Introduction
Corn poppy (Papaver rhoeas L.) is an agronomically important broadleaf weed in cereal crops across Europe, including Ireland (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025; Marshall et al. Reference Marshall, Hull and Moss2010; Torra et al. Reference Torra, Rojano-Delgado, Rey-Caballero, Royo-Esnal, Salas and De Prado2017). This competitive species exhibits extended germination, prolific seed production, and a persistent seedbank, making repeated herbicide use critical for effective management and crop yield protection (Bond et al. Reference Bond, Davies and Turner2007; Marshall et al. Reference Marshall, Hull and Moss2010).
In Ireland, cropping systems depend heavily on herbicides due to the Atlantic-influenced climate (Vijayarajan et al. Reference Vijayaraajan, Fealy, Cook, Onkokesung, Barth, Hennessy and Forristal2022). The main cereal crops are spring barley (Hordeum vulgare L.) (138,200 ha), winter barley (43,300 ha), winter wheat (Triticum aestivum L.) (40,600 ha), and oats (Avena sativa L.) (29,800 ha) (CSO 2024). Broadleaf weed control in winter cereals typically involves a pre-sowing glyphosate application; residual herbicides (e.g., diflufenican combined with pendimethalin, flufenacet, or prosulfocarb), applied pre- or early postemergence; and spring postemergence herbicides, primarily acetolactate synthase (ALS) inhibitors and auxin mimics (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025). Wetter autumns and fewer opportunities for preemergence spraying have increased reliance on spring postemergence herbicide use (Vijayarajan et al. Reference Vijayaraajan, Fealy, Cook, Onkokesung, Barth, Hennessy and Forristal2022). ALS-inhibiting sulfonylureas (SUs) and triazolopyramidines (TPs) are widely used to control broadleaf weeds, including P. rhoeas (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025). The combination of ALS inhibitors and auxin mimics provides a robust herbicide mixture with a broad application window for effective control (Rosario et al. Reference Rosario, Cruz-Hipolito, Smeda and De Prado2011). To date, auxin mimics have mainly been used to protect ALS inhibitor efficacy and, in some cases, to manage or eliminate ALS inhibitor–resistant populations (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025). However, because P. rhoeas is an outcrossing diploid, repeated use of auxin mimics, in combination with ALS inhibitors, especially at reduced rates or following inadequate control with ALS inhibitors, may increase the risk of multiple resistance developing on some farms (Kati et al. Reference Kati, Scarabel, Thiery-Lanfranchi, Kioleoglou, Liberopoulou and Délye2019; Koreki et al. Reference Koreki, Michel, Lebeaux, Trouilh and Délye2023; Palma-Bautista et al. Reference Palma-Bautista, Portugal, Vazquez-Garcia, Osuna, Torra, Lozano-Juste, Gherekhloo and De Prado2022).
Resistance to ALS inhibitors or auxin mimics in P. rhoeas can arise through target-site resistance (TSR) and non–target site resistance (NTSR). Of the nine known ALS gene mutation sites associated with TSR in weeds, four (Pro-197, Trp-574, Ala-205, Ala-122) have been identified in P. rhoeas, with Pro-197 being the most prevalent (Chtourou et al. Reference Chtourou, Osuna, Mora Marín, Hada, Torra and Souissi2024; Délye et al. Reference Délye, Pernin and Scarabel2011; Kaloumenos et al. Reference Kaloumenos, Dordas, Diamantidis and Eleftherohorinos2009; Koreki et al. Reference Koreki, Michel, Lebeaux, Trouilh and Délye2023; Palma-Bautista et al. Reference Palma-Bautista, Portugal, Vazquez-Garcia, Osuna, Torra, Lozano-Juste, Gherekhloo and De Prado2022; Stankiewicz-Kosyl et al. Reference Stankiewicz-Kosyl, Haliniarz, Wrochna, Obrępalska-Stęplowska, Kuc, Łukasz, Wińska-Krysiak, Wrzesińska-Krupa, Puła, Podsiadło, Domaradzki, Piekarczyk, Bednarczyk and Marcinkowska2023). Additionally, NTSR via cytochrome P450–mediated enhanced metabolism has been shown to contribute to ALS inhibitor resistance (Rey-Caballero et al. Reference Rey-Caballero, Menéndez, Osuna, Salas and Torra2017; Scarabel et al. Reference Scarabel, Pernin and Delye2015). Resistance to auxin mimics in P. rhoeas is primarily associated with different forms of NTSR (Palma-Bautista et al. Reference Palma-Bautista, Portugal, Vazquez-Garcia, Osuna, Torra, Lozano-Juste, Gherekhloo and De Prado2022; Rey-Caballero et al. Reference Rey-Caballero, Menendez, Gine-Bordonaba, Salas, Alcantara and Torra2016; Torra et al. Reference Torra, Rojano-Delgado, Rey-Caballero, Royo-Esnal, Salas and De Prado2017). For the widely used auxin-mimic herbicide 2,4-D, resistance mechanisms include P450-mediated metabolism and/or reduced translocation (Palma-Bautista et al. Reference Palma-Bautista, Portugal, Vazquez-Garcia, Osuna, Torra, Lozano-Juste, Gherekhloo and De Prado2022; Rey-Caballero et al. Reference Rey-Caballero, Menendez, Gine-Bordonaba, Salas, Alcantara