Introduction
Strongyloidiasis is the infection caused by the intestinal parasitic worm Strongyloides stercoralis. This parasite is widely distributed worldwide, with a global prevalence of infection estimated at about 600 million cases in 2017 (Buonfrate et al., Reference Buonfrate, Bisanzio, Giorli, Odermatt, Fürst, Greenaway, French, Reithinger, Gobbi, Montresor and Bisoffi2020). The highest burden is in southeast Asia, Africa and the Western Pacific region (Olsen et al., Reference Olsen, van Lieshout, Marti, Polderman, Polman, Steinmann, Stothard, Thybo, Verweij and Magnussen2009; Buonfrate et al., Reference Buonfrate, Bisanzio, Giorli, Odermatt, Fürst, Greenaway, French, Reithinger, Gobbi, Montresor and Bisoffi2020). Most infected individuals are asymptomatic, allowing the infection to remain undiagnosed and untreated for years. The highest risk of infection in the world is in rural areas where lack of access to sanitation and barefoot walking favour soil contamination by infective larvae (Buonfrate et al., Reference Buonfrate, Bradbury, Watts and Bisoffi2023). Strongyloidiasis can be found in non-endemic areas due to increases in travel and migration from endemic to non-endemic countries (Bethony et al., Reference Bethony, Brooker, Albonico, Geiger, Loukas, Diemert and Hotez2006; Montes et al., Reference Montes, Sawhney and Barros2010).
Three main clinical presentations of strongyloidiasis are acute infection, chronic infection, hyperinfection and dissemination. Chronic infection is characterized by low level of worm reproduction, and most infected people are asymptomatic. When symptoms are present, they are non-specific, allowing the infection to remain undiagnosed, thus untreated for years (Bisoffi et al., Reference Bisoffi, Buonfrate, Montresor, Requena-Méndez, Muñoz, Krolewiecki, Gotuzzo, Mena, Chiodini, Anselmi, Moreira and Albonico2013). Of note, in immunosuppressed people strongyloidiasis can cause a serious and most often fatal illness: the hyperinfection syndrome. It describes an accelerated autoinfection, and the diagnosis implies the presence of signs and symptoms attributable to increased larval migration to organs beyond the range of the pulmonary auto-infective cycle (dissemination). The invasion of helminths into the mucosa is often associated with Gram-negative bacterial sepsis (Keiser and Nutman, Reference Keiser and Nutman2004; Olsen et al., Reference Olsen, van Lieshout, Marti, Polderman, Polman, Steinmann, Stothard, Thybo, Verweij and Magnussen2009). Delayed presentation, misdiagnosis and reduced access to clinical care impact mortality (Keiser and Nutman, Reference Keiser and Nutman2004).
Conventional diagnosis is based on stool-based parasitological methods, such as direct-smear fecal microscopy, which has limited usefulness because of its extremely low sensitivity, Baermann sedimentation and agar plate culture. Several specimens should be collected on different days to improve the detection rate. The sensitivity of microscopic-based techniques might not be sufficient, especially in chronic infections where larval output is very low. However, the most sensitive techniques, the Baermann and agar plate methods, are too labour-intensive for use in an extensive population (Buonfrate et al., Reference Buonfrate, Bradbury, Watts and Bisoffi2023). Serology assays are also available but sensitivity is low in very early infections and hyperinfection in immunosupressed patients. These include commercial enzyme-linked immunosorbent assays and other in-house techniques (Zaha et al., Reference Zaha, Hirata, Kinjo and Saito2000; van Doorn et al., Reference van Doorn, Koelewijn, Hofwegen, Gilis, Wetsteyn, Wismans, Sarfati, Vervoort and van Gool2007; Bisoffi et al., Reference Bisoffi, Buonfrate, Sequi, Mejia, Cimino, Krolewiecki, Albonico, Gobbo, Bonafini, Angheben, Requena-Mendez, Muñoz and Nutman2014). Strongyloides DNA detection by real-time polymerase chain reaction is highly specific, with improved sensitivity compared to direct microscopy, but similar to that of Baermann and agar plate culture. As with other techniques, there is a lack of standardization of methods, including sample collection, preservation and DNA extraction methods (ten Hove et al., Reference ten Hove, van Esbroeck, Vervoort, van den Ende, van Lieshout and Verweij2009; Gordon et al., Reference Gordon, Utzinger, Muhi, Becker, Keiser, Khieu and Gray2024).
