The term Baltic amber is broad and can cover a range of different fossil resins from different areas (Bojarski et al. Reference Bojarski, Cierocka and Szwedo2025b), although here it is used to refer to succinite from the Samland Peninsula, Kaliningrad Oblast, Russia, which is the primary source of amber washed up on the southeastern Baltic coast (Kharin & Eroshenko Reference Kharin and Eroshenko2017). Thus, the use of the term Baltic amber in this paper excludes Lublin amber from southeastern Poland, Rovno amber from Ukraine and Bitterfeld amber from Germany, following Drohojowska et al. (Reference Drohojowska, Śladowska and Szwedo2024).
Baltic amber is well known for its exquisite preservation of small animals, plants and other organisms, particularly insects, spiders and other arthropods, also worms, snails, lizards, bird feathers, mammal hair, flowers, leaves, conifer cones, ferns, mosses, liverworts, lichens and fungi (Weitschat & Wichard Reference Weitschat and Wichard2002; Kaasalainen et al. Reference Kaasalainen, Schmidt and Rikkinen2017; Kettunen et al. Reference Kettunen, Sadowski, Seyfullah, Dörfelt, Rikkinen and Schmidt2018). For researchers studying the evolution of these organisms, it is important to know how old they are, particularly taxa used in phylogenetic trees. Unfortunately, the age of Baltic amber has been uncertain and a subject of controversy for many years (Sadowski et al. Reference Sadowski, Schmidt and Kunzmann2022; Bojarski et al. Reference Bojarski, Cierocka and Szwedo2025a; Drohojowska et al. Reference Drohojowska, Gorzelańczyk and Szwedo2025). Most amber specimens come into scientists’ hands via the amber trade after precise information on their locality and horizon has been lost – although for pieces washed up on beaches or extracted from reworked deposits that information is already lost (Ross et al. Reference Ross, Zhou, Hoffeins and Crighton2024). However, knowing the most likely source is useful for providing a probable age for the amber. The dating of amber can only be done by dating the bed that the amber is found in; however, this represents a minimum age as it is not known how long it took the amber to be transported from its source to its final site of deposition. The age of Baltic amber is notoriously imprecise because it is known to occur at several horizons.
The Upper Blue Earth Member from within the Prussian Formation has by far the highest concentration of amber and within it the concentration is highest towards the base, as indicated by Standke (Reference Standke, Rascher, Wimmer, Krumbiegel and Schmiedel2008). The Prussian Formation outcrops on the Samland (Sambian) Peninsula, part of Kaliningrad Oblast, an enclave of Russia sandwiched between Poland and Lithuania, and amber has been commercially mined there since the middle of the 19th century (Kostiashowa Reference Kostiashowa1999; Ostrowski et al. Reference Ostrowski, Pruszak, Babakov and Chubarenko2012). The Upper Blue Earth extends under the Baltic Sea so is also the primary source of amber washed up on southeastern Baltic beaches (Grigelis Reference Grigelis2001; Sivkov & Zhamoida Reference Sivkov, Zhamoida, Gritsenko, Sivkov, Yurov and Kostianoy2017). It is the most likely source of amber with inclusions in museum collections, particularly specimens from the Klebs collection, which were mined by the Stantien & Becker company from 1875 (when the Palmnicken mine opened, now at Yantarny) and are in the collections of the Georg-August-Universität Göttingen (Germany) and Natural History Museum, London (UK) (Ritzkowski Reference Ritzkowski2001; Ross Reference Ross2004; Sadowski et al. Reference Sadowski, Schmidt and Kunzmann2022). Klebs also produced small representative collections of Baltic amber mined by Stantien & Becker, of which examples are at the Bristol Museum & Art Gallery (UK) and Naturhistorisches Museum Wien (Austria) (Leyla Seyfullah pers. comm. 2025). More recently, a collection from the Primorsky opencast mine, Yantarny (Kaliningrad Oblast, Russia), was acquired by the Booth Museum of Natural History, Brighton (UK) (Perkovsky et al. Reference Perkovsky, Rasnitsyn, Vlaskin and Taraschuk2007). There are also important historical collections of Baltic amber from the Samland Peninsula at the Museum für Naturkunde, Berlin (Germany), but there is no evidence as to whether the specimens were mined and/or collected from the sea or shore (Christian Neumann pers. comm. 2025).
Amber is only slightly heavier than water so can easily be transported. Pieces that contain air bubbles have a lower specific gravity and can float (Ross Reference Ross2010). It has been suggested that the amber in the Upper Blue Earth has been reworked, and it has certainly been reworked into Quaternary sediments (Szwedo & Sontag Reference Szwedo and Sontag2009; Weitschat & Wichard Reference Weitschat, Wichard and Penney2010; Kharin & Eroshenko Reference Kharin and Eroshenko2017). Amber with inclusions mined from the Upper Blue Earth is generally fresh (lemon yellow), except for a thin amber-coloured crust, and has not been eroded into pebble shapes, so it has been suggested that this amber has not been reworked and would therefore be contemporaneous (Grimaldi & Ross Reference Grimaldi, Ross, Fraser and Sues2017). However, a detailed study of the amber in situ is required, before it goes through the mining process, to confirm this.
Although the age of Baltic amber has long been considered to be Eocene, there have been different opinions as to which stage or stages the amber came from, particularly for the Upper Blue Earth, with conflicting dates provided by absolute (radiometric) dating and biostratigraphical dating using microfossils. This has led to confusion. For instance, Sivkov & Zhamoida (Reference Sivkov, Zhamoida, Gritsenko, Sivkov, Yurov and Kostianoy2017, p. 39) referred to the Prussian Formation as Priabonian (late Eocene) but on the same page referred to the Upper Blue Earth as middle Eocene.
Based on aspects of the insect fauna, Wappler (Reference Wappler2005) considered Baltic amber was Lutetian in age, whereas Bukejs et al. (Reference Bukejs, Alekseev and Pollock2019) suggested a Bartonian age for the Baltic amber-producing forest, and others have assumed a Priabonian age for Baltic amber (e.g., Brazidec & Vilhelmsen Reference Brazidec and Vilhelmsen2022). Wappler (Reference Wappler2005) based his argument on the presence of four extinct insect genera found in both Baltic amber and at Eckfeld Maar (Germany) of Lutetian age. However, other extinct genera present in Baltic amber have been found in other Eocene deposits, e.g., Marjorietta (Hymenoptera: Ichneumonidae) and Cyttaromyia (Diptera: Cylindrotomidae) (Antropov et al. Reference Antropov, Belokobylskij, Compton, Dlussky, Khalaim, Kolyada, Kozlov, Perfilieva and Rasnitsyn2014; Krzemiński et al. Reference Krzemiński, Blagoderov, Azar, Lukashevich, Szadziewski, Wedmann, Nel, Collomb, Waller and Nicholson2019). There are many extant genera in Baltic amber and some even extend back into the Cretaceous (e.g., see Pełczyńska et al. Reference Pełczyńska, Blagoderov, Krzemiński and Soszyńska2025), so insect genera can persist for a long time and, given the incompleteness of the insect fossil record, they may not be reliable for dating. Finding the same species in Baltic amber and other fossil faunas is much more significant. A species of braconid wasp (Hymenoptera: Braconidae), Bassus quadrangularis (Brues Reference Brues1933), that was originally described from Baltic amber, was found in the Insect Limestone of the Bembridge Marls (Isle of Wight, England) of late Priabonian age (Antropov et al. Reference Antropov, Belokobylskij, Compton, Dlussky, Khalaim, Kolyada, Kozlov, Perfilieva and Rasnitsyn2014; Ross & Self Reference Ross and Self2014). However, the direct comparison of insects trapped in three-dimensional form in amber with those preserved as two-dimensional compressions in rock can be challenging due to the differences in how they are preserved and what diagnostic characters are visible for study. Often only the wing venation can be studied in compression fossils, whereas features of the genitalia are useful for identifying some species in amber. For instance, sciarid fungus gnats (Diptera: Sciaridae) are abundant in Baltic amber and the species are primarily identified based on the form of their male genitalia (Mohrig & Röschmann Reference Mohrig and Röschmann1994); however, their wing venation is conservative with only subtle differences, so fossil wings of this family are generally not useful taxonomically. There are also taphonomic biases: in particular, small insects are common in Baltic amber, whereas large insects, such as dragonflies, which are well represented as fossils in sedimentary rocks (see Kohli et al. Reference Kohli, Ware and Bechly2016), are very rare in amber. The Baltic amber ant fauna (Hymenoptera: Formicidae) is dominated by wingless workers due to their habit of running up and down tree trunks, whereas in sedimentary deposits the ant faunas are dominated by winged males and females due to them having drowned in water bodies while swarming. However, compositionally, at subfamily level, the Baltic amber ant fauna is similar to that of Florissant (Colorado, USA) of Priabonian age (Antropov et al. Reference Antropov, Belokobylskij, Compton, Dlussky, Khalaim, Kolyada, Kozlov, Perfilieva and Rasnitsyn2014; Ross et al. Reference Ross, Zhou, Hoffeins and Crighton2024). So, although there are some similarities of the Baltic amber insect fauna with other Eocene fossil insect faunas, there are also many differences, which could be due to different palaeoecological, palaeogeographical, palaeoenvironmental and taphonomic conditions, as well as age (compare Larsson Reference Larsson1978; Weitschat & Wichard Reference Weitschat and Wichard2002; Meyer Reference Meyer2003; Wappler Reference Wappler2003; Lutz et al. Reference Lutz, Kaulfuss, Wappler, Löhnertz, Wilde, Mertz, Mingram, Franzen, Frankenhäuser and Koziol2010; Ross Reference Ross2014; Ross & McGowan Reference Ross and McGowan2019). A wider holistic study is required to compare the insect fauna of Baltic amber with other Eocene insect faunas, but many taxa from Baltic amber and Eocene deposits described from the 19th and early 20th centuries require revision (Szwedo et al. Reference Szwedo, Drohojowska, Popov, Simon and Wegierek2019).
