Introduction
The family Trypetheliaceae Eschw. is an almost strictly tropical lineage of nearly exclusively corticolous (very rarely saxicolous) lichenized fungi (Aptroot & Lücking Reference Aptroot and Lücking2016). It is one of the predominant elements of crustose lichen communities in tropical forests, dry forests and savannahs (Harris Reference Harris1986; Makhija & Patwardhan Reference Makhija and Patwardhan1988; Komposch & Hafellner Reference Komposch and Hafellner2000, Reference Komposch and Hafellner2003; Aptroot et al. Reference Aptroot, Lücking, Sipman, Umaña and Chaves2008; Aptroot Reference Aptroot2009a, Reference Aptroot b; Lücking et al. Reference Lücking, Nelsen, Aptroot, Barillas de Klee, Bawingan, Benatti, Binh, Bungartz, Cáceres and Canêz2016).
Throughout its history, Trypetheliaceae has had several changes in systematic classification at genus level and its placement within the fungi, now known to belong in the ascomycete class Dothideomycetes O. E. Erikss. & Winka (Nylander Reference Nylander1863; Müller Reference Müller1884, Reference Müller1885; Vainio Reference Vainio1890; Zahlbruckner Reference Zahlbruckner1922, Reference Zahlbruckner1924, Reference Zahlbruckner1928; Letrouit-Galinou Reference Letrouit-Galinou1957, Reference Letrouit-Galinou1958; Eriksson Reference Eriksson1981; Harris Reference Harris1986, Reference Harris1993; Del Prado et al. Reference Del Prado, Schmitt, Kautz, Palice, Lücking and Lumbsch2006; Nelsen et al. Reference Nelsen, Lücking, Grube, Mbatchou, Muggia, Rivas Plata and Lumbsch2009; Schoch et al. Reference Schoch, Sung, López-Giráldez, Townsend, Miadlikowska, Hofstetter, Robbertse, Matheny, Kauff and Wang2009a, Reference Schoch, Crous, Groenewald, Boehm, Burgess, De Gruyter, de Hoog, Dixon, Grube and Gueidan b; Aptroot & Lücking Reference Aptroot and Lücking2016; Lücking et al. Reference Lücking, Nelsen, Aptroot, Barillas de Klee, Bawingan, Benatti, Binh, Bungartz, Cáceres and Canêz2016). A detailed synopsis based on phylogenetic, morpho-anatomical and chemical data of extensive material, including revision of type specimens, formally accepted 418 species distributed among 15 genera (Aptroot & Lücking Reference Aptroot and Lücking2016). Among these, Astrothelium Eschw. is by far the most speciose genus (242 species at the time of the synopsis), whereas several genera include only a small number of species, such as Aptrootia Lücking & Sipman (3), Distothelia Aptroot (3), Marcelaria Aptroot et al. (3), Nigrovothelium Lücking et al. (2), and Novomicrothelia Aptroot et al. (1) (Aptroot & Lücking Reference Aptroot and Lücking2016).
Members of Trypetheliaceae are characterized by a crustose, corticate or sometimes ecorticate thallus with trentepohlioid algae; perithecioid, simple to aggregate or compound ascomata with separate or fused ostioles, often immersed into distinct pseudostromata contrasting with the thallus in colour; branched and usually anastomosing hamathecial filaments (paraphysoids); bitunicate, cylindrical to oblong asci; disto- to more rarely euseptate ascospores occurring in numbers of 1–8 per ascus, distoseptate, usually IKI−, rarely IKI+ violet, with rounded or angular lumina, colourless, rarely becoming brown (Aptroot & Lücking Reference Aptroot and Lücking2016).
The genus Laurera Rchb. was traditionally recognized by the solitary to aggregate perithecia with an apical ostiole, in combination with muriform, hyaline ascospores (Letrouit-Galinou Reference Letrouit-Galinou1957, Reference Letrouit-Galinou1958). However, morphological inconsistencies in species included in Laurera have been recognized by various authors, who divided the genus into several groups (Upreti & Singh Reference Upreti and Singh1987). One of these was the L. benguelensis group, corresponding to three species (L. benguelensis (Müll. Arg.) Zahlbr., L cummingi (Mont.) Zahlbr. and L. purpurina (Nyl.) Zahlbr.), characterized by sessile, solitary to aggregate perithecial warts with a broad, flat ostiolar region and often a split between the proper ascoma wall and the surrounding thallus wart. The ascoma warts contain several layers, including a gelatinous cortex, a medulla filled with pigment crystals, and a proper, carbonized inner wall (excipulum), whereas algal cells are absent and restricted to the vegetative thallus (Aptroot et al. Reference Aptroot, Nelsen and Parnmen2013; Aptroot & Lücking Reference Aptroot and Lücking2016).
