Functional gastrointestinal disorders (FGID), or disorders of gut–brain interaction, are characterised by the spectrum of persistent and recurring symptoms attributable to the gastrointestinal (GI) tract, such as diarrhoea, constipation, abdominal pain, bloating, nausea and vomiting(Reference Duffy, Boggiano and Ganesh1). FGID comprise a heterogeneous group of disorders, which impose significant negative impacts on functional status, health-related quality of life and economic burden(Reference Palsson, Tack and Drossman2). It has been estimated that more than 40 % of the worldwide general population may meet the diagnostic criteria for at least one FGID at any given time(Reference Palsson, Tack and Drossman2). These disorders are more prevalent in females than males, and they are also more likely to have severe symptoms and comorbid conditions such as depressive and anxiety disorders(Reference Narayanan, Anderson and Bharucha3).
The exact pathophysiology of FGID remains poorly understood; however, visceral hypersensitivity, GI motility disturbance, dysbiosis, mucosal and immune dysfunction, inflammation, altered gut–brain axis, psychological disturbances and environmental factors play viable roles(Reference Black, Drossman and Talley4). Food is an important trigger of FGID symptoms and most people with FGID associate their symptoms with the consumption of specific food items including alcohol, caffeine, spicy foods, wheat or dairy products, dietary fibre, dietary fat, processed food and fried foods(Reference Simrén, Månsson and Langkilde5,Reference Hayes, Fraher and Quigley6) .
Amongst various dietary factors that contribute to the initiation of FGID, the advanced glycation end products (AGE) devoted considerable attention recently. AGE can potentially induce oxidative stress and inflammation, which can affect gut health(Reference de Graaf, Scheijen and Spooren7). These dietary products are a large and heterogeneous group of compounds produced by the spontaneous reaction between free amino groups of lipids, proteins or nucleic acids with reducing sugar such as glucose(Reference van Dongen, Linkens and Wetzels8).
Animal studies showed that high AGE diets could alter the gut microbiota composition and structure and induce gut inflammation(Reference Wang, Cai and Yu9). Furthermore, a previous study revealed that plasma levels of AGE were correlated with GI disorders in autistic children(Reference Ghodsi and Kheirouri10). In this context, a recent study on patients with inflammatory bowel disease (IBD) and irritable bowel syndrome (IBS) demonstrated that dietary intake of dicarbonyls and AGE was not higher in patients with IBD and IBS compared with healthy controls and these products were not associated with intestinal inflammation(Reference de Graaf, Scheijen and Spooren7).
To our knowledge, there is no evidence examining the association of dietary intake of AGE with FGID in adults. Given that the popularity of Western diet, a diet rich in processed food and thus AGE(Reference Bettiga, Fiorio and Di Marco11) is increasing in both developed and developing countries and the harmful effects of dietary AGE on GI health is still under debate, assessing the relation between dietary intake of AGE and risk of FGID would shed light on the management of the disease. Moreover, although sex-based differences exist in the prevalence, pathophysiology and management of FGID(Reference Narayanan, Anderson and Bharucha3), the effects of these biological differences on the development and management of FGID remain uncertain. Therefore, the aim of this study was to evaluate the association between dietary intake of AGE and the risk of FGID in an Iranian adult population. This study also aimed to explore potential sex-based differences in the association of AGE with FGID.
Methods and materials
Study population
This cross-sectional analysis was conducted using the baseline data collected for the Isfahan functional disorders cohort study. Isfahan functional disorders was designed to investigate the epidemiology, risk factors, course and prognosis of functional somatic syndrome in a sample of Iranian adults in Isfahan province. The study design, sampling procedures, characteristics of the participants and data collection procedure have been presented elsewhere(Reference Adibi, Ani and Vaez12). In total, 1930 seemingly healthy adults (aged between 18 and 65) living in the Kerdabad neighbourhood of Isfahan, Iran, were included using census-based sampling technique. Information regarding functional symptoms, psychological assessment, quality of life, socio-demographic and lifestyle factors were collected by self-administered questionnaires. Trained personnel followed the standard protocols for measuring the anthropometric data. Written informed consents were obtained from all participants prior to their participation. This study protocol was approved by the National Research Ethics Committee of the Iranian Ministry of Health and Medical Education and the Research Ethics Committee of Isfahan University of Medical Sciences (IR.MUI.REC.1395.1.149).
