Sleep disturbances during the early childhood years are tied to risk for a host of negative outcomes, including greater cardiovascular risk (Sun et al., Reference Sun, Wang, Yang, Zhao, Bovet and Xi2020), poorer mental health (Matricciani et al., Reference Matricciani, Paquet, Galland, Short and Olds2019), and poorer neurodevelopmental trajectories across childhood (Bernier et al., Reference Bernier, Beauchamp, Bouvette-Turcot, Carlson and Carrier2013; Dearing et al., Reference Dearing, McCartney, Marshall and Warner2001; Dionne et al., Reference Dionne, Touchette, Forget-Dubois, Petit, Tremblay, Montplaisir and Boivin2011; Kocevska, Muetzel, et al., Reference Kocevska, Muetzel, Luik, Luijk, Jaddoe, Verhulst, White and Tiemeier2017). As a result, better characterizing the developmental origins of sleep disturbance in early childhood can assist with targeted efforts to promote healthy sleep patterns early in life, with cascading benefits to children’s subsequent development (Masten & Cicchetti, Reference Masten and Cicchetti2010; Quach et al., Reference Quach, Nguyen, Williams and Sciberras2018; Williams et al., Reference Williams, Berthelsen, Walker and Nicholson2017). Refining our understanding of the early origins of sleep disturbance is particularly well-suited to developmental psychopathology frameworks (Cicchetti, Reference Cicchetti2016), which have widely focused on the prenatal environment as a programmer of vulnerability to postnatal stress later in life, and hence as potentially presaging offspring’s early sleep problems (Belsky & Pluess, Reference Belsky and Pluess2009; Hellemans et al., Reference Hellemans, Sliwowska, Verma and Weinberg2010; O’Donnell & Meaney, Reference O’Donnell and Meaney2017). At the same time, exploring putative outcomes of this vulnerability process within the early childhood period is particularly salient for identifying timely opportunities for effective early intervention.
The developmental psychopathology lens underscores the critical intersectionality of postnatal risks which can cascade from a stressful prenatal environment (Huizink & De Rooij, Reference Huizink and De Rooij2018; Masten & Cicchetti, Reference Masten and Cicchetti2010). This intersectionality emphasizes the likelihood of not only behavioral risks such as sleep problems, but also co-occurring contextual adversity, which is often also interwoven in offspring’s postnatal development. Recently, an intersectional developmental psychopathology lens emphasized the critical risks of maternal alcohol consumption for offspring’s postnatal outcomes in the context of postnatal adversity (Rockhold et al., Reference Rockhold, Handley and Petrenko2025); a risk which can be particularly challenging to unfurl given substantially variability in its impact, based on timing and dosage of maternal alcohol use (O’Leary et al., Reference O’Leary, Bower, Zubrick, Geelhoed, Kurinczuk and Nassar2010).
Maternal alcohol consumption during pregnancy is a relatively common prenatal stressor; but with variable effects on offspring, based on a combination of prenatal timing and level of use, from heavy drinking to more moderate use levels (Akison et al., Reference Akison, Hayes, Vanderpeet, Logan, Munn, Middleton, Moritz, Reid, Barnett, Crawford, Doney, Elliott, Friend, Goldsbury, Gullo, Harrington, Harris, Hewlett, Hutchinson and Walker2024; O’Leary et al., Reference O’Leary, Bower, Zubrick, Geelhoed, Kurinczuk and Nassar2010) Yet this has risk been surprisingly under-examined when considering its impacts on subsequent childhood sleep disturbance (Chandler-Mather et al., Reference Chandler-Mather, Occhipinti, Donovan, Shelton and Dawe2021; Lund & Ystrom, Reference Lund and Ystrom2022). At the same time, maternal alcohol consumption is well-understood to pose substantial developmental risk, by exposing the developing fetus to teratogens, termed prenatal alcohol exposure (PAE), which can have lifelong effects on growth and neurodevelopment (Popova et al., Reference Popova, Charness, Burd, Crawford, Hoyme, Mukherjee, Riley and Elliott2023; Wozniak et al., Reference Wozniak, Riley and Charness2019). The role of PAE on development has been well-considered as a direct influence on an array of developmental outcomes (Akison et al., Reference Akison, Hayes, Vanderpeet, Logan, Munn, Middleton, Moritz, Reid, Barnett, Crawford, Doney, Elliott, Friend, Goldsbury, Gullo, Harrington, Harris, Hewlett, Hutchinson and Walker2024). However, its role as a programmer of human vulnerability to early life stress, which cascades on to developmental difficulties, including sleep disturbance, has generally been empirically overlooked (Hellemans et al., Reference Hellemans, Sliwowska, Verma and Weinberg2010; Holman & Raineki, Reference Holman and Raineki2023).
PAE places infants and children at risk for a range of developmental difficulties (Jacobson et al., Reference Jacobson, Akkaya-Hocagil, Ryan, Dodge, Richardson, Olson, Coles, Day, Cook and Jacobson2021; Popova et al., Reference Popova, Charness, Burd, Crawford, Hoyme, Mukherjee, Riley and Elliott2023), including unhealthy sleep behaviors and patterns (Chandler-Mather et al., Reference Chandler-Mather, Occhipinti, Donovan, Shelton and Dawe2021; Inkelis & Thomas, Reference Inkelis and Thomas2018; Pesonen et al., Reference Pesonen, Räikkönen, Matthews, Heinonen, Paavonen, Lahti, Komsi, Lemola, Järvenpää, Kajantie and Strandberg2009). Preclinical work suggests that PAE may impact neurobiological systems involved in sleep regulation, via mechanisms including loss of glial cells and neurons that produce or interact with the neurotransmitter GABA, as well as loss and dysfunction of neurons that interact with acetylcholine and serotonin across the brain stem and basal forebrain (Wilson et al., Reference Wilson, Sullivan, Smiley, Saito and Raineki2024). A dysregulated circadian system may also be implicated (Chandler-Mather et al., Reference Chandler-Mather, Shelton, Donovan, Till, Theroux and Dawe2026). In keeping with a developmental cascade perspective, it has been hypothesized that PAE-induced sleep disturbance may in turn compound neurodevelopment impairments associated with PAE across the early developmental period (Chandler-Mather et al., Reference Chandler-Mather, Crichton, Shelton, Harris, Donovan and Dawe2024; Wilson et al., Reference Wilson, Masiello, Lewin, Hui, Smiley and Saito2016, Reference Wilson, Sullivan, Smiley, Saito and Raineki2024).
The role of the postnatal environment in modulating this risk conveyed by PAE for developmental outcomes, such as sleep in childhood, is somewhat unclear. However, there is converging indirect evidence to suggest that postnatal adversity may serve to exacerbate the effects of PAE on sleep and other developmental outcomes (Chandler-Mather et al., Reference Chandler-Mather, Betts, Donovan, Shelton and Dawe2023; Rockhold et al., Reference Rockhold, Kautz-Turnbull, Handley and Petrenko2023). Children with PAE who develop the most severe neurodevelopmental impairments, captured under the diagnostic of Fetal Alcohol Spectrum Disorder (FASD), experience high rates of postnatal adversity relative to the general population (Flannigan et al., Reference Flannigan, Kapasi, Pei, Murdoch, Andrew and Rasmussen2021; Tan et al., Reference Tan, Symons, Fitzpatrick, Connor, Cross and Pestell2022). Greater exposure to postnatal adversity has been associated with greater sleep disturbance and behavioral difficulties among children with FASD (Chandler-Mather et al., Reference Chandler-Mather, Betts, Donovan, Shelton and Dawe2023; Rockhold et al., Reference Rockhold, Kautz-Turnbull, Handley and Petrenko2023). A recent meta-analytic review found that the detrimental effects of PAE on neurodevelopment were attenuated in a middle-class cohort, suggesting a role for potentially more cognitively stimulating postnatal environments in modulating risk (Jacobson et al., Reference Jacobson, Akkaya-Hocagil, Ryan, Dodge, Richardson, Olson, Coles, Day, Cook and Jacobson2021). Moreover, PAE appears to alter neurobiological systems that respond to threat, including the structure and functioning of the amygdala (Cullen et al., Reference Cullen, Burne, Lavidis and Moritz2013; Raineki et al., Reference Raineki, Morgan, Ellis and Weinberg2019) and the regulation of HPA axis activity, which may lead to more dysregulated responses to threatening inputs from the environment (Haley et al., Reference Haley, Handmaker and Lowe2006; Maxwell et al., Reference Maxwell, DiDomenico, Roberts, Marquez, Rai, Weinberg, Jacobson, Stephen and Bakhireva2023; McLachlan et al., Reference McLachlan, Rasmussen, Oberlander, Loock, Pei, Andrew, Reynolds and Weinberg2016). Thus, PAE may negatively affect development in the context of postnatal adversity, but the role of postnatal adversity in exacerbating the effects of different levels of PAE on sleep has not been explicitly tested.
