Introduction
Khasi nightshade (Solanum khasianum C.B. Clarke; syn.: Solanum aculeatissimum Jacq.), is a perennial invasive herb. Native to Brazil, it is now widely distributed across tropical regions of Asia and Africa (Begum et al. Reference Begum, Munda, Pandey and Lal2022; Gogoi et al. Reference Gogoi, Sarma, Pandey and Lal2021). It is commonly found in pastures, orchards, grassy slopes, open forests, and along roadsides at elevations ranging from 1,300 to 2,300 m (Silva et al. 2015; Yang et al. Reference Yang, Liu, Zhang, Wang and Cheng2017). Solanum khasianum was initially introduced into China as a medicinal plant and has since spread to Yunnan, Guangxi, Guizhou, and other regions due to poor management (Yang et al. Reference Yang, Liu, Zhang, Wang and Cheng2017). As an invasive plant species, S. khasianum exhibits rapid growth, continuous flowering and fruiting, and a high reproductive capacity, all of which contribute to its strong competitiveness. The leaves and stems of the plant have thorns, which pose physical hazards to humans and livestock. Its berries contain high steroidal alkaloids, which can harm the digestive and nervous systems of livestock when ingested (Begum et al. Reference Begum, Munda, Pandey and Lal2022; Ma et al. Reference Ma, Wu, Zhao, Zhao, Cai, Zhou and Tang2023). Each mature berry releases hundreds of pale yellow seeds that are flat, nearly ovoid, and approximately 2.5 mm in diameter (ECFRPS 1978). Presently, S. khasianum is listed in the fourth batch of alien invasive species in China (MEEPRC 2016), indicating its significant economic and ecological impacts.
Seed germination marks the beginning of plant growth and development, which is critical for species reproduction (Durr et al. Reference Durr, Dickie, Yang and Pritchard2015; Zheng et al. Reference Zheng, Shi and Wang2024). Germination and seedling emergence are not only influenced by seed traits, but also by various abiotic environmental factors, including light, pH, osmotic stress, salt stress, high temperature, and burial depth (Baskin and Baskin Reference Baskin and Baskin1998; Javaid and Tanveer Reference Javaid and Tanveer2014). Several studies have investigated the germination characteristics of other Solanum species, including hairy nightshade (Solanum physalifolium Rusby; syn.: Solanum sarrachoides Sendtn.), eastern black nightshade (Solanum ptycanthum Dunal), Australian bush tomato (Solanum centrale J. M. Black), American black nightshade (Solanum americanum Mill.), black nightshade (Solanum nigrum L.), and silverleaf nightshade (Solanum elaeagnifolium Cav.) (Ahmed et al. Reference Ahmed, Johnson, Burchett and Kenny2006; Forte et al. Reference Forte, Nunes, Filho, Galon, Chechi, Roso, Menegat, Rossetto and Franceschetti2019; Kapiluto et al. Reference Kapiluto, Eizenberg and Lati2022; Ma et al. Reference Ma, Huang, Huang, Guo, Saeed, Jiang, Chen and Wei2021; Zhou et al. Reference Zhou, Deckard and Ahrens2005a, Reference Zhou, Deckard and Messersmith2005b). For instance, six populations of S. nigrum exhibited varying responses to temperature, with optimal germination occurring between 15 and 30 C (Ma et al. Reference Ma, Huang, Huang, Guo, Saeed, Jiang, Chen and Wei2021). Recently, temperature-based seed germination models have been developed for S. nigrum and S. elaeagnifolium to predict seed germination under different temperature regimes (Kapiluto et al. Reference Kapiluto, Eizenberg and Lati2022; Ma et al. Reference Ma, Huang, Huang, Guo, Saeed, Jiang, Chen and Wei2021). However, the impact of environmental factors on the germination of S. khasianum has not been reported. Existing research has primarily focused on medicinal properties (Gogoi et al. Reference Gogoi, Sarma, Pandey and Lal2021; Jarald et al. Reference Jarald, Edwin, Saini, Deb, Gupta, Wate and Busari2008), tolerance to heavy metals (Li Reference Li2008; Wu et al. Reference Wu, Sun and Li2024), disease resistance (Zhou et al. Reference Zhou, Bao, Liu and Zhuang2016; Zhuang et al. Reference Zhuang, Zhou and Wang2012), and gene functional identification (Yang et al. Reference Yang, Liu, Zhang, Wang and Cheng2017; Yi et al. Reference Yi, Cai, Yang, Shen, Sun and Li2024; Zhang et al. Reference Zhang, Yi, Xia, Jiang, Du, Yang and Yang2024).
Therefore, the objective of this study was to comprehensively determine the effects of various environmental factors such as temperature, light, pH, osmotic stress, salt stress, high-temperature pretreatment, and burial depth on the germination of S. khasianum. This information concerning germination ecology will serve as a key basis for formulating management strategies for this invasive plant.
Materials and Methods
Seed Collection and Preparation
Mature berries of S. khasianum were collected randomly from approximately 10 plants in grasslands from Malong District, Qujing City, Yunnan Province (25.31°N, 103.35°E) in July 2024. Seeds were extracted by removing the skin and pulp from the berries, then air-dried in a well-ventilated room at ambient temperature for 2 wk. The air-dried seeds were stored in paper bags under the same conditions until use. A preliminary germination test conducted 30 d after harvesting showed that seeds did not exhibit dormancy at 35 C, achieving a germination rate (GR) of 94% with deionized water alone. Seeds were not subjected to additional processing until they were used for the experiments.
General Germination Test
Thirty seeds of S. khasianum were placed in each 12 cm by 12 cm plastic culture dish containing a double layer of filter paper moistened with 6 ml of deionized water (control) or test solution (solutions with different pH values or different levels of osmotic stress or salt stress) for the experiment. An equal volume of deionized water was added to each control group. All petri dishes were secured with sealing film to reduce moisture loss and kept in artificial climate chambers at a constant temperature of 35 C under a 12/12-h (light/dark) photoperiod and a photosynthetic photon flux density of 200 μmol m⁻2 s⁻¹. Seeds were considered germinated when the radicle reached at least 2 mm in length (ISTA 2020). Germinated seeds were recorded and removed daily. The cumulative number of germinated seeds in each petri dish was counted over a 21-d period. The GR was calculated by dividing the cumulative number of germinated seeds by the total number of seeds. The culture system was strictly followed unless otherwise specified.
All experiments were arranged in a randomized complete block design with four replications and were conducted twice in succession during October and November 2024. Before the other experiments commenced, seeds were placed in petri dishes containing deionized water in the chamber as described earlier to test viability, and all GRs were greater than 90%.
Effect of Temperature on Germination
To determine the optimal germination temperature of S. khasianum, petri dishes containing 30 seeds each were incubated at one of six constant temperatures (25, 30, 33, 35, 38, and 40 C). All dishes were maintained in an artificial climate chamber under a 12/12-h (light/dark) photoperiod. The number of germinating seeds was observed daily, and deionized water was added to avoid seeds drying out.
Effect of Light on Germination
To determine the effect of photoperiod on germination, petri dishes containing 30 seeds each were placed under 0/24-, 6/18-, 12/12-, 18/6-, and 24/0-h light/dark regimes at a constant temperature of 35 C. Based on the results from the initial experiment on the effect of temperature on germination, 35 C was the most favorable temperature for the germination of S. khasianum and was adopted in all subsequent subexperiments. The petri dishes in complete darkness (0/24-h light/dark) were wrapped in a double layer of aluminum foil to avoid any light. Other light environments were precisely controlled using artificial climate chambers, and the photosynthetically active radiation intensity during the illumination period was 200 μmol m⁻² s⁻¹. The addition of water and daily counting were strictly carried out under the corresponding light conditions at 35 C.