There is no in situ evidence for the Baltic amber-producing forest, though it has been suggested that it was tropical/sub-tropical and persisted for 10 million years (Weitschat Reference Weitschat, Rascher, Wimmer, Krumbiegel and Schmiedel2008). It has also been suggested that the forest flourished in the early Eocene during a warm phase but then cooling in the mid-Eocene increased stress levels that led to increased resin production (Słodkowska et al. Reference Słodkowska, Kramarska, Kasiński, Kosmowska-Ceranowicz, Gierlowski and Sontag2013). However, Seyfullah et al. (Reference Seyfullah, Beimforde, Corso, Perrichot, Rikkinen and Schmidt2018) suggested that large deposits of resin can be built up over thousands of years, as demonstrated by the amount of resin produced by the Kauri pine (Agathis australis) in New Zealand over a relatively short time. Recent studies of the lichens and flora in Baltic amber were consistent in indicating that the forest was warm-temperate, not tropical (Kaasalainen et al. Reference Kaasalainen, Schmidt and Rikkinen2017; Sadowski et al. Reference Sadowski, Schmidt, Seyfullah and Kunzmann2017, Reference Sadowski, Seyfullah, Regalado, Skadell, Gehler, Gröhn, Hoffeins, Hoffeins, Neumann, Schneider and Schmidt2019, Reference Sadowski, Schmidt and Denk2020). A recent thorough study of the conifer flora revealed that it is unlike that of middle Eocene floras, and thus a Priabonian age was accepted by Sadowski et al. (Reference Sadowski, Schmidt and Kunzmann2022) for Baltic amber from the Upper Blue Earth.
There are a number of papers that discuss the stratigraphy of the Baltic amber-bearing beds, although they have produced differing interpretations as to the ages of these units. These papers are critically reviewed here and the different aspects of stratigraphy – lithostratigraphy, chronostratigraphy, absolute dating and biostratigraphy – are investigated with a view to updating them and bringing the information together to gain a better understanding of the most likely ages of the different Baltic amber-bearing beds and the amber itself. The preliminary results of this study were presented at the International Palaeoentomological Conference, Fossil Insects – Bridging Past and Present, Kraków, Poland on 23 May 2025 (Ross et al. Reference Ross, Bojarski and Szwedo2025).
1. Lithostratigraphy
For a detailed history of investigation into the geology of Kaliningrad Oblast since 1945 see Iakovleva (Reference Iakovleva2023). Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977) produced a detailed lithostratigraphy of the amber-bearing beds on the Samland Peninsula, though given this work is in Russian it has often been ignored by researchers in other countries. This work indicated that amber occurred at six horizons. Standke (Reference Standke, Rascher, Wimmer, Krumbiegel and Schmiedel2008) reviewed previous work on the lithostratigraphy by other researchers and also produced a detailed lithostratigraphy. Four amber-bearing horizons were referred to (oldest to youngest): Untere Blaue Erde, Wilde Erde, Blaue Erde and Grüne Mauer/Unterer Gestreifter Sand. Weitschat & Wichard (Reference Weitschat, Wichard and Penney2010) provided English translations of these beds: Lower Blue Earth, Wild Earth, Blue Earth and Green Wall. It is worth pointing out that Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977) did not record amber from the Wild Earth. Kharin & Eroshenko (Reference Kharin and Eroshenko2017) provided a summary of the occurrence of amber in the area and modified Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977)’s stratigraphy, with the units translated into English. They listed amber-bearing horizons at the base of the Sambian Formation, Lower Blue Ground within the Alksk Formation, Blue Ground, Krant quicksand facies and White Wall within the Prussian Formation, and Green Wall which equates to the Palvé Formation. They considered that the only two primary deposits were the Lower Blue Ground and Blue Ground and that the amber at the base of the Sambian Formation and above the Blue Ground was reworked, as well as amber being reworked into Quaternary deposits.
Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) produced a detailed lithostratigraphy of the sequence in the Yantarny P-1 borehole. Here they used the word ‘Earth’ instead of ‘Ground’ and ‘Lower’ and ‘Upper’ to distinguish the two wild earths and two blue earths and referred to the Alka Formation instead of ‘Alksk’. The borehole commenced in anthropogenic deposits, with the first in situ unit reached being the Upper Blue Earth, through other units and extending down into Cretaceous deposits. The base of the Prussian Formation was recorded at a depth of 18.5 m, the base of the Alka Formation at 37.2 m and the base of the Sambian Formation at 51.7 m. Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) also provided a modified version of Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977)’s stratigraphy in English, also using ‘Earth’ instead of ‘Ground’ and ‘Lower’ and ‘Upper’. They provided a detailed stratigraphy of the units exposed in Primorsky opencast mine, which is mined for amber.
According to Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), below the Sambian Formation lies the Lubava Formation; however, Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977) recognised another formation, referred to as the Zaostrovskaya Formation by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021), lying between the Sambian and Lubava formations. There appears to be a facies change at a depth of 60.5 m in the P-1 borehole which may mark the base of the Zaostrovskaya Formation, but this formation was not recognised by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020). Given that no amber has been recorded from below the Sambian Formation, the presence or absence of the Zaostrovskaya Formation does not affect the discussion in this paper; thus, here the names of the stratigraphic units from the Upper Blue Earth and below are taken from Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and the names of the units above the Upper Blue Earth are taken from Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). Seven of the stratigraphic units have been recorded as containing amber, within the Sambian, Alka, Prussian and Palvé formations.
Above the Palvé Formation lies the Kurshskaya Formation, of which the lowest 0.5 m is referred to as the Chocolate clays, which is overlain by the Brown sands (Iakovleva et al. Reference Iakovleva, Aleksandrova and Mychko2021). This formation does not contain amber.
Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977) showed that the stratigraphic units demonstrate considerable lateral variation – for instance, the Upper Blue Earth ranges from 0.5 m to 13 m in thickness. Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) provided accurate depths for the different stratigraphic units in the P-1 borehole, so accurate thicknesses can be calculated for these units in the borehole. While 5.5 m of Upper Blue Earth was measured, it is not known how much of this unit is missing at the top. Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) indicated that the Upper Blue Earth in Primorsky opencast mine ranges from 5.1 m to 6.2 m, so perhaps very little is missing from the P-1 borehole section. Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) only measured the top 2.7 m of the Upper Blue Earth in the Primorsky mine.