Additionally, the risk conveyed by PAE may depend on the distinct patterning of alcohol use across pregnancy (Bandoli et al., Reference Bandoli, Coles, Kable, Wertelecki, Yevtushok, Zymak-Zakutnya, Wells, Granovska, Pashtepa and Chambers2019, Reference Bandoli, Jones, Wertelecki, Yevtushok, Zymak-Zakutnya, Granovska, Plotka, Chambers and Cifasd2020; Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020; Muggli et al., Reference Muggli, Matthews, Penington, Claes, O’Leary, Forster, Donath, Anderson, Lewis, Nagle, Craig, White, Elliott and Halliday2017). PAE has typically been measured by average number of standard drinks or ounces of alcohol per week, averaged across pregnancy or by trimester (O’Leary et al., Reference O’Leary, Bower, Zubrick, Geelhoed, Kurinczuk and Nassar2010). However, this approach ignores the pattern of alcohol use across the trimesters, and how this might dynamically influence fetal brain development and programing. More recently, distinct patterns of alcohol use have been extracted from maternal-reported alcohol use across pregnancy, which have been associated with distinct postnatal dysmorphology and neurodevelopment (Bandoli et al., Reference Bandoli, Coles, Kable, Wertelecki, Yevtushok, Zymak-Zakutnya, Wells, Granovska, Pashtepa and Chambers2019, Reference Bandoli, Jones, Wertelecki, Yevtushok, Zymak-Zakutnya, Granovska, Plotka, Chambers and Cifasd2020; Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020; Muggli et al., Reference Muggli, Matthews, Penington, Claes, O’Leary, Forster, Donath, Anderson, Lewis, Nagle, Craig, White, Elliott and Halliday2017). For example, high and, surprisingly, low sustained use of alcohol across pregnancy was shown to predict mental and psychomotor development at 6 and 12 months (Bandoli et al., Reference Bandoli, Coles, Kable, Wertelecki, Yevtushok, Zymak-Zakutnya, Wells, Granovska, Pashtepa and Chambers2019). Both low and high sustained use patterns, along with moderate to heavy use at the beginning of pregnancy before reducing use, were associated with greater dysmorphology and other physical health-related outcomes at 6 to 12 months of age (Bandoli et al., Reference Bandoli, Jones, Wertelecki, Yevtushok, Zymak-Zakutnya, Granovska, Plotka, Chambers and Cifasd2020). Various alcohol use patterns have also been associated with poorer neurodevelopmental and behavioral outcomes during middle childhood, including those noted above, along with light use before reducing (Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020). As a result, taking a pattern-based approach to examining the effects of PAE on developmental outcomes may uncover distinct risk “profiles” that convey heightened risk for sleep disturbance in children.
Importantly, postnatal adversity may then exacerbate such risk arising from patterning of PAE. That is, the impact of risk associated with patterns of PAE may depend on the features or characteristics of adversity within the postnatal environment (McLaughlin et al., Reference McLaughlin, Sheridan, Humphreys, Belsky and Ellis2021; McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016). In the past, a dominant approach to operationalizing postnatal adversity has been to create a cumulative risk index that adds the presence or absence of a basket of risk factors for adverse development (Evans et al., Reference Evans, Li and Whipple2013; McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016). Illustratively, a popular index using this cumulative risk approach has been the adverse childhood experiences index, which adds exposures to discrete experiences of abuse, neglect, and household dysfunction into a single score (Felitti et al., Reference Felitti, Anda, Nordenberg, Williamson, Spitz, Edwards, Koss and Marks1998) and uses this score to predict outcomes. While this approach has been effective in highlighting the diffuse impacts of adversity on health and developmental outcomes (Evans et al., Reference Evans, Li and Whipple2013), it assumes that the noxious effects of adversity on outcomes are mediated by a single mechanism, often referring to allostatic load (Danese & McEwen, Reference Danese and McEwen2012) or a general stress response system, which disregards the potential differential impacts of these adverse experiences on developmental outcomes (McLaughlin et al., Reference McLaughlin, Sheridan, Humphreys, Belsky and Ellis2021).
In contrast, the dimensional model of adversity and psychopathology (DMAP), seeks to address the multifinality that can arise from early adversity exposure (McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014; McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016). DMAP parses adversity into two cardinal dimensions of adversity, including threat, characterized by experiences of harm or threat of harm to the child, and deprivation, characterized by the absence of expected physical or social inputs from the environment (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016). Here, the threat dimension is thought to affect child outcomes via threat processing systems, while the deprivation dimension is thought to affect child outcomes via disruptions to reward and pattern learning circuitry and executive and language-based systems (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016); though both may also activate a more general stress response system (McLaughlin et al., Reference McLaughlin, Sheridan, Humphreys, Belsky and Ellis2021).
Reviews and meta-analyses of DMAP research find that threat and deprivation are often associated with divergent outcomes across domains of neurodevelopment, such that threatening adversity demonstrates marked associations with amygdala volumes (McLaughlin et al., Reference McLaughlin, Weissman and Bitrán2019), psychopathology (Lee et al., Reference Lee, Kitagawa, Mirhashem, Rodriguez, Hilerio and Bernard2024), and fear conditioning with putative links to daily stress responses (Modecki et al., Reference Modecki, Murphy and Waters2020, Reference Modecki, Ryan and Waters2023). Reference Modecki, Murphy and Waters While deprivation demonstrates associations with prefrontal brain network activity, language, and executive functioning (McLaughlin et al., Reference McLaughlin, Sheridan and Nelson2017, Reference McLaughlin, Weissman and Bitrán2019). While a recent meta-analysis of 127 studies demonstrated stronger effects of threat exposure on internalizing and externalizing psychopathology than deprivation (Lee et al, Reference Lee, Kitagawa, Mirhashem, Rodriguez, Hilerio and Bernard2024), the application of this approach to understanding developmental impacts resulting from adversity during the sensitive period of infancy has been relatively under investigated compared to toddlerhood, middle childhood, and adolescence (Lee et al, Reference Lee, Kitagawa, Mirhashem, Rodriguez, Hilerio and Bernard2024). Similarly, the application of this approach to sleep outcomes has generally been limited (Chandler-Mather et al., Reference Chandler-Mather, Betts, Donovan, Shelton and Dawe2023; Vaughn-Coaxum et al., Reference Vaughn-Coaxum, Dhawan, Sheridan, Hart and Weisz2020), despite sleep representing a potent developmental precursor for internalizing and externalizing psychopathology (Quach et al., Reference Quach, Nguyen, Williams and Sciberras2018).
