Introduction
Phlebotomine sand flies (Diptera: Psychodidae) are small, blood-feeding insects of major medical and veterinary concern, primarily due to their role in transmitting Leishmania spp., as well as some viruses and bacteria (Maroli et al. Reference Maroli, Feliciangeli, Bichaud, Charrel and Gradoni2013; Ready, Reference Ready2013; Akhoundi et al. Reference Akhoundi, Kuhls, Cannet, Votýpka, Marty, Delaunay and Sereno2016; Jancarova et al. Reference Jancarova, Polanska, Thiesson, Arnaud, Stejskalova, Rehbergerova, Kohl, Viginier, Volf and Ratinier2025). Furthermore, the presence of Trypanosoma spp. DNA in sand flies indicates a possible role for these insects as vectors of the protozoan (Srisuton et al. Reference Srisuton, Phumee, Sunantaraporn, Boonserm, Sor-Suwan, Brownell, Pengsakul and Siriyasatien2019; Phumee et al. Reference Phumee, Wacharapluesadee, Petcharat, Tawatsin, Thavara and Siriyasatien2021a, Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024; Buatong et al. Reference Buatong, Dvorak, Thepparat, Thongkhao, Koyadun, Siriyasatien and Pengsakul2022; Sunantaraporn et al. Reference Sunantaraporn, Somwang, Khositharattanakool, Unchanam, Saenchaiban, Wongkhut, Sanum, Pataradool, Boonserm, Depaquit and Siriyasatien2024; Klaiklueng et al. Reference Klaiklueng, Kumlert, Moonmake, Ruang-Areerate, Siriyasatien, Sunantaraporn, Wanachiwanawin, Ruenchit and Wongkamchai2025). The distribution and population dynamics of sand flies are shaped by a range of ecological and climatic factors, including temperature, humidity, vegetation cover, altitude, wind speed, soil type and host availability (Maroli et al. Reference Maroli, Feliciangeli, Bichaud, Charrel and Gradoni2013). These influences contribute to a patchy distribution in tropical and subtropical regions such as Southeast Asia, where certain areas sustain higher sand fly densities (Shahar et al. Reference Shahar, Hassan, Lee and Salmah2011; Loyer et al. Reference Loyer, Depaquit and Gay2016; Vu et al. Reference Vu, Tran, Tran, Tran, Tran, Dang, Nguyen, Vu, Ngo, Nguyen and Hoàng2021; Khositharattanakool et al. Reference Khositharattanakool, Pathawong, Pongsiri, Pengsakul, Ponlawat and Somwang2024; Nguyen et al. Reference Nguyen, Nugraheni, Nguyen, Arnuphapprasert, Pengsakul, Thong, Ampol, Siriyasatien and Kaewthamasorn2024; Vongphayloth et al. Reference Vongphayloth, Randrianambinintsoa, Lakeomany, Phommavanh, Pongsanarm, Vungkyly, Luangamath, Chonephetsarath, Brey and Depaquit2024). Thailand, located within a biodiversity-rich zone, supports diverse phlebotomine fauna, including multiple sand fly genera. These flies inhabit a variety of ecological niches: forests, caves, rural landscapes and peri-domestic settings, reflecting their adaptability to different environments (Polseela et al. Reference Polseela, Apiwathnasorn and Samung2011b; Chusri et al. Reference Chusri, Thammapalo, Chusri, Hortiwakul and Silpapojakul2014; Renaux Torres et al. Reference Renaux Torres, Pellot, Somwang, Khositharattanakool, Vongphayloth, Randrianambinintsoa, Mathieu, Siriyasatien, Gay and Depaquit2023; Phumee et al. Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024; Sunantaraporn et al. Reference Sunantaraporn, Somwang, Khositharattanakool, Unchanam, Saenchaiban, Wongkhut, Sanum, Pataradool, Boonserm, Depaquit and Siriyasatien2024; Klaiklueng et al. Reference Klaiklueng, Kumlert, Moonmake, Ruang-Areerate, Siriyasatien, Sunantaraporn, Wanachiwanawin, Ruenchit and Wongkamchai2025).
While human and animal leishmaniasis cases have risen in the region (Chusri et al. Reference Chusri, Thammapalo, Chusri, Hortiwakul and Silpapojakul2014; Junsiri et al. Reference Junsiri, Wongnarkpet, Chimnoi, Kengradomkij, Kajeerum, Pangjai and Nimsuphan2017; Sarasombath, Reference Sarasombath2017; Rijal et al. Reference Rijal, Sundar, Mondal, Das, Alvar and Boelaert2019; Sriwongpan et al. Reference Sriwongpan, Nedsuwan, Manomat, Charoensakulchai, Lacharojana, Sankwan, Kobpungton, Sriwongpun, Leelayoova, Mungthin and Siripattanapipong2021), entomological data on vector ecology remain limited. Moreover, seasonal distribution data remain sparse, despite their relevance for predicting transmission peaks and informing timely vector control efforts (Maroli et al. Reference Maroli, Feliciangeli, Bichaud, Charrel and Gradoni2013; Alten et al. Reference Alten, Maia, Afonso, Campino, Jiménez, González, Molina, Bañuls, Prudhomme, Vergnes and Toty2016). Traditionally, this disease has been considered endemic to regions such as South America, Africa and the Mediterranean (Alvar et al. Reference Alvar, Vélez, Bern, Herrero, Desjeux, Cano, Jannin and and2012). However, the emergence of autochthonous cases of human leishmaniosis in Thailand suggests that the causative agents are effectively transmitted by native sand fly species (Kanjanopas et al. Reference Kanjanopas, Siripattanapipong, Ninsaeng, Hitakarun, Jitkaew, Kaewtaphaya, Tan-ariya, Mungthin, Charoenwong and Leelayoova2013; Chusri et al. Reference Chusri, Thammapalo, Chusri, Hortiwakul and Silpapojakul2014; Leelayoova et al. Reference Leelayoova, Siripattanapipong, Manomat, Piyaraj, Tan-Ariya, Bualert and Mungthin2017; Sriwongpan et al. Reference Sriwongpan, Nedsuwan, Manomat, Charoensakulchai, Lacharojana, Sankwan, Kobpungton, Sriwongpun, Leelayoova, Mungthin and Siripattanapipong2021; Srivarasat et al. Reference Srivarasat, Brownell, Siriyasatien, Noppakun, Asawanonda, Rattanakorn, Preativatanyou and Kumtornrut2022). Environmental transformation, urban expansion and climate variability are likely contributing to shifts in sand fly habitat and abundance (Ready, Reference Ready2013). Several studies in Thailand have recorded sand fly species in humid and shaded environments such as forests and caves (Chaniotis et al. Reference Chaniotis, Correa, Tesh and Johnson1974; Polseela et al. Reference Polseela, Apiwathnasorn and Samung2011b; Apiwathnasorn et al. Reference Apiwathnasorn, Samung, Prummongkol, Phayakaphon and Panasopolkul2011; Phumee et al. Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024), while others have highlighted their presence in villages, indicating potential for human-vector contact in peri-domestic areas (Kanjanopas et al. Reference Kanjanopas, Siripattanapipong, Ninsaeng, Hitakarun, Jitkaew, Kaewtaphaya, Tan-ariya, Mungthin, Charoenwong and Leelayoova2013; Chusri et al. Reference Chusri, Thammapalo, Chusri, Hortiwakul and Silpapojakul2014). Furthermore, asymptomatic infections are frequently detected in HIV-positive individuals in southern provinces such as Trang, indicating the need for targeted surveillance in high-risk populations (Manomat et al. 2017; Bualert et al. Reference Bualert, Ruang-Areerate, Mungthin, Leelayoova, Siripattanapipong, Naaglor, Hongsimakul, Sroythong, Rattanalertpaiboon, Tulpeng and Piyaraj2024).
