Introduction
Soil carbonate is a significant component of the global carbon cycle, composing 40–50% of total global soil carbon (Eswaran et al., Reference Eswaran, Reich, Kimble, Beinroth, Padamnabhan, Monchareon, Lal, Kimble, Eswaran and Stewart2000; Plaza et al., Reference Plaza, Zaccone, Sawicka, Méndez, Tarquis, Gascó, Heuvelink, Schuur and Maestre2018). The term “soil inorganic carbon” or “soil carbonate” is used to describe carbonate in soil that is pedogenic, detrital, or biogenic in origin (Monger et al., Reference Monger, Kraimer, Khresat, Cole, Wang and Wang2015; Zamanian et al., Reference Zamanian, Pustovoytov and Kuzyakov2016). Pedogenic carbonate refers to carbonate that precipitates in situ in soil pore water. Detrital carbonate refers to carbonate inherited from parent carbonate material, such as finely ground limestone or dust. Biogenic carbonate forms as animal or plant skeletons (e.g., shells or seeds). Amongst these three forms of soil carbonate, pedogenic carbonate is of particular interest because it represents active fluctuations in the pools of soil inorganic carbon. Furthermore, pedogenic carbonate is commonly used as a paleoclimate archive because its stable isotope composition is related to environmental conditions at the time of its formation (e.g., Cerling and Quade, Reference Cerling and Quade1993; Kelson et al., Reference Kelson, Huntington, Breecker, Burgener, Gallagher, Hoke and Petersen2020).
Pedogenic carbonate is most commonly found and studied in drylands, where the balance between limited rainfall and high evaporation is thought to promote the accumulation of calcites (e.g., Arkley, Reference Arkley1963; Royer, Reference Royer1999; Retallack, Reference Retallack2005; Breecker et al., Reference Breecker, Sharp and McFadden2009; Slessarev et al., Reference Slessarev, Lin, Bingham, Johnson, Dai, Schimel and Chadwick2016). Though less explored, humid environments host as much as 20% of the global stock of soil carbonate (Plaza et al., Reference Plaza, Zaccone, Sawicka, Méndez, Tarquis, Gascó, Heuvelink, Schuur and Maestre2018; United States Department of Agriculture, Natural Resources Conservation Service, 2022), some of which is interpreted to have a pedogenic origin (Cerling, Reference Cerling1984; Strong et al., Reference Strong, Giles and Wright1992; Wang et al., Reference Wang, Cerling and Effland1993; Railsback, Reference Railsback2021; Licht et al., Reference Licht, Kelson, Bergel, Schauer, Petersen, Capirala, Huntington, Dupont-Nivet, Win and Aung2022). An understanding of the origin and the processes driving the formation of pedogenic carbonate in humid environments would therefore help understand the factors that contribute to spatial variation in soil carbonate (Slessarev et al., Reference Slessarev, Lin, Bingham, Johnson, Dai, Schimel and Chadwick2016; Stanbery et al., Reference Stanbery, Pierce, Benner and Lohse2017; Pfeiffer et al., Reference Pfeiffer, Padarian and Vega2023). Understanding the distribution of pedogenic carbonate is important to help inform predictions of how its global distribution might evolve under climate change and anthropogenic management (Nyachoti et al., Reference Nyachoti, Jin, Tweedie and Ma2019; Ferdush and Paul, Reference Ferdush and Paul2021; Naorem et al., Reference Naorem, Jayaraman, Dalal, Patra, Rao and Lal2022; Stanbery et al., Reference Stanbery, Ghahremani, Huber, Will, Benner, Glenn and Hanif2023; Wani et al., Reference Wani, Kumar, Hussain, Wani, Babu, Alam, Rashid, Popescu and Mansoor2023). Furthermore, pedogenic carbonate can be either a net sink or source of carbon to the atmosphere (e.g., Monger et al., Reference Monger, Kraimer, Khresat, Cole, Wang and Wang2015; Sharififar et al., Reference Sharififar, Minasny, Arrouays, Boulonne, Chevallier, Van Deventer, Field and Sparks2023). If pedogenic carbonate is precipitating as an intermediate step during lithogenic carbonate weathering, it may slow the export of dissolved bicarbonate and alter carbon cycling rates (Szramek and Walter, Reference Szramek and Walter2004; Williams et al., Reference Williams, Szramek, Jin, Ku and Walter2007).
Here we document an occurrence of pedogenic carbonate in glacial drift in the humid continental climate of southern Michigan. Building on prior local work on carbonate weathering in the shallow vadose zone (Jin et al., Reference Jin, Hamilton and Walter2008a, Reference Jin, Williams, Szramek, Walter and Hamilton2008b, Reference Jin, Ogrinc, Hamilton, Szramek, Kanduc and Walter2009), we use environmental monitoring and isotope geochemistry to explore the origin and formation conditions of the soil carbonate at a site in southern Michigan. We infer the processes driving pedogenic carbonate formation at our site, and discuss its implications for paleoclimate reconstructions, regional chemical weathering, and carbon cycling.
Methods
We investigated soil carbonate formation in southern Michigan (USA) using soil stratigraphy, soil monitoring, water isotope analyses, and carbonate isotope analyses. We first describe the site and modern climate. Then we describe sampling, monitoring, and isotope analysis methods.
Site description
The study site is in the Edwin S. George Reserve (hereafter, “the Reserve”), which is a forest preserve maintained by the University of Michigan (UM), located in southern Michigan, USA (Fig. 1) (42°27.44′N, 83°59.87′W WGS84). Southern Michigan has a humid continental climate. Regional precipitation averages 860 mm/year, well above the ~500 mm/year threshold commonly used to delineate where soils contain abundant calcium carbonate (i.e., the Pedocal/Pedalfer boundary) (D'Avello et al., Reference D'Avello, Waltman, Waltman, Thompson and Brennan2019). Each month has >50 mm precipitation, with May through August being the wettest four months and precipitation primarily falling as snow from November through April. The mean annual air temperature is 8.8°C (winter mean = −3.4°C, summer mean = 20.8°C) (Arguez et al., Reference Arguez, Durre, Applequist, Vose, Squires, Yin, Heim and Owen2012).

Figure 1. Regional map showing the position of the Edwin S. George Reserve (ESGR) (red star) relative to the cities of Ann Arbor and Lansing, Michigan, with the U.S. state border in black (WGS84 datum used). The inset shows the position of the site in North America.
The geomorphology and topography of the region was strongly influenced by the growth and retreat of the Laurentide Ice Sheet (Dalton et al., Reference Dalton, Margold, Stokes, Tarasov, Dyke, Adams and Allard2020), as reflected in sand- and gravel-rich soils, numerous kettle lakes and swamps, and eskers (Rieck, Reference Rieck1976; Farrand and Bell, Reference Farrand and Bell1982; Schaetzl, Reference Schaetzl2001). Glacial sediments are derived from Canadian Shield bedrock and from the sedimentary sequence of Paleozoic and Mesozoic rocks in the Michigan Basin, which include limestones and dolomites (Milstein, Reference Milstein1987; Williams et al., Reference Williams, Szramek, Jin, Ku and Walter2007). The glacially reworked sediments in Michigan contain finely ground dolomites and limestones (Schaetzl, Reference Schaetzl1992; Williams et al., Reference Williams, Szramek, Jin, Ku and Walter2007; Jin et al., Reference Jin, Williams, Szramek, Walter and Hamilton2008b), resulting in soil series with mappable amounts of soil carbonate that are a global outlier for their high pH despite the humid climates (Slessarev et al., Reference Slessarev, Lin, Bingham, Johnson, Dai, Schimel and Chadwick2016; United States Department of Agriculture, Natural Resources Conservation Service, 2022). The study site is a knoll about 10 m high that was partially excavated during a gravel quarry operation initiated between 1950 and 1966 (Figs. 2 and 3). Most of the Reserve is forested, including the top of the knoll. The near-vertical, excavated slope itself is unvegetated. The native vegetation of the study site is a mixed oak forest with oak, tamarack, and willow (Roller, Reference Roller1974; Comer et al., Reference Comer, Albert, Wells, Hart, Raab, Price, Kashian and Corner1995). The forest is dominated by C3 vegetation: δ13C values of soil organic matter from the Reserve range from −29.4 to −24.7‰, with an average of −26.8‰ (Jin et al., Reference Jin, Ogrinc, Hamilton, Szramek, Kanduc and Walter2009).