The Centers for Disease Control and Prevention and the World Health Organization (WHO) recommend ivermectin as the drug of choice for the treatment of strongyloidiasis, based on several clinical trials and meta-analysis (Henriquez-Camacho et al., Reference Henriquez-Camacho, Gotuzzo, Echevarria, White, Terashima, Samalvides, Pérez-Molina and Plana2016). Albendazole has lower efficacy than ivermectin, hence is considered a second-line drug (The Medical Letter, 2013).
The control of strongyloidiasis as a public health problem has been recently recommended by the WHO. However, the treatment is not universally available, although ivermectin is included in the WHO essential medicines list.
Massive use of ivermectin has been already carried out for decades, in the context of the elimination programmes for onchocerciasis and lymphatic filariasis. This has raised concerns about the possible emergence of drug resistance, that has been documented so far in veterinary but not in human medicine (Cobb and Boeckh, Reference Cobb and Boeckh2009). New drugs are needed to face this possible, but uncertain threat.
Moxidectin is a macrocyclic lactone that belongs to the same family of ivermectin and has been widely used by veterinarians (Cobb and Boeckh, Reference Cobb and Boeckh2009). In addition, moxidectin was used in humans in a randomized controlled trial (RCT) (NCT00790998) involving 1472 patients with onchocerciasis. The trial was conducted at 4 sites in West Africa (Opoku et al., Reference Opoku, Bakajika, Kanza, Howard, Mambandu, Nyathirombo, Nigo, Kasonia, Masembe, Mumbere, Kataliko, Larbelee, Kpawor, Bolay, Bolay, Asare, Attah, Olipoh, Vaillant, Halleux and Kuesel2018). Although moxidectin belongs to the same family of ivermectin, it has a different resistance pattern (Hofmann et al., Reference Hofmann, Smit, Sayasone, Pfister and Keiser2022). This paves the way to also use it as an alternative treatment for strongyloidiasis.
The aim of this systematic review was to assess the efficacy of ivermectin vs moxidectin for treating S. stercoralis infection.
Materials and methods
Selection criteria
This systematic review was registered in the International prospective register of systematic reviews (PROSPERO) with the reference no. CRD42024494934, and it was conducted following the PRISMA 2020 statement criteria (Page et al., Reference Page, McKenzie, Bossuyt, Boutron, Hoffmann, Mulrow, Shamseer, Tetzlaff, Akl, Brennan, Chou, Glanville, Grimshaw, Hróbjartsson, Lalu, Li, Loder, Mayo-Wilson, McDonald, McGuinness, Stewart, Thomas, Tricco, Welch, Whiting and Moher2021). RCTs that evaluated the outcomes of moxidectin and ivermectin in S. stercoralis infection were included. The inclusion criteria were: participants aged 12 years and above; immunocompetent individuals; S. stercoralis infection confirmed by parasitological examination (at least 1 positive specimen). The following outcomes were considered: elimination of infection or parasitological cure, defined as any negative parasitological exam during follow-up period; mortality; serious adverse events, such as inpatient hospitalization or prolongation of existing hospitalization, persistent or significant disability/incapacity or life-threatening states. The search protocol followed the PRISMA guideline.
Search strategy and study selection
We identified all relevant trials, regardless of their publication status (published, unpublished), using the search terms detailed in Appendix 1 in the following databases: Ovid MEDLINE (from January 1966 to February 2024), Embase (from January 1980 to February 2024) and Web of Science. The reference lists of the included studies were identified by the mentioned methods. Two reviewers (CH-C, PR) used Rayyan software to check the titles and abstracts of the literature independently, searching for potentially relevant articles (Ouzzani et al., Reference Ouzzani, Hammady, Fedorowicz and Elmagarmid2016). We retrieved the full reports of potentially relevant trials and applied the inclusion criteria using an eligibility form. The third author (EG) resolved any disagreements. The eligible trials were scrutinized to exclude possible duplicates. In case of several publications reporting the same trial, the most recent was chosen. There were no restrictions based on language, sample size, age, gender, ethnicity or duration of follow-up.