Several studies have detected Leishmania DNA in Sergentomyia species, raising questions about their potential involvement despite unconfirmed vector competence for Leishmania species affecting mammals (Chusri et al. Reference Chusri, Thammapalo, Chusri, Hortiwakul and Silpapojakul2014; Siripattanapipong et al. Reference Siripattanapipong, Leelayoova, Ninsaeng and Mungthin2018; Sriwongpan et al. Reference Sriwongpan, Nedsuwan, Manomat, Charoensakulchai, Lacharojana, Sankwan, Kobpungton, Sriwongpun, Leelayoova, Mungthin and Siripattanapipong2021; Phumee et al. Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024). Importantly, the presence of Phlebotomus species, known vectors of Leishmania spp., in Thailand’s endemic regions, further underscores the importance of nationwide entomological surveys to clarify species distribution and infection status (Polseela et al. Reference Polseela, Apiwathnasorn and Samung2007; Apiwathnasorn et al. Reference Apiwathnasorn, Samung, Prummongkol, Phayakaphon and Panasopolkul2011; Phumee et al. Reference Phumee, Wacharapluesadee, Petcharat and Siriyasatien2021b). Also, Trypanosoma species in Thailand are gaining attention following their detection in sand flies and possible circulation in wildlife reservoirs. Although human-infective trypanosomes like T. brucei and T. cruzi have not been detected, recent studies have identified Trypanosoma spp., T. noyesi and amphibian-associated Trypanosoma lineages in sand flies relating to their blood meal preference, indicating a broader distribution than previously recognized (Buatong et al. Reference Buatong, Dvorak, Thepparat, Thongkhao, Koyadun, Siriyasatien and Pengsakul2022; Phumee et al. Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024; Sunantaraporn et al. Reference Sunantaraporn, Somwang, Khositharattanakool, Unchanam, Saenchaiban, Wongkhut, Sanum, Pataradool, Boonserm, Depaquit and Siriyasatien2024). These findings suggest sylvatic cycles in wildlife, which could potentially influence the local ecosystem health and pose zoonotic spillover risks. However, the direct impact of Trypanosoma on public health or livestock in Thailand remains unclear due to limited evidence of pathogenicity or transmission to humans or domestic animals. Ongoing monitoring and research are essential to understand the epidemiological significance of these parasites in Thailand (Preativatanyou et al. Reference Preativatanyou, Chinwirunsirisup, Phumee, Khositharattanakool, Sunantaraporn, Depaquit and Siriyasatien2023).
Ongoing nationwide and molecularly driven studies published between 2021 and 2025 reveal that phlebotomine sand flies in Thailand are widely distributed, genetically diverse and often harbour multiple parasites. In 2020, a study across 6 provinces identified at least 13 species of sand flies, suggesting hidden diversity that may compromise accurate surveillance (Phuphisut et al. Reference Phuphisut, Nitatsukprasert, Pathawong, Jaichapor, Pongsiri, Adisakwattana and Ponlawat2021). In addition, a study spanning eight provinces recorded 24 species, including Phlebotomus argentipes, a known vector of Leishmania, highlighting a wide distribution of potential disease vectors (Polseela et al. Reference Polseela, Vitta, Thammeepak and Thanwisai2024). Besides this, a survey in southern Thailand detected Leishmania and Trypanosoma DNA in Sergentomyia khawi, with co-infections suggesting complex transmission dynamics (Phumee et al. Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024). The most recent integrative taxonomic study uncovered two putatively novel phlebotomine species, emphasizing that undiscovered species may harbour unknown vector capacity (Soomro et al. Reference Soomro, Tuangpermsub, Ngamprasertwong and Kaewthamasorn2025). Another work also found Trypanosoma spp. DNA in sand flies, even in regions where Leishmania spp. have not been recorded (Klaiklueng et al. Reference Klaiklueng, Kumlert, Moonmake, Ruang-Areerate, Siriyasatien, Sunantaraporn, Wanachiwanawin, Ruenchit and Wongkamchai2025). In addition to this, sand flies are also known to transmit other pathogens such as phleboviruses (e.g. Toscana virus and sand fly fever Naples virus), which can lead to febrile illnesses and neurological complications (Maroli et al. Reference Maroli, Feliciangeli, Bichaud, Charrel and Gradoni2013; Depaquit et al. Reference Depaquit, Pesson, Augot, Hamilton, Lawyer and Léger2016). Occurrence of viral RNA in sand flies in Thailand has been reported recently (Phumee et al. Reference Phumee, Wacharapluesadee, Petcharat, Tawatsin, Thavara and Siriyasatien2021a, Reference Phumee, Wacharapluesadee, Petcharat and Siriyasatien2021b), as well as bacterial DNA (Sunantaraporn et al. Reference Sunantaraporn, Somwang, Khositharattanakool, Unchanam, Saenchaiban, Wongkhut, Sanum, Pataradool, Boonserm, Depaquit and Siriyasatien2024).
Given the increasing globalization, environmental changes and emerging infectious disease threats, continued surveillance of phlebotomine sand fly populations in Thailand is essential. This scoping review, therefore, aims to (1) consolidate the current knowledge on the abundance and distribution of phlebotomine sand fly species and (2) investigate their associated pathogens in Thailand, with the goal of identifying potential vectors, regional hotspots and research gaps relevant to public health surveillance and future research directions. While previous literature has addressed sand fly biodiversity and pathogen presence across the Greater Mekong Subregion (Hustedt et al. Reference Hustedt, Prasetyo, Fiorenzano, von Fricken and Hertz2022), this study serves as an initial investigation conducted in Thailand and will later be expanded to include other countries in Southeast Asia, particularly within the Greater Mekong Subregion. In this context, this review compiles fragmented data from diverse provinces, offering a comprehensive overview of sand fly species and their roles in pathogen transmission in Thailand.
Materials and methods
Search strategy and selection criteria
The search was performed in the PubMed, Scopus and Web of Science databases to identify articles published without restricting the time range that include the following combinations of terms in the title, abstract, or keywords: (‘prevalence’ OR ‘epidemiology’ OR ‘incidence’ OR ‘presence’ OR ‘abundant’ OR ‘occurrence’) AND (‘Thailand’) AND (‘phlebotomus’ OR ‘sand fly’ OR ‘sand fly’ OR ‘phlebotomine sand fly’ OR ‘phlebotominae’), following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting checklist for Scoping Reviews (Supplementary File 1) as described by Page et al. (Reference Page, McKenzie, Bossuyt, Boutron, Hoffmann, Mulrow, Shamseer, Tetzlaff, Akl, Brennan and Chou2021). These three databases were chosen based on systematic review recommendations and the following criteria: subject relevance, document availability, support for Boolean operators and parentheses and the ability to bulk download. In addition, the reference lists of selected studies were also reviewed to search for additional relevant publications. The collection of these documents was completed in June 2025, without any time limitation. The review protocol was registered at the OSF platform (https://osf.io/8rswz/).
Following the removal of duplicates, a screening was conducted at different levels (by title, abstract and full text) to determine whether they met the inclusion criteria: (1) epidemiological study; (2) research done in Thailand; (3) research originated on survey of sand fly; (4) international publication; (5) publications in English; (6) original research paper; and (7) available online. Two independent researchers carried out this selection process (Figure 1). Throughout the selection process, all data were verified through cross-checking, and any disagreements were resolved by a senior researcher.
