Introduction
Old World fruit bats (family Pteropodidae) are important pollinators and seed dispersers of over 163 economically useful plants (Fujita & Tuttle, Reference Fujita and Tuttle1991), notably pollinating species of the Fabaceae, Lythraceae, Malvaceae, Musaceae and Myrtaceae families, and dispersing seeds of Ficus, Syzygium and Diospyros species (Aziz et al., Reference Aziz, McConkey, Tanalgo, Sritongchuay, Low and Yong2021). Within the Old World fruit bat family, flying foxes (genera Acerodon, Desmalopex, Eidolon, Mirimiri, Neopteryx, Pteralopex, Pteropus, Styloctenium) are the larger-sized bats, able to carry large fruits and traverse great distances, making them prominent seed dispersers (Mwinyi & Nnko, Reference Mwinyi and Nnko2022). Despite their ecological importance, 51 of the 73 flying fox species are threatened because their wide-ranging habits make them susceptible to deforestation and habitat loss (Meyer et al., Reference Meyer, Struebig, Willig, Voigt and Kingston2016), hunting (Mildenstein et al., Reference Mildenstein, Tanshi, Racey, Voigt and Kingston2016) and land-use changes, including agricultural development and urbanization (Jung & Threlfall, Reference Jung, Threlfall, Voigt and Kingston2016). Unfortunately, few species of flying foxes have received sustained conservation attention, so little is known about population sizes and trends.
The Pemba flying fox Pteropus voeltzkowi is endemic to the island of Pemba, Zanzibar, an autonomous region of Tanzania. The species is important to Pemba because it is a significant pollinator and seed disperser of wild and cultivated fruit trees (Entwistle & Corp, Reference Entwistle and Corp1997b; Mwinyi & Nnko, Reference Mwinyi and Nnko2022; Seltzer et al., Reference Seltzer, Ndangalasi and Cordeiro2013). The population of P. voeltzkowi has been surveyed twice since 1995, allowing us to monitor its trajectory over the past 3 decades. In addition to this unusual long-term conservation attention, the species appears to have shown a population recovery from very low numbers during most of the 20th century (Andersen, Reference Andersen.1912). In 1992, an island-wide survey documented 2,400–3,600 individuals (Seehausen, Reference Seehausen1991), and a subsequent more comprehensive survey, in 1995, counted 4,608–5,450 individuals (Entwistle & Corp, Reference Entwistle and Corp1997a). Concerns about the small population led to an awareness and outreach programme in which initial awareness activities in 1992 were expanded in 1995 by the former Department of Commercial Crops, Fruit and Forestry, now the Department of Forestry, in conjunction with Flora & Fauna International and Lubee Bat Conservancy, to establish community-based environmental clubs, convene village-level meetings and secure commitments to ban shotgun hunting and reduce roost site disturbance (Entwistle, Reference Entwistle2002). These community-supported efforts collectively resulted in a 400% increase in the population, which reached 18,200–22,100 individuals, as assessed through two total population counts, in March and May 2008 (Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010). A partial count in 2012, based on censusing the four largest roosts at the time and extrapolating, estimated 9,360–10,630 individuals (Grose & Clemmens, Reference Grose and Clemmens2012).
Following cessation of direct conservation promotion activities with communities in 2007, it is unclear how far the shift in attitudes that favoured protection of the bats has been maintained. It is therefore unclear whether the core messages of the earlier conservation programme have been retained sufficiently to maintain a healthy population size. The objectives of this research were to provide current information on the population size and distribution of P. voeltzkowi colonies by visiting every roost site on the island, to examine current threats, and determine the extent to which Pembans still remember the conservation intervention programmes from 2 decades ago.
Study area
Pemba is a continental island 50 km off the coast of mainland Tanzania. Covering 988 km2, it is one of two major islands comprising the Zanzibar Archipelago. It is dominated in the west by heavily vegetated ridges and valleys of primary and secondary forest and clove trees, with baobab Adansonia digitata, palms and coastal bush in the flatter east. There are a few small cities on the island, mainly but not exclusively the four District headquarters, but the majority of residents live in the many small villages scattered across most of the island and outlying islets. We identified six roost sites in urban areas (i.e. locations with > 10,000 people). These were the three District headquarters Wete, Chake Chake and Mkoani, plus Ole, Kengeja and Tumbe. In addition to P. voeltzkowi, several other vertebrate species are endemic to the island including the Pemba sunbird Cinnyris pembae, Pemba green-pigeon Treron pembaensis, Pemba white-eye Zosterops vaughani and Pemba scops owl Otus pembaensis.
