Introduction
Cerebral small vessel disease (CSVD), defined as pathology of the small-sized (generally less than 500 micron diameter) arteries and veins, Reference Pantoni1 becomes extremely common with aging. CSVD has been linked to risk for ischemia, hemorrhage, white matter demyelination, perivascular space enlargement and brain atrophy. Reference Duering, Biessels and Brodtmann2 It is an important cause of stroke, but even more importantly it is the second most common cause of dementia. Reference Matthews, Brayne and Lowe3 In this review, I will describe the main types of CSVD, their diverse clinical consequences, their radiological manifestations and new pathophysiological targets for treatment.
Types of CSVD
Pantoni proposed that there are six subtypes of CSVD that can classified based on arterial and venous wall pathology (Table 1). Reference Pantoni1
Types of cerebral small vessel disease

The most common CSVD pathology is arteriolosclerosis. Its prevalence increases with aging, hypertension and other vascular risk factors. Reference Oveisgharan, Kim and Agrawal4 The molecular steps that mediate arteriolosclerosis are poorly understood, warranting more investigation.
Cerebral amyloid angiopathy (CAA) caused by accumulation of amyloid in the media and adventitia of small arteries of the brain and leptomeninges. Reference Charidimou, Boulouis and Frosch5 Although several types of amyloid can cause CAA, the only common, age-related, sporadic aggregate is beta-amyloid. Reference Revesz, Holton and Lashley6 Beta-amyloid is formed by aggregation of abeta peptides derived from proteolytic cleavage of the amyloid precursor protein. CAA is a cousin of Alzheimer’s disease (AD), which is caused by aggregation of beta-amyloid in the brain parenchyma in neuritic plaques. Reference Greenberg, Bacskai, Hernandez-Guillamon, Pruzin, Sperling and van Veluw7 Although neuritic plaques usually accompany CAA, Reference Greenberg, Bacskai, Hernandez-Guillamon, Pruzin, Sperling and van Veluw7 most patients diagnosed with CAA-related hemorrhagic stroke do not have dementia. A study using the Informant Questionnaire on Cognitive Decline in the Elderly to diagnose pre-stroke cognitive impairment found that only 10 out of 49 patients with CAA-related ICH were significantly impaired. Reference Viswanathan, Patel and Rahman8 However, many survivors of CAA-related ICH have mild cognitive impairment, Reference Case, Charlton and Zwiers9 and they are at risk of transitioning to dementia. Reference Biffi, Bailey and Anderson10,Reference Moulin, Labreuche and Bombois11 A study found that decreased cerebrovascular reactivity and white matter damage accounted mediated about half of the risk for impairment in CAA; Reference Durrani, Wang and Cox12 the other half was not accounted for and could be mediated by concomitant AD.
Pathologies of small veins and venules have received much less attention than arterial pathologies but may also be important. Venous collagenosis is defined as thickening of the walls of cerebral venules and small veins by collagen deposition. Reference Moody, Brown, Challa and Anderson13 It is hypothesized to cause impaired vascular drainage resulting in increased periventricular tissue water content, visible as white matter hyperintensity (WMH) on T2-weighted MRI. Unfortunately, it is difficult to diagnose accurately during life because WMH are nonspecific, are associated with arterial pathology as well, and the veins affected by collagenosis are too small to be visualized. More research is needed, including prospective community-based studies with autopsy, on the associations of venous collagenosis with brain injury and dysfunction, and its clinical correlates.
It is important to recognize that infarction in the perfusion territory of a small artery is not synonymous with cerebral small vessel disease. Infarction can also result from embolism or thrombosis on an atheromatous plaque in the parent artery (also termed branch atheromatous disease). Reference Deguchi and Takahashi14
Clinical manifestations of CSVD
CSVD can cause ischemic stroke due to small vessel occlusion, intracerebral hemorrhage, or atraumatic convexity subarachnoid hemorrhage. Overall, CSVD causes about 25% of all ischemic strokes Reference Sacco, Marini, Totaro, Russo, Cerone and Carolei15 and more than 90% of intracerebral hemorrhages. Reference Raposo, Zanon Zotin and Seiffge16 However, the greatest impact of CSVD is on age-related cognitive, behavioral and gait decline in the absence of overt clinical stroke. Autopsy studies show that CSVD is present in the brains of 75% of people with dementia, 17 often in conjunction with other age-related pathologies such as AD. Reference Boyle, Yu and Leurgans18 CSVD accounts for about 25% of the risk for dementia, making it the second most common cause of dementia after AD. Reference Matthews, Brayne and Lowe3 CAA and AD are related and occur together more often than by chance, while arteriolosclerosis and AD are not associated with each other. Reference Nichols, Merrick and Hay19 In community-based autopsy studies the severity of amyloid plaques is not correlated with the severity of arteriolosclerosis. Reference Nichols, Merrick and Hay19 However, when both AD and cerebral small disease are present together their combined adverse effects on cognition are substantial. Reference Schneider, Boyle, Arvanitakis, Bienias and Bennett20
Radiological diagnosis of CSVD
Only the very largest of the small arteries, the lenticulostriate branches arising from the middle cerebral artery, can be visualized directly on MRI and, even then, only at high field strength. Reference Yashiro, Kameda and Chida21 Consequently, CSVD must be diagnosed during life based on indirect evidence. Because the clinical symptoms of CSVD – stroke, cognitive and behavioral decline, and slowed gait – are nonspecific and have other causes, the diagnosis of CSVD rests on characteristic patterns of tissue injury demonstrated by neuroimaging.
