Introduction
The puma Puma concolor is a large felid that has a wide distribution in the American continent and is found in almost the entire territory of Argentina (De Angelo et al., Reference De Angelo, Llanos, Guerisoli, Varela, Valenzuela and Pía2019). Although it is declining in some parts of its range, the puma is categorized as Least Concern both globally (Nielsen et al., Reference Nielsen, Thompson, Kelly and Lopez-González2015) and in Argentina (De Angelo et al., Reference De Angelo, Llanos, Guerisoli, Varela, Valenzuela and Pía2019). Because of their adaptability, pumas occur in a wide variety of environments (Nowell & Jackson, Reference Nowell and Jackson1996) and are relatively tolerant of human-modified habitats (Nielsen et al., Reference Nielsen, Thompson, Kelly and Lopez-González2015; Guerisoli et al., Reference Guerisoli, Luengos Vidal, Franchini, Caruso, Casanave and Lucherini2017). However, their large territorial requirements and dependence on the availability of prey make them sensitive to anthropogenic disturbances (Quiroga et al., Reference Quiroga, Noss, Paviolo, Boaglio and Di Bitetti2016). Like most carnivores, pumas are mainly nocturnal and crepuscular, but human presence and the behaviour of their prey may affect their activity patterns (Sweanor et al., Reference Sweanor, Logan, Bauer, Millsap and Boyce2008; Paviolo et al., Reference Paviolo, Di Blanco, De Angelo and Di Bitetti2009). Additionally, anthropogenic threats create a so-called landscape of fear (or landscape of co-existence, sensu Oriol-Cotterill et al., Reference Oriol-Cotterill, Valeix, Frank, Riginos and Macdonald2015), where pumas tend to avoid people (Suraci et al., Reference Suraci, Smith, Clinchy, Zanette and Wilmers2019) and areas close to human infrastructure (Guerisoli et al., Reference Guerisoli, Caruso, Luengos Vidal and Lucherini2019).
In Argentina the transformation of natural environments into farmlands, the introduction of livestock and the decline in wild prey have led pumas to prey on livestock and, consequently, to the killing of pumas by farmers. In southern Buenos Aires Province, human–carnivore conflict is intense, especially between pumas and sheep ranchers (Luengos Vidal et al., Reference Luengos Vidal, Guerisoli, Caruso, Casanave and Lucherini2016). Although pumas are protected by the laws of Buenos Aires Province, their killing remains the most widespread solution to conflicts in this area (Guerisoli et al., Reference Guerisoli, Luengos Vidal, Franchini, Caruso, Casanave and Lucherini2017). However, lethal control is usually economically inefficient, requires ongoing effort and resources, frequently fails to eliminate specific problem individuals, and offers limited long-term effectiveness (Conover, Reference Conover2001; Rigg et al., Reference Rigg, Finďo, Wechselberger, Gorman, Sillero-Zubiri and Macdonald2011; Miller et al., Reference Miller, Stoner, Cejtin, Meyer, Middleton and Schmitz2016). In addition, the removal of a puma can facilitate the influx of new individuals, potentially increasing the local predator population and the risk of depredation and, ultimately, deepening tensions with ranchers (Crooks & Soulé, Reference Crooks and Soulé1999; McManus et al., Reference McManus, Dickman, Gaynor, Smuts and Macdonald2015). Although several approaches can be used to limit damage from wildlife, empirical evidence indicates that the implementation of strategies requires local evaluation (Dickman, Reference Dickman2010; Treves et al., Reference Treves, Krofel and McManus2016). Recently, ranchers in southern Buenos Aires Province started using livestock guarding dogs. Guarding dogs have been used for at least 6,000 years and can be very effective at reducing predation by carnivores (Miller et al., Reference Miller, Stoner, Cejtin, Meyer, Middleton and Schmitz2016; Bergstrom, Reference Bergstrom2017; Novaro et al., Reference Novaro, González, Pailacura, Bolgeri, Hertel, Funes and Walker2017) and increasing ranchers’ tolerance of large carnivores (Potgieter, et al., Reference Potgieter, Kerley and Marker2016). The dogs are raised with the flock, allowing them to establish a close bond with it and the urge to protect it (Bidinost et al., Reference Bidinost, Gaspero, Castillo, Villar, Garramuño and Bruno Galarraga2016). Supposedly, guarding dogs do not attack predators but rather intimidate or scare them away through non-lethal means. These include active behaviours such as barking and confronting intruders, as well as passive signals such as territorial scent marking (Allen et al., Reference Allen, Stewart-Moore, Byrne and Allen2017; van Bommel & Johnson, Reference van Bommel and Johnson2017). In direct encounters, intimidation through approach and vocalizations have been shown to dissuade predators such as wolves Canis lupus (Landry et al., Reference Landry, Borelli and Drouilly2020), highlighting the importance of perceived risk in the predator’s decision-making process. Even though ranchers appear to be satisfied by guarding dog performances, a sound understanding of the effectiveness of this tool and its effect on both predators and prey is required for its proper evaluation and to design long-term management plans based on the use of livestock guarding dogs (van Bommel & Johnson, Reference van Bommel and Johnson2016; Bromen et al., Reference Bromen, French, Walker, Silvy and Tomeček2019).