Methods
Fieldwork was conducted on Pemba over consecutive days during 22 July–4 August 2024 by TC, RSR, AIH and TM. We investigated 81 reported roost sites, of which 67 were visited in person and for 14 a knowledgeable person was questioned by telephone (none of the latter contained bats). We first identified colony locations from a list of 55 roost sites visited in the last survey, in 2008 (Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010). Following a snowball methodology, we added new colony locations to the list whenever these were reported by people whom we encountered at those sites. Two of the authors (RSR and AIH) are employees of the Department of Forestry; RSR knows the island intimately and was involved in previous surveys of the species. We are reasonably certain we visited nearly all occupied and unoccupied roosts. At each roost site, the village or town name, ward name, geographical coordinates, the number of bats in the roost and the characteristics of the roost were recorded; i.e. the trees in which bats were roosted, the forest type, the roost surroundings and reports or observations of any disturbance.
We were brought to the roosting areas by the Sheha (the government appointed executive officer) and local community members who were familiar with the bats’ roosting locations. With their help, we identified the best vantage points for surveying the roost and conducted a daytime roost count following a standardized direct censusing protocol for flying foxes (Mildenstein et al., Reference Mildenstein, Stier, Nuevo-Diego and Mills2005; Mildenstein, Reference Mildenstein, Newman, Field, Epstein and de Jong2010; Kunz et al., Reference Kunz, Betke, Hristov, Vonhof, Kunz and Parsons2009), optimized for single survey population abundance assessment on islands (Mickleburgh et al., Reference Mickleburgh, Waylen and Racey2009). The exact number of bats was counted using binoculars, and the colony size range was categorized as 0, 1–50, 51–100, 101–500, 501–1,000, 1,001–2,500, 2,501–5000 and > 5000 bats, to facilitate comparison with previous surveys. Roost counts are expected to have high probabilities of observing most individuals (Kunz et al., Reference Kunz, Betke, Hristov, Vonhof, Kunz and Parsons2009), but it is possible some bats were missed, and it was more difficult to see all the bats within the diurnal roosting area at larger roosting colonies. Thus, in addition to direct roost counts, we also conducted evening dispersal counts between 18.50 and 19.40 at the two largest roosts (Chake Chake and Wete), following a standardized exit count protocol (Mildenstein et al., Reference Mildenstein, Stier, Nuevo-Diego and Mills2005; Mildenstein, Reference Mildenstein, Newman, Field, Epstein and de Jong2010; Kunz et al., Reference Kunz, Betke, Hristov, Vonhof, Kunz and Parsons2009). Exit counts are known to be variable, and best practices for population abundance assessment recommend multiple evening dispersal counts across consecutive evenings (Mildenstein, Reference Mildenstein, Newman, Field, Epstein and de Jong2010), so our single evening dispersal counts of Chake Chake and Wete are likely to be minimum estimates.
We asked residents over 15 years of age living near each roost if they would like to take part voluntarily in a short interview, in Swahili, which they could leave at any time. Questions included the history of the occupancy of the site, whether there was roost disturbance by people, and what kind, such as tree felling, hunting, general harassment (noise and stone throwing) or fruit picking, usually by children; what foods they believed the bats to feed on; and whether they hunted and/or ate the bats. In situations where roosts were empty, interviewees were asked to approximate how many bats had roosted there in the past. Interviewees were also asked whether they remembered the previous fruit bat education campaign (that is, the initial 1992 education campaign and the large-scale engagement efforts led by the Department of Forestry during 1998–2007), and if a new campaign was needed.
Subsequently, we compared P. voeltzkowi population size and locations from our survey to surveys by the Department of Forestry personnel during 1992–2002, an island-wide survey in 1995 (Entwistle & Corp, Reference Entwistle and Corp1997a) and two island-wide surveys conducted in 2004 (Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010). The roost characteristics, threat assessments and interviews from our survey provide baseline information for 2024.
