Introduction
Human alveolar echinococcosis (AE) is a perilous parasitic illness resulting from infections caused by the metacestode stage of the dog/fox tapeworm Echinococcus multilocularis. AE is primarily confined to the Northern Hemisphere, with notable endemicity in Eurasia (Wen et al., Reference Wen, Vuitton, Tuxun, Li, Vuitton, Zhang and McManus2019). The present global toll of human AE surpasses 680 000 disability-adjusted life years, a figure that is likely underestimated (Torgerson et al., Reference Torgerson, Devleesschauwer, Praet, Speybroeck, Willingham, Kasuga, Rokni, Zhou, Fevre, Sripa, Gargouri, Furst, Budke, Carabin, Kirk, Angulo, Havelaar and de Silva2015; Woolsey and Miller, Reference Woolsey and Miller2021). The natural lifecycle of E. multilocularis is sustained through a cycle involving canid definitive hosts (primarily foxes) and small rodents as intermediate hosts (chiefly voles). Nevertheless, a diverse array of mammals, including humans, can accidentally contract the infection by ingesting the parasite eggs excreted by the definitive hosts during defecation. These E. multilocularis metacestodes proliferate in the liver, forming malignant tumour-like masses, and can infiltrate nearby organs and tissues, leading to distant metastases. If not appropriately treated, parasite expansion will eventually lead to organ failure or death (Kern et al., Reference Kern, Menezes, Akhan, Mullhaupt, Vizcaychipi, Budke and Vuitton2017).
Invasive surgical resection of the whole parasite tissues represents the only curative treatment of AE, but in reality, surgery is reserved for early-stage disease (Brunetti et al., Reference Brunetti, Kern and Vuitton2010). AE typically remains asymptomatic during the first 5–15 years, and most patients seeking medical help present with locally advanced or metastatic stage, which renders radical resection unfeasible. Albendazole-based antiparasitic therapy remains the only option for inoperable cases. Albendazole suppresses metacestode growth but does not act parasiticidally (Lundstrom-Stadelmann et al., Reference Lundstrom-Stadelmann, Rufener, Ritler, Zurbriggen and Hemphill2019); therefore, albendazole requires life-long and high-dose administration to avoid parasite growth after treatment discontinuation. Problems with adverse effects and compliance are common consequences of long-term regimens (Reuter et al., Reference Reuter, Buck, Manfras, Kratzer, Seitz, Darge, Reske and Kern2004; Grüner et al., Reference Grüner, Kern, Mayer, Gräter, Hillenbrand, Barth, Muche, Henne-Bruns, Kratzer and Kern2017). With increasing numbers of patients and no alternative to albendazole, the development of novel, and preferably better, treatment options has become increasingly urgent.
Given the relatively small target population, AE has not attracted the attention of pharmaceutical companies to engage in preclinical drug development. Therefore, an important strategy to find alternative drugs is to focus on the repurposing of existing drugs that were originally developed for other diseases. Drug repurposing could accelerate the drug development process because of reduced costs, lower risk of failure and decreasing the time to market, which offers hope to neglected diseases such as AE (Meier et al., Reference Meier, Antillon, Burri, Chitnis, Endriss, Keiser, Moore, Muller, Penny, Voss, Maser and Utzinger2023; Hamid et al., Reference Hamid, Mäser and Mahmoud2024). In the last 2 decades, drug repurposing efforts have been directed against E. multilocularis infections. In the experimental therapy of AE, various antineoplastics and anti-infectives with promising in vitro activity against metacestodes have been followed up in rodent models, but only very few of them have shown a significant impact on metacestode growth compared to albendazole treatment in infected mice (Lundstrom-Stadelmann et al., Reference Lundstrom-Stadelmann, Rufener, Ritler, Zurbriggen and Hemphill2019; Xu et al., Reference Xu, Qian, Gao, Pang, Zhou, Zhu, Wang, Pang, Wu, Yu, Kong, Shi, Guo, Su, Hu, Yan, Feng and Fan2022). To date, only the antifungal agent amphotericin B, the broad-spectrum antiparasitic nitazoxanide and antimalarial mefloquine have been studied in humans as salvage therapy, but they have not been pursued due to limited efficacy and significant side-effects (Siles-Lucas et al., Reference Siles-Lucas, Casulli, Cirilli and Carmena2018; Burkert et al., Reference Burkert, Peters, Bloehdorn and Gruner2022).
