Introduction
Non-human primates (NHPs) share many intestinal parasites with humans, and cross-species exchange of parasitic infections between NHPs and humans has been demonstrated in shared Central African tropical forest ecosystems (Medkour et al., Reference Medkour, Amona, Laidoudi, Davoust, Bitam, Levasseur, Akiana, Diatta, Pacheco, Gorsane and Sokhna2020). Under environmental pressure, NHPs may acquire and act as reservoirs for human parasitic infection, with a flow of interspecies infection occurring. This is specifically the case for the cross-species exchange of environmentally acquired intestinal parasites, such as soil-transmitted helminths (STH) and waterborne intestinal protozoa and helminths. Such exchange has been demonstrated for the waterborne helminth Schistosoma mansoni infections (Kebede et al., Reference Kebede, Bech, Allienne, Olivier, Erko and Boissier2020; Ketzis et al., Reference Ketzis, Lejeune, Branford, Beierschmitt and Willingham2020) and the STH Strongyloides fuelleborni (Janwan et al., Reference Janwan, Rodpai, Intapan, Sanpool, Tourtip, Maleewong and Thanchomnang2020), Ternidens deminutus (Bradbury, Reference Bradbury2019), Oesophagostomum spp. (Sirima et al. Reference Sirima, Bizet, Hamou, Červená, Lemarcis, Esteban, Peeters, Mpoudi Ngole, Mombo, Liégeois and Petrželková2021), Necator gorillae (Hasegawa et al., Reference Hasegawa, Modrý, Kitagawa, Shutt, Todd, Kalousová, Profousová and Petrželková2014; Pafčo et al., Reference Pafčo, Kreisinger, Čížková, Pšenková-Profousová, Shutt-Phillips, Todd, Fuh, Petrželková and Modrý2019) and some sub-clades of Trichuris trichiura (Rivero et al., Reference Rivero, Cutillas and Callejón2021).
In areas of West Africa where significant deforestation has occurred, there is increasing pressure on NHP populations to reside near human settlements. There has been significant recent work on the richness and diversity of intestinal parasites of NHPs in West Africa, specifically in Senegal (Medkour et al., Reference Medkour, Amona, Laidoudi, Davoust, Bitam, Levasseur, Akiana, Diatta, Pacheco, Gorsane and Sokhna2020; N’da et al., Reference N’da, Dahourou, Gbati and Alambedji2020, Reference N’da, Dahourou, Ndiaye, Lindshield, Gbati and Traore2022), Cote d’Ivoire and Sierra Leone (Köster et al., Reference Köster, Lapuente, Pizarro, Prieto-Pérez, Pérez-Tanoira, Dashti, Bailo, Muadica, González-Barrio, Calero-Bernal and Ponce-Gordo2022). However, there is a paucity of information from The Gambia, a country in this region nestled within Senegal and particularly affected by desertification. The Gambia lost almost 100 000 hectares of land to desertification between 1998 and 2009 (Food and Agriculture Organization, 2021). While forest management systems are now in place, such reduction and fragmentation of habitat has been shown to place increased pressure on African NHP populations and places many groups into closer contact with humans (Bloomfield et al., Reference Bloomfield, McIntosh and Lambin2020). Prior work in Uganda, East Africa (Gillespie et al., Reference Gillespie, Chapman and Greiner2005; Gillespie and Chapman, Reference Gillespie and Chapman2008; Zommers et al., Reference Zommers, Macdonald, Johnson and Gillespie2013) has demonstrated that the fragmentation of forests leads to an increase in parasite richness and diversity in resident NHPs. The combination of closer contact between humans and NHPs in shared environments and increased parasite richness and diversity in the NHP population presents a potential risk to human health (Wallis and Lee, Reference Wallis and Lee1999; Chapman and Peres, Reference Chapman and Peres2001; Chapman et al., Reference Chapman, Gillespie and Goldburg2005; Hopkins and Nunn, Reference Hopkins and Nunn2007; Devaux et al., Reference Devaux, Mediannikov, Medkour and Raoult2019, Reference Devaux, Mediannikov, Medkour and Raoult2019; Bloomfield et al., Reference Bloomfield, McIntosh and Lambin2020).