Dietary intake assessment
A self-administered, dish-based, 106-item semi-quantitative FFQ (DS-FFQ), which was designed and validated specifically for Iranian adults, was applied to collect habitual dietary intake of participants. The design, foods and validity of this questionnaire are available elsewhere(Reference Keshteli, Esmaillzadeh and Rajaie13). Subjects were asked to report the amount and frequency of consumption of each food item during the past year on a daily (e.g. bread), weekly (e.g. rice, meat) or monthly (e.g. fish) basis. Household measures were used to convert all the portion sizes of consumed foods to grams. Nutrient analysis of diets was done using Nutritionist IV software (Version 7·0; N-Squared Computing, Salem, OR, USA) with its database adapted for Iranian foods. Finally, the diurnal intakes of all food items were calculated and used in statistical analyses.
Assessment of advanced glycation end product
For the calculation of the AGE scores, we used the AGE content (kU/100 g) of food items which was reported previously(Reference Uribarri, Woodruff and Goodman14). Of the 106 food items of our FFQ, the AGE content of seventy-two food items was measured. Finally, the AGE values of all food items were summed to obtain the AGE score for each individual.
Assessment of functional gastrointestinal disorders
The diagnosis of FGID was done based on Rome IV criteria for adults. The Rome IV diagnostic questionnaires have been created and validated for screening of FGID and can be used in clinical trials as well as in epidemiological research. This criterion classifies the FGID based on symptoms rather than physiological criteria. The adult Rome IV Diagnostic Questionnaire has up to eighty-six questions and recommends the 90th percentile symptom frequency as the threshold of abnormality in GI. Diagnostic questions were subjected to criterion validity by clinical diagnostics using clinical experts through a recursive process. Twenty-two FGID diagnoses were assigned according to Rome IV criteria, based on responses to the Rome IV Diagnostic Questionnaire(Reference Drossman15,Reference Palsson, Whitehead and Van Tilburg16) . In this study, those who had one of the most prevalent upper or lower FGID (including functional dyspepsia, IBS, functional heartburn, globus, functional dysphagia, nausea and vomiting disorders, functional constipation, functional diarrhoea, functional abdominal bloating/distension) were considered as suffering from FGID.
Assessment of other variables
BMI was calculated by dividing weight (kg) to height (m2). Physical activity was determined using the International Physical Activity Questionnaire and expressed as the metabolic equivalent (MET) h/week(Reference Craig, Marshall and Sjöström17). Depression and anxiety symptoms were evaluated using the Hospital Anxiety and Depression Scale questionnaire which has been validated for the Iranian population(Reference Montazeri, Vahdaninia and Ebrahimi18). The Pittsburgh Sleep Quality Index questionnaire was used to assess the sleep quality of the study participants(Reference Buysse, Reynolds and Monk19). The validity and reliability of the complete Pittsburgh questionnaire for the Iranian population have been confirmed(Reference Chehri, Nourozi and Eskandari20).
Statistical analysis
Mean (sd) and percentages were used to summarise the continuous and categorical variables, respectively. Normality of continuous variables was evaluated using Kolmogorov–Smirnov test and Q–Q plots. Participants were categorised based on tertiles of dietary intake of AGE. The Chi-square test for categorical variables and independent samples t test (or Mann–Whiney U test) and ANOVA (or Kruskal–Wallis test) for continuous variables were conducted to compare the general characteristics and dietary intakes of study participants with and without FGID as well as across the tertiles of AGE. Univariate and multiple logistic regression analyses were performed to examine the association between tertiles of dietary AGE and the risk of FGID. Crude and adjusted OR and 95 % CI are presented. The lowest tertile of AGE intake was considered as the reference group. In the crude model, the relationship between the tertiles of AGE intake and FGID was estimated without adjustment of confounding variables. Sex and age were adjusted in the first model. Further adjustments were applied for BMI, socio-economic status and smoking in the second model. In the third model, the confounding effect of physical activity and suffering from physical and mental chronic disease were controlled. The fourth model was additionally adjusted for energy intake. Confounding variables selection was done based on a literature review and observed baseline incomparability across dependent and independent variables(Reference Jahromi, Tehrani and Teymoori21,Reference VanderWeele22) . The data were analysed using the Statistical Package for Social Sciences (version 20; SPSS Inc.). P < 0·05 was considered significant in all statistical analyses.
