Trial Registration: ROLO: ISRCTN54392969 registered at www.isrctn.com; MicrobeMom: ISRCTN53023014 at www.isrctn.com.
Ensuring adequate nutrient intake during pregnancy is critical for maternal well-being, fetal development and the long-term health of offspring(Reference Killeen, Donnellan and O’Reilly1). Pregnancy is a time of increased nutritional demands, yet these requirements often go unmet, contributing to adverse health outcomes for both mother and child(Reference Fanzo, Bellows and Spiker2,Reference Hanson, Bardsley and De-Regil3) . Addressing these nutritional needs must also align with the dual challenges of achieving universal nutrition and environmental sustainability; however, these goals remain largely unmet globally(Reference Fanzo, Bellows and Spiker2).
Poor nutrition represents a major contributor to the global disease burden(Reference Beal, Ortenzi and Fanzo4). Concurrently, food systems drive 34 % of human-induced greenhouse gas emissions, consume 70 % of global freshwater and contribute to habitat conversion and species extinction(Reference Beal, Ortenzi and Fanzo4,Reference Crippa, Solazzo and Guizzardi5) . Recognising this, the EAT–Lancet Commission proposed the ‘planetary health diet’ (PHD) as a model for achieving both health and sustainability(Reference Willett, Rockström and Loken6). This framework emphasises flexible intake limits across food groups, with a focus on reducing highly processed and animal-derived foods(Reference Willett, Rockström and Loken6). The planetary health diet index (PHDI) is a novel measure adapted to quantify alignment with the EAT–Lancet Commission’s PHD(Reference Frank, Jaacks and Adair7,Reference Bui, Pham and Wang8) .
Reduced intakes of animal-source foods (ASF) present notable challenges, particularly in meeting essential nutrient requirements. Limiting ASF can exacerbate existing micronutrient and protein deficiencies, making it more challenging to obtain adequate amounts of key nutrients such as folate, Fe, Ca and vitamin D(Reference Sebastiani, Herranz Barbero and Borrás-Novell9–Reference Beal, Ortenzi and Fanzo11). These nutrients are particularly important during pregnancy to support maternal health and fetal development, including neural tube formation, bone mineralisation, oxygen transport and immune function(Reference Oh, Keats and Bhutta12–Reference Schröder-Heurich, Springer and von Versen-Höynck14). Deficiencies in these nutrients can lead to adverse outcomes such as neural tube defects, preterm birth, low birth weight (bw) and maternal complications, emphasising the need for optimal dietary intake during this crucial period(Reference Hanson, Bardsley and De-Regil3,Reference Molloy, Kirke and Brody15,Reference Lindsay, Gibney and McAuliffe16) .
Supporting the increased nutritional demands of pregnancy often requires careful dietary planning, particularly within the context of dietary models that encourage plant-based eating(Reference Sebastiani, Herranz Barbero and Borrás-Novell9). Plant-based sustainable diets have raised concerns regarding potential challenges in obtaining essential nutrients(Reference Leonard and Kiely17). Emerging research suggests that higher PHDI scores are linked to a lower likelihood of Fe, fibre and potassium inadequacy(Reference Frank, Jaacks and Adair7). While the PHD has been widely studied for its role in reducing the risk of CVD, type 2 diabetes and asthma, its suitability during pregnancy remains an important and under-researched area(Reference Sawicki, Ramesh and Bui18,Reference Ojo, Jiang and Ojo19) .
The European Food Safety Authority (EFSA) uses scientific data to set dietary reference values (DRV) for many nutrients, including population reference intakes (PRI), average requirements (AR), adequate intakes (AI) and reference intakes (RI)(20). Some of these values are also adjusted for different life stages, including pregnancy, with increased reference values for specific nutrients including folate, vitamin D, Zn, Ca, Fe and protein(20).
The primary aim of this study was to examine how moderate alignment with the PHD relates to nutrient intake and adherence to EFSA pregnancy dietary guidelines in healthy pregnant women in early pregnancy. Rather than assessing strict adherence to the PHD, the PHDI was used to capture the degree to which an individual’s diet aligns with the PHD principles.
Materials and methods
Study design
This is an analysis of dietary data collected prior to intervention at baseline in early pregnancy (∼12–16 weeks) in participants recruited as part of two randomised controlled trials (RCT): the ROLO (Randomised cOntrol trial of a LOw glycaemic index diet in pregnancy to prevent macrosomia) study and the MicrobeMom study. Both trials involved healthy pregnant women attending the National Maternity Hospital, Dublin, Ireland. The ROLO study (ISRCTN54392969) was an RCT of a low glycaemic index diet during pregnancy to reduce recurrence of macrosomia(Reference Walsh, McGowan and Mahony21). The trial randomised 800 secundigravid women with a previous macrosomic baby (bw > 4 kg). Women in the control group received no dietary intervention, while those in the intervention group received low glycaemic index dietary advice during their pregnancy. The MicrobeMom RCT (ISRCTN53023014) randomised 160 women in early pregnancy to receive probiotic supplementation (Bifidobacterium breve 702 258) or placebo antenatally and up to 3 months postpartum to study the transfer to neonatal stool(Reference Moore, Feehily and Killeen22). Women were excluded from both trials if they had existing diabetes, previous gestational diabetes or any other medical conditions that required medication (online Supplementary Figure 1). Detailed methodology and results of ROLO and MicrobeMom RCT have been published previously(Reference Moore, Feehily and Killeen22,Reference Walsh, Mahony and Foley23) .