Data extraction and assessment of risk of bias
Two reviewers (DB and JAP-M) independently extracted data regarding the inclusion criteria, outcome data and adverse events. Any disagreement was resolved after evaluation by a third reviewer (EG). The risk of bias was assessed independently using a risk of bias Cochrane Collaboration form (Higgins et al., Reference Higgins, Altman, Gøtzsche, Jüni, Moher, Oxman, Savovic, Schulz, Weeks and Sterne2011), while the third reviewer resolved any disagreements. Generation of allocation sequence and allocation concealment was categorized as adequate, unclear or inadequate accordingly, using the criteria outlined in the Cochrane Handbook (Higgins et al., Reference Higgins, Savović, Page, Elbers, Sterne, Higgins, Thomas, Chandler, Cumpston, Li, Page and Welchn.d.). We considered the following domains: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective outcome reporting (reporting bias) and other sources of bias. We classified each domain as being at ‘low’, ‘high’ or ‘unclear’ risk of bias. A ‘Risk of bias’ graph was included in the analysis.
Outcome assessment
The data extracted were dichotomous variables. We recorded the absolute number of events and participants in each group for all outcomes. Pooled odds ratios (ORs) with 95% confidence intervals (CIs) were reported. We calculated the proportion of loss to follow-up in each group. We reported data about methodological quality of trials, characteristics of participants, characteristics of interventions, characteristics of outcome measures, date of trial, trial authors, location of trial, sponsor of trial (specified, known or unknown), design (described as randomized or not), interventions (treatment, days, doses), and outcomes, mortality and serious adverse events. We calculated the proportion of loss to follow-up in each group.
Statistical analysis
We calculated ORs with 95% CIs for dichotomous data. We planned to perform a pooled analysis by using a fixed or random effects model based on the level of heterogeneity. Heterogeneity was assessed using forest plots of study results to visually check for overlaps in CIs; Chi2 test for statistical heterogeneity (we considered trial results as heterogeneous with P < 0.10) and results of the I 2 statistic. We judged the importance of the observed value of I 2 according to the magnitude and direction of effects and the strength of evidence of heterogeneity (from 0 to 40% heterogeneity – might not consider important; from 30 to 60% heterogeneity – might be moderate; from 50 to 90% heterogeneity – might be substantial and from 75 to 100% heterogeneity – might be considerable) (Deeks et al., Reference Deeks, Higgins, Altman, Higgins, Thomas, Chandler, Cumpston, Li, Page and Welchn.d.). We used the Review Manager 5 package, provided by Cochrane for data synthesis and analysis (Collaboration NCCTC., n.d.).
For the analysis of adverse events, the number of participants who experienced the adverse events were included. Data from trials that only reported the number of adverse events were not included, as it was possible that the same individual reported more than 1 adverse event.
We assessed the certainty of the evidence using the GRADE approach (Guyatt et al., Reference Guyatt, Oxman, Akl, Kunz, Vist, Brozek, Norris, Falck-Ytter, Glasziou, DeBeer, Jaeschke, Rind, Meerpohl, Dahm and Schünemann2011). Our confidence in the estimate of effect was evaluated in terms of study limitations, inconsistency of effect or unexplained heterogeneity, imprecision of results, indirectness and publication bias. We planned to evaluate the risk of publication bias graphically if more than 10 studies were included for the considered outcome. We presented a ‘Summary of findings’ table to summarize the key results of our review using the GRADEpro Guideline Development Tool.