The Standards for Reporting Vascular Changes on Neuroimaging provide consensus terms and definitions for vascular brain changes caused by CSVD (Fig. 1). Reference Duering, Biessels and Brodtmann2 The most widely recognized lesions, which would typically be reported by radiologists on routine clinical imaging, are lacunes, white matter hyperintensities, microbleeds and cortical superficial siderosis. In most cases, no alternative cause of these lesions (Fig. 1) can be identified, and therefore they are presumed to be caused by CSVD. CSVD may also be diagnosed based on the occurrence of intracerebral hemorrhage or atraumatic convexity subarachnoid hemorrhage, after other competing causes (e.g., vascular malformations) have been investigated and excluded.
CSVD lesion types. Terms and definitions of these lesions can be found in the updated standards for reporting vascular changes on neuroimaging.Reference Duering, Biessels and Brodtmann2 CSVD = cerebral small vessel disease; DWI = diffusion-weighted imaging; PVS = perivascular space; RCVS = reversible cerebral vasoconstriction syndrome; WMH = white matter hyperintensity.

The Boston Criteria (Fig. 2) can be used to diagnose CAA based on characteristic patterns of hemorrhagic lesions and white matter features in patients with lobar ICH, cognitive decline, or CAA-related transient focal neurological episodes. Reference Charidimou, Boulouis and Frosch5 The criteria were recently re-derived to make a version ‘2.0’ and validated against neuropathology. The Boston Criteria 2.0 have good sensitivity and specificity (80% and 82%, respectively). Diagnostic accuracy against brain autopsy, the gold standard for CAA diagnosis is even higher in patients presenting with lobar ICH (sensitivity 90%, specificity 93%), probably because ICHs begin to occur at more severe stages of CAA. In contrast, in patients presenting with non-hemorrhagic manifestations of CAA the sensitivity of the Boston Criteria 2.0 is lower (55%), indicating that in this setting some patients who do not meet criteria nonetheless have CAA. This is probably because they have milder stage CAA, prior to the onset of the hemorrhagic features that are used for CAA diagnosis. In all scenarios in which the criteria were derived (lobar ICH, convexity SAH, cognitive decline, or TFNE) the criteria have a positive predictive value greater than 90%; i.e., patients meeting criteria are highly likely to have pathological evidence of CAA. However, caution is needed when extrapolating the criteria to persons without neurological symptoms as there are limited pathology data, and the positive predictive value is probably lower. For patients with lobar ICH who do not have an MRI, the computed tomography (CT)-based modified Edinburgh criteria can be used to rule in or rule out CAA with reasonable certainty in many patients. Reference Rodrigues, Samarasekera and Lerpiniere22
Boston criteria version 2.0. The criteria have been validated against neuropathological evidence of CAA, and in an external validation cohort had sensitivity of 80% and specificity of 82%.Reference Charidimou, Boulouis and Frosch5 CAA = cerebral amyloid angiopathy; CT = computed tomography; CMB: cerebral microbleed; cSS = cortical superficial siderosis; ICH = intracerebral hemorrhage; MRI = magnetic resonance imaging; PVS = perivascular space; SAH = subarachnoid hemorrhage; WMH = white matter hyperintensity.