The taxonomic coherence of the Laurera benguelensis group was confirmed by molecular phylogenetic analysis, placing sequences of L. purpurina and L. cummingi in a strongly supported clade (Nelsen et al. Reference Nelsen, Lücking, Aptroot, Andrew, Cáceres, Rivas Plata, Gueidan, da Silva Canêz, Knight and Ludwig2014; Lücking et al. Reference Lücking, Nelsen, Aptroot, Barillas de Klee, Bawingan, Benatti, Binh, Bungartz, Cáceres and Canêz2016). The group is one of the few morphologically characterized entities that form separate clades within Trypetheliaceae, distinct from both Trypethelium s. str. and the large, redefined Astrothelium clade, and therefore the new genus Marcelaria was proposed for this group (Aptroot et al. Reference Aptroot, Nelsen and Parnmen2013).
Several characters are important for the recognition of species within Marcelaria, including the pigments in the pseudostromata, which are bright red in M. purpurina (Nyl.) Aptroot et al. and yellow-orange in M. benguelensis (Müll. Arg.) Aptroot et al. and M. cumingii (Mont.) Aptroot et al. Additionally, M. purpurina has a clear hamathecium and rather large ascospores (110–170 μm long); it is distributed in the Neotropics and the African Paleotropics. In contrast, M. benguelensis and M. cumingii have a densely inspersed hamathecium and much smaller ascospores (50–80 μm long); both are restricted to the eastern Paleotropics. In the present paper, we describe a new species in the genus Marcelaria from Colombia which combines characters of both groups, apparently linking them morphologically, namely an inspersed hamathecium and large ascospores, as in M. purpurina, with yellow-orange pigments as in M. benguelensis and M. cumingii.
Materials and Methods
Fresh material was collected during a field expedition to the Piedemonte Llanero region, in the municipality of Villanueva (Casanare). The specimens were carefully collected using a knife and paper bags, following the conventional methodology for the collection of lichens (Hale Reference Hale1979). The material was air-dried at room temperature. The specimens were subsequently examined and measurements of all structures were carried out under an Olympus SZ60 dissecting microscope; hand-cut sections of thallus and perithecia were studied under an Olympus CH30 compound microscope. Microscope images were captured with a Motorola Moto G72 smartphone. Thin-layer chromatography (TLC) was performed according to Orange et al. (Reference Orange, James and White2010), using solvent C. The morphological, anatomical and chemical descriptions were assembled following the analysis of characters used for the description of the other Marcelaria species (Aptroot et al. Reference Aptroot, Nelsen and Parnmen2013). The distribution map was created using ArcGIS software.
New sequences of the mitochondrial small subunit rDNA (mtSSU) were generated for two separate pieces of the type collection, repeating the PCR and sequencing reaction twice. Attempts to also sequence the nuclear large subunit rDNA (nuLSU) failed. For DNA extraction, we used the Sigma REDExtract-N-Amp Plant PCR Kit (St Louis, Missouri, USA), following the manufacturer’s instructions but with lower proportions for lower amounts of material and DNA. The mtSSU was amplified using the primers SSU1R and SSU3R (Zoller et al. Reference Zoller, Scheidegger and Sperisen1999). The 13-μl PCR reactions consisted of 6.0 μl of water, 0.1 μl of each PCR primer, 3.5 μl of REDExtract-N-Amp PCR Ready Mix (Sigma-Aldrich) and 2 μl DNA. PCR cycling conditions were as follows: 95 °C for 5 min, followed by 35 cycles of 94 °C for 45 s, 50 °C for 1 min, 72 °C for 1.5 min, with a single 72 °C final extension for 10 min. Prior to assembly, the obtained sequence reads were evaluated using BLASTn (Chen et al. Reference Chen, Ye, Zhang and Xu2015) and subsequently combined with selected sequences of Trypetheliaceae from GenBank, following the study by Nelsen et al. (Reference Nelsen, Lücking, Aptroot, Andrew, Cáceres, Rivas Plata, Gueidan, da Silva Canêz, Knight and Ludwig2014), focusing on Marcelaria and related genera (Table 1). The sequences were arranged in BioEdit v. 7 (Hall Reference Hall2011) and aligned using MAFFT v. 7 (Katoh & Standley Reference Katoh and Standley2013). The phylogenetic tree was built by maximum likelihood, with 1000 bootstrap pseudoreplicates, using RAxML v. 8 (Stamatakis Reference Stamatakis2014). The best-scoring tree was visualized in FigTree v. 1.4 (Rambaut & Drummond Reference Rambaut and Drummond2012).
