Introduction
Extracellular polysaccharide substances (EPS) are complex polymers commonly produced by microbes (Reference DechoDecho, 2000) and composed mainly of neutral sugars with variable fractions of uronic acids, sulfates, amino sugars and proteins (Reference Nichols, Lardière, Bowman, Nichols, Gibson and GuézennecNichols and others, 2005). Some of these polymers are tightly bound to the microbial cell, whereas others are loosely attached, forming a more dispersed ‘slime’ (Reference Decho and BarnesDecho, 1990). Among many roles, EPS act as ligands for metal cations, participate in the formation of marine aggregates and contribute to biogeochemical cycles (Reference PassowPassow, 2002; Reference Nichols, Lardière, Bowman, Nichols, Gibson and GuézennecNichols and others, 2005). Regarding sea ice, EPS produced by algae and bacteria have been considered to provide cryoprotection within the ice matrix (Reference Krembs, Eicken, Junge and DemingKrembs and others, 2002; Reference Collins, Carpenter and DemingCollins and others, 2008; Reference Marx, Carpenter and DemingMarx and others, 2009), as well as a mechanism for organisms, particularly EPS-coated algae, to entrain selectively into the ice (Reference Meiners, Gradinger, Civitarese and SpindlerMeiners and others, 2003; Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007) and remain anchored within it (Reference Krembs, Deming, Margesin, Schinner, Marx and GerdayKrembs and Deming, 2008). Here we consider, via controlled laboratory tests, the entrainment of bacterial EPS into growing sea ice.
During the freezing process, sea ice retains a fraction of the microbial organisms, particles, salts and other solutes present in the source water; they are retained within liquid inclusions in the ice, brine-filled pores and channels (Reference Petrich, Eicken, Thomas and DieckmannPetrich and Eicken, 2010) which constitute the habitable portion of the ice (Reference Junge, Krembs, Stierle and EickenJunge and others, 2001). A larger fraction of solutes and particles, though, is expelled back into the water column. the proportion by which solutes are retained in sea ice can be described by ‘effective segregation coefficients’, the ratio between the concentration of solutes in the ice and the concentration of solutes in the source liquid from which the ice forms (Reference Eicken, Thomas and DieckmannEicken, 2003). Effective segregation coefficients, k eff, were initially defined for salts in sea water:
where S ice is the salinity of the melted ice (bulk salinity) and S source is the salinity of sea water away from the ice–water interface. Extending this definition to other solutes allows, for example, calculation of the segregation coefficient of dissolved exopolymers:
where [EPS]ice is the concentration of exopolymers in the melted ice and [EPS]source is the concentration of exopolymers in the source solution.
Solutes in sea water will segregate proportionally to dissolved salts unless they interact with the ice crystals, in which case the segregation of the interacting solute will diverge from the segregation of the salts. To assess this divergence, segregation coefficients of different solutes can be normalized to generate an enrichment index (Reference GradingerGradinger and Ikävalko, 1998), defined as:
where k effx is the segregation coefficient of the solute x. If I s = 1, the solute and the salts are expelled in the same proportion. If I s > 1, the solute is selectively retained (enriched) in the ice. In ocean environments that experience seasonal freezing, enrichment indices have been calculated from measurements of the concentrations of particles, cells and EPS in the ice and in underlying sea water (Reference Garrison, Close and ReimnitzGarrison and others, 1989; Reference Meiners, Gradinger, Civitarese and SpindlerMeiners and others, 2003; Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007).
The enrichment mechanism for organic ‘impurities’ (including microbes) present in sea water has usually been ascribed to a non-selective physical process, whereby suspended ice crystals, known as frazil ice, rise through the water column to consolidate an ice layer at the surface and collect particulate matter indiscriminately as they ascend (Reference Garrison, Ackley and BuckGarrison and others, 1983, Reference Garrison, Close and Reimnitz1989). Evidence for a selective mechanism in the enrichment of particles and microorganisms, affected by cell size and possibly EPS coatings, comes from enrichment indices calculated for a variety of organisms in new Arctic sea ice (Reference GradingerGradinger and Ikävalko, 1998; Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007) and for bacteria and algae in artificial sea ice (Reference Grossmann and GleitzGrossmann and Gleitz, 1993).