These gaps are surprising, given that postnatal adversity of threat and deprivation can lead to divergent developmental outcomes that are mediated by neural processing, including those related to sleep (McLaughlin et al., Reference McLaughlin, Weissman and Bitrán2019). That is, the context of threat versus deprivation within the subsequent postnatal environment may serve to condition the impact of distinct patterns of PAE on children’s sleep outcomes. In relation to early childhood sleep, it is plausible that threat may be especially salient in terms of disruptive mechanisms, with deprivation still moderating the impact via self-regulation and other skills, though perhaps to a lesser extent (Holman & Raineki, Reference Holman and Raineki2023). Notably, these hypotheses also align with an intersectional developmental psychopathology lens, given the potential heterogeneity in impact attributable to distinct aspects of early adversity experiences (Popova et al., Reference Popova, Lange, Probst, Gmel and Rehm2017).
In sum, the current study addresses this intersectionality of postnatal risks which can cascade from the stressful prenatal environment (Rockhold et al., Reference Rockhold, Handley and Petrenko2025). The study addresses the likelihood of subsequent early behavioral risks arising from PAE, in the form of sleep problems, as well also co-occurring early contextual adversity. In particular, we address how early life adversity (threat vs deprivation during infancy) modulates the risk that distinct patterns of PAE pose to sleep outcomes at 3 years of age, an important period for sleep regulation and neural growth and consolidation (Lokhandwala & Spencer, Reference Lokhandwala and Spencer2022). Sleep at the junction between the toddlerhood and preschool years has been demonstrated to shape emerging emotion regulation (Berger et al., Reference Berger, Miller, Seifer, Cares and Lebourgeois2012; Miller et al., Reference Miller, Seifer, Crossin and Lebourgeois2015), executive functioning (Bernier, Cimon-Paquet, & Tétreault, Reference Bernier, Cimon-Paquet, Tétreault, Berger, Harbourne and Scher2021), and academic achievement abilities (Bernier, Cimon-Paquet, Tétreault, et al., Reference Bernier, Cimon-Paquet, Tétreault, Berger, Harbourne and Scher2021), making this a salient outcome for investigation.
The study leverages a smaller birth cohort study focused on mothers living in an area of socio-economic disadvantage in Australia, to derive distinct patterns of PAE (based on previous work; see Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020; Muggli et al., Reference Muggli, Matthews, Penington, Claes, O’Leary, Forster, Donath, Anderson, Lewis, Nagle, Craig, White, Elliott and Halliday2017). We test the hypothesis that risks associated with specific prenatal alcohol patterns are exacerbated by experiences of postnatal adversity at 12 months of age, leading to poorer sleep outcomes at 3 years. Previous work has identified three patterns that conferred the greatest risk for development outcomes at age 12 months (Bandoli et al., Reference Bandoli, Coles, Kable, Wertelecki, Yevtushok, Zymak-Zakutnya, Wells, Granovska, Pashtepa and Chambers2019) i) heavy consumption throughout pregnancy; ii) heavy consumption in trimester 1 before reducing use; and iii) stable low alcohol consumption across pregnancy, Thus, we expected similar patterns would confer the greatest risk for poor sleep outcomes at 3 years in the context of postnatal adversity (namely threat exposure and to a lesser extent, deprivation). Additionally, given likely impacts of early life (age 12 months) adversity on developmental health, we hypothesized that early adversity would be directly associated with worse sleep outcomes in toddlerhood (age 3 years). We posited, again, that this relation might be potentially stronger in relation to threat exposure than in relation to deprivation (e.g. Lee et al., Reference Lee, Kitagawa, Mirhashem, Rodriguez, Hilerio and Bernard2024).
Method
Study sample
Data were derived from three relevant waves from the Environments for Healthy Living (EFHL) study, a prospective longitudinal birth cohort study designed to study mother-infant dyads from before birth to age 5 years across a range of eco-epidemiological factors (Cameron et al., Reference Cameron, Scuffham, Spinks, Scott, Sipe, Ng, Wilson, Searle, Lyons, Kendall, Halford, Griffiths, Homel and McClure2012). In the current study, prenatal (T0), 12-month (T1) and 36-month (T2) data were examined. In this case, 36 months (3 years) was selected as the key outcome age, given the inclusion of relevant sleep outcomes at this wave (sleep duration and waking).
Mothers aged 16 years and over were recruited by research trained nurses at their third trimester antenatal appointments at one of three public maternity hospitals across South-East Queensland and New South Wales, Australia. Mothers completed questionnaires at baseline (T0; M = 35.69 weeks, SD = 3.36), during their antenatal visit, and then at prospective follow ups at approximately 12 months (T1), 3 years (T2), and 5 years postnatal. Birth outcome data was also extracted from hospital records. Offspring follow-up measures were derived from maternal-report data at 12 months (T1) and 3 years (T2). Ethics approval for the EFHL was obtained from the University Human Research Ethics Committee (HREC) (Ref: MED/16/06/HREC, renewed MED/23/11/HREC) and for use of the data in the current study (Ref: 2023/228).
Within the current study, mothers were included from cohorts recruited 2006 to 2010 who provided complete baseline data (T0) for prenatal drug and alcohol exposures and basic demographics (Table 2 and Supplementary Table 1), given our focus on prenatal exposure to alcohol. In order to reduce potential confounds from emergent neurodevelopmental or medical conditions post-birth, data were further screened to exclude children who were reported to have neurodevelopmental or serious medical conditions by 3 years, including epilepsy, developmental delay, ADHD or ASD, meningitis, or a genetic disorder (n = 36), resulting in a final sample of n = 1952. Sensitivity checks indicated no substantive difference in results between the full sample and the screened sample.
Measures
Covariates
Key constructs collected at T0 were controlled for across analyses (see Table 2 and Supplementary Table 1). These included maternal baseline data related to number of weeks of pregnancy at recruitment, age, marital status, and pre-pregnancy body mass index (BMI). Maternal use of tobacco, marijuana, cocaine, and methamphetamine during pregnancy, reported at T0 (yes/no) were also controlled. Further, child sex, available infant birth weight, length, head circumference, and gestational age data were extracted from hospital records and included as relevant child covariates. Postnatal cigarette smoke exposure was reported at T1 (“how often is the baby in a room where other people are smoking?,”) any smoking coded as “yes,” not at all coded as “no.” Postnatal alcohol use was reported at T1 (“how often do you have 5 or more alcoholic drinks on one occasion?”), any binge use coded as “yes,” no use coded as “no.”
Zip code data were used to derive a standardized measure of socioeconomic disadvantage (ABS Index of Relative Socio-economic Disadvantage; SES index), which summarizes social and economic conditions across geographic zip codes via census variables of income, employment, education, and housing. The SES index is categorized by deciles (1 = most disadvantaged; 10 = least disadvantaged). Maternal education level was also recorded; as described in Supplementary Table 1, 51.6% of mothers reported a high school education or less and 80.2% of mothers reported completing trade school or less education. Hence mothers were characterized as having lower levels of education while living in heterogenous socioeconomic settings.
Independent variables
Patterns of PAE. Patterns of maternal alcohol use during pregnancy were constructed to address the role of varying levels and timing of PAE across pregnancy. At baseline, mothers provided estimates of their frequency (how often they drank per week) and quantity (how many standard drinks they consumed on each occasion) of alcohol use for each trimester of their pregnancy on a questionnaire (as per O’Leary et al., Reference O’Leary, Bower, Zubrick, Geelhoed, Kurinczuk and Nassar2010) using items from the National Drug Household Strategy survey that were adapted for pregnant women (Cameron et al., Reference Cameron, Davey, Kendall, Wilson and McClure2013). The questionnaire included images and information on different types of alcohol and what constitutes a standard drink in Australia (10 mg of pure alcohol).