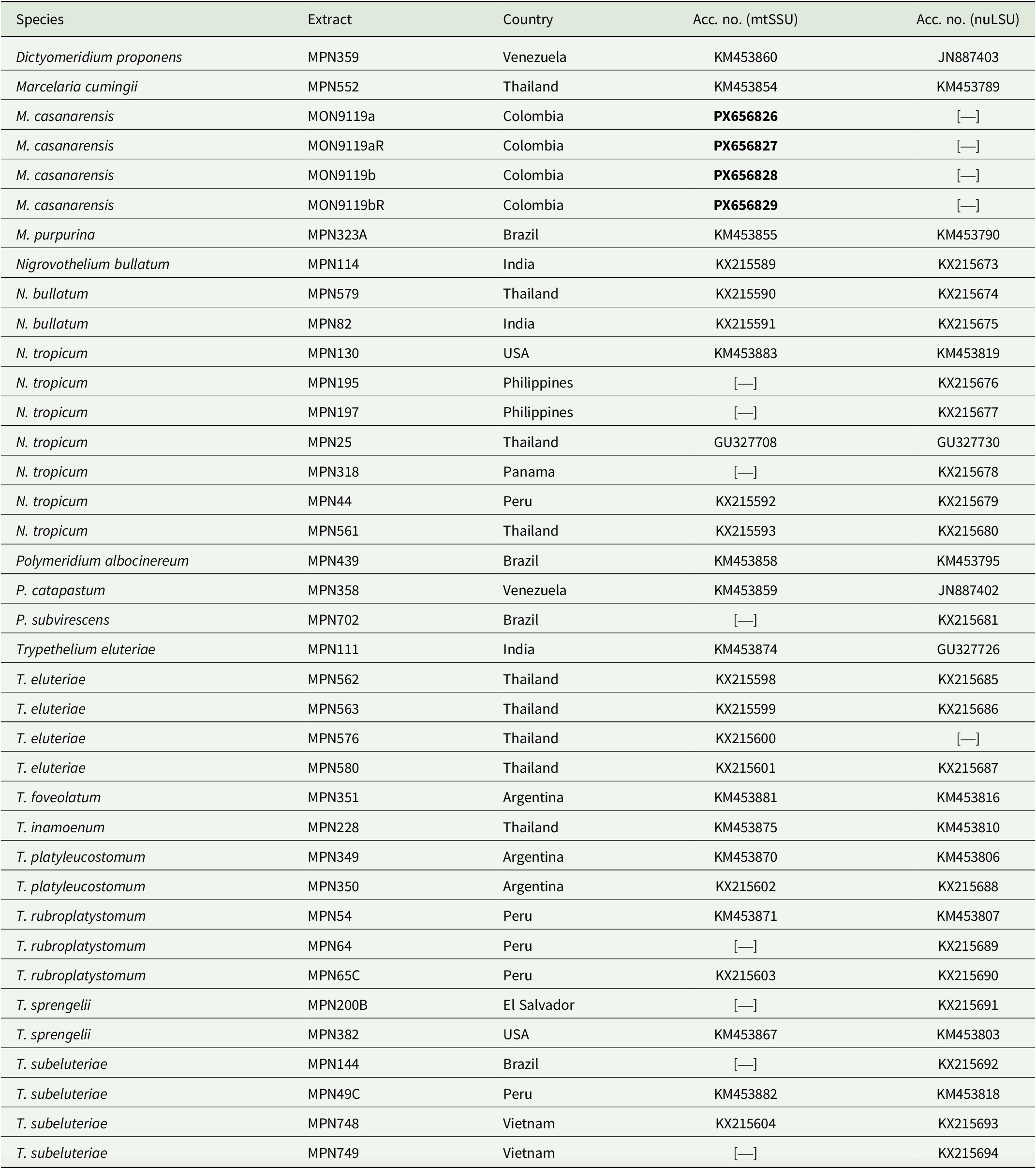
GenBank Accession numbers for Trypetheliaceae taxa and samples used in the phylogenetic analysis of the present study. Newly generated sequences are indicated in bold. The voucher information for Marcelaria casanarensis is: Colombia, L. Gonzalez 59 (UDBC, B).
Results
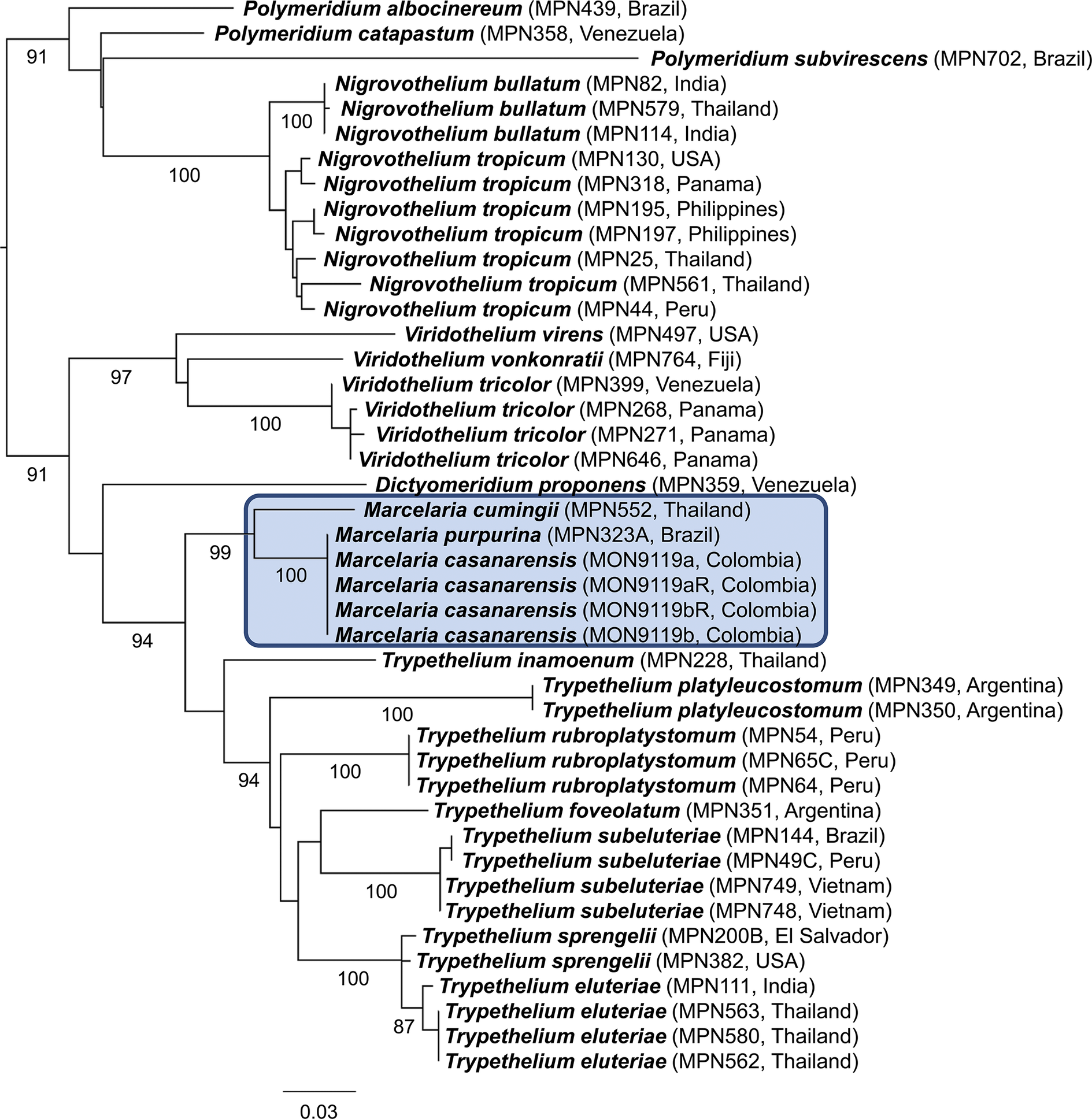
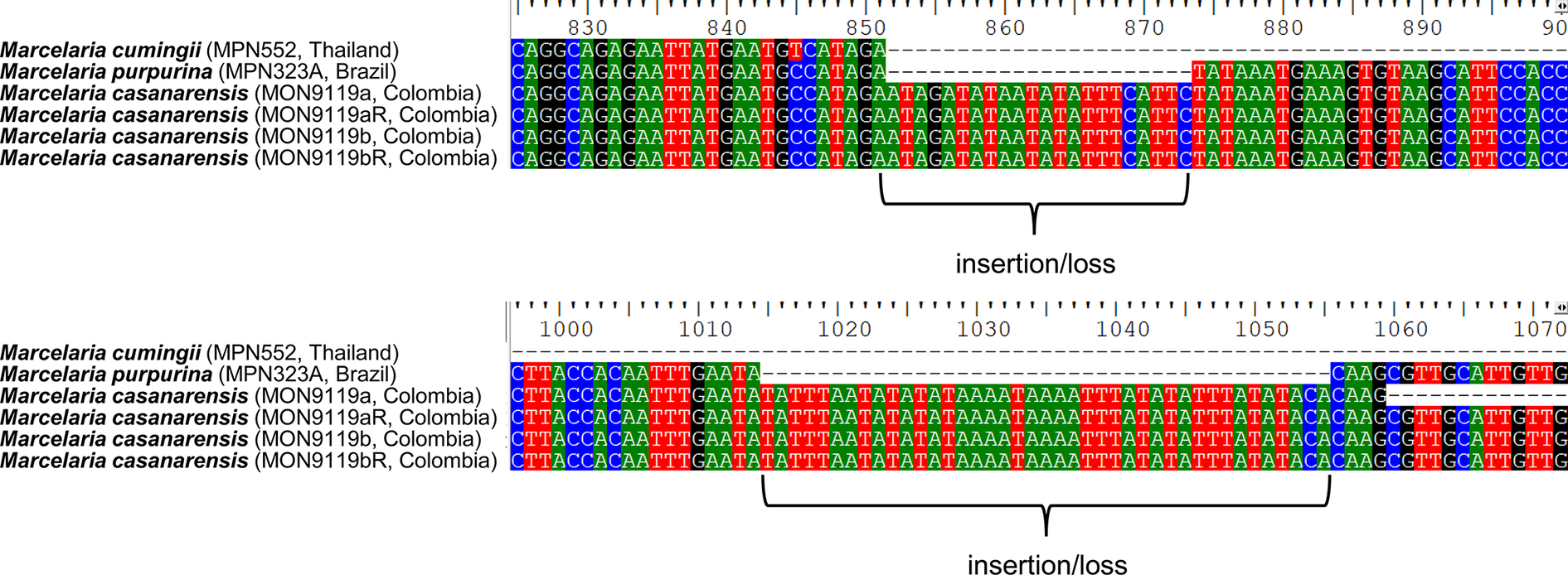
The phylogenetic analysis placed the new species, Marcelaria casanarensis J.-M. Torres, Lücking & Moncada, in a strongly supported clade corresponding to the genus Marcelaria, with absolute support close to M. purpurina (Fig. 1). The relationship with the latter was unresolved since both species share the same sequence pattern in the mtSSU marker, indicating a very close relationship. However, M. casanarensis consistently displayed two larger insertions (or deletions in M. purpurina) (Fig. 2), suggesting a very recent divergence. Unfortunately, the nuLSU marker, which provides better resolution in the Trypetheliaceae, could not be amplified.