Results
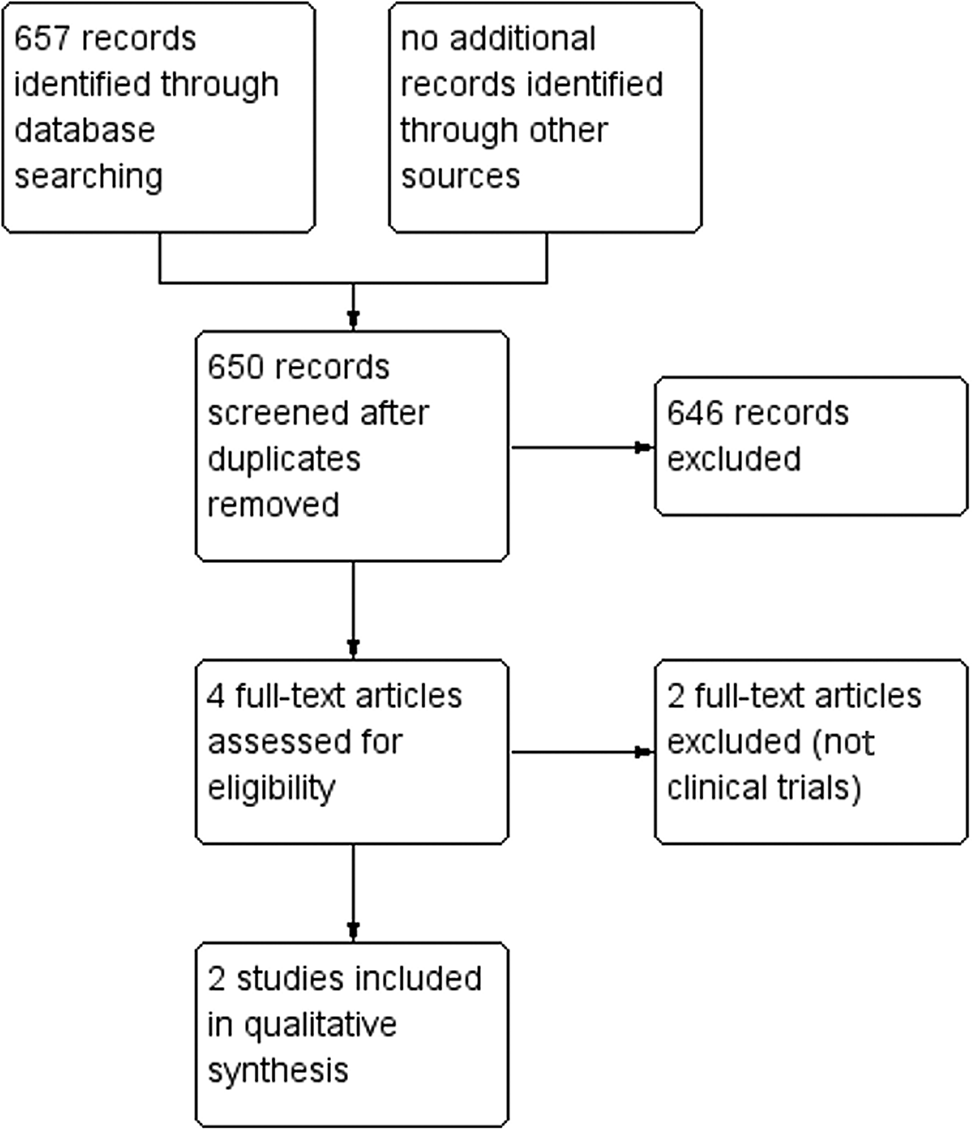
The electronic search identified 296 references (PubMed), 247 references (Web of Science) and 114 references (Embase). A total of 650 records were identified after removing duplicates, 646 references were excluded based on title and abstract screening and 4 were included after full-text screening. Finally, 2 trials (Barda et al., Reference Barda, Sayasone, Phongluxa, Xayavong, Keoduangsy, Odermatt, Puchkov, Huwyler, Hattendorf and Keiser2017; Sprecher et al., Reference Sprecher, Hofmann, Savathdy, Xayavong, Norkhankhame, Huy, Khieu, Sayasone, Hattendorf and Keiser2024) met the inclusion criteria (Fig. 1). We excluded 2 trials because they were not RCTs (Hürlimann et al., Reference Hürlimann, Hofmann and Keiser2023; Gandasegui et al., Reference Gandasegui, Muñoz, Fleitas, Mazzi and Bisoffi2024).
Study flow diagram.
Two RCTs, overall including 821 adult participants, were assessed in this review. Both studies were conducted in southeast Asia (Cambodia and Laos). None of the trials included immunocompromised patients. The mean age of the participants ranged from 40 to 45 years old, with a similar distribution of males and females. For all participants, S. stercoralis infection was confirmed by the Baermann method. Pregnant and breastfeeding women were excluded, as well as women planning to become pregnant within 3 months after treatment. Participants with concomitant chronic diseases were also excluded in the Barda et al.'s (Reference Barda, Sayasone, Phongluxa, Xayavong, Keoduangsy, Odermatt, Puchkov, Huwyler, Hattendorf and Keiser2017) trial (see Table 1).
