Gastroesophageal reflux disease (GERD) is frequently encountered by gastroenterologists, otorhinolaryngologists, surgeons and primary healthcare physicians(Reference Yim, Chiou and Ongkasuwan1). While GERD is primarily associated with lower oesophageal sphincter (LES) dysfunction, several other factors also contribute to its development(Reference Clarrett and Hachem2). The prevalence of GERD is significantly higher among individuals aged 50 years and older, smokers, chronic users of non-steroidal anti-inflammatory drugs, those with obesity(Reference Eusebi, Ratnakumaran and Yuan3) and individuals with lower education and income levels(Reference Nirwan, Hasan and Babar4).
When gastric contents ascend into regions above the oesophagus, laryngopharyngeal reflux may occur(Reference Johnston, Dettmar and Strugala5), potentially affecting the larynx, pharynx, paranasal sinuses and middle ear(Reference Lazarini and da Silva6). Recent evidence even links this condition to eye diseases(Reference Mayo-Yáñez, Viña-Vázquez and Lechien7). Left untreated, GERD can lead to complications ranging from erosive oesophagitis, bleeding and peptic strictures to pre-malignant and malignant lesions such as Barrett’s oesophagus and oesophageal adenocarcinoma(Reference Parasa and Sharma8).
Although proton pump inhibitors (PPI) alleviate symptoms in more than 70 % of GERD cases, a subset of patients do not achieve adequate relief(Reference Gyawali and Fass9). This suggests that components beyond HCl, such as pepsin, bile acids and trypsin, alongside, may play significant roles in GERD pathophysiology(Reference Li, Xu and Zhou10), in addition to factors