The prevalence of asthma in school children has rapidly increased over the past few decades, both worldwide and in Taiwan. Dietary pattern changes may be one of the contributing factors(Reference Allan and Devereux1). Observational studies have shown that intake of vitamin E, vitamin C, carotenoids, Se, polyphenols, vegetables, fruit and n-3 PUFA are negatively associated with asthma, while that of n-6 PUFA is positively associated with asthma(Reference Allan and Devereux1, Reference Lee, Yang and Chuang2). However, supplementation with antioxidants or n-3 PUFA alone in adults with asthma has either modest or no clinical benefits(Reference Allan and Devereux1, Reference Cassano3). The Dietary Approach to Stop Hypertension trial, an unprecedented study that combined multiple beneficial food components, demonstrated a significant and medication-equivalent effect on blood pressure reduction in hypertensive patients(Reference Appel, Moore and Obarzanek4). Therefore, we intended to adopt this combinatorial principle to examine the effect of multiple beneficial food factors on asthma control, using food concentrate to increase the feasibility.
A fruit plus vegetable concentrate, high in antioxidants, was used in the present study. A previous study has shown that fruit plus vegetable concentrate supplementation can increase plasma antioxidant levels and reduce CHD risk factors(Reference Samman, Sivarajah and Man5, Reference Plotnick, Corretti and Vogel6). As reactive oxygen species play a key role in inflammatory initiation and amplification in asthmatic airways(Reference Nadeem, Masood and Siddiqui7), supplementing fruit plus vegetable concentrate may dampen or prevent the inflammatory response in asthma by restoring the oxidant–antioxidant balance or through other potential mechanisms.
Fish oil is rich in EPA and DHA, which can inhibit cyclo-oxygenase and lipo-oxygenase enzyme activity, and decrease 2-series PGE2 and 4-series leukotrienes (leukotriene E4, leukotriene B4). The 2-series PG have an immunomodulatory function, which modifies the activity of macrophages and lymphocytes and suppresses the production of T-helper 1 (Th1)-related cytokines, promoting the expression of the T-helper 2 (Th2) phenotype associated with asthma. The 4-series leukotrienes have pro-inflammatory properties and are important mediators in the production of airway obstruction(Reference Chilton, Rudel and Parks8). Fish oil supplementation reduces the concentration of inflammatory mediators (PGE2, leukotriene E4 and leukotriene B4), eosinophils and TNF-α(Reference Mickleborough, Lindley and Ionescu9–Reference Moreira, Moreira and Delgado11). However, the beneficial effects on forced expiratory volume in 1 s (FEV1), bronchial challenge tests, asthma symptoms and medication use have not been consistent(Reference Mickleborough, Lindley and Ionescu9–Reference Villani, Comazzi and De Maria13). This is possibly due to the varied dosage and duration of the fish oil treatment.
The immunomodulatory effects of several probiotics have already been confirmed. Dendritic cells appear central to directing the beneficial immune system effects of probiotic bacteria, and in translating microbial signals from the innate to the adaptive immune system. Regulatory T cells are also emerging as potentially key players in probiotic-mediated responses(Reference Forsythe14). Recent clinical trials have shown that supplementation with Lactobacillus gasseri (Reference Chen, Jan and Lin15) and Bifidobacterium breve (Reference van de Pol, Lutter and Smids16) significantly improves pulmonary function and significantly reduces systemic production of Th2 cytokines in asthma patients. Therefore, supplementation with probiotics could potentially be of benefit to asthma control. The present study used Lactobacillus salivarius as the probiotic supplement. This particular probiotic has been shown to decrease allergen-induced airway hyperresponsiveness and elevate levels of interferon-γ in animal models(Reference Li, Lin and Hsueh17).
The aim of the present study was to investigate the combined effects of fruit plus vegetable concentrate, fish oil and probiotic supplements on asthma, related parameters and medication use.
Methods
The present study is a 16-week, double-blind, randomised, placebo-controlled trial in school children with asthma that tests the efficacy of combining the following supplements: two ‘fruit and vegetable’ capsules, three to five ‘fish oil’ capsules and two ‘probiotic’ capsules per d (FVFP supplement). The present study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human subjects/patients were approved by the institutional review board of Academia Sinica, Taiwan. The trial was registered in clinicaltrials.gov: NCT01366222. Written informed consent was obtained from all the subjects/patients.
Study participants
Participants who had ever been diagnosed with asthma were recruited from twenty-three elementary schools in metropolitan Taipei through parent conferences. All potential children and their parents had to sign the informed consent form before further eligibility screening was carried out by a paediatrician specialised in asthma. The inclusion criteria of the present trial were as follows: (1) age between 10 and 12 years and (2) with mildly or moderately persistent asthma based on the Global Initiative for Asthma guidelines. The exclusion criteria were as follows: (1) participation in other therapeutic studies in the previous 6 months; (2) regular use of oral corticosteroids, high-dose multivitamin supplements, fish oil supplements or probiotic supplements within 3 months before screening; (3) coagulation disorders or other serious illness; (4) inability to swallow capsules; (5) a resting FEV1 less than 1 litre and (6) being a vegetarian.