In this study, we assess the spatiotemporal dynamics of the puma in an agro-ecosystem in central Argentina, where a livestock guarding dog was used to mitigate conflict with carnivores. We expected that (1) pumas would avoid areas with high activity levels of people and dogs, and prefer sites with high natural vegetation cover, (2) segregation of the temporal activity patterns of pumas and the dog and the sheep it protected, and (3) a puma visit to a site would trigger a greater exploratory response by the dog than that by pumas in response to a dog visit, especially in the days immediately following the puma visit.
Study area
Fieldwork was carried out at Chacra Experimental Patagones, a 1,212 ha ranch in the Argentinean Espinal ecoregion of central Argentina (Fig. 1). The Chacra is primarily used for raising c. 800 sheep, which generally feed on planted pastures and crops near the main house (48.6% of the total area, 590 ha), are kept as a single flock, and are confined in corrals at night during lambing season. The property is divided into fenced paddocks, and the flock is rotated across them depending on forage availability.
The study area on a ranch in Patagones county, Buenos Aires Province, central Argentina.

The Argentinean Espinal ecoregion is mostly flat, at an average elevation of 700 m. Climate is temperate and semiarid (Fernández et al., Reference Fernández, Gil and Distel2009), with a mean annual temperature of 15.3 °C and mean total annual precipitation of 350–500 mm, concentrated in spring and autumn (Cabrera & Willink, Reference Cabrera and Willink1980). The natural vegetation includes xerophytic deciduous woodlands dominated by Neltuma caldenia, grassland prairies and prairies interspersed with extensive patches of scrubland (Cano & Movia, Reference Cano and Movia1967). Overgrazing, logging, the transformation of natural forests into grasslands and crops, and poor livestock management are threatening ecological processes, the diversity of species and the economic productivity of the area (Distel, Reference Distel2016).
Within this matrix, there are three main habitat types. Scrubland has the greatest coverage; it includes native woody species such as Geoffroea decorticans, Condalia microphylla, Neltuma flexuosa and Larrea divaricate. Grasslands are under light grazing or enclosed, dominated by native grasses such as Stipa clarazii, Poa ligularis, and Stipa tenuis and Piptochaetium napostaense in degraded areas. Pastures and crops include rotational cultivation areas with perennial forage (Thinopyrum ponticum, Vicia villosa) and winter crops (Hordeum vulgare, Avena sativa).
The region is characterized by sheep and cattle farming practiced both extensively and intensively. Since 2017, the Chacra has had a livestock guarding dog of the Maremmano breed, intended to reduce carnivore predation on sheep. The dog was a neutered male that moved with the sheep flock, typically following them. Ranch employees fed the dog using feeders placed far from the water source, to encourage the dog to move across the entire area.
Methods
Camera-trap sampling design
Data were collected from January 2018 to November 2022 (5 years in total). Twenty camera-trap stations were systematically distributed across the study area. To ensure coverage of the entire landscape, we overlaid a 1 × 1 km grid and deployed one camera in each grid cell, selecting a site based on the presence of a trail or puma pugmarks. Cameras (Bushnell Trophy HD, Bushnell, USA, and Reconyx Hyperfire, Reconyx, USA) were spaced 0.5–1.3 km apart, at a height of 40 cm above ground, and operated 24 h per day. We used motion and heat-triggered cameras set to record a sequence of three pictures in rapid succession once triggered, followed by a 3-minute lockout in which they remained inactive. Stations were checked at least twice per month, to replace batteries and memory cards as needed. To reduce human scent at the sites, field staff used gloves when handling cameras and avoided wearing perfumes, fragrances or insect repellents during fieldwork.
Data analysis
The camera-trap photographs were labelled using digiKam 7.2.0. The metadata (species, date, time, location and season) were used to identify independent puma and dog events. We considered consecutive records of a species obtained ≥ 30 min apart as independent events (Ávila–Nájera et al., Reference Ávila–Nájera, Chávez, Pérez–Elizalde, Palacios–Pérez and Tigar2020). Effort was calculated as the number of effective camera-trap days, excluding those days in which the cameras did not work or were not in the correct position (e.g. misplaced by animals or wind).