Best-scoring maximum likelihood tree of Marcelaria and related genera in the Trypetheliaceae based on the concatenated mtSSU and nuLSU markers (mtSSU only for M. casanarensis). Bootstrap support values are indicated below the branches. In colour online.
Sections of the mtSSU alignment of the Marcelaria casanarensis/purpurina clade, showing the positions of the two longer insertions/deletions (see also Supplementary Material File S1, available online). In colour online.
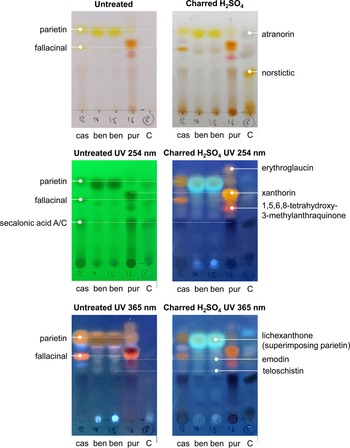
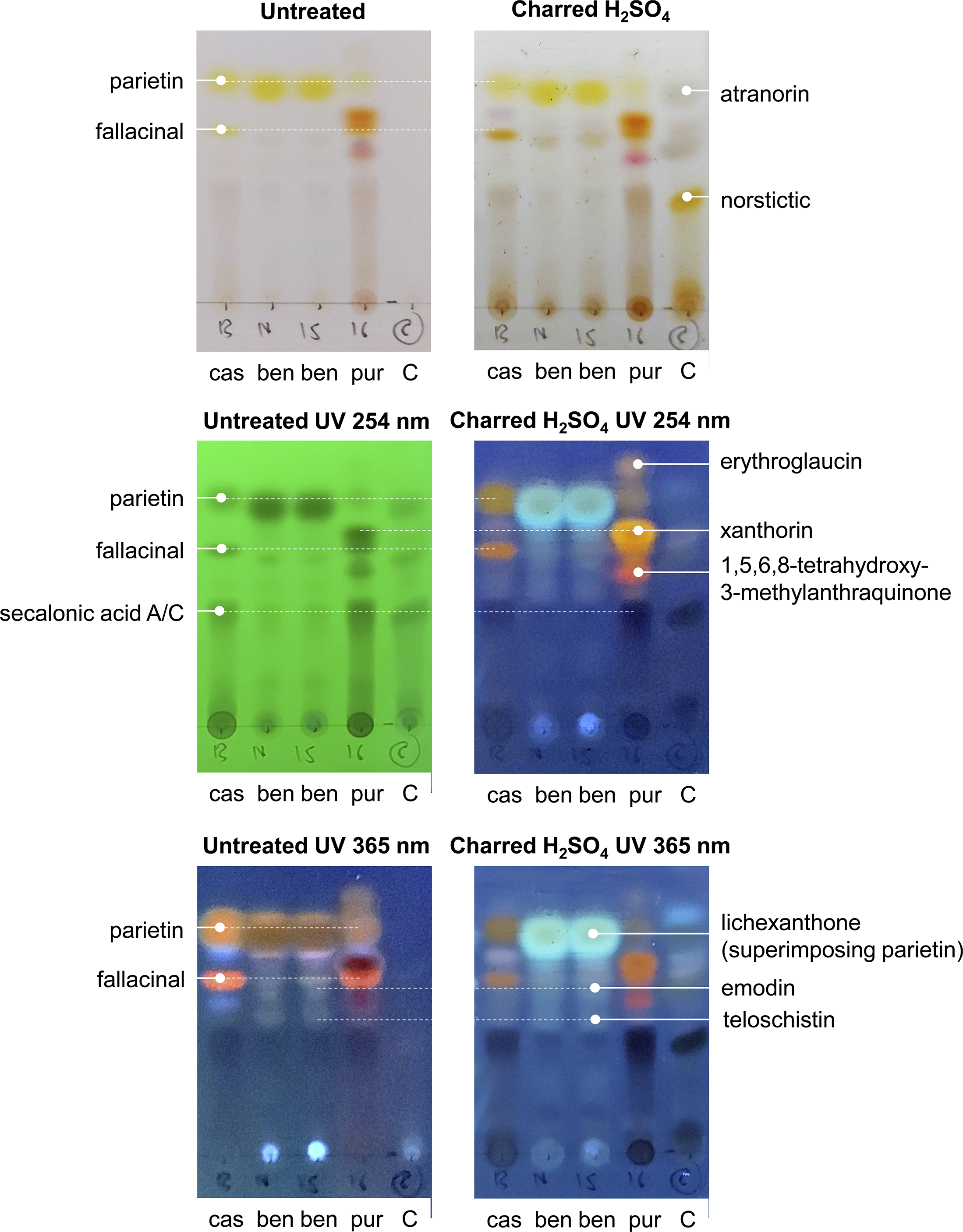
The comparative chemical analysis revealed a complex pigment pattern for the three species, M. casanarensis, M. benguelensis and M. purpurina (Fig. 3). All three species contain parietin and fallacinal; however, the concentration of parietin is much lower in M. purpurina compared to M. casanarensis and that of fallacinal is much lower in M. benguelensis compared to the other two species. Lichexanthone is present only in M. benguelensis, superimposing parietin. Both M. casanarensis and M. purpurina contain secalonic acid A/C, absent in M. benguelensis. The bright red colour of M. purpurina is caused