Figure 2. Overview of the study site and soil carbonate morphologies. (A) Photo of the excavated cut slope of the knoll with overlaid interpretational sketch. (B) Photograph of the soil horizons developed on the uppermost gravel unit 9 (see description in Fig. 3). Orange flags are spaced 20 cm apart. (C) Stringer (filaments) and a rhizolith at 90 cm depth. (D) Rhizolith encasing a root at 270 cm depth. (E) Carbonate rind coating a clast. This clast was found in the float at the base of the excavated knoll. Rinds on the undersides of clasts were found in situ at 40 cm and below. (F) Rinds coating clasts and diffuse carbonate in the matrix of the lower gravel unit (depths of 325–400 cm). Pencil for scale.

Figure 3. Site stratigraphy and soil carbonate stable isotope values. (A) Sedimentary composition and pedogenic features described on the excavated, cut slope of the knoll. (B) Stable isotope values (δ13C and δ18O) of soil and detrital carbonate samples that were collected from the excavated slope of the knoll and in soil pits dug from the top of the knoll (depth to carbonate differed by 20–40 cm between those locations). Lines connect data points of the same carbonate type.
Soil stratigraphy and sample collection
We characterized the Reserve site with soil stratigraphic techniques and monitoring of modern below-ground conditions. We described the stratigraphy in a hand-dug soil pit and in the existing cut slope after we cleared it of vegetation and then further machine-excavated. Field descriptions included depth and thickness of soil and sedimentary horizons, the morphology of clay films, and soil carbonate, grain size, and structure (Birkeland, Reference Birkeland1984) (Fig. 3).
We collected soil, sediment, and water samples. The soil and sediment samples included several morphologies of soil carbonate, including rhizoliths, rinds on the bottoms of clasts (<1 mm thick), thin filaments (“stringers”), diffuse carbonate in the soil matrix, bulk sediment (matrix), and limestone/dolostone clasts. We collected water samples from within and outside of the Reserve. Within the Reserve, the soil water and surface water from East Marsh were opportunistically collected (fall 2020 to fall 2022) (n = 12 marsh samples, n = 29 soil water samples collected on 12 unique events). We collected bulk soil for soil water sampling when soil pits were dug for monitoring installation and then subsequently with a hand auger (typical depths of 10, 25, and 50 cm). We extracted the soil water from the bulk soil samples via a custom-built, cryogenic vacuum extraction line at UM (largely following that of West et al., Reference West, Patrickson and Ehleringer2006). Outside of the Reserve, we implemented a longer-term precipitation and river monitoring program in nearby Ann Arbor, Michigan (30 km distant, Fig. 1) (started fall 2018, ongoing). Weekly precipitation samples were collected with a no-oil collector (after Gröning et al., Reference Gröning, Lutz, Roller-Lutz, Kralik, Gourcy and Pöltenstein2012). Weekly Huron River samples were initially collected from a dock upstream of the Argo Dam (May–October 2018) and subsequently downstream from the Fuller Street bridge (October 2018 to present, with a hiatus in summer 2020). We collected water and bulk soil samples into vials with polycone seal caps; caps were then wrapped with Parafilm as a secondary barrier to evaporation. We stored the vials upside down at room temperature at UM until analysis.
To further characterize the modern soil system, we monitored soil temperature and soil moisture at depths of 10, 30, and 60 cm and soil CO2 concentration (pCO2) at 60 cm (Fig. 4). We monitored soil temperature with a HOBO Pendant, soil moisture with a HOBO RX3000 Remote Monitoring Station equipped with HOBOnet Soil Moisture EC-5 Sensors, and soil CO2 concentration with a CO2meter.com K33 ELG sensor capable of analyzing 0–10,000 ppm CO2. The monitoring regime covered winter 2021 to winter 2022, with gaps due to equipment failure.

Figure 4. Below-ground soil monitoring data and precipitation data. (A) Soil temperature measured at depths of 10, 30, and 60 cm (lines). Colored horizontal stripes indicate TΔ47 values (±1 SE) for soil carbonate samples with Δ47 data (20ESGR-50, 20ESGR-slope-120, 20ESGR-slope-70-75, 22ESGR-rhizolith-180cm). (B) Soil water content at the site. (C) Daily total precipitation from nearby NOAA weather station US1MIWS0055 42.43423, −83.68679), accessed via Climate Data Online on August 18, 2023 (https://www.ncei.noaa.gov/cdo-web/). (D) Soil CO2 concentrations measured at 60 cm only. The sensor used reaches a maximum at 10,000 ppm; summertime pCO2 values exceed the maximum.
Isotope analytical methods
We analyzed water and carbonate isotope compositions to investigate the conditions of soil carbonate formation.
Water stable isotope analyses
We measured the oxygen and hydrogen (δ18O and δ2H) compositions of meteoric and water samples (Fig. 5). All water samples were introduced to instrumentation as liquid water. The δ18O and δ2H were measured via Cavity Ring Down Spectrometry primarily at UM and secondarily at Washington University in St Louis (WU). The UM system consists of a Picarro A0325 Autosampler, A0211 Vaporizer, and A0214 Micro-Combustion Module connected to a Picarro L2130-i Analyzer. We normalized the isotope values to the Vienna Standard Mean Ocean Water (VSMOW) scale using four in-house liquid water standards that are referenced to U.S. Geological Survey (USGS) water standards (USGS45, 46, 49, and 50). Precision is typically better than 0.1‰ in δ18O and 0.5‰ in δ2H based on repeat measurements of deionized water (Aron et al., Reference Aron, Poulsen, Fiorella, Matheny and Veverica2020). The WU system consists of a Picarro A0325 Autosampler, A02 Vaporizer, and A0214 Micro-Combustion Module connected to a Picarro L2140-i Analyzer that was run with two lasers activated (i.e., the 17O-mode). We normalized the samples to VSMOW2 and Standard Light Antarctic Precipitation 2 (SLAP2) using in-house liquid waters and international reference waters (Hutchings and Konecky, Reference Hutchings and Konecky2023). Typical root-mean-square error (RMSE) of known waters is 0.1‰ in δ18O and 0.5‰ in δ2H. For water samples with replicate measurements, typical standard deviations are 0.1‰ in δ18O and 0.2‰ in δ2H. We report d-excess as a secondary isotope parameter for all water samples (d-excess = δ2H – 8 × δ18O).

Figure 5. Stable isotope values (δ18O, δ2H) of surface and soil waters from southern Michigan and reconstructed soil waters (δ18Orsw) from soil carbonates. (A) δ18O–δ2H relationships for meteoric waters and soil waters. The local meteoric water line (LMWL) is calculated from precipitation data. (B) δ18O–d-excess values for of all water types. (C) Box plots of δ18O values of water, soil water, and reconstructed soil water. (D) δ18O throughout the collection period. Horizontal colored stripes (orange, red, gold, yellow) indicate the δ18Orsw values for soil carbonate samples with TΔ47 data (note that 20ESGR-slope-70-75 [gold] and 22ESGR-rhizolith-180cm [yellow] have overlapping values) and their relationship with warm season meteoric water samples.
Carbonate carbon, oxygen, and clumped stable isotope analytical methods
We processed several distinct carbonate morphologies for isotope analysis. We hand-picked gravels from the bulk sediment. For rinds and rhizoliths, we brushed the soil off the outer surface. We then carefully removed the rind material from the parent clast with a hand drill. We homogenized and crushed the cleaned rhizoliths. We hand-picked thin filaments of carbonate (“stringers”) out of the soil matrix and then homogenized the material. We cut open limestone/dolostone clasts and then subsampled the cut surface with a hand drill.
We measured carbonate δ13C and δ18O (hereafter δ13Cc and δ18Oc) using a Kiel IV automated preparation device connected online to an isotope ratio mass spectrometer (a Delta V or a ThermoFisher MAT 253) at UM, standardized with NBS-19 to the Vienna Pee Dee Belemnite (VPDB) scale. Typical standard deviations of known values are <0.1‰ for δ13C and δ18O.