In our previous studies, pyronaridine, an approved antimalarial, was identified as a promising drug candidate against cystic echinococcosis (CE) (Li et al., Reference Li, Wang, Yao, Wang, Li, Qi, Han, Ren, Dang, Han, Guo, Guo, Wang, Duan and Zhang2020). In this study, the anti-echinococcal effect of pyronaridine was further explored against the more aggressive and complicated AE in a clinically relevant hepatic AE murine model as a mimic of natural infection. The in vivo safety profile of the long-term administration of pyronaridine is also described in this report. In addition, a Food and Drug Administration (FDA)-approved drug library was screened in silico to discover diverse potential anti-echinococcal hits based on the structural similarity to pyronaridine. The obtained hits of interest were further tested against E. multilocularis protoscoleces and cultured small cysts.
Materials and methods
Materials
Pyronaridine tetraphosphate was synthesized in-house as previously described (Li et al., Reference Li, Wang, Yao, Wang, Li, Qi, Han, Ren, Dang, Han, Guo, Guo, Wang, Duan and Zhang2020). Amonafide, olopatadine hydrochloride, benzydamine hydrochloride, quinacrine hydrochloride, promethazine hydrochloride, lumefantrine and carazolol were purchased from Shanghai Aladdin Scientific Corporation (Shanghai, China); loratadine, pirenzepine hydrochloride, azasetron hydrochloride and alimemazine hemitartrate were purchased from TargetMol Chemicals Incorporated (Shanghai, China); and albendazole was purchased from Merck Limited (Beijing, China). RPMI 1640 culture medium (Cat# SH30809.01) and antibiotics (Cat# 15140122) were purchased from HyClone (Logan, UT, USA). Fetal calf serum (Cat# 10099148) was purchased from Gibco (Penrose, New Zealand).
Animals
Female BALB/c mice (aged 8–9 weeks, 21 ± 2 g) were provided by Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). The animals were housed in sterilized polycarbonate cages (25 cm × 35 cm) with sawdust bedding, and had free access to food and water. Mice were maintained in the animal centre of the First Affiliated Hospital of Xinjiang Medical University under a controlled temperature of 22–24°C, a relative humidity of 50–60% and a 12/12 h light/dark cycle.
Therapeutic efficacy study in a murine model of hepatic alveolar echinococcosis
The hepatic AE murine model was generated through liver portal vein injection of E. multilocularis protoscoleces as previously described (Li et al., Reference Li, Yang, Han, Li, Tian, Qi, An, Wu, Zhang, Han, Duan, Wang and Zhang2023). In brief, BALB/c mice were anesthetized with aerosolized isoflurane (4% in air). After shaving and sterilization with iodophor, a 2 cm longitudinal skin incision was made on the abdomens of the mice to expose the liver portal vein. A suspension containing E. multilocularis protoscoleces (500 viable protoscoleces in 200 μL saline per mouse) was injected into the portal vein. The incisions were then closed using absorbable silk suture. The animals were kept on a heat pad at 38°C for recovery. At 3 months post-infection, the animals were randomly allocated into the following groups (n = 8 per group): (1) unmedicated control, (2) 50 mg kg−1 albendazole, (3) 80 mg kg−1 albendazole, (4) 50 mg kg−1 pyronaridine and (5) 80 mg kg−1 pyronaridine. All treatments were administered once daily for 30 consecutive days by oral gavage in a volume of 0.2 mL, with pyronaridine dissolved in deionized water and albendazole suspended in 0.5% carboxymethyl cellulose (CMC). Unmedicated control individuals received an equal volume of vehicle (0.5% CMC). Uninfected mice (n = 4) were sham operated following the same surgical procedures as those for the infected animals, with the exception of protoscolex infection. During the entire course of treatment, animals were closely monitored for signs of hypoactivity, emaciation, anorexia and behavioural alterations.
At the end of the treatment period, the mice were allowed to recover for another 30 days, after which they were anesthetized with isoflurane, and blood samples were collected from the retro-orbital venous plexus. Plasma was separated by centrifugation at 4000 g for 15 min at 4°C and stored at −40°C until analysis. The animals were euthanized by CO2 and necropsy was conducted immediately thereafter. The treatment efficacy was evaluated by the average metacestode size and counts from each group.