by the additional pigments erythroglaucin, xanthorin, and 1,5,6,8-tretrahydroxy-3-methylanthraquinone, which are not present in the other three species. In contrast, M. benguelensis contains emodin and teloschistin, not present in M. casanarensis and M. purpurina (Fig. 3). Thus, although M. casanarensis shares the orange colour of the ascomata with M. benguelensis, its pigment chemistry is overall more closely related to that of M. purpurina, rendering the similarities in colour with M. benguelensis a remarkable homoplasy.
Results of the comparative TLC analysis of three species of Marcelaria, M. casanarensis (cas; isotype), M. benguelensis (ben; Thailand, Sipman 48523 (B 60 0165159), 48527 (B 60 0165159)), and M. purpurina (pur; Venezuela, Garcia s. n. (B 60 0101327)), with a control containing atranorin and norstictic acid, showing the different treatments without and with UV. In colour online.
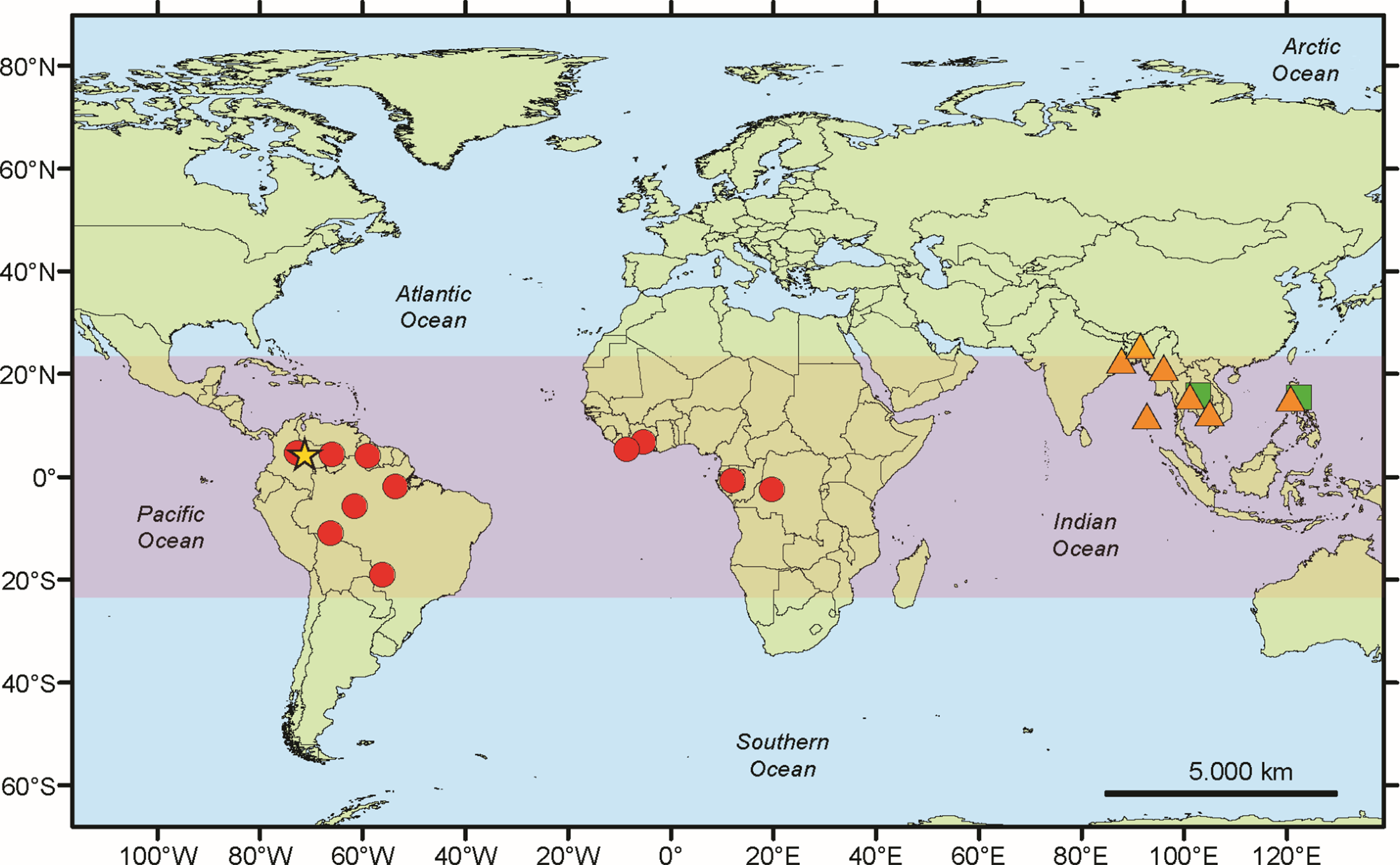
The new species is the fourth species known in the genus. The habitat in which the material was collected corroborates the preference of this genus for tropical forests and partially tropical woodlands (Fig. 4).