Maternal characteristics
Age, parity and BMI were recorded at the first study visit (∼12–16 weeks). Participants self-reported micronutrient supplement use, educational attainment, ethnicity and smoking status in early pregnancy (classified as current smoker in early pregnancy or non-smoker in early pregnancy). Residential address, retrieved from medical records, was used to calculate the Pobal Haase and Pratschke deprivation index for each participant. This index serves as an indicator of social deprivation, reflecting three dimensions of affluence and disadvantage(Reference Haase and Pratschke24). The scores are derived by comparing a specific location to other areas at the same time point based on the most recent census data(Reference Haase and Pratschke24).
Dietary assessment in early pregnancy
Baseline dietary data were obtained from 3-d food diaries completed prior to study intervention allocation at baseline visits for each study (∼12–16 weeks’ gestation). Three-day food diaries were chosen as the method of collecting information to capture regular eating on a diet, minimise recall bias and reduce the likelihood of dietary misreporting. All participants were instructed to record their typical food and beverage consumption over three consecutive days, ensuring the inclusion of at least one weekend day. Including at least one weekend day accounts for variations between weekday and weekend eating habits, ensuring a more representative assessment of usual dietary intake. They were instructed to document the type, amount and branding of all food and drink consumed during this period, quantifying their intakes using either the manufacturer’s weight on the food packaging or household measures (e.g. tablespoons). In the ROLO study, food diaries were analysed using the software NetWISP version 3.0 (Tinuviel Software) by a research dietitian using household measures and average portion sizes from the UK Food Standards Agency(Reference Crawley, Mills and Patel25). The NetWISP food composition database was derived from the 6th edition of McCance and Widdowson’s Food Composition Tables(Reference McCance and Widdowson26). For the MicrobeMom study, nutrient data were analysed using the Nutritics Research Edition V.7.3 software. Food groups were taken from McCance and Widdowson’s Food Composition Tables (fifth and sixth editions, including all supplementation editions) and Nutritics proprietary 2018 database (downloaded from Nutritics). Mean daily intake of macronutrients and micronutrients derived from dietary sources was generated for early pregnancy. All nutrient intakes reflect nutrient intake from dietary sources and do not account for supplemental contributions. Nutrient and total energy data, including both energy-yielding and non-energy-yielding components, were exported from Excel into SPSS version 29.0 for further statistical analysis. Food diary records were used to categorise foods into ASF groups (online Supplementary Figure 4).
The Goldberg equation was applied to assess dietary misreporting by evaluating the ratio of energy intake to estimated BMR(Reference Goldberg, Black and Jebb27). BMR was calculated using the Schofield equations, which are age-specific and rely on weight measurements taken at booking(Reference Schofield28). Dietary under-reporting was identified with an energy intake to BMR ratio of < 0·9, while plausible reporting was determined with a ratio of ≥ 0·9(Reference McGowan and McAuliffe29).
Calculation of the planetary health diet index score
PHDI scores were assigned to each participant based on the dietary data collected from 3-d food diaries. The PHDI score was computed based on alignment with the PHD recommendations(Reference Frank, Jaacks and Adair7). Detailed information about the scoring methods has been described previously(Reference Frank, Jaacks and Adair7). In brief, each participant’s score was derived from their dietary patterns, which were grouped into adequacy components and moderation components. Adequacy components include whole grains, whole fruits (excludes fruit juice), non-starchy vegetables, nuts and seeds, legumes and unsaturated oils. Moderation components include starchy vegetables, dairy products, red and processed meat, poultry, eggs, fish, saturated oils and trans-fats and added sugar and fruit juice. For moderation components, scores reflected proportional deviation from the EAT–Lancet reference range and represent relative dietary alignment rather than compliance with recommended PHD intake thresholds. Component scores from 0 to 10 were assigned with intakes between the minimum (0) and maximum (10) scored proportionately. Once component scores were assigned, the scores for all fourteen components were summed to create a total PHDI score ranging from 0 to 140(Reference Frank, Jaacks and Adair7). A higher PHDI score reflects better alignment with the PHD. For the present analysis, participants were dichotomised based on the median PHDI score in early pregnancy into a ‘Low’ (≤ 88·99) and ‘High’ (score > 88·99) PHDI group. The ‘High PHDI’ group does not reflect strict adherence to the PHD but rather a relatively higher alignment with its principles compared with those in the ‘Low PHDI’ group.
Comparison with European Food Safety Authority guidelines
All DRV were obtained from the EFSA DRV for Nutrients summary report (2017)(20), which includes specific reference values for pregnant women. The recommended intakes applied in this study are summarised in Tables 4 and 5.
Energy intake (kcal) adherence could not be calculated as the metabolic equivalent of task data for MicrobeMom participants was unavailable. Total sugars were not analysed as the EFSA guidelines do not specify an upper limit. Saturated fats are synthesised endogenously and are not essential in the diet; therefore, no reference value is established. Similarly, no reference values have been set for unsaturated fat intake. As per WHO (2017) guidelines, Na intake was not evaluated because the assessment of Na recommendations is ongoing. Zn evaluation requires data on the level of phytate intake, which was not available for this analysis.