and Torra2016; Torra et al. Reference Torra, Rojano-Delgado, Menéndez, Salas and de Prado2021). Crucially, both TSR and NTSR mechanisms conferring resistance to ALS inhibitor and auxin-mimic herbicides can coexist within a single field population of P. rhoeas, complicating its control, and multiple resistance has already been reported in several countries, including Spain, France, Greece, Italy, and Tunisia (Chtourou et al. Reference Chtourou, Osuna, Mora Marín, Hada, Torra and Souissi2024; El-Mastouri et al. Reference El-Mastouri, Košnarová, Hamouzová, Alimi and Soukup2025; Kati et al. Reference Kati, Scarabel, Thiery-Lanfranchi, Kioleoglou, Liberopoulou and Délye2019; Koreki et al. Reference Koreki, Michel, Lebeaux, Trouilh and Délye2023; Palma-Bautista et al. Reference Palma-Bautista, Portugal, Vazquez-Garcia, Osuna, Torra, Lozano-Juste, Gherekhloo and De Prado2022; Scarabel et al. Reference Scarabel, Pernin and Delye2015; Stankiewicz-Kosyl et al. Reference Stankiewicz-Kosyl, Synowiec, Haliniarz, Wenda-Piesik, Domaradzki, Parylak, Wrochna, Pytlarz, Gala-Czekaj, Marczewska-Kolasa, Marcinkowska and Praczyk2020).
Herbicide-resistance monitoring has only recently commenced in Ireland (Alwarnaidu Vijayarajan et al. Reference Alwarnaidu Vijayarajan, Forristal, Cook, Schilder, Staples, Hennessy and Barth2021). The program has confirmed ALS inhibitor resistance in suspected populations of grass weeds, including blackgrass (Alopecurus myosuroides Huds.), Italian ryegrass [Lolium perenne L. ssp. multiflorum (Lam.) Husnot], annual bluegrass (Poa annua L.), and rough-stalk bluegrass (Poa trivialis L.) (Alwarnaidu Vijayarajan et al. Reference Alwarnaidu Vijayarajan, Forristal, Cook, Schilder, Staples, Hennessy and Barth2021, Reference Alwarnaidu Vijayarajan, Morgan, Onkokesung, Cook, Hodkinson, Barth, Hennessy and Forristal2023; Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025) and in broadleaf weeds such as P. rhoeas, common chickweed [Stellaria media (L.) Vill.], corn marigold [Glebionis segetum (L.) Fourr.], and common field-speedwell (Veronica persica Poir.) (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025). In 2022, poor control with ALS inhibitor and auxin-mimic herbicides was reported in two P. rhoeas populations (PAPRH-R1 and PAPRH-R2), raising concerns about potential multiple resistance. Both fields had histories of ALS inhibitor use, with occasional application of auxin-mimic herbicides in the PAPRH-R1 field and consistent use in combination with auxin mimics (often at reduced rates) in the PAPRH-R2 field. Previously, no cases of broadleaf weed resistance to auxin mimics or to both ALS inhibitors and auxin mimics have been confirmed in Ireland. This study aimed to: (1) confirm and quantify resistance levels to ALS inhibitors and the auxin mimic 2,4-D, (2) identify the TSR and NTSR mechanisms underpinning this resistance, and (3) evaluate the resistance or sensitivity of PAPRH-R1 and PAPRH-R2 to other available postemergence herbicides.
Materials and Methods
Plant Materials
Two suspected herbicide-resistant P. rhoeas populations, PAPRH-R1 and PAPRH-R2, were sampled in August 2022 from winter wheat fields in County Meath (53.50°N, 6.90°W) and County Kildare (53.34°N, 6.61°W), respectively. PAPRH-R1 survived a field application of 15 + 15 g ai ha−1 of the ALS-SU inhibitors thifensulfuron + tribenuron (recommended rate of product Cameo Max SX, FMC Agro, Flintshire, UK). PAPRH-R2 survived a tank mix containing 2.9 + 14.1 g ai ha−1 of the ALS-TP inhibitors florasulam + pyroxsulam (Broadway Star, Corteva Agrisciences, Cambridge, UK) and 105 + 4.7 g ai ha−1 of the auxin mimics fluroxypyr + halauxifen (Whorl, Corteva Agrisciences), both applied at 75% of their respective recommended label rates. A sensitive reference population (S) was obtained from a commercial seed supplier (Fruit Hill Farm, County Cork, Ireland).
Growing Conditions and Herbicide Application
Seeds of the R and S populations were sown in 7 by 7 by 8 cm pots filled with a standard soil mix consisting of 70% loam, 20% horticultural grit, and 10% peat medium, supplemented with 2 g L−1 of Osmocote Mini™ (National Agrochemical Distributors, County Dublin, Ireland). Each replicate consisted of a plastic tray (23 by 17.6 by 5.5 cm) holding six individual pots, with one plant per pot at the time of herbicide application. Herbicides were applied using a Generation III Research Track Sprayer (DeVries Manufacturing, Hollandale, MN, USA) fit with a TeeJet® 8002-EVS flat-fan nozzle (TeeJet® Technologies, IL, USA), delivering a spray volume of 200 L ha−1 at a speed of 1.2 ms−1 and spraying pressure of 250 kPa. Plants were grown in a glasshouse with an 18 /12 C (day/night) temperature regime and a 16-h photoperiod supplemented with artificial lighting to maintain a minimum light intensity of 250 μmol m−2 s−1.