Results
Population status
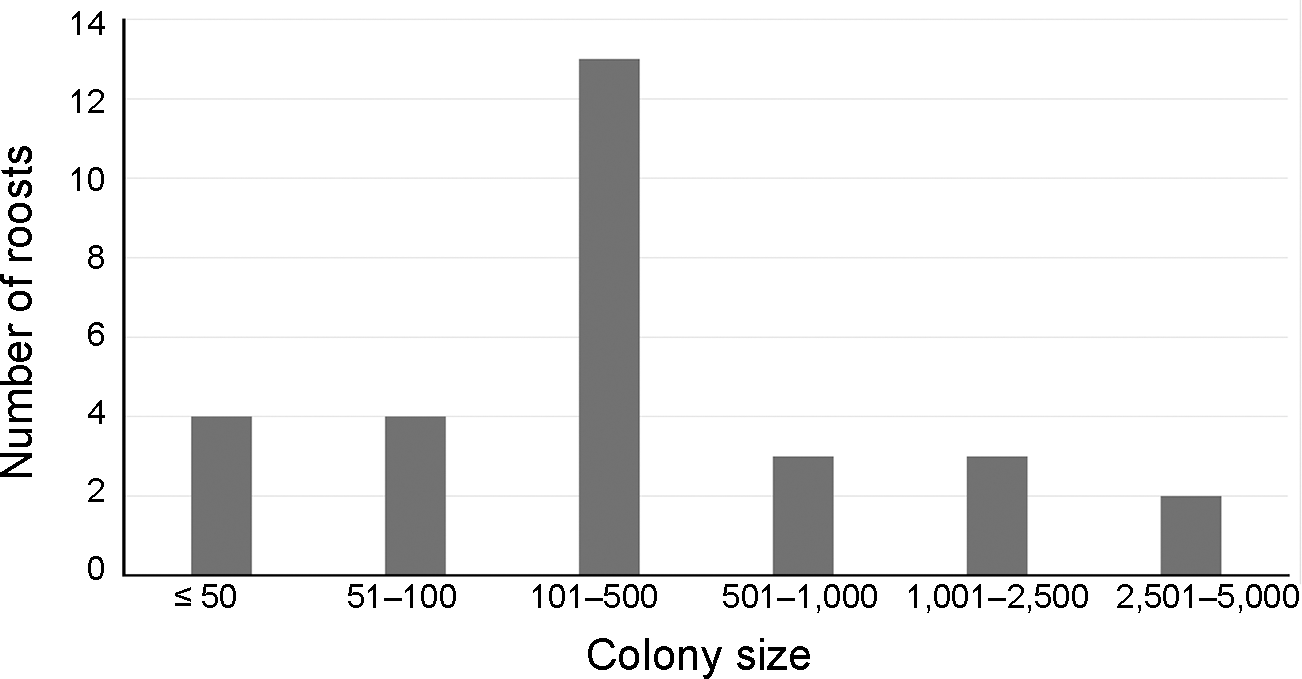
We counted a total of 25,328 P. voeltzkowi individuals across 81 roosting locations, 29 of which were inhabited and 52 empty (Supplementary Material 1). The modal colony size category was 101–500 bats (13 sites), eight sites had < 100 individuals, and two contained 2,500–5,000 bats (Fig. 1). These two large roosts were in the cities of Chake Chake (human population size 52,000 in 2022) and Wete (36,000 in 2022) that together held 58.7% of the island’s total P. voeltzkowi population at the time of the survey. Daytime counts at these two sites were 3,098 and 3,926 bats, respectively, whereas evening dispersal counts were 8,649 and 7,560 bats, respectively, indicating daytime counts were lower by a minimum of 64% and 48%, respectively (see Discussion).
Number of roosts in each Pemba flying fox Pteropus voeltzkowi colony size category on Pemba Island, Zanzibar, in 2024.