The only previous studies to analyse the impact of human activity on the richness and diversity of intestinal parasitic fauna in NHPs were conducted in a lush river forest of Kenya in East Africa (Mbora and McPeek, Reference Mbora and McPeek2009) and a variety of climates in South Africa (Gaetano et al., Reference Gaetano, Danzy, Mtshali, Theron, Schmitt, Grobler, Freimer and Turner2014). These are geographically and environmentally quite different from the Sahelian environment of The Gambia. This study serves to provide baseline information about the prevalence of intestinal helminths and protozoa among diurnal primate species found in The Gambia and determines the association of richness and diversity of these parasites with closeness to human activity.
Methods
Sample and data collection
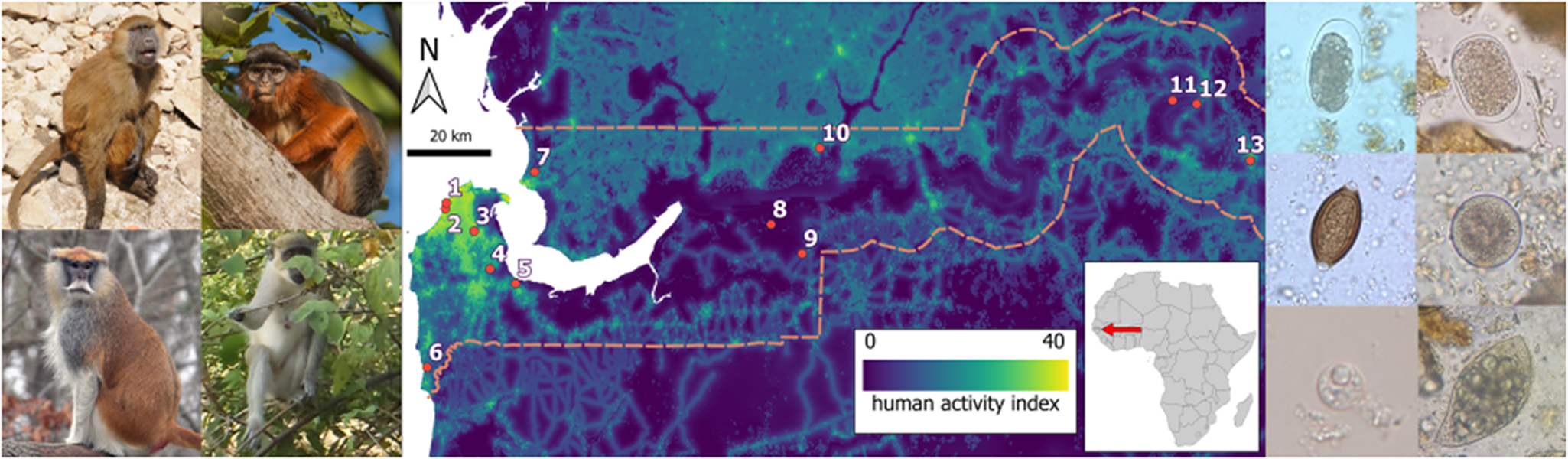
We collected fresh faecal samples from 4 diurnal monkey species (Chlorocebus sabaeus, Erythrocebus patas, Papio papio and Piliocolobus badius) spread across 13 sampling sites (Senegambia, Bijilo Park, Abuko, Makasutu, Pirang, Kartong, Niumi, Kiang-West, Kiang-West HQ, dia Fula, River Gambia, Touba and Janjanbureh) in The Gambia with a mix of Aw-Savannah, BSh – Arid hot steppe and localized Af – Tropical rainforest climate Köppen−Geiger Climate Classifications (Peel et al., Reference Peel, Finlayson and McMahon2007) and diverse surrounding physical environment (Table 1) and degree of human activity (Figure 1). Sampling was undertaken during the early onset of the wet season in June 2017, June 2018 and June 2019.
Heat map of human activity within The Gambia (map becomes lighter in colour with increased human activity), with the location of the 13 study sites superimposed (1, Senegambia; 2 Bijilo Park; 3, Abuko; 4, Makasutu; 5, Pirang; 6, Kartong; 7, Niumi; 8, Kiang-West; 9, Kiang-West HQ; 10, dia Fula; 11, River Gambia; 12, Touba; 13, Janjanbureh). The dashed line represents the political border between The Gambia and Senegal.

Site numbers, site names, position, Köppen–Geiger climate classification and written description of the immediate environment of the 13 sites samples were collected from in this study

To minimize the risk of duplicate sampling from a single individual, single-day troop follows were conducted by a qualified primatologist and wildlife officers. These monkey species are quite morphologically distinct, as is their faeces composition; thus, it was easy to clearly distinguish between stools from the different monkey species.