Clumped isotope (Δ47) geochemistry refers to the temperature-dependent clumping between 13C and 18O in a carbonate mineral (Eiler, Reference Eiler2007; Huntington and Petersen, Reference Huntington and Petersen2023). For Δ47 analyses, we prepared samples on a custom, automated vacuum extraction line connected to a Nu Perspective at the Isotopologue Paleosciences Laboratory at UM. First, 6–9 mg of carbonate equivalent of sample material was acidified in a common bath of phosphoric acid held at 90°C. The resulting CO2 was passed with helium through cryogenic water traps and a Poropak Q column. We calculated the Δ47 values using the 17O parameters of Brand et al. (Reference Brand, Assonov and Coplen2010) (Daëron et al., Reference Daëron, Blamart, Peral and Affek2016; Schauer et al., Reference Schauer, Kelson, Saenger and Huntington2016) and projected them into the ICDES90 reference frame (Bernasconi et al., Reference Bernasconi, Daëron, Bergmann, Bonifacie, Meckler, Affek and Bajnai2021; Daëron, Reference Daëron2021) using online equilibrated and heated gases (30°C and 1000°C) and carbonate standards ETH1–3. Typically, the standard deviation of Δ47 values of each standard is <0.02‰ within a session, and the long-term standard deviation of an in-house standard (102GCAZ01) is 0.014‰. We calculated temperatures (hereafter TΔ47) using the empirical calibration of Anderson et al. (Reference Anderson, Kelson, Kele, Daëron, Bonifacie, Horita and Mackey2021) (Δ47 = 0.0391*106/T2 + 0.154).
Strontium isotope analyses
We selected two carbonate rind samples for strontium isotope analysis (87Sr/86Sr), which was carried out at the University of Utah's inductively coupled plasma mass spectrometry (ICP-MS) Metals and Strontium Isotope Facility. About 50 mg of ground carbonate was digested in 0.5 mL of concentrated HNO3 at room temperature and then diluted with Type I grade water. Strontium concentration in acid digest was determined using an external calibration curve prepared from a single element Sr standard (Inorganic Ventures, Christiansburg, VA, USA) in a triple quadrupole ICP-MS (Agilent 8900, Santa Clara, CA, USA). Aliquots from the digests containing 200 ng of Sr were then mixed with concentrated HNO3 and water to 2 M HNO3 and purified using an automated system (PrepFAST MC, Elemental Scientific, Omaha, NE, USA). 87Sr/86Sr ratios were corrected for mass bias using an exponential law and normalizing to 86Sr/88Sr = 0.1194 (Steiger and Jäger, Reference Steiger and Jäger1977). To correct for isobaric interferences (e.g., from 87Rb and 86Kr), 85Rb and 83Kr were simultaneously monitored using the corresponding invariant ratios of 87Rb/85Rb = 0.385706 and 86Kr/83Kr = 1.502522 (Steiger and Jäger, Reference Steiger and Jäger1977). Measurement accuracy was assessed via multiple analyses of standard reference material SRM 987 in each run, with an analyzed value of 0.710297 ± 0.000006 (n = 8; mean ± standard deviation) compared to the certified value of 0.71034 ± 0.00026. Analytical precision (standard error) of 87Sr/86Sr for all samples was <0.00001.
Radiocarbon dating
We selected two carbonate rinds from the soil pit (50 and 70–75 cm depth) for radiocarbon analysis; 10 mg of chipped samples were sent to the University of Arizona's Accelerator Mass Spectrometry Laboratory where CO2 was extracted and purified, graphitized, and analyzed. We calculated the calibrated radiocarbon ages (cal yr BP) using the CALIB 8.0 software with the IntCal09 calibration curve (Stuiver and Reimer, Reference Stuiver and Reimer1993; Reimer et al., Reference Reimer, Baillie, Bard, Bayliss, Beck, Blackwell and Ramsey2009) (Table 1, Supplementary Table S1).
Table 1. Radiocarbon and strontium isotope data for soil carbonates from the Reserve.

Conceptual models for soil carbonate genesis and predictions for its stable isotope composition
We used previously established relationships between the stable isotopic composition of pedogenic carbonate (δ13C, δ18O, and Δ47) and its formation environment to predict the stable isotope composition of soil carbonate under three scenarios that involve formation in equilibrium with climate conditions at the Reserve (open and closed system) and its mixing with detrital carbonate (Fig. 6). We employed a simple mixing model to explore the possibility that the soil carbonate is pedogenic carbonate mixed with detrital carbonate particles (Amundson et al., Reference Amundson, Chadwick, Sowers and Doner1988; West et al., Reference West, Drees, Wilding and Rabenhorst1988a; Kraimer and Monger, Reference Kraimer and Monger2009; Zhou and Chafetz, Reference Zhou and Chafetz2010; Michel et al., Reference Michel, Driese, Nordt, Breecker, Labotka and Dworkin2013) (Fig. 6A). We modeled linear mixing in δ18O–δ13C and δ13C–Δ47 between various potential endmembers and with a varying fraction of pedogenic carbonates (F) in a Monte Carlo framework (Fig. 7). An example mixing line is shown in Figure 6 (schematically in A and B, calculated in G and H). In the Monte Carlo mixing framework (Fig. 7), we allowed for a range of endmember compositions (Supplementary Code). We allowed a range in detrital endmember compositions that was informed by the measured isotopic composition of limestone clasts at the site and the secular isotopic trends through the Phanerozoic (Veizer et al., Reference Veizer, Ala, Azmy, Bruckschen, Buhl, Bruhn and Garden1999): −8 ≤ δ18O ≤ −5, −2 ≤ δ13C ≤ +3‰, and 0.448 ≤ Δ47 ≤ 0.495‰ (65 ≤ TΔ47 ≤ 91°C). For the pedogenic carbonate endmember, we allowed for a wide potential range of compositions that could be reasonably found for the vast majority of pedogenic carbonates found globally: −15 ≤ δ18O ≤ −5‰, −14 ≤ δ13C ≤ 0‰, 0.594 ≤ Δ47 ≤ 0.678‰ (0 ≤ TΔ47 ≤ 25°C) (e.g., Cerling, Reference Cerling1984; Kelson et al., Reference Kelson, Huntington, Breecker, Burgener, Gallagher, Hoke and Petersen2020). Δ47 does not mix linearly, but the effect of nonlinearity in our range of values is ~0.0002‰ in Δ47 (White and Defliese, Reference White and Defliese2023), which is much smaller than our external precision in Δ47 (the long-term standard deviation of our in-house standard, 102GCAZ01, is 0.014‰). Note, however, that there is nonlinearity between temperature and Δ47; we perform the mixing calculations in Δ47 and then calculate temperature. We only used samples with δ13C, δ18O, and Δ47 data, and the feasible endmembers for each sample were those capable of reproducing the observed isotope composition within analytical error for all three isotopes (i.e., ±0.1‰ for δ13C/δ18O, ±5°C for TΔ47). For visual clarity we calculated F values in increments of 0.1 and emphasize differences between increments by plotting their average values.

Figure 6. Expected and measured isotope patterns for three scenarios explaining the origin of the soil carbonate. Scenario 1 (A and B): A physical mixture between pedogenic and detrital carbonate. In this scenario, δ13C and TΔ47 vary with the fraction of detrital material. δ18O is uniform because the δ18O of carbonate formed in equilibrium with meteoric waters is similar to (within 1–2‰ of) the δ18O values of detrital carbonates. Scenario 2 (C and D): Pedogenic carbonate in an open system. In this scenario, soil carbonate has a relatively small range of δ18O, δ13C, and TΔ47 values that reflects isotopic equilibrium with the environment during a single season. Scenario 3 (E and F): Pedogenic carbonate in a closed system with respect to soil CO2. In this scenario, δ13C varies while δ18O and TΔ47 are constant. Measured stable isotope values of the distinct carbonate types from the Reserve (G and H). One example of a mixing line calculated between a set of potential pedogenic and detrital endmembers is shown with black lines and arrows (i.e., one iteration of scenario A). Possible ranges of pedogenic and detrital endmember compositions at the Reserve are shown as rectangles outlined in black, filled in green and purple, respectively. These ranges are only illustrative and differ from the ranges used to constrain the mixing model shown (Fig. 8). The illustrated possible ranges of the pedogenic endmember δ13C values are calculated based on δ13C–CO2 values measured in the Huron River watershed (Jin et al., Reference Jin, Ogrinc, Hamilton, Szramek, Kanduc and Walter2009). The illustrated range of the pedogenic endmember δ18O values is based on Huron River waters and a carbonate growth temperature of 15°C. The possible range in δ18O and δ13C values of the limestone endmember is based on secular trends from Vezier et al. (Reference Veizer, Ala, Azmy, Bruckschen, Buhl, Bruhn and Garden1999) and measured limestone clast values. Where error bars are not visible, they are smaller than symbol size. The slight curvature in the δ13C–TΔ47 mixing line is due to nonlinearity in the relationship between temperature and Δ47.

