Introduction
Manganese (Mn) is the third most abundant transition metal in the Earth’s crust(Reference Wang, Lineweaver and Ireland1). It is widely present in soil (450–4000 mg/kg(Reference Wang, Wang and Zhang2)), where it is involved in the processing and recycling of soil organic carbon(Reference Zhuang, Zhu and Shi3), and is used as a raw material in industrial fields, including mining, welding and battery manufacturing(Reference Parmalee and Aschner4). For all living organisms, Mn is an essential trace and nutrient element for cellular metabolism(Reference Chen, Bornhorst and Aschner5,Reference Kawahara, Kato-negishi and Tanaka6) . Plants need Mn for the synthesis of chlorophyll, the promotion of photosynthesis and maintenance of the normal colour of the blade(Reference Schmidt, Jensen and Husted7). Mn widely exists in animals and functions as an enzyme component and activator(Reference Spears8). As an active cofactor, it participates in a series of biological reactions of a variety of key metalloenzymes, including arginase, pyruvate carboxylase, acetylcholinesterase, glutamine synthetase (GS) and manganese superoxide dismutase (MnSOD/SOD2),(Reference Chen, Bornhorst and Aschner5) to support the normal physiological activities of cells. In the human body, it is involved in fat metabolism, bone growth and the development and normal operation of the nervous system(Reference Tarnacka, Jopowicz and Maślińska9–Reference Xia, Wang and Zhang11).
Normal Mn levels range from 4 to 12 μg/l in whole blood, from 1 to 8 μg/l in urine, and from 0·4 to 0·85 μg/l in serum(Reference Martins, Ruella Oliveira and Barbosa12). To maintain the balance of Mn in the human body, the human body absorbs Mn from the environment and diet(Reference Miah, Ijomone and Okoh13). The majority of Mn intake comes from water and foods (for example, beans, rice, nuts, whole grains, seafood, seeds, chocolate, tea, etc.(Reference Peres, Schettinger and Chen14)), with rice (>42%) as the main food source of Mn(Reference Zhou, Su and Su15). Mn intake from environmental exposure or other external sources, such as air pollution or occupational exposure, is relatively rare. Dietary intake of Mn is mainly absorbed in the intestine(Reference Einhorn, Haase and Maares16). Excess Mn is delivered to the liver through the portal vein and excreted with bile through the faeces, while a small amount can be reabsorbed by the intestine. Urinary excretion of Mn is less than 10% of total excretion(Reference Myers, Thompson and Naik17). In certain populations, such as those with abnormally elevated Mn consumption or gastrointestinal absorption disorders, obtaining sufficient Mn from dietary sources or the environment may be challenging. In such cases, intravenous supplementation of Mn may be necessary to meet the body’s requirements. In mice, intravenous administration of Mn has been shown to produce higher concentrations of Mn in most organ tissues compared with inhalation(Reference Wooten, Aweda and Lewis18). The disruption of Mn homeostasis leads to a variety of diseases in humans(Reference Parmalee and Aschner4). Mn is needed for the biosynthesis of mucopolysaccharides in bone matrix formation and is a cofactor for enzymes in bone tissue(Reference Palacios19). Long-term dietary Mn deficiency can cause osteoporosis, and its mechanism is related to the increase of serum calcium and phosphorus levels and the decrease of bone calcium levels(Reference Strause, Hegenauer and Saltman20). Despite its importance for human health (it is a cofactor for normal cellular functioning enzymes), Mn is also toxic to human organs in excess. High levels of Mn are most likely to accumulate in the brain, especially in the basal ganglia, leading to neurotoxicity(Reference Wang, Li and Zheng21–Reference Dobson, Erikson and Aschner23). The main mechanisms of neurotoxicity in the human brain include oxidative stress, mitochondrial dysfunction, transporter dysregulation, metal imbalances, neuroinflammation and protein trafficking pathway dysregulation(Reference Harischandra, Ghaisas and Zenitsky24). Specifically, Mn destroys DOPA decarboxylase in central nerve cells, resulting in a decrease in L-DOPA content. The balance of L-DOPA and acetylcholine (ACH) is disrupted, and the content of ACH is temporarily increased, resulting in conduction dysfunction in the central nervous system(Reference Finkelstein, Milatovic and Aschner25). Excess Mn induces oxidative stress response and causes dopaminergic (DA) neuron degeneration, leading to a series of neuropsychiatric symptoms and signs(Reference Santos, Milatovic and Andrade26). The main symptoms are psychological and emotional disturbances, as well as many motor symptoms, including gait disturbance, tremor, stiffness and bradykinesia(Reference Bouabid, Tinakoua and Lakhdar-Ghazal27).