Distribution and roost characteristics
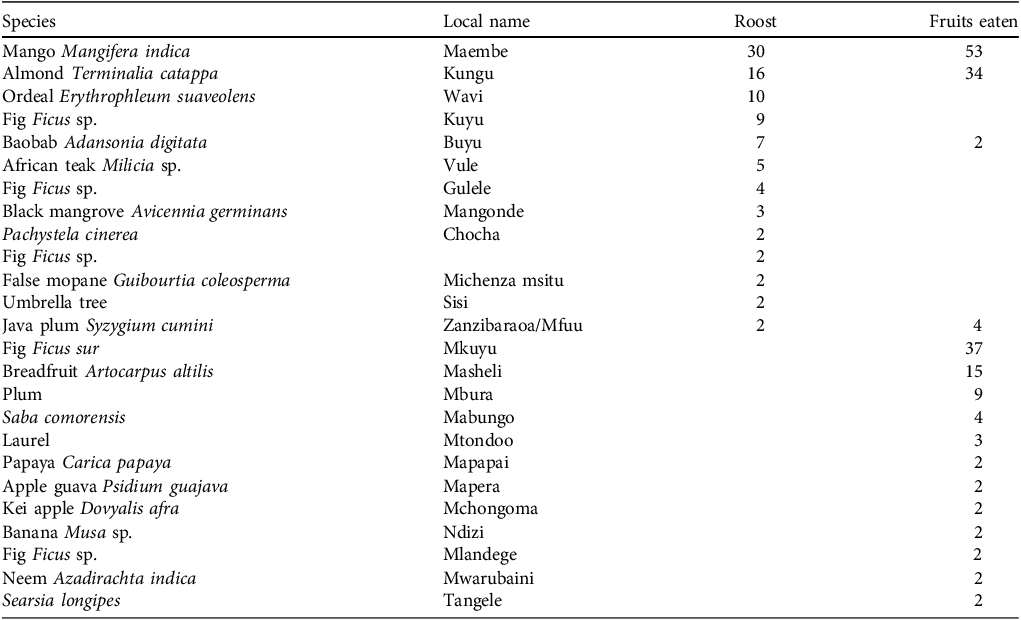
We found P. voeltzkowi across Pemba and on outlying islands (Fig. 2). Roost size was significantly larger in urban centres (n = 6, mean = 3,062) than in rural areas (n = 55, mean = 1,276; Mann–Whitney U = 303, P < 0.001). The roosts in Chake Chake and Wete had > 2,501 bats. One roost with 1,001–2,500 bats was in the centre of the small city of Mkoani; another was on agricultural land at Kidike. There were three roosts with 501–1,000 bats, just outside the town of Kengeja, and in Ngezi Forest Reserve (Supplementary Material 1). Bats were observed roosting in 32 species of tree, with 13 of these at more than one roost site (Table 1). Mango trees Mangifera indica were the most common roost tree species (n = 30) and, with tropical almond Terminalia catappa and ordeal Erythrophleum suaveolens trees, comprised 49.6% of 113 roost trees. Bats were reported to feed on 25 food species (16 reported more than once; Table 1). Mango was the most reported food species eaten (n = 52) and, with the fruit from the broom cluster fig Ficus sur and almond, comprised 66.9% of 181 reports.
Locations and size categories of P. voeltzkowi roosts sites recorded on Pemba in 2024. Note that some closely adjacent sites are aggregated here for the purpose of illustration.

Number of reports of tree species used as roosts and as a source of fruits by the Pemba flying fox Pteropus voeltzkowi, as indicated by interviewees on Pemba in 2024. Scientific names are provided where known.

Changes from previous surveys
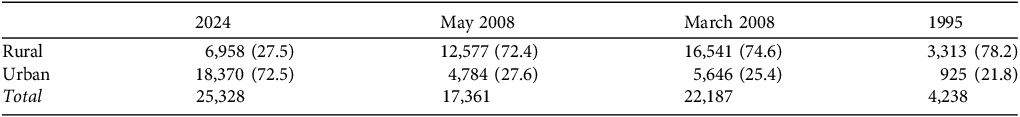
In 1995 bats were found in relatively low numbers at relatively few sites across Pemba but in 2008 and in our survey in 2024 they were found in greater numbers and at more locations (Fig. 3). The 2008 survey revealed large aggregations of bats at five sites, Mgogoni (south of Ngezi Forest Reserve), two outside the city of Mkoani, and in Kidike and Wete; all except Wete were rural sites. In 2024, we found bats at the six large sites described earlier, two of which were the same as in 2008. The number of bats in urban and rural roosts changed between 2008 and 2024 (Table 2), with 72.5% of bats found in urban roosts in 2024, compared with 27.6% and 25.4% in the two 2008 surveys, and 21.8% in the 1995 survey. In contrast to our 2024 survey, there were no significant differences in the number of bats counted at urban and rural sites in either of the 2008 surveys (May: n = 54 sites, U = 105.5, P = 0.288; March: n = 54, U = 105, P = 0.306), or in 1995 (n = 38, U = 64, P = 0.071).