Dose Response to ALS Inhibitors and the Auxin Mimic 2,4-D
At the 4 to 6 true-leaf stage (BBCH 14-16), the R and S populations were treated with a range of ALS inhibitors and 2,4-D doses (Table 1). For the R populations, dose rates ranged from 0.25 to 8 times the recommended label rate, while for the S population, doses ranged from 0.0625 to 2 times the recommended label rate. Each treatment included three replicates of six plants and was repeated at least once. At 28 d after treatment (DAT), plants were visually scored as alive (new active growth) or dead (no new growth, severe stunting, curling, epinasty, chlorosis or necrosis) compared with untreated controls. Aboveground shoots from each replicate were harvested, and both fresh and dry weights were recorded (dried at 70 C for 72 h). Dry weight measurements (data not shown) showed trends similar to those of fresh weight; therefore, fresh weight data were used to distinguish R and S populations. Shoot biomass for each replicate was expressed as a percentage of the mean fresh weight of untreated controls. The percentage of plant survival and shoot fresh weights were assessed at 30 DAT.
Table 1. Acetolactate synthase (ALS) and 2,4-D herbicides used for dose–response assays in resistant (R) and sensitive (S) populations of Papaver rhoeas.

a The recommended label rate for each herbicide is highlighted in bold.
b Cameo Max SX, FMC Agro, Flintshire, UK.
c Pacifica Plus, Bayer CropScience, Cambridge, UK. Treatments were applied with 1% v/v Biopower (alkylethersulfate sodium salt) adjuvant (Bayer CropScience, Cambridge, UK).
d Ally SX, FMC Agro, Flintshire, UK.
e Broadway Star, Corteva Agrisciences, Cambridge, UK. Treatments were applied with 1% v/v Kantor (alkoxylated triglycerides) adjuvant (Interagro, Hertfordshire, UK).
f D50, Nufarm UK, West Yorkshire, UK.
DNA Extraction and ALS Gene Sequencing
A total of 16 plants per population were grown to the 4 to 6 true-leaf stage and treated with the recommended label rate of ALS-SU inhibitors thifensulfuron + tribenuron (15 + 15 g ai ha−1). At 30 DAT, fresh leaf tissue was sampled from surviving plants of PAPRH-R1 and PAPRH-R2 populations, as well as from untreated control plants of the S population. Samples were freeze-dried for 72 h and ground to a fine powder using a Retsch Mixer Mill MM 400 (Retsch GmbH, Haan, Germany). Genomic DNA was extracted from 48 samples using the Macherey Nagel NucleoMag Plant kit (Lab Unlimited–Carl Stuart Group, County Dublin, Ireland) and the ThermoScientificTM Pharma KingfisherTM flex 96 Deep-well head magnetic particle processor (Lab Unlimited–Carl Stuart Group). DNA samples were stored at −20 C until further use.
Allele-specific PCRs were conducted using primer sets targeting the two most common mutation sites in the ALS gene: Pro-197 and Trp-574 (Murphy and Tranel Reference Murphy and Tranel2019), as listed in Table 2 (Délye et al. Reference Délye, Pernin and Scarabel2011). Each 25-μl PCR reaction contained 12.5 µl of Classic++TM Taq DNA Polymerase Master Mix (Tonbo Biosciences, San Diego, CA, USA), 0.2 or 0.4 µM of each primer (depending on the target site), 2 µl of genomic DNA, and PCR-grade water to a final volume of 25 μl. Thermal cycling included: initial denaturation at 95 C for 3 min, 37 cycles of 95 C for 5 s, annealing at the primer-specific temperature (65 C for Pro-197, 60 C for Trp-574) for 10 s, and extension at 72 C for 30 s. Amplification products were visualized on a 1.2% agarose gel and purified using ExoSAP-ITTM PCR Product Cleanup Reagent (Life Technologies Europe, County Dublin, Ireland). Purified products were standardized to 10 ng µl−1 and sent to LGC Genomics GmbH (Ostedstr.25/TGS, Berlin, Germany) for Sanger sequencing. Sequences were analyzed using Geneious Prime software (v. 2025.2.2) (Dotmatics, Hertfordshire, UK) to identify mutations in R plants relative to S plants.
Table 2. Primer sequences used to amplify the ALS gene to detect target-site resistance in Papaver rhoeas.

Effect of P450 Inhibitors on ALS Inhibitor and 2,4-D Resistance
A preliminary test evaluated the potential synergistic effects of two common P450 inhibitors, malathion and piperonyl butoxide (PBO), with ALS inhibitors and 2,4-D. Based on the results, PBO was selected for subsequent trials.