Algae have been reported early and often as enriched in sea ice (Reference Grossmann and GleitzGrossmann and Gleitz, 1993; Reference GradingerGradinger and Ikävalko, 1998; Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007), with larger algal cells (>5 μm) presenting a higher average index (I s = 63.0) than smaller algal cells (I s = 4.6) or bacteria (I s = 5.1) as reported in the most recent study (Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007). Reference Meiners, Gradinger, Civitarese and SpindlerMeiners and others (2003) reported an exception for pennate diatoms in nilas ice (I s = 0.5).
Bacteria, on the other hand, were not reported as enriched in early studies of this subject (Reference Grossmann and GleitzGrossmann and Gleitz, 1993; Reference Grossmann and DieckmannGrossmann and Dieckmann, 1994), and, when enrichment was reported later, the indices were always lower than those of algae (Reference GradingerGradinger and Ikävalko, 1998; Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007). As a result, the process of bacterial enrichment has been attributed to their pre-existing associations with larger cells (Reference Grossmann and GleitzGrossmann and Gleitz, 1993; Reference Grossmann and DieckmannGrossmann and Dieckmann, 1994), with algal EPS (Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007) or with exopolymer particles (EPS >0.4 μm, of indeterminate origin; Reference Meiners, Gradinger, Civitarese and SpindlerMeiners and others, 2003). In the cases where bacterial enrichment was not observed, the bacterial population of the ice had been inferred from the bacterial population measured in brines collected by centrifugation (as opposed to bulk sea-ice melting), an approach that would bias results towards a lower enrichment index if bacteria had remained within the ice matrix. the latter scenario is plausible given that some bacteria produce ice-affine substances that remain attached to the ice fraction after brine removal by centrifugation (Reference Raymond and FritsenRaymond and Fritsen, 2000).
EPS have also been reported as enriched in sea ice, with the average index for particulate EPS (>0.4 μm, of indeterminate origin) being of intermediate value and correlating positively with the enrichment of both algal cells and bacteria (Reference Meiners, Gradinger, Civitarese and SpindlerMeiners and others, 2003; Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007). These results have suggested that cellular coatings of EPS or EPS particles may play a role in the selective entrainment (or retention) of at least some microorganisms in the ice (Reference Meiners, Gradinger, Civitarese and SpindlerMeiners and others, 2003; Reference Riedel, Michel, Gosselin and LeBlancRiedel and others, 2007). the enrichment of dissolved organic materials, also observed in new sea ice (Reference Giannelli, Thomas, Haas, Kennedy and DieckmannGiannelli and others, 2001; Reference ThomasThomas and others, 2001), suggests that ‘dissolved’ EPS (<0.4 μm) not yet examined in this way may have a similar fate.
The high enrichment indices for particulate EPS and (assumed) EPS-coated cells may be due to interactions between the extracellular polymers, or some component of them, and the ice crystals. Changes in ice-crystal morphology have been associated with the interaction between ice-affine molecules, particularly proteins, and ice crystals (Reference Raymond and FritsenRaymond and Fritsen, 2000). Sea-ice growth experiments in which the pore morphology was significantly altered in ice enriched with algal EPS relative to EPS-free ice (Reference Krembs, Deming, Margesin, Schinner, Marx and GerdayKrembs and Deming, 2008; C. Krembs and others, unpublished information) thus provide support for linking ice affinity to ice enrichment. In these studies, the ice-altering property of algal EPS has been ascribed to a glycoprotein component, although ice activity has also been recently observed in polysaccharides with no detectable protein fraction (in xylomannans from insects; Reference Walters, Serianni, Sformo, Barnes and DumanWalters and others, 2009).