The size and distribution of occupied P. voeltzkowi roosts on Pemba Island recorded in (a) 1995 (Entwistle & Corp, Reference Entwistle and Corp1997a), (b) 2008 (Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010) and (c) in our survey in 2024.

Total numbers (and per cent) of P. voeltzkowi counted in rural and urban roosts on Pemba (Fig. 2) in our survey in 2024 and in three earlier surveys, in May and March 2008 (Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010) and 1995 (Entwistle & Corp, Reference Entwistle and Corp1997a). Comparisons: 2024 vs March 2008 χ 2 = 8,393.5, df = 1, P < 0.00001; 2024 vs May 2008 χ 2 = 1,1621, df = 1, P < 0.00001; 2024 vs 1995 χ 2 = 4,116.7, df = 1, P < 0.00001.

Disturbance
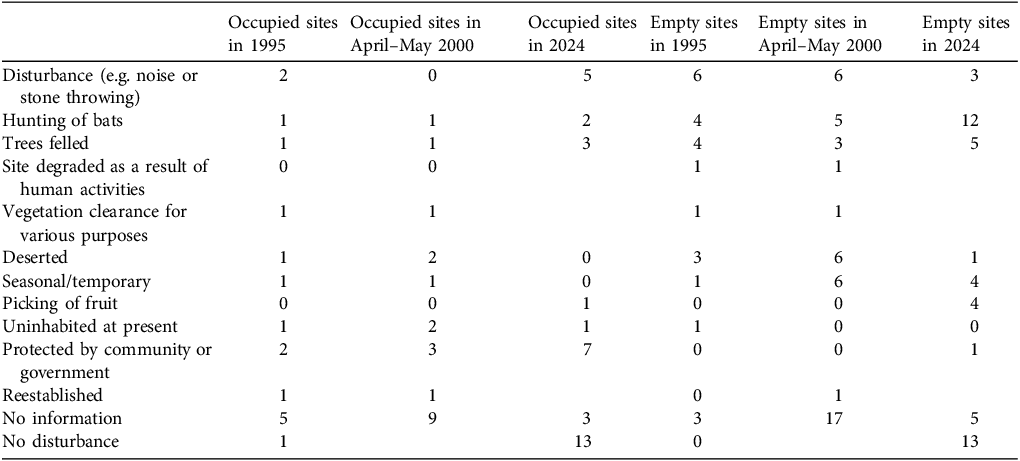
In 2024, interviewees reported disturbance of bats at 42.2% of the 81 roost sites (83 reports, as two people were interviewed for each of two roosts; Table 3). Empty roosts seemed to have suffered more from disturbance than occupied roosts (50.0% vs 31.4%, respectively). Hunting had occurred at 16.9% of roosts, with more reported at now empty roosts than at occupied roosts (25.0% vs 5.7%, respectively). Disturbance appeared to have fallen from 43.9% in 1995 to 23.9% in 2000 following the onset of the conservation project and adoption of local byelaws to protect the bats (Table 3). Hunting occurred at 12.2% and 12.8% of roosts in 1995 and 2000, respectively, and at 16.9% of roost sites in 2024.
Notes recorded at roost sites, with numbers of roosts at which each issue was observed in 1995 (Entwistle & Corp, Reference Entwistle and Corp1997a), 2000 (Entwistle, Reference Entwistle2002) and during our survey in 2024. Blank cells indicate data were not recorded.

Conservation project
When interviewees were asked about their recollection of the original education campaign and/or environmental club engagement, 28.4% remembered the project, 45.7% did not, and 13.6% had only heard of it indirectly, with some not answering. When asked whether a new campaign was needed, 88.9% answered yes. Reasons given for desiring a new campaign were to protect bats (22.2% of respondents), to protect the environment (25.9%), to improve education about bats (19.8%) to attract visitors to Pemba (8.6%) and because bats benefit the environment (11.1%).
Discussion
Population size
As a result of obstructions and limited visibility, counting bats while they are roosting can be challenging. Some previous surveys have used patch counts (Thomas & Laval, Reference Thomas and Laval1988), and then scaled up estimates at roosts with poor visibility. We used direct counts at small sites and evening dispersal counts at the two largest roost sites. With more time, evening dispersal counts at additional sites would have enabled us to generate a more accurate correction factor to apply to the other roosts, but evening dispersal counts over many nights (only one roost can be counted per night) would have lengthened the survey and could result in bats being double-counted as a result of inter-roost movement.