In addition, changes in Mn content are one of the characteristic markers of cancer progression and metastasis. Mn increases in a time-dependent manner during the development of carcinoma in situ within 3–5 weeks, and a Mn-rich niche is formed in distant metastatic carcinomas(Reference Stelling, Soares and Cardoso28). Mn mainly impacts cancer by affecting the body’s anti-cancer immune response. Mn can promote the survival and proliferation of immune T cells and effectively promote the cancer killing ability of natural killer T cells (NKT) (Reference Golara, Kozłowski and Guzik29–Reference Zhang, Qi and Cai32). In summary, Mn plays an important role in anti-cancer immunity in the body. Clarifying the role and mechanism of Mn in cancer is expected to provide new directions for cancer treatment in the future.
As an important nutritional element, appropriate amounts of Mn help to maintain human health and resist diseases, but the imbalance of Mn homeostasis can lead to the development of diseases. Therefore, it is necessary to study its physiological function in human body. We will further review the anti-cancer mechanism of Mn and the precise delivery method of Mn to explore new options for the treatment of tumours.
Pathways of Mn uptake, excretion and distribution
Pathways of Mn uptake
Mn is an important dietary element, and plant foods such as whole grains, nuts and vegetables are rich in Mn, with the most abundant content in tea(Reference Martins, Krum and Queirós33). Although the content of Mn in animal food (meat, fish and milk) is not high, its absorption and retention are both high, and it is a good source of Mn intake for humans. The efficiency of Mn absorption by the small intestine is affected by many factors, including intestinal pH, divalent metal transporter-1 (DMT1), competition from divalent metals (such as iron, copper, zinc or calcium) and chelators such as phytic acid(Reference Teeguarden, Gearhart and Clewell34). Mn is absorbed by intestinal cells in the form of Mn3+ through its binding to transferrin. Subsequently, endocytosis internalises the formed complex (Mn3+–transferrin) and facilitates the dissociation of Mn3+ into Mn2+(Reference Gunter, Gerstner and Gunter35). Mn2+ is then transported into the cytosol by DMT-1(Reference Gruenheid, Canonne-Hergaux and Gauthier36). Mn absorbed from the small intestine enters the blood through small intestinal epithelial cells. Most Mn is bound to Mn-transporting proteins in the plasma for transport, and a small part of the Mn directly enters erythrocytes.
Exposure of the body to the intake of Mn in the occupational environment is the main cause of the toxic side effects of Mn. With the rapid development of modern society, most industrial production involves the metal field, and large amounts of Mn are applied in industrial production. We found that Mn levels were higher in most workers than in the general population,(Reference Martins, Krum and Queirós33,Reference Leavens, Rao and Andersen37) and inhalation exposure to airborne Mn was common in welders and smelters(Reference Bowler, Beseler and Gocheva38). In the occupational environment, humans absorb Mn mainly through the lungs, and only particles with an aerodynamic diameter of less than 5 μm can reach the alveoli. Then, Mn is absorbed in the form of ions.
After people ingest Mn from the external environment, a portion of Mn enters the blood for transport to different tissues(Reference Bouabid, Tinakoua and Lakhdar-Ghazal27), and part is transported from the nose to the brain along the olfactory nerve, from which it can bypass the blood-brain barrier (BBB) and enter the central nervous system through the brain(Reference Lucchini, Dorman and Elder39). Many ways have been identified for Mn to enter the brain, including the following three main routes: the capillary endothelial cells of the BBB, the choroid plexus of the blood-cerebrospinal fluid barrier, and through the olfactory nerve in the nasal cavity directly to the brain(Reference Crossgrove and Zheng40). When the plasma concentration of Mn is within the physiological range (0·076–78 μM), Mn is mainly transported into the brain through saturation of cerebral capillaries. Beyond this range, however, the unsaturated transport of Mn into the brain through cerebrospinal fluid (CSF) occurs more rapidly(Reference Murphy, Wadhwani and Smith41). The third route is more important because most reports of the toxic side effects of Mn have occurred through inhalation exposure.
Pathways of Mn excretion
The pathways of Mn excretion from the body are as follows: the main pathway is excretion from the liver and gallbladder in the form of faeces, followed by excretion in the form of urine(Reference Klaassen42). In addition, a small amount of Mn can be excreted through sweat(Reference Omokhodion and Howard43). Regardless of the level of Mn absorption, adults can maintain the balance of Mn concentrations in tissues by regularly regulating the absorption rate and excretion rate of Mn. In the liver, Mn is separated from the blood and bound to bile before being re-secreted into the intestine. A small fraction of Mn is reabsorbed in the intestine, forming the hepato-enteric circulation. In the brain parenchyma, rapid accumulation of Mn occurs in brain structures such as the amygdala, end grain, hippocampus and pale sphere structure, and Mn has a half-life of approximately 5–7 d. However, Mn has the longest retention time in the periaqueductal grey matter, amygdala and endothelium(Reference Omokhodion and Howard43).