Katinas (Reference Katinas1971, p. 128) provided detailed information on quantities, size fractions and specific gravities of the amber from the different units in the Yantarny opencast mine and at Cape Taran (NW point of the Samland Peninsula). In a 9 m sequence of Upper Blue Earth (though with no data from the top metre) the lowest metre contained 45 g per m3 of amber, which increased to a maximum concentration of 2,677 g per m3 at 6–7 m from the top and then decreased to 379 g per m3 at 1–2 m from the top. In other units the quantity was much lower; for the Lower Blue Earth it was 92 g per m3, Upper Quicksand was 143 g per m3, White Wall 68 g per m3 and Green Wall 22 g per m3, with only a trace of amber recorded from the Upper Wild Earth. Regarding size fractions, from the Lower Blue Earth all pieces were less than 20 mm in size, with 63 % less than 6 mm. From the Upper Blue Earth, in the lowest metre and 1–2 m from the top, more than 72 % was less than 20 mm in size, whereas more than 48 % was more than 20 mm in size from 3–7 m from the top, of which 25–29 % was more than 32 mm. From the Upper Quicksand more than 76 % was more than 20 mm in size, of which 48 % was more than 32 mm. In the White Wall 90 % was less than 20 mm, whereas the Green Wall had varying amounts of different sizes ranging from 15–26 % in each of the size fractions. Regarding specific gravity (SG), all of the amber from the Lower Blue Earth had a high SG of over 1.05. From the Upper Blue Earth, in the lowest 4 m more than 78 % had an SG of 1–1.05, whereas at 1–5 m from the top 52–66 % had an SG of 1–1.05, with 32–43 % over 1.05. The Upper Quicksand had 50/50 amber with SG of 1–1.05 and over 1.05. The White Wall was similar to the upper part of the Upper Blue Earth, whereas the Green Wall had 30 % amber with an SG of less than 1.0, 44 % with 1–1.05 and 26 % with more than 1.05. These figures give an indication of relative energy levels and sorting during deposition. The highest relative energy was when the Lower Blue Earth was deposited but the amber pieces were small, so either these pieces were preferentially sorted or this may reflect the size of resin pieces being produced at that time. The next highest energy level was during the deposition of the Upper Quicksand with large pieces being concentrated, though at a much lower quantity than in the Upper Blue Earth. The lowest energy was during the deposition of the Green Wall, but the energy level was fluctuating as pieces of a variety of different sizes and specific gravities were deposited in this unit. However, given that there is considerable lateral variation in these units then the depositional environment would have been changeable.
For dating it is important to have type sections (stratotypes) with accurate thicknesses of the different units, though unfortunately the stratotype of the Sambian Formation is lost and the ‘combined stratotype’ of the Alka Formation provided varying thicknesses of its members (Iakovleva Reference Iakovleva2023). The stratotype of the Prussian Formation is the Primorsky mine section, although again the thicknesses of its members are variable within the mine, and the Upper Wild Earth and lower part of the Upper Blue Earth have not been sampled for detailed microfossil analysis. The stratotype of the Palvé Formation is also in the Primorsky mine (Iakovleva Reference Iakovleva2023). For the purpose of this study, the thicknesses of the Upper Blue Earth and units below are taken from Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and the thicknesses of the units above the Upper Blue Earth are taken from Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). See Figure 1 for the combined lithostratigraphy of the Baltic amber-bearing horizons from the Yantarny P-1 borehole and Primorsky opencast mine.
The lithostratigraphy of the Baltic amber-bearing horizons on the Samland Peninsula, based on the Yantarny P-1 borehole and Primorsky opencast mine sections from Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). Units containing amber are shaded, with the Upper Blue Earth having the highest concentration.

2. Chronostratigraphy
For any study where stratigraphical dating is required, the latest chronostratigraphy should be utilised, whereby stage boundaries have been given ages in millions of years. Today the internationally agreed chronostratigraphy is provided by the International Commission on Stratigraphy, and the latest Chronostratigraphic Chart is available from their website (https://stratigraphy.org/chart). The latest version available at the time of writing this paper is v2024/12. The ages of the stage boundaries are regularly being refined but the changes are slight. For instance, the base and top of the Eocene Epoch are currently considered to be 56.00 and 33.9 million years old (Ma) respectively, whereas they were given as 54.5 Ma and 33.7 Ma respectively by Weitschat & Wichard (Reference Weitschat, Wichard and Penney2010). Given that time moves forwards, geological time ranges are referred to using the oldest age first, so the Eocene Epoch is 56–33.9 Ma.
3. Absolute dating
Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977) were the first to provide absolute dates of the Baltic amber-bearing beds based on the Potassium-Argon (K-Ar) dating of glauconite from the sediments. They provided dates of 41 Ma for the Alka Formation, 37(±1.5) Ma for the Prussian Formation and 34(±3.7) Ma for the Palvé Formation. However, later, Ritzkowski (Reference Ritzkowski1997) provided much older absolute dates of 47.0(±1.2) Ma for the Upper Wild Earth, 44.1(±1.1) Ma for the Upper Blue Earth (Prussian Formation) and 38.1–8(±1.4) Ma for the Green Wall (Palvé Formation) respectively. The first two dates from Ritzkowski (Reference Ritzkowski1997) fall within the Lutetian Stage, which has been widely accepted as the age of Baltic amber, or even as a minimum age (e.g., Wolfe et al. Reference Wolfe, Tappert, Muehlenbachs, Boudreau, Mckellar, Basinger and Garrett2009). However, this method of dating can be unreliable because if the glauconite is thermally altered it can produce younger results (Obradovich Reference Obradovich1988). Conversely, glauconite can also be reworked or, if it is contaminated with non-glauconised mica/illite, then older results are produced (Clauer et al. Reference Clauer, Huggett and Hillier2005). The glauconite sample has to be unaltered and pure to provide an accurate result (Wu et al. Reference Wu, Bai, Shi, He and Qiu2023). Given that there have only been two studies to date the amber-bearing beds using this method, which provided widely different results from each other, and given that these results have not been replicated by other labs, then they can only be considered as unreliable. More dating is required, particularly of the Upper Blue Earth, using pure samples of glauconite and reproducibility tested by independent labs.
4. Biostratigraphy
There are also differing (though not too dissimilar) opinions on the ages of the amber-bearing formations based on microfossils, primarily dinoflagellate cysts. However, there should be caution when interpreting the results as there is a risk of reworking, and this is certainly known in Denmark where it is difficult to interpret where the Priabonian/Rupelian boundary lies (King Reference King, Gale and Barry2016b). Therefore, the first occurrence (FO) of a species is often more reliable than its last occurrence (LO). Some species can be diachronous, appearing or disappearing at different times in different regions.
Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) provided the first detailed investigation in which they identified many different taxa of dinoflagellate cyst (dinocysts) from the P-1 borehole core. This included 213 taxa (128 species and subspecies) from the base of the Sambian Formation up to the top of the Upper Blue Earth Member. It should be noted that the published tables provided a summary of the data. Not all the samples were listed so perhaps not all the samples were analysed, and they were not taken at regular intervals. Also, this study did not provide details on how much material was sampled, nor how many specimens of each taxon were recovered, though a general indication of abundance was provided. They correlated the occurrences with dinocyst ‘beds’ and the traditional numbered dinocyst zonation scheme and concluded that the Sambian Formation corresponded to the Alisocysta margarita and Deflandrea oebisdfeldensis ‘beds’ of Thanetian to Ypresian age, the Alka Formation corresponded to the Areosphaeridium diktyoplokum ‘bed’ within the Lutetian and the Prussian Formation to the Rhombodinium perforatum and Charlesdowniea clathrata angulosa ‘beds’ of Bartonian to Priabonian age (Aleksandrova & Zaporozhets Reference Aleksandrova and Zaporozhets2008b).
Lukashina (Reference Lukashina2010) recorded foraminifera from the P-1 borehole at depths of 52.5–60.8 m and 37.6–51 m, but most were only identified to genus level and detailed depth data were not provided. She considered the ‘Alkskaya’ (Alka) Formation to be upper Middle Eocene in age.
Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) also investigated the dinocysts, though recorded fewer taxa than Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a). They only recorded 107 taxa (59 species) from the base of the Sambian Formation up to the top of the Upper Blue Earth. Their samples were taken from the same depths as for Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and all samples were listed in the appendix, indicating which records were reworked. Like Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), they did not mention the size of the samples, nor record the numbers of specimens recovered of each taxon. They considered that the Sambian Formation was Lower Eocene, the Alka Formation was Middle Eocene and the Prussian Formation was Upper Eocene, but did not refer to the international stages.
Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) undertook a detailed investigation of dinocysts from the Primorsky opencast mine and took samples every 30 or 60 cm from the top 2 m of the Upper Blue Earth up to the bottom 1.5 m of the Kurshskaya Formation. This time they recorded how many specimens of each taxon were recovered and separated out specimens that they considered were reworked. They recorded 171 taxa (164 species) and considered that the Upper Blue Earth correlated with the Rhombodinium perforatum zone, the rest of the sequence correlated with the Thalassiphora reticulata zone and that the entire sequence was Priabonian in age.
Kuzmina & Iakovleva (Reference Kuzmina and Iakovleva2023) studied the palynomorphs from the Primorsky mine, from the same samples as Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). However, they only provided detailed results for the Palvé and lower part of the Kurshkaya formations. Only two samples from the Upper Blue Earth and only one sample each from the Upper Quicksand and White Wall were analysed. Like Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021), they also considered that the entire sequence was Priabonian in age.
Iakovleva (Reference Iakovleva2023) reviewed previous work on the microfossils, including reproducing the dinocyst data from Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). She also provided information on foraminifera and other fossils that have been recorded from the different formations from the Kaliningrad Oblast region and attempted to correlate them with the International Geological Time Scale. Although she listed many species of foraminifera, most were benthic and of the few planktonic species mentioned, none of them are zonal markers as outlined in the zonal scheme in Spiejer et al. (Reference Spiejer, Pälike, Hollis, Hooker, Ogg, Gradstein, Ogg, Schmitz and Ogg2020). Iakovleva (Reference Iakovleva2023) considered the Sambian Formation to be Ypresian, the Alka Formation to be Lutetian–Bartonian and the Prussian and Palvé formations to be Priabonian in age. She indicated that there were hiatuses between the Sambian, Alka and Prussian formations, though with some uncertainty.
None of the dinocyst studies have been correlated with the benchmark of stratigraphy, the Geologic Time Scale 2020 (Gradstein et al. Reference Gradstein, Ogg, Schmitz and Ogg2020), which contains detailed correlation charts for the dinocysts, based primarily on the work of King (Reference King, Gale and Barry2016a) for the North Sea. This included both the traditional numbered dinocyst zonation scheme and a new numbered scheme. Both schemes are based on the first and last occurrences of different species.
Herein we attempt to correlate the published dinocyst taxon records from Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) with the zonation schemes in the Geologic Time Scale 2020 to provide more accurate and up-to-date dating of the amber-bearing beds. To do this, all the key zonal and subzonal marker taxa and other useful taxa from the Paleocene, Eocene and Oligocene listed in King (Reference King, Gale and Barry2016a) and Spiejer et al. (Reference Spiejer, Pälike, Hollis, Hooker, Ogg, Gradstein, Ogg, Schmitz and Ogg2020) were searched for in the published data of the above three papers. The occurrences of each taxa were then investigated to determine whether they would be useful in determining where the zones lie. The following list of species is of those whose FO and/or LO are considered to be useful zonal/subzonal markers or other datum markers by King (Reference King, Gale and Barry2016a) and Spiejer et al. (Reference Spiejer, Pälike, Hollis, Hooker, Ogg, Gradstein, Ogg, Schmitz and Ogg2020), oldest to youngest, that have been recorded from the Samland Peninsula:
Damassadiniium californicum. FO marks the base of D1a and DP1, LO is within DP7. FO marks the base of the Danian in Denmark but higher up in Belgium (King Reference King, Gale and Barry2016b). Damassadiniium cf. californicum was recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from the Lubava Formation. Given that the identification is not certain, it may not be reliable.
Spongodinium delitiense. LO marks the top of D1b and DP2. Not well calibrated according to King (Reference King, Gale and Barry2016b). Recorded by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) from throughout the Lubava Formation.
Senoniasphaera inornata. LO marks the top of DP3b. FO marks the base of the Danian in Denmark though is also known from the Cretaceous (King, Reference King, Gale and Barry2016b). Recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from low down in the Lubava Formation.
Cerodinium striatum. FO marks the base of DP4b, LO is within DP11a. Recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from the upper half of the Lubava Formation and reworked into the base of the Sambian Formation.
Spinidinium densispinatum. FO marks the base of DP6, LO marks the top of DP8a. Also recorded from the Cretaceous (King Reference King, Gale and Barry2016b). Magallanesium (Spinidinium) densispinatum was recorded by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) from below the Lubava Formation. Spinidinium cf. densispinatum was recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from the top of the Lubava Formation.
Cerodinium speciosum. FO marks the base of DP7. FO higher in Germany than Denmark (King Reference King, Gale and Barry2016b). Recorded by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) from the Lubava (upper half) and Sambian formations and Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from the Lubava Formation (single depth). Cerodinium cf. speciosum was recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) reworked into the Alka and Prussian formations.
Isabelidium? viborgense. FO marks the base of DP9, LO marks the top of DP9. Recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from the lower half of the Lubava Formation and by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) from a single depth in the Sambian Formation. Given the short range of this taxon, it could be an important zonal marker, but the records are conflicting. The record from the Sambian Formation appears to be more reliable.
Alisocysta margarita. FO marks the base of D4a, LO marks the top of DP11c. May be diachronous according to King (Reference King, Gale and Barry2016b). Recorded by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) throughout the Sambian Formation and a single depth of the Alka Formation. Recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from the Lubava Formation and considered to be reworked into the base of the Sambian Formation. It is odd that Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) recorded different ranges. The Alka Formation record appears to be spurious but the latest Sambian Formation record may be significant.
Palaeoperidinium pyrophorum. LO marks the top of DP10b. May occur rarely higher up according to King (Reference King, Gale and Barry2016b). Recorded by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) from within and below the Lubava Formation with a single depth occurrence within the Sambian Formation.
Muratodinium fimbratum. FO corresponds with or is just above the base of D5a and DE1. Recorded from the Prussian (Upper Blue Earth and White Wall) and Palvé formations by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). Although its FO is discussed by King (Reference King, Gale and Barry2016b), it is not a zonal marker and in the Samland sequence it occurs much higher up so is not useful here.
Deflandrea oebisfeldensis. A high abundance (influx) of this species marks the base of DE2b to the top of DE2b, though the top of the influx is diachronous (King, Reference King, Gale and Barry2016b). Its LO marks the top of zones D7b and DE6. It is recorded throughout the Sambian Formation and all three members of the Alka Formation by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and the bottom two members by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), though the record from the Lower Quicksand is anomalous and may be reworked. Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) recorded a high abundance in the Lower Blue Earth but this was not seen by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020). It was also recorded from the Prussian and Palvé formations by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) but considered to be reworked.
Deflandrea phosphoritica. FO marks the base of DE4b, LO marks the top of DM1a. It is recorded from single depths from the Lubava Formation and Alka Formation (Lower Blue Earth and Lower Quicksand) by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), and throughout the Prussian and Palvé formations and all samples from the Kurshskaya Formation by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). This taxon has a very long range so is not useful here and the single record from the Lubava Formation is spurious.
Charlesdowniea coleothrypta. FO marks the base of D8a and DE8a. Although it was recorded from the Prussian Formation (Upper Wild Earth and Upper Blue Earth) by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), records from the Upper Blue Earth and the Palvé Formation were considered to be reworked by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Areospaeridium diktyoplokum. FO marks the base of D9a and DE9a, LO marks the top of zones D12c and DE20b, though considered unreliable due to reworking in the Oligocene (King, Reference King, Gale and Barry2016b). It is recorded from the Alka: Lower Quicksand Member by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), throughout the Prussian and Palvé formations and into Kurshskaya Formation by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Areoligera senonensis. FO in the Cretaceous, though there is an influx, the top of which marks the top of DE9a. It is recorded from the Sambian Formation by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), and from the Prussian (Upper Blue Earth and Upper Quicksand) and Palvé formations by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). Given that this species is long-ranging and is not recorded in high abundance, it is not useful here.
Cleistosphaeridium diversispinosum. An influx commenced within zones D9 and DE9b and ended at the top of D9e and DE14. It is only recorded from a single depth in the Prussian Upper Blue Earth Member by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and from throughout the rest of the Prussian Formation, Palvé Formation, and into the Kurshskaya Formation by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). As there is no indication of an influx then it is not useful here.