Metadata for individuals and sites was collected as follows. When possible, the sex and age class (infant, juvenile, subadult and adult) of the individual were also noted, along with GPS coordinates for each troop location, which were collected using a Garmin eTrex 10 handheld navigator. The group size for each NHP species was determined by visual inspection. Endangered animal categories were sourced from the International Union for the Conservation of Nature (IUCN)’s Red List (International Union for the Conservation of Nature, 2021).
Sampling was conducted during morning hours, beginning when monkeys were at sleeping sites from the previous night through the first 3–4 h of departing their sleeping sites. Faecal samples were collected using fresh wooden spatulas or plastic spatula inside the collection tubes. Where possible, the whole faecal sample was collected, or a portion of the centre of the faecal sample was collected, taking care not to collect portions of the stool sample contaminated by urine or environmental debris. Faecal samples were immediately stored in fixative/reagents. In 2017, samples were only stored in Total-Fix® (Medical Chemical Company, Torrance, CA). In 2018 and 2019, samples were collected in both Total-Fix® and 10% formalin (Medical Chemical Company, Torrance, CA). All the samples were stored at ambient temperature (20 to 25 °C) prior to analysis.
Human activity index
Each site was assigned a human activity index score using the 2019 global human footprint map for terrestrial environments by Gassert et al. (Reference Gassert, Ventner, Watson, Brumby, Mazzariello, Atkinson and Hyde2023). For this map, the index was calculated at a 100 m2 resolution and can range from 0 to 50, a higher score indicative of a greater human activity. Within The Gambia, human activity scores ranged from 3.2 to 41.1. To account for the range of values an individual NHP experiences within its home range, we used the mean human activity index calculated from all grid cells within a 2.5 km radius of each sample site for all analyses.
Sample shipment
Total-Fix® preserved samples were shipped at ambient temperature to the Centers for Disease Control and Prevention (CDC) in the United States. These did not require a U.S. Department of Agriculture import permit as they had been specifically treated and rendered non-infectious. The 10% formalin preserved samples were shipped at ambient temperature to Federation University in Australia. An Australian Department of Agriculture, Fisheries and Forestry (DAFF) import permit was not required as these samples have been preserved and fixed correctly by a department-approved method (10% formalin; reference DAFF BICON case: preserved and fixed animal and human specimens, effective: 07 January 2021).
Laboratory processing
The entire Total-Fix® or 10% formalin preserved samples were homogenized by shaking, then a 2 mL aliquot was diluted with 9 mL 0.85% saline and filtered through a 1 mm wire mesh, followed by centrifugation at 500 g for 5 min.
Wheatley’s trichrome stain
The packed faecal deposit was used to prepare smears for staining by mixing 5:1 with Mayers albumin (Meridian Biosciences, Cincinnati, OH) followed by smearing onto the surface of a microscope slide before being allowed to dry in ambient air at room temperature. The dried faeces and Mayer’s albumin smears were fixed in methanol for 5 min prior to staining using Wheatley’s trichrome stain (Garcia, Reference Garcia2009). This was followed by examination for protozoa under ×1000 oil immersion magnification.
Cyclospora detection by autofluorescence
A wet mount of the packed faecal deposit was made in one drop of 0.85% saline, a 18 × 18 mm coverslip placed on top and the entire coverslip scanned under ×400 magnification using ultraviolet microscopy at wavelength 350 nm to identify auto-fluorescing oocysts of Cyclospora spp.
Formalin ethyl-acetate concentration
The remaining packed faecal samples were corrected to a volume of 0.5 g, followed by resuspension in 9 mL of 10% formalin. The resuspended formalin solution was allowed to diffuse into the faecal matter for at least 60 min. Following this, parasite concentration of formalin-resuspended stool was performed using the formalin ethyl-acetate concentration (FEC) method (Garcia, Reference Garcia2009). Two 0.85% saline wet mounts of the resultant FEC deposit were prepared using 18 × 18 mm coverslips and both were scanned by light microscopy under ×100 magnification for helminth eggs and ×400 magnification for protozoa cysts and trophozoites.