Results
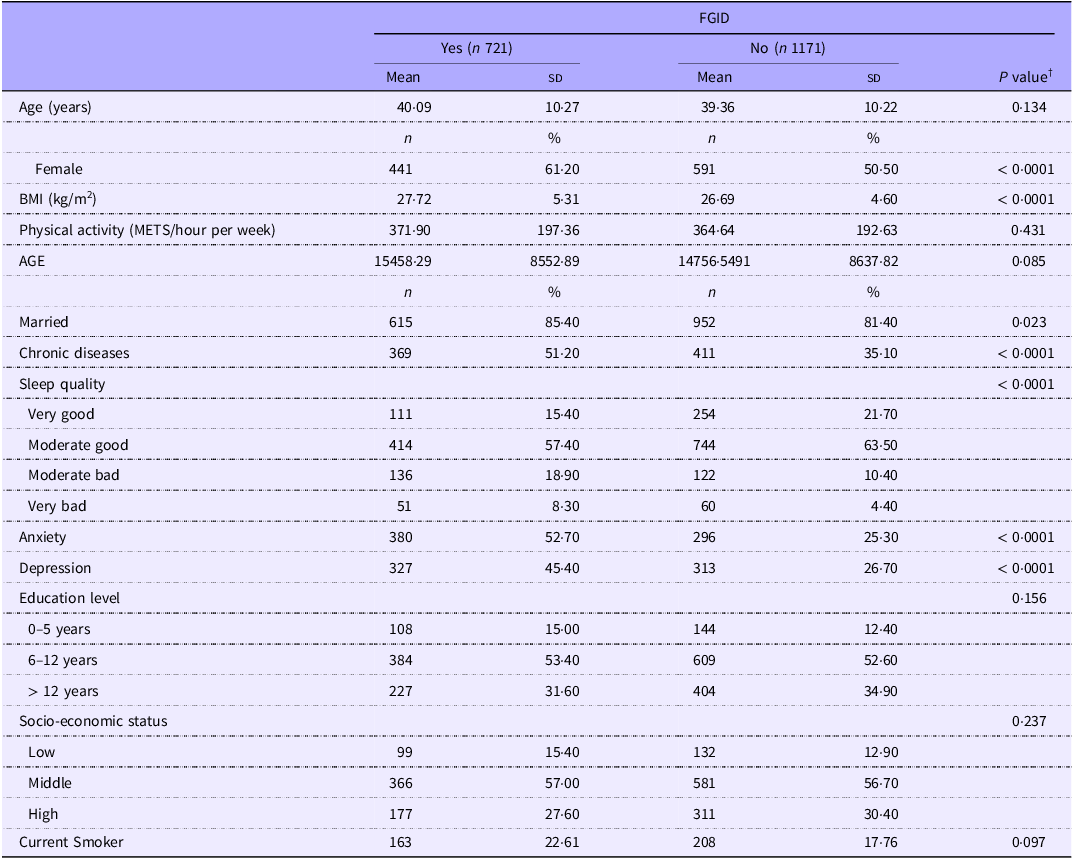
The study included a total of 1892 participants, among them 721 (38 %) subjects had at least one of the most prevalent FGID. FGID were prevalent among 42·70 % of females and 32·60 % of males. The mean age and BMI of the study participants were 39·64 (sd 10·24) years and 27·08 (sd 4·91) kg/m2, respectively. The mean of AGE was 14 690·10 (sd 8797·25) (kU/gr). The general characteristics of participants according to the presence or absence of FGID are reported in Table 1. Subjects with FGID were more likely to be female, married, depressed and anxious. They also had a higher BMI and history of chronic diseases and lower sleep quality. No other statistically significant differences were observed.