Instead, we tried an alternative method to address the issue of potential undercounting at large roosts. Specifically, we used a quadratic equation (y = 0003947x2 + 0.92106x) to account for undercounting at any roost site with > 200 bats, except Chake Chake and Wete, where we used the evening dispersal counts we made there. A quadratic function was employed because we reasoned that errors would increase disproportionately with roost size. This meant 14 of 81 roost sites had an adjusted estimate. At sites with ≤ 200 bats, we were confident of our direct daytime counts and used them. Using these plus the adjusted counts, we estimated there were 27,412 bats on Pemba. In summary, the 2024 P. voeltzkowi population on Pemba lies between 25,328 and 27,412 individuals, comparable to the estimate in 2008 (18,200–22,100).
Distribution
Both previously published surveys concluded that P. voeltzkowi roosts were most likely to be found in forest habitats (Entwistle & Corp, Reference Entwistle and Corp1997a; Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010). In the 1995 survey, nine of the 10 roosts containing > 100 bats were in primary or secondary forest (Entwistle, Reference Entwistle2002). Our survey suggests that by 2024 there had been a significant movement of bats into towns, with 58.7% of the population in two large city roosts and with mean roost size in towns twenty times greater than in forests (Table 2).
Many of the roost sites we visited were also surveyed in 1995 and 2008. In 2008, four sites (Kidike, Mbiji 1, Mbiji 2 and Wete), accounted for 87% of the total bat population on Pemba. Of these, only Wete has retained its large population, which has also increased. At Kidike, once a significant forest roost and former tourist attraction, there has been a substantial population decline, with a count of only 1,491 bats in 2024 compared to 3,394 and 4,978 in the two 2008 counts. The two forest roosts of Mbiji 1 and Mbiji 2 were empty in 2024 but had counts of 4,678 and 7,001, respectively, in 2008. Local reports attributed this decline to disturbance. These shifts illustrate the abandonment of forest roosts and movement to urban environments. Movement to cities was also implicated in changes in a 2012 study on Pemba (Grose & Clemmens, Reference Grose and Clemmens2012).
Other Pteropus species are also moving into urban areas, and species can now be found in multiple cities in Australia (Markus & Hall, Reference Markus and Hall2004; Parris & Hazell, Reference Parris and Hazell2005; Williams et al., Reference Williams, McDonnell, Phelan, Keim and Van der Ree2006), Egypt (Egert-Berg et al., Reference Egert-Berg, Handel, Goldshtein, Eitan, Borissov and Yovel2021), Singapore (Chan et al., Reference Chan, Aziz, Clare and Coleman2021) and throughout Southeast Asia (T. Mildenstein, unpubl. data). Although increased food availability can in some cases account for the movement of bats into urban areas (Williams et al., Reference Williams, McDonnell, Phelan, Keim and Van der Ree2006), the primary food species of P. voeltzkowi, mango, breadfruit Artocarpus altilis and Ficus spp. (Entwistle & Corp, Reference Entwistle and Corp1997b; Table 1), are not more abundant in cities on Pemba, and this is therefore an unlikely explanation for bat movements on the island. The most likely explanation for this phenomenon in areas where bats are hunted or disturbed is the added protection afforded in urban settings, where hunters are not tolerated by city dwellers (Mildenstein, Reference Mildenstein, Newman, Field, Epstein and de Jong2010).
Ephemeral nature of roosts
Many of the roosts we visited were empty (see also Grose & Clemmens, Reference Grose and Clemmens2012) but were reportedly occupied recently (corroborated by evidence of bat droppings in the understorey). Temporary roost occupancy appears to be a characteristic of P. voeltzkowi on Pemba, although the bats show strong fidelity to some roost sites, such as those in Ngezi forest and Kengeja (Pakenham, Reference Pakenham1984; Kingdon, Reference Kingdon1974; Bentjee, Reference Bentjee1990). Up to eight bat censuses were conducted by Department of Forestry personnel from just before 1992 up to February 2002, with numbers available for 46 sites visited more than once. Seasonal shifts were pronounced: bats were always present at 12 of these sites, and were always absent at six, 15 were vacated over the course of the censuses, five were established, two were vacated and then re-established, one was established but then vacated, and five flip-flopped between bats being present and absent (Entwistle, Reference Entwistle2002).