Effect of pH on Germination
To evaluate the influence of pH on the germination of S. khasianum, 30 seeds were placed on filter paper in petri dishes. Buffer solutions with seven pH values (4, 5, 6, 7, 8, 9, and 10) were prepared according to the method described by Reddy and Singh (Reference Reddy and Singh1992). A volume of 6 ml of one of the seven pH solutions was added to soak the filter paper in each petri dish. The filter paper was replaced every 3 d, and the same volume of solution was added simultaneously. The petri dishes were kept in a chamber at 35 C with a photoperiod of 12 h d⁻¹. Other conditions were the same as those described for the general germination test.
Effect of Osmotic Stress on Germination
To explore the effect of osmotic stress on the germination of S. khasianum, aqueous solutions with osmotic potentials of 0, −0.05, −0.1, −0.2, −0.4, and −0.6 MPa were prepared by dissolving appropriate amounts of polyethylene glycol 6000 in deionized water (Michel and Kaufmann Reference Michel and Kaufmann1973). Petri dishes containing seeds were kept in a chamber at 35 C with a photoperiod of 12 h d⁻¹. Other conditions were the same as those described for the general germination test.
Effect of Salt Stress on Germination
To explore the impact of salt stress, the concentrations of sodium chloride (NaCl) were 0, 10, 20, 30, 40, 50, 60, 80, and 100 mM. Other conditions were the same as those in the general germination test. Petri dishes were kept in a chamber at 35 C.
Effect of High-Temperature Pretreatment on Germination
To simulate the effect of high temperature caused by crop residue combustion on seed germination, seeds were pretreated at 40, 60, 80, 100, or 120 C for 5 min (Atabaki et al. Reference Atabaki, Gherekhloo, Ghaderi-Far, Ansari, Hassanpour-Bourkheili and Prado2023; Wang et al. Reference Wang, Zhao, Li, Chen, Liu and Wang2020). Subsequently, seed germination test was carried out as mentioned in the general germination test. Seeds stored at room temperature (25 C) were used as a control.
Effect of Burial Depth on Germination
To study the effect of burial depth on germination, seeds were planted in soil with burial depths ranging from 0 to 10 cm (0, 1, 2, 4, 6, 8, and 10 cm). The tested soil was collected from the area where S. khasianum grows. It was red heavy loam soil with an organic matter content of 3.39% and a physical clay content of 52.58% (Li et al. Reference Li, Zhang, Lv, Wang and Li2018). The collected soil was sieved and autoclaved to remove impurities, then used to fill plastic circular pots (height: 12 cm; diameter: 15 cm). To achieve the corresponding burial depths, the initial heights of soil in pots were 12, 11, 10, 8, 6, 4, and 2 cm, respectively. Subsequently, 30 seeds were evenly sown on the soil surface of each pot, and the pots were filled with soil to the same height (12 cm). All pots were kept in a chamber at a constant temperature of 35 C with a photoperiod of 12 h and watered every other day to maintain moisture. Coleoptiles exceeding 2 mm above the soil surface were considered emerged. The number of seeds that broke through the soil was counted daily for 21 d.
Statistical Analyses
Data from repeated experiments were subjected to ANOVA using SPSS software (v. 16.0, IBM, New York, NY, USA). Due to the statistically nonsignificant difference (P > 0.05) between the two runs for all the experiments, data were pooled for final analysis. Before ANOVA, the assumptions of normality and homogeneity of variance were investigated by the Kolmogorov-Smirnov test and Levene’s test with SPSS software, respectively. Data meeting assumptions of normality and homogeneity of variance were subjected to ANOVA. The mean comparison was performed using Fisher’s protected LSD test at the 5% significance level.
The time of first germination under different treatments was recorded as “onset of germination.” Equation 1 was used to evaluate the effect of different temperatures on the time required for germination percentage to reach 90% (t 90). L is the last day before 90% germination is reached, L p is the observed germination percentage on day L, and H p is the observed germination percentage on the day when germination reaches or exceeds 90% (Yue et al. Reference Yue, Jin, Lu, Gong, Han, Liu and Wu2021).
 $${t_{50}} = {t_i} + \left[ {{{\left( {N + 1} \right)/2 - {n_i}} \over {{n_j} - {n_i}}}} \right] \cdot \left( {{t_j}\; - \;{t_i}} \right)$$
$${t_{50}} = {t_i} + \left[ {{{\left( {N + 1} \right)/2 - {n_i}} \over {{n_j} - {n_i}}}} \right] \cdot \left( {{t_j}\; - \;{t_i}} \right)$$
Time to obtain 50% cumulative germination (t 50) was calculated according to Equation 2. N is the total number of germinated seeds, n j and n i represent the cumulative number of germinated seeds on adjacent sampling dates, day t j and t i respectively, where n i < (N + 1)/2 < n j (Coolbear et al. Reference Coolbear, Francis and Grierson1984).
Data from light and pH experiments were subjected to ANOVA using the general linear model procedure in SPSS. Nonlinear regression analysis was more appropriate for data from temperature, osmotic stress, salt stress, high-temperature pretreatment, and burial depth. Pseudo-R², often abbreviated to R², was calculated to assess the goodness of fit of the appropriate regression analysis (Koger et al. Reference Koger and Reddy2004).
The GRs at different temperatures were fit using a three-parameter gaussian model (Equation 3). Here, a represents the highest percentage (%) of germination, b represents the specific temperature that achieves the highest germination, and c represents the variance of the Gaussian distribution.
Data from high-temperature pretreatment were best fit to the quadratic polynomial Equation 4. G 0 is the estimated model intercept; a and b are coefficients (Koger et al. Reference Koger and Reddy2004).
The GRs of osmotic stress, salt stress, and burial depth at different levels were fit using a four-parameter sigmoid model (SigmaPlot v. 13, Systat Software, San Jose, CA, USA). In logistic Equation 5, x represents the osmotic potential, NaCl concentration, or burial depth in the corresponding test, respectively; x 50 is the osmotic potential, NaCl concentration, or burial depth required to achieve 50% germination; b is the slope at x 50; G is the cumulative germination percentage (%) at x; and D and C are the upper and lower limits of G, respectively (Amini et al. Reference Amini, Hasanfard, Ahmadian and Yuzband2024; Seefeld et al. Reference Seefeld, Jensen and Fuerst1995).