Activity patterns
Prior to analysis, the time of day of each photograph was converted first to decimal time and then to radians, to account for the circular nature of this variable (Zar, Reference Zar2010). To describe daily activity and temporal overlap, we fitted kernel density functions (Ridout & Linkie, Reference Ridout and Linkie2009) to the time of independent puma and guarding dog events. The smoothing factor was set to 2 to reduce noise and highlight general trends. We applied a Rayleigh’s test of uniformity (Zar, Reference Zar2010) to test whether the data were uniformly distributed or indicated a preference for a particular time of the day. To assess temporal overlap, we also used the time of day from independent puma and guarding dog events. We calculated the coefficient of overlap (
![]() $\hat \Delta $
1), which ranges from 0 (no overlap) to 1 (complete overlap), and is recommended for small sample sizes (Ridout & Linkie, Reference Ridout and Linkie2009). Overlap values were interpreted as low (< 0.5), medium (0.5–0.75) or high (> 0.75), following Yang et al. (Reference Yang, Zhao, Han, Wang, Mou, Ge and Feng2018). Then, we estimated the 95% confidence intervals (CI) for the overlap coefficient from 10,000 bootstrap samples (Meredith & Ridout, Reference Meredith and Ridout2021). As the coefficient of overlap is purely descriptive (Monterroso et al., Reference Monterroso, Alves and Ferreras2014), we used the Mardia–Watson–Wheeler test to compare activity patterns. Values of this test (W) furthest away from zero indicate the greatest differences between two distributions of events across the diel cycle. These analyses were carried out with the overlap (Ridout & Linkie, Reference Ridout and Linkie2009) and circular (Agostinelli & Lund, Reference Agostinelli and Lund2024) packages of R 4.1.1 (R Core Team, 2021). For all tests, a value of P ≤ 0.05 was considered significant.
$\hat \Delta $
1), which ranges from 0 (no overlap) to 1 (complete overlap), and is recommended for small sample sizes (Ridout & Linkie, Reference Ridout and Linkie2009). Overlap values were interpreted as low (< 0.5), medium (0.5–0.75) or high (> 0.75), following Yang et al. (Reference Yang, Zhao, Han, Wang, Mou, Ge and Feng2018). Then, we estimated the 95% confidence intervals (CI) for the overlap coefficient from 10,000 bootstrap samples (Meredith & Ridout, Reference Meredith and Ridout2021). As the coefficient of overlap is purely descriptive (Monterroso et al., Reference Monterroso, Alves and Ferreras2014), we used the Mardia–Watson–Wheeler test to compare activity patterns. Values of this test (W) furthest away from zero indicate the greatest differences between two distributions of events across the diel cycle. These analyses were carried out with the overlap (Ridout & Linkie, Reference Ridout and Linkie2009) and circular (Agostinelli & Lund, Reference Agostinelli and Lund2024) packages of R 4.1.1 (R Core Team, 2021). For all tests, a value of P ≤ 0.05 was considered significant.
We first evaluated whether daily activity patterns were consistent across years (2018–2022) by comparing interannual activity distributions for both pumas and the guarding dog using kernel density functions, the coefficient of overlap (
![]() $\hat \Delta $
1) and the Mardia–Watson–Wheeler test. When overlap values were high and no significant differences were detected, we considered it appropriate to pool data across years. We then compared the activity patterns of pumas and the dog using the same approach. We also used this method to examine whether puma activity differed between camera-trap stations that were used or not used by the dog or sheep. We defined a station as ‘used’ if at least one detection of the dog or sheep occurred during the entire survey period.
$\hat \Delta $
1) and the Mardia–Watson–Wheeler test. When overlap values were high and no significant differences were detected, we considered it appropriate to pool data across years. We then compared the activity patterns of pumas and the dog using the same approach. We also used this method to examine whether puma activity differed between camera-trap stations that were used or not used by the dog or sheep. We defined a station as ‘used’ if at least one detection of the dog or sheep occurred during the entire survey period.
Spatio-temporal interaction
We counted the frequency of puma–dog interactions on a weekly basis. We considered a puma–dog interaction to occur when both species were detected at the same camera-trap station within a defined survey period. This period began with the detection of one species (e.g. the puma, in this example, the triggering species) and ended either when the triggering species reappeared or when 21 days had elapsed without re-detection (Ferry et al., Reference Ferry, Dupont, Bender and Heurich2024; Fig. 2). Within this period, if the second species (i.e. the dog in this example) was detected, the event was classified as an interaction. Thus, one or more interactions could be triggered by the same dog/puma detection (Fig. 2).
Illustration of the approach used to determine interactions between pumas Puma concolor and the livestock guarding dog on a ranch in central Argentina (Fig. 1). In this example, a puma is the triggering species. Letters a, b and c represent different survey periods with one (a & c) or two (b) interactions.