Content and distribution of Mn
Mn exists in the human body in the form of a variety of proteins and enzymes such as manganese superoxide dismutase, GS and arginase(Reference Wang, Guan and Lv44), and acts as a coenzyme in a variety of biological processes such as skeletal system development, energy metabolism, enzyme activation, and functions of the nervous system, immune system and reproductive hormones(Reference Kawahara, Kato-negishi and Tanaka6). Mn is involved in a variety of biological processes such as nutrient metabolism, bone formation, the free radical defence system, ammonia clearance and neurotransmitter synthesis in the brain. Mn is most commonly stored in mitochondria after uptake, and therefore the highest concentration is found in mitochondria-rich organs such as liver, kidney and pancreas(Reference Aschner and Erikson45). The normal concentration of manganese varies in different human tissues: 1 mg/kg in bone, 1·04 mg/kg in the pancreas(Reference Barceloux46), 0·98 mg/kg in the renal cortex, 1·2–1·3 mg/kg in the liver and 0·15–0·46 mg/kg in the brain(Reference Chen, Bornhorst and Aschner5) (Fig. 1). We summarise the normal range of Mn content in various organs of the human body in Table 1. Excessive Mn can easily accumulate in the brain, leading to the onset of neuropsychiatric symptoms. In the brain, Mn preferentially accumulates in the caudoputamen, globus pallidus, substantia nigra and subthalamic nucleus(Reference Chakraborty and Aschner47). Under normal conditions, intracellular Mn mainly accumulates in mitochondria and participates in the process of oxidative phosphorylation (OXPHOS) in mitochondria. Chronic excessive exposure to Mn has been confirmed to induce mitochondrial dysfunction, which is related to oxidative damage(Reference Liu, Jing and Liu48). In fact, high concentrations of Mn can cause neurotoxicity and lead to psychiatric diseases(Reference Khan, Wasserman and Liu49,Reference Oulhote, Mergler and Barbeau50) , such as Parkinson’s disease and gamma-aminobutyric acid (GABA) system-related diseases(Reference Martins, Ruella Oliveira and Barbosa12). The daily intake of Mn per adult should be approximately 2–6 mg/d(Reference Freeland-Graves and Mousa51), and the World Health Organization/Food and Agriculture Organization (WHO/FAO) of the United Nations recommends Mn levels in drinking water <400 μg/l(Reference Martins, Krum and Queirós33).
Mn in the human body. Mn contents in various tissues of the human body and the pathways of Mn intake and excretion.

Mn homeostasis. The concentration of Mn in human organs and body tissues ranked from lowest to highest

The effects of Mn homeostasis on microbial survival
The number of bacteria present in the human body is almost equal to the ratio of human cells (1·3:1). The intestinal flora (99%) is the most important group of bacteria in human body, and erythrocytes (84%) are the most important cells(Reference Sender, Fuchs and Milo52). As a metabolic ‘organ’ in the human body, the interaction between bacterial communities is a key factor affecting human health. Mn is not only an indispensable catalytic centre and structural core for various enzymes but also involved in a variety of biological processes, including OXPHOS, glycosylation and signal transduction. It is also an essential metal element for pathogenic microorganisms to maintain basic biochemical activity and virulence. Hosts can utilise Mn to participate in the process of nutritional immunity to prevent pathogen invasion(Reference Wu, Mu and Xia53).
Role of Mn in pathogenic microbes
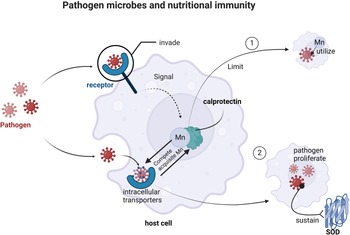
Usually, the survival environment of pathogenic microorganisms in the host body is brutal, and they need the participation of metal ions to maintain survival. The oxidative defence system of human body can kill pathogenic bacteria, but manganese can improve the tolerance of pathogenic bacteria to reactive oxygen intermediates (ROIs). In addition, Mn is involved in the adaptation process of pathogens to the human body by acting as a cofactor for enzymes involved in intermediary metabolism and cell signalling pathways, maintaining the expression of virulence related genes and other pathways(Reference Zaharik and Finlay54). A defence strategy called ‘nutritional immunity’ is employed by host cells invaded by pathogenic microorganisms. It aims to prevent invading pathogens from acquiring metal ions, such as Mn, from the host(Reference Hood and Skaar55). In brief, during an inflammatory episode, Mn binds to calprotectin to compete for or strip the metal from pathogen metalloproteins, inactivating them and weakening their defence against the host immune response. In some cases, pathogens have developed the ability to compete with calprotectin-mediated metal starvation in the gut(Reference Zackular, Chazin and Skaar56). In patients with various types of infection, it can be observed that their serum and tissue Mn levels are significantly reduced(Reference Domej, Krachler and Goessler57). A series of experiments have shown that once host cells have phagocytosed pathogens, Mn transporters, including natural resist-associated macrophage protein 1 (NRAMP1) encoded by the human SLC11A1 gene, and other metal transporters, limit the intracellular utilisation of Mn by pathogens(Reference Sheldon and Skaar58,Reference Cellier, Courville and Campion59) .