At the 4 to 6 true-leaf stage, R and S populations were pretreated with 2,222.2 g ha−1 PBO plus 0.1% v/v Tween-80 (Merck Life Science, County Wicklow, Ireland) and left at room temperature for approximately 3 h. Subsequently, a full dose response was conducted using florasulam + pyroxsulam (ALS-TP) and 2,4-D, with application rates ranging from 0.25 to 8 times the recommended label rate for R populations and 0.0625 to 2 times for the S population (Table 1). As R and S populations responded similarly to ALS-SUs, additional PBO assays were conducted with thifensulfuron + tribenuron applied at the recommended rate and 1.5 times that rate. The experiment included four treatments: untreated control, herbicide only, PBO only, and PBO + herbicide. Each treatment consisted of three replicates of six plants and was repeated once. Plant survival and shoot weights were assessed at 30 DAT, with fresh weight data used for analyses.
Absorption and Translocation of [14C]2,4-D
The absorption and translocation methodology for [14C]2,4-D followed Rey-Caballero et al. (Reference Rey-Caballero, Menendez, Gine-Bordonaba, Salas, Alcantara and Torra2016). Radioactive solutions of 14C-labeled 2,4-D dimethylamine salt (specific activity of 5.995 Mbq mg−1; Institute of Isotopes, Budapest, Hungary) were mixed with commercial 2,4-D to a final concentration of 5 g L−1, equivalent to the recommended label rate of 1,000 g ai ha−1, assuming a spray volume of 200 L ha−1. To mimic the herbicide concentration normally applied at the recommended label rate, six plants from the R and S populations, grown to the 6 true-leaf stage, were each treated with four droplets of 0.5 µl (2 µl per plant), delivering a total of 0.92 kBq of radioactivity per plant. Plants were harvested at 0, 4, 24, 48, and 72 h after treatment. Unabsorbed herbicide was removed by rinsing the treated leaves with 2 ml of acetone/water (1:1 v/v). The rinse solution was mixed with 15 ml of scintillation fluid (Ultima GoldTM, Revvity Health Sciences, Gronigen, The Netherlands) and analyzed by liquid scintillation spectrometry (LSS) (Beckman LS 6000 TA scintillation counter, Beckman Instruments, CA, USA) for 10 min per sample. The plants were then separated into treated leaf, shoot, and root sections, which were dried for 48 h at 70 C. Dried samples were combusted in a sample oxidizer (OX 500, R.J. Harvey Instrument, New York, NY, USA) and the 14C radioactivity was trapped using Oxysolve C-400 (Zinisser Analytic, Eschborn, Germany) and quantified using LSS. Percentage foliar absorption was calculated as:
and percentage translocation was calculated as:
To qualitatively assess translocation, three treated plants per population were removed from their pots at 72 h after treatment. Roots were rinsed, and whole plants were dried and pressed against a 25 cm by 12.5 cm phosphor storage film for 6 h and scanned using a storage phosphor imager (Cyclone, Perkin-Elmer, Packard Bioscience BV, MA, USA) to visualize radiolabel distribution.
Sensitivity Screenings to Other Postemergence Herbicides
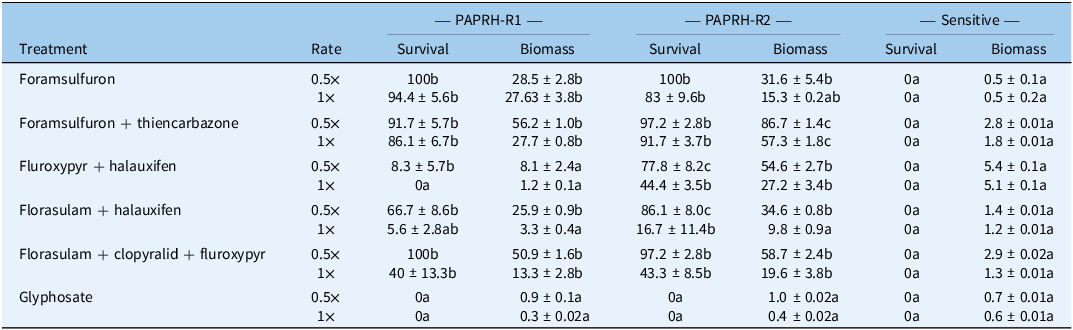
At the 4 to 6 true-leaf stage, the R and S populations were treated with half and full recommended label rates of six herbicides. Two ALS inhibitors were tested: foramsulfuron (Cubix, Bayer CropScience, Barcelona, Spain; not registered in Ireland) at 22.5 and 45 g ai ha−1, and foramsulfuron + thiencarbazone (Conviso One, Bayer CropScience, Cambridge, UK; for use only in the Conviso-Smart system) at 25 + 15 and 50 + 30 g ai ha−1. Two ALS inhibitor + auxin-mimic co-formulations were also tested: florasulam + halauxifen (Zypar, Corteva Agrisciences) at 1.9 + 2.4 and 3.8 + 4.8 g ai ha−1, and florasulam + clopyralid + fluroxypyr (Galaxy, Corteva Agrisciences) at 1.3 + 40 + 72 and 2.5 + 80 + 144 g ai ha−1. An auxin-mimic co-formulation, fluroxypyr + halauxifen (Pixarro EC, Corteva Agrisciences) was applied at 100.9 + 3 and 201.8 + 6 g ai ha−1. The EPSPS (5-enolpyruvylshikimate-3-phosphate synthase) inhibitor glyphosate (Roundup Flex, Bayer) was applied at 270 and 540 g ai ha−1. For foramsulfuron, a spray volume of 250 L ha⁻1 was used following label recommendations. Each treatment included six replicates, with six plants per replicate for each population. Plant survival and shoot weights were assessed at 30 DAT, with fresh weight data used for analyses.