Puma habitat use
We used generalized linear mixed models (GLMMs) with the number of independent puma events as the response variable. Predictive vegetation variables were the proportion of scrubland, grassland, and pasture and crop cover within a 250 m buffer around each camera-trap station. Vegetation cover was determined using high-resolution satellite images from Google Earth 7.3.6.10441 (Google, USA) and field validation. For the latter, we recorded the surrounding vegetation from the back of a pickup truck, noting all visible vegetation types as far as sight allowed. As proxies for anthropogenic activity, we used three predictor variables: the distances to a rural house (frequent human and dog activity, vehicle movement) and two water mills used by livestock. Mill 1 was located near the house and frequently used, whereas mill 2 was used less often. Sheep presence and the number of independent livestock guarding dog events were also included as predictive variables. Study year was included as a random effect.
To avoid multicollinearity between independent variables, we checked the variance inflation factors using the check_collinearity function in R package performance. Where multicollinearity was detected, the variables considered less biologically relevant were eliminated from the model. Prior to the analysis, we normalized the proportional variables using the angular transformation (arcsine √p; Cohen et al., Reference Cohen, Jarvis and Fowler2013), and applied the R scale function with default settings to normalize all predictor variables to mean 0 and standard deviation 1. To account for differences in sampling effort across sites, we incorporated the natural logarithm of the sampling days as offset in all models. We optimized models using the bobyqa function in the R package Ime4, with 100,000 iterations (Powell, Reference Powell2009). We modelled the number of independent puma detections using GLMMs with either a Poisson or negative binomial distribution, two commonly applied alternatives for ecological count data. To account for potential overdispersion, we compared both distributions by fitting models using the glmer and glmer.nb functions in the R package lme4. We retained the distribution that minimized the Akaike information criterion (AIC; Burnham & Anderson, Reference Burnham and Anderson2004), a standard approach when evaluating competing model structures. Once the appropriate distribution was selected, we explored combinations of predictors using the dredge function in the R package MuMIn. Models with ΔAICc > 2 (AIC corrected for small sample sizes) were excluded, and we averaged the remaining models using the full average method to obtain a consensus GLMM (Table 2). Once the final model was obtained, we estimated the 95% CI of each of the variables and used it to select those that were significantly affecting puma capture rates and exclude the variables whose CI included 0 (stable direction of relationship; Zeller et al., Reference Zeller, Nijhawan, Salom-Pérez, Potosme and Hines2011).
Results
The total survey effort of 23,738 trap days over 5 years produced 212 independent records of pumas (mean 8.5 captures/1,000 trap days) and 166 of the guarding dog (7.2 captures/1,000 trap days). Pumas were photographed at 95% and the dog at 50% of the trapping stations.
The combined activity patterns of pumas recorded in each year showed a high overlap between years. Overlap values averaged
![]() $\hat \Delta $
1 = 0.76 and ranged from a minimum of
$\hat \Delta $
1 = 0.76 and ranged from a minimum of
![]() $\hat \Delta $
1 = 0.70 between 2019 and 2020 (CI 0.53–0.86) and 2021 and 2022 (CI 0.55–0.84) to a maximum of
$\hat \Delta $
1 = 0.70 between 2019 and 2020 (CI 0.53–0.86) and 2021 and 2022 (CI 0.55–0.84) to a maximum of
![]() $\hat \Delta $
1 = 0.90, between 2018 and 2019 (CI 0.78–0.99). Because 70% of interannual comparisons showed no significant differences between puma activity distributions, we pooled all puma activity data obtained over the 5 years for the subsequent analyses. Puma events were distributed heterogeneously throughout the day (N = 212, Z = 0.46, P < 0.01). The pattern was markedly nocturnal: activity started around sunset (17.00–18.00) and decreased sharply after sunrise (06.00–07.00; Fig. 3).
$\hat \Delta $
1 = 0.90, between 2018 and 2019 (CI 0.78–0.99). Because 70% of interannual comparisons showed no significant differences between puma activity distributions, we pooled all puma activity data obtained over the 5 years for the subsequent analyses. Puma events were distributed heterogeneously throughout the day (N = 212, Z = 0.46, P < 0.01). The pattern was markedly nocturnal: activity started around sunset (17.00–18.00) and decreased sharply after sunrise (06.00–07.00; Fig. 3).
Daily activity (represented as kernel density across the 24-hour cycle) of pumas (212 events) and the livestock guarding dog (LGD; 166 events), with overlap indicated by the grey shaded area; the coefficient of overlap (
![]() $\hat \Delta $
1) and the statistic of the Mardia–Watson–Wheeler (W) test and its probability are indicated. The dark and light grey stripes show twilight in summer and winter, respectively.
$\hat \Delta $
1) and the statistic of the Mardia–Watson–Wheeler (W) test and its probability are indicated. The dark and light grey stripes show twilight in summer and winter, respectively.