To prevent themselves from being killed, pathogenic microorganisms have evolved a complicated system to obtain metal ions from the host for improved survival(Reference Sheldon and Skaar58,Reference Martínez, Delgado-Iribarren and Baquero60) . Among a variety of pathogens, such as Staphylococcus aureus (Reference Horsburgh, Wharton and Cox61), Staphylococcus pneumoniae (Reference Marra, Lawson and Asundi62), Salmonella typhimurium (Reference Kehres, Zaharik and Finlay63,Reference Kehres, Janakiraman and Slauch64) and Yersinia pestis (Reference Perry, Craig and Abney65,Reference Bearden and Perry66) , two manganese transport systems have been identified that are closely related to host pathogenesis and involve intracellular transporters such as MntABC, MntH, NRAMP, BmtA and MntX(Reference Green, Todd and Johnston67–Reference Ouyang, He and Oman70). They control the uptake of Mn and thus the virulence of pathogens. For example, the S. aureus proteins MntABC and MntH compete with the host manganese-binding protein calprotectin for Mn acquisition, thereby enabling the pathogen to proliferate and maintain its SOD activity(Reference Kehl-Fie, Zhang and Moore71) (Fig. 2). The functions of different transport proteins in pathogen microbes/cells are described in Table 2.
Pathogen microbes and nutritional immunity. Host cells and pathogenic microorganisms compete for Mn. Calprotectin in host cells compete for the Mn in pathogen metalloproteins, inactivating them and impairing their defence against host immune responses. Pathogenic microorganisms, however, compete with the host calprotectin to obtain Mn, thereby allowing the pathogen to proliferate and maintain its SOD activity.

Transport proteins. The functions of different transport proteins in pathogenic microbes/cells

Excessive Mn may be harmful to bacteria
Various Mn efflux proteins, such as MntE, MntP and MntX (which have the same names as the proteins listed above but represent different proteins), control the export of excess Mn from bacteria to prevent toxicity caused by excessive Mn(Reference Zeinert, Martinez and Schmitz72). For example, in Staphylococcus aureus (S. aureus), the Mn export protein MntE is necessary for the pathogen to exert its full virulence during infection, indicating the presence of Mn toxicity in the pathogen, whereas a mutant strain of S. aureus lacking the MntE has reduced resistance to oxidative stress and lower oxidative stress levels(Reference Grunenwald, Choby and Juttukonda73).
In addition, Mn plays a role in regulating the activity of viral enzymes at the host–virus interface, and the most important finding is that Mn contributes to the assembly and integration of human immunodeficiency virus (HIV) (Reference Wolfe, Felock and Hastings74). Mn has been shown to increase the number of DNA nucleotide triphosphate errors in pathogens and retroviruses in vitro (Reference Vartanian, Sala and Henry75). Thus, in the presence of excess Mn, the mutation rate of HIV increases significantly. In the DNA-based herpes simplex virus, for example, Mn ions seem to act as DNA polymerase super catalysts to promote DNA replication(Reference Villani, Tanguy Le Gac and Wasungu76).
In conclusion, pathogens such as bacteria and fungi have robust self-protection systems to regulate Mn homeostasis, thereby maintaining virulence and evading Mn toxicity. Impaired Mn metabolism impairs pathogen proliferation and virulence, and this process could be a potential research target for the prevention of pathogen invasion in the host.
Mn regulates mitochondrial function
At the subcellular level, most of the metabolism of manganese occurs in mitochondria. It enters the mitochondria via DMT1 present in the outer mitochondrial membrane, while efflux is very slow(Reference Wolff, Garrick and Zhao77). In addition to high storage concentrations, Mn enters mitochondria more rapidly, with intracellular mitochondria increasing Mn at a higher rate than nuclei after chronic Mn treatment(Reference Morello, Canini and Mattioli78). Mitochondria are the site of manganese metabolism and energy conversion in cells. Its main function is OXPHOS to synthesise ATP and supply life activities. In addition to being a key organelle for intracellular energy generation, mitochondria are also involved in metabolic processes such as apoptosis, lipid metabolism and free radical production. The inhibitory effects of excess Mn on the respiratory chain and calcium efflux in cellular mitochondria, and the anti-cancer effects of the antioxidant enzyme MnSOD, are summarised in Fig. 3.
Role of Mn in mitochondria in human cells. Mn is involved in the regulation of mitochondrial function by regulating SOD2 activity. A disruption of Mn homeostasis in mitochondria impairs the composition and function of mitochondrial RNA particles, leading to a disruption of mitochondrial transcriptional processes that results in impaired assembly and function of the mitochondrial respiratory chain. If Mn accumulates in mitochondria, it inhibits calcium efflux and triggers mitochondrial dysfunction.