Statistical Analysis
All data analyses were conducted using R (v. 3.6.3; R Core Team 2020). Treatment by experiment interactions were assessed using linear mixed-effects models (lmer function, lme4 package) (Bates et al. Reference Bates, Mächler, Bolker and Walker2015). ANOVA revealed no significant treatment by experiment interactions (P > 0.05); therefore, dose–response data (±P450 inhibitor) were pooled across replicates. Dose–response models were fit to plant survival and shoot fresh weight data using the drc package through the lack-of-fit F-tests (P > 0.05) (Ritz et al. Reference Ritz, Baty, Streibig and Gerhard2015). Two-parameter and four-parameter log-logistic models were used to model survival and biomass data, respectively, of ALS inhibitors and 2,4-D. Box-Cox transformations were applied as necessary to address non-normality and variance heterogeneity. Fitted models estimated the ED50 (effective dose causing 50% mortality) and GR50 (effective dose causing 50% growth reduction relative to untreated plants). The resistance index (RI) was calculated as the ratio of the ED50 or GR50 of each resistant population to that of the sensitive population.
Results and Discussion
Resistance Mechanisms of Papaver rhoeas to ALS inhibitors
The S population was effectively controlled by all ALS-SU and TP inhibitors at rates equal to or below 0.25 times the recommended label rate, resulting in 100% mortality and greater than 90% biomass reduction (Figure 1; Supplementary Figure S1). Compared with the S population, the two R populations showed distinct responses to ALS chemistries (Figure 1; Supplementary Figure S2).

Figure 1. Dose–response curves for surival and shoot biomass of sensitive (S) and resistant (R; PAPRH-R1 and PAPRH-R2) Papaver rhoeas populations treated with varying rates (proportion of recommended label) of differing acetolactate synthase (ALS) inhibitors: thifensulfuron + tribenuron (A and B), mesosulfuron + iodosulfuron + amidosulfuron (C and D), metsulfuron (E and F) and florasulam + pyroxsulam (G and H).
Both PAPRH-R1 and PAPRH-R2 exhibited high resistance to ALS-SU inhibitors (Figure 1A–F). Plant survival and growth remained well above 50%, even at eight times the recommended label rates of 15 g ha−1 thifensulfuron + 15 g ha−1 tribenuron (Figure 1A and 1B), 15 g ha−1 mesosulfuron + 5 g ha−1 iodosulfuron + 25 g ha−1 amidosulfuron (Figure 1C and 1D), and 6 g ha−1 metsulfuron (Figure 1E and 1F). Accordingly, ED50 and GR50 values for both R populations exceeded the highest dose tested, resulting in high resistance indices (RI >120) relative to the S population (Table 3). In contrast, both R populations showed low or evolving resistance to ALS-TP inhibitors, with a few surviving plants above the recommended label rate (Figure 1G and 1H). ED50 and GR50 remained below the recommended rate of 3.8 g ha−1 florasulam + 18.8 g ha−1 pyroxsulam, with RI ranging from 4 to 13 (Table 3). Despite curve fitting, model-based pairwise comparisons of ED50 or GR50 values (expressed as ratios or fold differences) among PAPRH-R1, PAPRH-R2, and the S population using the compParm() function in the drc package could not be performed for ALS inhibitors due to the extreme sensitivity of the S population (ED50 and GR50 below the lowest dose tested) and the strong resistance of the R populations.
Table 3. Estimated survival ED50 and shoot fresh weight GR50 values (SEs) of sensitive (S) and resistant (R; PAPRH-R1 and PAPRH-R2) Papaver rhoeas populations treated with varying rates of acetolactate synthase (ALS)-inhibiting herbicides. a

a Resistance index (RI) was calculated as the ratio of ED50 or GR50 values of R and S, and only one RI value is presented.
Application of the P450 inhibitor PBO alone had no effect on survival or biomass in either R or S populations. Co-application of PBO with thifensulfuron + tribenuron did not alter control in PAPRH-R1 or PAPRH-R2 (Supplementary Figure S3). In contrast, PBO cotreatment with florasulam + pyroxsulam increased plant survival and biomass relative to the herbicide alone, particularly in PAPRH-R2, indicating higher resistance under PBO co-application (Supplementary Figure S4; Supplementary Table 1). As expected, the S population remained fully controlled at the recommended label rate regardless of PBO treatment.