A composite method of prenatal alcohol classification was then used to assign categories of risk to trimester 1 (Tri1), trimester 2 (Tri2), and trimester 3 (Tri3) of pregnancy for each mother (O’Leary et al., Reference O’Leary, Bower, Zubrick, Geelhoed, Kurinczuk and Nassar2010). Based on previous research which was guided by the cutoffs from the National Health and Medical Research Council (O’Leary et al., Reference O’Leary, Bower, Zubrick, Geelhoed, Kurinczuk and Nassar2010), within each trimester, mothers were assigned to an alcohol use category: abstainer (no alcohol use), low (<7 drinks per week and a maximum of 1–2 drinks per occasion), moderate (< = 7 drinks per week and a maximum of 3–4 drinks per occasion), binge drinking (drinking up to 1–2 times per week and >= 5 drinks per occasion), and heavy (>7 drinks per week). Next, overall patterns of prenatal alcohol use across all three trimesters were constructed in accordance with patterns previously identified in the literature (Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020) and based on observed patterns within the sample in order to achieve adequate sample sizes for each group (Dalmaijer et al., Reference Dalmaijer, Nord and Astle2022).
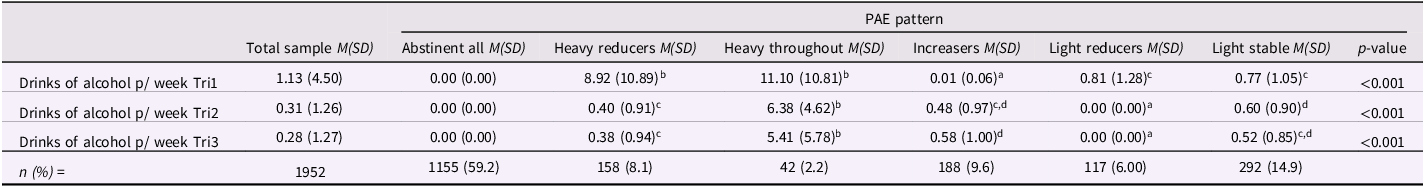
The six derived PAE patterns are characterized in Table 1, including descriptive statistics summarizing of alcohol use by trimester. The six PAE groups were as follows: (1) Abstinent (Abstinent across all three trimesters; 59.2%) (2) Light reducers (Low in Tri1, abstinent Tri2 and Tri3; 6.0%) (3) Heavy reducers (Moderate, heavy, or binge in Tri1, abstinent in Tri2 and Tri3 (n = 88); or else low in Tr2 and/or Tri3 (n = 70); 8.1%) (4) Stable light (Low in Tri1, and drinking at low levels in at least one of Tri2 and Tri3; 14.9%) (5) Increasers (Abstinent or low in Tri1, abstinent, low, or moderate in Tri2 and Tri3 (n = 182); or else low in Tri1 and abstinent, low, or moderate in Tri2 and Tri3 (n = 6); 9.6%) (6) Heavy throughout (Moderate, heavy, or binge in all Tris (n = 18) or else Moderate, heavy, or binge in Tri1 and in at least one other Tri (n = 15); or else abstinent or low in Tri1 and Moderate, heavy, or binge in at least one other Tri (n = 9); 2.2%).
Table 1. Average standard drinks of alcohol per week across each trimester by PAE pattern

a,b,c,d. Within rows, means without a common superscript significantly differ at p < .05.
Moderating variables
Threat and deprivation indices. Indices of threat and deprivation were constructed via data provided at the first follow-up at 12 months post-partum (T1). Postnatal adversity indices were then operationalized based on published examples within DMAP (McLaughlin et al., Reference McLaughlin, Sheridan, Humphreys, Belsky and Ellis2021; McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016) and following studies that have constructed similar indices from indicator variables (e.g., Colich et al., Reference Colich, Platt, Keyes, Sumner, Allen and McLaughlin2020; Usacheva et al., Reference Usacheva, Choe, Liu, Timmer and Belsky2022; Vogel et al., Reference Vogel, Perry, Brandes-Aitken, Braren and Blair2021).
To index threat exposure, seven different indicators were employed. Five indicators (coded as 1 for yes and 0 for no) were derived from the Family Environment Scale (Moos & Moos, Reference Moos and Moos1994), capturing experiences of threat in the home environment: 1) “Do family members sometimes hit each other,?” 2) “Do family members sometimes get so angry they throw things,?” 3) “We fight a lot in our family,” 4) “Family members rarely become openly angry” (reverse coded), and 5) “Family members hardly ever lose their tempers” (reverse coded). Further, one indicator captured experiences of threats to bodily integrity (Child taken to doctor, emergency department, etc. due to accident/injury? Coded as 0 for no, 1 for once or more). Lastly, one indicator captured threatening experiences at the community level (“I feel safe in this community,” ratings of “disagree” and “strongly disagree” rated as a 1 and “neither” to “strongly agree” rated as 0).
Likewise, to index deprivation exposure, five separate indicators were employed. Two indicators captured risk factors for challenging or insufficient provision of caregiving: postnatal depression (“Did you experience postnatal depression for a period of 2 weeks or long?”; yes coded as 1), and a period of being a single carer since birth (yes coded as 1). A single indicator captured financial security: “How well are you managing financially since birth?”, with ratings of “quite difficult” and “very difficult” coded as a 1 and “just getting by” to “comfortable” coded as a 0. Lastly, the deprivation index also included two indicators which captured insufficient access to resources within the community: 1) “I am able to get the services I need in my community” (ratings of “disagree” and “strongly disagree” rated as a 1 and “neither” to “strongly agree” rated as 0), and 2) “I can get help from friends & neighbors when I need it” (ratings of “disagree” and “strongly disagree” rated as a 1 and “neither” to “strongly agree” rated as 0).
Overall, threat index scores ranged from 0 to 6 (M = 1.03, SD = 1.21) and the deprivation index scores ranged from 0 to 4 (M = 0.48, SD = 0.75). The threat index score was significantly, but moderately, correlated with the deprivation index score (r(1238) = .22, p < .001) and was marginally correlated with SES based on zip-code (r(1256) = − .06, p = .051). The deprivation score was not significantly associated with zip-code SES (r(1239) = −.021, p = .466).
Outcome variables
Child sleep outcomes were assessed at T2 (3 years of age) and are characterized in Table 2. Mothers provided information on their child’s total sleep duration (sum of responses to “on average, how many hours does your child sleep at night?” and “on average, how many hours does your child sleep during each day?,”) night waking (“on average, how many times does your child wake during the night?,”) and nightmares (“does your child have bad dreams or nightmares?”; rated as a frequency as either never, rarely, sometimes, often, or almost always).