Excessive accumulation of Mn can inhibit calcium efflux
Excess Mn is detrimental to mitochondrial function both in vivo and in vitro, leading to dysfunction. Mn is involved in the antioxidant system of mitochondria and can interfere with calcium metabolism in mitochondria(Reference Zhang, Zhou and Fu79). Experimental studies have shown that Mn exposure in the brain, especially in the striatum and hypothalamus, leads to Mn accumulation while simultaneously affecting Ca2+ metabolism in mitochondria. It inhibits the outflow of Ca2+ from mitochondria such that Ca2+ gradually accumulates in mitochondria, affecting the transmission of excitation in mitochondria and causing a series of dysfunctions(Reference Ijomone, Aluko and Okoh80). Elevated calcium levels lead to reactive oxygen species (ROS) production and to the opening of the mitochondrial permeability transition pore (mPTP). This process leads to a loss of the inner membrane potential, mitochondrial swelling, impaired OXPHOS and inhibition of ATP synthesis. All of these processes further generate ROS associated with Mn neurotoxicity and aggravate mitochondrial dysfunction(Reference Miah, Ijomone and Okoh13).
Dysregulation of Mn homeostasis impairs the assembly and function of the respiratory chain
Mn can be harmful to human health when it is present in excess or deficient in the body. Mn exposure impaired the composition and function of the mitochondrial ribosomal proteins DHX30 and MRPS18B, and the mitochondrial RNA particle FASTKD2, disrupting the mitochondrial transcription process. FASTKD2 is a component of the mitochondrial RNA granule required for the processing of polycistronic mitochondrial RNA, a step necessary for mitochondrial protein synthesis. Notably, genetic disruption of the mitochondrial RNA granule or pharmacological inhibition of mitochondrial transcription–translation in cells is protective against acute Mn exposure in vitro (Reference Werner, Gokhale and Ackert81–Reference Antonicka and Shoubridge83). FASTKD2 and other mitochondrial RNA binding proteins (including DHX30, GRSF1 and the mitochondrial ribosomal subunit) participate in protein complexes, such as MRPS18B(Reference Antonicka and Shoubridge83,Reference Antonicka, Lin and Janer84) .
The disruption of Mn homeostasis results in interference with RNA particles within mitochondria. RNA particles are important intermediates for protein synthesis in mitochondria. They are responsible for the transcription of genetic information from DNA into RNA and further direct protein synthesis. The disruption of Mn homeostasis interferes with the formation and stability of RNA particles, thereby affecting protein synthesis within mitochondria. Werner et al. tested whether Mn exposure could induce these RNA granule phenotypes in induced pluripotent stem cell (iPSC)-derived human astrocytes and glutamatergic neurons and reported that iPSC-derived neurons presented reduced levels of DHX30, GRSF1 and MRPS18B when exposed to low Mn concentrations. These results suggested that a disruption of Mn homeostasis could reduce the composition of mitochondrial RNA particles and thus affect mitochondrial protein synthesis(Reference Werner, Gokhale and Ackert81).
Effect of MnSOD activity on mitochondrial redox balance
Active MnSOD is a mature antioxidant enzyme in mitochondria. MnSOD activity is essential for maintaining the redox balance in mitochondria and can protect cells from oxidative stress damage caused by excessive reactive oxygen species (ROS)(Reference Liu, Liu and Liu85,Reference Suski, Lebiedzinska and Bonora86) . In normal cells, MnSOD can maintain mitochondrial integrity and promote cell regeneration. In cancer cells, MnSOD attenuates the deleterious effects of ROS stimulation, including the promotion of cancer development and maintenance.
Specifically, hypoxia and hypermetabolism are the common characteristics of cancer cells, and their daily energy requirements are far higher than normal cells. This means that cancer cells need to produce more mitochondria to meet their energy demand. However, the mitochondria produced by cancer cells often have abnormal phenomena such as wrinkled outer membranes, membrane structural integrity defects, mitochondrial distribution around the nucleus, and hollow mitochondria. An important cause of abnormal mitochondrial structure and morphology is the frequent mutation of mitochondrial DNA (mtDNA) in cancer cells(Reference Cannino, Ciscato and Masgras87). mtDNA mutations not only cause changes in mitochondrial appearance but also directly cause dysfunction, increase ROS production and redox imbalance in mitochondria, and stimulate the proliferation and invasion of cancer cells(Reference Polyak, Li and Zhu88–Reference Hochhauser90). During carcinogenesis, tumour cells typically exhibit increased reactive oxygen species (ROS) production(Reference Thiagalingam and Nelkin91,Reference Sundaresan, Yu and Ferrans92) , increased ROS accumulation and dysregulation of antioxidant enzymes(Reference Nie, Chen and He93). ROS derived from the electron transport chain in mitochondria can activate signalling pathways related to carcinogenesis. For example, H2O2 (a form of ROS) can activate receptor tyrosine kinase(Reference Kamata and Hirata94,Reference Sundaresan, Yu and Ferrans95) , Ras-mitogen-activated protein kinase (Ras-MAPK)(Reference Guyton, Liu and Gorospe96,Reference Rao97) and phosphatidylinositol 3-kinase (PI3K) pathways(Reference Kamata and Hirata94). As an antioxidant enzyme, MnSOD can convert superoxide into hydrogen peroxide (H2O2) and oxygen, and further convert it into harmless water and oxygen during cancer progression, thereby reducing oxidative damage to cells and playing a protective antioxidant role(Reference Ekoue, He and Diamond98). Animal experiments have revealed that mice with a monoallelic knockout of MnSOD (SOD2+/−) exhibit increased superoxide levels, which leads to the occurrence of cancer(Reference Zhang, Zhang and Shi99). Compared with wild-type control, SOD2+/− mice are more likely to develop cancer and have a shortened lifespan, suggesting that the loss of MnSOD activity promotes carcinogenesis. In addition, an analysis of mitochondrial DNA from patients with breast cancer has shown a common deletion of MnSOD in the blood, resulting in impaired MnSOD activity and increased oxidative damage(Reference Nie, Chen and He93).
