
a a dopamine reuptake inhibitor with antidepressant effects
-
b a metabolite of progesterone that acts as a positive allosteric modulator at GABAA receptors
-
c a synthetic progestin used in most oral contraceptives
-
d an antagonist at the mu-opioid receptor implicated in reward regulation
-
e a serotonin 2A receptor partial agonist contributing to psychedelic effects.
-
-
5 Which of the following clinical populations may be most sensitive to hormone-related disruptions in reward processing?
-
a Women with a history of adverse childhood experiences
-
b Women using copper IUDs
-
c Men with alcohol use disorder
-
d Postmenopausal women on transdermal oestradiol
-
e Adolescent girls with high baseline anxiety.
-
MCQ answers
-
1 b
-
2 c
-
3 b
-
4 b
-
5 a
Data availability
Data availability is not applicable to this article as no new data were created or analysed in this study.
Author contributions
A.M.N. conceptualised the article and participated in the writing of the original draft as well as the review and editing of the final manuscript. C.N.E. and E.R.W. participated in the writing of the original draft as well as the review and editing of the final manuscript.
Declaration of interest
A.M.N. received grant support from the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) (grant K23HD11043) during the writing of this article. C.N.E. received grant support from the NICHD (grant R01HD105771) during the writing of this article; she was an investigator for a multisite clinical trial conducted by Sage Therapeutics, and is a consultant to EmbarkNeuro, Skyland Trail and Health Rhythms and a member of the scientific advisory board of Babyscripts. No funding or involvement from these entities was used to support the current work, and all views expressed are solely those of the authors.



eLetters
No eLetters have been published for this article.