Phthanoperidinium comatum. FO is within DE9b. It is rare in Denmark and its first occurrence is poorly defined there (King, Reference King, Gale and Barry2016b). Its last occurrence is within zone D14, at the top of DO2. It has been recorded from the Alka Formation and all members of the Prussian and Palvé formations, and most samples from the Kurshkaya Formation, by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Heteraulacacysta porosa. FO is within D9e and DE13, though its last occurrence is at or above the top of D12 and DE20b (King Reference King, Gale and Barry2016b). Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) recorded it from the Alka (Lower Quicksand) and Prussian (Upper Blue Earth) formations, though only at a single depth from the former. Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) recorded it from the Prussian (Upper Blue Earth, Upper Quicksand and White Wall), Palvé (Green Wall) and Kurshskaya formations. Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) recorded H. cf. porosa from the Prussian Formation (Upper Wild Earth Member).
Diphyes ficusoides. LO is within zones D9e and DE14. Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) recorded it from the Prussian formation, but only from single samples in the Upper Blue Earth and White Wall members. These records are higher up than its LO so are anomalous and may be reworked.
Glaphyrocysta intricata. FO marks the base of zones D10a and DE15. It is only recorded from one sample by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021), from the Prussian Upper Quicksand Member, higher than its FO so is not useful here.
Rhombodinium draco. FO marks the base of D10b and DE16, LO marks the top of D14 and DO5c. It was recorded by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) from the Prussian (Upper Blue Earth, Upper Quicksand and White Wall) and Palvé formations. Given it has a long range and was not recorded by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) then it is of little use here.
Corrudinium incompositum. LO marks the top of D10b and DE17. It is recorded from one depth from the Alka Lower Wild Earth Member by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), and all members from the Prussian, Palvé and Kurshskaya formations by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021). There is a problem in that it is only used as a zonal marker by Spiejer et al. (Reference Spiejer, Pälike, Hollis, Hooker, Ogg, Gradstein, Ogg, Schmitz and Ogg2020), not by King (Reference King, Gale and Barry2016a) and is not discussed by King (Reference King, Gale and Barry2016b). In the Samland sequence it occurs consistently with species that are younger than its LO as given by Spiejer et al. (Reference Spiejer, Pälike, Hollis, Hooker, Ogg, Gradstein, Ogg, Schmitz and Ogg2020). It seems that Spiejer et al. (Reference Spiejer, Pälike, Hollis, Hooker, Ogg, Gradstein, Ogg, Schmitz and Ogg2020) made an error in placing its LO at the top of zones D10 and DE17.
Rhombodinium porosum. FO marks the base of D11a and DE18. LO near the top of D11b and DE18 but is also considered to coincide with the LO of Heteraulacacysta porosa which is at the top of or above D12 and DE20b (King Reference King, Gale and Barry2016b), so the position of its LO is uncertain. It is recorded from all members of the Prussian and Palvé formations by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Rhombodinium perforatum. FO marks the base of D12a and DE19. LO marks the top of DE20a, though can coincide with the last occurrence of A. diktyoplokum at the top of DE20b (King Reference King, Gale and Barry2016b). This species has a relatively short range so its occurrence is significant. It was recorded from all members of the Prussian and Palvé formations by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021), but was not recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020). Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) also recorded it from the base of the Kurshkaya Formation.
Charlesdowniea (Kisselovia) clathrata angulosa. FO corresponds with the base of D12a and DE19, though can be above or below the FO of R. perforatum (King Reference King, Gale and Barry2016b). It is recorded from the Prussian Formation (near the top of the Upper Wild Earth, and Upper Blue Earth) by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), however this subspecies was not recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), nor Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Thalassiphora fenestrata. FO marks the base of D12b within DE19, though King (Reference King, Gale and Barry2016b) considers it roughly corresponds with that of R. perforatum (base of D12a and DE19). Its LO is poorly defined, before the last occurrence of A. diktyoplokum (King Reference King, Gale and Barry2016b). This species has been recorded from the Prussian (Upper Blue Earth, Upper Quicksand and White Wall) and Palvé formations by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Thalassiphora reticulata. FO marks the base of D12c and DE20a, though this requires confirmation (King, Reference King, Gale and Barry2016b). It has been recorded from a single depth in the Alka: Lower Quicksand Member by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), though this record is spurious. It was recorded from the Prussian (Upper Quicksand and White Wall) and Palvé formations by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Melitasphaeridium pseudorecurvatum. LO near the base of D12c and DE20a. It was recorded from the Prussian Upper Wild Earth Member by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and remaining members of the Prussian and Palvé formations by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Areoligera michoudii. LO within subzones D12c and DE20a. It was recorded from all members of the Prussian and Palvé formations by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Glaphyrocysta semitecta. LO marks the top of DO1b. It was recorded from the Prussian (Upper Blue Earth, Upper Quicksand and White Wall) and Palvé formations and most samples from the Kurshkaya Formation by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
Enneadocysta pectiniformis. Abundant up to the top of zones D14a and DO3 though can be reworked into younger deposits. It has been recorded from the Alka (Lower Wild Earth and Lower Quicksand) Formation and throughout the Prussian and Palvé formations into the Kurshkaya Formation by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) and Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021).
To try to work out the ages of the formations and the amber-bearing horizons, the species occurrences were plotted against the lithostratigraphy for each of the three papers (Aleksandrova & Zaporozhets Reference Aleksandrova and Zaporozhets2008a, Kasiński et al. Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020, and Iakovleva et al. Reference Iakovleva, Aleksandrova and Mychko2021) (Figs 2–4). The occurrences of the useful species were then combined, with gaps and anomalous/spurious records removed (Fig. 5). Their ranges were then examined relative to each other to look for reliable first and last occurrences that could mark zonal and subzonal boundaries (Fig. 6). From this the most likely positions of the stage boundaries were then added (Fig. 7).
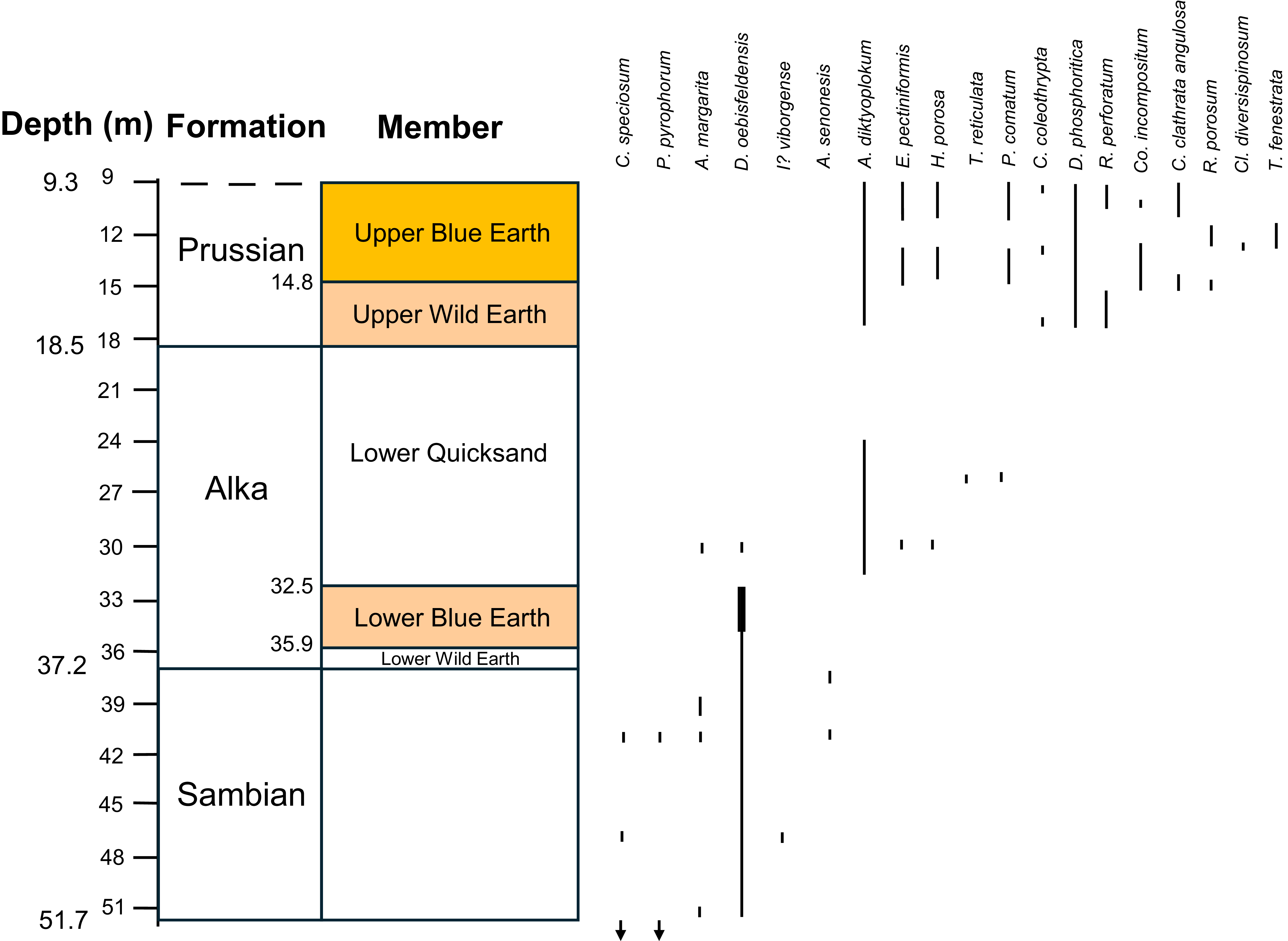
The occurrences of dinocyst species and subspecies that have been used for dating, recorded by Aleksandrova & Zoporozhets (Reference Aleksandrova and Zaporozhets2008a) from the P-1 borehole. The thicker line for D. oebisfeldensis indicates high abundance.