Perhaps because the majority of EPS found in polar sea ice is produced by algae (Reference Krembs, Eicken, Junge and DemingKrembs and others, 2002; Reference Meiners, Gradinger, Civitarese and SpindlerMeiners and others, 2003; Reference Riedel, Michel and GosselinRiedel and others, 2006), few studies have addressed the potential role of bacterially produced EPS in the entrainment of these smaller microorganisms into sea ice. Information from other (warm) environments, however, shows that bacterial EPS can bind directly to mineral surfaces, altering their crystalline structures, or can develop strong complexes with inorganic ions, preventing mineral formation. In the former case, evidence suggests that a protein component of the EPS mediates the EPS–mineral interaction (Reference Kawaguchi and DechoKawaguchi and Decho, 2002; Reference PerryPerry and others, 2005). As an example of the latter case, the presence of bacterial EPS inhibited calcium carbonate precipitation (Reference Dupraz, Reid, Braissant, Decho, Norman and VisscherDupraz and others, 2009), an EPS–mineral interaction that, if translatable to subzero temperatures in brine, may be significant to the fate of CO2 in sea ice (Reference Delille, Jourdain, Borges, Tison and DelilleDelille and others, 2007; Reference Rysgaard, Glud, Sejr, Bendtsen and ChristensenRysgaard and others, 2007).
On the other hand, some bacteria are known to produce proteins that interact at the molecular level with ice crystals (Reference Wilson, Kelley and WalkerWilson and others, 2006; Reference Raymond, Fritsen and ShenRaymond and others, 2008), and in non-saline laboratory systems the selective partitioning of organic solutes into ice has been used to indicate ice affinity in proteins (Reference MarshallMarshall and others, 2004). of particular relevance to this study is the recent isolation of an ice-binding protein (IBP) from the spent culture medium of an Antarctic sea-ice bacterium of the genus Colwellia (Reference Raymond, Fritsen and ShenRaymond and others, 2007), suggesting that EPS produced by cold-adapted marine bacteria may include a protein fraction interactive with ice crystals that might contribute to the retention of bacterial cells in sea ice.
Here we propose that bacteria may be enriched in sea ice not only by their interaction with algae or algal EPS but also by the interaction of their own EPS with growing sea-ice crystals. To address this hypothesis, we examined the retention in artificial sea ice of EPS produced by the cold-adapted marine gammaproteobacterium, Colwellia psychrerythraea strain 34H (hereafter strain 34H), a genus and species often found in cold sea water and sea ice (Reference Deming, Thomas and DieckmannDeming, 2010). In a departure from studies of particulate EPS, we examined the ‘dissolved’ (<0.4 μm) fraction of EPS. the goal was to determine whether or not these EPS are selectively retained in the ice and, if so, whether that selective retention is attributable to a specific component of the EPS. We tested this hypothesis by determining and comparing the enrichment indices of three sources of EPS: EPS present in the spent culture media of strain 34H; a split of that same EPS first subjected to heating to denature its heat-labile components; and EPS present in the uninoculated (also pre-heated) culture medium (derived from yeast extract). We also measured temperature and volume of these experimental ices for an initial investigation of potential physical alteration of the ice by EPS of bacterial origin.
Methods
Ice was grown in triplicate experiments, completely randomized, from three source saline solutions containing high molecular weight polymers of different origin (Fig. 1). To obtain the first source solution, hereafter called native exopolymers or simply exopolymers, Colwellia psychrerythraea strain 34H was grown at 2˚C, with shaking, in Marine Broth 2216 (Difco Laboratories) diluted to half organic strength with artificial sea water (ASW) as in Reference Marx, Carpenter and DemingMarx and others (2009), until the culture reached maximum optical density (0.74 at A600). ASW contained the four major sea-water salts (0.4M NaCl, 9mM KCl, 26mM MgCl2, 28mM MgSO4) and KH2PO4 buffer (salinity 32, pH 7). A cell-free solution of exopolymers was obtained from the batch culture by low-speed centrifugation (20 min, 3000g, 2˚C) to pellet most of the cells, and filtration of the recovered supernatant (0.45 μm filter, Millipore-HAWG 047 00) to remove cells remaining in suspension, according to Reference Huston, Methe and DemingHuston and others (2004), then stored at 2˚C until use. This gentle method for cell removal, validated microscopically during protocol development, reduces the risks of cell lysis, leakage of intracellular molecules or disruption of macromolecules, collecting only the loosely attached exopolymers but not the capsular EPS tightly bound to the cells (Reference Nielsen, Jahn, Wingender, Neu and FlemmingNielsen and Jahn, 1999). Furthermore, as the filtration step excludes what is typically considered the particulate fraction of EPS, the EPS examined here corresponds to the dissolved fraction of loosely attached 34H exopolymers.