General characteristics of participants according to the presence or absence of FGID (Mean values and standard deviations; numbers and percentages) *

AGE, advanced glycation end products; FGID, functional gastrointestinal disorders.
* Values are mean (sd) for continuous variables and frequency (percentage) for categorical variables.
† Derived from independent samples t test for continuous and chi-square test for categorical variables.
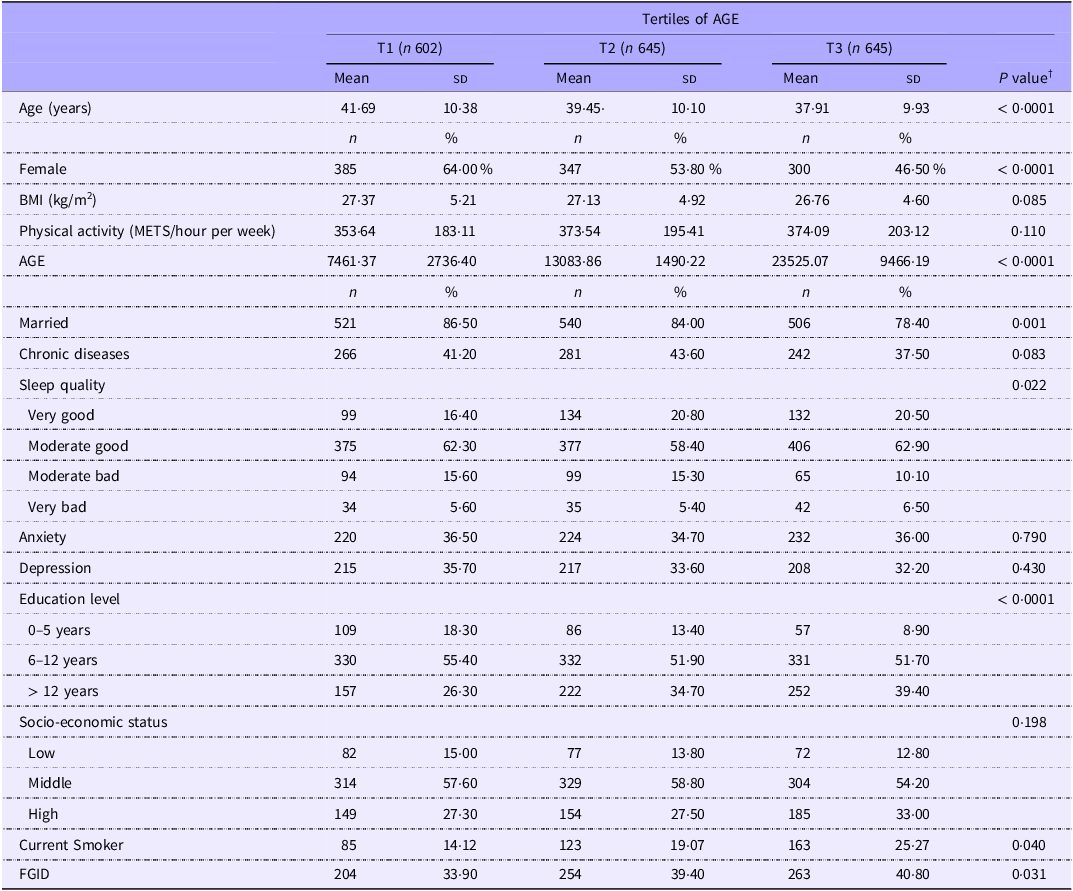
Table 2 presents the general characteristics of participants across tertiles of AGE. Compared with individuals in the lowest tertile of the AGE, those in the highest tertile were younger, more likely to be smoker, highly educated and have FGID, but less likely to be female and married. Furthermore, sleep quality was lower in the highest tertile of AGE in comparison with the lowest tertile. No other significant differences were observed across the tertiles of AGE.