Dietary supplement design and preparation
The FVFP supplement includes three kinds of dietary supplements. Each ‘fruit and vegetable’ capsule contained a 400 mg concentrate derived from grapes, plums, blueberries, raspberries, cranberries, cherries, cowberries, strawberries, artichokes, beets, carrots, broccoli, white cauliflower, kale, celery, spinach and tomatoes (Schiff Nutrition Group, Inc.). Each capsule contained 986 Oxygen Radical Antioxidant Capacity (ORAC) units, according to Brunswick Laboratory data, which is equal to approximately two servings of fruits and vegetables. One serving of fruits or vegetables contains 350 ORAC units(Reference Beer, Myers and Sorenson18). Each ‘fish oil’ capsule (500 mg/capsule) contained 230 mg of EPA and 125 mg of DHA (DSM Nutritional Products Limited). The number of fish oil capsules given to individual children was determined according to their body weight: three capsules per d for those weighing 24·8–32·6 kg, four capsules for those weighing 34–41·4 kg and five capsules for those weighing 45·3–59·2 kg. The daily dosage of EPA plus DHA was 1065–1775 mg, which is equal to 1–1·5 servings of n-3-rich oily fish. Each ‘probiotic’ capsule (1000 mg/capsule) contained 2 × 109 colony-forming units of L. salivarius PM-A0006 (ProMD Biotech Company, Limited).
The placebo capsules were indistinguishable in appearance from the supplement capsules. ‘Fruit and vegetable’ placebo capsules (400 mg/capsule) were composed of maize starch (Nihon Shokuhin Kako Company, Limited) and had 79 ORAC units. ‘Fish oil’ placebo capsules contained 500 mg of maize oil, which was rich in 58 % n-6 PUFA (God-bene Company, Limited). ‘Probiotic’ placebo capsules (1000 mg/capsule) were composed of yogurt-flavoured maize starch without L. salivarius (ProMD Biotech Company, Limited).
Intervention
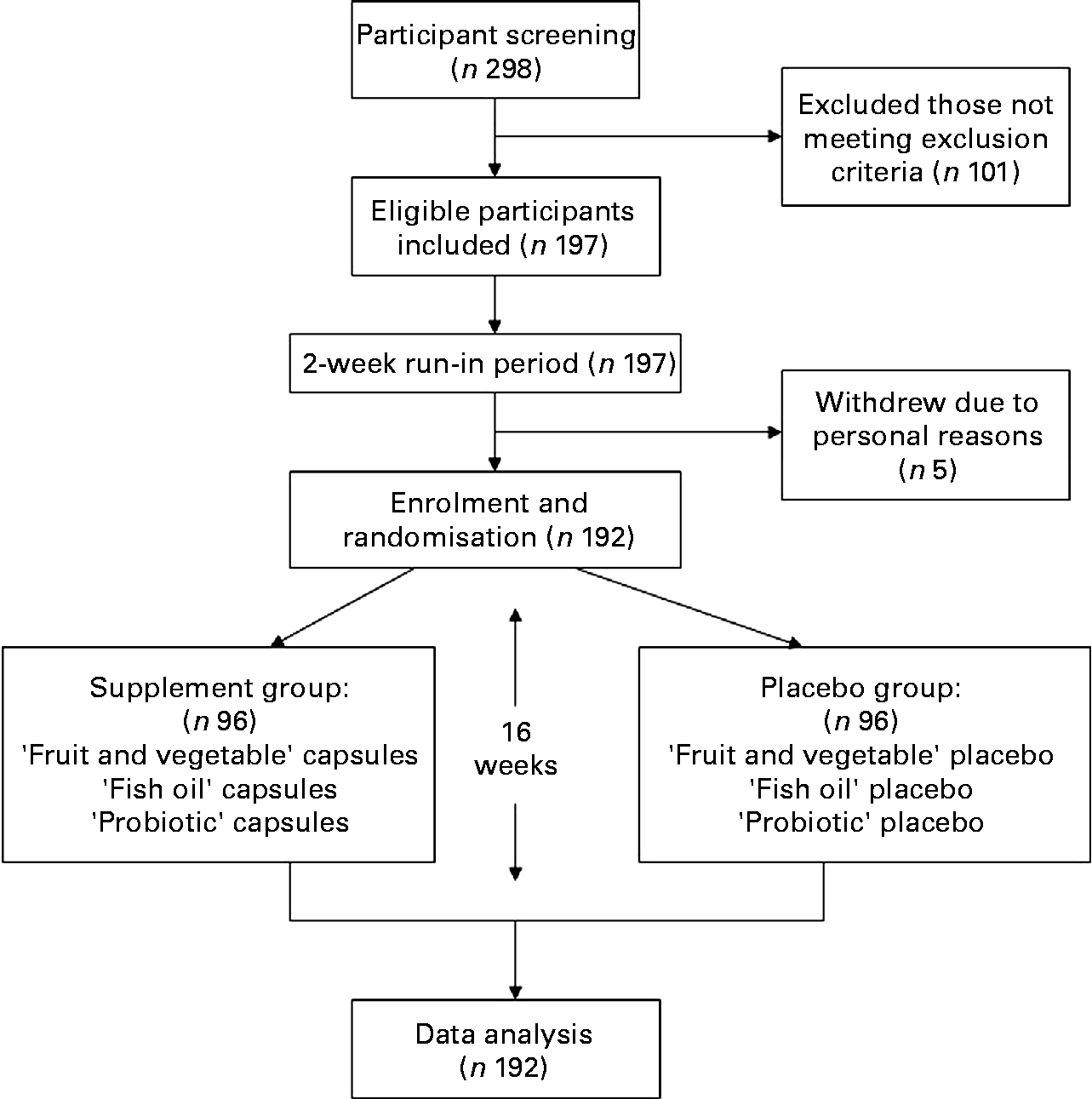
All 197 eligible participants were given placebo capsules during the 2-week run-in period. Of the 197 children, five dropped out of the trial due to personal reasons. The remaining 192 children were randomly assigned to either the supplement group or the placebo group (Fig. 1). Stratified randomisation was carried out with stratification by sex, age and clinical site (children's school), as boys or younger children are known to have a higher risk of asthma. In addition, local environmental exposures such as weather, allergens and air pollution needed to be balanced between the experimental group and the control group.