The plasma sample from each animal was analysed as follows: the counts of red blood cells, blood platelets, white blood cells, lymphocytes and monocytes were measured using an automatic blood cell analyser (BC-5300Vet, Mindray Bio-Medical Electronics Co., Ltd., Shenzhen, China); and the levels of liver transaminases alanine aminotransferase (ALT), aspartate aminotransferase (AST), urea nitrogen (Urea) and creatinine (Crea) were determined using a fully automatic biochemical analyser (BS-120, Mindray Bio-Medical Electronics Co., Ltd.).
Histopathology
Liver tissue samples from each group were collected and fixed in 4% paraformaldehyde for 36 h, before dehydrating the samples and embedding in paraffin. Sections were cut using an ultramicrotome (Reichert-Jung, Vienna, Austria) and stained with haematoxylin and eosin. The sections were sealed with neutral balsam and observed microscopically.
Similarity searching
The 2D structures of an FDA-approved drug library (Cat# L1000) were obtained from TargetMol Chemicals Incorporated, with pyronaridine serving as the reference drug. The library was searched against their structural similarity to pyronaridine using the Tanimoto similarity coefficient, which represents the similarity between 2 compounds based on the fingerprint bits they match (Gimeno et al., Reference Gimeno, Ojeda-Montes, Tomas-Hernandez, Cereto-Massague, Beltran-Debon, Mulero, Pujadas and Garcia-Vallve2019). The similarity coefficient ranges remain between 0 and 1, where the higher the value, the more similar the compounds. The similarity calculation was performed using the open-source cheminformatics software RDKit (http://www.rdkit.org/).
Protoscolex killing assay
The E. multilocularis Xinjiang strain was maintained and propagated in the peritoneum of Kunming mice. From the metacestode material collected from infected mice, E. multilocularis protoscoleces were isolated and prepared as previously described (Li et al., Reference Li, Yang, Han, Li, Tian, Qi, An, Wu, Zhang, Han, Duan, Wang and Zhang2023). Protoscoleces (viability >95%) were aliquoted into 96-well plates with each well containing 200 protoscoleces. Eleven approved drugs and albendazole were dissolved in DMSO or deionized water and applied to the protoscoleces (final concentrations: 5, 10, 20, 30 or 40 μ m). Control protoscoleces received DMSO (0.1% v/v) only. The plates were incubated under 5% CO2 at 37°C for 24 h. The viability of the protoscoleces was evaluated using a methylene blue staining test. The experiments were performed in triplicate.
In vitro cultured small cysts killing assay
E. multilocularis protoscoleces were cultured in RPMI 1640 culture medium supplemented with 20% fetal calf serum and antibiotics at 37°C under 5% CO2 for 4–6 weeks to obtain in vitro cultured small cysts (diameter: 0.2–1 mm). In vitro cultured small cysts killing assays were conducted in 12-well plates (5–10 small cysts per well). Eleven approved drugs and albendazole were incubated with these cultured small cysts (final concentrations: 10, 20 or 30 μ m). After 24 h of incubation, the small cysts of each well were collected, washed with phosphate-buffered saline 3 times and observed microscopically. A cultured small cyst is scored as collapsed when its germinal layer collapses and shrinks, becoming clearly detached from the laminated layer (Li et al., Reference Li, Wang, Yao, Wang, Li, Qi, Han, Ren, Dang, Han, Guo, Guo, Wang, Duan and Zhang2020; Lopez et al., Reference Lopez, Pensel, Fabbri, Albani, Elissondo, Gambino and Elissondo2022).
Statistical analysis
GraphPad Prism software (version 9.4.0, GraphPad Software, Boston, MA, USA) was used for data processing and statistical analysis. The data are expressed as the mean ± s.e.m. Multiple comparisons between more than 2 groups were analysed using one-way analysis of variance (ANOVA) followed by Tukey's post hoc test. In the case of heterogeneity of variance, the Welch and Brown–Forsythe ANOVA test was performed. A P value < 0.05 was considered statistically significant.