Figure 7. Solutions for the model of two-component mixing between detrital and pedogenic carbonate. Plots show solutions for carbonate in sample groups separated as rinds (A and C) and rhizoliths or stringers (B and D) in δ13C–δ18O and δ13C–TΔ47 space. We only modeled samples with δ13C, δ18O, and Δ47 data to provide maximum constraint on solutions for F. The dark gray box outlines the possible detrital endmember values, the light gray box outlines the full range of possible pedogenic carbonate endmember values on a global basis, and the red triangles are observed sample values. The small colored dots are the feasible pedogenic carbonate endmembers that can mix with the detrital endmember to create the observed soil carbonate isotope composition at the assigned fraction of pedogenic carbonate (F). Different colors denote mixing model solutions for different samples. For visual clarity, we show simulations in 10% increments of the fraction of pedogenic carbonate; the large circles are averages for each increment and lines connect the average values. The set of endmember solutions for the assumed fraction of pedogenic carbonate are only labeled for the bottom right sample in each panel (e.g., “solutions for F = 0.6”), but all samples have the same pattern. Note that when the assumed fraction of pedogenic carbonate is 100% (F = 1), the endmember solutions necessarily match the observed sample isotope composition.

Figure 8. Environmental conditions capable of creating observed soil carbonate δ13C values assuming samples are pedogenic carbonate formed in an open system. Theoretically feasible conditions for the δ13C of soil respiration and soil CO2 concentration are shown for three sample groups: (A) rinds, (B) rhizoliths and stringers, and (C) bulk soil samples. Colored dots denote solution spaces (10,000 iterations each) for different samples. Solutions that are consistent with modern conditions must fall within the black box, which outlines modern conditions for δ13C of soil respiration in a C3 dominated landscape (Tipple and Pagani, Reference Tipple and Pagani2007; Jin et al., Reference Jin, Ogrinc, Hamilton, Szramek, Kanduc and Walter2009) and the observed soil CO2 concentrations during our monitoring period.
In a canonical calcic soil system, the soil pore water and CO2 are open to isotopic exchange with incoming meteoric waters and the soil gas reservoir, resulting in a relatively small range in pedogenic carbonate isotope values that reflect formation in equilibrium with soil climate conditions (Fig. 6B) (Cerling, Reference Cerling1984; Cerling and Quade, Reference Cerling and Quade1993; Quade et al., Reference Quade, Eiler, Daëron and Achyuthan2013). To model the pedogenic carbonate system, we first calculated the δ18O values of the carbonate-parent water (i.e., reconstructed soil water, δ18Orsw) using the temperature-dependent isotope fractionation factor informed by TΔ47 data (Kim and O'Neil, Reference Kim and O'Neil1997) (Fig. 5C). For samples without TΔ47 data, we used the average TΔ47 value to calculate δ18Orsw. Choice of calibration for the fractionation factor does not materially change our interpretations (i.e., Coplen, Reference Coplen2007 vs. Kim and O'Neil, Reference Kim and O'Neil1997). We compared the δ18Orsw to measured δ18O values of meteoric and soil waters.
Then, we modeled the set of environmental conditions potentially capable of producing the measured soil carbonate δ13C values using established equations that relate pedogenic carbonate to soil respiration, the overlying vegetation, and the atmosphere. For each sample, we first calculated the δ13C of soil gas (δ13Cs) from the measured δ13C of soil carbonate (δ13Cc) and its formation temperature (Romanek et al., Reference Romanek, Grossman and Morse1992):
where ɛcl-CO2 is the mineral-gas fractionation factor for calcite (i.e., δ13Cs = (δ13Cc + 1000)/(ɛcl-CO2/1000 + 1) − 1000). Then, we modeled the δ13C of soil respiration (δ13Cr) (Cerling, Reference Cerling1984; Cerling and Quade, Reference Cerling and Quade1993; Davidson, Reference Davidson1995):
where Ca is atmospheric pCO2 and Cs is soil pCO2. This equation is solved independently for each measured δ13Cc value, each representing a specific depth. The calculations were performed 10,000 times, assuming varying conditions within a plausible range for the Late Quaternary (atmospheric CO2 concentration: 180 to 280 ppm, soil CO2 concentration: atmospheric concentration to 10,000 ppm; soil temperature: 0 to 25°C; δ13C of atmospheric CO2: −6 to −7‰) (Cerling, Reference Cerling1984; Davidson, Reference Davidson1995; Bereiter et al., Reference Bereiter, Eggleston, Schmitt, Nehrbass-Ahles, Stocker, Fischer, Kipfstuhl and Chappellaz2015; Eggleston et al., Reference Eggleston, Schmitt, Bereiter, Schneider and Fischer2016; Huth et al., Reference Huth, Cerling, Marchetti, Bowling, Ellwein, Passey, Fernandez, Valley and Orland2020) (Supplementary Code). The modeled conditions that were required to produce the measured δ13Cc values were then compared to modern conditions (Fig. 8).
Results
Soil profile developed in carbonate-rich glacial drift
We documented a soil profile developed in stratified glacial drift. The soil profile is about 2.4 m thick and consists of two A horizons, a Bt horizon, and a Bk horizon (Figs. 2 and 3). The parent material is fine–medium, poorly sorted sand with 10−15% gravel; evidence of primary sedimentary bedding was not identified. Soil horizons (A1, A2, Bt, Bk, and Ck) were differentiated based on organic content, color, pedogenic structure, and secondary mineral composition (Fig. 3). Notably, the Bk horizon was identified based on the presence of carbonate (i.e., the first depth at which the matrix reacted vigorously with hydrochloric acid application) and the sharp color change relative to the overlying layer. Within the Bk horizon, carbonate was found as stage I–II rinds on the bottom of clasts, rhizoliths (some surrounding still-intact roots), stringers, and as diffuse fine-grained material within the matrix (Fig. 2). The depth to the Bk horizon varied by up to 50 cm at several sampled locations (instrumented soil pit, auger collections, and the excavated slope) and at other test pits in glacial drift in the region; this variation could be partially due to alteration of the ground surface elevation during historical quarry operations and/or natural variation in leaching depths.
Below the soil profile, we found stratified sands and gravels consistent with a fluvio-glacial drift deposit, such as a kame or an esker (Fig. 3). A layer of cross-bedded sand appears at 2.4 m below the surface. The sand unit is very fine to very coarse and well rounded, with some grain-size separation in the beds. There were abundant carbonate rhizoliths throughout the sand unit, and the matrix reacted vigorously with hydrochloric acid. The gravel unit 3.2 m below the ground surface consisted of lenses of sorted sediments, ranging from rounded–subrounded gravels to medium sand. The gravel unit reacted with HCl vigorously throughout, and many of the clasts displayed incipient (stage I) carbonate rinds (Figs. 2 and 3).
Calcite mineralogy was confirmed with X-ray diffraction analysis of rind material drilled from clasts collected at depths of 50 and 70 cm in a soil pit adjacent to the slope. The 87Sr/86Sr values of ≈0.709 for the rinds collected at 50 cm and 70–75 cm are consistent with marine limestone as the parent material (Table 1). The radiocarbon ages are 23,691 cal yr BP and 17,366 cal yr BP for the same rinds collected at 50 cm and 70–75 cm (Table 1).