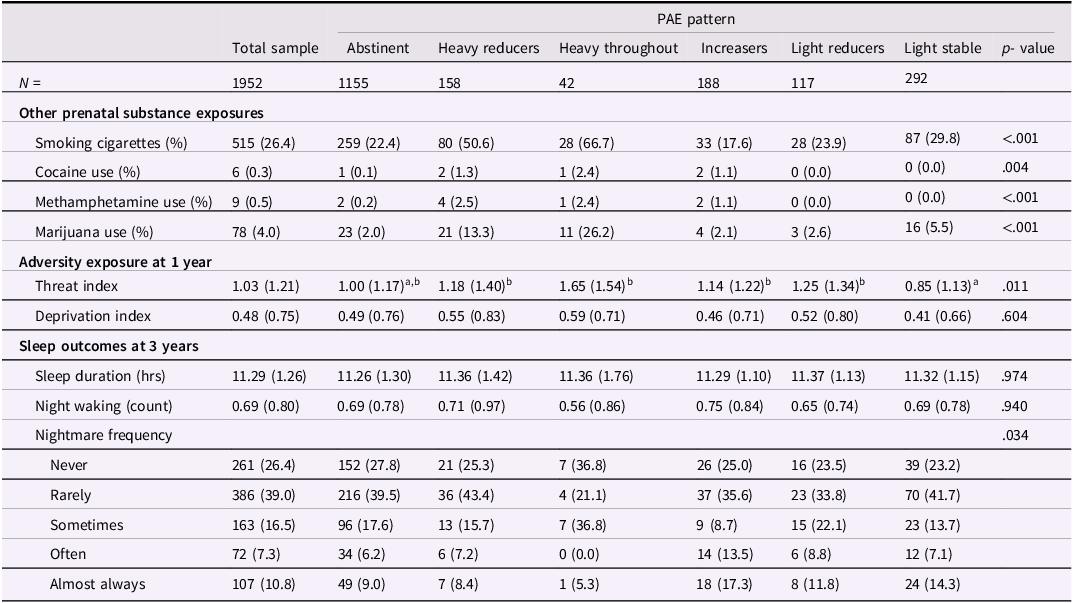
Table 2. Other prenatal exposures, adversity index scores, and sleep outcomes for the total sample and by PAE pattern

Note. Other prenatal substance exposures across pregnancy, 1 = yes, 0 = no; Threat index range from 0 to 6 (M = 1.03, SD = 1.21) and the deprivation index range from 0 to 4 (M = 0.48, SD = 0.75).
Planned analyses
Statistical analyses were conducted using R statistical software version 4.2.3 (R Project for Statistical Computing) (R Core Team, 2023). Analysis scripts are available at OSF: https://osf.io/mpt8u. Researchers requesting access to the data may contact the corresponding authors for assistance in accessing data and materials, given current restrictions related to public data access.
ANOVA was used to test for significant differences in drinking at each trimester by PAE pattern; Welch two sample t-tests were used to compare two patterns. Separate regression models (detailed next) then examined the effects of PAE and threat and deprivation indices on subsequent child sleep (total sleep duration, waking, and nightmares). Sleep duration was modeled using linear models, number of night wakings using negative binomial models, and frequency rating of nightmares using ordinal models. Benjamin-Hochberg correction was applied across all main effect and omnibus moderated regression models to control the false discovery rate at 5%.
Next, we took a gated approach to address whether PAE was moderated by postnatal adversity, following previous research investigating patterning effects of PAE (Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020). First, in an omnibus test, PAE was examined as a binary variable (exposed vs unexposed reference group) in relation to each sleep outcome. Two separate omnibus models were conducted as follows: i) the PAE (binary) x threat index term, conditional main effects, and covariates, and ii) the PAE (binary) x deprivation index term, conditional main effects and covariates. Across all regression models, covariates controlled for number of weeks of pregnancy at recruitment, maternal education, SES index, maternal age, maternal pre-pregnancy BMI, marital status, cocaine use in pregnancy, marijuana use in pregnancy, methamphetamine use in pregnancy, postnatal cigarette smoke exposure, postnatal alcohol use, child sex, birth weight, birth length, head circumference, and gestational age. Second, when a given interaction term was statically significant (with FDR correction applied) and significantly improved model fit (relative to the main effects model), follow-up analyses examined whether there were different effects associated with varying patterns of alcohol use through pregnancy (i.e., PAE pattern) conditioned by threat and/or deprivation. This gated approach was applied to further safeguard against inflated Type 1 error (see Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020). Thus, follow-up models addressed specific PAE patterns and included five dummy coded variables representing each of the PAE pattern groups with children of mothers characterized by the abstinent pattern serving as the reference group. All analyses controlled for key study covariates. Across all models, variance inflation factors did not exceed 2 indicating very low collinearity among predictors.
Missing data
Data were missing on several baseline variables and at follow-up time points, T1 and T2. Little’s MCAR test was significant, indicating that data did not meet the MCAR assumption (χ 2(2366) = 6,069.262, p < .001). Missingness was related to lower maternal age, less married/de facto partnerships, less maternal education, lower zip code SES, lower birth weight and gestational age, smaller birth head circumference, and greater cigarette and marijuana use reported during pregnancy (ps < .05), all of which were included in our models as covariates (Enders, Reference Enders2022). Further there was no significant difference between those with complete data across all time points and those with missing data at T1 who reengaged in T2 across the sleep outcome measures (p > .05). Given this, data were considered to be consistent with an assumption of Missing at Random (MAR). In total, there were 910 (46.62%) participants that returned data at T1 and T2, 597 (30.58%) that dropped out at T1, 350 (17.93%) that dropped out at T2, but 95 (4.87%) that had dropped out at T1 and reengaged at T2.
Multiple imputation by chained equations (MICE) was implemented using the “mice” package (Buuren & Groothuis-Oudshoorn, Reference Buuren and Groothuis-Oudshoorn2011) to estimate missing values and to avoid biased estimates of regression parameters owing to missing data. All variables included in the regression models, including those that predicted missingness, were used in missing data models. To avoid statistical power falloff and to generate standard error distributions that are robust to the rate of attrition present in this longitudinal study, 100 datasets were imputed (Graham et al., Reference Graham, Olchowski and Gilreath2007). Because the analysis model included interaction terms between PAE pattern and the threat and deprivation indexes, the imputation dataset was divided into strata defined by PAE pattern, then the separate datasets were imputed 100 times each and remerged. This approach ensures that the imputation model preserves the assumed correlational structure of the data and avoids biased parameter estimates (Tilling et al., Reference Tilling, Williamson, Spratt, Sterne and Carpenter2016). The threat and deprivation index scores were constructed after imputation to further reduce estimate bias (Eekhout et al., Reference Eekhout, de Vet, Twisk, Brand, de Boer and Heymans2014). Subsequently, the three sets of regression models (for sleep duration, night waking, and nightmares) were run separately on each imputed dataset. The coefficient and standard error estimates from each model were pooled across datasets and are reported. Model fit was compared using the “D1” function in the “mice” package, akin to a multivariate Wald test (Buuren & Groothuis-Oudshoorn, Reference Buuren and Groothuis-Oudshoorn2011).
Results
Descriptive characteristics and birth outcomes
Other prenatal exposures, threat and deprivation index scores, and sleep outcomes by PAE pattern are presented within Table 2, while demographic characteristics and birth outcomes are presented in Supplementary Table 1. Maternal participants represented the SES spectrum, though over 40% of participants belonged to low SES households based on national zip code data (lowest 40 percentile). There was also notable variability in PAE, with trimester 1 characterized by the most variation. On average, cohort mothers in the study were 29.20 years (SD = 6.02), and the majority (51.6%) reported an education level of high school or less; 87.4% of maternal participants were born in Australia or the UK (England, Scotland, Wales, Ireland), and 2.1% of maternal participants identified as Aboriginal and/or Torres Strait Islander.
Infants exposed to heavy throughout drinking patterns in utero exhibited significantly smaller head circumferences at birth relative to children of mothers with no PAE (t(36.35) = 2.221, p = .033, Cohen’s d = 0.27). Whereas smaller birth length and lower birth weight among infants exposed to heavy throughout drinking patterns compared to children of mothers with no PAE reached only trend-level significance (t(32.45) = 2.021, p = .052, Cohen’s d = 0.44; t(46.11) = 1.942, p = .058, Cohen’s d = 0.24, respectively). No other significant differences emerged in birth outcomes by pattern of PAE.