General characteristics of participants according to tertiles of dietary advanced glycation end products (AGE) (Mean values and standard deviations; numbers and percentages) *

AGE, advanced glycation end products; FGID, functional gastrointestinal disorders.
* Values are mean (sd) for continuous variables and frequency (percentage) for categorical variables.
† Derived from ANOVA for continuous and chi-square test for categorical variables.
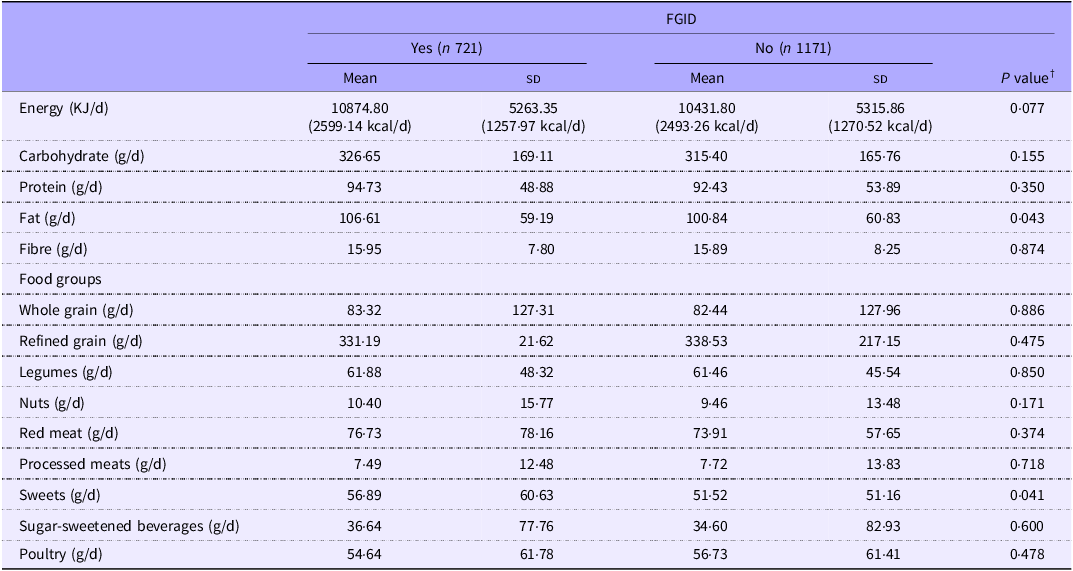
Dietary intakes of participants according to the presence or absence of FGID are depicted in Table 3. Sweets were consumed in greater amounts by the subjects with FGID (P = 0·041). There were no other statistically significant differences between groups.
Dietary intakes of participants according to the presence or absence of FGID (Mean values and standard deviations) *

FGID, functional gastrointestinal disorders.
* Values are mean (sd).
† Derived from independent samples t test or Mann–Whitney U test.
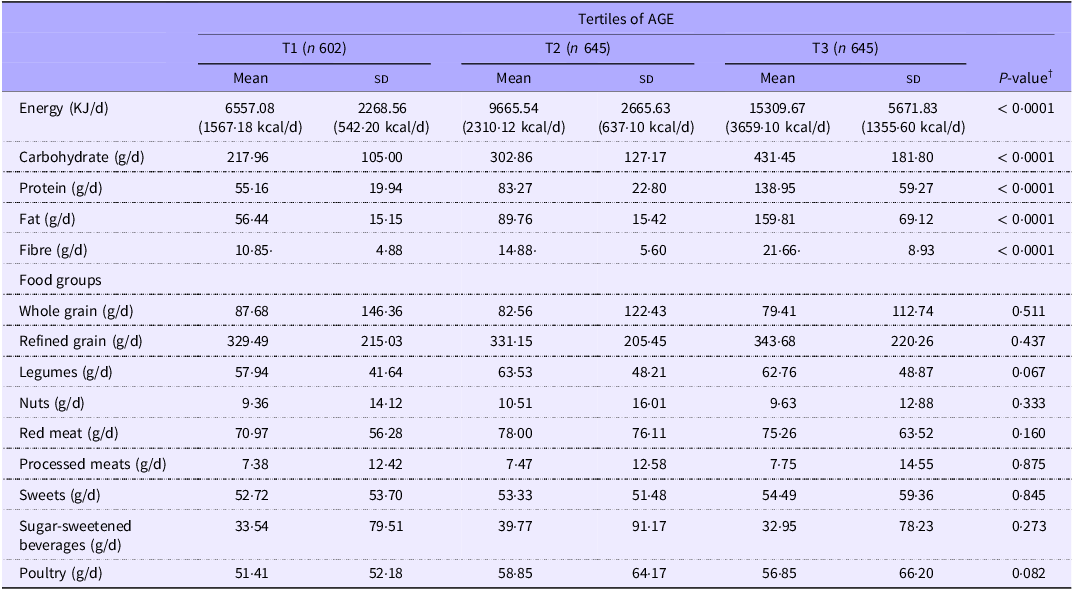
Table 4 illustrates the dietary intakes of study participants across the tertiles of AGE. Energy, carbohydrates, proteins, fat and fibre were consumed in greater amounts by individuals in the highest tertile of AGE compared with those in the lowest tertile. No other statistically significant differences were seen between the different tertiles of AGE in terms of food group consumption.