Results of below-ground soil monitoring
Over the year of monitoring we conducted, we observed seasonal cycles in soil temperature, water content, and pCO2. At carbonate-relevant depths of 60 cm, winter temperatures are near-freezing in January and February (0.6°C). The soil begins to thaw in mid-March, and temperatures reach a maximum temperature of 21.4°C in late August (Fig. 4). Soil water content remains at 0.2–0.3 m3/m3 for winter and spring (December through June), with minor fluctuations due to infiltration of snowmelt and precipitation. Initial snowmelt occurred in late February, causing an increase in water content at all depths that was followed by several cycles of increasing and decreasing soil moisture heading into the summer (Fig. 4). The soil pCO2 values were between 4300 and 4500 ppm midwinter (January to February). In late March, pCO2 concentrations started to rise above winter lows, and then remained >5000 ppm with spikes of >8000 ppm through early summer. Mid-late summer pCO2 values (July to August) exceeded 10,000 ppm (the limit of our sensor). The pCO2 record ended on August 11, 2021. Even considering the relatively short period of monitoring, these data capture the major seasonal trends (Fig. 4).
Results of stable isotope analyses of carbonates and water and of predictions for pedogenic carbonate
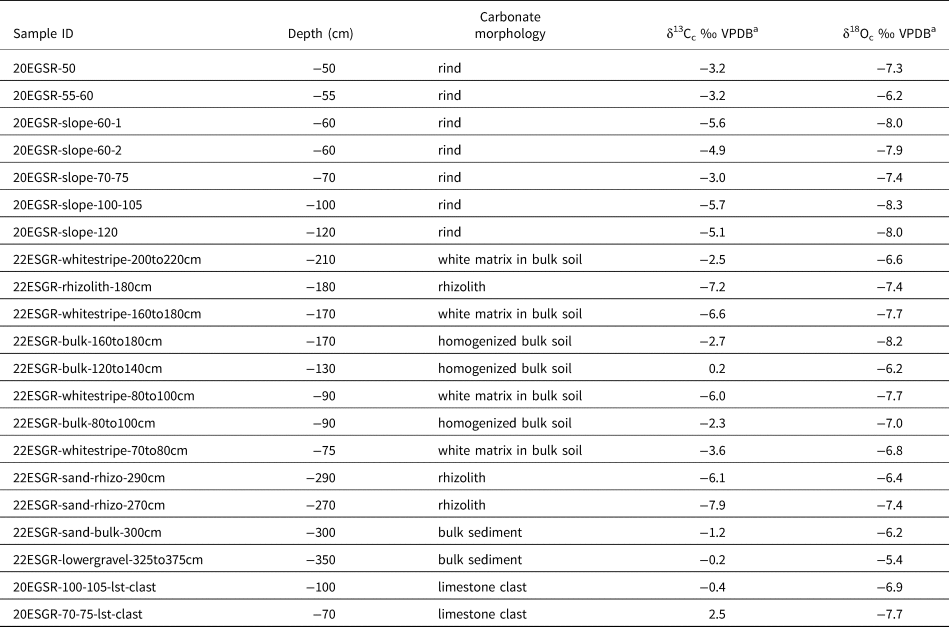
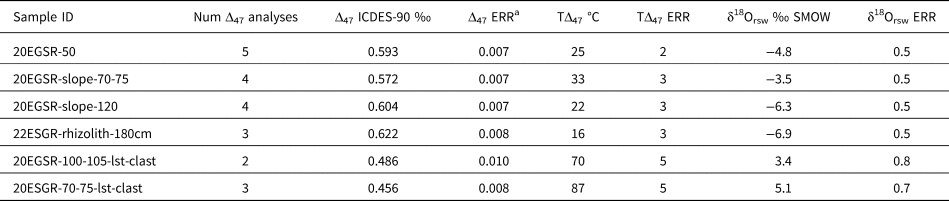
The δ13Cc values of all carbonates are −7.9 to 2.5‰ (VPDB), spanning a 10.4‰ range (Table 2, Figs. 3 and 8). The δ13Cc values of the rhizolith, stringer, and rind samples are generally lower than those of the bulk matrix and parent limestone clasts (Figs. 3 and 6). The δ18Oc values of all carbonates are −8.3 to −5.4‰ (VPDB), spanning a 2.9‰ range (Table 2, Figs. 3, 6, and 8). There are not marked differences in δ18Oc values amongst carbonate morphologies (Fig. 6). A subset of the samples was measured for Δ47, and those values range from 0.4562 to 0.6219‰ (ICDES-90), corresponding to temperatures (TΔ47) of 87 to 16°C (Table 3). The TΔ47 values of the measured rinds and rhizoliths are lower than the TΔ47 values of the limestone clasts (range of 15.9–32.9°C vs. 70.2–86.6°C) (Table 3). There is significant covariation between δ13Cc and TΔ47 (r2 = 0.89, P = 0.004) amongst all the carbonate morphologies, but there is not covariation between δ18Oc and δ13Cc or TΔ47 (Fig. 6D).
Table 2. Stable carbon and oxygen isotope data from carbonates in the Reserve.

a δ13C and δ18O data are generated via CO2 on a Kiel Device. Typical precision is ±0.1.
Table 3. Clumped isotope (Δ47) data and calculated δ18Orsw values for select soil carbonates from the Reserve.

a Δ47 ERR is calculated as the larger of 1 SD of sample measurements or 0.014 (the long term SD of in-house standard 102GCAZ01) divided by the square root of the number of measurements.
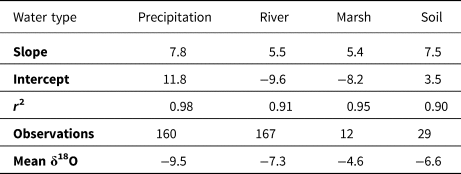
We observed seasonal variation in the isotopic values (δ18O, δ2H, and d-excess) of meteoric waters (Fig. 5), typical of continental locations with seasonal fluctuations in air temperature (Rozanski, Reference Rozanski, Araguás-Araguás, Gonfiantini, Swart, Lohmann, McKenzie and Savin1993). There is covariation between δ18O and d-excess for samples with δ18O > ~−10‰, indicative of evaporation. The local meteoric water line (i.e., δ18O–δ2H linear regression) defined by precipitation has a slope of 7.8 that is close to the canonical value of 8 (Table 4) (Putman et al., Reference Putman, Fiorella, Bowen and Cai2019). The isotopic composition of the precipitation is quite variable (δ18O ranges from −23.6 to −0.14‰ VSMOW; d-excess ranges from −8.5 to 25.8‰), though the isotopically light precipitation events tend to occur only in the winter and the isotopically heaviest rain occurs in the summer. The Huron River water has less variability in δ18O and δ2H than precipitation, but does have a sinusoidal pattern of seasonal variation (Fig. 5), with low δ18O values (high d-excess values) in the winter (typical minimum δ18O of −9 ‰ and maximum d-excess of +15) and high δ18O values (low d-excess values) in the summer (typical maximum δ18O of −5 and minimum d-excess of +5) (Pelletier, Reference Pelletier2020). East Marsh in the Reserve has δ18O values that are consistently higher than the contemporaneous river water and the δ18O–δ2H relationship has a slope of 5.4, indicating evaporative influences (Table 4). The soil water isotope values generally reflect the seasonal patterns delineated by the river and precipitation water isotopes. The slope of the soil water line (i.e., δ2H–δ18O trend) is 7.4, indicating only minor evaporative influences.
Table 4. Summary of meteoric and soil water isotope (δ18O, δ2H) data.

We predicted stable isotope values and patterns that we would have observed if the soil carbonate formed only via pedogenesis and if pedogenic carbonate was mixed with detrital carbonate (Figs. 6–8). The mixing model between the stable isotope compositions of detrital and pedogenic carbonate can satisfactorily explain the observed δ13C, δ18O, and Δ47 data for all sample types if samples have pedogenic fractions of approximately 0.6 ≤ F ≤ 1 (Fig. 7). The calculated δ18Orsw values overlap with the highest observed δ18O values of precipitation, river, marsh, and soil water isotope values (Fig. 5). The environmental conditions required to match the measured δ13Cc have no or minimal overlap with modern conditions (Fig. 8). Only one rhizolith, with the lowest δ13C value, could potentially be consistent with modern soil conditions.