Cyclospora 18S rRNA gene sequencing
Total-Fix® preserved aliquots of samples that tested positive for Cyclospora spp. by microscopy were subjected to PCR and sequencing of the Cyclospora 18S rRNA gene for species identification. We utilized the UNEX-buffer method for total genomic DNA extraction (Qvarnstrom et al., Reference Qvarnstrom, Benedict, Marcet, Wiegand, Herwaldt and da Silva2018). A portion of the 18S rRNA gene was amplified using PCR primers CRYPTOFL and cycR2 (da Silva et al., Reference da Silva, Cacciò, Williams, Won, Nace, Whittier, Pieniazek and Eberhard2003; Eberhard et al., Reference Eberhard, Owens, Bishop, de Almeida and da Silva2014). PCR products were Sanger sequenced using the BigDye V3.1 chemistry on an ABI Prism 3100 sequence analyser (Life Technologies).
Statistical analysis
A χ2 test in Microsoft Excel (version 2208) was used to compare the prevalence of any intestinal parasite, intestinal protozoa and STH infection in each NHP species sampled. All other statistical analyses were performed using the R software environment (version 4.1.2). For the 2 most surveyed species of NHP, P. papio (n = 33) and P. badius (n = 28), we fitted a generalized linear mixed model (GLMM) to identify the effect of human activity and group size on parasite richness within an individual using the glmer function in the lme4 package (Bates et al., Reference Bates, Mächler, Bolker and Walker2015). We fitted each GLMM with a Poisson error distribution and a logarithmic link function, which is appropriate when the response variable is count data (Zuur et al., Reference Zuur, Ieno, Walker, Saveliev and Smith2009). The human activity index and group size were included in each model as fixed effects, whereas sample site was included as a random effect to account for observations at each site not being statistically independent (Zuur et al., Reference Zuur, Ieno, Walker, Saveliev and Smith2009). Given that collinearity among predictor variables can influence coefficient estimates and statistical power in GLMM, models were inspected for multicollinearity by measuring the variance inflation factor (VIF) using the vif function in the car package (Fox and Weisberg, Reference Fox and Weisberg2019). We also tested for spatial autocorrelation within our models by calculating Moran’s I using residuals grouped by sample site using the testSpatialAutocorrelation in the DHARMa package (Hartig, Reference Hartig2020). There was no evidence of strong collinearity among predictor variables (VIF < 5.0 in all cases) or spatial autocorrelation in model residuals (Moran’s I between −1 and 1 in all cases), and thus, we proceeded without further consideration of either phenomenon. Residual vs. fitted value plots were inspected to ensure model residuals did not violate the statistical assumptions required for parametric tests. For each model, we report the parameter coefficients, z-scores and associated P-values for human activity and group size.
Results
NHP distribution across sampling sites
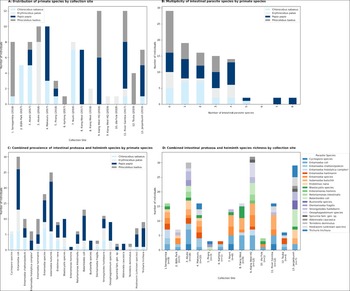
A total of 99 NHPs from all 4 diurnal species indigenous to The Gambia were sampled over the sampling period as follows: 43 in 2017, 44 in 2018 and 12 in 2019. Not all NHP species were observed at each sample site. We observed and collected samples from C. sabaeus (IUCN species of least concern; n = 24) at 8 sites, E. patas (IUCN near threatened species; n = 14) at 6 sites, P. papio (IUCN near threatened species; n = 33) at 8 sites and P. badius (IUCN endangered species; n = 28) at 9 sites (Figure 2a).
Results of faecal samples collection and parasite screening of four diurnal non-human primate (NHP) species conducted in The Gambia, West Africa, in June 2017, June 2018, and June 2019. (A) Distribution of specimens collected at each sampling site; (B) Multiplicity of infection in NHP species; (C) Prevalence of intestinal protozoa and helminth species in NHP species; (D) Combined intestinal protozoa and helminth richness by study site. *The Entamoeba histolytica complex includes Entamoeba nuttali, E. histolytica, E. dispar, E. moshkovski and E. bangladeshi, which cannot be differentiated by microscopy alone.