Study design.

The capsules were distributed to the children in schools by on-site workers. The supplements were given to the children personally in the morning, to ensure that the children took the capsules from Monday to Friday. In addition, the personnel reminded the children to take their capsules at home on weekends. The children were asked to take all unused capsules back to school. During the intervention period, children saw doctors and maintained their regular lifestyle and exercise habits as usual. Participants were monitored via an interview for serious adverse events and side effects such as prolonged epistaxis, bleeding tendency and gastrointestinal symptoms.
Clinical assessment and outcomes
Assessments were carried out at baseline, and at weeks 8 and 16 of the trial. Information on socio-demographic characteristics, risk factors for asthma, medications, usual dietary intake and the use of dietary supplements was collected at baseline. Dietary intake was measured using a twenty-eight-item FFQ to compare the frequency distribution of fruits, vegetables, n-3-rich oily fish and probiotic-related food intake between the two groups.
Asthma-related quality of life was measured using the Paediatric Asthma Quality of Life Questionnaire (PAQLQ) that consists of twenty-three items(Reference Juniper, Guyatt and Feeny19). The total score ranges from 1 to 7 (1 indicates maximum impairment and 7 indicates no impairment).
The Childhood Asthma Control Test(Reference Chen, Wang and Jan20) (CACT) was used to evaluate how well the children's asthma was controlled. The reliability and validity of this test have been demonstrated previously in Chinese children(Reference Chen, Wang and Jan20). The CACT was completed jointly by children and their parent(s) or caregiver(s). The CACT includes seven items and has a total score ranging from 0 to 27. The higher the score, the better the asthma control. Inadequately controlled asthma was defined as a CACT score less than 19(Reference Chen, Wang and Jan20).
Pulmonary function was measured using a portable spirometer (MiniSpir, Medical International Research, Inc.) according to the American Thoracic Society guidelines. The highest values of FEV1, forced vital capacity (FVC) and peak expiratory flow rate from the ‘American Thoracic Society best test’ were used in the analyses(21). FEV1 is an important clinical index of pulmonary function. The ratio of FEV1:FVC (or FEV1%) should be approximately above 80 % in healthy adults and above 90 % in healthy children. When the airway is obstructed, FEV1 and FEV1% are lower than the normal limit(Reference Bateman, Hurd and Barnes22). Participants were asked to avoid taking bronchodilators within 4 h of the test (8 h for long-acting bronchodilators). Pulmonary function was measured at approximately the same time of day at baseline and follow-up.
During the intervention period, daily medication usage was recorded by the parent/caregiver. The recorded information was checked by the study coordinator and was clarified with parents whenever necessary. Collected data included name, frequency and dosage of the medications and date of consumption. Information on short-acting inhaled bronchodilators, inhaled corticosteroids (ICS), oral steroids and leukotriene receptor antagonists was included in the statistical analysis.
The erythrocyte membrane arachidonic acid, EPA and DHA compositions were analysed using GC. The procedure has been described in detail elsewhere(Reference Chiu, Huang and Su23).
Sample size estimation
Differences in PAQLQ and FEV1 between the two groups were used to estimate the sample size. The standard deviation for the change in PAQLQ score was assumed to be 0·6(Reference Basaran, Guler-Uysal and Ergen24). We estimated that fifteen participants in each group would give 95 % power at 5 % significance level to detect a mean difference of 0·78 between the supplement and placebo group for the change in PAQLQ score between baseline and the end of the trial. We also used FEV1 to calculate the sample size. Assuming a standard deviation for the change in FEV1 of 0·41 litres(Reference Bousquet, D'Urzo and Hebert25), we estimated that 102 participants in each group would give 80 % power at the 5 % significance level to detect a mean difference of 0·16 litres between the supplement and the placebo group for the change in FEV1 between baseline and the end of the trial. Therefore, we aimed for a sample size of about 100 participants in each group.
Statistical analysis
Student's t test for continuous variables and χ2 test for categorical variables were used to compare characteristics between the treatment and control groups. For the outcome analysis, the Student's t test was used to compare the change from baseline to week 8 and week 16 between the two groups in PAQLQ score (Table 2), CACT score (Table 2), pulmonary function parameters (Table 3) and the mean frequency of medication use (Fig. 3). Mixed models were used to control for potential confounding factors including age, sex, BMI, cluster effect (children's school) and seasonal effect. McNemar's test was used to examine the change in proportion of children using medication during the 1st period (1st to 8th week) and the 2nd period (1st to 16th week) of the trial. The proportional Z test was used to compare the difference in the change in proportion using medication between the two groups (Fig. 2).