Preparation of source solutions: exopolymers: cell-free supernatant of the spent culture medium after growth of Colwellia psychrerythraea strain 34H; HT exopolymers: a split of the native exopolymer solution, heated to 90˚C for 10 min; and broth: uninoculated culture medium containing (autoclaved) polymers from yeast extract. Prior to use, all solutions were dialysed (3500 MWCO).

The second source solution, called heat-treated (HT) exopolymers, was a split of the first solution subjected to 90˚C for 10 min (before storage at 2˚C). These heating conditions denature the secondary structure of proteins (even in the presence of hydrocolloids; Reference IbanogluIbanoglu, 2005), but should not affect the concentration of polysaccharides given the short exposure time (according to rate constants of depolymerization for polysaccharides in Reference Lai, Lii, Hung and LuLai and others, 2000). the third source solution, called broth, was a split of the uninoculated medium used to culture strain 34H. This Marine Broth 2216, diluted to half organic strength, had been autoclaved (protein-denaturing conditions) prior to use. All three source solutions were dialysed with pleated dialysis tubing (3500 MWCO) in ASW for 2 days at 4˚C to ensure that all polymers tested were above a minimum size. After dialysis, 200mL samples of the solutions were stored frozen at –15˚C until use.
Ice was generated using a cold-finger apparatus (Fig. 2a), first described by Reference Kuiper, Lankin, Gauthier, Walker and DaviesKuiper and others (2003) for recovering ice-affine proteins from freshwater solutions, modified for use with saline solutions (corrodible metal parts were replaced with glass). the apparatus consisted of a glass tube connected to a circulating bath that kept antifreeze in the tube at the desired temperature, which in this study was –5˚C (Fig. 2a) as representative of sea-ice-forming conditions in the fall. Samples were placed in a 250 mL beaker immersed in a saline-water ice bath (salinity = 30) that kept the external temperature at –1.5˚C. For each experiment the cold finger was placed into the sample, and ice allowed to form, for a period of 6 hours. the temperature of the unfrozen source solution was monitored during this period (Fig. 2b). Experiments were conducted under sterile conditions and using aseptic technique to the extent possible in a 4˚C cold room, which helped to maintain reproducible experimental conditions.
Operation of the cold-finger apparatus: (a) the apparatus, as modified from Reference Kuiper, Lankin, Gauthier, Walker and DaviesKuiper and others (2003) for use with saline solutions; (b) cold finger placed into source solution for 6 hours, with temperature of the solution monitored during ice growth; (c) formed ice removed; and (d) ice immediately photographed from three angles, then allowed to melt at room temperature.

At the end of each experiment, the cold finger was removed from the remaining liquid solution (Fig. 2c) and the ice was released from it and immediately photographed from three different angles (Fig. 2d): the top (Fig. 3a) and two sides (Fig. 3b and c). the top photograph was used to verify circularity. the contours of the lateral photographs were extracted manually using an open-source vector graphics editor (Inkscape), and the volume was integrated using circular slabs of 0.125 cm thickness (Fig. 3d). To account for the small asymmetries in the ice hemispheres evidenced by the top photograph, the volumes calculated from the two lateral photographs were averaged. the volume occupied by the cold finger (2.4cm3), calculated from measured dimensions, was subtracted from the total.
Calculation of ice volume by photographic approach: (a) the formed ice hemisphere photographed from the top, with axis perpendicular to underlying surface; (b) the ice photographed from the side, with axis parallel to underlying surface; (c) the ice photographed from the side after a 90˚C rotation; and (d) extrapolation of the ice volume from the contour of the hemisphere, integrating the volumes of slabs 0.125 cm wide (and subtracting the volume occupied by the cold finger itself). Bars indicate 1.25 cm.