PRISMA flow chart of records selection process.

Quality assessment
Two independent authors assessed the risk of bias (RoB) in all the articles that met the inclusion criteria. Since there is no established RoB tool specifically designed for studies on vector distribution, the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Analytical Cross-Sectional Studies was used to assess study quality (Moola et al. Reference Moola, Munn, Tufanaru, Aromataris, Sears, Sfetcu, Currie, Qureshi, Mattis, Lisy, Mu, Aromataris and Munn2020), as the included articles were predominantly cross-sectional in nature. To ensure relevance to entomological fieldwork and enhance the evaluation of methodological rigour, 2 additional domains were incorporated: (1) whether the study adequately processed sand fly specimens, such as through appropriate identification and preservation methods; and (2) whether a standardized trapping method was used for sample collection. These domains were added to address specific aspects critical to the validity and comparability of vector distribution studies, which are not fully captured by existing generic tools. Any disagreements were resolved by consensus, and when information was insufficient, the corresponding author was contacted. The recruitment of data was performed in an Excel sheet with the ‘Yes,’ ‘No,’ or ‘Unclear’ statements. Articles were classified into low (no ‘No’ or ‘Unclear’ statements), medium (1 ‘No’ or ‘Unclear’ statement) or high (2 or more ‘No’ or ‘Unclear’ statements) RoB. Those articles with a high RoB were excluded from this scoping review.
Data extraction and analysis
A qualitative approach was used to accommodate the varied publication styles, research methods and research purposes. Briefly, relevant information was initially extracted from the full texts of eligible studies and then verified for accuracy. Subsequently, a second reviewer evaluated the extracted data for clarity and completeness. Any disagreements were resolved through discussion until consensus was achieved at every step of selection. The extracted data focused on: (1) bibliometrics; (2) material and methods; and (3) results (Table 1). Studies’ sampling periods were categorized into three time periods: before 2010, 2011–2020 and 2021–2025. Data were collected using a standardized Microsoft Excel sheet (Version 2017). Metrics and graphics for the descriptive analysis were performed by Microsoft Excel. The distribution map of study provinces for sand fly studies involved in selected publications within the three different time zones was created using QGIS (version 3.44). The interaction matrix (Sankey diagram) between sand fly species and detected pathogens was performed using RAWGraphs 2.0 (https://app.rawgraphs.io/).
Categories, variables and factor labels studied from the 33 articles

Results
Selection of articles and quality assessment
A total of 113 articles were identified in the different scientific databases and the review of the reference lists (Figure 1). After removing duplicates and applying the inclusion criteria, 33 articles were selected for quality assessment. The RoB of the 33 included studies was assessed based on the identified domains (Figure 2). Most studies fulfilled the majority of the JBI Critical Appraisal Checklist criteria. All studies clearly defined their inclusion criteria and provided sufficient detail on study subjects and sampling settings. Exposure measurements were conducted using valid and reliable techniques, and standardized criteria were consistently applied for sample collection in line with the study objectives. However, 7 articles (Kongkaew et al. Reference Kongkaew, Siriarayaporn, Leelayoova, Supparatpinyo, Areechokchai, Duang-Ngern, Chanachai, Sukmee, Samung and Sridurongkathum2007; Apiwathnasorn et al. Reference Apiwathnasorn, Samung, Prummongkol, Phayakaphon and Panasopolkul2011; Sukra et al. Reference Sukra, Kanjanopas, Amsakul, Rittaton, Mungthin and Leelayoova2013; Chusri et al. Reference Chusri, Thammapalo, Chusri, Hortiwakul and Silpapojakul2014; Polseela et al. Reference Polseela, Jaturas, Thanwisai, Sing and Wilson2015; Somwang et al. Reference Somwang, Khositharattanakool, Pathawong, Pongsiri, Davidson and Ponlawat2021; Polseela et al. Reference Polseela, Vitta, Thammeepak and Thanwisai2024) did not describe in detail how to address the confounding factors, particularly in relation to distribution and abundance, occurrence of anthropophilic behaviour, occurrence of known vectors for Leishmania, remarkably low species, and either abundance or density of cavernicolous species. In contrast, the remaining studies acknowledged relevant confounding variables such as sampling season, habitat characteristics, species distribution, trap type and discussed their potential impact, along with strategies to minimize bias. All 33 studies reported outcome measurements adequately, conducting standardized techniques for sand fly identification to ensure reliable results and applying appropriate data analysis methods. In addition, standardized trapping methods were used consistently across the sampling periods. Notably, one study (Somwang et al. Reference Somwang, Khositharattanakool, Pathawong, Pongsiri, Davidson and Ponlawat2021) tested different trapping approaches to assess whether trap placement or the use of CO₂ influenced the number of sand flies collected. Overall, the studies demonstrated good methodological quality and consistent practices in assessing sand fly distribution and abundance, supporting the reliability of the current evidence base. So, the 33 articles finally included in this scoping review are listed in Supplementary file 2.
Risk of bias for 33 included studies for sand flies distribution and abundance with a complete description of domains and signalling questions using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Analytical Cross-Sectional Studies.

Descriptive data
Regarding bibliometrics, overall, 42.4% (14/33) of articles were published in the first quartile, while 12.1% (4/33) and 12.1% (4/33) appeared in the second and third quartile, respectively, according to the Journal Citation Report (JCR). In addition, 30.3% (10/33) were published in the fourth quartile, and only 3.1% (1/33) were not published in a JCR-indexed journal (Figure 3A). The analysis of the ‘Materials and Methods’ section from the selected articles revealed a wide variety of experimental designs. The study duration was variable: 21.2% (7/33) of articles lasted ≤ 1 month, 24.2% (8/33) between 1 and 3 months, only 6.1% (2/33) of studies lasted between 3 and 6 months, 27.3% (9/33) lasted between 6 months and 1 year, and 18.2% (6/33) lasted more than 1 year. Only 1 article did not mention the exact sampling period. Not all publications examined the temporal distribution of sand flies; available data on their temporal distribution were collected only in 21.2% (7/33) of the articles.
(A) Journal quartile rank of the 33 included studies. (B) Diagnostic techniques used to identify sand flies in the 33 included studies.

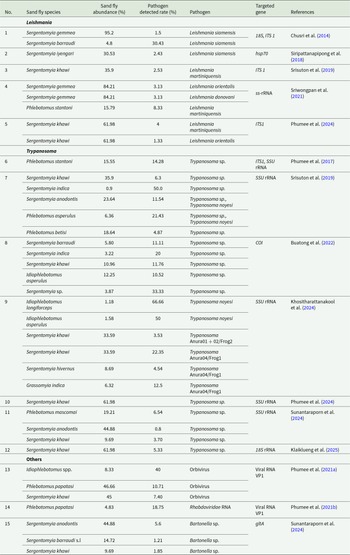
Regarding the collection method, most of the articles (90.9%; 30/33) used CDC (Centers for Disease Control and Prevention) light traps, and only the remaining 9.0% (3/33) of the articles used several methods (one used light traps, Disney traps, human-bait collectors and cow-bait net; other used CDC light traps and aspirators; and the last one used CDC light traps, CDC Updraft Blacklight trap, Encephalitis Vector Survey trap and Leitner-Kirk trap). The sand fly traps were placed in different sites: caves (60.6%; 20/33), urban/peri-urban areas (36.4%; 12/33), rural areas (21.2%; 7/33), forest/natural areas (12.1%; 4/33) and islands (6.1%; 2/33). About 27.3% of the articles (9/33) used several of these sampling sites. Species identification was primarily based on taxonomic keys. Specifically, 72.7% (24/33) of the articles used morphological characteristics, 21.2% (7/33) used both morphological characteristics and molecular approaches (cytb gene amplification 18.2%, 6/33; DNA barcoding 3.0%, 1/33), and the remaining 6.1% (2/33) used only molecular techniques (cytb gene amplification 3.0%, 1/33; DNA barcoding 3.0%, 1/33) for species identification (Figure 3B). In addition, only 9.1% (3/33) of the articles reported for host blood meal analysis. On the other hand, 36.4% (12/33) of the articles analysed the presence of different pathogens in the sand flies. Concretely, they used molecular methods to detect Leishmania spp. (41.7%; 5/12), Trypanosoma spp. (58.3%; 7/12), Bartonella spp. (8.3%; 1/12), Orbivirus (8.3%; 1/12) and Rhabdovirus (8.3%; 1/12). Three out of these 12 articles analysed several pathogens (Leishmania spp. and Trypanosoma spp. and Trypanosoma spp. and Bartonella spp., respectively). The targeted genes employed for each molecular analysis are summarized in Table 2.