Discussion
Physical evidence of pedogenic carbonate
The carbonate morphology and underlying sedimentology supports post-glacial pedogenic carbonate formation (Fig. 2). Our initial observations of carbonate morphology are strongly suggestive of in situ pedogenic carbonate precipitation (Gile et al., Reference Gile, Peterson and Grossman1966; West et al., Reference West, Wilding and Hallmark1988b; Gocke and Kuzyakov, Reference Gocke and Kuzyakov2011; Zamanian et al., Reference Zamanian, Pustovoytov and Kuzyakov2016) (Fig. 2). Calcite rinds indicate in situ recrystallization of calcite, though they are typically described in deserts (Gile et al., Reference Gile, Peterson and Grossman1966). Rhizoliths cross cut sedimentary bedding structures and often surround roots of living plants. Filaments of calcite-rich material, called stringers, are also indicative of pedogenic processes. The diffuse carbonate in the matrix throughout the sedimentary sequence is equally likely to be finely ground detrital limestone or secondary carbonate (Kraimer and Monger, Reference Kraimer and Monger2009; Li et al., Reference Li, Chen and Chen2013; Zamanian et al., Reference Zamanian, Lechler, Schauer, Kuzyakov and Huntington2021).
Isotopic evidence for pedogenic carbonate and its formation processes
We examined the stable isotope data in the context of three scenarios to explain the origin of the soil carbonate: (1) a mixture of pedogenic and detrital carbonate; (2) pedogenic carbonate formed under equilibrium, open-system conditions; and (3) pedogenic carbonate formed in closed-system conditions (Fig. 6). Each of these scenarios would yield distinct isotope patterns (in TΔ47, δ18O, and δ13C) in the resulting soil carbonate (Fig. 6). The isotope and monitoring data are most consistent with the scenario that the soil carbonate is a mix between pedogenic and detrital carbonate.
A physical mixture of pedogenic and detrital carbonate
A mix between pedogenic and detrital endmembers can parsimoniously explain the observed isotopic values (δ18O, δ13C, TΔ47, and 14C) of the carbonate (Figs. 6 and 7). Because the δ18O value of the detrital limestone overlaps with the δ18O value of pedogenic carbonate formed in equilibrium with modern waters, δ18O is almost invariant in this scenario (Fig. 6), matching the measured δ18Oc/δ18Orsw values (Fig. 5). TΔ47 and δ13C covary because both are dependent on the mixing fraction (Fig. 6). The detrital endmembers (limestone clasts) have high TΔ47 values of 70–86°C and high δ13C values. The sampled rhizolith has a low TΔ47 of 16°C and a low δ13C value, while the rinds have intermediate TΔ47 values of 21–33°C and intermediate δ13C values (Fig. 6). Because of this variation, the TΔ47 values provide a strong constraint on the fraction of pedogenic carbonate within the samples (0.5 ≤ F ≤ 1) (Fig. 7). A mix between detrital and pedogenic carbonates is also consistent with the radiocarbon ages that predate glacial retreat; the radiocarbon-dead-detrital component would make 14C ages appear older than their true formation age.
The distinctions in stable isotope values with carbonate morphology mostly fit with our expectation given a physical mix. The bulk samples have δ13C and δ18O values that overlap with that of the detrital limestone (Fig. 6 G and H, Table 2), which is expected given the higher potential for detrital contamination in bulk carbonate. For carbonate morphologies that are more likely to be composed of a higher proportion of pedogenic carbonate (rinds, rhizoliths, and stringers), the isotopic composition is explained via a mix of pedogenic and geogenic carbonate (Figs. 6 and 7). This framework groups rhizoliths, rinds, and stringers as having a higher proportion of pedogenic carbonate than bulk samples (Fig. 6), as also observed by Zamanian et al. (Reference Zamanian, Lechler, Schauer, Kuzyakov and Huntington2021) and Gocke et al. (Reference Gocke, Pustovoytov, Kühn, Wiesenberg, Löscher and Kuzyakov2011). Using this framework, the isotopic data suggest that the rhizoliths tend to have a higher proportion of pedogenic carbonate than rinds or stringers. This pattern could be a sampling artifact: The rinds were thin (~1 mm) and separating them from carbonate-cemented matrix was subjective. In comparison, the rhizoliths were more straightforward to isolate from matrix because they were thicker (~1 cm), firm, had a predictable, cylindrical geometry, and were whiter than the surrounding matrix. This apparent difference in pedogenic component could be a true result that relates to their formation mechanisms. We might expect rinds to have matrix contamination if they form as hypocoatings in pore space that grow towards the clasts, rather forming gravitationally like a pedothem in arid soils (Ducloux et al., Reference Ducloux, Butel and Dupuis1984; Durand et al., Reference Durand, Monger, Canti, Verrecchia, Stoops, Marcelino and Mees2018). In comparison, rhizoliths have been previously shown to be purely secondary (pedogenic) carbonate (Gocke et al., Reference Gocke, Pustovoytov, Kühn, Wiesenberg, Löscher and Kuzyakov2011). In one model of rhizolith formation, the first step is that acidic root exudate dissolves the surrounding matrix (including detrital carbonate and non-carbonate minerals). Subsequently, the root selectively takes up Ca2+ in solution, then rhizolith carbonate precipitates from the remnant Ca2+ in solution and CO2 from microorganism respiration (Barta, Reference Barta2011; Brazier et al., Reference Brazier, Schmitt, Gangloff, Pelt, Gocke and Wiesenberg2020; Huguet et al., Reference Huguet, Bernard, El Khatib, Gocke, Wiesenberg and Derenne2021). Therefore, the mixing model can explain the composition and origin of the soil carbonate at the Reserve.
The Monte Carlo mixing model gives predictions for the isotopic composition of the pedogenic carbonate endmembers (Fig. 7). The model predicts a range of δ13C values for the pedogenic carbonate endmember, consistent with formation under mixed C3–C4 vegetation regimes or at substantially lower soil CO2 concentrations than observed today. Our results are therefore consistent with, but do not uniquely identify, a substantial component of C4 vegetation in southern Michigan in the late deglacial/Early Holocene (Nelson et al., Reference Nelson, Hu, Grimm, Curry and Slate2006; Chapman and Brewer, Reference Chapman and Brewer2008).
Alternative scenarios: pedogenic carbonate formed in modern open- or closed-system conditions
Given that the soil carbonate appears morphologically to be pedogenic, the simplest hypothesis is that the soil carbonate formed via pedogenic processes under post-glacial environmental conditions. However, the isotope data (δ18O, δ13C, TΔ47, and 14C) and monitoring data are not fully compatible with this scenario (Figs. 5, 6, and 8).
The only evidence that aligns with the pedogenic carbonate scenario is that the δ18Orsw values overlap with the highest observed δ18O values of precipitation, river, marsh, and soil water. These data could be consistent with pedogenic carbonate formation from parent soil waters during the warm half of the year (Fig. 5), but the other considered scenarios could also explain the measured δ18Oc values (Fig. 6), so the information from δ18O is nonunique at this site.
All other isotope data do not match our predictions for open-system, modern pedogenic carbonate. First, the radiocarbon ages of 17,366 and 23,691 cal yr BP predate glacial retreat from the region (Table 1) (Dalton et al., Reference Dalton, Margold, Stokes, Tarasov, Dyke, Adams and Allard2020), which would require pedogenic carbonate formation while the region was covered with an ice sheet. Second, the TΔ47 values of 16 to 33°C overlap or exceed the maximum measured soil temperatures (25°C) at 10 to 60 cm (Fig. 4). A simple interpretation of these data would suggest pedogenic carbonate formation during the warmest months (but even so, temperatures of 33°C are not reasonable). This interpretation is inconsistent with the δ13C modeling that, in a C3-dominated environment, requires formation at pCO2 ~<2500 ppm, and the fact that summer pCO2 exceeds 8000 ppm. Finally, the δ13C values of the soil carbonate samples are generally inconsistent with predictions of values of pedogenic carbonate formed under modern conditions (colored dots in Fig. 8). The δ13C values of most of the soil carbonate samples are higher than would be expected if they were pedogenic. The environmental conditions capable of recreating the measured soil carbonate δ13C values are either low soil pCO2 (~<2500 ppm) or δ13C values of respired CO2 of ~−20 to −15‰ (colored dots in Fig. 8). Neither of these conditions are consistent with modern conditions (black box in Fig. 7), where the minimum measured soil pCO2 is 4000 ppm and the overlying vegetation is dominantly C3 (forested) (measured δ13Corg ranges from −29.4 to −24.7‰; Jin et al., Reference Jin, Ogrinc, Hamilton, Szramek, Kanduc and Walter2009). The only samples for which there is some overlap in modeled and observed conditions are rhizoliths, which is consistent with stratigraphic evidence supporting that these are primarily modern (Figs. 6 and 8).