Results
Oral treatment with pyronaridine reveals significantly better therapeutic efficacy than albendazole in a clinically relevant AE model
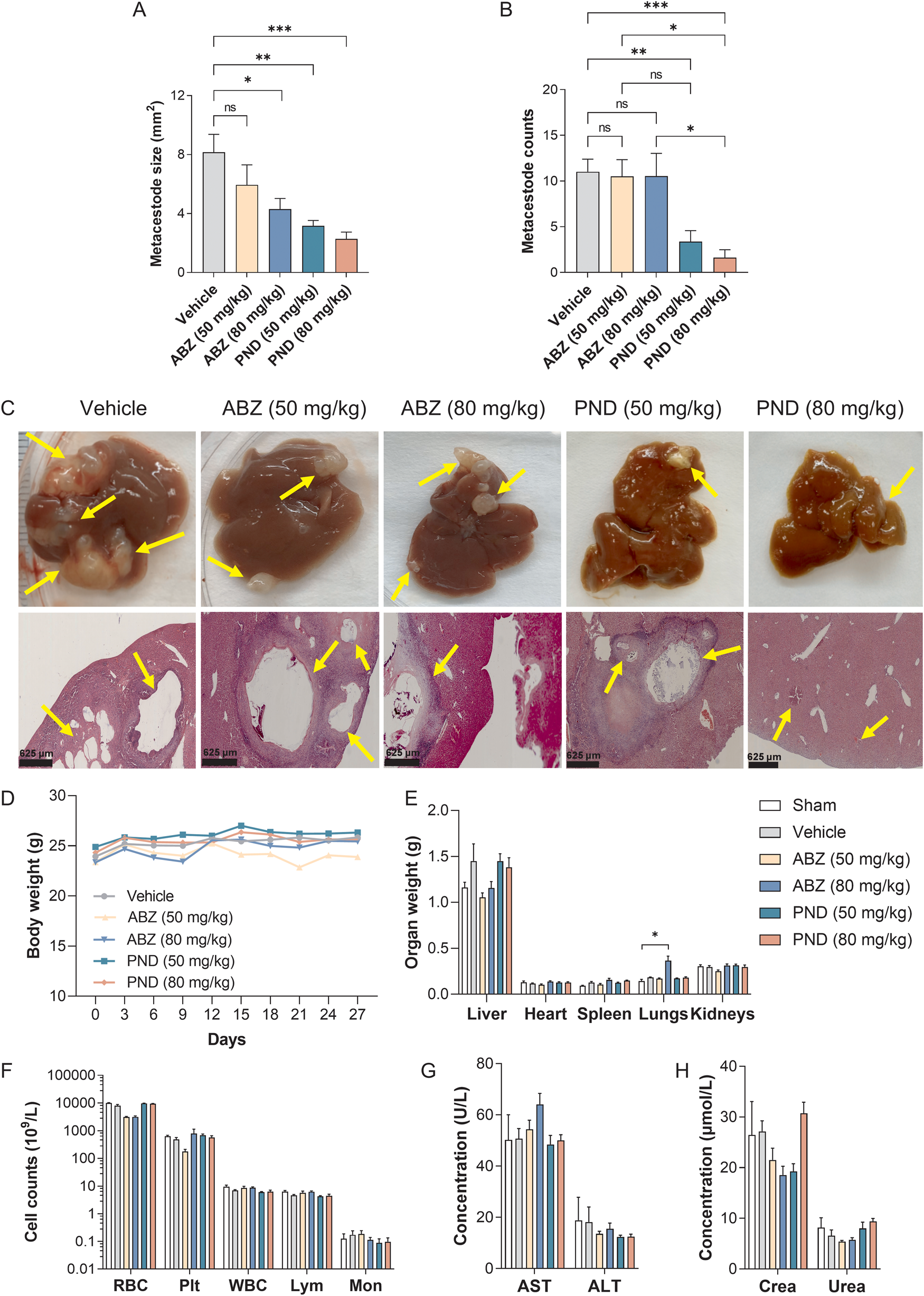
The therapeutic efficacy of pyronaridine was investigated in a hepatic AE murine model to reproduce the natural infection of intermediate hosts as best as possible. E. multilocularis metacestodes developed precisely in the liver and disseminated on the surface. At 3 months post-infection, metacestodes of various shapes and sizes were observed, with a maximum surface area of 40 mm2. Because the metacestodes were embedded in the liver tissue and could not be excised clearly, the in vivo drug efficacy was evaluated by measuring the metacestode size and counts of the infected animals. Pyronaridine proved orally active, demonstrating significant reductions in parasite burden and dose-dependently suppressing the dilatation of parasite-induced lesions. Following treatment with pyronaridine at doses of 50 and 80 mg kg−1, the average metacestode size of the infected animals shrank by 61.3% (P < 0.01) and 72.0% (P < 0.001), respectively, showing a significant reduction compared to that of unmedicated controls (Fig. 1A). Treatment with albendazole at a dose of 80 mg kg−1 resulted in a significant decrease in the metacestode size by 47.3% (P < 0.05) compared to the vehicle controls. In terms of metacestode counts, oral administration of pyronaridine at both doses resulted in a significant decrease in 69.3% (P < 0.01) and 85.2% (P < 0.001) compared to the vehicle controls, whereas in albendazole-treated animals, no significant difference was observed (Fig. 1B). Notably, a significant reduction in metacestode counts was observed in pyronaridine-treated mice compared to that in albendazole-treated individuals at an equal dose of 80 mg kg−1 (P < 0.05), which demonstrated the remarkable superiority of pyronaridine on the treatment outcome.