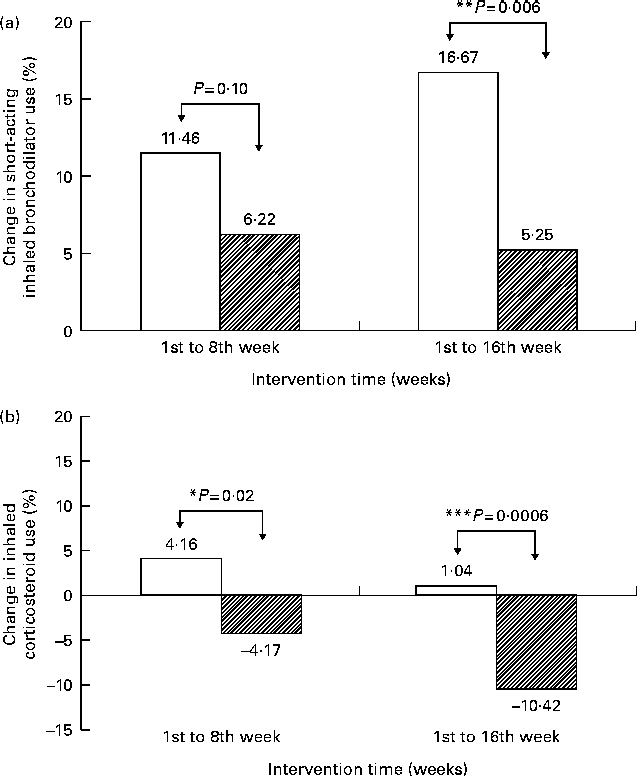
Change in percentage of children using (a) short-acting inhaled bronchodilators and (b) inhaled corticosteroids from baseline by supplementation status. The percentage of children using (a) and (b) was 15·6 and 21·9 % at baseline in the supplement (□) group, and 19·8 and 16·7 % in the placebo (![]() ) group. McNemar's test was used to examine the change in proportion of children using medication during the 1st period (1st to 8th week) and the 2nd period (1st to 16th week) of the trial. The proportional Z test was used to compare the difference in the change in proportion using medication between the two groups. * P< 0·05, ** P< 0·01, *** P< 0·001.
) group. McNemar's test was used to examine the change in proportion of children using medication during the 1st period (1st to 8th week) and the 2nd period (1st to 16th week) of the trial. The proportional Z test was used to compare the difference in the change in proportion using medication between the two groups. * P< 0·05, ** P< 0·01, *** P< 0·001.

Results
A total of 192 participants completed the trial. The follow-up rate achieved 100 % and no serious adverse events or side effects were reported (Fig. 1).
Baseline characteristics and participant compliance
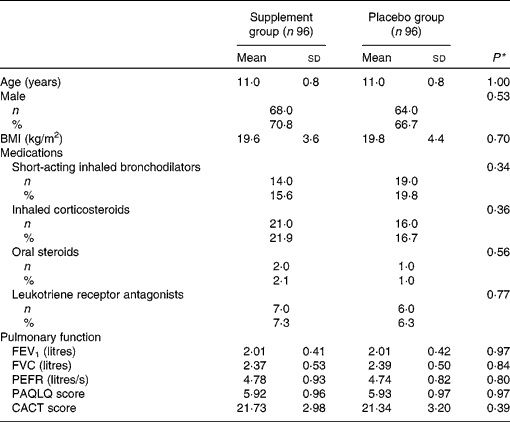
At baseline, the distributions of age, sex, BMI, medication usage, pulmonary function, PAQLQ scores and CACT scores were similar in the supplement and placebo groups (Table 1). The frequency of FVFP formula-related foods (vegetables, fruit, oily fish, Yakult and yogurt) and other foods (milk, cheese, eggs, fish, shellfish, meat, soya bean milk, soya bean products, sweet drinks, desserts and candy) were non-significantly different between the two groups at baseline (Table S1, available online) as well as during the intervention (Table S2, available online). On average (Table S1, available online), these children consumed vegetables about three times per d and fruit once per d. In addition, they consumed candy, sweet drinks or desserts about ten times per week, milk five times per week, yogurt or Yakult three times per week and cheese 1·5 times per week, and fish four times per week. Around one-third of these fish were oily fish. Information on portion sizes was not requested. According to the 24 h dietary recall data from the Nutrition and Health Survey in Taiwan, elementary school children consume about 33 g of vegetables (raw and uncooked form), 150 g of fruits and 40 g of fish each time. Therefore, children in the present study ingested one serving of vegetables and 1·5 servings of fruit per d, and 1·5 servings of oily fish per week, but relatively little Lactobacillus-containing foods.
Baseline socio-demographic and clinical characteristics in the supplement and placebo groups (Mean values and standard deviations; number of subjects and percentages)

FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; PEFR, peak expiratory flow rate; PAQLQ, Paediatric Asthma Quality of Life Questionnaire; CACT, Childhood Asthma Control Test.
* P value was estimated using the Student's t test for continuous variables and χ2 test for categorical variables.