Dietary intakes of study participants across the tertiles of AGE (Mean values and standard deviations) *

AGE, advanced glycation end products.
* Values are mean (sd).
† Derived from ANOVA or Kruskal–Wallis tests.
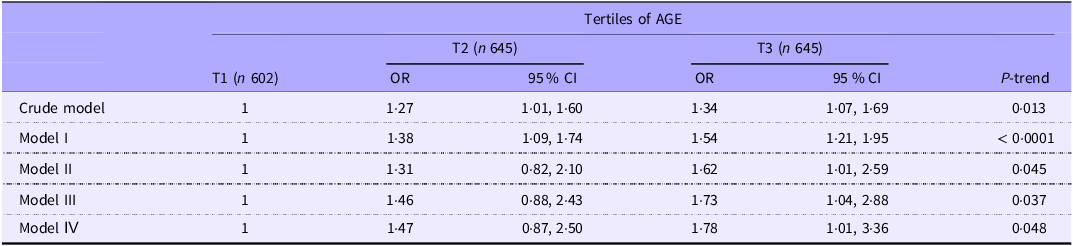
The crude and multivariable-adjusted OR and 95 % CI for FGID across tertiles of AGE are shown in Table 5. In the crude model, being in the highest v. lowest tertile of AGE was associated with increased odds of FGID (OR = 1·34; 95 % CI: 1·07, 1·69), and this association remained significant and strengthened after adjustment for demographic confounders, lifestyle confounders, mental, chronic diseases and energy intake in multivariable-adjusted model (OR = 1·78; 95 % CI: 1·01, 3·36).
Crude and multivariable-adjusted OR and 95 % CI for FGID across the tertiles of dietary AGE in total sample (OR and 95 % CI) *

AGE, advanced glycation end products; FGID, functional gastrointestinal disorders.
Model I: Adjusted for age and sex.
Model II: Additionally adjusted for BMI, socio-economic status and smoking.
Model III: Additionally adjusted for physical activity, mental and chronic diseases.
Model IV: Additionally adjusted for energy intake.
* Values are OR and 95 % CI.
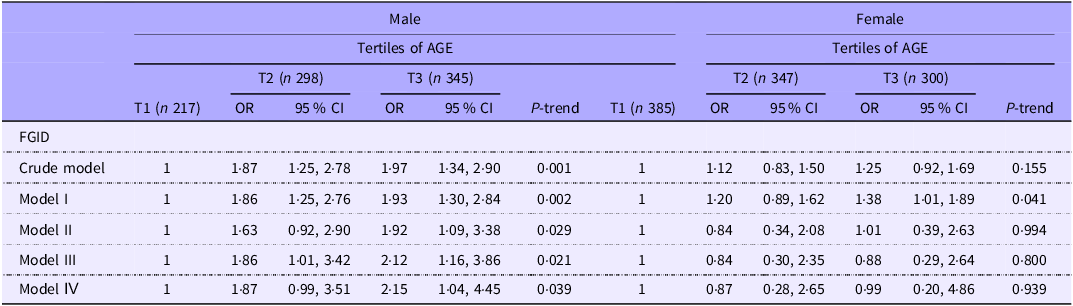
Table 6 presents the crude and multivariable-adjusted OR and 95 % CI for the association of AGE with FGID stratified by sex. In males, individuals in the highest tertile of AGE had higher odds of FGID than those in the lowest tertile either in crude or adjusted models. In the fully adjusted model, being in the highest v. the lowest tertile of AGE almost doubled the risk of FGID (OR = 2·15; 95 % CI: 1·04, 4·45).