The ice was then allowed to melt at room temperature. Bulk salinity, volume and EPS content were determined for both the melted ice and the liquid solution that had remained unfrozen throughout the experiment. Salinity was measured at room temperature with a hand-held YSI-30 conductivity meter (accuracy of ±0.1 psu). EPS was measured as total concentration of hexoses, expressed as glucose equivalents, by the phenol sulphuric acid assay, as described by Reference Dubois, Gilles, Hamilton, Rebers and SmithDubois and others (1956). To determine whether or not the liquid inclusions of the ice we grew contained exopolymers disproportionately to the salt also retained, we calculated the segregation coefficients, k effs and k effe, and the enrichment index, I s, for EPS.
Results and Discussion
Because growing ice rejects the salts present in a source solution, k effs should always be <1.0. In our experiments, we observed an average value of 0.39±0.01 (± standard error, n = 3) for k effs in all three treatments (Table 1). There was no significant difference among treatments (p = 0.88, Kruskall–Wallis test, nonparametric test for three independent variables), indicating that salts were rejected similarly from the ice grown in all three cases.
Salinity (mean±S.E., n = 3) of EPS-containing source solutions at the start of an ice-growing experiment, of the ice formed after 6 hours (melted for analysis), and of the unfrozen solution remaining at 6 hours, with calculated segregation coefficents for the salt (k effs)

If EPS in the source solution are expelled from the growing ice in the same proportion as the salts, then k effe should be similar to k effs; if EPS are retained preferentially in the ice, then k effe should be greater than k effs. Our experimental results show that k effe for the HT exopolymers (0.38±0.1) and the broth (0.40±0.1) were similar to k effs (0.39±0.01), indicating no preferential retention of EPS. In contrast, the native (unheated) bacterial exopolymers had a higher k effe of 0.63±0.1 (Table 2), indicating their tendency to be retained in the ice. Using a Mann–Whitney U test (nonparametric test for small datasets, to compare two independent variables), we determined that the k effe values for the HT exopolymers and the broth were not significantly different from each other (p = 0.83), indicating that these two types of exopolymers segregated from the growing ice in a similar way. the k effe of the native exopolymers, however, differed from the k effe of the HT exopolymers and the broth (p = 0.13), indicating with an 87% confidence level that the native bacterial exopolymers segregated differently in the saline ice than either the HT or the broth exopolymers.
EPS concentration (mean±S.E., n = 3) in source solutions at the start of an ice-growing experiment, in ice formed after 6 hours, and in solution remaining at 6 hours, with calculated segregation coefficients for exopolymers (k effe) and enrichment indices for exopolymers relative to salt (I s)

The selective retention of EPS is quantified by I s (Table 2), with I s > 1 indicating selective retention. Neither the I s values for EPS in the HT exopolymers (I s = 0.97±0.23) nor the broth (I s = 1.02±0.14) were significantly greater than 1.0 (one-tailed t test, 57% level, p = 0.43). the only enrichment index that could be considered greater than 1 (with a confidence level of 89%, p = 0.11) was for the native bacterial exopolymers (I s = 1.61±0.30).
From these results we suggest that the loosely attached EPS produced by strain 34H can be retained preferentially in sea ice and that such retention is attributable to a heat-labile fraction of the EPS. If the selective retention of EPS from strain 34H can be generalized, this ice-adaptive characteristic could be exploited in the environment. Bacteria may use their EPS to associate directly with frazil ice crystals as they rise through the water column, or to be retained in the sea ice once formed, without depending on an association with algae, other larger particles or algal EPS. In a laboratory setting, selective retention of EPS may be exploited to develop physical isolation methods of such polymers (e.g. by freeze selection) without altering their chemical properties, as has been done for proteins (Reference Kuiper, Lankin, Gauthier, Walker and DaviesKuiper and others, 2003).