Pathogens identified from individual sand fly species in the 12 studies that analysed their presence

Spatiotemporal coverage of sand fly studies
The distribution map of study provinces where sand flies were collected in the 33 selected publications within the three different time periods (before 2010, between 2011 and 2020 and between 2021 and 2025) is presented in Figure 4. During the period before 2010, sand fly surveys were conducted in 11 provinces, primarily in the south (4 provinces: Songkhla, Surat Thani, Trang and Nakhon Si Thammarat), followed by central (3 provinces: Bangkok, Ratchaburi and Saraburi), north (2 provinces: Nan and Phitsanulok) and one province each in the west (Kanchanaburi) and the Andaman coast (Phang-nga) (Figure 4A). Between 2011 and 2020, the number of surveyed provinces increased to 20, with notable expansion in the northern region (5 provinces: Chiang Rai, Chiang Mai, Lampang, Phitsanulok and Tak), northeastern region (Si Sa Ket), eastern region (4 provinces: Sa Kaeo, Chanthaburi, Prachinburi and Rayong), southern region (6 provinces: Chumphon, Songkhla, Trang, Nakhon Si Thammarat, Yala and Phatthalung), central (Nonthaburi, Kamphaeng Phet) and western region (2 provinces: Kanchanaburi and Ratchaburi) (Figure 4B). From 2021 to 2025, surveys were conducted in 17 provinces, including the south (8 provinces: Songkhla, Chumphon, Ranong, Surat Thani, Nakhon Si Thammarat, Phatthalung, Narathiwat and Trang), north (3 provinces: Chiang Rai, Phayao and Uthai Thani), northeast (2 provinces: Sakhon Nakhon and Bueng Kan), central (2 provinces: Lopburi and Saraburi), western (Kanchanaburi) and eastern (Chachoengsao) (Figure 4C). Several provinces, such as Songkhla, Trang and Nakhon Si Thammarat, were surveyed repeatedly, reflecting sustained research interest. However, certain parts of central and western Thailand remained underrepresented.
Distribution maps of provinces sampled in the included studies published within three different periods of time: (A) before 2010; (B) between 2011 and 2020; and (C) between 2021 – 2025.

Recorded sand fly species and their abundance
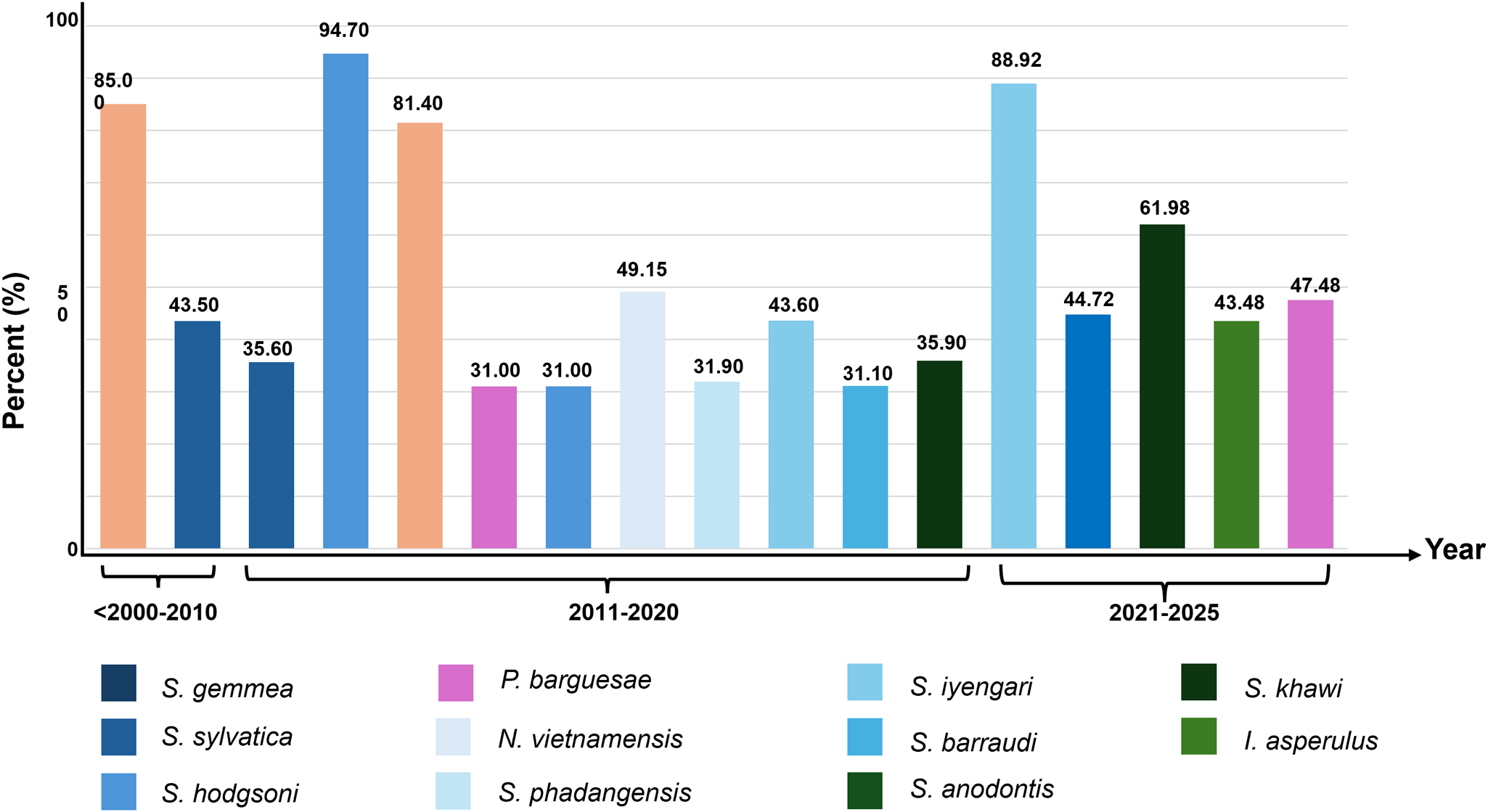
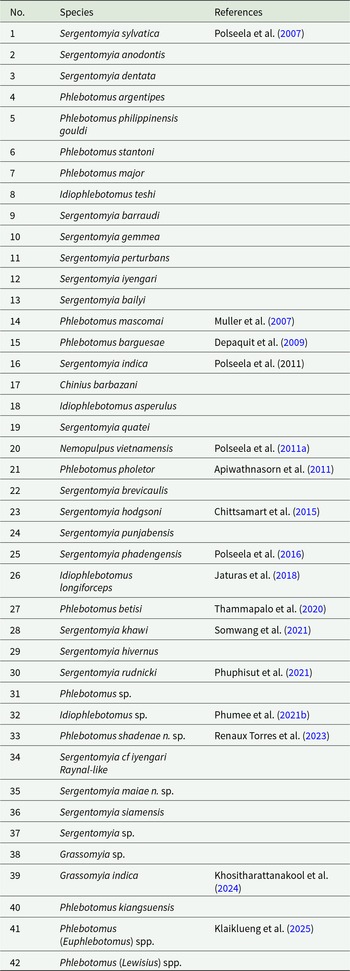
All reviewed publications included a total of 6 genera of sand flies (Sargento, Phlebotomus, Idiophlebotomus, Grassomyia, Chinius and Neopulpus), and 42 species. The first descriptions of each of these 42 species are chronologically ordered in Table 3. Briefly, there are 21 and 13 species of the genera Sergentomyia and Phlebotomus, respectively. Other identified species included Idiophlebotomus teshi, Idiophlebotomus asperulus, Idiophlebotomus longiforceps, Idiophlebotomus sp., Grassomyia sp., Grassomyia indica, Chinius barbazani and Neopulpus vietnamensis (Table 3). About 21 out of 33 articles (63.6%) were surveyed for the abundance rate of sand fly species. According to these articles, the sand flies’ abundances were grouped regarding three periods of time. Briefly, in publications between 2007 and 2010, two species, Sergentomyia gemmea (85%) and Sergentomyia sylvatica (43.5%), were abundantly found. From 2011 to 2020, the number of abundant species increased to 10, including Sergentomyia hodgsoni (94.7%), S. gemmea (81.4%), N. vietnamensis 49.2%, Sergentomyia iyengari (43.6%), Sergentomyia khawi (35.9%), S. sylvatica (35.6%), Sergentomyia phadangensis (31.9%), Sergentomyia barraudi (31.1%), S. hodgsoni (31%) and Phlebotomus barguesae (31%). Most recently, between 2021 and 2025, 5 species S. iyengari 88.9%, S. khawi 60.3%, P. barguesae 47.5%, Sergentomyia anodontis 44.7% and I. asperulus 43.5% were predominantly recorded. The abundant distribution rates from which each species with an over 30% abundance rate in the three different periods of selected publications were presented in Figure 5. From the articles that analysed the temporal distribution of sand flies, 71.4% (5/7) articles reported that sand flies were most abundant during the early rainy season (June and July), while the remaining 28.6% (2/7) detected a peak in the dry season (March–May).
The abundant distribution rate from which each species with an over 30% abundance rate in the included studies published within the three different periods of time.

Chronological list of the first descriptions of sand fly species detected in the included articles

Sand fly associated pathogens
The interaction matrix between sand fly species and detected pathogens is shown in Figure 6 using a Sankey diagram. The detailed distribution of pathogens that were detected in sand fly species across provinces is shown in Figure 7.