In vivo therapeutic efficacy and safety profile of pyronaridine in a hepatic alveolar echinococcosis murine model. The medicated groups (n = 8) were orally administered with pyronaridine (PND, 50 and 80 mg kg−1) or albendazole (ABZ, 50 and 80 mg kg−1) once a day for 30 days. The unmedicated vehicle control (n = 8) received only 0.5% CMC. Uninfected control mice (n = 4) were sham operated. The average metacestode size (A) and counts (B) of each group were measured. Liver tissues collected from each group were stained with haematoxylin and eosin and presented (C). Metacestodes are indicated by yellow arrows. The body weight (D), organ weight (E), haematological indices [red blood cells (RBC), blood platelets (Plt), white blood cells (WBC), lymphocytes (Lym) and monocytes (Mon)] (F), liver transaminases [aspartate aminotransferase (AST) and alanine aminotransferase (ALT)] (G) and renal function indices [creatinine (Crea) and urea nitrogen (Urea)] (H) of mice were determined. Data are shown as the mean ± s.e.m. *P < 0.05; **P < 0.01; ***P < 0.001; ns, not statistically significant (one-way ANOVA or Welch and Brown–Forsythe ANOVA test).
Histopathological analysis confirms the strong parasiticidal ability of pyronaridine against E. multilocularis metacestodes
Morphological alterations induced by pyronaridine on E. multilocularis metacestodes were thoroughly investigated by histopathological analysis (Fig. 1C). Parasitic tissues recovered from unmedicated mice showed intact germinal and laminated layers, around which, severe infiltration of inflammatory cells was observed. Albendazole treatment (80 mg kg−1) had an obvious effect on the metacestode tissues, with a major portion of the germinal layers being largely distorted. This drug-induced damage on germinal layers was also observed in albendazole treatment (50 mg kg−1), albeit to a lesser extent. Metacestodes collected from pyronaridine-treated (50 mg kg−1) mice suffered devastating damage: the germinal layers completely detached from the laminated layers, with only a few residues present. Following treatment with pyronaridine at 80 mg kg−1, metacestode integrity was completely lost. These collapsed metacestodes turned into calcified foci, and the surrounding liver parenchyma almost recovered to a normal state, with no signs of inflammatory infiltrates or fibroplastic proliferation.
Pyronaridine exhibits a good in vivo safety profile
The behaviour and appearance of pyronaridine-treated mice were normal throughout the entire course of treatment, and no adverse effects were noted. Additionally, there were no significant changes in body weight and organ weight between pyronaridine-treated and shamed animals (P values > 0.05), whereas a significant elevation of lung weight was observed in albendazole-treated (80 mg kg−1) individuals (P < 0.05) (Fig. 1D and E). The haematological indices, liver transaminases (AST and ALT) and renal function indices (Crea and Urea) were all within normal limits (Fig. 1F–H).
In silico similarity search identifies promising anti-echinococcal agents
Encouraged by the satisfactory outcome of pyronaridine repurposing, a screening campaign combined with computational approaches was performed to discover potential anti-echinococcal hits from the perspective of structural similarity to pyronaridine. The 2863 compounds that comprised the approved drug library were virtually screened using the Tanimoto similarity coefficient. The top 20 drugs (similarity threshold >0.4) were manually refined to exclude central nervous system drugs to avoid potential adverse effects. A set of 11 drugs with diverse clinical indications were then selected as potential hits to be further examined for their in vitro anti-echinococcal potency against protoscoleces and cultured small cysts (Table 1 and Fig. S1). The majority of the hits exhibited evident parasiticidal activity, including the antiallergic drugs loratadine and olopatadine, the antimalarials quinacrine and lumefantrine, and the β-blocker carazolol, all of the drugs killed 100% of the protoscoleces at a concentration of 40 μ m. Notably, some of the screening hits, such as benzydamine and carazolol, were only effective against one stage (protoscolex or cultured small cyst) but not the other. Pirenzepine, a gastric acid-inhibiting drug, exhibited pronounced parasiticidal activity and a rapid onset of action. Pirenzepine killed all protoscoleces and cultured small cysts within 24 h at concentrations of 20 or 30 μ m.