Characteristics of the included trials

s.d., standard deviation.
Interventions
In both trials, the dose of ivermectin was 200 μg kg−1 single dose (Stromectol® from Merck Sharp & Dohme or IverP® from Elea, Argentina), and the dose of moxidectin was fixed at 8 mg . In Sprecher et al. (Reference Sprecher, Hofmann, Savathdy, Xayavong, Norkhankhame, Huy, Khieu, Sayasone, Hattendorf and Keiser2024), moxidectin formulation was 2 mg tablets (Development for Global Health; Melbourne, VIC, Australia), whereas in Barda et al. (Reference Barda, Sayasone, Phongluxa, Xayavong, Keoduangsy, Odermatt, Puchkov, Huwyler, Hattendorf and Keiser2017) an oral suspension was used (Cydectin® 0.1%; Zoetis, Switzerland). The analysis was per-protocol analysis. The follow-up period ranged from 14 to 25 days. The assessment of the outcome measures was based on the parasitological cure rate (see Table 2).
Recruitment and following period of the included trials

sd, single dose.
Summary of findings
The evidence was moderate for parasitological cure rate (see Appendix 2). Certainty was downgraded by 1 level because of imprecision (wide CIs). Regarding risk of bias, both RCTs reported adequate methods of allocation concealment and random sequence generation. Only 1 trial was double blinded for the participants and outcome assessment. Nevertheless, the lack of blinding may not have affected the results of the other study, because the primary outcome (parasitological cure and death) was objectively measured (see Fig. 2). We cannot evaluate the risk of publication bias due to the low number of studies.
Risk of bias Summary.

Effect of interventions
The outcome was communicated in all reports. Both trials measured the parasitological cure at 14–21 days. Moxidectin was not inferior to ivermectin: OR 0.67, 95% CI 0.36–1.25 (P = 0.21), I 2 = 0%, 821 participants (Fig. 3). No deaths were reported in any trial.
Comparison between moxidectin vs ivermectin for Strongyloides stercoralis infection. Outcome: parasitological cure rate.

Adverse events
One trial (Sprecher et al., Reference Sprecher, Hofmann, Savathdy, Xayavong, Norkhankhame, Huy, Khieu, Sayasone, Hattendorf and Keiser2024) reported mild adverse events that were unrelated to the study intervention. In total, 153/726 (21%) participants had an adverse event, which were equally distributed among the 2 treatment groups. The most reported symptoms were abdominal pain and headache. None of the patients stopped therapy or discontinued normal daily activities.
Discussion
This systematic review aimed to summarize all the evidence from RCTs relating to the effectiveness of moxidectin compared to ivermectin in strongyloidiasis to provide current best evidence to base decisions for practice and further research.
The results suggest that there is evidence of moderate quality that moxidectin is non-inferior to, and as safe as ivermectin. We found no difference in the parasitological cure, although this result is based on only 2 trials with few patients.
Abdominal pain and headache emerged as the predominant adverse events in the trials reviewed. Given that ivermectin and moxidectin are members of the same pharmacological family, it is reasonable to hypothesize that they may exhibit similar adverse event profiles, despite potential differences in efficacy. The current review indicates that the adverse events associated with these drugs were generally mild and transient.
Both RCTs included immunocompetent participants over 11 years of age, with S. stercoralis infection detected by the Baermann method. Thus, we have no data comparing the efficacy of ivermectin or moxidectin on other clinical stages (acute strongyloidiasis or hyperinfection syndrome) because of the difficulty in finding patients in these stages or in severe cases of dissemination who agreed to undergo experimental treatment. Most patients have chronic infections. In the literature, there are case reports and case series describing the use of ivermectin for severe cases, but a robust estimate of its efficacy for these cases is not possible due to the lack of RCTs. There are no reports of moxidectin used in severe cases. Also, although evidence about the efficacy of ivermectin for treatment of strongyloidiasis in immunocompromised people is scarce, there are no data on the use of moxidectin in this vulnerable population.