The heat-labile fraction of the exopolymers responsible for their selective retention in the ice may correspond to a protein fraction with ice-affinity properties. EPS from Antarctic bacterial isolates (near relatives of our test organism) have been shown to contain variable levels of protein, in some cases reaching 50% of the total EPS composition (Reference Nichols, Lardière, Bowman, Nichols, Gibson and GuézennecNichols and others, 2005). Furthermore, IBPs have been described for a number of cold-adapted microorganisms including sea-ice bacteria (Reference Raymond, Fritsen and ShenRaymond and others, 2007), glacial-ice bacteria (Reference Raymond, Christner and SchusterRaymond and others, 2008), sea-ice diatoms (Reference Janech, Krell, Kang and RaymondJanech and others, 2006) and snow molds (Reference HoshinoHoshino and others, 2003); the IBP domain of the protein is similar in all cases. Because Colwellia psychrerythraea strain 34H does not have a gene encoding this IBP (Reference Raymond, Fritsen and ShenRaymond and others, 2007), the biochemical basis for the detected EPS enrichment is not yet clear. If the heat-labile fraction responsible for the selective retention of the native exopolymers from strain 34H is a protein with ice affinity, it may represent a different class of IBP not yet identified genomically for strain 34H (Reference MethéMethé and others, 2005). the ice-affinity property may also be dependent on the polysaccharide material itself, as happens for the thermal hysteresis-producing antifreeze from the freeze-tolerant beetle Upis ceramboides (Reference Walters, Serianni, Sformo, Barnes and DumanWalters and others, 2009).
An alternative explanation for the observed segregation coefficients is that exopolymers from the various sources were differentially expelled from the ice due to (1) differences in size or three-dimensional configuration of the polymers, or (2) changes in the structure of the ice matrix (e.g. more tortuous channels) due to interaction between the heat-labile component in the native exopolymers and the ice crystals. Our dialysis step, however, ensured that all solutions contained a similar concentration and size spectrum of polymers, all above 3500 Da (Table 2). Although we cannot rule out the possibility that differences in the three-dimensional configuration of the polymers influenced polymer expulsion from the ice, if changes in the physical microstructure of the ice contributed to a differential expulsion or retention of the polymers, those changes themselves would be attributable to the presence of ice-interactive polymers.
As a final point, molecules with surface affinity can modify the structure of the crystals to which they attach (Reference Kawaguchi and DechoKawaguchi and Decho, 2002; Reference PerryPerry and others, 2005). In particular, ice-binding proteins produced by several different microbes, including sea-ice bacteria, are known to modify the structure of ice crystals (Reference Raymond, Fritsen and ShenRaymond and others, 2007; Reference Kawahara, Margesin, Schinner, Marx and GerdayKawahara, 2008). A similar function for bacterial extracellular polysaccharides is not yet clear, but there is evidence to suggest that ice-algal EPS contain a glycoprotein fraction responsible for altering the pore morphology of sea ice (C. Krembs and others, unpublished information). To address the potential alteration of sea-ice properties by native exopolymers from strain 34H, we examined two physical parameters in our experiments: the change in temperature of the source solution over the course of the ice-growing period, reflecting the temperature gradient in the ice, and the volume of ice that formed. Although more detailed work is needed, both sets of results suggest that preferential retention of bacterial exopolymers may be associated with an increase in the liquid fraction of the ice.
Specifically, the temperature change in the source solution over the 6 hour experiment (Table 3) was similar (agreed within errors) between the broth and HT exopolymer solutions (p = 0.57, Mann–Whitney U test) but was lower for the native exopolymers (p = 0.13, Mann–Whitney U test). the smaller change in the solution of native exopolymers suggests a stronger temperature gradient through the ice, which can be explained by a lower thermal conductivity due to an increased liquid fraction (liquid brine has lower thermal conductivity than frozen water; Reference Pringle, Ingham, Eicken, Gradinger, Shirasawa and PerovichPringle and Ingham, 2009). To evaluate alternative explanations (e.g. convection in the brine pores) would require detailed information on differences in ice–brine distribution.