The interaction matrix (Sankey diagram) between sand fly species and detected pathogen. The number of occurrences were shown in brackets.

Map of Thailand showing provinces where sand flies were collected and the corresponding pathogens detected. Each province is marked with the identified pathogen(s) found in sand flies based on the included studies.

Sand fly species and Leishmania spp. detection
As previously said, 12 out of 33 included articles (36.4%) analysed the presence of pathogens from the collected sand flies. The detailed information on the abundance of sand fly species (calculated based on collected number of sand flies collected in each study), the detected pathogen rate (calculated based on collected number of sand flies and the number of pathogens detected in each), and their associated pathogen are listed in Table 2. As a result, Leishmania species DNA was detected from a total of 5 sand fly species in Thailand. Briefly, S. gemmea, S. barraudi, S. iyengari, Phlebotomus stantoni and S. khawi were found to be infected with Leishmania siamensis, Leishmania orientalis, Leishmania donovani and Leishmania martiniquensis. Interestingly, S. gemmea has been documented for presenting three Leishmania species DNA (L. siamensis, L. orientalis and L. donovani) and S. khawi for two Leishmania species DNA (L. orientalis and L. martiniquensis). Moreover, S. iyengari, P. stantoni and S. barraudi harboured DNA of a single Leishmania species. Among the positive records, L. siamensis was the most frequently detected species. All positive sand fly specimens were collected using CDC light traps. In terms of regional distribution, Leishmania spp. DNA-positive sand flies were reported from both southern and northern Thailand, specifically, two southern provinces (Trang and Songkhla) and one northern province (Chiang Rai). The majority of studies performed polymerase chain reaction (PCR) targeting the ITS or small subunit rRNA (ss-rRNA) genes for Leishmania pathogen detection (Table 2).
Sand fly species and Trypanosoma detection
In Thailand, Trypanosoma spp. DNA were detected in 13 sand fly species reported across the 33 included studies. These comprised S. khawi, Sergentomyia hivernus, S. anodontis, S. barraudi, S. indica, Sergentomyia sp., P. stantoni, Phlebotomus mascomai, I. asperulus, I. longiforceps, Phlebotomus asperulus, Phlebotomus betisi and G. indica, which were found to harbour Trypanosoma spp., Trypanosoma noyesi, Trypanosoma Anura01 + 02/Frog2 and Trypanosoma Anura04/Frog1. Notably, S. khawi tested positive for three Trypanosoma species (Trypanosoma sp., Trypanosoma Anura01 + 02/Frog2, Trypanosoma Anura04/Frog1) while the remaining species harboured a single Trypanosoma species. Among the detections, Trypanosoma sp. was the most frequently reported. All Trypanosoma-positive sand flies were collected using CDC light traps in the northern, eastern and southern regions of Thailand across 10 provinces: Chiang Rai, Chanthaburi, Sa Kaeo, Songkhla, Lampang, Narathiwat, Satun, Phatthalung, Chumphon and Phang Nga. Most of the studies employed PCR targeting the SSU rRNA gene of Trypanosoma species (Table 2).
Other detected pathogens
Regarding bacteria, the only pathogen evaluated across the reviewed studies was Bartonella spp. Detection of Bartonella spp. DNA was reported in three Sergentomyia species: S. barraudi s.l., S. anodontis and S. khawi. These positive samples were collected from sand flies captured by CDC light traps in both northern and southern Thailand, specifically in Lampang, Chiang Rai and Songkhla provinces. Molecular detection was performed using PCR targeting the gltA gene, a citrate synthase gene commonly used for Bartonella spp. identification (Table 2). No other bacteria were reported in the surveyed sand fly populations. Regarding viruses, Phlebotomus papatasi collected from Trang province tested positive for Rhabdoviridae RNA, and Orbivirus RNA was detected in Idiophlebotomus spp., P. papatasi and S. khawi also from Trang province. Both studies used CDC light traps to collect the sand flies.
Host blood meal analysis
From the three studies that analysed the host blood meal, two studies identified blood from humans in S. khawi, S. iyengari, S. barraudi, S. hodgsoni, S. sylvatica, Sergentomyia hamidi, S. indica, P. argentipes, P. stantoni, I. asperulus and Sergentomyia sp., one in frogs (Micryletta inornate and Microhyla fissipes) inside S. khawi, and one from insects inside S. gemmea.