When defining recommended nutrient intake, protein PRI and AR were specified by trimester, so for this analysis, the 2nd trimester recommendation was applied (PRI: ≥ 0·83 g/kg bw (+) 9 g/d and AR: ≥ 0·66 g/kg bw + 7·2 g/d). The RI for carbohydrates is 45–60 % of total energy and 20–35 % of total energy for fat. Ca intake was analysed by age group: for women aged 18–24, the RI was 1000 mg/d and AR: 860 mg/d; for those aged ≥ 25, the RI was 950 mg/d and AR: 750 mg/d. All other micronutrient recommendations are displayed in Table 6.
Statistical analysis and justification of sample size
This study analysed dietary data from pregnant women in the MicrobeMom and ROLO cohorts at baseline prior to any study intervention. Participants were eligible for inclusion in this analysis if they had completed a 3-d food diary in early pregnancy (∼12–16 weeks) from which a PHDI score could be calculated, leading to a total sample size of 678 women (online Supplementary Figure 1). All statistical analyses were performed using SPSS version 29.0 for Mac (Macintosh). Assessment of normality for all continuous variables was based on a conceptual understanding of the data, visual inspection of histograms and interpretation of the Kolmogorov–Smirnov test. Results were presented as mean and sd for normally distributed continuous variables or median and interquartile range (IQR 25th–75th percentile) for non-normally distributed continuous variables. Categorical variables were presented as frequency and percentage (n (%)).
Differences in maternal baseline characteristics and nutritional status between ‘High’ and ‘Low’ PHDI groups were assessed using independent t tests for normally distributed variables, Mann–Whitney U tests for non-normally distributed variables and χ 2 tests for categorical variables. The comparison of median nutrient intakes between the ‘High’ and ‘Low’ PHDI groups allows for a straightforward assessment of the raw differences between groups.
In addition, unadjusted and adjusted linear regression models were created using a forced entry approach, comparing the ‘High PHDI’ group with the ‘Low PHDI’ group. The PHDI was also analysed as a continuous variable (‘Per 1-point increment in PHDI score’) to preserve information and improve precision. Regression analyses allow adjustment for potential confounding factors. Confounders were chosen a priori based on previous knowledge of relevant published literature and author consensus(Reference McGowan and McAuliffe29,Reference Rhee, Cho and Willett30) . ‘Unadjusted’ results present the association between the PHDI score and nutrient intake without controlling for covariates. ‘Adjusted’ results control for total energy intake (kcal), dietary misreporting status (under reporter, yes/no), maternal age, BMI, Pobal Haase and Pratschke index score, education status and trial group (MicrobeMom or ROLO). Pairwise deletion of missing data was used due to complete data for most variables. A two-sided P-value < 0·05 was considered statistically significant for all analyses.
Average daily nutrient intakes derived from 3-d food diaries for participants in the ‘High PHDI’ and ‘Low PHDI’ groups were compared with EFSA reference values for pregnancy (PRI, AR, AI and RI, where available). Participants were considered to meet or exceed the guideline if their mean intake was equal to or greater than the corresponding EFSA reference value.
Results
Maternal characteristics
In total, 678 women who completed 3-d food diaries in early pregnancy were included in this study (Table 1 and online Supplementary Figure 1). Significant differences in maternal characteristics were noted between the ‘High’ PHDI’ and ‘Low PHDI’ groups. Women in the ‘High PHDI’ group were significantly older, had lower weight and BMI measures in early pregnancy, were of a higher socio-economic background and had higher educational attainment, compared with those in the ‘Low PHDI’ group. A descriptive overview of nutrient intake in early pregnancy in the total cohort is provided in online Supplementary Table 1.
Maternal characteristics of the total cohort in early pregnancy and a comparison of maternal characteristics between the ‘Low PHDI’ and ‘High PHDI’ groups

PHDI, planetary health diet index; IQR, interquartile range; HP, Haase and Pratshke.
BMI – maternal BMI categories as per the WHO BMI classification.
P-value < 0·05 is statistically significant.
* Non-normal distribution – compared using the Mann–Whitney U test. Expressed as median (interquartile range, IQR).
† Categorical variables – compared with χ 2 tests. Expressed as n (% denominator).
Associations between the planetary health diet index score and macronutrient intake
Differences in energy and macronutrient intake according to ‘High’ and ‘Low’ alignment with the PHD are displayed in online Supplementary Table 2, and unadjusted and adjusted associations are presented in Table 2. In adjusted models, there was a significant positive association between the ‘High PHDI’ score and higher intakes of carbohydrates (g/d), total sugars (g/d) and dietary fibre (g/d) and lower intakes of protein (g/d) and saturated fat (g/d) compared with the ‘Low PHDI’ group. When examining the score continuously rather than in ‘High’ and ‘Low’ groups, each 1-unit increase in PHDI score was associated with higher intakes of carbohydrates (g/d), total sugars (g/d) and dietary fibre (g/d) and lower intakes of protein (g/d), total fat (g/d) and saturated fat intake (g/d).
Unadjusted and adjusted associations between the planetary health diet index and energy and macronutrient intakes in early pregnancy

PHDI, planetary health diet index score.
Adjusted model is controlled for energy intake (kcal), dietary misreporting status (under reporter, yes/no), maternal age, BMI, Pobal Haase and Pratshke index score, education status and trial group (MicrobeMom or ROLO).
P < 0·05 is considered statistically significant.