TSR analysis confirmed that all plants from both R populations carried amino acid substitutions exclusively at Pro-197 (Table 4), whereas no mutations were detected at either Pro-197 or Trp-574 in the S population. In PAPRH-R1, thirteen plants carried the Pro-197-Leu substitution (11 homozygous and 2 heterozygous) and three plants were trans-heterozygous for Pro-197-Leu/His (Table 4). Chromatogram analysis, supported by allele-specific PCR, indicated that 68.8% of individuals were homozygous, 12.5% heterozygous, and 18.7% trans-heterozygous, with overlapping peaks reflecting the presence of two different mutant alleles. In PAPRH-R2, three plants were homozygous for the Pro-197-His substitution, two were heterozygous for Pro-197-His, three had Pro-197-Thr (1 homozygous and 2 heterozygous), and the remaining eight plants were trans-heterozygous: five with Pro-197-Leu/His, two with Pro-197-Thr/His, and one with Pro-197-Thr/Arg (Table 4). Overall mutations in PAPRH-R2 comprised 25% homozygous (n = 4), 25% heterozygous (n = 4), and 50% trans-heterozygous (n = 8).
Table 4. Amino acid substitutions identified at position Pro-197 in the ALS genes of resistant (PAPRH-R1 and PAPRH-R2) populations of Papaver rhoeas.

a Sixteen plants per population were analyzed.
b Frequency of each amino acid substitution is shown relative to the total number of plants analyzed.
c Individuals with each amino acid substitution (homozygous, heterozygous, or trans-heterozygous) are listed, with frequencies and percentage (%) given in parentheses.
Although, herbicide control failures with broadleaf weeds in this region have been less serious than those involving grass weeds (Vijayarajan et al. 2022), reports are increasing. The resistance patterns observed in this study—high SU resistance and low TP resistance in both R populations—correspond with Pro-197 TSR mutations. In the only previously reported case of P. rhoeas in Ireland, ALS inhibitor resistance involved the uncommon Trp-574 mutation alongside Pro-197 (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025). Multiple amino acid substitutions at Pro-197 (e.g., Thr, Ser, Leu, Phe, Ala, Arg, His, Asn, Ile, Tyr) have been reported to confer strong resistance to SUs, while their effects on TP herbicides range from moderate to negligible (Chtourou et al. Reference Chtourou, Osuna, Mora Marín, Hada, Torra and Souissi2024; Koreki et al. Reference Koreki, Michel, Lebeaux, Trouilh and Délye2023; Palma-Bautista et al. Reference Palma-Bautista, Portugal, Vazquez-Garcia, Osuna, Torra, Lozano-Juste, Gherekhloo and De Prado2022; Kaloumenos et al. Reference Kaloumenos, Adamouli, Dordas and Eleftherohorinos2011). In this study, PAPRH-R1 was characterized exclusively by the Pro-197-Leu substitution in homozygous, heterozygous, or trans-heterozygous forms, whereas PAPRH-R2 exhibited a broader spectrum of Pro-197 variants, including His and Thr, and multiple allelic combinations. The presence of Thr substitutions in PAPRH-R2 (ED50 and GR50 RIs of 13 and 5.5, respectively) may explain its slightly higher ALS-TP resistance compared with PAPRH-R1 (ED50 and GR50 RIs of 11 and 4, respectively), consistent with Rey-Caballero et al. (Reference Rey-Caballero, Menéndez, Osuna, Salas and Torra2017). PBO assays provided no evidence of metabolism-mediated resistance to either SUs or TPs. Preliminary tests with another P450 inhibitor, malathion (applied at 1,000 g ha⁻1; Supplementary Figure S5) produced identical results, supporting the absence of P450-mediated ALS-SU resistance. In contrast, responses to ALS-TPs were inconclusive due to variability in survival and growth (data not shown), preventing a definitive assessment of potential NTSR involvement alongside TSR. Collectively, these findings indicate that Pro-197 TSR mutations are the primary mechanism of ALS inhibitor resistance in these Irish P. rhoeas populations, with minimal contribution from metabolic NTSR under the conditions tested.
Resistance Mechanisms of Papaver rhoeas to the Auxin Mimic 2,4-D
Dose–response parameters for 2,4-D alone did not differ significantly between experiments (P > 0.05), so the data were pooled for comparison with 2,4-D + PBO treatments (Figure 2). The S population was well controlled by 2,4-D at the recommended label rate, resulting in 100% mortality and greater than 85% biomass reduction (Supplementary Figure S1). In contrast, both R populations were resistant to 2,4-D (Supplementary Figure S2), with PAPRH-R2 showing higher resistance than PAPRH-R1 (Figure 2A and 2B; Table 5). In PAPRH-R1, complete control was achieved only at four times the recommended label rate of 1,000 g 2,4-D ha−1, with ED50 and GR50 values above and below this rate, respectively (RI: 3.4 and 3.9). PAPRH-R2 showed little control even at eight times the recommended label rate, with both ED50 and GR50 values exceeding this rate (RI: 9.1 and 14.2). Pairwise comparisons of ED50 and GR50 values using the compParm() function indicated the ED50 and GR50 values of both R populations were significantly higher than those of the S population (P < 0.05), and PAPRH-R2 was significantly more resistant than PAPRH-R1 (P < 0.001) (Supplementary Table 3).