Results and Discussion
Effect of Temperature on Germination
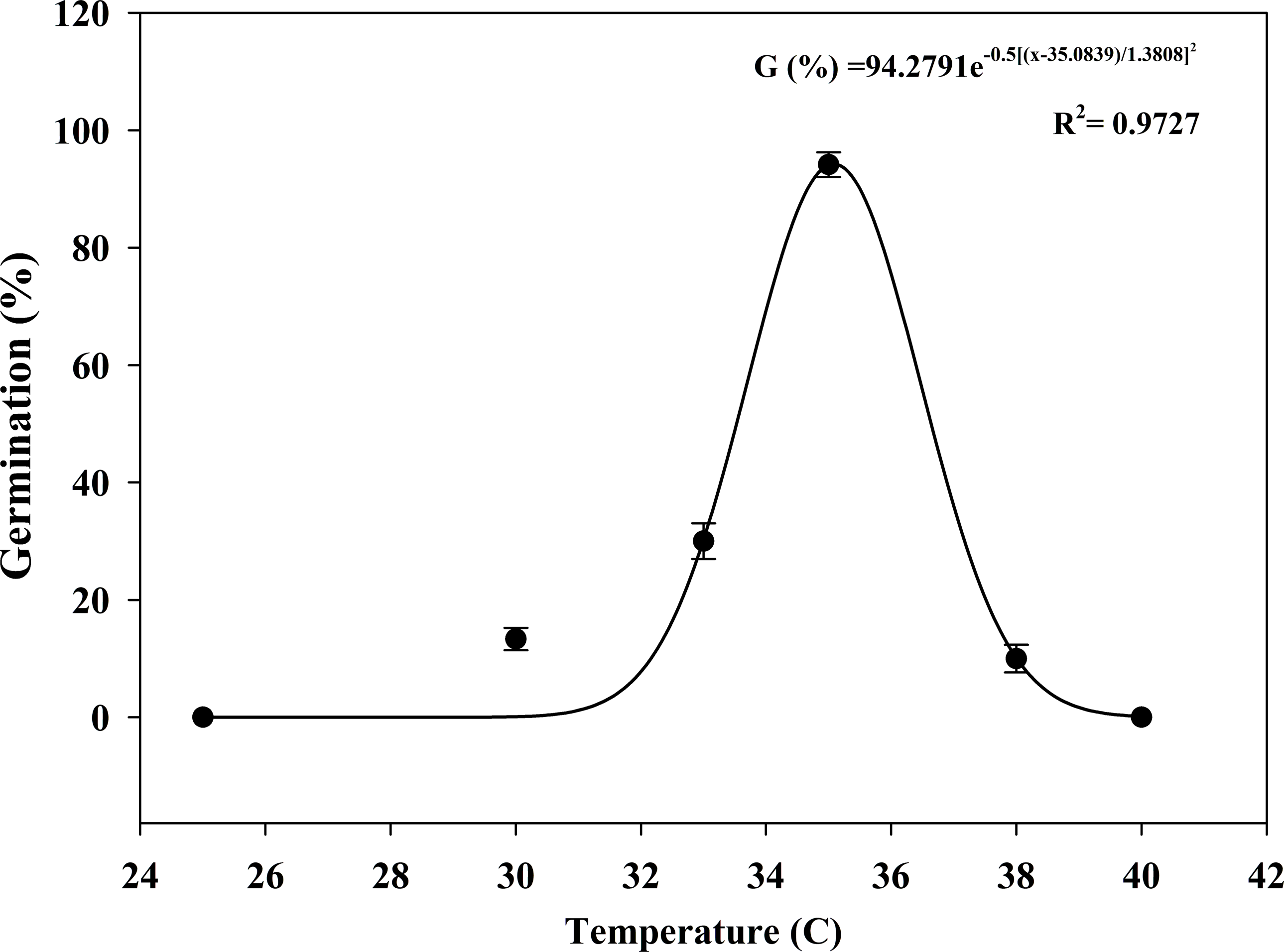
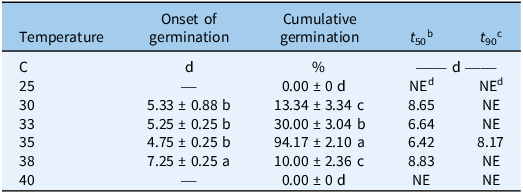
When exposed to constant temperature, S. khasianum germinated over a narrow temperature range of 30 to 38 C (Figure 1). Seed germination increased as temperatures increased from 25 to 35 C, then decreased as temperatures increased to 40 C. The optimum germination temperature was 35 C, and the GR value was 94%. The time to start germination (4.75 d) and t 50 (6.42 d) of S. khasianum were recorded at 35 C, and the t 90 was 8.17 d. Ten percent germination was observed at 38 C, whereas no germination occurred at 25 C or 40 C. Compared with 35 C, the time to start germination and t 50 were also delayed at 30, 33, or 38 C (Table 1).
Effect of temperature on the seed germination of Solanum khasianum under a 12-h photoperiod. Each data point shown is mean ± SE, and the curve represents a gaussian model (Equation 3) fit to the data.

The germination of Solanum khasianum under different temperature regimes within 21 d. a

a Data shown as the mean ± SE. Different lowercase letters in the same column indicate significant differences among different temperatures based on Fisher’s protected LSD (P < 0.05).
b t 50, time to obtain 50% of cumulative germination.
c t 90, time required to reach 90% germination.
d NE, not estimated, because germination rates did not reach 90% in all replications.
Temperature is an important factor affecting weed seed germination. In this study, the most favorable temperature for the germination of S. khasianum was 35 C, which was above the temperature normally considered optimal for other Solanum species. For example, the optimum constant germination temperature for S. sarrachoides was 30 C (Zhou et al. Reference Zhou, Deckard and Ahrens2005a), for S. nigrum, it was 26 or 30 C (Taab and Andersson Reference Taab and Andersson2009), and for S. ptycanthum, it ranged from 28 to 33 C (Zhou et al. Reference Zhou, Deckard and Messersmith2005b). Solanum khasianum is primarily distributed in tropical and subtropical regions with relatively high temperatures (Aubriot and Knapp, Reference Aubriot and Knapp2022), which might be an adaptation to the high temperatures in its native environment (Ouahzizi et al. Reference Ouahzizi, Elbouny, Sellam, Alem and Bakali2023). The invasion area of S. khasianum may expand in response to the progress of global warming. Similarly, germination of carpetweed (Mollugo verticillata L.), eclipta [Eclipta prostrata (L.) L.], and small flower morningglory [Jacquemontia tamnifolia (L.) Griseb.] was also strongly driven by relatively high temperatures, with the optimum temperature range for these species being 35 to 40 C (Sharpe and Boyd Reference Sharpe and Boyd2019; Shaw et al. Reference Shaw, Smith, Cole and Snipes1987). However, temperatures exceeding 40 C can completely inhibit germination by affecting enzymatic activities in weeds (Yue et al. Reference Yue, Jin, Lu, Gong, Han, Liu and Wu2021), which is consistent with the results of this study. Unlike the strict temperature requirements for seed germination, the seedlings of S. khasianum exhibit cold tolerance and can survive in environments with an average temperature of −7.39 C (Han et al. Reference Han, Liu, Gao and Lv2025). Furthermore, Yang et al. (Reference Yang, Liu, Zhang, Wang and Cheng2017) identified numerous cold-responsive miRNAs in S. khasianum, providing a molecular basis for its cold tolerance. This may explain why S. khasianum has also invaded high-altitude and colder areas in China, including the Tibet Autonomous Region and Sichuan Province (MEEPRC 2016). This study only investigated the effect of constant temperatures on S. khasianum seed germination. In fact, alternating day/night temperature regimes are more consistent with natural conditions. In addition, a temperature-based study on the germination dynamics of S. elaeagnifolium involving multiple biological parameters has been conducted, including base temperature, optimal temperature, and ceiling temperature. These parameters enabled accurate prediction of its seed germination under variable temperature regimes across different ecosystems, providing insights for future research on S. khasianum (Kapiluto et al. Reference Kapiluto, Eizenberg and Lati2022).