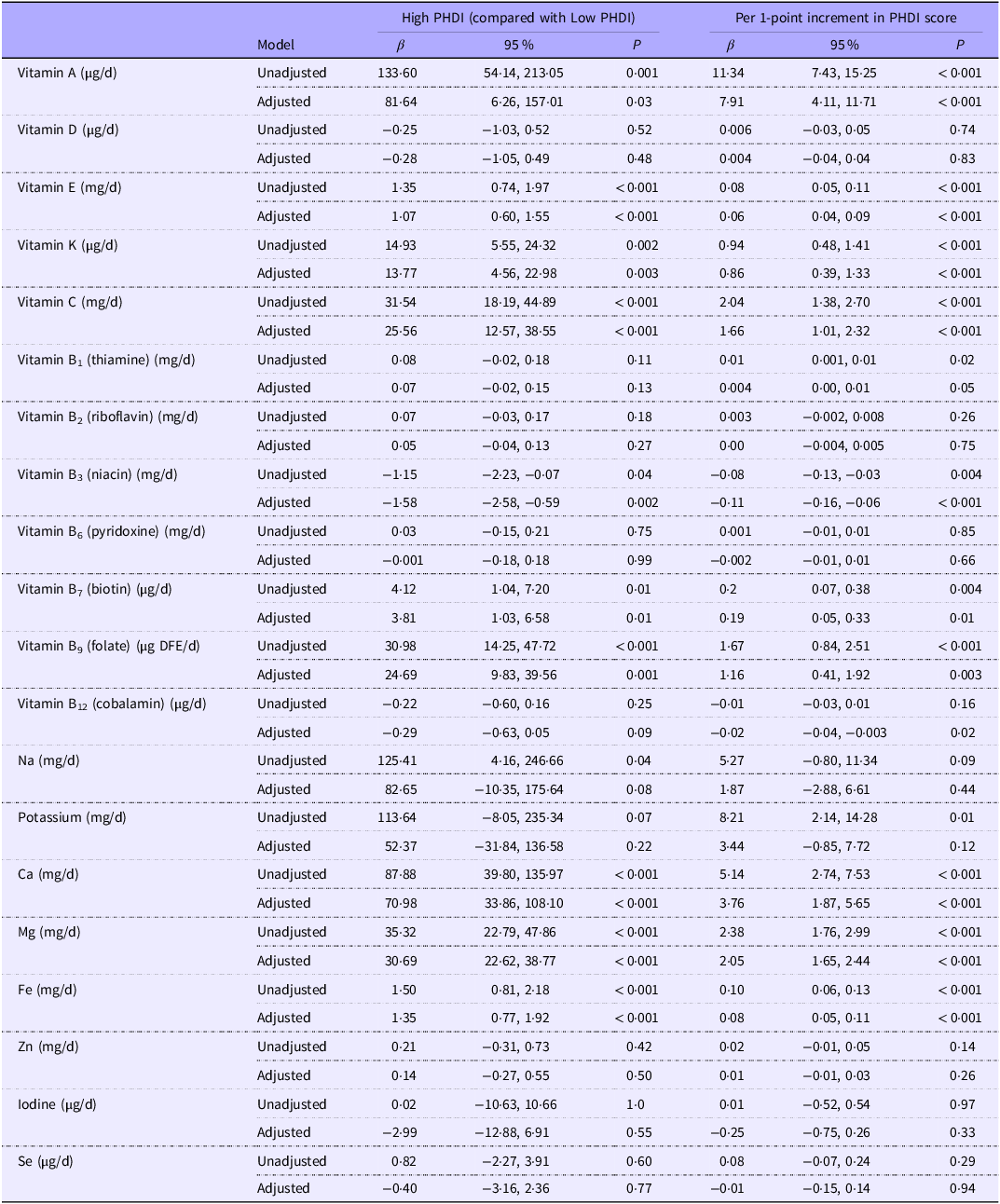
Associations between the planetary health diet index score and micronutrient intake
Differences in energy and micronutrient intake according to ‘High’ and ‘Low’ PHDI alignment are displayed in online Supplementary Table 3, with unadjusted and adjusted associations presented in Table 3. In the adjusted models, a ‘High PHDI’ score was positively associated with intakes of vitamin A (µg/d), vitamin E (mg/d), vitamin K (µg/d), vitamin C (mg/d), vitamin B7 (µg/d), vitamin B9 (µg dietary folate equivalent per d), Ca (mg/d), Mg (mg/d) and Fe (mg/d) and negatively associated with intake of vitamin B3 (mg/d). Continuous analyses showed a similar pattern, with each 1-unit increase in the PHDI score associated with higher intakes of these same micronutrients and with lower intakes of vitamin B3 (mg/d) and B12 (µg/d).
Unadjusted and adjusted associations between the planetary health diet index and energy and micronutrient intakes in early pregnancy

PHDI, planetary health diet index score; DFE, dietary folate equivalent.
Adjusted model is controlled for energy intake (kcal), dietary misreporting status (under reporter, yes/no), maternal age, BMI, Pobal Haase and Pratshke Index score, education status and trial group (MicrobeMom or ROLO).
P < 0·05 is considered statistically significant.