such as low adherence to treatment and the presence of functional heartburn(Reference Mermelstein, Chait Mermelstein and Chait11). Concerns regarding the long-term use of PPI, particularly their effects on the absorption and homeostasis of key micronutrients (e.g. vitamin B12, Ca, Fe and Mg), have further complicated GERD management(Reference Fossmark, Martinsen and Waldum12).
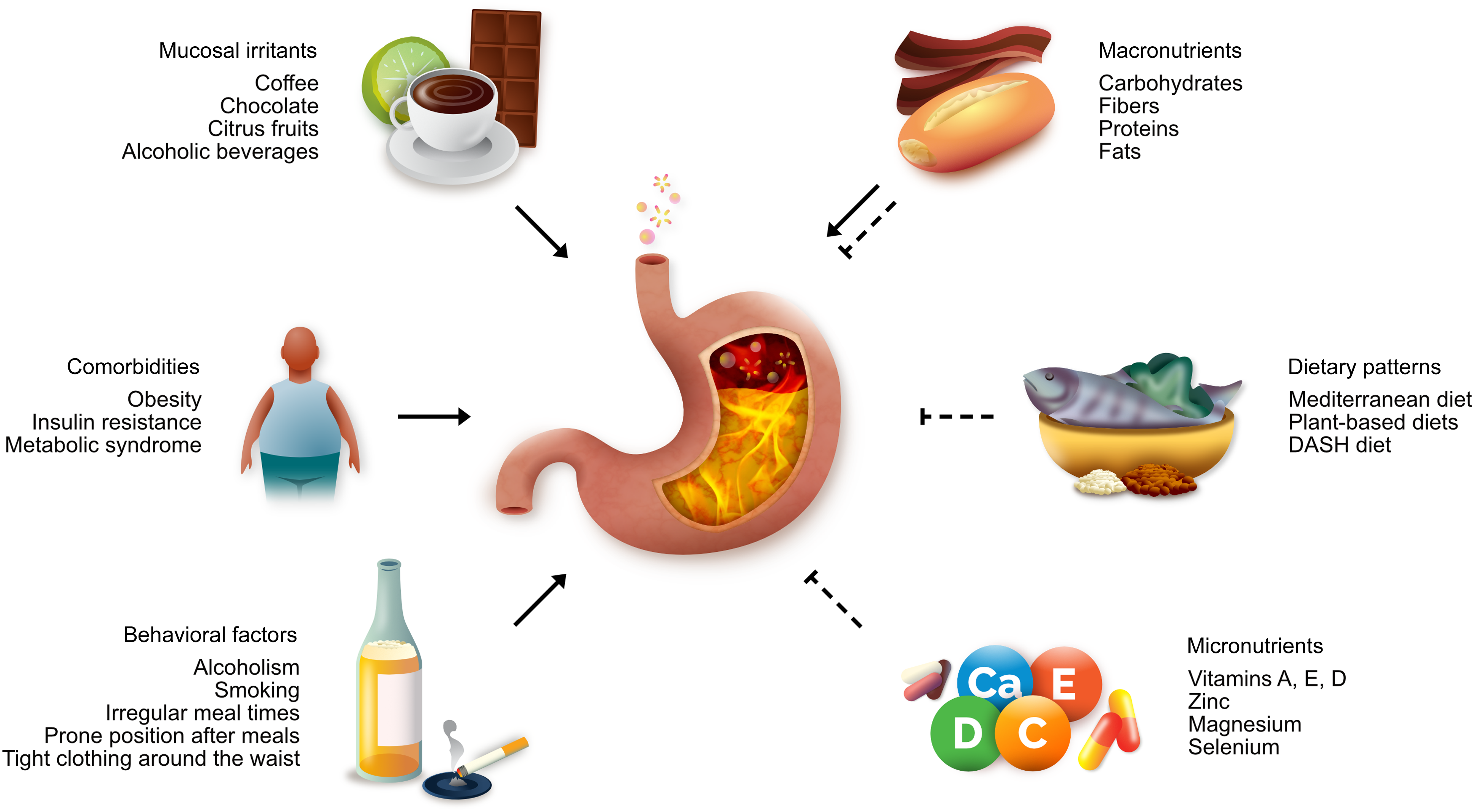
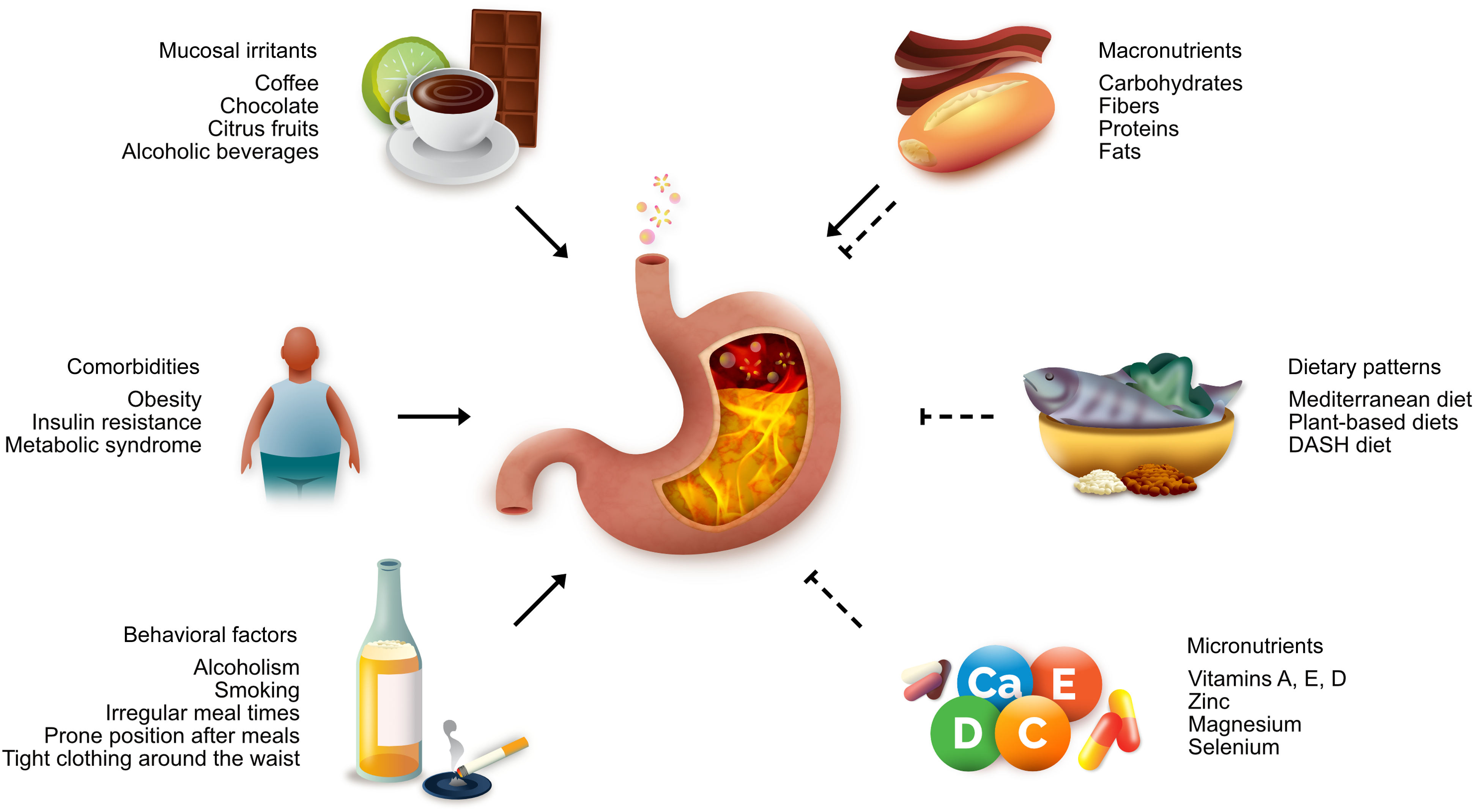
Given these complexities, non-pharmacological approaches, particularly dietary and nutritional interventions, have gained increasing attention in GERD management. This is partly driven by the observation that mucosal irritants in gastroduodenal contents are secreted in response to food intake and can be modulated by altering the diet’s nutritional composition (Figure 1)(Reference Newberry and Lynch13). Additionally, the strong association between obesity and GERD has further emphasised the role of dietary interventions, as addressing obesity through healthy dietary patterns not only aids in weight management but also directly contributes to GERD symptom control(Reference Nirwan, Hasan and Babar4,Reference Hwalla and Jaafar14) .
Nutritional and lifestyle factors associated with GERD.

Pathophysiology of gastroesophageal reflux disease and oesophageal mucosal injury
At the oesophagus–stomach junction lies the circular smooth muscle structure called the LES(Reference Hershcovici, Mashimo and Fass15). During swallowing, mechanoreceptors in the pharynx stimulate the LES to relax, allowing the food bolus to enter the stomach. Subsequently, the LES contracts to prevent the retrograde