The coefficient of overlap between the annual records of the guarding dog was moderate in general (average
![]() $\hat \Delta $
1 = 0.66) and high in only 2020–2022 (
$\hat \Delta $
1 = 0.66) and high in only 2020–2022 (
![]() $\hat \Delta $
1 = 0.90, CI 0.73–1.02). We found no significant differences between the distributions of livestock guarding dog activity in 80% of interannual comparisons. When pooled, the dog events were heterogeneously distributed throughout the day (N = 166, Z = 0.12, P = 0.07). Guarding dog activity was mainly diurnal, with two peaks, one in the first part of the morning and a less pronounced one at 12.00–18.00 (Fig. 3).
$\hat \Delta $
1 = 0.90, CI 0.73–1.02). We found no significant differences between the distributions of livestock guarding dog activity in 80% of interannual comparisons. When pooled, the dog events were heterogeneously distributed throughout the day (N = 166, Z = 0.12, P = 0.07). Guarding dog activity was mainly diurnal, with two peaks, one in the first part of the morning and a less pronounced one at 12.00–18.00 (Fig. 3).
Daily activity patterns of pumas and the dog overlapped moderately (Fig. 3;
![]() $\hat \Delta $
1 = 0.60, CI 0.52–0.68), with distinct peaks of activity and a significant dissimilarity between their respective distributions (W = 62.89, P < 0.01). Puma activity patterns in camera stations used (N = 132) and not used by the livestock guarding dog (N = 80; Fig. 4a) overlapped (
$\hat \Delta $
1 = 0.60, CI 0.52–0.68), with distinct peaks of activity and a significant dissimilarity between their respective distributions (W = 62.89, P < 0.01). Puma activity patterns in camera stations used (N = 132) and not used by the livestock guarding dog (N = 80; Fig. 4a) overlapped (
![]() $\hat \Delta $
1 = 0.79, CI 0.68–0.88) and the two curves did not differ (W = 4.40, P = 0.11). The overlap of puma activity patterns was also relatively high (
$\hat \Delta $
1 = 0.79, CI 0.68–0.88) and the two curves did not differ (W = 4.40, P = 0.11). The overlap of puma activity patterns was also relatively high (
![]() $\hat \Delta $
1 = 0.79, CI 0.68–0.89) between camera stations used (N = 46) and not used by sheep (N = 166; Fig. 4b), but in this case with a significant difference between the two curves (W = 6.89, P = 0.03).
$\hat \Delta $
1 = 0.79, CI 0.68–0.89) between camera stations used (N = 46) and not used by sheep (N = 166; Fig. 4b), but in this case with a significant difference between the two curves (W = 6.89, P = 0.03).
Daily activity (represented as kernel density across the 24-hour cycle) of pumas recorded by camera stations (a) used (n = 132 events) or not used (n = 80) by the livestock guarding dog (LGD), and (b) used (n = 46) or not used (n = 166) by sheep, with overlap indicated by the grey shaded area; the coefficient of overlap (
![]() $\hat \Delta $
1) and the statistic of the Mardia–Watson–Wheeler (W) test and its probability are indicated. The dark and light grey stripes show twilight in summer and winter, respectively.
$\hat \Delta $
1) and the statistic of the Mardia–Watson–Wheeler (W) test and its probability are indicated. The dark and light grey stripes show twilight in summer and winter, respectively.

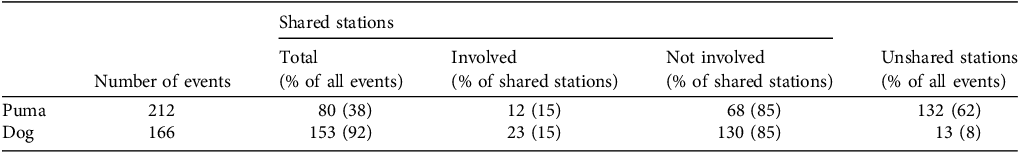
Pumas, the guarding dog and sheep shared 40% of the stations (n = 8). We found no camera stations where sheep occurred without the guarding dog (i.e. the dog was recorded at all sites where sheep were recorded), but we found that one of the nine stations used by the dog and sheep was not used by pumas. Most of the dog events (153, 92%) occurred in the stations also used by pumas, but only 80 (38%) of the puma events occurred in the stations where the dog was also recorded (Table 1).
Total number of puma Puma concolor and livestock guarding dog events (i.e. camera-trap records) and number of events (and per cent of total) at shared and unshared camera-trap stations on a ranch in central Argentina (Fig. 1). A station was defined as shared when both pumas and the dog were recorded and unshared when only one species was recorded. For shared stations, we report the events that involved or did not involve puma–dog interactions. An interaction occurred when a puma and the dog were recorded at the same camera, not necessarily together but within a fixed period, initiated when a puma or dog event occurred and ended when the triggering species reappeared or when 21 days had elapsed (Fig. 2).