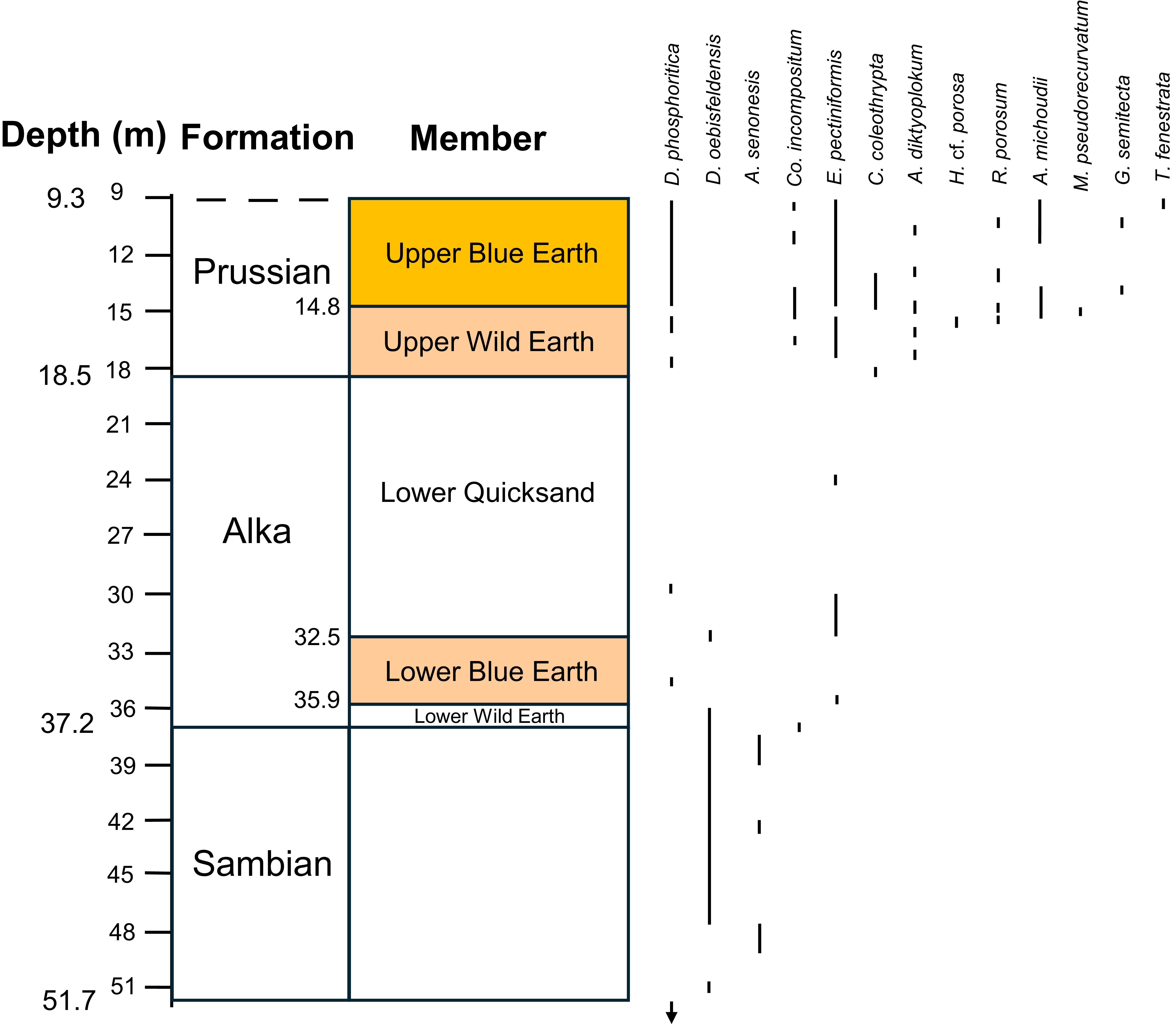
The occurrences of dinocyst species that have been used for dating, recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) from the P-1 borehole.

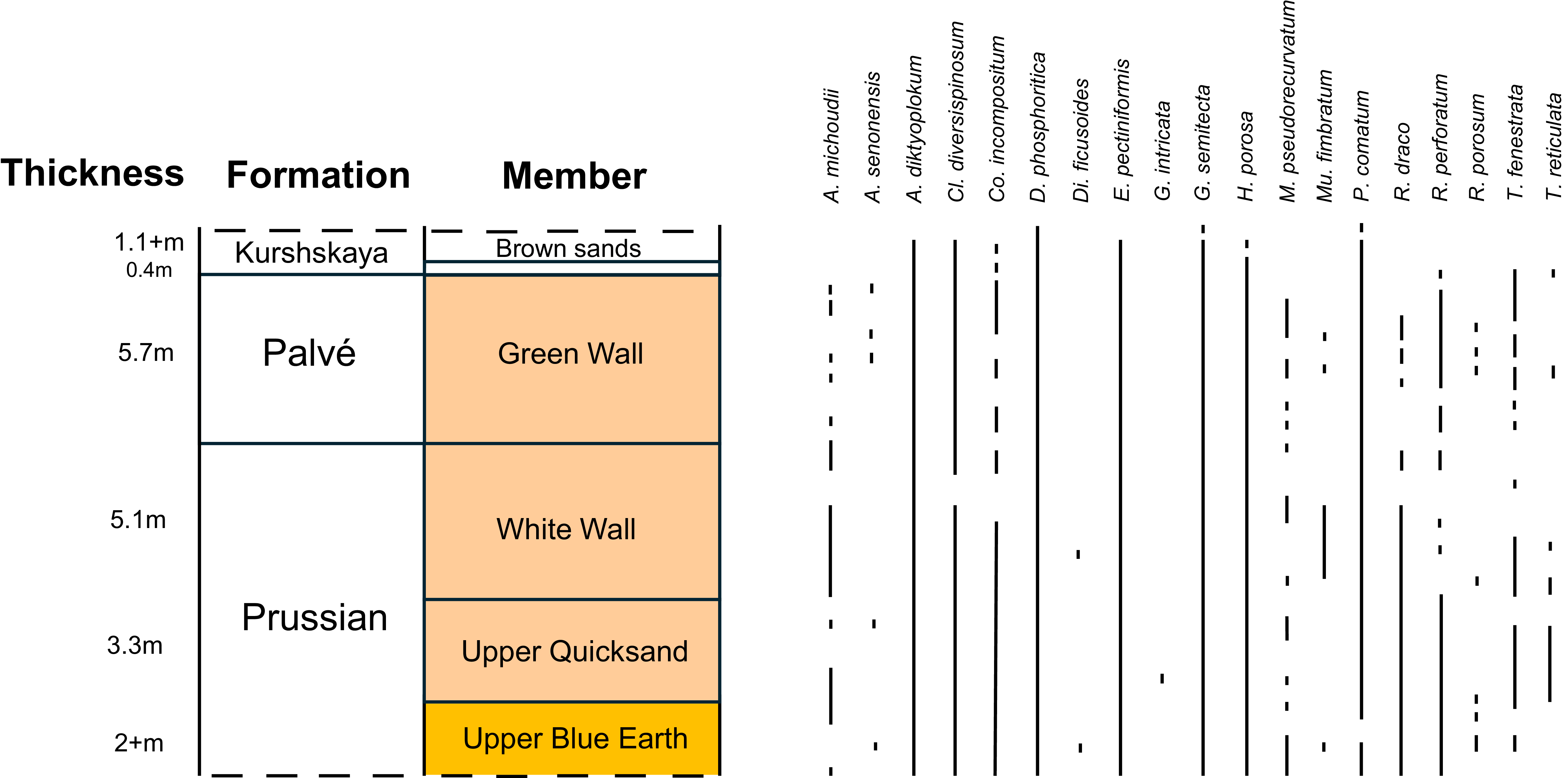
The occurrences of dinocyst species that have been used for dating, recorded by Iakovleva et al. (Reference Iakovleva, Aleksandrova and Mychko2021) from Primorsky opencast mine.