Associations between the planetary health diet index score and adherence to dietary recommendations in early pregnancy
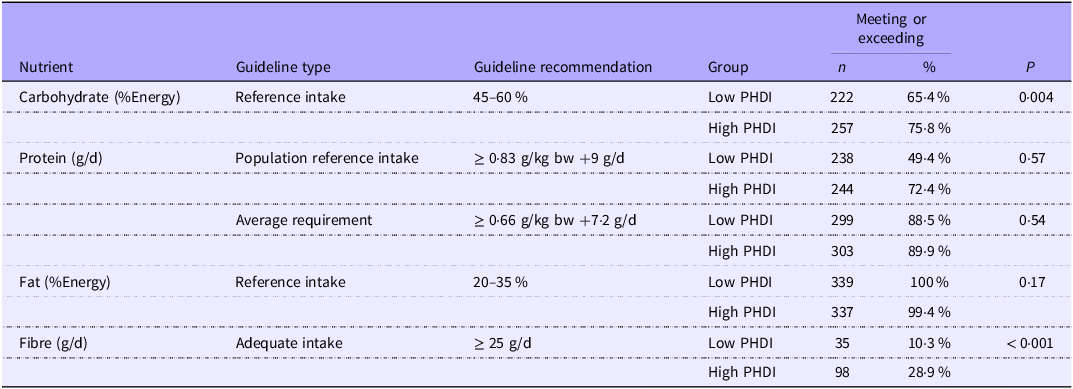
Associations between the PHDI score and adherence to dietary recommendations in early pregnancy are shown in Tables 4 and 5. For macronutrients, there were significantly more women with a ‘High PHDI’ score who met/exceeded RI recommendations for carbohydrates (percentage of total energy intake) and fibre AI recommendations (g/d), compared with those with a ‘Low PHDI’ score. For micronutrients, there were significantly more women with a ‘High PHDI’ score that met EFSA guidelines for vitamin A, vitamin E, vitamin K, vitamin C, vitamin B2, Ca, Mg and Fe, compared with those with a ‘Low PHDI’ score. Significantly more women with a ‘Low PHDI’ score met EFSA guidelines for vitamin B3 and iodine, compared with those with a ‘High PHDI’ score.
Associations between the planetary health diet index and adherence to macronutrient guidelines in early pregnancy

%Energy, percentage of total energy intake; PHDI, planetary health diet index; bw, birth weight.
Meeting (n %) = Participants meeting or exceeding the European Food Safety Authority guidelines for the specified nutrient.
P-values determined using χ 2 tests for categorical variables. P < 0·05 is considered statistically significant.
Population reference intake (PRI): The amount of nutrient required to meet the needs of virtually all individuals in a specific population group.
Average requirement: The nutrient intake level that is sufficient for half of the people in a population, assuming a normal distribution of requirements.
Adequate intake: The value estimated when a PRI cannot be established because the average requirement is unknown. It represents the average daily intake level observed in healthy individuals, assumed to be adequate.
Reference intake: The recommended intake levels for macronutrients, expressed as a percentage of total energy intake, which support health and minimise the risk of chronic diseases.
Associations between the planetary health diet index and adherence to micronutrient guidelines in early pregnancy

PHDI, planetary health diet index; NE, niacin equivalent; DFE, dietary folate equivalent.
Meeting (n %) = Participants meeting or exceeding the European Food Safety Authority guidelines for the specified nutrient.
P-values determined using χ 2 tests for categorical variables. P < 0·05 is considered statistically significant.
Population reference intake (PRI): The amount of nutrient required to meet the needs of virtually all individuals in a specific population group.
Average requirement: The nutrient intake level that is sufficient for half of the people in a population, assuming a normal distribution of requirements.
Adequate intake: The value estimated when a PRI cannot be established because the average requirement is unknown. It represents the average daily intake level observed in healthy individuals, assumed to be adequate.
Reference intake: The recommended intake levels for macronutrients, expressed as a percentage of total energy intake, which support health and minimise the risk of chronic diseases.
Associations between the planetary health diet index score and animal-source food intake
Online Supplementary Table 4 highlights the differences in ASF intake between the ‘High’ and ‘Low’ PHDI groups. In the adjusted analysis, those in the ‘High PHDI’ group had significantly lower intake of animal products, including meat and egg products (g/d). No significant difference in dairy product (g/d) intake was noted between the groups.
Discussion
Main findings
This study found that women with a ‘High PHDI’ score in early pregnancy had a higher intake of several key pregnancy-related nutrients, including folate, Fe, Ca, Mg, vitamin D, vitamin E and dietary fibre. Although intakes were higher in the ‘High PHDI’ group for certain nutrients such as folate and vitamin D, supplementation is still necessary to ensure that adequate levels are achieved in pregnancy. Concerns are often raised about the potential for lower intakes of key micronutrients when adopting diets that reduce environmental impact and limit ASF consumption(Reference Leonard, Leydon and Arranz31). However, our results indicate that women with a higher PHDI score have higher intakes of key pregnancy-related nutrients.
Macronutrient intakes
These findings indicate distinct differences in macronutrient intake among those with higher PHDI scores in early pregnancy. Participants in the ‘High PHDI’ group exhibited significantly higher intakes of carbohydrates, sugars and dietary fibre and lower intakes of protein. Those in the ‘High PHDI’ group were also significantly more likely to meet/exceed EFSA guidelines for carbohydrates and dietary fibre. These patterns align with the plant-based dietary emphasis of the PHD, which prioritises whole grains, fruits and vegetables while limiting ASF. The increased fibre intake is particularly notable, as dietary fibre plays a crucial role in digestive health and has been linked to improved pregnancy outcomes, such as reduced risk of pre-eclampsia(Reference Perry, Stephanou and Rayman32).