Moreover, there are no data comparing the efficacy of the 2 drugs in children below 12 years of age, specifically because moxidectin is not registered for use in younger children. The ideal dose of ivermectin or moxidectin is unknown for very young or very old people as most of the trials did not include information about effectiveness in relation to age.
The effect of moxidectin or ivermectin in preventing new infections is not assessed. The trials included in this systematic review were not primarily designed to evaluate the effectiveness of moxidectin in preventing long-term new infections of strongyloidiasis and this outcome was not reported more than 25 days of following. Furthermore, moxidectin has a longer half-life in the plasma and a broader distribution based on its lipophilic properties (Hürlimann et al., Reference Hürlimann, Hofmann and Keiser2023). This facilitates a longer-acting formulation, independent of the weight of the patient, and is widely used by veterinarians because of its long plasma half-life of 20–43 days (Prichard et al., Reference Prichard, Ménez and Lespine2012).
For Barda et al. (Reference Barda, Sayasone, Phongluxa, Xayavong, Keoduangsy, Odermatt, Puchkov, Huwyler, Hattendorf and Keiser2017), we have considered that lack of blindness has a low risk of bias because the measurement of the outcome (parasitological cure) was done objectively. For both RCTs, there was insufficient information to assess the attrition bias of the trials included.
Publication bias is a major threat to the validity of systematic reviews. To minimize the risk of publication bias, we conducted a comprehensive search across numerous clinical trial databases. However, we consider it is unlikely to have occurred in this study because negative or non-conclusive RCTs are unlikely to remain unpublished.
We have identified a similar study, a Cochrane systematic review, published in 2011 comparing efficacy and safety of ivermectin and benzimidazoles for S. stercoralis infection (Henriquez-Camacho et al., Reference Henriquez-Camacho, Gotuzzo, Echevarria, White, Terashima, Samalvides, Pérez-Molina and Plana2016). The previous review did not include moxidectin, as this is drug has only recently been tested for this infection.
The main advantage of moxidectin over ivermectin is its weight-independent dosing, being an alternative treatment in patients with extreme low weight (mainly in poor settings) or obesity (in developed countries). This characteristic can be of interest in the context of mass drug administration, as it avoids the need of using scales or height poles. Ivermectin is currently employed for mass treatment in the context of the elimination programmes targeting onchocerciasis and lymphatic filariasis (Heukelbach et al., Reference Heukelbach, Winter, Wilcke, Muehlen, Albrecht, de Oliveira, Kerr-Pontes, Liesenfeld and Feldmeier2004). Indirect evidence from those programmes demonstrated a significant reduction in strongyloidiasis prevalence in endemic communities (Traore et al., Reference Traore, Sarr, Badji, Bissan, Diawara, Doumbia, Goita, Konate, Mounkoro, Seck, Toe, Toure and Remme2012), posing the basis for the use of this drug for public health control of S. stercoralis infection (Stroffolini et al., Reference Stroffolini, Tamarozzi, Fittipaldo, Mazzi, Le, Vaz Nery and Buonfrate2023). Based on the results of clinical trials it is likely moxidectin will be similarly approved in the future for strongyloidiasis.
The main limitation of this study is the low number of RCTs, which did not permit us to carry out a meta-analysis. The overall evidence was deemed of good quality; however the study by Sprecher presented some discrepant data between study sites that would deserve further evaluation, at least on efficacy of moxidectin in different settings and populations.
Data availability statement
Dataset used and/or analysed during the current study is available upon reasonable request to the corresponding author.
Acknowledgements
We acknowledge Universidad Francisco Vittoria Library for bibliographic support.
Author contributions
C. H.-C., B. L. and P. R. carried out the systematic search. D. B. and J. A. P.-M. extracted the data. C. H.-C. and M. N. P. analysed and interpreted the data and critically revised the manuscript for intellectual content. C. H.-C., D. B., M. N. P., E. G. and J. A. P.-M. drafted the manuscript.
Financial support
This research was partly funded by the Italian Ministry of Health (‘Ricerca corrente’ Linea 2) with fund to IRCCS Sacro Cuore Don Calabria hospital.
Competing interests
None.
Appendix 1. Search strategy

Appendix 2. Summary of findings for the main comparison: moxidectin vs ivermectin: cure rate