Prevalence and diversity of intestinal helminths and protozoa
We observed 21 intestinal parasite species from at least 17 separate genera, one genus of coccidia (Cyclospora spp.), 4 species of Entamoeba, 2 ciliate protozoa (Balantioides coli and Buxtonella sp.) and 7 other genera of protozoa. In 18 cases, the species of Entamoeba could not be determined due to morphological ambiguity. At least 5 genera of STH (including hookworms, which may represent multiple genera) were identified, as well as 2 insect-intermediate-host-transmitted spirurid nematode genera (Spirurina fam. gen. sp.) (Figure 3).
Selected intestinal parasites identified in four diurnal non-human primate species specimens collected at each sampling site in The Gambia, June 2017 June 2018, and June 2019. In wet preparation microscopy; (A) hookworm egg, (B) Oesophagostomum egg, (C) Strongyloides fuelleborni fuelleborni egg, (D) Trichuris trichiura egg, (E) Spirurina fam. gen. sp. egg type 1, (F) Spirurina fam. gen. sp. egg type 2, (G) Balantioides coli cyst, (H) Balantioides coli trophozoite; (I) Buxtonella sp. cyst, (J) Buxtonella sp. trophozoite, (K) Entamoeba coli cyst; (L) autofluorescent Cyclospora sp. oocyst at wavelength 350 nm, and, in trichrome stain; (M) Entamoeba chattoni/polecki cyst, (N) Iodamoeba buetschlii cyst, (O) Blastocystis sp. trophozoite and, (P) Endolimax nana cyst.

At least one intestinal parasite was detected in 82% of P. papio, 79% of C. sabaeus, 64% of E. patus and 54% of P. badius, with a mean average intestinal parasite prevalence of 71% across all NHP species examined (Figure 2b). The difference in overall parasite prevalence between the 4 NHP species was not statistically significant (χ2 P = 0.105). Regarding the average richness of parasitic infections, P. papio had the highest mean average richness for individuals (mean average = 2.8, median = 3); followed by E. patas (mean average = 2.0, median = 1.5); then C. sabaeus (mean average = 1.7, median = 1.5) and P. badius (mean average = 1.1, median = 1.0). Guinea baboons (P. papio) were most likely to harbour more than one intestinal parasite (Figure 2b).
The prevalence of intestinal protozoan infection was 82% in P. Papio, 75% in C. sabaeus, 64% in E. patus, 50% in P. badius and 69% across all NHP species examined. These differences in protozoan prevalence by species were not statistically significant (χ2 P = 0.051). The most common species of intestinal protozoa were Entamoeba coli (30%), followed by I. buetschlii (25%) (Figure 2c). Five (83%) Cyclospora infections identified were in C. sabaeus. A single E. patas was also identified (Figure 2c). Cyclospora 18S rRNA PCR was performed on 6 positive samples. Two samples (both from C. sabaeus at site 7) were positive by PCR and generated DNA sequences almost identical (one base pair difference in 1014 base pairs total; GenBank accession number OR699281) to Cyclospora cercopitheci 18S rRNA.
The prevalence of STH infection was 39% in P. papio, 43% in E. patas, 21% in C. sabaeus, 14% in P. badius and 28% across all NHP species examined; however, these differences were not statistically significant (χ2 P = 0.078). The most prevalent species of helminth was Strongyloides fuelleborni subsp. fuelleborni (11%), then Oesophagostomum spp. (9%), T. trichiura (9%) and hookworms (7%). Five of eight Oesophagostomum spp. infections (60%) were from a small geographic region of Kiang West province (Figure 1). The hookworms, T. trichiura and S. f. fuelleborni infections showed no apparent geographic clustering (Figure 2d).
The effect of human activity and group size on parasite richness
The results of the GLMM for P. badius indicated that there was no significant association with human activity (β = 0.604, z = 1.401, P = 0.161) or group size (β = 0.158, z = 0.520, P = 0.603) on parasite richness. Similarly, the GLMM for P. papio indicated that there was no significant effect of human activity (β = −0.048, z = −0.232, P = 0.817) or group size (β = −0.098, z = −0.514, P = 0.607) on parasite richness in individuals. There were insufficient observations from insufficient sites to fit a GLMM to E. patas or C. sabaeus.