Adequate dietary protein intake throughout pregnancy is essential to support the normal growth and development of the fetus. Current recommendations advise 0·83 g/kg/d with an additional 1 g, 9 g and 28 g/d in the first, second and third trimesters, respectively(20). Although those in the ‘Low PHDI’ group consumed higher amounts of protein, likely due to higher intake of ASF, neither group was more likely to meet EFSA guidelines. Importantly, plant-based proteins can provide all essential amino acids when consumed in sufficient quantity and variety, and those following the PHD can increase their protein intake by increasing consumption of plant-based protein sources such as cereals, legumes, oil seeds, nuts and tubers. This approach to meeting protein requirements aligns with the PHD emphasis on minimally processed plant foods and may help to address concerns regarding protein adequacy in plant-forward dietary patterns.
No difference in total energy intake was observed between the ‘High’ and ‘Low’ PHDI groups. Although energy requirements increase across pregnancy to support maternal tissue expansion, fetal growth and rising metabolic demands, these needs vary depending on factors such as maternal BMI, age and activity level. The lack of group differences indicates that following a more plant-forward diet does not appear to reduce the overall energy intake. Monitoring gestational weight gain alongside energy intake remains important in pregnancy, particularly for women at the lower or higher end of the BMI range.
Micronutrient intakes
In the current analysis, those in the ‘High PHDI’ group were also significantly more likely to meet/exceed EFSA guidelines for Mg, Ca, vitamin C, vitamin A, vitamin K and vitamin E intakes but significantly less likely to meet vitamin B3 and iodine EFSA guidelines in early pregnancy. This is consistent with prior studies that have also reported that a higher PHDI score correlates with greater intakes of vitamin E, vitamin C and folate(Reference Cacau, Hanley-Cook and Huybrechts33). The lower intake of vitamin B3 is not surprising, as decreased consumption of ASF has previously been associated with lower intakes of B vitamins(Reference Bakaloudi, Halloran and Rippin34). This is important to note when following the PHD as vitamin B3 plays a protective role against gestational diabetes mellitus, and inadequate intake has been associated with an increased risk of fetal congenital anomalies(Reference Qin, Song and Jiang35,Reference Palawaththa, Islam and Illic36) .
Pregnancy significantly increases the risk of negative Fe balance due to greatly amplified Fe demands(Reference Georgieff, Krebs and Cusick37). Ensuring Fe sufficiency during pregnancy helps reduce maternal morbidity, supports fetal health and provides the newborn with adequate nutrient stores for early postnatal life(Reference Georgieff, Krebs and Cusick37). The observation of increased Fe intake among participants with higher PHDI scores alleviates some concerns that the PHD may be associated with poorer Fe status due to reduced meat consumption. However, although total Fe intake was higher among women with a ‘High PHDI’, the bioavailability of non-haem Fe from plant-based sources is lower (1–20 %) compared with haem Fe from animal products (15–35 %)(Reference Uriza-Pinzón, Verstraete and Franco38,Reference Qin, Pang and Hu39) . Fe requirements increase substantially during pregnancy, and inadequate Fe absorption has been associated with anaemia and adverse pregnancy outcomes such as low bw and preterm birth(Reference Bhattacharjee, Puthumana and Khan40). During the first trimester, the body’s ability to increase absorption of Fe begins at about 8 % and gradually rises to about 37 % by 36 weeks’ gestation(Reference Whittaker, Lind and Williams41). However, this physiological adaptation alone may not be sufficient to eliminate the risk of anaemia in all mothers. Vitamin C enhances non-haem Fe absorption, and in this cohort, the ‘High PHDI’ group also consumed significantly more vitamin C (P < 0·001), likely due to higher intake of fruit and vegetables. Increased vitamin C intake in this group may also help to offset the lower bioavailability of non-haem Fe, suggesting that those with higher PHDI scores may achieve more efficient Fe absorption despite the reduced haem Fe intake from animal products. Nevertheless, individuals with high intakes of non-haem Fe may remain at risk of anaemia due to the substantially larger quantities of non-haem Fe sources required to meet physiological demands during pregnancy.
Conversely, high meat intake is linked to an elevated risk of CVD, type II diabetes and certain cancers, while the production of meat and dairy products contributes substantially to greenhouse gas emissions, water use, land use and biodiversity loss. These findings are encouraging, as they suggest that public health and environmental benefits could be achieved by reducing the consumption of ASF without increasing the risk of anaemia(Reference Frank, Jaacks and Adair7).
Unsurprisingly, only 2·7 % of women in this cohort met the daily recommended intake of dietary folate (≥ 600 µg dietary folate equivalent per d) in early pregnancy. However, those in the ‘High PHDI’ group had significantly higher dietary folate intake compared with those in the ‘Low PHDI’ group. Within the PHD, sources of folate include vegetables, especially dark leafy greens, kale, spinach and broccoli; fruits, such as oranges and avocados; legumes; and whole grains. The higher folate intake observed likely reflects a greater consumption of these plant-based foods, which are naturally rich in dietary folate. Despite this, higher alignment with the PHD principles alone was insufficient to meet the EFSA-recommended dietary folate intake during pregnancy. Therefore, folate supplementation remains essential to ensure AI. All women of child-bearing age following the PHD should be advised to supplement with 400 µg folate for at least 3 months prior to conception and during pregnancy to reduce the risk of neural tube defects and other adverse outcomes, including preterm birth, low bw and developmental delays(Reference Li, Zhang and Peng42,Reference Obeid, Holzgreve and Pietrzik43) .