Effect of Light on Germination
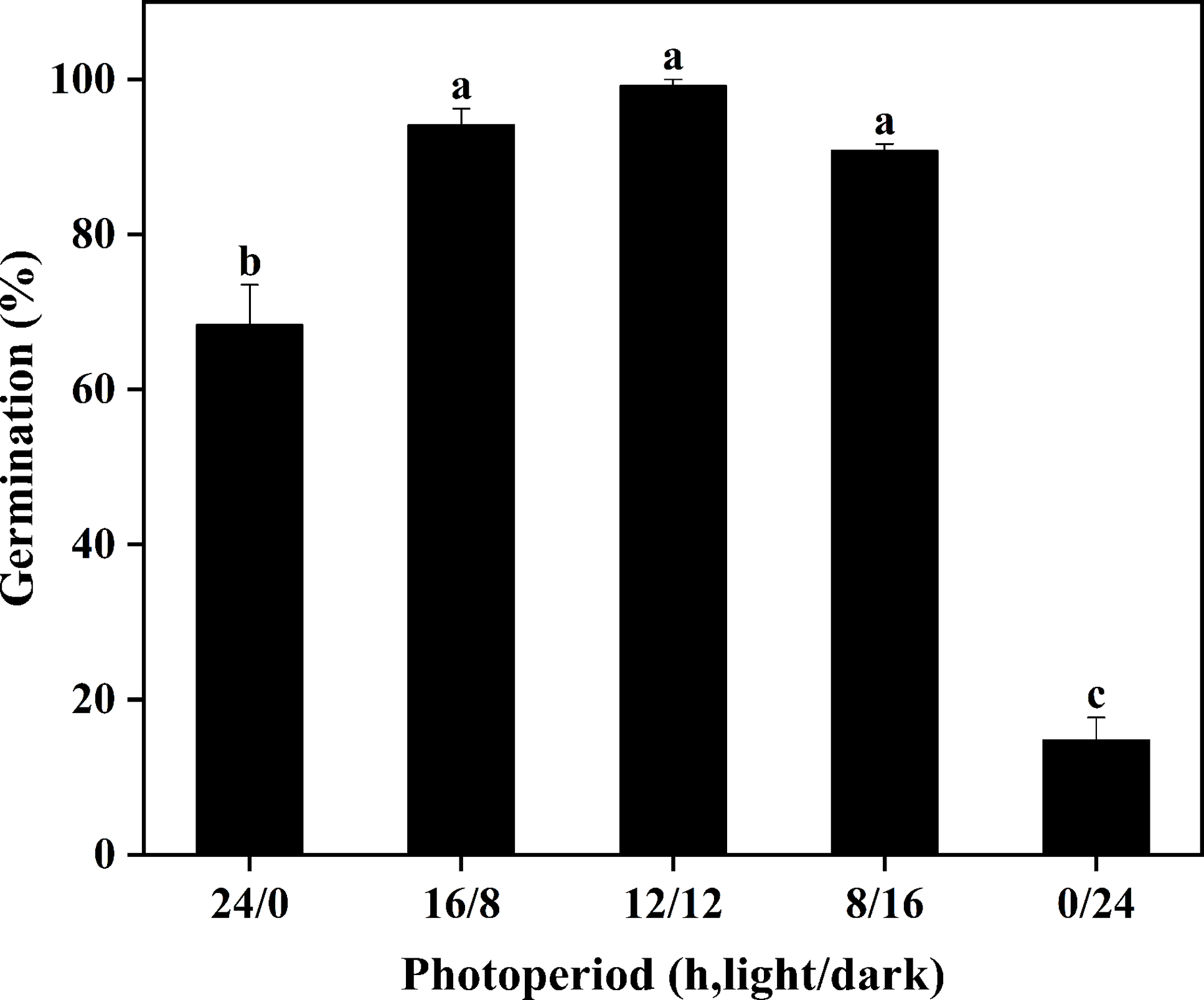
The GR was 15% under complete darkness (0/24 h), whereas exposure to continuous light (24/0 h) increased germination to 68% (Figure 2). Compared with complete darkness and continuous light, 16/8-h, 12/12-h, and 8/16-h light/dark cycles significantly promoted the seed germination with GRs of 94%, 99%, and 91%, respectively. This indicated that the germination of S. khasianum requires alternating light and darkness, but is not sensitive to the time change of photoperiod.
Effects of photoperiod on the seed germination of Solanum khasianum at 35 C. Each data point shown is mean ± SE. Different lowercase letters indicate significant differences among different photoperiods (P < 0.05).

Similar to our study, continuous light or darkness also significantly reduced the GR of fireweed (Senecio madagascariensis Poir.) (Hooda and Chauhan Reference Hooda and Chauhan2024), common lambsquarters (Chenopodium album L.) (Tang et al. Reference Tang, Guo, Yin, Ding, Xu, Wang, Yang, Xiong, Zhong, Tao and Sun2022), green galenia [Galenia pubescens (Eckl. and Zeyh.) Druce] (Mahmood et al. Reference Mahmood, Florentine, Chauhan, McLaren, Palmer and Wright2016), spiny threecornerjack [Emex spinosa (L.) Campd.], and southern threecornerjack (Emex australis Steinh.) (Javaid and Tanveer Reference Javaid and Tanveer2014). However, the response of seed germination to light varies across different weed species. Many studies have shown that light does not affect the germination of certain weeds, such as drunken horse grass [Achnatherum inebrians (Hance) Keng] (Yue et al. Reference Yue, Jin, Lu, Gong, Han, Liu and Wu2021), muskweed (Myagrum perfoliatum L.) (Honarmand et al. Reference Honarmand, Nosratti, Nazari and Heidari2016), and Japanese brome (Bromus arvensis L.) (Li et al. Reference Li, Tan, Yuan, Du, Ma and Wang2015).
Effect of pH on Germination
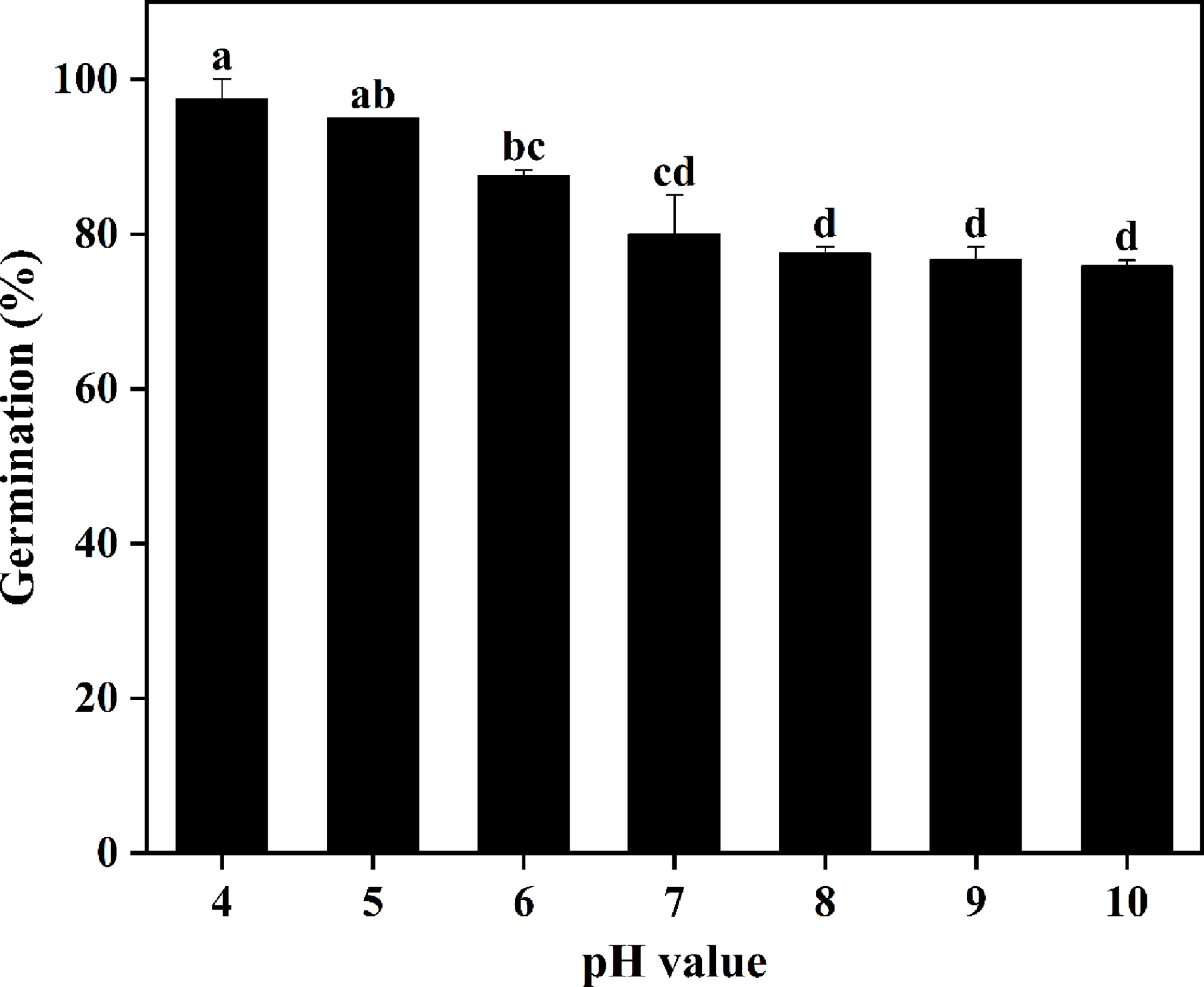
Seeds of S. khasianum germinated at all pH ranges evaluated in the study (Figure 3). The GR gradually decreased with increase in pH value, with the highest (98%) and lowest (74%) germination observed at pH 4 and pH 10, respectively. In general, the germination of seeds preferred a strongly acidic condition, whereas neutral or strongly alkaline conditions slightly inhibited the germination.