flow of stomach contents into the oesophagus and underlying regions(Reference de Carlos, Cobo and Macías16,Reference Sidhu and Lopoulos17) . This complex regulatory process is mediated by the vagus nerve(Reference Chen18) and also responds to the hormonal action of gastrin and cholecystokinin (CCK). Gastrin increases LES tone(Reference Rattan, Coln and Goyal19,Reference Wright and Castell20) , while CCK promotes its relaxation(Reference Babaei and Mittal21).
Under normal conditions, LES’s tonic pressure surpasses that of the stomach, effectively preventing the reflux of gastric contents(Reference Mittal, Fisher and McCallum22). However, dysfunction in this protective mechanism allows the retrograde flow of gastric or gastroduodenal contents into the distal portions of the oesophagus and, in cases of laryngopharyngeal reflux, into the laryngopharynx, oropharynx or even the nasopharynx, triggering the characteristic symptoms of this condition. This is because these regions lack the protective mechanisms of the stomach, making them vulnerable to mucosal injury from gastric juice, digestive enzymes and other irritants(Reference Argüero and Sifrim23–Reference Runggaldier, van Schie and Marti25).
Reflux is classified based on pH(Reference Jodorkovsky, Katzka and Gyawali26)pH falls below 4, characterised by a predominance of HCl. Reflux with a pH between 4 and 7, containing mixed contents, is classified as lightly acidic reflux. Lastly, slightly alkaline reflux is defined by a pH above 7, where the gastroduodenal content is primarily composed of pepsin and bile acids(Reference Boeckxstaens and Smout27,Reference Kowalik and Krzeski28) .