Combined records of the useful dinocyst species and subspecies, with gaps and spurious/anomalous records removed.

The most useful dinocyst species with first and last occurrences, marked by a horizontal bar, that indicate where zonal and subzonal boundaries probably lie.

Correlated lithostratigraphy, dinocyst biostratigraphy and chronostratigraphy of the Baltic amber-bearing horizons of the Samland Peninsula.

5. The ages of the formations
The Lubava Formation (sensu Kasiński et al. Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) was recorded from 66.4–51.7 m (14.7 m thick) in the P-1 borehole, below the Sambian Formation. Although no amber has been recorded for this formation it is worth discussing its age. Based on the dinocysts, the presence of Senoniasphaera inornata, Cerodinium striatum, Cerodinium speciosum and Palaeoperidinium pyrophorum indicate that this formation is Danian–Selandian in age. However, there are conflicting records. The records of Spongodinium delitiense occurring throughout the Lubava Formation and Spinidinium densispinatum occurring below the Lubava Formation, as recorded by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a), are odd given that the LO of S. delitiense (early Danian) is before the FO of S. densispinatum (late Danian); however, King (Reference King, Gale and Barry2016b) considers the LO of S. delitiense is not well calibrated and that S. densispinatum is also known from the late Cretaceous. The records of Alioscysta margarita and Isabelidium viborgense from low in the Lubava Formation, as recorded by Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), are odd given that the FOs of A. margarita and I? viborgense are late Selandian, conflicting with the early Danian LOs of S. delitiense and S. inornata. From these conflicting records it can only be assumed that not all of the species have been identified correctly, or the last occurrences are reworked or the species ranges are diachronous (or a combination of all three). The records of C. speciosum at a depth of 59 m in the P-1 borehole by both Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008a) and Kasiński et al. (Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020) are significant. The FO of this species marks the base of zone DP7 and the position of the Danian–Selandian boundary, so the boundary could be at a depth of 59 m in the P-1 borehole.
The Sambian Formation was recorded from 51.7–37.2 m in the P-1 borehole. The presence of Cerodinium speciosum, Palaeoperidinium pyrophorum, Alisocysta marginata and Isabelidium? viborgense suggest that this formation is Selandian–Thanetian in age. Although useful records are sparse, given that I? viborgense has a very short range, its record at a depth of 47 m is significant and would therefore mark the position of zone DP9. The base of the Sambian Formation would therefore be early Selandian, about 61 Ma. The subsequent records of P. pyromorphum and A. marginata (excluding the spurious record from the Alka Formation) at depths of 41 m and 38.5 m respectively, could mark the tops of DP10 and DP11. The Selandian–Thanetian boundary is just above the base of DP11, so if this interpretation is correct then the boundary occurs at a depth of about 40.5 m in the P-1 borehole.
The Alka Formation was recorded from 37.2–18.5 m in the P-1 borehole. The high abundance of Deflandrea oebisfeldensis and presence of Areospaeridium diktyoplokum and Heteraulacacysta porosa suggest that this formation is Ypresian–Lutetian in age. Again, useful records are sparse although the high abundance of D. oebisfeldensis in the upper part of the Lower Blue Earth could correspond with subzone DE2b, of early Ypresian age, and suggests that the Thanetian–Ypresian boundary lies at the base of the Alka Formation or close to it. The appearance of A. diktyoplokum at a depth of 32 m (immediately above the high abundance of D. oebisfeldensis), which has its FO at the base of D9/DE9 (late Ypresian), indicates that there is a hiatus, so the FO of A. diktyoplokum is probably missing. This hiatus was also deduced by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008b). Although there is only the single depth record of Heteraulacacysta porosa at a depth of 30 m from this formation, this could be within DE13 within the Lutetian, and thus the hiatus may have ceased at or near the Ypresian–Lutetian boundary. Unfortunately, the top 5 m of the Alka Formation has not yielded any useful records so the age of this part is uncertain. Rhombodinium porosum, which ranges from the Bartonian–Priabonian, is recorded from the overlying formation, along with taxa that are solely Priabonian in age, which implies that the top of the Alka Formation is Bartonian, but there is no indication as to where the Lutetian–Bartonian boundary lies.
The Prussian Formation is 17.6 m thick, based on the combined sections from the P-1 borehole and Primorsky mine. The presence of Rhombodinium perforatum, Areoligera michoudii, Melitasphaeridium pseudorecurvatum, Thalassiphora fenestrata and Thalassiphora reticulata indicate that this formation is Priabonian in age. The FOs of R. perforatum, T. fenestrata and T. reticulata are staggered, occurring in the Upper Wild Earth, Upper Blue Earth and Upper Quicksand respectively. These are significant as they mark the bases of D12a/DE19, D12b and D12c/DE20a respectively, within the Priabonian. The Bartonian–Priabonian boundary occurs within D11b/DE18 so occurs at or near the base of the Prussian Formation.
The Palvé Formation was recorded from the Primorsky mine as being 5.7 m thick. As for the Prussian Formation, the presence of Rhombodinium perforatum, Areoligera michoudii, Melitasphaeridium pseudorecurvatum, Thalassiphora fenestrata and Thalassiphora reticulata indicate that this formation is Priabonian in age.
Only the lowest 1.5 m of the Kurshkaya Formation was sampled. Species were recorded that have their LOs near the Priabonian–Rupelian boundary or extend into the Rupelian. The LOs of Rhombodinium perforatum and Areospaeridium diktyoplokum mark the tops of DE20a and DE20b respectively and the Priabonian–Rupelian boundary lies between the two. Their highest occurrences were recorded at the base of and 1 m above the base of the Kurshkaya Formation respectively, thus the Priabonian–Rupelian boundary lies at about 0.5 m above the base of the formation, which corresponds with the boundary between the Chocolate clays and Brown sands. The LO of Glaphyrocysta semitecta marks the top of DO1b and this species was recorded at 1.5 m above the base of the formation, from the highest sample, so the top of DO1b is at this level or above. Phthanoperidinium comatum was also recorded from the highest sample and, given its LO is at the top of DO2, then it probably extends higher up the sequence.
Comparing the interpretation as outlined here with that of Iakovleva (Reference Iakovleva2023), both studies agree that the Prussian and Palvé formations are Priabonian in age; however, there are disagreements about ages of the lower formations. Iakovleva (Reference Iakovleva2023) considered the Sambian Formation was Ypresian and the Alka Formation was Lutetian–Bartonian, though here the Sambian Formation is considered to be Selandian–Thanetian and the Alka Formation is Ypresian–Bartonian. Iakovleva (Reference Iakovleva2023) indicated that there were hiatuses between the Sambian, Alka and Prussian formations, but with uncertainty. According to Spiejer et al. (Reference Spiejer, Pälike, Hollis, Hooker, Ogg, Gradstein, Ogg, Schmitz and Ogg2020), dinocyst zone DP10 had a duration of about 0.75 Ma and both zones DP11 and DP12 were about 1.75 Ma in duration, so it appears that the sedimentation rate decreased towards the top of the Sambian Formation and may have stopped completely during zone DP12, which means that there could be a short hiatus and that the top of the Thanetian may be missing from the sequence. Data is lacking from the top 5 m of the Alka Formation so it is uncertain if there is a hiatus between the Alka and Prussian formations. The dinocyst data indicates that there is a hiatus of about 7 Ma duration between the Lower Blue Earth and Lower Quicksand within the Alka Formation, as indicated by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008b). As Iakovleva (Reference Iakovleva2023) suggested, more boreholes are required, which should be subjected to consistent sedimentological and microfossil sampling to elucidate regional variation.