Another possibility is that the rind and bulk samples formed pedogenically immediately after glacial retreat, when the sparse vegetation coverage could lead to high δ13C–CO2 values in the soil. This possibility could explain their relatively high δ13C values (Fig. 8). However, it is difficult to explain why the post-glacial rinds would have higher formation temperatures than the modern rhizoliths (Tables 2 and 3, Fig. 6) given that post-glacial climates are cooler than the present day (c.f. varying seasonality of soil carbonate formation; Kelson et al., Reference Kelson, Huntington, Breecker, Burgener, Gallagher, Hoke and Petersen2020). Furthermore, we might expect that the δ18O of meteoric water and pedogenic carbonate would be different in post-glacial versus present-day southern Michigan, but we measured uniform δ18O values amongst the sample types. The mixing model (scenario 1) is a simpler explanation that unifies all the data.
Theoretically, another possibility is that the soil carbonate is forming in situ, but rather than following the typical pedogenic model described above, the carbonate is re-precipitated from detrital carbonate in a closed system. Ultimately, this scenario is not consistent with the isotope and monitoring data (Fig. 6) and our understanding of shallow vadose zones. In a closed system, meteoric water equilibrates with the gas reservoir and dissolves pre-existing carbonate (i.e., detrital limestone) without further replenishment of CO2 or water. Jin et al. (Reference Jin, Ogrinc, Hamilton, Szramek, Kanduc and Walter2009) reported that the vadose zone in the Reserve might be partially closed based on elevated values of δ13C of dissolved inorganic carbon at depths >1.7 m, but those data could also represent equilibrium values at a high pH. A closed system would yield variable δ13C values and invariant δ18O values: The δ13C of carbonate increases with cycles of dissolution and re-precipitation but the δ18O of carbonate retains the δ18O value derived from meteoric water (Salomons and Mook, Reference Salomons, Mook, Fritz and Fonts1986; Lohmann, Reference Lohmann, James and Choquette1988) (Fig. 6). This predicted δ18O versus δ13C pattern is displayed by the soil carbonates in the Reserve but could be explained by other scenarios (Fig. 6). Closed-system precipitation could also explain the “too-old” 14C ages, whereby 14C in the soil pore waters would be partially derived from dissolved limestone and/or pre-glacial, inherited organic matter (Wang et al., Reference Wang, McDonald, Amundson, McFadden and Chadwick1996). However, under closed-system precipitation we would expect uniform clumped isotope temperatures resembling mean annual air temperature at the depths of carbonate formation (>50 cm) (Quade et al., Reference Quade, Eiler, Daëron and Achyuthan2013; c.f. seasonal biases in Kelson et al., Reference Kelson, Huntington, Breecker, Burgener, Gallagher, Hoke and Petersen2020) (Fig. 6). Instead, we document variation in TΔ47 that correlates with δ13C (Fig. 6). Furthermore, under a closed or partially closed system, we might expect an increase in δ13C with depth, where shallow carbonates form under a more open system. We do not find a depth–δ13C pattern (Fig. 3), though this pattern may be difficult to detect given that we observe distinct carbonate morphologies with depth (i.e., the deepest carbonates are rhizoliths, which are unlikely to be closed system given their adjacency to roots). Previous workers have shown open-system behavior at depths of ~<1 m in soils: Even in limestone terrane, pedogenic δ13C values match predictions of the pedogenic model of mixed atmospheric and respired CO2 with a negligible contribution of CO2 dissolved from parent limestone (Amundson et al., Reference Amundson, Chadwick, Sowers and Doner1989; Cerling and Quade, Reference Cerling and Quade1993). Given these inconsistencies, pedogenic formation in a partially closed vadose zone is unlikely to be the dominant mode of carbonate formation at the Reserve.
Processes driving pedogenic carbonate formation in southern Michigan and implications for carbon cycling
We can confidently determine that pedogenic carbonate has formed in our field site in southern Michigan, even if it is physically mixed with detrital carbonate, and our data allow us to consider the process(es) driving its formation.
In arid settings, it is thought that pedogenic calcite precipitation is driven by wetting and drying cycles. Infiltrating precipitation dissolves Ca-bearing minerals and brings Ca2+ ions (e.g., from dust; Reheis, Reference Reheis2006) from the surface to depths in the soil profile. When the soil pore water dries via evapotranspiration, it reaches supersaturation with respect to calcite, and calcite precipitates (e.g., Breecker et al., Reference Breecker, Sharp and McFadden2009; Tabor et al., Reference Tabor, Myers, Gulbranson, Rasmussen, Sheldon, Driese and Nordt2013; Gallagher and Sheldon, Reference Gallagher and Sheldon2016; Huth et al., Reference Huth, Cerling, Marchetti, Bowling, Ellwein and Passey2019; Kelson et al., Reference Kelson, Huth, Passey, Levin, Petersen, Ballato and Beverly2023). However, soil drying is probably not the major mechanism driving pedogenic carbonate formation in the Reserve because (1) we observed little variation in soil water content, outside of minor increases after summer rain (Fig. 4), and (2) there is little isotopic evidence of evaporation in the soil waters (Fig. 5, Table 4). The δ18O–δ2H composition of the soil waters falls near the local meteoric water line and has a slope of 7.5 (Table 4); typical soil water evaporation slopes in arid places are 2–3 (Benettin et al., Reference Benettin, Volkmann, Von Freyberg, Frentress, Penna, Dawson and Kirchner2018; Bowen et al., Reference Bowen, Putman, Brooks, Bowling, Oerter and Good2018). The isotopic composition of the soil waters reflects seasonal patterns of precipitation. For example, we observed wholesale isotope reset to the measured depths of 50 cm after precipitation events between September 3, 2021 and October 28, 2021, indicating significant infiltration of unevaporated soil water. In this setting, the formation of pedogenic carbonate is not controlled by a balance between incoming precipitation and soil drying. And, unlike desert settings where carbonate accumulates with time into progressively mature Bk horizons (Gile, Reference Gile1961; Gile et al., Reference Gile, Peterson and Grossman1966), soil carbonate may not be accumulating in the Reserve.
Instead, we propose that in the Reserve the pedogenic carbonate is forming ultimately because of an abundant supply of aqueous Ca2+ derived from glacially ground limestone and dolostone in the parent till and seasonal cycles in soil respired CO2 (Jin et al., Reference Jin, Hamilton and Walter2008a). Fine-grained carbonates, like those ground up by glaciers, are particularly susceptible to dissolution and recrystallization (Anderson et al., Reference Anderson, Drever and Humphrey1997; Gallagher and Breecker, Reference Gallagher and Breecker2020). We hypothesize that the detrital limestone is dissolving and re-reprecipitating on a seasonal basis, resulting in pedogenic carbonate formation as an intermediate product (also described by West et al., Reference West, Drees, Wilding and Rabenhorst1988a). The 87Sr/86Sr values of ≈0.709 suggest that the source of Ca2+ is marine limestone (or dust derived from marine limestone). In the nearby Huron and Kalamazoo watersheds, Jin et al. (Reference Jin, Hamilton and Walter2008a) found that the saturation index of calcite in pore water is constant throughout the year, while the Ca2+ concentrations increased during the summer via calcite dissolution at higher pCO2. In the surface water, lakes, and wetlands, however, secondary calcite precipitates due to degassing (Szramek and Walter, Reference Szramek and Walter2004). Our data also show that pCO2 increases in the summer, probably due to soil respiration. Put together, this evidence suggests that the in situ precipitation of pedogenic carbonate in the soil is driven by cycles of soil-respired pCO2. Higher soil temperatures enable increased respiration, which increases soil pCO2 and dissolves existing fine-grained calcite while maintaining a constant saturation index. Subsequently, cooling in the fall results in decreased respiration, decreased pCO2, and decreased Ca2+ ions in the pore water as the calcite re-precipitates. This timing and mechanism are consistent with the stable isotope composition of the rhizolith sample that we hypothesize best represents the pedogenic endmember (i.e., the sample with the lowest δ13C and TΔ47 values). The TΔ47 of the rhizolith is 16 ± 3°C, matching soil temperatures in September to October when pCO2 is likely decreasing. Our observations support that soil respiration rates, which influence the acid-carrying capacity of soil pore waters, are an important lever in controlling the dissolution and re-precipitation of carbonate in the soil profile, and ultimately the export of bicarbonate to streams (Calmels et al., Reference Calmels, Gaillardet and François2014; Romero-Mujalli et al., Reference Romero-Mujalli, Hartmann and Börker2019).