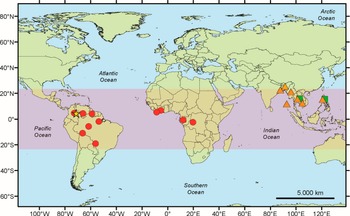
Geographical distribution of Marcelaria species. The orange triangles correspond to M. benguelensis, the green squares to M. cumingii, the red dots to M. purpurina, and the yellow star to M. casanarensis. In colour online.
Taxonomy
Marcelaria casanarensis J.-M. Torres, Lücking & B. Moncada sp. nov.
MycoBank No.: MB 862136
Differing from M. purpurina in the orange instead of red pseudostromata and from M. benguelensis and M. cumingii in the clear hamathecium and the larger ascospores (110–170 μm vs 50–80 μm long).
Type: Colombia, Casanare, Municipio de Villanueva, Vereda La Camarga Lechemiel, 4°35′59.060″N, 72°54′34.373″W, 256 m alt., corticolous, 29 April 2024, L. Gonzalez 59 (UDBC—holotype; B—isotype).
(Fig. 5)
Thallus corticate, olive green but with patches of yellow-orange pruina. Photobiont a species of Trentepohlia, with rounded to somewhat irregular cells.
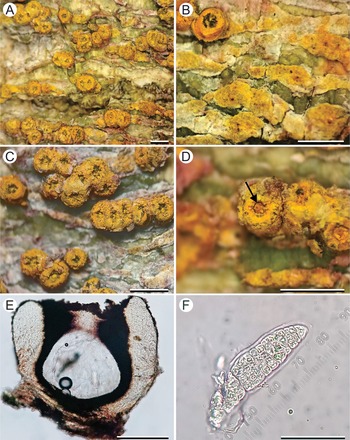
Marcelaria casanarensis (L. Gonzalez 59). A, thallus with ascomata. B, detail of thallus surface with patches of yellow-orange pruina. C, clusters of 2–8 ascomata. D, detail of ascoma showing split between the proper excipulum and the enclosing thalline wart (arrow). E, transverse section of ascoma showing clear hamathecium. F, ascospore. Scales: A–D = 1 mm; E = 0.5 mm; F = 50 μm. In colour online.

Ascoma warts solitary to aggregate, prominent to sessile, forming bright yellow-orange warts, 0.7–1.2 mm diam. and up to 0.7 mm high, clusters of 2–8 warts up to 3 mm diam.; surface rough due to patches of pruina, slightly shiny, in section with a 5–10 μm thick, prosoplectenchymatous upper cortex, and a gelatinous main cortex filled with yellow-olive to orange-brown pigment granules, 50–70 μm thick at the base, 70–150 μm thick in lateral portion and 150–185 μm in apical portion. Proper wall (excipulum) 50–60 μm thick at the base, 70–150 μm thick in lateral portion, 150–210 μm thick in apical portion, brown-black, carbonized. Nucleus 0.65–0.85 mm diam. and 0.5–0.8 mm high. Hamathecium clear. Ascospores 8 per ascus, their wall IKI+ violet, densely muriform with 25–35 transverse septa and 4–6 longitudinal septa per segment, 110–170 × 20–26 μm, surrounded by a 5–10 μm thick gelatinous sheath.
Conidia not observed.
Chemistry
Thallus UV−, K− except for the patches of yellow-orange pruina, these K+ purple; ascoma warts UV+ orange, K+ purple. TLC: parietin (major), fallacinal (major), secalonic acid A/C (major).
Etymology
The epithet refers to the department of Casanare, the type region of the new species.
Ecology and distribution
Neotropical (so far known only from Colombia); growing on tree bark in open areas near to the road.