Prevalence of CSVD lesions on neuroimaging with aging
The prevalence of CSVD lesions increases dramatically with aging (Fig. 3). Covert (i.e., clinically unrecognized) CSVD is the most common incidental finding on brain imaging in the elderly. Reference Vernooij, Ikram and Tanghe23 These lesions are not benign. Covert infarcts and WMH, in particular, have been associated with increased future risk for stroke and dementia, controlling for vascular risk factors and prior history of symptomatic stroke. Reference Debette, Schilling, Duperron, Larsson and Markus24 The relevance for routine clinical practice was recently demonstrated by a study that analyzed electronic health records from a large health maintenance organization and showed that patients with brain infarcts or WMH reported on their CT or MRI scans had 1.8–3.0 times higher risk for future stroke and 1.2–2.9 times higher risk for future dementia, adjusting for age and vascular risk factors. Reference Kent, Leung and Zhou25,Reference Kent, Leung and Zhou26
Prevalence of cerebral small vessel disease lesions by age decade. Because there are no meta-analyses, the prevalences shown here are approximate and are based on extrapolations from representative population-based studies. Reference Smith, O’Donnell and Dagenais62–Reference Shoamanesh, Akoudad and Himali67 For WMH the prevalence of beginning confluent or confluent WMH, equivalent to Fazekas grade 2 or 3, is provided. For PVS, the prevalence of grade 3 or 4 PVS according to the Wardlaw scale is provided. CMI = cortical microinfarct, cSS = cortical superficial siderosis, DWI+ = incidental DWI positive lesion detected on magnetic resonance imaging without symptoms, PVS = perivascular space, WMH = white matter hyperintensity.

Vascular and tissue consequences of CSVD
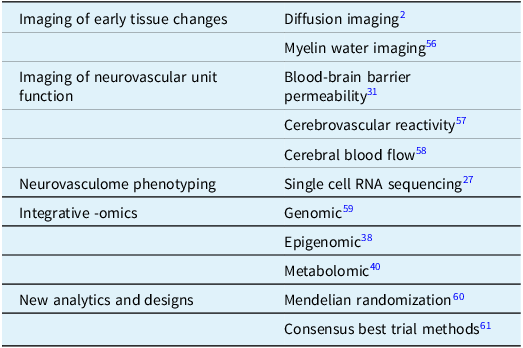
A more comprehensive picture of the evolution of CSVD is emerging, informed by epidemiological, physiological and quantitative neuroimaging studies, complemented by an enhanced understanding of the diversity of the neurovasculome and integrative multi-omic studies (Table 2).
Selected advanced research methods for investigating cerebral small vessel disease

In the most common form of CSVD, arteriolosclerosis results from collagen depositionin the vessel media in response to different types of insults, most commonly chronic hypertension. These histopathological changes are accompanied by disrupted functioning of the cells of the neurovasculome i.e., the multiple cell types that comprise the vascular tissues of the brain including endothelium, smooth muscle cells and pericytes. Reference Iadecola, Smith and Anrather27 These vascular cells are in very close proximity to the other cells of the neurovascular unit: astrocytes, neurons and microglia. Indeed, the pericytes and vascular smooth muscle cells are in direct contact with astrocytic end feet, which envelope the small vessels from the level of the penetrating arterioles to the ascending venules. Consequently, in addition to being a conduit for blood flow and maintaining hemostasis, the neurovasculome has been implicated in critical roles for trophic support of the neurovascular unit, immune cell trafficking and clearance of wastes. Reference Iadecola, Smith and Anrather27
Quantitative neuroimaging has improved our understanding of early vascular dysfunction in CSVD. Alteration in cerebrovascular reactivity is a measurable consequence of CSVD in living persons, occurs in both arteriolosclerosis Reference Blair, Doubal, Thrippleton, Marshall and Wardlaw28 and CAA Reference Beaudin, McCreary and Mazerolle29 , and is probably an early consequence. Reference van Opstal, van Rooden and van Harten30 Increased permeability of the blood brain barrier is another functional consequence that probably occurs at an early stage. Reference Thrippleton, Backes and Sourbron31 From there, tissue damage proceeds along two main pathways: increased permeability and hemorrhaging, and altered blood delivery and thrombosis. Reference Wardlaw, Smith and Dichgans32
Insights into the time course and progression of CSVD have come from rare monogenic disorders. By studying presymptomatic mutation carriers the early stages of CSVD development, prior to symptoms, can be investigated. These studies suggest that CSVD may evolve over 1–3 decades, with a long prodromal phase followed by late clinical manifestations. In cerebral autosomal dominant arteriopathy with subcortical ischemic leukoencephalopathy, migraine headaches typically being in the 20s or 30s, small subcortical infarcts in the 50s and vascular dementia in the 60s (Fig. 4). Reference Chabriat, Joutel, Dichgans, Tournier-Lasserve and Bousser33 Radiological manifestations of WMH begin even before any symptoms. In CAA, progression is proposed to occur in four stages, starting with asymptomatic vascular beta-amyloid deposition (Fig. 4). Reference Koemans, Chhatwal and van Veluw34 Hemorrhaging, which is the foundation of the diagnosis of probable CAA during life, occurs only in the last stage.