Effect of pH solutions on the seed germination of Solanum khasianum at 35 C. Each data point shown is mean ± SE. Different lowercase letters indicate significant differences at different pH levels (P < 0.05).

Like many invasive weeds, such as Tausch’s goat grass (Aegilops tauschii Coss.) (Wang et al. Reference Wang, Zhao, Li, Chen, Liu and Wang2020), C. album (Tang et al. Reference Tang, Guo, Yin, Ding, Xu, Wang, Yang, Xiong, Zhong, Tao and Sun2022), and common sowthistle (Sonchus oleraceus L.) (Chauhan et al. Reference Chauhan, Gill and Preston2006), S. khasianum could germinate across a wide range of pH values. Unlike S. elaeagnifolium, which prefers alkaline conditions (Balah et al. Reference Balah, Hassany and Mousa2021), the results showed that strongly acidic conditions were more suitable for the germination of S. khasianum, which may be related to the generally acidic soil in its collection area (Yang et al. Reference Yang, Jia, Yang, Li, Duan, Hu and Cui2023). This characteristic could also explain why S. khasianum has severely invaded grasslands in Yunnan Province, where acidic soil conditions are prevalent. Moreover, S. khasianum seedlings can also grow normally in acidic or aluminum-rich soils, and they still maintain growth under stress via intrinsic physiological and photosynthetic protection mechanisms (Li Reference Li2008; Ma et al. Reference Ma, Wu, Zhao, Zhao, Cai, Zhou and Tang2023). In recent years, severe soil acidification has occurred in major croplands worldwide, especially in tropical and subtropical regions (Goulding Reference Goulding2016; Guo et al. Reference Guo, Liu, Zhang, Shen, Han, Zhang, Christie, Goulding, Vitousek and Zhang2010). The area of strongly acidic soil (pH < 5.5) in China has increased by approximately 4 million ha over the past 30 yr (Yang et al. Reference Yang, Jia, Yang, Li, Duan, Hu and Cui2023). This trend may be more favorable for the further invasion of S. khasianum.
Effect of Osmotic Stress on Germination
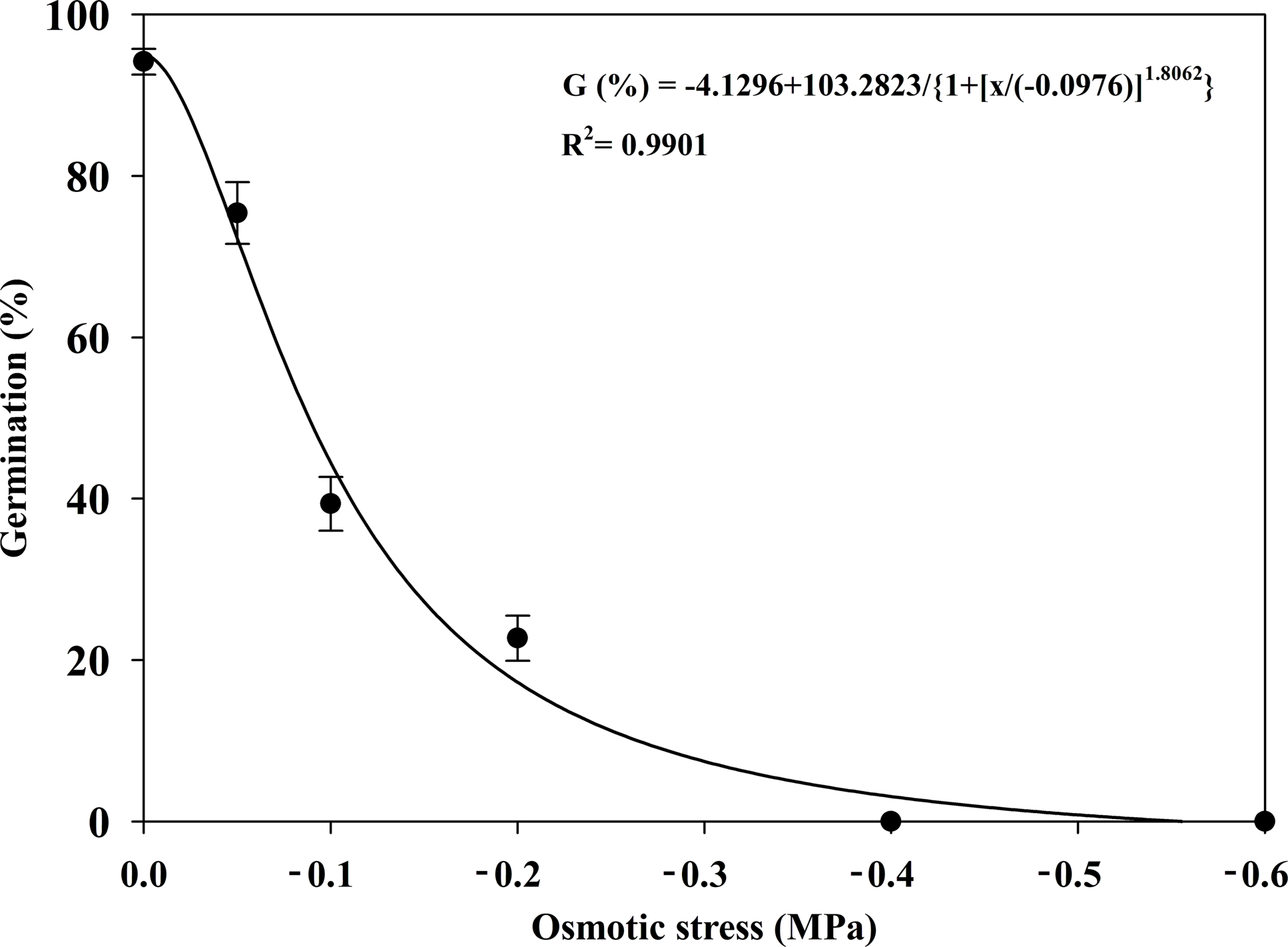
Osmotic stress significantly affected the germination of S. khasianum (Figure 4). Seeds germinated only within a narrow range of osmotic potentials. The GRs decreased sharply from 94% to 23% when the osmotic potential decreased from 0 to −0.2 MPa, while no seeds germinated at −0.4 MPa. The osmotic potential required to reduce the maximum GR by 50% was estimated to be approximately −0.10 MPa.