Change in temperature of source solutions (mean±S.E., n = 3) over 6 hours (ΔT = | T 6 – T 0|) and volume of ice (mean±S.E., n=3) grown by 6 hours. Temperature was measured while the cold finger was in the solution. Volume of melted ice was measured at room temperature; volume of unmelted ice was extrapolated from photographs taken immediately upon removal of the ice from the source solution
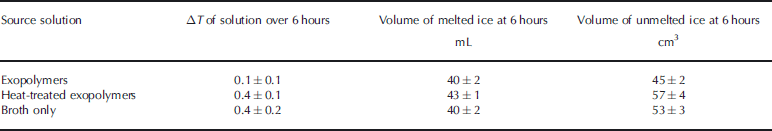
Lower thermal conductivity is also associated with higher ice density (Reference Pringle, Ingham, Eicken, Gradinger, Shirasawa and PerovichPringle and Ingham, 2009). Although we did not measure the density of the ice directly, comparison of our ice volume measurements (bulk and unmelted) suggests a higher density in the ice formed from the source solution of native exopolymers. the bulk volume (pore water plus melted ice) of the melted ice (Table 3) agreed to within errors (p = 0.49, Kruskal–Wallis test) for the three test solutions. Because ice is less dense than water, we expected the volume of the unmelted ice, determined photographically, to be uniformly greater than the volume of melted ice. the photographic estimates, however, indicated differences between the native exopolymer experiment (smaller volume) and the other treatments (p = 0.05, Mann–Whitney U test), with no difference between the HT and broth exopolymers (p = 0.83, Mann–Whitney U test) (Table 3). Although formation of higher-density ice from the solution of native exopolymers accounts for this difference, confirming that the results represent differences in the liquid fraction of the ice requires more detailed work.
Overall, the selective retention of native exopolymers produced by strain 34H and their apparent association with physical changes in the ice suggest that bacteria, like algae, have the ability to modify the sea-ice environment and make it more habitable by increasing its liquid fraction. This finding agrees with previous evidence that algal EPS increases sea-ice habitability on the microscopic scale (Reference Krembs, Deming, Margesin, Schinner, Marx and GerdayKrembs and Deming, 2008) and that bacterial production of EPS in winter sea ice, in the absence of ice algae, serves a cryoprotectant function (Reference Collins, Carpenter and DemingCollins and others, 2008). Although algal EPS production dominates during the biologically productive seasons in polar regions and thus may be expected to mediate physical alterations of sea ice on a larger scale, bacterial EPS production may nevertheless alter the physical properties of sea ice on a scale beneficial to these smallest of organisms that live throughout the ice in all seasons.
Conclusions
When artificial sea ice was formed from a solution containing dissolved native EPS from Colwellia psychrerythraea strain 34H, the EPS tended to be retained selectively in the ice compared to the salts also entrained (I s = 1.6). the ability to be retained in the ice was lost if the exopolymers were first heated. This ability was also absent from other polysaccharides present in the culture medium used to grow strain 34H. We interpret these results as an ice-affinity property of the EPS likely due to a protein component, though the nature of that component is not yet identified and changes in the three-dimensional structure of the exopolymers cannot be ruled out. Our results also suggest differences in the liquid fraction of the ice formed from the solution of native bacterial exopolymers, but more work is needed to understand in detail the effects of bacterial EPS on the physical properties of ice and their specific roles as cryoprotectants and possibly osmoprotectants. the selective retention of bacterial EPS has implications for the segregation and retention of microorganisms in ice and for beneficial habitat alteration on the microscale. It may also contribute to developing methods for isolating chemically unaltered exopolymers for applications in cryopreservation or bioremediation.
Acknowledgements
This research was supported by a Washington State Sea Grant award NAO70AR4170007 to J.W. Deming and a Vatican Observatory Fellowship to M. Ewert. We thank the University of Washington School of Oceanography for additional support, Z. Adams and S. Carpenter for valuable assistance in the laboratory, D. Grunbaum, J. Baross, R. Woodgate and B. Light for critical discussion, and two anonymous reviewers for their constructive comments and suggestions.