Participant compliance was assessed by capsule counting and by the level of EPA and DHA in the erythrocyte membrane. The capsule consumption rate was 91 % (no. of consumption days/total no. of intervention days) in both groups. The levels of EPA, DHA and arachidonic acid in the erythrocyte membrane were non-significantly different between the two groups at baseline. However, at the end of the trial, the levels of EPA and DHA in the supplement group were significantly higher than those in the placebo group (EPA: 1·66 v. 0·77 %, P< 0·001; DHA: 6·77 v. 5·97 %, P= 0·001). In contrast, the arachidonic acid:EPA ratio (2·10 (sd 0·49) v. 2·70 (sd 10·61), P< 0·001) and n-6:n-3 fatty acid ratio (7·32 (sd 3·84) v. 16·40 (sd 7·56), P< 0·001) were significantly lower in the supplement group than in the placebo group. These results demonstrate that participants had good compliance.
Paediatric Asthma Quality of Life Questionnaire and Childhood Asthma Control Test evaluation
The change in PAQLQ and CACT scores from baseline to week 8 and week 16 was non-significantly different between the two groups (Table 2).
Mean change in the Paediatric Asthma Quality of Life Questionnaire (PAQLQ) and the Childhood Asthma Control Test (CACT) scores from baseline in the supplement and placebo groups (Mean values and 95 % confidence intervals)

* Mean values were significantly different between the two groups (Student's t test).
† A mixed model was used to adjust for age, sex, BMI, cluster effect (children's school) and seasonal effect (intervention phase) when comparing the two groups.
‡ The mean PAQLQ score was 5·92 (sd 0·96) at baseline in the supplement group and 5·93 (sd 0·97) in the placebo group; the mean CACT score was 21·73 (sd 2·98) at baseline in the supplement group and 21·34 (sd 3·20) in the placebo group.
Assessment of change in medication usage
The supplement group demonstrated a smaller increase in the percentage of children using short-acting inhaled bronchodilators than the placebo group during the 1st to the 8th week (P= 0·10) and during the 1st to the 16th week (P= 0·006) (Fig. 2(a)). In addition, the proportion of children using ICS decreased in the supplement group, but increased in the placebo group. This difference in the change in use of ICS between the supplement and placebo groups was statistically significant for the 1st to the 8th week (P= 0·02) and for the 1st to the 16th week (P= 0·0006) (Fig. 2(b)). Use of oral steroids and leukotriene receptor antagonists was relatively infrequent compared with the earlier two medicines, and the percentage change in their use was non-significantly different between the two groups (data not shown).
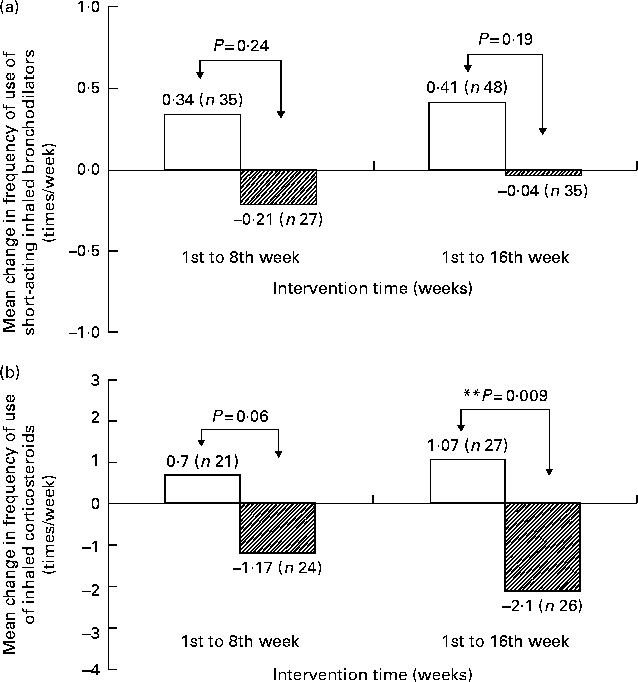
We also evaluated the frequency of medication use in children who were taking medication during the trial period (Fig. 3). Use of short-acting inhaled bronchodilators during the 1st to the 8th week (P= 0·24) declined by 0·21 times per week in the supplement group, while it increased by 0·34 times per week in the placebo group. Use of this medication during the 1st to the 16th week (P= 0·19) declined by 0·04 times per week in the supplement group, while it increased by 0·41 times per week in the placebo group (Fig. 3(a)). In addition, the supplement group demonstrated a downward trend in the frequency of use of ICS, whereas the placebo group demonstrated an upward trend. The change in frequency of the use of ICS from the 1st to the 8th week (P= 0·06) and from the 1st to the 16th week (P= 0·009) was significantly different between the two groups (Fig. 3(b)). This difference remained in the mixed model after controlling for age, sex, BMI, cluster effect (children's school) and seasonal effect.
Change in mean frequency of medication use in children using medication by supplementation status. (a) Short-acting inhaled bronchodilators and (b) inhaled corticosteroids. The mean frequency of use of (a) and (b) was 0·82 (sd 1·51) and 5·70 (sd 6·80) times/week at baseline in the supplement (□) group, and 0·85 (sd 1·14) and 2·86 (sd 4·22) times/week in the placebo (![]() ) group. Student's t test was used to compare the difference between the two groups of the change from baseline to week 8 and week 16. ** P< 0·01.
) group. Student's t test was used to compare the difference between the two groups of the change from baseline to week 8 and week 16. ** P< 0·01.

Pulmonary function evaluation
In the analysis of pulmonary function, the supplement group showed a significantly higher increment in FEV1 (P= 0·01), FVC (P= 0·01) and FEV1:FVC ratio (P= 0·008) in the 16th week compared with the placebo group. These significant associations remained, even after controlling for confounders (Table 3).