Sleep duration
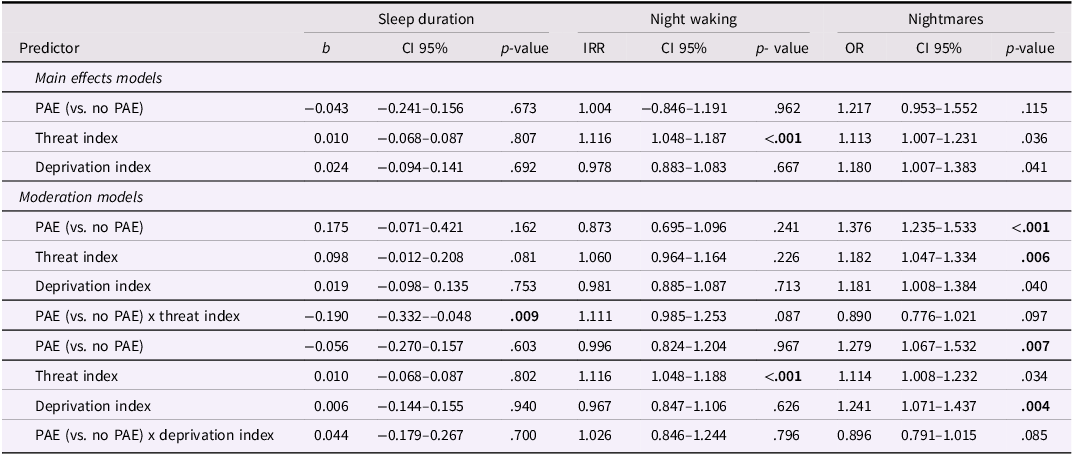
There were no significant main effects of PAE (binary), threat, or deprivation on sleep duration at three years (see Table 3). The PAE x deprivation index interaction term was statistically non-significant; whereas the PAE x threat index was significant (b = –0.190, p = .009) and significantly improved model fit relative to the main effects model F(1,250.259) = 6.930, p = .009. Probing the significant interaction term indicated that the effect of any PAE on sleep duration was moderated by threat index, whereby children with any PAE experienced a 5.52-minute reduction in sleep duration at three years for each additional threat exposure at one year, relative to children with no PAE (the effect of threat index for children with no PAE (i.e. the reference group) b = 0.098, p = .081).
Table 3. Summary of results from main effects and moderation models

Note. Significant p values after Benjamin-Hochberg correction are bolded. Horizontal lines separate models. All models included covariates listed in methods.
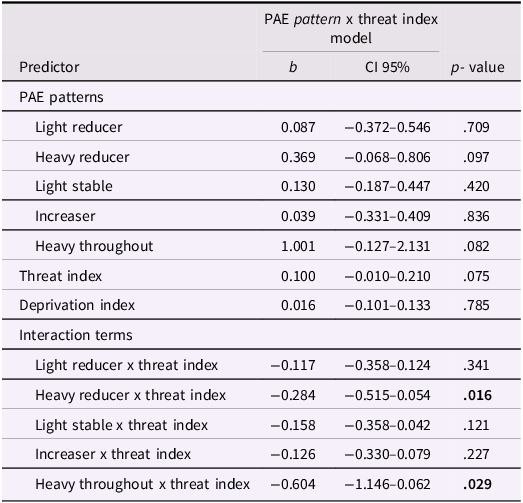
Next, we followed up the omnibus binary moderation model with a PAE pattern x threat index moderation model, to determine whether particular patterns of PAE were driving the finding (see Table 4 and Figure 1). Follow-up analysis indicated that the heavy reducer and heavy throughout patterns were significantly moderated by postnatal threat exposure. Children with maternal heavy reducer pattern exhibited a 11.04-minute reduction in sleep duration at three years with each additional threat exposure at one year, relative to children with no PAE. Children with the heavy throughout pattern exhibited a 30.24-minute reduction in sleep duration at three years with each additional threat exposure at one year relative to children with no PAE. The slope of the threat index did not significantly differ between the heavy throughout and heavy reducer patterns, Δb = −0.320, t(df = 192) = −1.114, p = .267.

Figure 1. The effect of PAE pattern on sleep duration (age 3) conditioned by threat exposure (12 months). The interactions with the heavy reducer (yellow) and heavy throughout (red) groups were statistically significant (p < .05).
Table 4. Results from PAE pattern x threat index predicting sleep duration at three years

Note. All terms entered into the model concurrently. p < .05 bolded.
Night waking
Th main effect of PAE on the night waking frequency at three years was statistically non-significant (see Table 3). However, there was a significant main effect for threat index, indicating an estimated 11.6% increase in the frequency of night wakings at three years for each additional threat exposure at one year of age. The main effect of deprivation index on night waking at 3 years was statistically non-significant. Additionally, neither the PAE (binary) x threat index model nor the PAE (binary) x deprivation index model were statistically significant; hence no follow-up models to examine PAE patterns were conducted.
Nightmares
There main effect of PAE on the frequency of nightmares at three years was statistically non-significant (see Table 3). Additionally, the main effects of threat index and deprivation index no longer reached statistical significance once the Benjamin-Hochberg correction was applied. Neither the PAE (binary) x threat index model nor the PAE (binary) x deprivation index model significantly improved model fit, F(1,699.845) 2.766, p = .097 and F(1,956.633) = 2.978, p = .085, respectively; hence no follow-up models to examine PAE patterns were conducted.
Sensitivity analyses
Sensitivity analyses were conducted to test whether the threat x PAE effect on sleep duration was isolated to alcohol consumption patterns during pregnancy and were not also present for other substance use patterns in pregnancy or post-partum. The threat index x prenatal cigarette, threat index x prenatal marijuana, threat index x postnatal alcohol use, and threat index x postnatal smoke exposure effects (all run in separate models with covariates used in main analyses along with PAE (binary)) on sleep duration at 3 years were all statistically non-significant (ps > .05).
Discussion
An intersectional developmental psychopathology lens underscores children’s co-occurring postnatal risks, which often cascade from stressful prenatal environments (Rockhold et al., Reference Rockhold, Handley and Petrenko2025). By focusing a lens on potential co-occurring risks, such as PAE and coinciding contextual adversity (such as experiences of threat and/or adversity), the current study sought to address the heightened risks that can be interwoven into offspring’s postnatal development. Within the study, we adopted this intersectional lens to address the ongoing risks of maternal alcohol consumption patterns for offspring’s postnatal outcomes (Rockhold et al., Reference Rockhold, Handley and Petrenko2025). In particular, we hypothesized that negative impacts on toddler sleep outcomes resulting from PAE patterns would be stronger among children who were also exposed to adversity in the form of threat or deprivation in the first year of life.
Overall, we found preliminary support for such intersectionality of risks in predicting toddler sleep outcomes–early exposure to threat worsened risk for shortened sleep duration in the context of risky maternal alcohol use patterning, albeit with small to moderate effect sizes. Notably, too, distinct PAE profiles showed specific validity (albeit with modest effect sizes), highlighting the role of both timing and dosage of maternal alcohol use for offspring outcomes three years later. In the context of threat exposure, heavy throughout and heavy reducer patterns were associated with shortened sleep duration, whereas lighter use patterns were not. Additionally, early threat exposure was independently associated with increased night waking (moderate effect size) at age three years, further highlighting the pernicious effect of early threatening environments, even in the context of major co-occurring risk.