Effect of osmotic potential on the seed germination of Solanum khasianum at 35 C. Each data point shown is mean ± SE, and the curve represents a four-parameter logistic model (Equation 5) fit to the data.

In contrast to S. khasianum, S. sarrachoides and tropical soda apple (Solanum viarum Dunal) showed higher tolerance to osmotic potentials, with GRs of 17% and 69% at −1.0 MPa, respectively (Akanda et al. Reference Akanda, Mullahey and Shilling1996; Zhou et al. Reference Zhou, Deckard and Ahrens2005a). Generally speaking, low osmotic potentials exhibited a strong effect on the germination of S. khasianum. This might also explain why S. khasianum tends to invade regions with moist soil. For example, the average annual precipitation at the seed collection site in 2023 was 1,050.3 mm (YPDWR 2024), which is classified as a typical humid region with relatively high soil moisture content. This sensitivity to low osmotic potentials has also been observed in tall morning glory [Ipomoea purpurea (L.) Roth] (Singh et al. Reference Singh, Ramirez, Sharma and Jhala2012), Asia Minor bluegrass (Polypogon fugax Nees ex Steud.) (Wu et al. Reference Wu, Li, Xu and Dong2015), M. verticillata, Carolina geranium (Geranium carolinianum L.) (Sharpe and Boyd Reference Sharpe and Boyd2019), and motherwort (Leonurus cardiaca L.) (Susko 2025). In contrast, tolerance to low osmotic potentials has been reported in some weed species. For example, B. japonicus and A. tauschii could germinate at an osmotic potential as low as −1.2 MPa (Li et al. Reference Li, Tan, Yuan, Du, Ma and Wang2015; Wang et al. Reference Wang, Zhao, Li, Chen, Liu and Wang2020).
Effect of Salt Stress on Germination
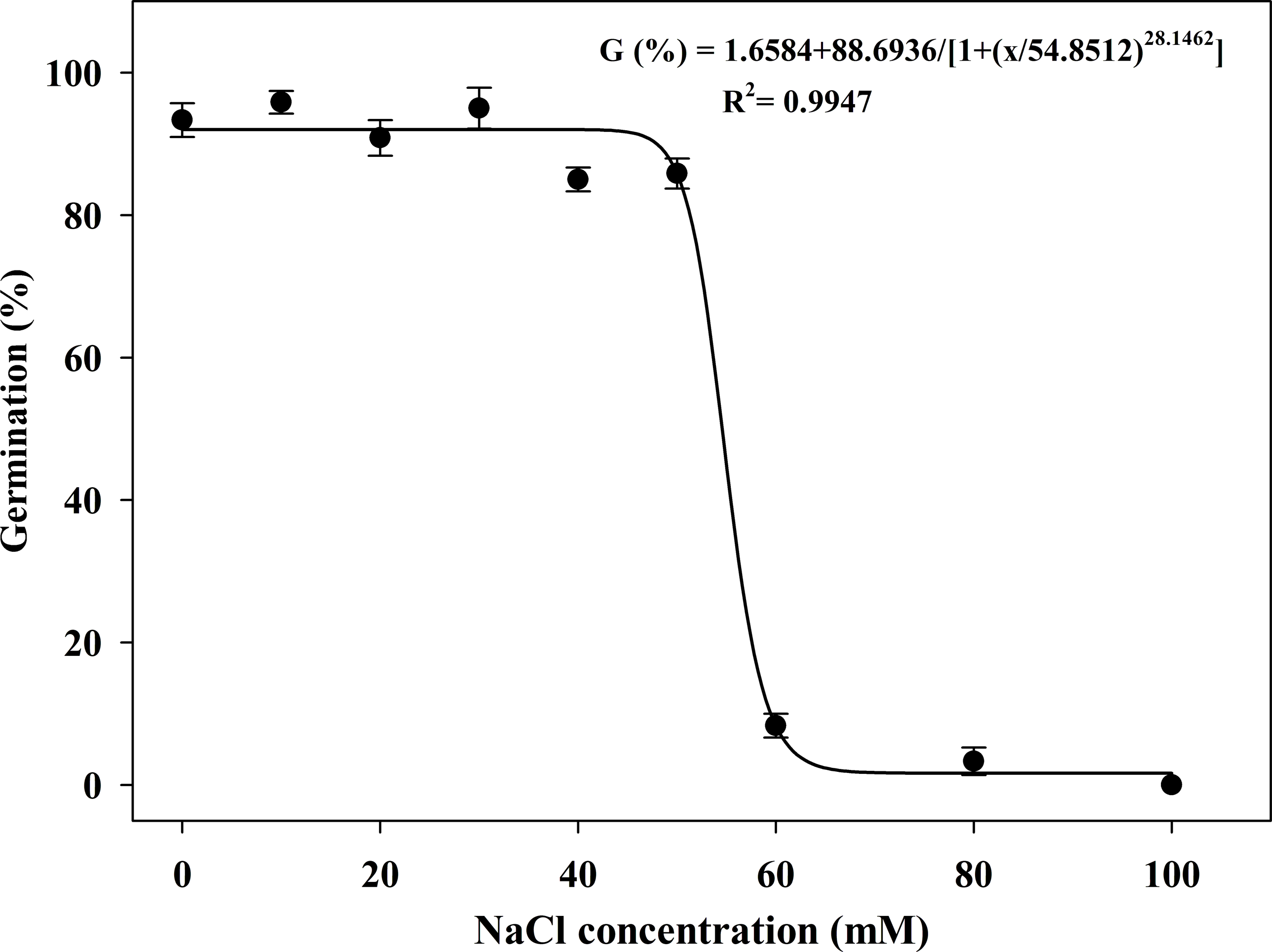
Salinity tolerance could be observed in germination within concentrations of up to 50 mM NaCl. Subsequently, GRs decreased rapidly with increasing salt concentration. The GR was 8% at 60 mM NaCl, and no seed germination was observed at 100 mM NaCl. The estimated NaCl concentration required for 50% inhibition of germination was 55.10 mM (Figure 5). In addition, salt stress also significantly affected the initial germination time. The shortest (5.00 d) and longest (16.00 d) initial germination times were recorded at 0 mM and 80 mM NaCl, respectively.
Effect of NaCl concentration on the seed germination of Solanum khasianum at 35 C. Each data point shown is mean ± SE, and the curve represents a four-parameter logistic model (Equation 5) fit to the data.