Mean change in pulmonary function parameter estimates from baseline in the supplement and placebo groups (Mean values and 95 % confidence intervals)

FVC, forced vital capacity; FEV1, forced expiratory volume in 1 s; PEFR, peak expiratory flow rate.
* Mean values were significantly different between the two groups (Student's t test).
† A mixed model was used to adjust for age, sex, BMI, cluster effect (children's school) and seasonal effect when comparing the two groups.
‡ At baseline, the FVC was 2·39 (sd 0·50) litres in the supplement group and 2·37 (sd 0·53) litres in the placebo group; the FEV1 was 2·01 (sd 0·42) litres in the supplement group and 2·01 (sd 0·41) litres in the placebo group; the PEFR was 4·78 (sd 0·93) litres/s in the supplement group and 4·74 (sd 0·82) litres/s in the placebo group; the FEV1:FVC ratio was 85·4 (sd 6·9) % in the supplement group and 84·8 (sd 7·7) % in the placebo group.
Discussion
This double-blind, 16-week, randomised intervention trial showed that FVFP supplementation had a significantly beneficial effect on asthma symptom control in children residing in metropolitan Taipei. Although there was no significant improvement in PAQLQ and CACT scores, the FVFP supplement reduced the usage percentage and frequency of short-acting inhaled bronchodilators and ICS, and improved several measures of pulmonary function (FEV1, FVC and FEV1:FVC).
To date, there have been few trials investigating the supplemental effect of multiple beneficial food components on asthma control. Findings have been controversial, as to the effects of single supplements such as antioxidative vitamins, minerals, vegetables or fruits, fish oil and probiotics alone(Reference Mickleborough, Lindley and Ionescu9–Reference Villani, Comazzi and De Maria13, Reference Chen, Jan and Lin15, Reference Fogarty, Lewis and Scrivener26–Reference Stockert, Schneider and Porenta37). A randomised, double-blind clinical trial of supplementation with vitamin C(Reference Fogarty, Lewis and Scrivener26) (1 g, 16 weeks), vitamin E(Reference Pearson, Lewis and Britton27) (500 mg, 6 weeks), vitamins C+E(Reference Hernandez, Zhou and Zhou28) (1 g+500 mg, 12 weeks), Mg(Reference Fogarty, Lewis and Scrivener26, Reference Kazaks, Uriu-Adams and Albertson29) (450 mg for 16 weeks or 340 mg for 6·5 months) and Se(Reference Shaheen, Newson and Rayman30) (100 mg, 24 weeks) in adults aged 16–60 years showed no beneficial effects on pulmonary function, asthma symptoms or medication use, despite high levels of antioxidative vitamins or minerals being observed in the blood. Only one trial with Mg(Reference Gontijo-Amaral, Ribeiro and Gontijo31) supplementation (300 mg, 2 months) in children aged 7–19 years (n 37) found that Mg reduced bronchial reactivity to methacholine (PC20 (provocative concentration of methacholine causing a 20 % fall in FEV1), P< 0·05), the number of days of asthma exacerbation episodes (P= 0·002) and use of inhaled salbutamol (P< 0·0001). However, there was no significant change in FEV1, FVC or FEV1:FVC ratio. In another study, asthmatic adults supplemented with tomato products for 7 d(Reference Wood, Garg and Powell32), or asthmatic children supplemented with fruits (apples, oranges, pears and pear) for 1 year(Reference Fogarty, Antoniak and Venn33) demonstrated no improvement. Supplementation with purple passion fruit peel powder for 4 weeks has been reported to reduce the prevalence of wheeze (P< 0·001), cough (P< 0·001) and shortness of breath (P< 0·05) in asthmatic adults aged 18–60 years(Reference Watson, Zibadi and Rafatpanah34). Short-term trials in asthmatic adults failed to demonstrate improvement in pulmonary function, asthma symptoms or medication use with low-dose fish oil(Reference Schubert, Kitz and Beermann10, Reference Moreira, Moreira and Delgado11) (EPA+DHA 780 mg for 2 weeks or 690 mg for 5 weeks), whereas long-term (1 year) supplementation with low-dose fish oil(Reference Dry and Vincent35) (1 g) demonstrated significant improvement in FEV1 up to 9 months after the intervention. High-dose, short-term fish oil(Reference Mickleborough, Lindley and Ionescu9, Reference Villani, Comazzi and De Maria13) supplementation (EPA+DHA 5·4 g for 3 weeks or 2·58 g for 4 weeks) can decrease bronchial hyperreactivity (P< 0·05) in adults with exercise-induced bronchoconstriction or seasonal asthma. Using a dose of fish oil similar to that in the present study, a long-term (10 months) randomised controlled trial in twenty-nine Japanese asthmatic children found that fish oil supplementation reduces bronchial reactivity to methacholine (PC20, P< 0·05) and asthma score (P< 0·05) at 24 weeks(Reference Nagakura, Matsuda and Shichijyo12). In trials of probiotic supplements, a short-term (2 months) trial using L. gasseri A5 supplements in asthmatic children aged 6–12 years found that probiotics can improve peak expiratory flow rate levels and CACT score (P< 0·05), and can significantly decrease levels of TNF-α, interferon-γ, IL-12 and IL-13 production by peripheral blood mononuclear cells(Reference Chen, Jan and Lin15). However, supplementation with L. casei (Reference Giovannini, Agostoni and Riva36) for 12 months or non-pathogenic Enterococcus faecalis (Reference Stockert, Schneider and Porenta37) for 17 weeks had no beneficial effect on asthma control in asthmatic children.