Figure 2. Dose–response curves for surival (A) and shoot biomass (B) of sensitive (S) and resistant (R; PAPRH-R1 and PAPRH-R2) Papaver rhoeas populations treated with varying rates (proportion of recommended label) of the 2,4-D herbicide, with and without the cytochrome P450 inhibitor piperonyl butoxide (PBO).
Table 5. Estimated survival ED50 and shoot fresh weight GR50 values (SEs) of sensitive (S) and resistant (R; PAPRH-R1 and PAPRH-R2) Papaver rhoeas populations treated with a range of rates of 2,4-D herbicide, with and without the cytochrome P450 inhibitor piperonyl butoxide (PBO). a

a Resistance index (RI) was calculated as the ratio of ED50 or GR50 values of R and S.
Co-application of PBO reversed resistance in both R populations, although to different extents (Figure 2A and 2B; Supplementary Figure S6). In PAPRH-R1, reversal was complete: ED50 decreased from 2,019.8 to 251.6 g ha−1 (RI from 3.4 to 1.1) and GR50 from 680.8 to 84.9 g ha−1 (RI from 3.9 to 1), achieving complete control at the recommended label rate (Table 5). In PAPRH-R2, reversal was only partial: ED50 decreased from 5,366.7 to 1,512.8 g ha−1 (RI from 9.1 to 6.9), remaining above the recommended label rate (Figure 2A and 2B), while GR50 decreased from 2,508.9 to 456.8 g ha−1 (RI from 14.2 to 5.2), falling below it (Table 5). Growth was affected in a dose-dependent manner, with complete control achieved only at higher 2,4-D doses. The S population responded similarly to PARPH-R1, achieving total control at half the recommended label rate following PBO treatment (Figure 2; Table 5), limiting the relevance of direct R/S comparisons. Pairwise comparisons confirmed that PBO significantly reduced ED50 and GR50 in both R populations and in the S population (P < 0.05), although the capacity of S population to metabolize 2,4-D was smaller than that of the R populations (Supplementary Table 3).
Additional NTSR mechanisms were investigated by examining 2,4-D absorption and translocation. No significant differences (P > 0.05) in absorption were observed between R and S populations at 4, 24, 48, and 72 h, with over 83% of the herbicide was absorbed into the leaf tissue within 4 h (Supplementary Table 2). Similarly, more than 97% of [14C]2,4-D remained in the treated leaf across all populations, consistent with the generally low translocation in P. rhoeas (Supplementary Figure S7).
Although 2,4-D is not widely used in Irish arable crops, it was selected to investigate resistance mechanisms in P. rhoeas, as resistance to this auxin mimic is well characterized. Both R populations exhibited confirmed 2,4-D resistance, with PAPRH-R2 showing higher resistance than PAPRH-R1. The differential reversal of resistance by PBO pretreatment suggests variable contributions of P450-mediated metabolism. In PAPRH-R1, resistance was fully reversed, restoring sensitivity at the recommended rate; in contrast, in PAPRH-R2, although both ED50 and GR50 decreased, the more pronounced reduction in the relatively sensitive GR50 parameter, as also reported by Burgos et al. (Reference Burgos, Tranel, Streibig, Davis, Shaner, Norsworthy and Ritz2017); Mora et al. (Reference Mora, Manicardi, Osuna, Alwarnaidu Vijayarajan, Llenes, Montull, Recasens and Torra2025) and Yu and Powles (Reference Yu and Powles2014), suggests only partial inhibition of detoxification. Given that reduced absorption or translocation is unlikely to contribute to 2,4-D resistance in either PAPRH-R1 or PAPRH-R2, these findings collectively indicate that 2,4-D resistance in Irish P. rhoeas populations is largely driven by metabolism-based NTSR. Furthermore, as 2,4-D resistance in PAPRH-R2 was only partially reversed by PBO, this population should be investigated further for a potential TSR-based resistance mechanism. Preliminary malathion tests increased survival and growth in both R and S populations rather than restoring 2,4-D activity (Supplementary Figure S8), suggesting that P450 involvement is family or isoform specific (Gaines et al. Reference Gaines, Duke, Morran, Rigon, Tranel, Kupper and Dayan2020; Torra et al. Reference Torra, Rojano-Delgado, Menéndez, Salas and de Prado2021; Zhu et al. Reference Zhu, Wang, Gao, Liu, Li, Feng and Dong2023). In Irish populations, P450s appear to synergize primarily with PBO, contrasting with some European populations, particularly in Spain (Torra et al. Reference Torra, Rojano-Delgado, Menéndez, Salas and de Prado2021), where P450 enzymes enhance 2,4-D activity in combination with both malathion and PBO.
Sensitivity Screenings of Papaver rhoeas to Other Postemergence Herbicides
Control efficacy, in terms of survival and biomass, varied significantly (P < 0.05) in both R populations depending on the herbicide and application rate, whereas the S population was well controlled even at half the recommended label rate (Table 6).