Explanatory models of puma habitat use (number of independent events per site) in a ranch in central Argentina. The table shows the estimated model coefficients of the variables included, degree of freedom (df), Akaike information criterion adjusted for small sample size (AICc) and the different in AICc compared to the best-performing model (ΔAICc). Only models with ΔAICc < 2 are included; these models have equivalent empirical support. Blank cells indicate that the corresponding variable was not included in the model.

Interactions between pumas and the livestock guarding dog occurred in only two of the nine camera stations they shared (i.e. where both puma and the dog were recorded). Additionally, interactions occurred only in a small portion of the events. Of the 80 puma events in shared stations, only 12 events were involved in the interactions (Table 1), 6% of all 212 puma events. Twenty-three of the 153 guarding dog events in shared stations were involved in the interactions (Table 1), i.e. 14% of all 166 dog events.
We recorded 27 interactions, 18 triggered by pumas and nine by the guarding dog. For both species, interactions were concentrated within the first week after the triggering event, but the response remained high in the following weeks in the case of the dog (Fig. 5). In one camera station, we detected one sheep predation by a puma (Plate 1), where seven interactions occurred after the predation (25.9% of all interactions).
Frequency of interactions between pumas and the livestock guarding dog (LGD) 1, 2 and 3 weeks after the triggering event (Fig. 2), based on camera-trap detections. An interaction occurred when both species (pumas and the dog) were recorded at the same camera, not necessarily together, but within a period initiated by a detection of one of the species (Fig. 2).

A puma Puma concolor predating on sheep, recorded on a camera trap on a ranch in central Argentina (Fig. 1).

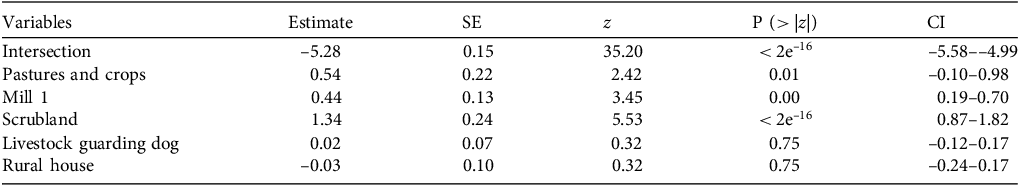
When analyzing the factors affecting habitat use by pumas, we found that our data fitted better to a negative binomial than a Poisson distribution model (AICPoisson = 368, AIC NegativeBinomial = 337) and that vegetation cover variables showed multicollinearity. Thus, we excluded grassland because previous studies showed that this habitat has the lowest impact on habitat use by pumas in the area (Guerisoli et al., Reference Guerisoli, Caruso, Luengos Vidal and Lucherini2019). Three models had ΔAICc values < 2 (Table 2). The proportion of scrubland and the distance to mill 1 were the only variables that affected the use of habitat by pumas (Table 3). Guarding dog events and the distance to the rural house had no significant effect (Table 3). The number of puma events increased in sites with greater proportions of scrubland and greater distance from mill 1.
Average (obtained through the full average method) of the estimated values of the variables included in the top explanatory model of puma habitat use (Table 2), with standard error (SE) and 95% confidence interval (CI). The z value is for the Wald statistical test.

Discussion
We report the first data for Argentina on the activity patterns and interactions of a livestock guarding dog with pumas, the most common top predator in the southern cone of South America. Our findings show that the activity patterns of pumas and the dog differed, thus reducing the chances of encounters. Pumas were primarily nocturnal throughout the year, whereas guarding dog activity was more variable among seasons and essentially cathemeral, in line with the limited information available about the activity patterns of guarding dogs (Kinka et al., Reference Kinka, Schultz and Young2021; Martinez, Reference Martinez2023). Although the activity of the dog was primarily dictated by that of the sheep flock it protected, the nocturnal movements could also be in response to a greater predation risk of livestock (Mosley et al., Reference Mosley, Roeder, Frost, Wells, McNew and Clark2020).