In silico similarity screening hits and their in vitro parasiticidal activity against E. multilocularis protoscoleces and cultured small cysts

Discussion
AE is an aggressive and infiltrative disease with very limited treatment options. To date, the pharmaceutical industry has not provided the incentive to develop novel drugs against AE, largely because the market has not been properly evaluated given the high number of asymptomatic carriers and long latency period before diagnosis. Commercial support for neglected diseases, such as echinococcosis, is modest. Thus, the repurposing of currently available drugs represents a shortcut to drug discovery for AE in an academic environment. Approved drugs have the highest potential to be used as salvage therapy, particularly for patients with AE who respond poorly to albendazole or suffer from severe side-effects. In this context, we repurposed pyronaridine as an anti-AE candidate, and many other approved drugs, such as pirenzepine, as promising lead compounds for the treatment of AE.
Considerable efforts have been made to identify alternative anti-AE therapeutic options. Numerous anti-infectives, antineoplastics and antiparasitics have been tested in vitro against the larval stage of E. multilocularis (Siles-Lucas et al., Reference Siles-Lucas, Casulli, Cirilli and Carmena2018). For most of these repurposing initiatives, a major problem has been that promising in vitro activity does not translate into antiparasitic efficacy in vivo, whereas antiprotozoals, such as nitazoxanide (Stettler et al., Reference Stettler, Rossignol, Fink, Walker, Gottstein, Merli, Theurillat, Thormann, Dricot, Segers and Hemphill2004), mefloquine (Kuster et al., Reference Kuster, Stadelmann, Rufener, Risch, Muller and Hemphill2015; Rufener et al., Reference Rufener, Ritler, Zielinski, Dick, Da Silva, Da Silva Araujo, Joekel, Czock, Goepfert, Moraes, de Souza, Müller, Mevissen, Hemphill and Lundström-Stadelmann2018b) and atovaquone (Enkai et al., Reference Enkai, Kouguchi, Inaoka, Irie, Yagi and Kita2021), have shown superiority either alone or in combination with albendazole. Pyronaridine, also an antiprotozoal, was first approved in China in the 1980s for the treatment of malaria and was most recently licensed along with artesunate by the European Medicines Agency to treat uncomplicated malaria (Stone et al., Reference Stone, Mahamar, Sanogo, Sinaba, Niambele, Sacko, Keita, Youssouf, Diallo, Soumare, Kaur, Lanke, ter Heine, Bradley, Issiaka, Diawara, Traore, Bousema, Drakeley and Dicko2022). Apart from Plasmodium, pyronaridine is also active against a range of pathogens, with promising activity against schistosomiasis in mice (Koehne et al., Reference Koehne, Zander, Rodi, Held, Hoffmann, Zoleko-Manego, Ramharter, Mombo-Ngoma, Kremsner and Kreidenweiss2021) and humans (Zoleko-Manego et al., Reference Zoleko-Manego, Okwu, Handrich, Dimessa-Mbadinga, Akinosho, Ndzebe-Ndoumba, Davi, Stelzl, Veletzky, Kreidenweiss, Nordmann, Adegnika, Lell, Kremsner, Ramharter and Mombo-Ngoma2022) and against SARS-CoV-2 (Puhl et al., Reference Puhl, Gomes, Damasceno, Godoy, Noske, Nakamura, Gawriljuk, Fernandes, Monakhova, Riabova, Lane, Makarov, Veras, Batah, Fabro, Oliva, Cunha, Alves-Filho, Cunha and Ekins2022) and Ebola virus infection (Lane et al., Reference Lane, Massey, Comer, Freiberg, Zhou, Dyall, Holbrook, Anantpadma, Davey, Madrid and Ekins2020) in vivo.