Table 6. Effects of other available postemergence herbicides at half (0.5×) and full (1×) recommended label rates on resistant (R) and sensitive (S) Papaver rhoeas populations a

a Survival and biomass expressed as a percentage (%) of untreated controls for each population with SE. Differences among populations within each herbicide treatment and dose were assessed using ANOVA. Estimated marginal means for survival and biomass were computed using the emmeans package in R (v. 3.6.3), and pairwise comparisons among populations were performed with Tukey adjustment. Letter groupings indicating significant differences (P < 0.05) were generated using the cld() function. Model assumptions were checked by visual inspection of residual plots. Means followed by different letters within each treatment and dose are significantly different.
Both R populations showed inadequate control with ALS inhibitors, especially PAPRH-R2 (Table 6), consistent with the presence of Pro-197 TSR mutations conferring broad resistance to SUs (Tranel et al. Reference Tranel, Wright and Heap2025). The most 2,4-D-resistant population, PAPRH-R2, survived nearly all auxin-mimic herbicide combinations at recommended label rates (Supplementary Figure S9). In contrast, PAPRH-R1 was totally controlled by fluroxypyr + halauxifen and showed >94% control with florasulam + halauxifen. Surviving plants from the R populations at the recommended label rates exhibited marked biomass reduction, indicating severe stunting or injury despite survival (Table 6). The comparatively lower efficacy of the three-way formulation (florasulam + clopyralid + fluroxypyr) is likely due to the natural tolerance of P. rhoeas to clopyralid, as previously reported by Palma-Bautista et al. (Reference Palma-Bautista, Portugal, Vazquez-Garcia, Osuna, Torra, Lozano-Juste, Gherekhloo and De Prado2022). Both R populations remained totally sensitive to glyphosate.
Despite indirect evidence of P450-mediated NTSR to 2,4-D, selected auxin-mimic (halauxifen-based) herbicides remain effective for PAPRH-R1. In contrast, in PAPRH-R2, the P450-mediated NTSR conferring 2,4-D resistance may extend to other auxin mimics, thereby limiting reliable in-crop control options.
Resistance to ALS inhibitors in broadleaf weeds is already present in Ireland, whereas auxin-mimic herbicides have generally remained effective against such populations (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025). However, continued dependence on these modes of action has increased selection pressure, leading to the emergence of populations resistant to more than one herbicide mode of action on some cereal farms. This study provides the first confirmation of such multiple resistance in Irish P. rhoeas populations (PAPRH-R1 and PAPRH-R2) originating from continuous winter wheat systems. Specifically, resistance was underpinned by two distinct mechanisms within the same populations: ALS inhibitor resistance conferred by TSR Pro-197 mutations and 2,4-D resistance likely mediated by enhanced metabolism (NTSR). Unlike Spanish populations, where a common P450-mediated system confers resistance to both 2,4-D and ALS inhibitors such as imazamox (Torra et al. Reference Torra, Rojano-Delgado, Menéndez, Salas and de Prado2021), resistance reversal in Irish populations was observed only for 2,4-D following PBO treatment, suggesting that the lack of response to ALS inhibitors likely reflects TSR, while the effect on 2,4-D indicates possible involvement of distinct enzymes or isoform-specific activity. Although such multiple-resistant populations are currently isolated, reduced sensitivity to newer auxin-mimic herbicides—particularly when applied at reduced rates, a common practice among Irish growers—limits reliable chemical control.
Given the limited effectiveness of cultural and nonchemical measures against P. rhoeas, future management will continue to rely on the judicious use of non–ALS inhibitor herbicides, including glyphosate, preemergence or autumn-applied residual herbicides, and auxin-mimic mixtures, while avoiding sole reliance on ALS chemistry (Alwarnaidu Vijayarajan et al. Reference Vijayarajan, Torra, Runge, de Jong, van de Belt, Hennessy and Forristal2025; Torra et al. Reference Torra, Cirujeda, Taberner and Recasens2010). Effective stewardship practices, such as stacking and sequencing residual herbicides in winter cereals, selecting appropriate herbicide combinations, and applying full recommended label rates will be essential to reduce the weed seedbank and protect remaining effective chemistries. Herbicide-based control options are further constrained by regulatory changes. The recent phaseout of metribuzin (2024), the imminent withdrawal of flufenacet (2026), and regulatory pressure on key actives such as diflufenican and pendimethalin—cornerstones of broadleaf weed control—are progressively narrowing available options (DAFM 2025). These actives are listed as candidates for substitution under EU legislation or are affected by proposed restrictions on per- and polyfluoroalkyl substances (ECHA 2025; European Commission 2025). Together, these developments will further complicate cereal weed management, highlighting the importance of proactive resistance monitoring, early detection, and careful stewardship of available herbicides.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/wsc.2026.10095
Acknowledgments
We are grateful to Gerard Nolan for glasshouse technical support and to Daniel Camacho Calero for support in translocation and absorption trials.
Funding statement
This research was supported by funding from the project EVOLVE (Evolving Grass-Weed Challenges and Their Impact on the Adoption of Carbon Smart-Tillage Systems, grant no. 2021R528), which is funded by the Department of Agriculture, Food, and the Marine (DAFM) under the Research Stimulus Fund (RSF) Programme. This work is a part of the Teagasc Climate Centre, which is a virtual center that coordinates and disseminates agricultural climate change and biodiversity research and innovation in Ireland.
Competing interests
The authors declare no conflicts of interest.