Discussion
This scoping review synthesized 18 years of publications (January 2007–June 2025) on sand fly species in Thailand, encompassing the vector trends in abundance, geographical distribution and associated pathogens detection. No new publications were found after June 2025 up to the submission date. This review incorporated findings from multiple regions, including temples, caves, forests, islands and also both husbandry and peridomestic areas across the country, highlighting variation in sand fly diversity and temporal dynamics. By combining qualitative synthesis with supporting quantitative data, our work addresses critical knowledge gaps and provides a clearer picture of sand fly ecology and vector–pathogen relationships in Thailand. Analysis of the 33 collected documents revealed significant data gaps across many regions of Thailand, preventing a comprehensive assessment of sand fly presence in those areas. Interestingly, a total of 13 publications appeared within 2021–2025 (Figure 2), with integrated diagnostic techniques suggesting an increased interest in sand flies and their role in pathogen transmission.
Regarding publication in JCR-indexed journals, a notable dominance of first quartile publications (42.4%) suggests strong academic recognition of sand fly research in Thailand, compared to the far fewer studies published in either lower-tier journals or non-JCR-indexed journals.
According to this comprehensive review, Thailand exhibits considerable sand fly diversity, a total of six genera: Sergentomyia, Phlebotomus, Idiophlebotomus, Chinius, Grassomyia and Neopulpus, comprising 42 sand fly species that were identified across Thailand. This level of diversity seems greater than that reported in other nearby countries in the Greater Mekong Subregion. Briefly, Vietnam, Malaysia and Cambodia have documented fewer species, which may result from actual biological variations or differences in sampling techniques (Depaquit et al. Reference Depaquit, Muller and Léger2009; Shahar et al. Reference Shahar, Hassan, Lee and Salmah2011; Depaquit et al. Reference Depaquit, Pesson, Augot, Hamilton, Lawyer and Léger2016; Hustedt et al. Reference Hustedt, Prasetyo, Fiorenzano, von Fricken and Hertz2022; Nguyen et al. Reference Nguyen, Nugraheni, Nguyen, Arnuphapprasert, Pengsakul, Thong, Ampol, Siriyasatien and Kaewthamasorn2024). In contrast, a recent study found 20 different species belonging to five genera (Chinius spp., Idiophlebotomus spp., Phlebotomus spp., Sergentomyia spp. and Grassomyia spp.) in Laos (Vongphayloth et al. Reference Vongphayloth, Randrianambinintsoa, Lakeomany, Phommavanh, Pongsanarm, Vungkyly, Luangamath, Chonephetsarath, Brey and Depaquit2024). The genus Sergentomyia dominates in Thailand, as similar as other Southeast Asian countries, but some species, such as S. khawi, S. gemmea and S. barraudi have shown a wide ecological distribution and might represent species complexes. For instance, molecular data suggest that morphologically similar species, such as S. gemmea and S. iynegari, may include cryptic taxa, highlighting the need for genetic confirmation (Phuphisut et al. Reference Phuphisut, Nitatsukprasert, Pathawong, Jaichapor, Pongsiri, Adisakwattana and Ponlawat2021). Higher species diversity may reflect a broad range of ecological adaptability and host preferences, enhancing the likelihood of vector-pathogen interactions. This complexity may hinder vector control efforts, as a single approach may not effectively target all competent vectors. Moreover, diverse sand fly populations could support overlapping transmission cycles of zoonotic pathogens, heightening the risk of disease emergence in both rural and peri-urban areas (Ready, Reference Ready2013).
Notably, Sergentomyia spp. and Phlebotomus spp. were the most represented, with 21 and 13 species, respectively. Comparing species distributions is challenging due to regional differences in previous studies, so data were analysed by species and grouped into three main categories: low abundance, abundant but understudied and abundant species. Across studies till 2025, the most abundant sand fly species were S. gemmea, S. khawi, S. iyengari and S. hodgsoni. Species identification was primarily done using taxonomic keys, despite a few studies using an integrative approach. Phuphisut et al. (Reference Phuphisut, Nitatsukprasert, Pathawong, Jaichapor, Pongsiri, Adisakwattana and Ponlawat2021) highlighted that combining morphological taxonomy with DNA barcoding enhances species resolution. Studies using both approaches have achieved a higher level of accuracy in identification, especially in cryptic sand fly complexes (Muller et al. Reference Muller, Depaquit and Léger2007; Renaux Torres et al. Reference Renaux Torres, Pellot, Somwang, Khositharattanakool, Vongphayloth, Randrianambinintsoa, Mathieu, Siriyasatien, Gay and Depaquit2023). On seasonality, only 21.21% (7/33) of studies reported seasonal trends, with sand fly peaks in the early rainy months (June–July) or at the end of the dry season (March–May). Similar temporal patterns have been previously observed in Laos, Vietnam and the Mekong Subregion, where sand fly populations tend to peak during the early rainy season, when humidity and temperature create favourable breeding conditions (Loyer et al. Reference Loyer, Depaquit and Gay2016; Hustedt et al. Reference Hustedt, Prasetyo, Fiorenzano, von Fricken and Hertz2022; Nguyen et al. Reference Nguyen, Nugraheni, Nguyen, Arnuphapprasert, Pengsakul, Thong, Ampol, Siriyasatien and Kaewthamasorn2024; Vongphayloth et al. Reference Vongphayloth, Randrianambinintsoa, Lakeomany, Phommavanh, Pongsanarm, Vungkyly, Luangamath, Chonephetsarath, Brey and Depaquit2024). Temporal analysis showed a shift in species patterns: S. gemmea and S. sylvatica were dominant in 2007–2010; in 2011–2020, there was an increased abundance of S. gemmea, S. hodgsoni, S. khawi and N. vietnamensis, while S. iyengari, S. khawi, S. anodontis, P. barguesae and I. asperulus emerged in 2021–2025. Due to inconsistent reporting about seasonal patterns and the frequent omission of phenological data, the presence of multiple dominant species may be underscored, leading to the need for standardized, long-term studies.