Individuals in the ‘High PHDI’ group were less likely to meet EFSA iodine requirements, which is expected, given that the key sources of iodine include fish, eggs and milk(Reference Kent, Kehoe and Flynn44). Iodine is essential for thyroid hormone synthesis and plays a critical role in fetal neurodevelopment. Deficiency is associated with impaired fetal thyroid development, reduced neurocognitive function and, in severe cases, cretinism(Reference Skeaff45). Therefore, iodine supplementation, or other practical dietary alternatives such as iodised salt or seaweed, may be recommended alongside adherence to the PHD.
Additionally, both groups were found to have low vitamin D levels. These findings align with nationally representative data from the National Adult Nutrition Survey at a similar time point, which found that dietary intake in Irish adults averages only ∼4·0 μg/d, significantly less than recommended(Reference Cashman, Muldowney and McNulty46). The survey findings also showed that only 17·5 % of adults use vitamin D supplements(Reference Cashman, Muldowney and McNulty46). Vitamin D is mainly sourced from sunlight exposure, with only a low contribution from dietary sources such as eggs, oily fish and fortified food products. As a result of the limited sun exposure in Ireland, the population is dependent on dietary sources of vitamin D, and most of the population fails to meet the RDA. Given that both PHDI groups did not meet the recommended intakes, supplementation with vitamin D, alongside an increase in these foods, may also be necessary to ensure optimal health outcomes.
Surprisingly, no significant differences in vitamin B12 intake were observed between the ‘High PHDI’ and ‘Low PHDI’ scoring groups. This finding may reflect the limited inclusion of ASF in the diets of women in the ‘High PHDI’ group as women can still achieve ‘High PHDI’ scores without fully eliminating animal products from their diets. However, when examined continuously, each one-point increment in the PHDI score was negatively associated with vitamin B12 intake, suggesting that greater alignment with the PHD may be accompanied by a gradual reduction in B12 consumption. As plant-based diets are often associated with inadequate B12 status, which can contribute to adverse maternal and neonatal outcomes such as developmental anomalies, pre-eclampsia and low bw, it is essential that women following the PHD ensure sufficient B12 intake through fortified foods or supplementation if necessary, aiming for at least 4·5 μg/d(20,47) .
Additionally, vitamin B7 intake was higher in the ‘High PHDI’ scoring group. Research indicates that mild biotin deficiency can occur naturally in a significant number of pregnancies and may be linked to an increased risk of birth defects(Reference Mock48). This suggests that a higher PHDI score may offer an additional benefit by promoting greater biotin intake from plant-based sources, such as nuts, seeds, legumes and potatoes, which may play a role in promoting maternal and fetal health(Reference Cervantes and Daley49).
It is important to acknowledge that some plant-based foods contain anti-nutritional factors, including phytates and tannins, which can reduce the bioavailability of essential nutrients like Fe, Zn and protein(Reference Shaw, Zello and Rodgers50). Therefore, plant-based diets must be carefully planned to ensure sufficient intake and absorption of energy and essential nutrients(Reference Shaw, Zello and Rodgers50). Strategies such as combining non-haem Fe sources with vitamin C or incorporating minimal haem Fe sources may help enhance Fe absorption(Reference Shaw, Zello and Rodgers50). As mentioned above, a higher PHDI is also associated with increased vitamin C intake, which could mitigate some of these bioavailability concerns. Although this study did not quantify the effect of vitamin C on Fe absorption, previous research indicates that approximately 50 mg of vitamin C per meal optimises absorption, and even 50 ml of orange juice (∼25 mg vitamin C) can increase Fe absorption by about 2·5 fold(Reference Barrett, Whittaker and Williams51,Reference Hallberg, Brune and Rossander52) . Evidence from dietary interventions in pregnant women also shows that combining plant-based Fe sources with vitamin C-rich fruits can improve Fe status(Reference Wijaya-Erhardt, Muslimatun and Erhardt53).
Even among those whose diets aligned more closely with the PHD, many did not meet EFSA guidelines, emphasising that dietary intake alone may not always be sufficient to achieve recommended nutrient levels. While women can be encouraged to adopt a more sustainable diet during pregnancy, our research does not negate the need for targeted supplementation before and throughout pregnancy. Supplementation is particularly important during the prenatal and antenatal stages to ensure AI of essential nutrients such as Fe, vitamin D and folate.
Strengths and limitations
A major strength of this study is the use of 3-d food diaries in both cohorts, which offer a reliable method for evaluating average dietary intake(Reference Lynch, Killeen and O’Brien54). This approach allows for more accurate quantification of portion sizes compared with FFQ and helps minimise recall bias due to real-time data collection(Reference Lynch, Killeen and O’Brien54). The relatively large sample size enhances statistical power and enables more robust analysis of dietary patterns. This study examined the PHD in a healthy pregnant population, which increases the generalisability of the findings. Inclusion of all EFSA DRV (PRI, AI, AR, RI) gives a nuanced view of adherence levels in this cohort.