In general, enhanced MnSOD activity reduces mitochondrial oxidative stress and inhibits cancer development. Taking advantage of the antioxidant effects of manganese in mitochondria, it has also been used in cancer therapy in some cases. For example, single-atom manganese anchored on carbon dots can effectively concentrate in mitochondria, interfere with their oxidation–reduction balance, and exhibit excellent anti-cancer performance and good magnetic resonance imaging (MRI) signal responsiveness under visible light irradiation, showing its application potential in the integration of tumour diagnosis and treatment(Reference Wang, Ma and Liang100).
Mn enhances anti-tumour immunity through the cGAS–STING pathway
Mn, an indispensable trace element in the human body, is not only an important cornerstone for maintaining health but also plays a non-negligible role in the anti-cancer process. We summarise the key pathways and mechanisms through which Mn exerts its anti-cancer effects by enhancing immune responses (Fig. 4).
cGAS-STING signalling pathway. Mn increases the sensitivity of cGAS to dsDNA in cancer cells and regulates the cGAS–STING pathway to promote the anti-cancer immunity of immune cells. Mn binds to cGAS and enhances its anti-cancer effect by inhibiting the binding of immune receptors such as PD-1 and CTLA-4 on the surface of immune cells to ligands such as PDL-1 and BT on the surface of cancer cells. In the STING pathway, Mn stimulates IRF-3 in tumour cells to produce IFN, thereby activating the anti-cancer effect of immune cells such as NKT and CTL.

Increased dsDNA sensitivity
Further studies showed that Mn-mediated anti-tumour immune responses were dependent on the cyclic GMP-AMP synthase-stimulator of interferon genes (cGAS–STING) pathway. Mn can directly interact with cGAS, increase the sensitivity of dsDNA, significantly stimulate the expression of interferon (IFN) regulatory factor (RF3/IRF7) and type I IFN(Reference Wang, Guan and Lv44), and then stimulate T cells to exert immune function. cGAS activity is affected by a variety of factors, of which the sensitivity of double-stranded DNA (dsDNA) is a key factor. As an important immune antigen, dsDNA can stimulate the activity of cGAS and trigger a strong immune response. The sensitivity of dsDNA may be insufficient, resulting in a weakened immune response and thus affecting the efficacy of disease treatment. Mn can act on cancer cells, directly bind to cGAS, stabilise cGAS activity, increase its sensitivity to dsDNA(Reference Lv, Chen and Zhang101) and thus stimulate the cGAS–STING pathway to participate in human immune responses. This implies that, in the presence of manganese, even low concentrations of dsDNA are effective in stimulating cGAS activity to elicit a strong immune response.
Inhibition of immunosuppressive molecules
More importantly, Mn could directly bind to cGAS and reduce surface programmed cell death protein-1 (PD-1) and cytotoxic T-lymphocyte associated protein 4 (CTLA-4) protein concentrations. PD-1 and CTLA-4 are two important immunosuppressive molecules(Reference Yang, Li and Gu102), and their elevated expression inhibits T-cell function. By activating the cGAS–STING pathway, manganese ions can reduce the expression of these inhibitory molecules, thereby relieving immunosuppression and enhancing anti-tumour immune responses(Reference Yang, Li and Gu102–Reference Buchbinder and Desai106). At present, anti-PD-1/PD-L1 antibodies have been successfully used to treat various types of cancer(Reference Herbst, Giaccone and De Marinis107–Reference Qiu, Chen and Zhang114). However, due to individual differences, most patients cannot benefit from this therapy(Reference Yi, Jiao and Xu115,Reference Niu, Yi and Li116) . Divalent manganese, as a natural STING agonist, can cooperate with anti-PD-1 /PD-L1 antibodies in cancer immunotherapy(Reference Ghaffari, Peterson and Khalaj117). Even in low immunogenic tumour models such as B16, the combination of STING agonists with anti-PD-1/PD-L1 antibody treatment significantly prolonged the survival of mice compared with the corresponding monotherapy(Reference Pan, Perera and Piesvaux118). Preliminary clinical evidence suggests that Mn supplementation, when administered as Mn chloride via intranasal (0·05–0·1 mg/kg/d) or inhalation (0·1–0·4 mg/kg/d) routes in combination with subsequent intravenous chemotherapy (day 2) and anti-PD-1 antibody (2–4 mg/kg, day 3) on a 3-week cycle, demonstrates promising efficacy in treatment-refractory advanced metastatic solid tumours. The observed objective response rate reached 45·5% with a disease control rate of 90·9% in patients who had failed standard anti-cancer therapies including chemoradiotherapy and prior PD-1 blockade(Reference Lv, Chen and Zhang101). These findings need further confirmation in randomised controlled trials. In addition, Mn could significantly increase the expression of CD80 and CD86 in Dendritic cells (DCs) by activating the cGAS–STING pathway. CD80 and CD86 are costimulatory molecules that bind to their respective receptors on the surface of T cells and induce T-cell activation, thereby enhancing the adaptive immune response.