Salinity is one of the most important abiotic limiting factors for seed germination. Germination is delayed under critical concentrations of salt. The critical concentration range of NaCl for S. khasianum was 50 mM to 60 mM, which was lower than the concentrations tolerated by some other weeds. In addition to GR, the initial germination time of seeds was also significantly affected. For example, the onset of germination was rapidly delayed to 11.33 d at 60 mM NaCl. Soils with 40 to 100 mM NaCl are considered moderately saline (Chauhan and Johnson Reference Chauhan and Johnson2009), which poses challenges for S. khasianum germination. The soil in Qujing City is nonsaline-alkali, with a salt content of approximately 1.65 g kg−1 in the 0- to 20-cm soil layer, which is feasible for the growth of S. khasianum (Shi et al. Reference Shi, Deng, Zhang and Su2009). In fact, the significantly reduced germination in response to salinity has also been confirmed in other Solanum species (Ahmed et al. Reference Ahmed, Johnson, Burchett and Kenny2006; Balah et al. Reference Balah, Hassany and Mousa2021). In contrast to S. khasianum, S. centrale showed considerable salinity tolerance and germinated in solutions with NaCl concentrations up to 200 mM (Ahmed et al. Reference Ahmed, Johnson, Burchett and Kenny2006). Salinity was believed to exert osmotic stress by inhibiting water uptake in seeds, thereby preventing germination (DiTommaso Reference DiTommaso2004). In this study, the results indicated S. khasianum was susceptible to salt stress, which was consistent with its response to osmotic stress. This feature means that it is difficult for S. khasianum to invade dry and high-salinity areas.
Effect of High-Temperature Pretreatment on Germination
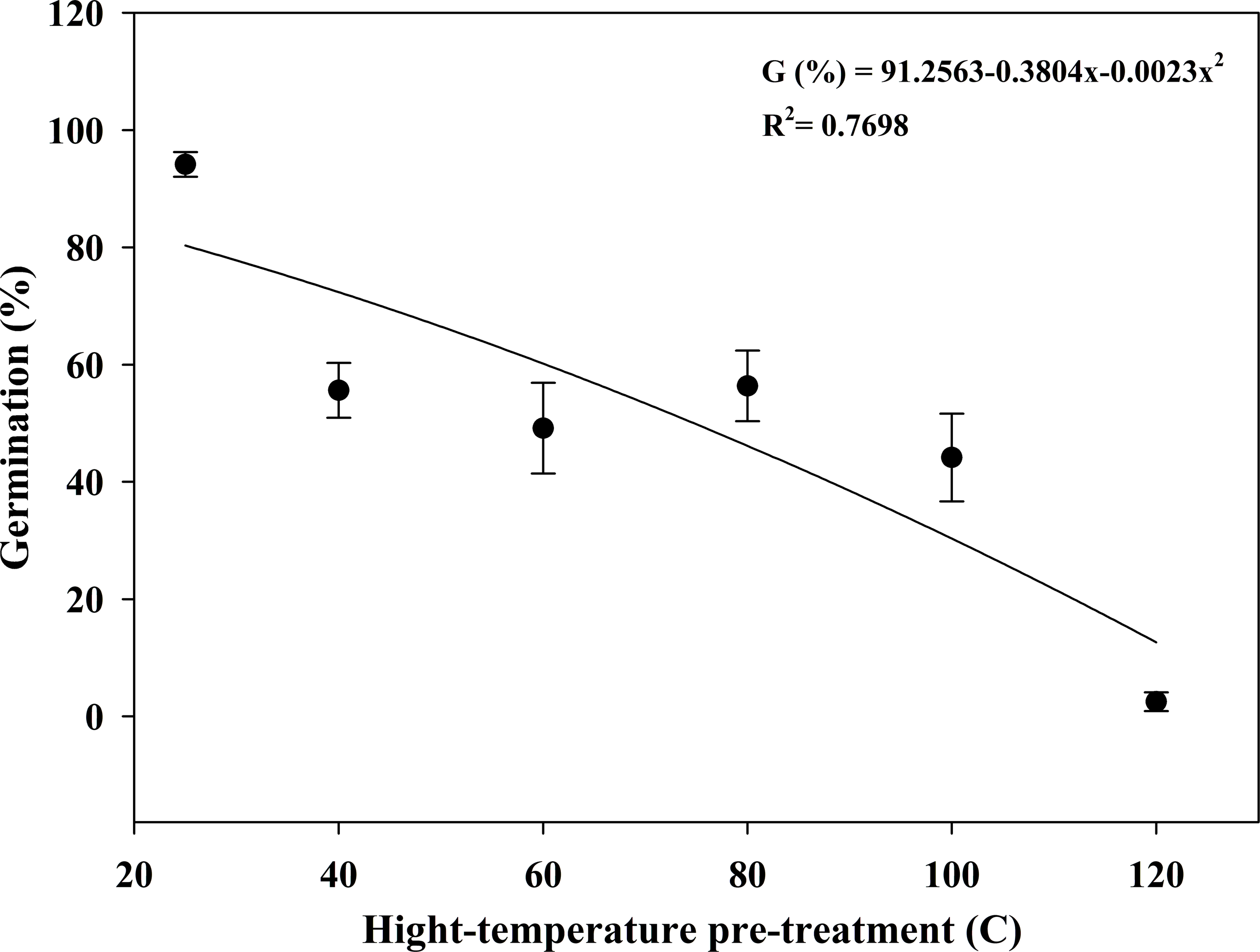
High-temperature pretreatment greatly reduced the GR of the seeds. The 40 C pretreatment alone decreased the GR from 94% to 56%, and then the GR values were maintained at approximately 44% under pretreatment temperatures up to 100 C. However, seeds hardly germinated at 120 C with a GR value of 3% (Figure 6).
Effect of high-temperature pretreatment for 5 min on the seed germination of Solanum khasianum at 35 C. Each data point shown is mean ± SE, and the curve represents a nonlinear model (Equation 4) fit to the data.

Similarly, Asian spiderflower (Cleome viscosa L.) (Akbari-Gelvardi et al. Reference Akbari-Gelvardi, Siahmarguee, Ghaderi and Gherekhloo2021), cutleaf geranium (Geranium dissectum L.) (Atabaki et al. Reference Atabaki, Gherekhloo, Ghaderi-Far, Ansari, Hassanpour-Bourkheili and Prado2023), and A. tauschii (Wang et al. Reference Wang, Zhao, Li, Chen, Liu and Wang2020) lost the ability to germinate following pretreatment at 100 C, 120 C, and 130 C, respectively. Burning in grasslands can increase soil surface temperatures, which may considerably reduce or eliminate seeds in the soil seedbank (Morgan Reference Morgan1999). Burning has also been shown to effectively control weeds in Japanese lawngrass (Zoysia japonica Steud.) grasslands (Hu et al. Reference Hu, Zhang, Yang, Qi and Jia2007). In China, grassland burning authorized by designated departments is allowed for production purposes. For instance, the Inner Mongolia Forestry and Grassland Bureau had launched pilot projects for the comprehensive utilization of straw and its planned burning (NFGA 2024). In addition, many factors such as burial depth also affected seed survival in soil following fire. Therefore, as a simple and effective weed control technology, crop residue burning could serve as a method to control weed seeds on or near the soil surface (Spoth et al. Reference Spoth, Haring, Everman, Reberg-Horton, Greene and Flessner2022; Walsh et al. Reference Walsh, Newman and Powles2013).