Two trials have investigated the beneficial effects of multiple nutrient supplements, which combine fish oil and antioxidative vitamins and minerals, on asthma control. A randomly assigned, 6-week, cross-over intervention, placebo, self-controlled trial in sixty children (aged 4–11 years) with moderately persistent asthma investigated the effects of supplementation with each of fish oil (EPA+DHA 300 mg), vitamin C (200 mg) and Zn (15 mg) and a combination of the three. The study found that supplementation with a single nutrient significantly improved the CATC score, FEV1 and sputum inflammatory markers (P< 0·001). Significant improvements were also observed in those taking combined supplements compared with any single supplement (P< 0·001)(Reference Biltagi, Baset and Bassiouny38). The second randomised, double-blind, placebo-controlled, parallel group trial in forty-three children (aged 6–14 years) with mildly to moderately persistent asthma investigated the supplementation of a novel nutritional formula (rich in EPA+DHA 300 mg, antioxidative vitamins and minerals) and found that it reduced bronchial reactivity to methacholine (PC20, P< 0·05) and decreased inflammatory markers (serum IgE, eNO and eosinophil cationic protein). However, asthma symptoms and bronchodilator usage were non-significantly different compared with the placebo group. In addition, although the FEV1 and FEV1:FVC ratio increased to 130 ml (placebo group: 20 ml) and 3·8 % (0 %) from baseline to the 12th week, the difference between the treatment and control groups was not statistically significant(Reference Covar, Gleason and Macomber39).
The present study is the first to combine multiple beneficial food components in the forms of food extract concentrates rather than pure synthetic compounds. The present findings are superior to other studies investigating a single(Reference Mickleborough, Lindley and Ionescu9–Reference Moreira, Moreira and Delgado11, Reference Villani, Comazzi and De Maria13, Reference Fogarty, Lewis and Scrivener26–Reference Shaheen, Newson and Rayman30, Reference Wood, Garg and Powell32, Reference Fogarty, Antoniak and Venn33, Reference Dry and Vincent35–Reference Stockert, Schneider and Porenta37) or multiple nutrient supplements(Reference Hernandez, Zhou and Zhou28, Reference Covar, Gleason and Macomber39), as not only did we demonstrate a reduction in the percentage and frequency of use of short-acting inhaled bronchodilators and ICS, but we also demonstrated that use of our FVFP supplement significantly increased pulmonary function. The PAQLQ and CACT scores showed no significant difference between the two groups in the present study. This could be because children in both groups were prescribed with medication whenever needed, as the national health insurance scheme in Taiwan covers routine paediatrician visits, resulting in diminished difference in scores between the two groups.
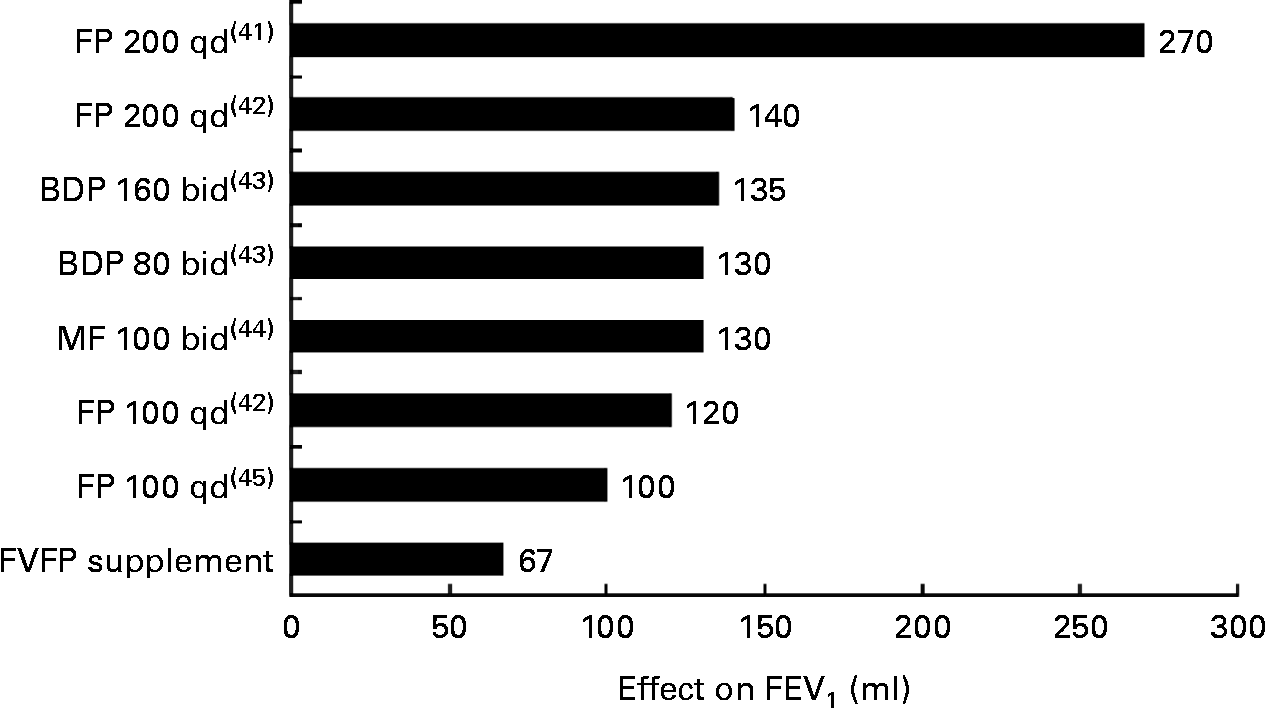
The global guideline for asthma management of the Global Initiative for Asthma recommends that rapid-acting inhaled β2-agonist (when required) and low-dose ICS are used for asthma control as the first step; and high-dose ICS or high-dose ICS plus long-acting inhaled β2-agonist combinations are used to augment asthma control for patients with poor control(Reference O'Byrne40). The FVFP supplement for 16 weeks was able to improve FEV1 by 67 ml and FEV1:FVC ratio by 3·2 % in asthmatic children. The improvement effect was almost a half in magnitude compared with various low-dose ICS(Reference LaForce, Pearlman and Ruff41–Reference Pauwels, Pedersen and Busse46) (Fig. 4). Although more clinical trials are needed to confirm the present finding and to compare the effects of FVFP supplementation or a diet rich in FVFP with various asthmatic medications, a FVFP supplement or diet may be used as an adjuvant non-pharmacological therapy to the regular asthma medication. It may reduce the dosage of ICS and diminish side effects.