Amplify the activities of cGAS and STING
Mn can also amplify the activation of cGAS and STING by increasing cyclic guanosine monophosphate–adenosine monophosphate (cGAMP) production and increasing the binding affinity of cGAMP-STING. STING proteins trigger downstream signals on the endoplasmic reticulum (ER) surface by activating and recruiting interferon regulatory factor 3 (IRF-3), TANK-binding kinase 1 (TBK-1) and nuclear factor kappa B (NF-κB), followed by the expression and secretion of IFNs. Type I IFNs stimulate adaptive and innate immunity to cancer by promoting DC maturation and antigen presentation to stimulate T cells(Reference Li, Li and Niu119). IFNs can also promote NKT recruitment and CTL infiltration(Reference Cheng, Zhang and Liu120) to exert anti-cancer effects. One study showed that STING knockout (Sting gt/gt) mice had a faster growth rate of tumour cells (such as RMA-S lymphoma and B16-BL6 melanoma) than wild-type mice, indicating that STING plays an important role in anti-tumour response. In Rag2 knockout (Rag2−/−) mice lacking both T and B cells, when STING was knocked out (Rag2−/− Sting gt/gt), these mice were significantly more sensitive to RMA-S and B16-BL6 tumours. This suggests that STING also plays a key role in NK cell-mediated anti-tumour responses. Knockout of cGAS gene (Cgas−/−) in tumour cells by CRISPR/Cas9 technology can reduce the activation of NK cells, further confirming the importance of the cGAS–STING pathway in NKT activation. These experimental results suggest that cGAS in cancer cells recognise their own DNA to produce cGAMP molecules. The cGAMP molecule is recognised by the STING protein of the host cell and activates STING. Activated STING proteins trigger an interferon response and produce cytokines such as IFN-β. These cytokines can activate NK cells to enhance anti-cancer immune responses and improve the ability to recognise and kill cancer cells(Reference Marcus, Mao and Lensink-Vasan121).
On the basis of the mechanism that Mn promotes anti-tumour effects in a cGAS–STING-dependent manner, a bovine serum albumin/ferritin-based nanoagonist incorporating Mn ions (Mn2+) and β-lapachone has been developed. This protein-based cGAS–STING nanoagonist was able to efficiently activate T-cell mediated anti-cancer immune responses in in vitro cell experiments and inhibit cancer growth and also showed significant therapeutic effects in animal models(Reference Wang, Liu and Xue122).
Mn delivery strategies
Mn, as an essential trace element, plays an essential role in anti-cancer immunity under physiological conditions. Studies have demonstrated that the growth of cancer cells and lung metastasis are significantly accelerated in Mn-deficient mice. Exogenous Mn2+ effectively activates the cGAS–STING pathway, significantly promotes the ability of host antigen-presenting cells to present cancer antigens, promotes the infiltration of cytotoxic T cells into tumour tissues, and enhances the specific killing of tumour cells(Reference Lv, Chen and Zhang101). Given these findings, targeted delivery of Mn to cancer sites represents a promising therapeutic strategy for supporting anti-cancer immunity and enhancing the efficacy of cancer treatments. Consequently, the development of efficient Mn delivery systems has become the focus of current research.
Intelligent responsive delivery systems
Due to the immunosuppressive microenvironment (ISME) in cancer tissues(Reference Hayes, Donohoe and Davern123), the clinical efficacy of cancer immunotherapy often falls short of expectations. As a result, responsive nanocarriers for Mn delivery have emerged, particularly targeting the low pH conditions of the tumour microenvironment (TME). The Warburg effect indicates that cancer cells predominantly rely on aerobic glycolysis for energy, resulting in the accumulation of lactate and creating an acidic environment within cancer tissues(Reference Ippolito, Morandi and Giannoni124). In contrast to the pH of approximately 7·4 in normal tissues, the extracellular environment of cancer cells typically exhibits a pH of 6·8, with even lower pH values in endosomes and lysosomes, around 5·0–5·5(Reference Lee, Gao and Bae125). MnO x nanomaterials remain stable in a neutral environment (pH 7·4)(Reference Zhang, Xu and Mu126) but can effectively catalyse endogenous H2O2 to generate O2 in the acidic TME(Reference Luo, Yang and Chen127), alleviating cancer hypoxia. The catalytic reaction is as follows: MnO2 + 2H+ → Mn2+ + 2H2O + ½O2.