In this study, we pursued a new application of pyronaridine against hard-to-treat AE. A hepatic AE murine model was generated through the portal vein infection of protoscolex, where metacestodes developed precisely in the liver, with the same manifestations of primary infection through ingestion of parasite eggs (Enkai et al., Reference Enkai, Kouguchi, Inaoka, Irie, Yagi and Kita2021). The clinically relevant hepatic AE model represents a feasible option to reflect the natural infection situation in humans, without the need for high-level biosafety experimental conditions. Following a 30-day oral regimen of 80 mg kg−1 day−1, pyronaridine achieved an excellent therapeutic outcome in infected mice, showing a significant reduction in both metacestode size (72.0%) and counts (85.2%) compared to unmedicated infected mice, which revealed significantly more potent anti-echinococcal potency than albendazole treatment at an equal dose. The potent parasiticidal activity of pyronaridine was further validated by the destructive damage to metacestode tissues observed pathologically. The collapsed metacestodes were calcified, and an almost complete recovery of the surrounding liver parenchyma cells was observed.
The effective anti-AE oral regimen (80 mg kg−1 day−1, 30 days) of pyronaridine was comparable to that of nitazoxanide (3 mg day−1, approximate 150 mg kg−1 day−1, 35 days) and mefloquine (100 mg kg−1, twice per week, 12 weeks). Based on the human equivalent dose (HED) formula (Nair and Jacob, Reference Nair and Jacob2016), this dose is equivalent in humans to 6.5 mg kg−1 of pyronaridine, which is much lower than the recommended average dose of albendazole for humans (10–15 mg kg−1 day−1) in clinical practice. When compared to the anti-CE regimen in intraperitoneally infected mice (57 mg kg−1 day−1, 30 days) (Li et al., Reference Li, Wang, Yao, Wang, Li, Qi, Han, Ren, Dang, Han, Guo, Guo, Wang, Duan and Zhang2020), a higher dose of pyronaridine was required to achieve a significant treatment outcome against AE, probably because of the aggressive and infiltrative features of AE and/or the influence of intraperitoneal or intraparenchymatous parasite lesions. Indeed, the same is true for other anti-echinococcal agents, e.g. isoprinosine, ivermectin and cyclosporin A (Siles-Lucas et al., Reference Siles-Lucas, Casulli, Cirilli and Carmena2018).
Typically, toxicity is not a concern for the repurposing strategy because the approved drugs have known safety profiles. However, changes in therapeutic doses and periods applied for different indications should be considered. Pyronaridine was approved for human use to treat malaria at a dose of 24 mg kg−1 given in 4 divided doses over 3 days (Croft et al., Reference Croft, Duparc, Arbe-Barnes, Craft, Shin, Fleckenstein, Borghini-Fuhrer and Rim2012). Although the effective anti-AE therapeutic dose in this study was within the safe dose range of its original indication, chronic and complex AE usually requires long-term drug therapy. Fortunately, our in vivo toxicity assessment revealed that the 30-day regimen of pyronaridine showed a good tolerability profile at both dose levels, with no safety concerns. Plasma biochemical indices demonstrated that the treatments did not result in liver or kidney toxicity. In addition, previous sub-acute toxicity study indicated that oral gavage of pyronaridine at a dose of 24 mg kg−1 day−1 (HED = 13.0 mg kg−1 day−1) for 30 days was well tolerated by dogs with no obvious side-effects (Chen and Zheng, Reference Chen and Zheng1992; Bailly, Reference Bailly2021), which offered proof of its safety in long-term and high-dose regimens.