Segregation between pumas and the dog we monitored could potentially occur at different, non-exclusive spatiotemporal levels. Spatially, pumas could avoid the areas frequented by the dog at the scale of the study area; at a finer scale, pumas could reduce their activity in sites where the dog was most active. Temporally, pumas may avoid being active when the dog is, or avoid sites recently used by the dog. Different patterns of segregation have been consistently observed in other studies. Avoidance of areas used by guarding dogs was observed for bobcats Lynx rufus and grey foxes Urocyon cinereoargenteus in Texas, USA (Bromen et al., Reference Bromen, French, Walker, Silvy and Tomeček2019), red foxes Vulpes vulpes in Australia (van Bommel et al., Reference van Bommel, Magrath, Coulson and Johnson2024), and white-tailed deer Odocoileus virginianus in the USA (Gehring et al., Reference Gehring, VerCauteren, Provost and Cellar2010). In France, livestock guarding dogs have been observed to repel wolves (Landry et al., Reference Landry, Borelli and Drouilly2020). Predator–predator avoidance, including pumas avoiding jaguars Panthera onca and coyotes Canis latrans avoiding pumas, has also been reported (Wang et al., Reference Wang, Allen and Wilmers2015; Elbroch & Kusler, Reference Elbroch and Kusler2018), suggesting a general mechanism of spatiotemporal partitioning to reduce the risk of encounters with larger or dominant species.
Unsurprisingly, we found that the guarding dog’s movements were limited to the areas where sheep were allowed to forage (Bromen et al. Reference Bromen, French, Walker, Silvy and Tomeček2019), whereas pumas were detected in all camera stations except the one closest to the farmhouse. Accordingly, puma habitat use was not affected by the dog, and showed avoidance of areas with higher human presence. This may allow pumas to scout the entire area for prey and take advantage of opportunities when some sheep are not under direct dog protection.
At finer scales, interactions between pumas and the dog were infrequent and occurred at only a few sites. Most interactions occurred when the dog visited sites where pumas had been detected previously, suggesting a monitoring behaviour by the dog rather than avoidance by pumas. However, we acknowledge that 21 days may be a relatively long interval and that our results should be interpreted cautiously and viewed as a first step in quantifying interactions between these species.
We argue that the potential for interactions between pumas and the livestock guarding dog may have been primarily limited by their temporal segregation. In other words, if pumas and the dog were rarely active at the same time, the chances for them to interact were necessarily small. This hypothesis is also in line with the presence of the dog having no effect on the habitat use by pumas. However, this does not rule out a potential indirect effect of the dog, mediated by its scent marking. Pumas, like many large carnivores, rely heavily on olfactory and auditory cues to assess the presence and activity of competitors and potential threats (Allen et al., Reference Allen, Stewart-Moore, Byrne and Allen2017). Livestock guarding dogs often leave scent marks (urine, feces, hair) and vocalize (barking), which can serve as persistent indicators of their presence in the landscape (van Bommel & Johnson, Reference van Bommel and Johnson2016; Kinka et al., Reference Kinka, Schultz and Young2021). Even in the absence of direct encounters, such cues may be sufficient to induce spatial or temporal avoidance by pumas, contributing to a form of niche partitioning or predator–predator displacement (Wang et al., Reference Wang, Allen and Wilmers2015). This suggests that livestock guarding dogs could perform as dominant competitors for large carnivores either directly (by their presence) or indirectly (through their urine or faeces; Kinka et al., Reference Kinka, Schultz and Young2021), even though they do not kill or attack wild carnivores.
In agreement with our predictions, the puma in our study area was primarily nocturnal and crepuscular, with little activity during daylight. These findings are in accordance with most studies from a variety of habitats across the Americas (e.g. California, USA: Sweanor et al., Reference Sweanor, Logan, Bauer, Millsap and Boyce2008; central Argentina: Zanón-Martínez et al., Reference Zanón-Martínez, Kelly, Mesa-Cruz, Sarasola, DeHart and Travaini2016; México: Gutiérrez-González & López-González, Reference Gutiérrez-González and López-González2017). In southern Buenos Aires Province, anthropogenic pressures probably reinforce this nocturnal pattern as a result of greater human presence and hunting activity during daylight, which increases the risk of puma mortality (Guerisoli et al., Reference Guerisoli, Caruso, Luengos Vidal and Lucherini2019). Puma behaviour can be affected by the presence of apex predators and humans (Foster et al., Reference Foster, Hamsem and Doncaster2010; Elbroch & Kusler, Reference Elbroch and Kusler2018). The difference we found between the activity of pumas and the dog could be interpreted as evidence of pumas avoiding the dog, but could also reflect a more general avoidance of human activity rather than a direct response to the dog. In neighbouring La Pampa Province, pumas in hunting areas avoided daytime activity, whereas those in protected areas were active during all hours (Zanón-Martínez et al., Reference Zanón-Martínez, Kelly, Mesa-Cruz, Sarasola, DeHart and Travaini2016). Additionally, the nocturnal habits of local key prey species are also likely to shape puma activity patterns (Guerisoli et al., Reference Guerisoli, Gallo, Martinez, Luengos Vidal and Lucherini2021), to optimize foraging efficiency while minimizing anthropogenic threats (Lima & Dill, Reference Lima and Dill1990).