Discussion
This study represents the first survey of such parasites in NHPs from The Gambia, and 1 of only 6 surveys performed in the semi-arid Sudanian environmental zone of West Africa (McGrew et al., Reference McGrew, Tutin, Collins and File1989; Howells et al., Reference Howells, Pruetz and Gillespie2011; Joshua et al., Reference Joshua, Yidawi, Sada, Msheliza and Turaki2020; Medkour et al., Reference Medkour, Amona, Laidoudi, Davoust, Bitam, Levasseur, Akiana, Diatta, Pacheco, Gorsane and Sokhna2020; N’da et al., Reference N’da, Dahourou, Gbati and Alambedji2020). Our results reveal a greater diversity of parasite species than many others from the Sudanian climate region, with Gambian NHPs having a richness of intestinal parasite fauna only matched by chimpanzees in the nearby south-eastern region of Senegal (McGrew et al., Reference McGrew, Tutin, Collins and File1989; Howells et al., Reference Howells, Pruetz and Gillespie2011). This may reflect increased habitat pressure on Gambian NHPs in comparison to those elsewhere. The Gambia lost almost 100 000 hectares of land between 1998 and 2009 to desertification (Food and Agriculture Organization, 2021), and while forest management strategies are now in place, this reduction in habitat may have placed increased parasitic disease pressure on the Gambian NHP population.
Habitat loss and an increasing human population in this region have led to greater contact between NHPs and humans in some regions of West Africa and elsewhere (Chapman and Peres, Reference Chapman and Peres2001). Furthermore, primate populations have been restricted to tightly confined areas of remnant habitat (Wallis and Lee, Reference Wallis and Lee1999; Chapman and Peres, Reference Chapman and Peres2001). Both factors increase stress on individual NHPs, reduce population size and expose them to a higher risk of disease acquisition and transmission between humans and animals (Wallis and Lee, Reference Wallis and Lee1999; Chapman et al., Reference Chapman, Gillespie and Goldburg2005). Both humans and NHPs are likely to share water sources and are thereby exposed to the same gastrointestinal protozoa. Similarly, humans and NHPs will be exposed to the same infective stages of STH in the soil. For such parasites, it is deemed likely that such cross-species parasite transmission will occur within a given geographical environment (Chapman et al., Reference Chapman, Gillespie and Goldburg2005). However, our study did not observe any significant increase in the richness of intestinal parasite species in P. badius and P. papio in sites with higher human activity indices than in sites with lower human activity. Erythrocebus patas and C. sabaeus were not assessed due to insufficient sample size. Gaetano et al. (Reference Gaetano, Danzy, Mtshali, Theron, Schmitt, Grobler, Freimer and Turner2014) similarly reported no effect of anthropogenic activity (as determined by reference to a global human population density grid similar to the human activity index employed in our study) on intestinal parasitism in 78 South African vervet monkeys (Chlorocebus aethiops). In contrast, Mbora and McPeek (Reference Mbora and McPeek2009) compared human activity, measured by the basal area of cut stems at a given site, with the richness and diversity of intestinal parasites in 2 species of NHP within the Tana River Primate National Reserve in Kenya. In this study, Tana River red colobus (Piliocolobus rufomitratus) (n = 150) and mangabeys (Cercocebus galeritus) (n = 81) demonstrated increased parasite prevalence and richness associated with greater population density and crowding due to decreased and fragmented habitat consequent to human activity (Mbora and McPeek, Reference Mbora and McPeek2009). It remains possible that the much larger sample size employed in this Kenya study is responsible for the variation in findings of their work when compared to that of our work and that of Gaetano et al. (Reference Gaetano, Danzy, Mtshali, Theron, Schmitt, Grobler, Freimer and Turner2014).
The greatest richness in parasite infection was observed in P. papio, consistent with increased richness of parasites in Papio spp. in other studies (Frias and MacIntosh, Reference Frias, MacIntosh, Knauf and Jones-Engel2020). Being terrestrial, highly mobile and more likely to be in close proximity to humans during foraging are considered favourable factors leading to higher parasite prevalence in Guinea baboons from Senegal (N’da et al., Reference N’da, Dahourou, Ndiaye, Lindshield, Gbati and Traore2022). We consider this to be the likely reason for the increased parasite richness also observed in Gambian P. papio Guinea baboons.