Forced expiratory volume in 1 s (FEV1) improvement (the improvement effect was the effect of intervention group minus that of the placebo group. More detailed information of the improved FEV1 and FEV1% with asthma medication are provided in Table S3 (available online)) after ‘fruit and vegetable’ capsules, ‘fish oil’ capsules and ‘probiotic’ capsules (FVFP supplementation) compared with asthma medications in mildly to moderately asthmatic children. FP, fluticasone propionate; qd, once per d; BDP, beclomethasone dipropionate; bid, twice per d; MF, mometasone furoate.

The nutrients contained in these dietary supplements can be equated to natural food intake in the diet. The two capsules of ‘fruit and vegeables’ are equivalent to a total of four servings of fruits and vegetables. The three to five capsules of ‘fish oil’ (EPA+DHA: 1065–1775 mg) are equivalent to one serving of Pacific saury (35 g, EPA+DHA = 1598 mg), 0·5–1 serving of tuna (30 g, EPA+DHA = 2132 mg) or 1·5–2·5 servings of salmon sashimi (30 g, EPA+DHA = 673 mg). The amount of probiotic provided is equivalent to one to two servings of yogurt. Other fermented foods are also rich in probiotics such as cheese, pickles or sauerkraut. The FVFP supplements that we provided are more comparable with what the children consumed on average, i.e. about one serving of vegetables and 1·5 servings of fruits per d, 1·5 servings of oily fish per week and three cups of either yogurt or Yakult per week. However, adding together the original dietary intake and supplements, the total amount of vegetables and fruits (6·5 servings), yogurt (1·5 servings) and oily fish (one serving) required are achievable through dietary means. A portion of common antioxidant-poor, but energy-rich foods served in the everyday life of a Taiwanese may be replaced, e.g. deep-fried pork chop or chicken breast (replaced by oily fish), pickled vegetables (by fresh vegetables), diluted and sweetened soya milk (by yogurt) and polished white rice (by brown rice).
The present study has several limitations. We did not use a factorial design to study the effects of the single items and the combined supplement, as it would have required a large sample size. Further studies may be needed to investigate the magnitude of the effects in individual items and whether there are interactions among the multiple food components. Moreover, future studies could investigate the beneficial effects of the FVFP supplement over a wider age range and in asthmatic patients with moderate-to-severe persistent asthma.
Conclusion
Combining vegetable and fruit concentrate, fish oil and probiotics significantly reduces the usage percentage and frequency of short-acting inhaled bronchodilators and ICS, and improved pulmonary function in asthmatic children. The reduction in medication use could help to avoid medication side effects and to reduce medical expenditure. The present study suggests that a dietary pattern featuring adequate amounts of vegetables and fruit, fish and probiotic foods should be recommended in asthma prevention guidelines.
Supplementary material
To view supplementary material for this article, please visit http://dx.doi.org/10.1017/S0007114512004692
Acknowledgements
We are indebted to the children and their families for participating in the present scientific investigation. We also thank Schiff Nutrition Group, Inc. for supplying the fruit plus vegetable concentrate and ProMD Biotech Company, Limited for supplying the probiotic capsules. The authors' responsibilities were as follows: S.-C. L. designed the research, conducted the research, analysed and interpreted the data and wrote the manuscript; W.-H. P. designed the research, interpreted the data and had responsibility for the final content; Y.-H. Y. collected and interpreted the data; S.-Y. C. provided statistical consultation and interpreted the data. S.-Y. H. analysed the fatty acid composition of erythrocyte membranes. All of the authors read and approved the final manuscript. None of the authors has any conflicts of interest to report. The present study was supported by a grant (NSC96-2628-B-001-025-MY2) from the National Science Council, Taiwan and by intramural funds from Institute of Biomedical Science, Academia Sinica and from the Institute of Population Health Sciences, National Health Research Institutes, Taiwan.