Progression of monogenic cerebral small vessel diseases. By studying mutation carriers before they develop symptoms, the pre-symptomatic and symptomatic phases of cerebral autosomal dominant arteriopathy with subcortical leukoencephalopathy (CADASIL) and Dutch-type hereditary CAA have been elucidated. In both monogenic diseases, asymptomatic radiological changes precede the onset of clinical symptoms by as much as 20–30 years. Sporadic age-related cerebral small vessel disease may have a similar lag time between disease onset and clinical symptoms, based on general population studies showing that MRI signs of covert cerebral small vessel disease begin to emerge in some persons in their 40s. Reference Smith, O’Donnell and Dagenais62 CMI = cortical microinfarct, cSS = cortical superficial siderosis, ICH = intracerebral hemorrhage, PVS = perivascular space, SAH = subarachnoid hemorrhage, TIA = transient ischemic attack, WMH = white matter hyperintensity.

It is tempting to posit that individuals with CSVD, hereditary or sporadic, progress in an orderly fashion through the different stages. However, clinical experience shows that there is variability in the order and progression of lesions, both across individuals and within the same brain within the same individual. For example, some people present with a single small subcortical infarct without WMH, some have microbleeds without other CSVD lesions and so on. This heterogenous progression of CSVD contrasts with the more orderly temporal and spatial evolution of some of the common neurodegenerative diseases of aging such as AD and Parkinson’s disease.
New clues to CSVD progression from multi-omics studies
The molecular basis of CSVD remains poorly understood. Gaining more understanding will be key to developing new therapeutic strategies.
Probably the least well understood lesion type is WMH. Radiologically, the appearance of WMH is nonspecific and simply indicates increased concentration of water in the affected voxels, either because the water content has increased or the non-water content has decreased (e.g. from loss of cells, axons, or myelin). Histopathologically, the main correlate is loss of myelin. Reference Humphreys, Smith and Wardlaw35 It is possible that there are multiple pathways to WMH, and that the pathogenesis of WMH varies across CSVDs or stages within a single CSVD. A little recognized fact, with important implications, is that vascular risk factors account for less than 15% of the variation in WMH volume. Reference Wardlaw, Allerhand and Doubal36,Reference Koohi, Harshfield and Markus37 (By contrast, vascular risk factors account for 70% of the variation in atherosclerosis burden). Reference Wardlaw, Allerhand and Doubal36 This strongly suggests a role for factors that are not yet discovered.
Clues to the origin of WMH are beginning to be found via large scale genomic, proteomic and metabolomic studies. WMH volume is highly heritable (more than 50%), suggesting that analysis of genetic variation may be fruitful. In the largest integrative genomic, transcriptomic, epigenetic and proteomic analysis to date, researchers of the NeuroCHARGE consortium identified two families of expressed genes that contributed to WMH variation: one family comprised genes related to vessel function (extracellular matrix organization, immune cytokine signaling, G-protein coupled receptor binding and smooth muscle contraction) and another family comprised genes involved in lipoprotein metabolism. Reference Yang, Knol and Wang38 Additional work using metabolomics has identified alterations in metabolites related to cell membrane and myelin components, further implicating myelin loss and repair as key processes in pathogenesis of WMH. Reference Smith39–Reference Harshfield, Sands, Tuladhar, de Leeuw, Lewis and Markus41 These promising avenues of research should be pursued, with the goal of identifying processes targetable by drugs.
Targets for CSVD treatment
There is little evidence to guide treatment of CSVD, with few trials. A guideline committee of the European Stroke Organization could make only one strong recommendation, for controlling blood pressure. Reference Wardlaw, Debette and Jokinen42
Many fundamental questions, such as whether aspirin should be given to patients with an incidentally discovered covert lacune, have not been answered. Controlling blood pressure is probably helpful, as demonstrated by an MRI substudy of the SPRINT trial which showed that intensive blood pressure control to a target of less than 120 mmHg systolic resulted in less increase in WMH Reference Nasrallah, Pajewski and Pajewski43 and reduced risk of mild cognitive impairment or dementia. Reference Williamson, Pajewski and Auchus44 This study shows that WMH progression is modifiable and confirms hypertension as a causal factor. However, as noted above, hypertension and other vascular risk factors account for only 15% of the variation in WMH volume in the population. Reference Wardlaw, Allerhand and Doubal36,Reference Koohi, Harshfield and Markus37 Progression of WMH in clinical trial cohorts, despite full application of conventional secondary stroke prevention, has been repeatedly described. Reference Cavalieri, Schmidt and Chen45 This suggests that there are probably other, undiscovered, pathways to WMH prevention that may be unique to CSVD and not blocked by treatment with antithrombotics, antihypertensives and statins.