MnO2 nanomaterials are unique TME-responsive nanomaterials that have garnered considerable attention due to their high biocompatibility, structural stability and easily modifiable surfaces(Reference Ning, Yang and Yan128). Nanoshells constructed from MnO2 can rapidly decompose in acidic TME, releasing Mn2+ and the anti-cancer drugs loaded within the shell(Reference Zhou, Liang and Zhao129). Polydopamine (PDA), a novel polymer inspired by mussels, offers natural advantages such as good biocompatibility, adhesion and multiple drug response releases. The PDA shell remains stable at pH 7·4 but ruptures upon reaching the cancer site (pH 6·8) due to acidic conditions(Reference Zheng, Zhang and Wang130). Therefore, coating an additional layer of polydopamine (PDA) on the MnO2 nanoshell can serve as a ‘gate’ to control drug release, reducing premature drug release in the bloodstream(Reference Liu, Guo and Li131).
Targeted delivery of Mn
In addition to utilising TME characteristics for Mn delivery, targeting the biological characteristics of cancer cells has also become a significant strategy. Hyaluronic acid (HA), recognised for its excellent biocompatibility and CD44 receptor-mediated cancer-targeting capability, has become a widely used delivery vehicle(Reference Wang, Wang and Jiang132,Reference Catania, Rodella and Vanvarenberg133) . Research indicates that modifying HA on the surface of MnO2 nanosheets enables successful targeting of Mn2+ to cancer sites and its release into the cancer microenvironment(Reference Liang, Wang and Zhao134). Research also found that decorating MnO2 surfaces with gene-engineered exosomes carrying CD47 exhibits good cancer-targeting capabilities(Reference Cheng, Zhang and Liu120). Another common targeted delivery strategy involves loading MnO2 onto nanoparticles coated with the same kind of cancer cell membrane, where the cell membrane coating ensures active cancer targeting, resulting in efficient endocytosis and high accumulation(Reference Luo, Li and Lin135). Furthermore, a study has proposed targeting Mn delivery to the ER, since STING signalling activation and subsequent immune responses are primarily associated with the ER. It reported the design of ER-targeted Mn-based nanocomplexes (NC) by complexing Mn2+ with a zwitterionic polymer, poly(2-(N-oxide-N,N-dimethylamino) ethyl methacrylate) (OPDMA). In mouse models of colon and hepatocellular carcinoma, intravenously injected Mn/OPDMA NC delayed cancer growth rates by 2·4–5 times compared with free-Mn2+-treated mice and extended the survival period of the mice(Reference Zhu, Xu and Geng136).
In recent years, a variety of delivery methods have been developed to precisely deliver Mn to cancer sites, including intelligent responsive delivery systems and targeted delivery systems. These strategies are expected to provide novel therapeutic approaches for patients with cancer. By exploiting the low pH properties of the tumour microenvironment and targeted delivery strategies, scientists are designing vectors that can deliver manganese precisely to the cancer site. These innovative delivery systems not only enhance treatment precision but may also lead to new breakthroughs in cancer treatment in future clinical applications.
Conclusions
In conclusion, Mn is an indispensable trace element in the human body, and an imbalance of Mn homeostasis can lead to changes in human physiological states and many diseases. Mn positively regulates host immunity, mitochondria and microbes to exert anti-cancer effects. In recent years, a variety of delivery methods have been developed to precisely supplement manganese nutrition to cancer sites, including intelligent responsive delivery systems and targeted delivery systems. This is expected to provide new therapeutic strategies for cancer patients. However, the specific mechanism of manganese inhibition of tumours is still unclear, and further studies are needed in the future to explore the therapeutic strategies that manganese can be applied to the clinical treatment of cancer. It is hoped that this review can provide new ideas and programmes for scholars to conduct more in-depth research on Mn.
Availability of data and material
Data sharing is not applicable to this article as no new data were created or analysed in this study.
Acknowledgements
Not applicable.
Authors Contributions
Conceived and drafted the manuscript: Han Shuwen and Liu Jiang. Wrote the paper: Zhuang Jing and Wang Yingchen. Reviewed and compiled the literature: Li Jinyou and Wu Yinhang. Designed and drew figures: Li Jinyou. All authors read and approved the paper.
Financial support
This work was supported by Public Welfare Technology Application Research Program of Huzhou (no. 2022GZB04), Zhejiang Medical and Health Technology Project (nos. 2025KY1531 and 2025KY327) and Zhejiang Province Traditional Chinese Medicine Science and Technology Project (no. 2024ZL1018).
Competing Interests
The authors declare that no potential conflicts of interest exist.
Ethical standards
Not applicable.
Consent for publication
Not applicable.