Within the genus Sergentomyia, over 60% of the abundances of S. gemmea, S. iyengari and S. khawi were recorded throughout this systematic review. Sergentomyia gemmea was documented in southern Thailand as the most prevalent sand fly species in leishmaniasis-affected areas (Polseela et al. Reference Polseela, Apiwathnasorn and Samung2007; Sriwongpan et al. Reference Sriwongpan, Nedsuwan, Manomat, Charoensakulchai, Lacharojana, Sankwan, Kobpungton, Sriwongpun, Leelayoova, Mungthin and Siripattanapipong2021; Phumee et al. Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024). This abundant species was collected using CDC light traps in peridomestic and cave environments, which suggests that they exhibit high levels of anthropophilia and aggressive behaviour toward humans (Sukra et al. Reference Sukra, Kanjanopas, Amsakul, Rittaton, Mungthin and Leelayoova2013). Sergentomyia gemmea is also known to be a potential vector for Leishmania orientalis (formerly referred to as L. siamensis) within the subgenus Mundinia in Thailand (Leelayoova et al. Reference Leelayoova, Siripattanapipong, Manomat, Piyaraj, Tan-Ariya, Bualert and Mungthin2017), with PCR-based detection of Leishmania spp. DNA alongside human blood meals supporting its potential vector status (Srisuton et al. Reference Srisuton, Phumee, Sunantaraporn, Boonserm, Sor-Suwan, Brownell, Pengsakul and Siriyasatien2019). Its large size, consistent abundance and ease of sampling make S. gemmea one of the most intensively studied sand fly species. Another abundant species, S. iyengari, was recorded at lower frequencies in early surveys but gained prominence in later collections. In a multi-province survey using both morphology and DNA barcoding, S. iyengari emerged as the most common identified species (Phuphisut et al. Reference Phuphisut, Nitatsukprasert, Pathawong, Jaichapor, Pongsiri, Adisakwattana and Ponlawat2021). Notably, the blood meal analysis of S. iyengari from Trang province revealed both human and reptile blood meal, indicating its diverse host-feeding behaviour (Siripattanapipong et al. Reference Siripattanapipong, Leelayoova, Ninsaeng and Mungthin2018). However, morphological identification can lead to misidentification in previous studies, as shown by species initially classified as S. iyengari that were later confirmed to be S. gemmea via DNA barcoding (Phuphisut et al. Reference Phuphisut, Nitatsukprasert, Pathawong, Jaichapor, Pongsiri, Adisakwattana and Ponlawat2021). In fact, over the previous decades, S. khawi was historically understudied or misidentified (Kongkaew et al. Reference Kongkaew, Siriarayaporn, Leelayoova, Supparatpinyo, Areechokchai, Duang-Ngern, Chanachai, Sukmee, Samung and Sridurongkathum2007; Polseela et al. Reference Polseela, Apiwathnasorn and Samung2007). On the other hand, the recent detection of S. khawi in diverse habitats, including caves, forests, surrounding residential areas, enclosures, animal pens, barns, rubber plantations, palm plantations and rural areas, highlights their ecological versatility and potential role of pathogen transmission (Sriwongpan et al. Reference Sriwongpan, Nedsuwan, Manomat, Charoensakulchai, Lacharojana, Sankwan, Kobpungton, Sriwongpun, Leelayoova, Mungthin and Siripattanapipong2021; Phumee et al. Reference Phumee, Wacharapluesadee, Petcharat, Tawatsin, Thavara and Siriyasatien2021a; Buatong et al. Reference Buatong, Dvorak, Thepparat, Thongkhao, Koyadun, Siriyasatien and Pengsakul2022).
Although P. argentipes and P. papatasi, known vectors of some Leishmania species, were identified with low abundance in certain provinces (Saraburi, Satun, Phitsanulok, Phang Nga, Surat Thani, Trang and Phayao), P. argentipes was reported more frequently than P. papatasi. Interestingly, these two species were documented in studies conducted between 2007 and 2024, but have not yet been recorded in recent publications of 2025. However, the absence of these sand fly species in recent studies should not be interpreted as species disappearance, but rather as a result of limited or non-targeted surveys performed recently. These anthropophilic species, known vectors of L. donovani and Leishmania major, had been identified in patients from Trang province (southern Thailand), but were rarely detected lately (Suankratay et al. Reference Suankratay, Suwanpimolkul, Wilde and Siriyasatien2010; Charoensakulchai et al. Reference Charoensakulchai, Bualert, Manomat, Mungthin, Leelayoova, Tan-Ariya, Siripattanapipong, Naaglor and Piyaraj2020). The potential for local transmission of Leishmania spp. is created by the coexistence of infected individuals and the presence of known vectors, creating a well-established scenario in endemic regions (Thammapalo et al. Reference Thammapalo, Pawestri, Kolaeh, Boondej, Benarlee, Apiwathnasorn and Kumlert2020). In addition to environmental shifts, human migration and population movements can introduce infected individuals into areas with established vector populations, further increasing autochthonous transmission risk (Grifferty et al. Reference Grifferty, Shirley, McGloin, Kahn, Orriols and Wamai2021).
Based on reported pathogen occurrences, S. khawi and S. gemmea were found to be infected with multiple Leishmania species, while S. khawi carried three Trypanosoma species including amphibian-associated types. These findings suggest higher possibilities of S. khawi and S. gemmea in pathogen transmission, though vector competence has yet to be confirmed. Regionally, Leishmania spp. DNA-positive sand flies were detected in both southern and northern Thailand, particularly in Trang and Songkhla provinces in the south, and Chiang Rai province in the north. These provinces are recognized as endemic areas for leishmaniasis, with multiple lines of evidence supporting local transmission (Renaux Torres et al. Reference Renaux Torres, Pellot, Somwang, Khositharattanakool, Vongphayloth, Randrianambinintsoa, Mathieu, Siriyasatien, Gay and Depaquit2023). In Chiang Rai, there was at least one autochthonous cutaneous leishmaniasis case (L. martiniquensis) in an immunocompetent person, and additional asymptomatic infection was detected via molecular and serological testing in the community (Sriwongpan et al. Reference Sriwongpan, Nedsuwan, Manomat, Charoensakulchai, Lacharojana, Sankwan, Kobpungton, Sriwongpun, Leelayoova, Mungthin and Siripattanapipong2021). These findings reinforce the endemic status of these provinces and underscore the need for ongoing vector and host surveillance.
Several sand fly species have been implicated as potential vectors of Trypanosoma spp., which have been detected in both endemic and non-endemic areas of leishmaniasis in Thailand (Phumee et al. Reference Phumee, Tawatsin, Thavara, Pengsakul, Thammapalo, Depaquit, Gay and Siriyasatien2017, Reference Phumee, Sutthanont, Chitcharoen, Sawaswong, Boonserm, Ayuyoe, Cantos-Barreda and Siriyasatien2024; Buatong et al. Reference Buatong, Dvorak, Thepparat, Thongkhao, Koyadun, Siriyasatien and Pengsakul2022; Khositharattanakool et al. Reference Khositharattanakool, Pathawong, Pongsiri, Pengsakul, Ponlawat and Somwang2024; Klaiklueng et al. Reference Klaiklueng, Kumlert, Moonmake, Ruang-Areerate, Siriyasatien, Sunantaraporn, Wanachiwanawin, Ruenchit and Wongkamchai2025). For Trypanosoma species, T. noyesi, Trypanosoma Anura01/02, Trypanosoma Anura04 and Trypanosoma sp. were identified from sand fly specimens collected in Thailand. Notably, T. noyesi is a wildlife-associated species described in Australian marsupials and classified within the T. cruzi clade; although its DNA has also been detected in sand flies in Thailand (Srisuton et al. Reference Srisuton, Phumee, Sunantaraporn, Boonserm, Sor-Suwan, Brownell, Pengsakul and Siriyasatien2019; Khositharattanakool et al. Reference Khositharattanakool, Pathawong, Pongsiri, Pengsakul, Ponlawat and Somwang2024), there is no evidence of its involvement in human or livestock infection (Botero et al. Reference Botero, Cooper, Thompson, Clode, Rose and Thompson2016). Besides, Trypanosoma Anura01/02 and Anura04, also known as Frog1 and Frog2 lineages, have been identified in amphibian hosts and frog-feeding vectors, without evidence of zoonotic potential (Spodareva et al. Reference Spodareva, Grybchuk-Ieremenko, Losev, Votýpka, Lukeš, Yurchenko and Kostygov2018). Detection of Anuran-type Trypanosoma DNA in sand flies and in the flat-tailed house gecko in southern Thailand suggests possible ecological overlap (Toontong et al. Reference Toontong, Sunantaraporn, Tiawsirisup, Pengsakul, Boonserm, Phumee, Siriyasatien and Preativatanyou2022). Therefore, the presence of Trypanosoma spp. DNA in diverse sand fly species raises concerns about their role in parasite transmission and suggests broader ecological and public health implications beyond known Leishmania spp. affected areas. Briefly, all Trypanosoma-positive sand flies involved 13 species and were collected across 10 provinces: Chiang Rai, Lampang, Chanthaburi, Sa Kaeo, Songkhla, Narathiwat, Satun, Phatthalung, Chumphon and Pang Nga. Moreover, the molecular diversity of trypanosomes highlights the need for further research on their transmission dynamics, reservoir hosts and potential public health implications.