Escalating the intensity of antithrombotics is probably not the answer to preventing stroke and functional decline in CSVD. MRI substudies of two clinical trials showed that the addition of either low dose or full dose rivaroxaban did not reduce the incidence of new small subcortical infarcts. Reference Sharma, Smith and Pearce46,Reference Sharma, Hart and Smith47 The Secondary Prevention of Small Subcortical Strokes (SPS3) clinical trial showed that the addition of clopidogrel to aspirin did not reduce the incidence of new strokes or cognitive decline. Reference Benavente, White and Pearce48,Reference Pearce, McClure and Anderson49 It is uncertain whether aspirin should be used in patients with covert brain infarcts or lacunes, and this should be tested in a future clinical trial.
The Framework for Clinical Trials in Cerebral Small Vessel Disease group has provided consensus recommendations for good practices for trials for CSVD, including precisely defining the CSVD type under study, using neuroimaging biomarkers for patient selection and for measuring surrogate outcomes in early phase trials, and selection of appropriate cognitive and functional endpoints. Reference Markus, van Der Flier and Smith61 Early phase, proof-of-concept trials that have recently been completed or are underway are testing a range of interventions including lifestyle modification, physical activity, neuroprotectants and vasodilating antihypertensives. Reference Smith and Markus50 The Canadian Consortium on Neurodegeneration in Aging is coordinating a pilot trial of the adherence and tolerability of remote ischemic conditioning for mild cognitive impairment due to CSVD. Reference Ganesh, Barber and Black51 In remote ischemic conditioning, blood flow is temporarily interrupted in a body part (in this case, the arm is rendered ischemic via inflation of a sphygmomanometer cuff) which conditions remote organs, including the brain, to protect themselves via induction of anti-inflammatory, anti-oxidant and anti-apoptotic pathways. Reference Hess, Blauenfeldt and Andersen52 Small trials have previously been conducted in patients with CSVD with some suggestion of WMH regression, Reference Mi, Yu, Ji, Sun and Qu53,Reference Wang, Meng and Song54 but more research is needed.
The recent phase 2 LACI-2 trial showed encouraging trends for the potential efficacy of isosorbide dinitrate and cilostazol in patients with clinical lacunar ischemic stroke. Reference Wardlaw, Woodhouse and Mhlanga55 LACI-2 used innovative secondary clinical endpoints, including a composite endpoint with cognitive impairment as one of the components (along with stroke, cardiovascular events and death) and a seven-level ordinal rating of cognitive function based on telephone Montreal Cognitive Assessment and functional scales (Barthel Index and modified Rankin scale). These promising new endpoints, integrating cognitive function with major adverse cardiovascular events and death, deserve more investigation.
Conclusions
CSVDs are among the most common age-related pathologies of the brain and are a significant cause of dementia and stroke. Arteriolosclerosis and CAA are the best recognized CSVDs. The clinical and radiological manifestations are diverse. The inability to visualize small arteries and veins directly makes research on them more difficult than for large arteries and veins. However, advances in quantitative imaging methods, including physiological imaging such as measurement of cerebrovascular reactivity and increased vascular permeability, are beginning to allow investigation of the early effects of CSVD in living people. Because the small vessels are in such close contact with other cells of the neurovascular unit, they are implicated in many unique functions of the brain. This makes CSVDs important targets for research on new mechanisms to promote brain health and prevent dementia.
Author contributions
Dr. Smith conceived of and wrote the article.
Funding statement
Dr Smith has received grant funding from the Canadian Institutes of Health Research, Brain Canada, Weston Brain Institute and Weston Family Foundation to study cerebral small vessel disease. The author wishes to acknowledge Aurora Smith for medical illustrations used in Figure 4. Dall-E (OpenAI, San Francisco) was used to generate clip art for Figure 4.
Competing interests
Dr Smith reports personal consulting for Alnylam Pharmaceuticals, Eisai and Eli Lilly.