Remarks
Marcelaria casanarensis is only the fourth species in the genus Marcelaria, characterized by its prominent to sessile, brightly yellow-orange pigmented ascomata with a flat top, often forming a split between the proper perithecium and the enclosing thalline wart, combined with a clear hamathecium and muriform ascospores (Fig. 5). The only neotropical species known so far, M. purpurina, was clearly set apart from the two paleotropical species, M. benguelensis and M. cummingii, in the bright red pigmentation, the clear hamathecium and the much larger muriform ascospores. The new species shares the anatomical features with M. purpurina and the colour with the two paleotropical taxa, but the pigment chemistry is more similar to that of M. purpurina.
Additional specimens examined
Colombia: Casanare: Municipio de Villanueva, Vereda La Camarga Lechemiel, 4°35′59.060″N, 72°54′34.373″W, 256 m alt., corticolous, 2024, J.-M. Torres 1650 (UDBC).
Key to the species currently accepted in Marcelaria

Discussion
The new species recognized in this paper confirms Marcelaria as a strictly tropical genus, with two species each now known from the Neotropics and from the eastern Paleotropics, and one from the African Paleotropics (Aptroot & Lücking Reference Aptroot and Lücking2016; Lücking et al. Reference Lücking, Nelsen, Aptroot, Barillas de Klee, Bawingan, Benatti, Binh, Bungartz, Cáceres and Canêz2016). Marcelaria casanarensis may prefer tropical savannah habitats of low altitudes, similar to M. purpurina (Aptroot et al. Reference Aptroot, Nelsen and Parnmen2013), but is possibly restricted to the Piedemonte Llanero in the Colombian territory, since this conspicuous taxon has not been found at other localities in the western Amazon or the eastern Andean foothills.
The new species appears to present a striking link in its morphological and anatomical features between the neotropical-African Marcelaria purpurina and the eastern paleotropical M. benguelensis and M. cumingii, sharing the anatomical details (clear hamathecium, large muriform ascospores) with M. purpurina but the yellow-orange pigmentation with the two paleotropical species. At first glance, this supports the close relationship between these taxa as evident from molecular data (Aptroot et al. Reference Aptroot, Nelsen and Parnmen2013; Nelsen et al. Reference Nelsen, Lücking, Aptroot, Andrew, Cáceres, Rivas Plata, Gueidan, da Silva Canêz, Knight and Ludwig2014; Lücking et al. Reference Lücking, Nelsen, Aptroot, Barillas de Klee, Bawingan, Benatti, Binh, Bungartz, Cáceres and Canêz2016). However, the orange colour shared between M. casanarensis and the two paleotropical species is better interpreted as homoplasy, since the underlying pigment chemistry is different: all three feature parietin, but M. casanarensis additionally contains fallacinal and secalonic acid A/C (as in M. purpurina), whereas the two paleotropical species contain emodin and teloschistin as accessory pigments.
Our results also highlight the importance of integrative taxonomy taking into account morphological, anatomical and chemical characters. Even before obtaining molecular sequence data, our comparative analysis of Laurera purpurina with L. benguelensis and L. cumingii led us to conclude that these species may be closely related. This conclusion now receives strong support with the discovery of the new species, its placement within Marcelaria also being confirmed with molecular data.
Supplementary Material
The Supplementary Material for this article can be found at http://doi.org/10.1017/S0024282926101388.
Acknowledgements
The authors thank Geocol Consultores S.A. for donating and depositing in the UDBC herbarium the material collected during the field phase of the project ‘Environmental impact study for the development area Llanos 87’, carried out in the municipalities of Villanueva, Monterrey and Tauramena (Casanare). The workshop at the UDBC herbarium during which the material was taxonomically assessed was supported by the DRYLICH project ‘Establishing a Network of Expertise for the Lichen Biota of the Tropical Dry Forest’; this is a bilateral project between the Botanical Garden and Botanical Museum of Berlin and the Universidad del Norte, Barranquilla, funded by the Federal Ministry of Education and Research (Bundesministerium für Bildung und Forschung – BMBF; Förderkennzeichen 01DN23013) and the Ministry of Science, Technology and Innovation (MinCiencias; contract no. 80740-032-2023), within the programme ‘Promoting Scientific and Technological Cooperation Projects with Colombia’ (Förderung von Projekten der wissenschaftlich-technologischen Zusammenarbeit mit Kolumbien). We especially thank John Elix for assistance with the interpretation of the TLC analysis.
Author Contribution
Conceptualization, methodology and validation: JMT, RL; fieldwork: JMT; morphological analyses: JMT; molecular and chemical analyses: BM, RL; writing of original draft preparation, JMT, RL; writing, review and editing: JMT, RL, BM. All authors reviewed and approved the final version of this manuscript.
Author ORCIDs
Jean-Marc Torres, 0000-0002-6703-338X; Robert Lücking, 0000-0002-3431-4636; Bibiana Moncada, 0000-0001-9984-2918.
Competing Interests
The authors declare none.
Data Accessibility
Nomenclatural novelties have been deposited in MycoBank.