Before unpacking these findings in further detail, it is worth re-iterating the challenges associated with examining outcomes of PAE due to substantial heterogeneity in its impact on children, based on difference in timing and dosage of maternal use (Popova et al., Reference Popova, Lange, Probst, Gmel and Rehm2017). In the current study, with a goal of greater developmental specificity, we adopted a relatively unique approach to this challenge, through the generation of distinct maternal alcohol use patterns based on the pregnancy trimesters. Indeed, recent studies using a similar pattern-based approach to the classification of PAE have found that patterns comparable to heavy throughout and heavy reducer were associated with significantly greater dysmorphology, delayed physical growth (Bandoli et al., Reference Bandoli, Jones, Wertelecki, Yevtushok, Zymak-Zakutnya, Granovska, Plotka, Chambers and Cifasd2020), and lower cognitive developmental (Bandoli et al., Reference Bandoli, Coles, Kable, Wertelecki, Yevtushok, Zymak-Zakutnya, Wells, Granovska, Pashtepa and Chambers2019) at 12 months. In the current study, we detected direct effects of the heavy throughout pattern on neonatal growth restriction, an established effect of heavy PAE (Akison et al., Reference Akison, Hayes, Vanderpeet, Logan, Munn, Middleton, Moritz, Reid, Barnett, Crawford, Doney, Elliott, Friend, Goldsbury, Gullo, Harrington, Harris, Hewlett, Hutchinson and Walker2024). Additionally, though we did find evidence of negative impacts from our heavy throughout and heavy reducer pattern, this was found only in the context of exposure to early experience of threat.
Our central study findings is that the impact of these risky patterns of PAE (heavy throughout and heavy reducer) on reduced sleep duration at three years, is found only in the context of early threat exposure. When experiencing two or more threating experiences at one year of age, among children experiencing these risky PAE patterns, sleep duration evidences steep declines. Additionally, while statistically similar, the slope was relatively steeper for those experiencing the heavy throughout pattern compared to the heavy reducer pattern. This may suggest that reducing drinking in trimesters two and three may somewhat mitigate postnatal biological sensitivity to threat, although harmful effects still persist. The biological pathways that might underpin the conditioning of heavy PAE patterns by threat exposure are unclear, but may involve the effects of PAE on the HPA axis and amygdala, which exhibit dysfunction following PAE (Cullen et al., Reference Cullen, Burne, Lavidis and Moritz2013; McLachlan et al., Reference McLachlan, Rasmussen, Oberlander, Loock, Pei, Andrew, Reynolds and Weinberg2016). These findings require replication among additional samples; should they be replicated, this suggests that heavy patterns of alcohol exposure may potentially be considered fetal programmers of susceptibility to threatening environmental inputs in infancy (Hellemans et al., Reference Hellemans, Sliwowska, Verma and Weinberg2010; O’Donnell & Meaney, Reference O’Donnell and Meaney2017).
Notably, early threat exposure directly predicted greater night waking at three years regardless of PAE. Together with the finding that threat appears to condition the effect of PAE on sleep duration, results are consistent with studies highlighting that children are highly sensitive to environmental exposures to threat during infancy (Opendak et al., Reference Opendak, Gould and Sullivan2017; Opendak & Sullivan, Reference Opendak and Sullivan2019). The neural systems responsible for responding to stress and threat undergo rapid development in the first year of life. Thus, infancy arguably represents an especially sensitive period for stressful events, and in particular, exposure to threatening events (Gabard-Durnam & McLaughlin, Reference Gabard-Durnam and McLaughlin2019). For example, threatening experiences that involve the caregiver appear to markedly impinge on development of neural systems that regulate threat processing and arousal (Opendak et al., Reference Opendak, Gould and Sullivan2017). These potential pathways may have mediated the effects on toddler’s sleep regulation and waking in the current study, as well as other developmental domains that were not assessed.
There was no evidence to suggest that deprivation conditions the effect of PAE. This finding suggests that the effect of PAE on sleep is not conditioned by postnatal experiences of deprivation. It might be that threat impinges on the HPA-axis and amygdala more directly than deprivation (Stroud et al., Reference Stroud, Chen, Navarro, Gim, Benjamin and Doane2024), thus more directly moderating vulnerabilities programed by PAE. This could explain a lack of conditioning of PAE by early life deprivation found in the current study. That said, emerging theoretical models of early life adversity also argue that deprivation experiences in infancy, specifically related to caregiver neglect, represent more salient threatening experiences than exposures traditionally classified as threatening, which are characterized by attacking behaviors such as caregiver hostility or injury (Lyons-Ruth, Reference Lyons-Ruth2025). As a result, further research in warranted to more closely address dimensions and definitions of threat and deprivation in the early developmental period, and how the postnatal environment might condition prenatally embedded vulnerabilities.
Importantly, such exposure to threatening environments in infancy, without subsequent intervention, may represent an early trigger for a cascade of poorer developmental outcomes and greater risk for psychopathology (Masten & Cicchetti, Reference Masten and Cicchetti2010; Rockhold et al., Reference Rockhold, Handley and Petrenko2025). There is considerable evidence demonstrating early childhood sleep shapes trajectories of brain structure and functioning across development. For example, poorer sleep from two years of age has been associated with thinner prefrontal cortical development (Kocevska, Muetzel, et al., Reference Kocevska, Muetzel, Luik, Luijk, Jaddoe, Verhulst, White and Tiemeier2017) and poorer performance on tasks assessing executive functioning (Bernier, Cimon-Paquet, & Tétreault, Reference Bernier, Cimon-Paquet, Tétreault, Berger, Harbourne and Scher2021), which similarly depend on prefrontal cortex activity (Yuan & Raz, Reference Yuan and Raz2014). Similar findings have been found for language, nonverbal intelligence (Kocevska, Rijlaarsdam, et al., Reference Kocevska, Rijlaarsdam, Ghassabian, Jaddoe, Franco, Verhulst and Tiemeier2017), and attention (O’Callaghan et al., Reference O’Callaghan, Al Mamun, O’Callaghan, Clavarino, Williams, Bor, Heussler and Najman2010). Moreover, sleep duration and waking also have cascading effects on externalizing and internalizing symptoms across childhood (Quach et al., Reference Quach, Nguyen, Williams and Sciberras2018). Of note, persistent nightmares starting at 2.5–3 years of age and continuing to middle childhood have been associated with psychopathology later in adolescence and early adulthood, including increased psychotic (Thompson et al., Reference Thompson, Lereya, Lewis, Zammit, Fisher and Wolke2015) and borderline personality disorder (Lereya et al., Reference Lereya, Winsper, Tang and Wolke2017) symptoms. Our findings–that greater threat exposure is associated with shorter sleep duration in children exposed to heavy alcohol consumption in trimester one or throughout pregnancy and that threat is associated with more night waking at 3 years of age regardless of PAE–arguably represent the beginning of such a cascade early in development.
It is also worth noting the co-occurrence of pre-and postnatal risk factors found in this study, which highlights the pervasiveness of a broader pattern that consists of polysubstance use during pregnancy (Jarlenski et al., Reference Jarlenski, Barry, Gollust, Graves, Kennedy-Hendricks and Kozhimannil2017) and postnatal adversity (Debowska et al., Reference Debowska, Willmott, Boduszek and Jones2017). This polysubstance use and postnatal adversity pattern appears to confer substantial developmental risk (e.g., Garrison-Desany et al., Reference Garrison-Desany, Hong, Maher, Beaty, Wang, Pearson, Liang, Wang and Ladd-Acosta2022) and has been demonstrated to sensitize children to postnatal adversity compared to children with no exposure to polysubstance use in utero (Yumoto et al., Reference Yumoto, Jacobson and Jacobson2008). Thus, while this study underscores the unique role of alcohol exposure in embedding vulnerability to threatening postnatal environments, future work could consider the impacts of polysubstance use which could further enhance the ecological utility of our models.
Additionally, within the current study, our focus was on considering experiences during the prenatal and infant periods when modeling burgeoning developmental risk. Environmental influences in later developmental stages, such as adolescence, may also condition the effects of PAE and other prenatal adversities on developmental outcomes, including sleep, and thus may present opportune avenues for intervention. At this developmental stage, experiences within their broader ecology, beyond the caregiving environment, may particularly intensify the impacts of prenatal embedding; but this requires more investigation (Fuhrmann et al., Reference Fuhrmann, Knoll and Blakemore2015).