When we arrived at an empty roost, we asked people how many bats were there previously. This was an open question but assuming any answer referred to the last time the bats were present, 27 of 46 empty roosts had reportedly housed 100–500 bats, and four sites were reported to have contained > 1,000 bats. Only rural habitats contained empty roosts; all town roosts were occupied, again suggesting bats were concentrated in towns at the time of this study in 2024.
Reasons for movements appeared twofold: disturbance and seasonal movements as a result of changing food distribution. We found that disturbance, hunting, tree felling and fruit picking were associated with 50% of empty roost sites (Table 3), and an earlier survey found a negative correlation between proximity to human settlements and colony size (Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010). Absence of bats was associated with disturbance, hunting and tree cutting in both 2000 and 1995 (Table 3; Entwistle, Reference Entwistle2002). Our information regarding changing food distribution was anecdotal, taken from people’s opinions, but mango trees, an important food source, occur across the island and bats will reportedly travel up to 15 km to food sources each night, suggesting food distribution may not necessarily be a major cause of changes in roost occupancy.
Hunting of bats for food on Pemba remains a problem, with bats captured at c. 17% of roosts, mostly by boys using long sticks with sticky seed pods or catapults (Walsh, Reference Walsh1995). Hunting has been a consistent threat to this species (Entwistle & Juma, Reference Entwistle and Juma2016), exacerbated by the introduction of shotguns (Entwistle & Corp, Reference Entwistle and Corp1997a). Hunting was more prevalent than other disturbances: it was reported at 12 of 48 empty roosts, and 18.5% of 81 local people admitted to hunting bats and 28.4% said they had eaten them. These statistics, along with two reports stating that people had visited roost sites to hunt bats with shotguns in the week previous to our survey, indicate hunting is still a threat to this species.
We believe that hunting, felling of trees, and throwing stones to knock down fruit in rural areas is driving P. voeltzkowi into large urban roosts where these activities are suppressed by the disapproval of passersby, and by policing. For example, there was a large roost at Msitu wa Mbiji, 2.5 km outside Mkoani, in the 2008 census, but this was empty in 2024, with the bats having likely taken up residence in trees within the District Government residence in the centre of Mkoani, reportedly as a result of hunting and human disturbance at Mbiji (see also Grose & Clemmens, Reference Grose and Clemmens2012).
Study limitations
Our study suffered from at least two limitations. Firstly, it was a single, dry season study, which limits our ability to make unequivocal statements regarding whether rural roosts have been abandoned permanently for urban sites; bats may roost in urban areas temporarily, influenced by temporal variation in food sources. In 2025, numbers at Chake Chake were lower than they were in 2024 as a result of movement from the roost (RSR, pers. obs.). Secondly, we did not take measures of disturbance, proximity to human habitation or roost tree species into account in our analyses. It is challenging to measure disturbance quantitatively as it varies over time and in extent, requiring frequent visits to sites.
Conservation implications
The 1992 education campaign to conserve P. voeltzkowi was centred on the species’ role in seed dispersal and forest regeneration, with the campaign attempting to encompass both formalized and community-based educational outlets (Robinson et al., Reference Robinson, Bell, Saleh, Suleiman and Barr2010). Initial awareness activities in 1992 were built upon in 1995, with surveys in schools demonstrating high understanding of the ecological role of the bats, but a lack of recognition of its endemism or threatened status (Entwistle & Corp, Reference Entwistle and Corp1997a; Trewhella et al., Reference Trewhella, Rodriguez-Clark, Corp, Entwistle, Garrett and Granek2005). Interviews demonstrated the cultural importance of this species locally (Bowen-Jones & Entwistle, Reference Bowen-Jones and Entwistle2002). Subsequent work by the Department of Forestry and NGOs extended education and awareness activities (including production of a video), established community-based environmental clubs in several villages and convened village-level meetings that resulted in commitments to protect the bats, including bans on hunting with shotguns, and reducing roost site disturbance (Entwistle, Reference Entwistle2002). The campaign showed the potential for protection of bats to be internalized within village environmental clubs, and some villages established local byelaws to protect bats and their roosts, enforced by club members (Entwistle, Reference Entwistle2001, Rice & Hamdan, Reference Rice and Hamdan2002); children were used as ambassadors to take the message to their parents. Evaluations of the project indicated an increase in bat population size over the period (albeit with some caution with regard to survey rigour), and decreased hunting at roost sites protected by environmental clubs (Rice & Hamdan, Reference Rice and Hamdan2002). Further project funding during 2003–2007 supported ongoing awareness raising activities, community-led monitoring activities and development of bat ecotourism at one environmental club, Kidike, along with ongoing survey work. Further population increases were reported over this period (Fauna & Flora International, 2007), leading to a recategorization of the species on the IUCN Red List from Critically Endangered (Mickleburgh et al., Reference Mickleburgh, Hutson, Bergmans and Howell2004) to Vulnerable (Entwistle & Juma, Reference Entwistle and Juma2016).