As in other fields of drug discovery, phenotypic screening of whole organisms is a basic and efficient strategy to identify novel compounds with efficacy against AE (Aulner et al., Reference Aulner, Danckaert, Ihm, Shum and Shorte2019). Previous screening campaigns have been conducted against Medicines for Malaria Venture (MMV) Malaria Box and Pathogen Box compound libraries, leading to the identification of 2 interesting compounds, MMV665807 and MMV689480 (buparvaquone); however, both failed to reduce the parasite burden in mice (Stadelmann et al., Reference Stadelmann, Rufener, Aeschbacher, Spiliotis, Gottstein, Hemphill and Kortagere2016; Rufener et al., Reference Rufener, Dick, D'Ascoli, Ritler, Hizem, Wells, Hemphill and Lundstrom-Stadelmann2018a). Given the satisfactory treatment outcome of pyronaridine, we assumed that its chemical structure could serve as a privileged anti-echinococcal template. Therefore, computer-aided virtual screening was performed from the unique perspective of identifying potential lead compounds sharing structural similarity with pyronaridine from an approved drug library. The strategy combined high-throughput virtual screening and experimental validation and, as expected, proved very effective. Over 10 drugs with diverse scaffolds and clinical indications were demonstrated to be effective against protoscoleces and/or cultured small cysts, which provided abundant starting points for subsequent optimization, allowing us to gain more insights about their valuable targets. Among them, pirenzepine, which possesses a pyridobenzodiazepine scaffold, represents the most promising lead compound. Pirenzepine is an M1 receptor-selective muscarinic antagonist that was originally developed as an orally delivered drug to reduce gastric acid secretion and gastric motility (Carmine and Brogden, Reference Carmine and Brogden1985). Besides its classical applications, pirenzepine is effective for controlling myopia progression (Kang, Reference Kang2018) and treating peripheral neuropathy (Calcutt et al., Reference Calcutt, Smith, Frizzi, Sabbir, Chowdhury, Mixcoatl-Zecuatl, Saleh, Muttalib, Van der Ploeg, Ochoa, Gopaul, Tessler, Wess, Jolivalt and Fernyhough2017). The recommended adult dosage of pirenzepine in the treatment of duodenal or benign gastric ulceration is 100–150 mg daily for 4–8 weeks. In human subjects, a single oral dose of pirenzepine at 150 mg resulted in a peak plasma concentration of 210 μg L−1 (equivalent to 0.6 μ m) (Carmine and Brogden, Reference Carmine and Brogden1985), which is significantly lower than the effective parasiticidal concentration (20 μ m) tested in this study. Considering that echinococcosis typically requires long-term medication, it is possible that multiple doses could lead to an improved steady-state plasma concentration of pirenzepine. However, further animal experimentation will be necessary to provide evidence regarding the potential efficacy of pirenzepine for in vivo treatment of E. multilocularis infections.
In conclusion, the drug repurposing strategy identified the established drug pyronaridine as a promising drug candidate for the AE treatment, with significantly better treatment outcomes than albendazole in a clinically relevant hepatic AE murine model. The drug is already in widespread clinical use and has been extensively characterized in terms of toxicity, pharmacokinetics and clinical tolerability. For long-term therapy in clinical practice, pyronaridine meets the necessary criteria of a candidate, including a long track record of safety, suitable pharmacokinetic properties and cost-effectiveness. Our findings underline the importance of carrying out further studies to determine the significance of applying pyronaridine on human patients with AE and/or CE. The anti-echinococcal therapeutic application of pyronaridine could translate rapidly to clinical use through ‘off-label’ prescription as an alternative or salvage treatment option for patients with severe side-effects or resistance to albendazole treatment. However, contrary to our expectations, the combination treatment of albendazole and pyronaridine did not yield positive results (data not shown). Our preliminary combination treatment of the 2 drugs for mouse AE did not demonstrate synergistic effects and even led to increased side-effects on liver function, probably due to drug–drug interactions. Given the intricacy involved in combining these 2 drugs, a comprehensive analysis of their interaction will be conducted in future studies to elucidate the underlying reasons.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0031182024001124
Data availability statement
All relevant data are within the manuscript and its supplementary files.
Acknowledgements
Graphic abstract was created with BioRender.com.
Author contributions
W. W., J. L. and W. Z. designed and coordinated the study. Y. C. and L. D. synthesized and identified the compounds. W. Q., M. T., C. W., Y. Z. and Y. Y. carried out the drug efficacy tests. W. W., S. H. and X. H. performed the data analyses and virtual screening. W. W. and J. L. wrote the article. L. D. and W. Z. critically revised the paper.
Financial support
This work was supported by the National Key R&D Program (grant number: 2023YFD1801204), National Natural Science Foundation of China (grant numbers: 81830066, 32072886, 82473743 and U1803282), the Ministry of Science and Technology of China (grant number: 2022YFE114600), Clinical Research Project of Healthcare Industry of Shanghai Municipal Health Commission (grant number: 202040054) and 2021 Kunlun Talents Project-High-Level Innovative and Entrepreneurial Talents.
Competing interests
None.
Ethical standards
Animal experimental procedures and welfare were conducted in compliance with the Chinese Laboratory Animal Administration Act 2017 as well as the Guidelines for the Ethical Review of Laboratory Animal Welfare People's Republic of China National Standard GB/T 35892-20181. All animal experiments and procedures were approved by the Animal Ethics Committee of the First Affiliated Hospital of Xinjiang Medical University (approval number: K202102-18).