Crude and multivariable-adjusted OR and 95% CI for FGID across the tertiles of dietary AGE stratified by sex (OR and 95 % CI) *

AGE, advanced glycation end products; FGID, functional gastrointestinal disorders.
Model I: adjusted for age.
Model II: additionally adjusted for BMI, socio-economic status and smoking.
Model III: additionally adjusted for physical activity, mental and chronic diseases.
Model IV: additionally adjusted for energy intake.
* Values are OR and 95 % CI.
In females, the AGE intake was not related to the risk of FGID in the crude model (OR: 1·25; 95 % CI: 0·92, 1·69). Adjusting for age led to a significant increase in the risk of FGID in subjects in the highest tertile of AGE compared with the lowest tertile (OR = 1·38; 95 % CI: 1·01, 1·89). However, after further control for lifestyle confounders, the association was no longer significant in model II (OR = 1·01; 95 % CI: 0·39, 2·63). The association also remained non-significant in model III (OR = 0·88; 95 % CI: 0·29, 2·64) and the fully adjusted model (OR = 0·99; 95 % CI: 0·20, 4·86).
Discussion
In this cross-sectional study, we found an independent positive relationship between high AGE consumption and the risk of FGID. In sex-stratified analysis, this association was evident only among men and no significant association in the fully adjusted model was observed in women.
There is only limited and conflicting evidence in terms of AGE in relation to GI disorders. Most of the available studies have shown that AGE are a contributing factor to the development of IBD(Reference Ciccocioppo, Vanoli and Klersy23–Reference Chen, Zhao and Gregersen25). Contrarily, a recent examination of 238 patients with IBD, 261 patients with IBS and 195 healthy controls failed to find any association between the amounts of dietary advanced glycation end products and intestinal inflammation(Reference de Graaf, Scheijen and Spooren7). The reason for their findings might be related to the limited number of assessed AGE (only three components) while there are many Maillard reaction productions. Although the AGE–FGID relationship has been poorly studied, some AGE-rich food products like ultra-processed foods have been investigated in more detail. The results of these studies demonstrated that a higher intake of ultra-processed foods was associated with higher odds of IBD or IBS incidence(Reference Chen, Wellens and Kalla26–Reference Lo, Khandpur and Rossato31). In contrast, a large prospective cohort study in eight European countries revealed a null association between ultra-processed foods and IBD but a lower risk for IBD development in those with higher consumption of unprocessed/minimally processed foods(Reference Meyer, Dong and Casagrande27).
The results of our study suggested a sex-specific association between dietary advanced glycation end products and FGID. One possible explanation behind the increased odds of FGID in males, but not females, with higher AGE intake might be attributable to sex and gender variations in prevalent FGID symptoms(Reference Narayanan, Anderson and Bharucha3,Reference Kim and Kim32) . FGID in women usually coexist with anxiety and depression and are more severe owing to differences in response to stress hormones and sexual hormones(Reference Narayanan, Anderson and Bharucha3,Reference Kim and Kim32) . This may suggest the superiority of biological factors over environmental factors for FGID in women compared with men. On the other hand, men who have lower biological disturbances than women may be more vulnerable to the interaction between biological and environmental factors and therefore more likely to be influenced by dietary factors. However, the reasons for sex-specific association between AGE and FGID need to be investigated by future studies.