6. The ages of the amber-bearing horizons
Assuming that the above interpretation of the positions of the FOs and LOs of the zonal and subzonal markers are correct, then the depth range of 59–40.5 m in the P-1 borehole is Selandian in age. The base of the Sambian Formation is at 51.7 m, so the lowest amber-bearing horizon, at the bottom of this formation within zone DP8, is early Selandian, about 61 Ma. Kharin & Eroshenko (Reference Kharin and Eroshenko2017) considered the amber at this horizon was reworked so it is probably older, either from earlier in the Paleocene or even Late Cretaceous.
The Lower Blue Earth Member is the next lowest amber-bearing horizon, with its top corresponding with the top of subzone DE2b. If the base of the Alka Formation does correlate with the Thanetian–Ypresian boundary (56 Ma), then the base of the Lower Blue Earth could correspond with the base of subzone DE2a. Zone DE2, and thus the Lower Blue Earth Member, is early Ypresian, 55.7–55 Ma. The amber in this member was considered to be a primary deposit by Kharin & Eroshenko (Reference Kharin and Eroshenko2017).
If the Bartonian–Priabonian boundary (37.71 Ma) corresponds with the Alka–Prussian Formation boundary then the base of the Upper Wild Earth Member is 37.71 Ma. The top of DE18 occurs within this member and is about 36.3 Ma. So the Upper Wild Earth Member is early Priabonian, 37.71–36 Ma.
The Upper Blue Earth Member is the primary source for Baltic amber and lies immediately above the Upper Wild Earth. Its top corresponds with the top of DE19, which is 35 Ma. Thus, the Upper Blue Earth is mid-Priabonian, 36–35 Ma. The amber was considered to be a primary deposit by Kharin & Eroshenko (Reference Kharin and Eroshenko2017) and a detailed study of the conifer flora preserved in the amber by Sadowski et al. (Reference Sadowski, Schmidt and Kunzmann2022), along with the fresh lemon-yellow colour of pieces with inclusions (Grimaldi & Ross, Reference Grimaldi, Ross, Fraser and Sues2017), indicates that the amber is contemporaneous.
The next three amber-bearing members lying immediately above – Upper Quicksand, White Wall and Green Wall, respectively – correspond to subzone DE20a, which is late Priabonian, 35–34 Ma, though the amber from these members could be reworked.
Given that Baltic amber (succinite) from the Prussian Formation (Upper Wild Earth, Upper Blue Earth, Upper Quicksand and White Wall members) and Palvé Formation (Green Wall Member) is now shown to be considerably younger than the amber from the Lower Blue Earth Member and base of the Sambian Formation, there is a possibility that they are chemically distinct and the latter two may not be succinite. Amber from the lowest two horizons needs to be analysed to find out, and if they do prove to be different that would constrain the age of Baltic amber as defined here to Priabonian only.
7. The likely age of the amber washed up on southeastern Baltic beaches
The maximum water depth in the Gulf of Gdańsk is about 110 m (Chubarenko et al. Reference Chubarenko, Sokolov and Dikii2023), so at what depths could amber be washed out of sediments and be washed up on nearby beaches? The maximum tidal height is less than 10 cm in the Gulf of Gdańsk (see Medvedev et al. (Reference Medvedev, Rabinovich and Kulikov2016), so this has a negligible effect. Borehole P-1 commenced at 2.73 m above sea level (Kasiński et al. Reference Kasiński, Kramarska, Słodkowska, Sivkov and Piwocki2020), with the Upper Blue Earth reached at a depth of 9.3 m; thus, the top of this unit is about 6.6 m below sea level, with its base at about 12.1 m below sea level. The effective wave base in fair-weather conditions on a marginal sea coast is 9.5 m (Flemming Reference Flemming2024), so amber from the Upper Blue Earth and overlying units could be washed out of the sediments by normal wave activity, and much more so during a storm. The effective wave base during a storm is 26 m so amber could also be washed out of the Upper Wild Earth (12.1–15.8 m below sea level) in stormy conditions but not in fair-weather conditions, though given that this unit only contains a little amount of amber then the chance of any of it subsequently being washed up on beaches is remote. The Lower Blue Earth is 29.8–33.2 m below sea level, so it is unlikely that any amber is washed out of this bed during a storm today. However, during the Quaternary, at times when the sea level was lower (see Uścinowicz Reference Uścinowicz2003), amber from the Lower Blue Earth and the base of the Sambian Formation could have been washed out and reworked into Quaternary deposits. Amber from Quaternary deposits that were subsequently eroded could end up back in the sea or be destroyed by weathering, though given the low concentration of amber in the Lower Blue Earth and at the base of the Sambian Formation then the chance of any of this amber subsequently being washed up today is remote, but cannot be ruled out. Therefore, the probable age of Baltic amber (succinite) washed up on beaches is Priabonian. In the Gulf of Gdańsk, although today longshore drift transports sediment south from the Samland Peninsula, then southwest along by the Vistula Spit, Poland (see Ostrowski et al. Reference Ostrowski, Pruszak, Babakov and Chubarenko2012), it is more likely that the amber found today on eastern Polish beaches is reworked and came from Quaternary coastal or nearshore sediments that have undergone successive episodes of erosion, redeposition and longshore transport during the late Quaternary and Holocene.
8. Conclusions
Given that an impure glauconite sample can provide an incorrect date, then the Lutetian age of the Upper Blue Earth provided by Ritzkowski (Reference Ritzkowski1997), which does not correspond to biostratigraphical dating, has to be regarded as unreliable. More absolute dating is required by independent labs using pure glauconite samples to see what results are produced. That said, the first glauconite dates by Kaplan et al. (Reference Kaplan, Grigelis, Strelnikova and Glikman1977) for the Alka, Prussian and Palvé formations fit within the biostratigraphical dating as outlined here.
Biostratigraphy is more reliable and, based on dinoflagellate cyst records, the Sambian Formation is likely to be Selandian to Thanetian (middle to late Paleocene), the Alka Formation is Ypresian to Bartonian (early to mid-Eocene) and the Prussian and Palvé Formations are Priabonian (late Eocene) in age. Regarding the amber-bearing horizons, the base of the Sambian is early Selandian, the Lower Blue Earth is early Ypresian and the other five horizons, including the Upper Blue Earth, are Priabonian in age.
The dinocyst data provides evidence of a hiatus between the Lower Blue Earth and Lower Quicksand within the Alka Formation, as indicated by Aleksandrova & Zaporozhets (Reference Aleksandrova and Zaporozhets2008b). There may also be a short hiatus between the Sambian and Alka formations. Data is lacking from the top 5 m of the Alka Formation so it is uncertain if there is a hiatus between the Alka and Prussian formations.
Given that the top five out of the seven amber-bearing horizons on the Samland Peninsula are Priabonian in age, including the highest concentration in the Upper Blue Earth, and that amber from these five horizons can be washed up on southeastern Baltic beaches, it can be considered that pieces of succinite with unknown provenance are probably Priabonian in age, 37.7–34 Ma. Pieces that are known to have come from the Upper Blue Earth, which are contemporaneous, are mid-Priabonian in age, 36–35 Ma. This dating also applies to any inclusions that are trapped in the amber. Given that the amber is concentrated towards the base of the Upper Blue Earth, then the age of Baltic amber can be considered to be 36 Ma for dating taxa used in phylogenetic trees.
9. Acknowledgements
Thanks go to the organisers of the International Palaeoentomological Conference, Fossil Insects – Bridging Past and Present for inviting A.J.R. to present this research and to the delegates for their encouragement to complete this paper. Thanks also go to Leyla Seyfullah (Universität Wien) and Alexander Schmidt (Georg-August-Universität Göttingen) for constructive comments, and to Christian Neumann (Museum für Naturkunde, Berlin) for information about historical amber collections in his institute.
10. Competing interests
The authors declare none.