We conclude that the effects of humans and guardian dogs may combine to create a landscape of fear that induces pumas to limit their activity to nocturnal hours. The possible presence of this landscape of fear in our study area is supported by the analysis of the factors affecting puma habitat use, which indicated that pumas preferred the scrubland and avoided the sites most frequently used by people. These findings are in line with those from other studies in ranchlands in central Argentina, suggesting that pumas avoided habitats altered by human intervention (Guerisoli et al., Reference Guerisoli, Caruso, Luengos Vidal and Lucherini2019; Zanón-Martínez et al., Reference Zanón-Martínez, Iranzo, Travaini, McNitt, Mansilla, Llanos and Kelly2023). This behaviour can enable pumas to shelter from people and move without being seen (Suraci et al., Reference Suraci, Smith, Clinchy, Zanette and Wilmers2019; Littlefair, Reference Littlefair2023).
Conclusions
There is growing evidence that non-lethal tools can be at least as effective as lethal control in reducing livestock predation (van Eeden et al., Reference van Eeden, Crowther, Dickman, Macdonald, Ripple and Ritchie2018), and that guarding dogs are a promising strategy (e.g. Andelt & Hopper, Reference Andelt and Hopper2000; Gehring et al., Reference Gehring, VerCauteren, Provost and Cellar2010; Rigg et al., Reference Rigg, Finďo, Wechselberger, Gorman, Sillero-Zubiri and Macdonald2011; van Bommel & Johnson, Reference van Bommel and Johnson2016; Mosley et al., Reference Mosley, Roeder, Frost, Wells, McNew and Clark2020). Our work provides insights into how a guarding dog performs as a non-lethal measure to mitigate human–carnivore conflicts and how its presence may influence wildlife in a modified rural landscape where livestock farming is the main economic activity. Our findings suggest that the potential for direct interactions with pumas was low, probably as a result of temporal and spatial segregation. Although our study focused on a single dog in one location, the habitat and land management in our study area are representative of large areas of central Argentina.
Even though evaluating the effectiveness of the dog in reducing predation was beyond the scope of this study, the Chacra staff perceived a reduction in sheep losses in the presence of the dog. Prior to the introduction of the dog, puma attacks were reported more frequently (7–10 animals lost per year), whereas no confirmed predation events were reported except for a single case documented by our cameras. Additionally, we did not record evidence of direct negative effects of the dog on pumas in the Chacra. Although this evidence should be interpreted cautiously, it suggests a potential deterrent effect worth exploring in future research.
From a management perspective, the relatively low cost and constant presence of trained guarding dogs make them a promising non-lethal strategy to mitigate human–carnivore conflicts, especially when integrated with improved husbandry practices (Green et al., Reference Green, Woodruff and Tueller1984; Miller et al., Reference Miller, Stoner, Cejtin, Meyer, Middleton and Schmitz2016; Novaro et al., Reference Novaro, González, Pailacura, Bolgeri, Hertel, Funes and Walker2017). Nonetheless, implementation should be accompanied by monitoring to ensure effectiveness and animal welfare (Villar et al., Reference Villar, Bidinost, Bruno Galarraga, Cancino, Castillo and Cueto2014; Novaro et al., Reference Novaro, González, Pailacura, Bolgeri, Hertel, Funes and Walker2017). Further research, ideally involving multiple dogs and replicated sites, is needed to assess the general effectiveness of guarding dogs across different landscapes and to better understand their impacts on native wildlife.
Author contributions
Study design, fieldwork: SDM, EMLV, ML; data analysis, writing: MLRG, NC, ML, EMLV; revision: all authors.
Acknowledgements
We thank the Chacra employees, the volunteers and students of Universidad Nacional del Sur, and Université de Sherbrooke, Canada, and the staff of Instituto Nacional de Tecnología Agropecuaria who facilitated our work. This study was supported by Proyectos de Investigación Plurianuales (CONICET; PIP 11220200100839CO); Secretaría de Ciencia y Técnica-Universidad Nacional del Sur (PGI 24/B243 and 24/B318); Proyectos de Investigación Científica y Tecnológica (PICT 2018-04134); Wild Felid Research and Management Association; and the Neotropical Grassland Conservancy.
Conflicts of interest
None.
Ethical standards
This research abided by the Oryx guidelines on ethical standards. Formal ethical approval was not required for this study, as it was based exclusively on non-invasive observational methods and did not involve animal capture, handling or experimentation. Camera traps were deployed on the Chacra Experimental Patagones, a research facility belonging to the Ministerio de Desarrollo Agrario of Buenos Aires Province, with permission from the authority responsible for the site. Camera traps were deployed without the use of bait or attractants, to reduce behavioral alteration. Some images of people were inadvertently captured; these images were not analysed, and were not used for any purpose.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.