The exact mechanisms underlying the association of AGE with GI disorders have not been well-established. However, it seems that the effects of dietary advanced glycation end products on GI disorders are mediated through two main pathways, that is, a direct effect after absorption by the gut and an alteration in gut microbiota(Reference Nie, Li and Qian33). Intestinal cells are continuously exposed to AGE. The binding of AGE with transmembrane receptors for AGE in the intestinal epithelial cells may induce pro-inflammatory pathways, causing the development and progression of a variety of diseases(Reference Nie, Li and Qian33). Furthermore, AGE can modify gut microbiota and thereby influence the integrity of the intestinal barrier(Reference Nie, Li and Qian33,Reference Jansen, Fogliano and Rubert34) . In a rat-model intervention study, higher AGE consumption was associated with the depletion of goblet cells, impaired tight junctions zonulin-1 expression in the colon and increased gut permeability(Reference Nie, Li and Qian33,Reference Jansen, Fogliano and Rubert34) . Increased intestinal permeability, also known as leaky gut, besides dysbiosis and modified gut–brain axis may cause FGID(Reference Uribarri, Woodruff and Goodman14,Reference Camilleri, Lasch and Zhou35,Reference Barbara, De Giorgio and Stanghellini36) . In addition, due to limited absorption of AGE, their accumulation in the ileum and colon is associated with increased levels of oxidative stress and pro-inflammatory cytokines as a result of downregulating enzymatic antioxidative and stimulating pro-inflammatory cytokines production(Reference Nie, Li and Qian33). Elevated levels of oxidative stress and inflammatory cytokines adversely affect long-lived proteins and also result in the development of various diseases(Reference Nie, Li and Qian33).
Our study is one of the first nutritional epidemiologic studies on human subjects which explored the association of AGE with FGID using a large sample size and considering some relevant confounders like depression. However, the findings should be interpreted with caution. The lack of detailed information on the cooking method in our FFQ is the principal limitation of our study as it may impact the AGE content of foods(Reference Uribarri, Woodruff and Goodman14). Furthermore, causal inferences cannot be shown by our cross-sectional study. However, reverse causation bias seems unlikely in our study since given that foods are the main triggers of symptoms in GI disorders, patients with FGID would not tend to increase the consumption of foods rich in AGE over time. Moreover, dietary intake was evaluated using self-administered questionnaire, possibly leading to misclassification of participants and different associations. Estimating AGE and nutrient and intake using tools which have not been specifically for Iranian foods might be another limitation of the current study. However, since our findings are in line with earlier studies, which is a way for validation, our study results can be reliable. Finally, we adjusted energy intake as a covariate and did not use the residual method. However, there is evidence suggesting that both models can lead to comparable results and therefore our applied approach can consider any issues raised by energy intake(Reference Day, Wong and Bingham37).
In conclusion, our findings provide evidence to support a direct link between dietary advanced glycation end products intake and the risk of FGID. Stratified analysis by sex showed that this association was more evident among men. Longitudinal studies are required to confirm our findings in other populations. Moreover, the impact of AGE reduction on the amelioration or prevention of FGID warrants further examination in clinical trials.
Acknowledgements
We wish to thank all participants and their families for their commitment to this study. We acknowledge the management of Isfahan province health center and also Isfahan health center #1 for their administrative support. We also acknowledge the staff and health workers at Kerdabad health center for their gracious role in recruiting and following the participants.
This work has been supported by the Iranian Ministry of Health and Medical Education (700/144), Isfahan University of Medical Sciences (195149), the Iranian National Institute for Medical Research Development (NIMAD) (972 953), and the Ethics Committee of Isfahan University of Medical Sciences (IR.ARI.MUI.REC.1403.213).
PA, HR were responsible for the organization and coordination of the study, developed the study design and provided feedback on methods, results and discussion parts of all manuscript revised versions. FH, PH contributed to the writing the first draft of the manuscript and the reviewing and editing of the final manuscript. AF performed data analysis and contributed to the reviewing and editing of the final manuscript. TG contributed to the reviewing and editing of the final manuscript. FH, PH and AF designed and developed the figures and tables. HSh, AA contributed to the management or administration of the study. All authors approved the final version of the manuscript.
The authors declare that there are no conflicts of interest.