Gambian P. badius showed a lower prevalence of overall intestinal parasite, intestinal protozoan and STH infections when compared to the other NHP species sampled, although this did not reach significance. This is consistent with Mbora and McPeek (Reference Mbora and McPeek2009), which also found a higher prevalence of parasites in terrestrial mangabeys than in arboreal Tana River red colobus monkeys, but unlike our data, their findings did reach significance (P < 0.01). Mbora and McPeek (Reference Mbora and McPeek2009) postulated that NHP species behaviour leads to these findings, and we agree. The more arboreal nature of P. badius may contribute to this phenomenon. Observational studies in The Gambia have noted that P. badius monkeys rarely descend to the ground to obtain water, and only do so during the dry season (Hillyer et al., Reference Hillyer, Armstrong and Korstjens2015). This species mostly obtains water from leaves, collections in tree trunk depressions, or by licking their bodies after rain (Hillyer et al., Reference Hillyer, Armstrong and Korstjens2015). The primary use of relatively ‘cleaner’ water sources by P. badius, and their aversion to terrestrial activity, likely reduces the opportunities for exposure to waterborne protozoa and STH from both humans and other NHP species. While the lower intestinal parasite prevalence observed in P. badius was not statistically significant in our study, additional investigation with larger sample sizes would better elucidate this hypothesis.
This study provides the first identification of Cyclospora infection in C. sabaeus and E. patas hosts. Since the morphology of all primate Cyclospora species is the same or very similar, the determination of the various Cyclospora species requires sequencing. Two of these infections (occurring in C. sabaeus hosts) were possible to identify by sequencing as C. cercopitheci. Originally, these were identified in C. aethiops from Ethiopia (Eberhard et al., Reference Eberhard, da Silva, Lilley and Pieniazek1999) and later Kenya (Eberhard et al., Reference Eberhard, Njenga, DaSilva, Owino, Nace, Won and Mwenda2001). This report expands both the known hosts and the geographical range of this parasite, as all prior reports were restricted to East Africa (Eberhard et al., Reference Eberhard, da Silva, Lilley and Pieniazek1999, Reference Eberhard, Njenga, DaSilva, Owino, Nace, Won and Mwenda2001; Lopez et al., Reference Lopez, Manglicmot, Schmidt, Yeh, Smith and Relman1999; Li et al., Reference Li, Kiulia, Mwenda, Nyachieo, Taylor, Zhang and Xiao2011), or much further south in Equatorial Guinea (Eberhard et al., Reference Eberhard, Owens, Bishop, de Almeida and da Silva2014).
Wild NHP stool is difficult to obtain. The use of only one stool per individual likely reduced the recovery of intestinal parasites in this study (Garcia, Reference Garcia2009) but was unavoidable. The choice of preservative was changed during the study due to Australian biosecurity importation rules, and this may have had a minor influence on the recovery rate of low-intensity parasite infections. This may have affected the findings of samples tested between 2017 and 2018–2019. A larger study, incorporating more individuals and collection of samples from both humans and monkeys in the same area, with associated genotyping, is indicated.
In summary, our study provides insight into the richness and diversity of intestinal parasites in NHPs from The Gambia, West Africa. Despite increased contact between NHPs and humans due to desertification, our results suggest that the richly diverse population of parasites in these diurnal monkeys is not influenced by human activity or group size in this setting; further investigation with a larger dataset is required to better elucidate these findings.
Acknowledgements
The authors would like to thank Mr Mark Fox of the Division of Parasitic Diseases and Malaria, CDC for trichrome staining on the 2018 samples discussed in this paper.
Author contributions
J.D.C., Y.Q., A.R.O. and R.S.B. conceived of the study. J.D.C. and M.J. collected samples. J.D.C. and M.J. collected site and NHP-specific data. J.D.C., E.F.N. and M.A. performed initial sample processing and shipment. S.G.H.S, Y.Q., I.S.P. and R.S.B. performed laboratory analyses. R.S.B., J.D.C. and A.R.O. collated and analysed data, and performed statistical analyses. A.R.O., E.F.-N. and R.S.B. undertook data visualization. R.S.B. wrote the first draft of the manuscript. All authors contributed to, reviewed and approved the final draft of the manuscript.
Financial support
This study was funded by the Division of Parasitic Diseases and Malaria, CDC.
Competing interests
The authors declare no competing interests. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Agency for Toxic Substances and Disease Registry or the Centers for Disease Control and Prevention.
Ethical standards
Ethical review of this work was conducted by the Medical Research Council Unit The Gambia at the London School of Hygiene and Tropical Medicine (SCC 1536v1.1). This was reciprocally reviewed by the Office of the Associate Director for Science, Center for Global Health and CDC. Approval for primate research in National Parks was granted by the Department of Parks and Wildlife Management, The Gambia [Ref. AHB 159/192/01(91)].