Limitations
The current study includes several notable strengths, including a focus on mothers with predominantly low levels of education (80% with trade school education or less). Additionally, the study addressed developmental patterning of maternal alcohol use across pregnancy trimesters and experiences of threat and deprivation at 12 months, both in relation to sleep experiences of their offspring, from prenatally to the toddler years (age 3). We also controlled for other critical exposures during pregnancy, such as nicotine (e.g., Stone et al., Reference Stone, LaGasse, Lester, Shankaran, Bada, Bauer and Hammond2010) and marijuana (e.g., Scher et al., Reference Scher, Richardson, Coble, Day and Stoffer1988), which have been directly associated with sleep disturbances in children. Further, additional sensitivity analyses to confirm that a moderating influence of threat exposure in infancy was limited only to risky patterns of alcohol use in the prenatal period (as opposed to postnatal alcohol exposure or prenatal/postnatal cigarette exposure) represents an additional strength.
However, the study is not without limitations. First, although maternal report of alcohol use during pregnancy is the most common way to ascertain PAE (Popova et al., Reference Popova, Lange, Probst, Gmel and Rehm2017), it can lead to error in estimates, as recall of use can be difficult. Systematic underestimates of reported alcohol use can also occur due to stigma associated with alcohol use in pregnancy (Zelner et al., Reference Zelner, Shor, Lynn, Roukema, Lum, Eisinga and Koren2012). In addition, longitudinal data did not record the time of pregnancy recognition nor whether alcohol consumption was adjusted after recognition, which would have further enhanced the ecological validity of the patterns employed in this study. Regardless, we showed validity in our PAE patterning. Illustratively, offspring with mothers characterized by the heavy throughout pattern had significantly smaller head circumferences at birth than those in the abstainer group, as well as marginally lower birth weight and length, common birth outcomes for children with heavy PAE (Akison et al., Reference Akison, Hayes, Vanderpeet, Logan, Munn, Middleton, Moritz, Reid, Barnett, Crawford, Doney, Elliott, Friend, Goldsbury, Gullo, Harrington, Harris, Hewlett, Hutchinson and Walker2024).
Second, sleep measures in our study were all via carer-report. Although carer reported sleep duration, waking, and nightmares broadly align with estimates derived from more objective measures, these methods can lead to divergent findings (O’Sullivan et al., Reference O’Sullivan, Bissell, Hamilton, Bagshaw and Richards2023). Caregivers tend to overestimate sleep duration compared to estimates derived from objective measures and underestimate night wakings in children with neurodevelopmental disorders (O’Sullivan et al., Reference O’Sullivan, Bissell, Hamilton, Bagshaw and Richards2023). That said, assessment of sleep duration, night waking, and nightmares using carer report are relatively common within longitudinal studies (e.g., Cai et al., Reference Cai, Tham, Xu, Fu, Goh, Gluckman, Chong, Yap, Shek, Hoe Teoh, Gooley, Yam-Thiam Goh, Meaney, Schneider, Rifkin-Graboi and Broekman2022; Tesfaye et al., Reference Tesfaye, Wright, Zaidman-Zait, Bedford, Zwaigenbaum, Kerns, Duku, Mirenda, Bennett, Georgiades, Smith, Vaillancourt, Pickles, Szatmari, Elsabbagh and Team2021), as objective measures are generally more resource intensive. Additionally, maternal reports can be confounded by other factors such as heavy drinking and variations in postnatal environments across socio-economic status (Lupini et al., Reference Lupini, Daniel, Mindell and Williamson2023). Hence, reliance on caregiver report may have contributed to higher error and associated lower variance explained within our reported models. Further, sleep onset latency was not measured within our longitudinal data, thought this is a common concern for children with PAE (Chandler-Mather et al., Reference Chandler-Mather, Betts, Donovan, Shelton and Dawe2023). Future research will ideally include objective sleep constructs across each of these dimensions, using actigraphy recording devices or coded from in-home technology data.
Third, analyses addressed outcomes at only a single time-point (age 3), given available sleep measures. Future research would benefit from examining earlier developmental time points, which may perhaps be more sensitive to effects tested here. This would provide additional granularity and may better reveal early developmental sensitivity to postnatal environments in relation to sleep. Likewise, following young children later across developmental would better characterize whether these risks potentially fade or intensify with time.
Lastly, our hypotheses in this case were not formally preregistered. While we originally aimed to recreate PAE patterns based solely on prior research (Bandoli et al., Reference Bandoli, Coles, Kable, Wertelecki, Yevtushok, Zymak-Zakutnya, Wells, Granovska, Pashtepa and Chambers2019; Lees et al., Reference Lees, Mewton, Jacobus, Valadez, Stapinski, Teesson, Tapert and Squeglia2020; Muggli et al., Reference Muggli, Matthews, Penington, Claes, O’Leary, Forster, Donath, Anderson, Lewis, Nagle, Craig, White, Elliott and Halliday2017), sample sizes within certain prenatal alcohol use timings required some combining of patterns. While the patterns we extracted align with established patterns of PAE, their relations to threat and sleep outcomes may be sample specific and require replication. Additionally, effect sizes were only modest across the study; this is not altogether surprising given the complexity of multiple developmental exposures over time, and measuring impacts of exposures three years (threat, deprivation) and four years (PAE) prior. That said, our sample size of over 1900 higher risk young children represents a considerable strength, and overall study findings provide early exploratory evidence of the impacts of intersectional risks prenatally and during the early childhood years.
Conclusions
Prenatal adversity can not only confer risk to the developing fetus but may also confer cascading risks, exacerbated by their future developmental contexts. As a result, this study adopted an intersectional developmental psychopathology lens, with a goal of disentangling the intersection of PAE patterns and postnatal adversity on children’s sleep early in development.
We found some support for such intersectionality of risks in predicting toddler sleep outcomes–early exposure to threat worsened risk for shortened sleep duration. In the context of early threat exposure, children characterized by heavy throughout and heavy reducer patterns of PAE showed shortened sleep at age three. Additionally, early threat exposure was independently associated with increased night waking, above and beyond the negative impacts of prenatal alcohol use.
Bridging understanding of adversities in the prenatal and postnatal environments is a critical way forward. This will arguably uncover additional pre- and post-natal pairings that confer important vulnerabilities across development. In turn, such investigations help to illuminate opportunities–both prenatal and postnatal–for more targeted intervention to promote positive developmental cascades and mitigate risk.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0954579426101321.
Data availability statement
TOP standards were followed, with all code and relevant study materials available at OSF link: https://osf.io/mpt8u. Researchers requesting access to the data may contact the corresponding authors for assistance in accessing data, given current restrictions related to public data access.
Acknowledgements
We would like to acknowledge the children, families, researchers, and staff involved in the (EFHL; Australian and New Zealand Clinical Trials Registry: ACTRN12610000931077) study and the Gold Coast Biobank, Institute for Biomedicine and Glycomics.
Funding statement
K.L.M’s research contributions were supported in part by the following: Partnership Award #0059 from the Stan Perron Charitable Foundation; Distinguished Professoriate Fellowship DF2022-23/1 from the Future Health and Innovation Fund, Government of Western Australia; ARC Discovery Award #DP210103986. N.C.M. and S.D.’s contributions were supported in part by the Australian Government Department of Health Alcohol Program: FASD Diagnostic Services and Models of Care Grant Opportunity (H1617G038) awarded to a consortium led by S.D. Additional funding was provided to K.L.M, S.D., and N.C. by a Center for Mental Health Strategic Investment Award.
Competing interests
The author(s) declare none.
Preregistration statement
The study was not preregistered.
AI statement
AI was not employed in conducting this study.