However, several limitations should be acknowledged. The analysis combined data from two separate cohorts (ROLO and MicrobeMom), which may introduce variability due to differences in study design, recruitment periods and inclusion/exclusion criteria. This analysis may have introduced a degree of selection bias as the criteria for the original RCT meant all women included were healthy pregnant women (MicrobeMom) and/or healthy pregnant women with a previous macrosomic baby (ROLO).
Although 3-d food diaries offer a robust method for recording average dietary intake, they may be insufficient alone for assessing long-term compliance with specific eating habits and dietary guidelines; therefore, DRV comparisons should be interpreted with caution. Only one dietary assessment was performed in early pregnancy, and this analysis could have been strengthened by regular repeated assessments to enable usual intake modelling in the first trimester. Dietary intake was analysed at a single time point in pregnancy, while longitudinal studies tracking dietary patterns from preconception through postpartum periods would provide more detailed insights into the sustained impact of the PHD on nutritional status. Despite all participants included in this study completing a 3-d food diary, nutrient values were unavailable for folate and unsaturated fat intake for a small number of participants, resulting in missing data for fourteen participants. Additionally, under-reporting could only be calculated for 674 participants as complete data for this estimation was missing for four participants.
Furthermore, the demographic homogeneity of the cohort may limit the generalisability of the findings for more diverse populations. Another limitation is the inconsistency in evaluation and scoring methodologies for the PHD across studies. While the EAT–Lancet reference diet provides intake recommendations for each food group, differences in scoring approaches make it challenging to compare findings across the literature. Additionally, this study did not analyse full adherence to the PHDI but rather compared nutrient intakes between two groups dichotomised by the median PHDI score in relation to pregnancy dietary guidelines. The ‘High PHDI’ group does not represent strict adherence to the PHD but rather a moderate alignment with its principles. Participants were not provided with any guidance to follow the PHD. Future intervention studies could help clarify whether intentional adherence to the PHD meets dietary requirements more effectively.
Clinical implications
This study highlights the potential benefits of the PHD in pregnancy, showing that women with higher PHDI scores had greater intakes of key nutrients, including Fe, folate and Ca. These findings suggest that the PHD can support maternal and fetal health while reducing the environmental impact of diets high in ASF. However, supplementation with folate and iodine remains necessary, as some women following the PHD may not meet EFSA guidelines for these nutrients.
Future research
Future research should aim to explore the long-term health outcomes associated with adherence to the PHD during pregnancy, particularly concerning maternal and neonatal health. Further studies should investigate the associations between adherence to the PHD and specific pregnancy outcomes, including the incidence of hypertensive disorders of pregnancy, anaemia, gestational diabetes mellitus, preterm birth and fetal growth restriction. Understanding these relationships can help determine whether higher PHD adherence confers protective effects against adverse pregnancy outcomes. Future research could also explore alternative methods such as usual intake modelling, which may provide more detailed insights into habitual intake rather than relying on self-reported intakes at a single time point.
Given the evolving food environment and cultural dietary shifts, future studies should include more diverse populations across various socio-economic and ethnic backgrounds to enhance the generalisability of the PHD. When investigating protein intake, future research should investigate specific amino acids associated with optimal maternal health and how their intake changes alongside adherence to the PHD. Moreover, evaluating the effectiveness of supplementation in conjunction with the PHD during pregnancy is essential to ensure nutritional adequacy. Research should also consider the environmental and economic feasibility of adopting the PHD during pregnancy, identifying potential barriers and facilitators to adherence in real-world settings. Future research should also explore how national dietary guidelines can be refined to better align with both nutritional needs and environmental sustainability, ensuring that they are tailored to the unique food systems and cultural contexts of individual countries.
Conclusion
In conclusion, this study highlights the potential benefits of following the PHD in pregnancy, such as its positive effects on folate, Fe and Ca intakes. Women with higher PHDI scores demonstrated better adherence to EFSA’s recommended intake levels, suggesting that the PHD, alongside appropriate supplementation, can support maternal and fetal health while reducing environmental impacts associated with high ASF consumption.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114526106874.
Acknowledgements
The authors would like to thank all the women who took part in the ROLO and MicrobeMom studies. They also thank the staff of the National Maternity Hospital for facilitating our research.
This research was supported by the Health Research Board Ireland, the Health Research Centre for Health and Diet Research, the National Maternity Hospital Medical Fund and the European Union’s Seventh Framework Programme (FP7/2007–2013), project EarlyNutrition under grant agreement no. 289346, Science Foundation Ireland (12/RC/2273, 16/SP/3827 and 22-FFA-10302), which included a research grant from PrecisionBiotics Group Ltd.
A.D. designed the study, analysed the data and drafted the initial manuscript. S.C. advised on statistical analysis and contributed to the manuscript preparation. G.A.C. and E.O.B. were involved in study design and contributed to the manuscript preparation. A.P.D. was responsible for the data management and curation. F.M.McA. designed the study and had primary responsibility for the final content. All authors approved the final version of the manuscript and agreed to be accountable for all aspects of the work.
There are no conflicts of interest.
The data used and/or analysed in these studies are available from the corresponding author on reasonable request.
Both trials were carried out in accordance with the Declaration of Helsinki of 1975 as revised in 1983. Institutional ethical approval was obtained from the Ethics Committee of the National Maternity Hospital in December 2006 for the ROLO study (GEN/279/12) and in February 2016 for MicrobeMom (EC 35.2015). Informed written consent was obtained from all participants.