Another factor contributing to pedogenic carbonate formation may be the mixed carbonate mineralogy in the watershed. Dolomite is less soluble and dissolves more slowly than calcite in the temperate climate of southern Michigan. Ground waters and surface waters in the Huron River watershed are undersaturated with respected to dolomite (MgCa(CO3)2) but supersaturated with respect to calcite (CaCO3) (Williams et al., Reference Williams, Szramek, Jin, Ku and Walter2007; Jin et al., Reference Jin, Williams, Szramek, Walter and Hamilton2008b). Continued dissolution of dolomite (which releases Ca2+ and Mg2+) after soil water is saturated with respect to calcite may contribute to calcite supersaturation in the watershed, setting the stage for calcite re-precipitation.
The pedogenic carbonate at the Reserve may represent a geologically transient feature. The carbonate has been leached from the top ~0.5 to 1.5 m at the Reserve after glacial retreat and at similar profiles described in the region (Figs. 2 and 3) (Jin et al., Reference Jin, Hamilton and Walter2008a, Reference Jin, Ogrinc, Hamilton, Szramek, Kanduc and Walter2009). There is net dissolution of carbonate minerals in the Huron River watershed (Williams et al., Reference Williams, Szramek, Jin, Ku and Walter2007; Jin et al., Reference Jin, Williams, Szramek, Walter and Hamilton2008b). It is likely that the observed pedogenic carbonate will ultimately be dissolved and carried away by ground water on geologic timescales. Thus, the morphology, depth, and amount of pedogenic carbonate in this profile are transient, adjusting to post-glacial conditions. It is unlikely that such soil profiles with chemistry adjusting to changing climate conditions would be preserved in the geologic record as paleosols. If they were preserved, paleosols analogous to those currently found in the Reserve would be poor records of paleoclimate because the stable isotopic composition does not perfectly reflect environmental conditions in all samples. Even as a transient feature, the pedogenic carbonate represents an important intermediate step in watershed-scale carbonate weathering. The back-precipitation of carbonates in soils could help explain the missing Ca2+ in solute chemistry of rivers (Cavazza et al., Reference Cavazza, Zuffa, Camporesi, Ferretti, Johnsson and Basu1993; Erlanger et al., Reference Erlanger, Rugenstein, Bufe, Picotti and Willett2021; Bufe et al., Reference Bufe, Cook, Galy, Wittmann and Hovius2022) and could delay the export of bicarbonate from parent material to the ocean.
Conclusion
We presented physical and isotopic evidence for pedogenic carbonate formation in the humid, temperate climate of southern Michigan. The isotope values of the soil carbonate are most simply interpreted as a mixture of pedogenic and detrital carbonate. Because of the detrital component in this carbonate-rich glacial drift, paleoclimate reconstructions based on primary pedogenic carbonate material would require finer-scale techniques like laser ablation or secondary ion mass spectrometry (Passey and Cerling, Reference Passey and Cerling2006; Oerter et al., Reference Oerter, Sharp, Oster, Ebeling, Valley, Kozdon and Orland2016; Huth et al., Reference Huth, Cerling, Marchetti, Bowling, Ellwein, Passey, Fernandez, Valley and Orland2020). For hand-drilled samples, it is preferable to avoid limestone terranes when developing soil-based paleoclimate records (Kraimer and Monger, Reference Kraimer and Monger2009; Sheldon and Tabor, Reference Sheldon and Tabor2009; Cotton and Sheldon, Reference Cotton and Sheldon2012; Michel et al., Reference Michel, Driese, Nordt, Breecker, Labotka and Dworkin2013).
The apparent in situ precipitation of rinds and rhizoliths has implications for terrestrial pools of inorganic carbon and carbon cycling. First, this study demonstrates the existence of pedogenic carbonate, at least transiently, in a wider range of ecosystems than is typically recognized (Railsback, Reference Railsback2021; Licht et al., Reference Licht, Kelson, Bergel, Schauer, Petersen, Capirala, Huntington, Dupont-Nivet, Win and Aung2022). Though there is net dissolution of carbonates in the glacial till in southern Michigan (Szramek and Walter, Reference Szramek and Walter2004), the timescale of dissolution is mediated by open-system re-precipitation of calcite in the shallow weathering zone, driven by seasonal fluctuations in soil respired CO2. Because the formation of pedogenic calcite is likely driven by soil-respired CO2, changes in land use and vegetation cover could affect the rates and amount of net calcite precipitation in this setting. Given that much of the Midwest is deeply mantled in glacial till that may also be rich in finely ground detrital carbonates, it is possible that re-reprecipitation of pedogenic carbonate may be occurring across much of the landscape. Our results raise the question of how the rates and magnitudes of this calcite re-precipitation mediate the net dissolution of parent limestone and dolomite and thus slow the export of dissolved bicarbonate. If back-precipitation as pedogenic carbonate is occurring on a sufficiently large scale, it could be relevant to considerations of post-glacial chemical weathering rates (Szramek and Walter, Reference Szramek and Walter2004), orogen-scale carbonate weathering rates (Erlanger et al., Reference Erlanger, Rugenstein, Bufe, Picotti and Willett2021; Bufe et al., Reference Bufe, Cook, Galy, Wittmann and Hovius2022), and the efficacy of enhanced weathering in carbonate-rich till (Knapp and Tipper, Reference Knapp and Tipper2022). Back-precipitation of detrital carbonate as pedogenic carbonate may alter the timing of regional carbon cycling, potentially acting to slow the glacial-enhancement of chemical weathering and attendant CO2 sequestration that is a negative feedback on climate on medium (~thousand-year) timescales (Sharp et al., Reference Sharp, Tranter, Brown and Skidmore1995; Anderson et al., Reference Anderson, Drever, Frost and Holden2000; Williams et al., Reference Williams, Szramek, Jin, Ku and Walter2007), even if it does not alter the geologically long-term (~million year) sequestration (Berner et al., Reference Berner, Lasaga and Garrels1983).
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/qua.2024.41.
Acknowledgments
We thank the Edwin S. George Reserve for hosting our research activities, and especially acknowledge Alex Wenner's assistance in removing vegetation and excavating the slope and Nate Sanders as director at the time of our study. We acknowledge field assistance from Natalie Packard, Margaret Rudnick, Million Mengesha, and Jungpyo Hong. The Ann Arbor precipitation and Huron River water collections were initiated by Phoebe Aron and Alex Thompson and supported by Chris Poulsen. Many thanks to those who made the laboratory analyses possible: Ben Passey for assistance with clumped isotope measurements and all activities relating to the Isotopologue Paleosciences Laboratory, Kacey Lohmann and Lora Wingate for measuring the δ13C and δ18O values of carbonate, Jack Hutchings for measuring the δ18O/δ2H values of select water samples, and Diego Fernandez for measuring the Sr-isotope values. We acknowledge funding from the National Science Award Number 2122023 to Levin and Award Number 1854873 to Kelson. This manuscript was improved by comments from Brenden Fischer-Femal, Jeremy Rugenstein, and Associate Editor Kathleen Johnson.
Data availability
Clumped isotope (Δ47) data at the replicate-level for samples and standards are available from EarthChem (doi: https://doi.org/10.60520/IEDA/113108) and in Supplementary Table S2. Water isotope data will be available at the University of Utah Water Isotope Database upon publication: https://wateriso.utah.edu/waterisotopes/index.html and in Supplementary Table S3.