Yet, in 2024, less than half of interviewees had heard of these educational efforts, but nearly everyone thought a new campaign was necessary and that most people would listen and learn from it. Pembans recognize that fruit bats play important ecological roles as pollinators and seed dispersers on the island (Mwinyi & Nnko, Reference Mwinyi and Nnko2022); 37% of interviewees talked, either directly or indirectly, about the role of bats in the environment and this being a reason why a new campaign is needed.
Although the P. voeltzkowi population appears to have been stable since 2008, there are indications that bats are moving from rural to urban locations. Our working hypothesis is that this movement is driven by human activities, namely hunting and roost disturbance, which are less frequent in cities because of widespread disapproval of these activities. Interview responses indicated that a substantial number of local people are likely to be receptive to a new conservation project. Any campaign should continue to educate the younger generation in schools, not only because children hunt the species, but to emphasize the key roles of bats in plant pollination and seed dispersal, both of value to humans (Fujita & Tuttle, Reference Fujita and Tuttle1991), and to remind people that the species is endemic to Pemba and is therefore a threatened cultural icon. Given that bats are now an unavoidable presence in towns, there is an opportunity for the Department of Forestry to reinstate the community-level environmental clubs, which were previously successful, as an effective mechanism to discourage interference with bat roosts in urban environments but also in rural areas where they are more threatened by disturbance.
Recommendations for future surveys
Our examination of several surveys conducted by different people and organizations revealed nine challenges involved in counting P. voeltzkowi. (1) There is inconsistency in the time of year in which surveys are conducted. (2) There is inconsistency in choice of sites surveyed, with number and localities changing both between and within years (e.g. Kisiwa Panza and Matumbini, see Supplementary Material 1). (3) There can be confusion with site names, and where there is more than one site within a community area the roosts are not currently distinguished between each other; e.g. Mgelema (Mgelima); this could result in incorrect recording of data. (4) There is a lack of reconnaissance, training and survey experience among surveyors conducting the counts, which introduces observer error. (5) There is a lack of time allocated for replication using direct and exit counts. (6) Surveys are usually conducted just once and at one time of year, but multiple island-wide counts across seasons are required to better understand bat roosting choices and movements. (7) At sites with small numbers of P. voeltzkowi it is possible to count each individual, but at large roosting sites this is infeasible, and no standardization has been employed in techniques to count these populations, increasing the margin for error. (8) In some surveys, each team is allocated a section of the island to survey; this extends the length of time each team spends in one geographical area, which could increase the risk of double-counting individuals or groups if they move between roost sites. (9) Although assessment of habitat and disturbance is made at roost sites during surveys, there is scope for improving assessment methods and collecting more detailed information. Addressing these challenges could lead to more robust surveys in the future.
Supplementary material
The supplementary material for this article is available at doi.org/10.1017/S0030605325102238
Author contributions
Project design: TC, TM; fieldwork: TC, RSR, AIH, TM; analyses, map production: SO, IJ; writing: TC; addition of historical text: AE, JR; revision: TM.
Acknowledgements
We thank Bat Conservation International for funding, Matt Clark for advice on mapping, and two anonymous reviewers for comments.
Conflicts of interest
None.
Ethical standards
Observational research was conducted with permission from the Government of Zanzibar, ethical approval was obtained from the University of Bristol (UIN-24-056), and the study abided by the Oryx ethical standards.
Data availability
Data are provided in the supplementary material.