The emergence of Bartonella tamiae as a human pathogen in Thailand has raised concerns regarding its transmission dynamics (Kosoy et al. Reference Kosoy, Bai, Sheff, Morway, Baggett, Maloney, Boonmar, Bhengsri, Dowell, Sitdhirasdr and Lerdthusnee2010). Herein, unidentified Bartonella DNA was also detected in sand fly specimens from southern Thailand (Sunantaraporn et al. Reference Sunantaraporn, Somwang, Khositharattanakool, Unchanam, Saenchaiban, Wongkhut, Sanum, Pataradool, Boonserm, Depaquit and Siriyasatien2024). The genus Bartonella includes multiple pathogenic species associated with a wide range of clinical manifestations in humans, such as cat scratch disease (B. henselae), trench fever (B. quintana) and endocarditis or chronic bacteremia (B. henselae and B. quintana) (Harms and Dehio, Reference Harms and Dehio2012). Notably, B. bacilliformis, transmitted by Lutzomyia sand flies in South America, causes Carrion’s disease, which may present in acute (Oroya fever) or chronic (Verruga peruana) forms and can be fatal without appropriate treatment (Pons et al. Reference Pons, Gomes, Del Valle-mendoza and Ruiz2016). Although the vector of B. tamiae remains identified, recent molecular evidence has detected the Bartonella spp. DNA in three sand fly species (S. khawi, S. anodontis and S. barraudi) within the endemic areas of leishmaniasis (Sunantaraporn et al. Reference Sunantaraporn, Somwang, Khositharattanakool, Unchanam, Saenchaiban, Wongkhut, Sanum, Pataradool, Boonserm, Depaquit and Siriyasatien2024). However, detection of DNA alone is insufficient to establish vector competence. Further entomological, molecular and xenodiagnostic studies are required to elucidate the role of sand flies in the ecology and transmission of zoonotic Bartonella species.
Although identifying host-virus associations remains a major challenge in virus research in Thailand, the detection of Rhabdovirus and Orbivirus in several sand fly species in southern Thailand, in endemic areas for leishmaniasis, highlights the need to develop vector control strategies targeting sand fly-borne viruses (Phumee et al. Reference Phumee, Wacharapluesadee, Petcharat, Tawatsin, Thavara and Siriyasatien2021a, Reference Phumee, Wacharapluesadee, Petcharat and Siriyasatien2021b). Briefly, Orbivirus which is closely related to Changuinola virus (CGLV), known to cause febrile illness (Changuinola fever) in Central and South America (Peralta et al. Reference Peralta, Shelokov, Vogel and Longfellow1966; Travassos da Rosa et al. Reference Travassos da Rosa, Tesh and Pinheiro1984; Phan et al. Reference Phan, Tesh, Guzman and Delwart2020), was identified in sand flies: S. khawi, P. papatasi, Idiophlebotomus sp. in Thailand (Phumee et al. Reference Phumee, Wacharapluesadee, Petcharat, Tawatsin, Thavara and Siriyasatien2021a). In addition to this, Rhabdovirus-like sequences, closely related to insect-specific Bactrocera dorsalis sigmavirus strain BDSV.abc5 and the lineages of insect-specific Rhabdoviridae, underscoring considerable viral diversity in sand flies in Thailand (Phumee et al. Reference Phumee, Wacharapluesadee, Petcharat and Siriyasatien2021b).
The host blood meal analyses of sand flies in Thailand have been done in only three articles (Siripattanapipong et al. Reference Siripattanapipong, Leelayoova, Ninsaeng and Mungthin2018; Buatong et al. Reference Buatong, Dvorak, Thepparat, Thongkhao, Koyadun, Siriyasatien and Pengsakul2022; Khositharattanakool et al. Reference Khositharattanakool, Pathawong, Pongsiri, Pengsakul, Ponlawat and Somwang2024). Interestingly, the blood meal of S. khawi was found to come from humans (Buatong et al. Reference Buatong, Dvorak, Thepparat, Thongkhao, Koyadun, Siriyasatien and Pengsakul2022) and from frogs, specifically from Micryletta inornate and Microhyla fissipes (Khositharattanakool et al. Reference Khositharattanakool, Pathawong, Pongsiri, Pengsakul, Ponlawat and Somwang2024). Moreover, human blood was detected in a wide variety of sand fly species, including S. iyengari, S. barraudi, S. hodgsoni, S. sylvatica, S. hamidi, S. indica, P. argentipes, P. stantoni, I. asperulus and Sergentomyia sp., while insects’ blood was identified inside S. gemmea (Siripattanapipong et al. Reference Siripattanapipong, Leelayoova, Ninsaeng and Mungthin2018; Buatong et al. Reference Buatong, Dvorak, Thepparat, Thongkhao, Koyadun, Siriyasatien and Pengsakul2022). These findings underscore that several sand fly species, particularly those of the genus Sergentomyia, exhibit opportunistic feeding behaviour that includes humans, suggesting potential implications for zoonotic transmission. Moreover, multiple pathogens including viral RNA detected in S. khawi and previous evidence of human host blood meal should be considered for further clarification.
This review summarizes current data on phlebotomine sand fly distribution and pathogen detection in Thailand, highlighting species abundance trends, identification methods and potential vector-pathogen links. Our study focusing on Thailand enables deeper insight into the regional patterns often missed in broader analyses. This comprehensive review will later be expanded to include other Southeast Asian countries within the Greater Mekong Subregion, where regional awareness will support the development of effective control measures and monitoring plans.
Conclusion
This scoping review provides an updated picture of sand fly species diversity, distribution and their associations with pathogens in Thailand. Molecular evidence confirmed the presence of Leishmania spp., Trypanosoma spp., Bartonella spp. and viruses in multiple sand fly species, including those previously overlooked as potential vectors. Notably, human blood meals were detected in several Sergentomyia species, of which S. khawi also hosted several pathogens, underscoring their possible role in zoonotic transmission. Continued entomological surveillance, vector-pathogen interaction studies and blood meal analysis are vital for vector control strategies and understanding disease risks in Thailand. In addition, this study contributes to updated knowledge through publicly accessible interactive maps of sand fly distribution and their pathogen occurrence.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0031182025101091.
Author contribution
Babi Kyi Soe: Writing – review & editing, Writing – original draft, Validation, Methodology, Investigation, Resources, Formal analysis, Data curation, Conceptualization. Apinya Arnuphapprasert: Investigation, Resources, Data curation. Poom Adisakwattana: Writing – review & editing, Supervision, Investigation, Resources, Data curation, Conceptualization. María Ortuño Gil: Resources, Investigation, Validation. Ana Huertas-Lopez: Writing – review & editing, Supervision, Validation, Methodology, Investigation, Resources, Formal analysis, Data curation, Conceptualization.
Financial support
The authors would like to thank the Graduate Scholarship Program for ASEAN and NON-ASEAN countries, Chulalongkorn University, the Erasmus+ International Mobility Program (University of Murcia) and the Veterinary Pathology Program, Faculty of Veterinary Science, Chulalongkorn University for funding the research stay in Thailand (2019–2021), which enabled the development of this collaborative study.
Competing interests
The authors declare no financial or personal relationships with individuals or organizations that could inappropriately influence or bias the content of this paper.
Ethical standards
Not applicable.