Effect of Burial Depth on Emergence
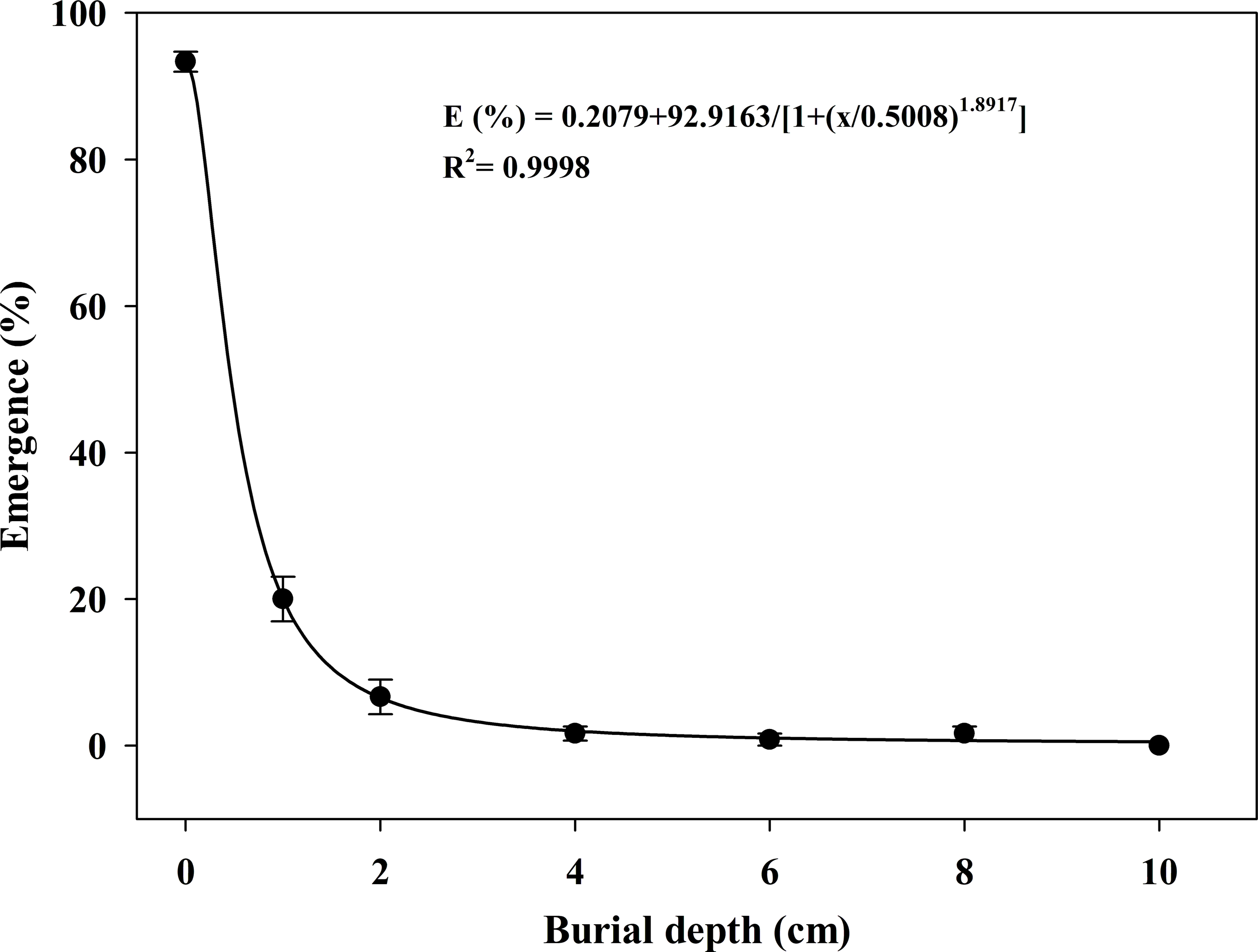
The effect of burial depth on seedling emergence is shown in Figure 7, which also conformed to the four-parameter sigmoid model. The optimum predicted percentage of seedling emergence was 93%, which occurred at 0-cm depth. Seedling emergence sharply decreased as burial depth increased from 0 to 2 cm, and almost no seedlings emerged as the depth continued to increase (4 to 8 cm). No seedlings emerged when seeds were sown at a depth of 10 cm. Meanwhile, the burial depth required to achieve 50% of maximum seedling emergence was estimated to be 0.49 cm (Figure 7).
Effect of burial depth on the seedling emergence of Solanum khasianum at 35 C. Each data point shown is mean ± SE, and the curve represents a four-parameter logistic model (Equation 5) fit to the data.

In this study, S. khasianum exhibited the highest emergence rates when seeds were sown on the surface, a pattern similar to that of S. sarrachoides (Zhou et al. Reference Zhou, Deckard and Ahrens2005a) and S. americanum (Forte et al. Reference Forte, Nunes, Filho, Galon, Chechi, Roso, Menegat, Rossetto and Franceschetti2019). In contrast, the optimum predicted emergence rate of S. viarum was observed at a depth of 5.6 cm (Akanda et al. Reference Akanda, Mullahey and Shilling1996). Generally, some large seeds are believed to have the ability to emerge from greater depths due to more seed reserves (Baskin and Baskin Reference Baskin and Baskin1998; Wu et al. Reference Wu, Li, Xu and Dong2015). For example, citronmelon [Citrullus lanatus (Thunb.) Matsum. & Nakai var. citroides (L.H. Bailey) Mansf.], with a seed diameter of 6 mm, achieved an emergence rate of over 60% even when buried at 10 cm (Ramirez et al. Reference Ramirez, Jhala and Singh2014). In contrast, small-seeded species such as mucronate sprangletop [Leptochloa panicea (Retz.) Ohwi; syn. Dinebra panicea var. brachiata (Steud.)] (Weller et al. Reference Weller, Florentine and Chauhan2019) and camelina [Camelina sativa (L.) Crantz] (Sanehkoori et al. Reference Sanehkoori, Pirdashti and Bakhshandeh2023), with seed lengths of approximately 1 to 2 mm, only emerged from extremely shallow burial depths. Although seeds of S. khasianum are larger than those of many weeds, seedlings hardly appeared when seeds were buried deeper than 2 cm. This poor tolerance to burial depth might be attributed to the light requirement for seed germination (Forte et al. Reference Forte, Nunes, Filho, Galon, Chechi, Roso, Menegat, Rossetto and Franceschetti2019), which further confirms the earlier results regarding light (Figure 2). As a consequence, tillage operations that bury seeds deeper than 2 cm might be beneficial for controlling S. khasianum in grassland.
This study comprehensively explored the effects of multiple environmental factors on the germination of S. khasianum. However, studies have shown that different populations of the same weed exhibit distinctly different germination behaviors under identical environmental conditions (Kapiluto et al. Reference Kapiluto, Eizenberg and Lati2022; Ma et al. Reference Ma, Huang, Huang, Guo, Saeed, Jiang, Chen and Wei2021). The seeds in this study were collected from a single population in Yunnan Province, which is insufficient to fully demonstrate the germination characteristics at the species level. Therefore, further studies involving more populations from other regions are needed to comprehensively reveal the germination traits of S. khasianum.
In summary, the results indicated that the germination of S. khasianum was affected by various environmental factors. Specifically, the germination was strongly driven by relatively high temperatures and alternating light/dark cycles. Acidic conditions favored germination more than the neutral to alkaline conditions. In addition, seeds were sensitive to osmotic stress and salt stress, and they hardly germinated at a NaCl concentration of 60 mM or an osmotic potential of −0.4 MPa. Burying seeds at a depth of 2 cm or more or pretreating them at 120 C for 5 min effectively suppressed germination. This study partially revealed the adaptive mechanism of invasive S. khasianum in Yunnan, China. Whether deep tillage or burning is applicable in grasslands where native species had already been established for managing this invasive weed warrants further investigation.
Funding statement
This work was supported by National Natural Science Foundation of China (no. 32302407), China Agricultural Research System (no. CARS-34), Qingdao Agricultural University Doctoral Start-Up Fund (no. 6631123002) and Graduate Innovation Program of Qingdao Agricultural University (QNYCX24040).
Competing interests
The authors declare there are no conflicts of interest.