The oesophageal mucosa responds to acid injury by increasing bicarbonate secretion via carbonic anhydrase-III activity, in an attempt to neutralise the acid(Reference Nortunen, Väkiparta and Parkkila29). However, HCl downregulates the expression of E-cadherin, a transmembrane glycoprotein essential for cellular junction integrity, resulting in increased intercellular permeability and, thus, damage to the oesophageal mucosa(Reference Jovov, Que and Tobey30). Increased proton pump expression in oesophageal tissue may also contribute to local acid secretion, causing inflammation, mitochondrial damage and, ultimately, carcinogenesis(Reference Stabenau, Samuels and Lam31).
Pepsins, a group of proteases secreted by gastric chief cells, are released in their inactive precursor form, pepsinogen(Reference Stanforth, Wilcox and Chater32). Under acidic conditions, pepsinogen is converted into its active form through the cleavage of acid-labile bonds, initiating protein digestion(Reference Al-Janabi, Hartsuck and Tang33). This enzyme, along with other gastric contents, can damage the mucous membranes and epithelial barrier it contacts, digesting intercellular connections(Reference Kowalik and Krzeski28).
Another mechanism by which pepsin can perpetuate its harmful effects is by reducing carbonic anhydrase-III levels, which play a fundamental role in local protection against the deleterious effects of stomach acid content(Reference Gill, Johnston and Buda34). Carbonic anhydrase-III promotes the secretion of bicarbonate, leading to the alkalinisation of the oesophageal environment and the consequent deactivation of pepsin activity(Reference Bardhan, Strugala and Dettmar35). Thus, decreased levels of carbonic anhydrase-III may favour the action of pepsin by maintaining an acidic pH favourable to its action(Reference Stanforth, Wilcox and Chater32). Although pepsin operates optimally at a pH of 2–3·2, it remains active at pH levels of 6–7·2, values compatible with those of the oral cavity and respiratory tract (whose pH is around 6·4–7·2). This means that pepsin can inflict damage even in non-acidic environments(Reference Kowalik and Krzeski28).
Pepsin can also negatively regulate E-cadherin levels and increase the release of β-catenin into the cytoplasm, thereby increasing the risk of tumour cell infiltration and metastasis(Reference Yin, Zhang and Zhong36,Reference Lyros, Rafiee and Nie37) . This occurs because β-catenin accumulated in the cytoplasm can migrate to the nucleus and promote the transcription of various oncogenes associated with carcinogenesis and tumour progression through the Wnt/β-catenin pathway(Reference Yu, Yu and Li38,Reference Jung and Park39) . Furthermore, pepsin can be reactivated in acidic environments or in cells with low pH. In these circumstances, pepsin is internalised by cells through endocytosis, stored in vesicles and transported to organelles like the Golgi complex, causing mitochondrial damage and promoting the expression of genes related to carcinogenesis(Reference Bardhan, Strugala and Dettmar35). In summary, the harmful effects of pepsin on the oesophageal mucosa include increased intercellular permeability, local accumulation of reactive oxygen species, oxidative stress, inflammation, mitochondrial injury and an increased risk of neoplastic development(Reference Kowalik and Krzeski28,Reference Bardhan, Strugala and Dettmar35,Reference Yu, Yu and Li38,Reference Jung and Park39) .
Bile acid reflux is another contributor to inflammatory damage of the oesophageal mucosa(Reference Shi, Chen and Yang40). Physiologically, the function of bile acids secreted together with bile is to facilitate the digestion and absorption of fats and fat-soluble nutrients in the small intestine(Reference Shulpekova, Shirokova and Zharkova41). However, when refluxed, bile acids can induce epithelial-to-mesenchymal cell transformation via vascular endothelial growth factor signalling(Reference Zhang, Agoston and Pham42) and NF-κB activation, leading to local inflammation and the abnormal expression of tumour factors(Reference Sasaki, Hajek and Doukas43,Reference Zhang, Ran and Wu44) . Additionally, trypsin activates the protease-2 receptor, inducing the secretion of IL-8, a neutrophil chemotactic factor involved in the inflammatory response(Reference Kandulski, Wex and Mönkemüller45,Reference Yoshida, Katada and Handa46) . These mechanisms collectively increase oxidative stress(Reference Lee, Oh and Ahn47) and pro-inflammatory cytokine expression in the oesophageal mucosa(Reference Yoshida, Katada and Handa46).
Certain factors are known to decrease LES tone or increase intra-abdominal pressure, contributing to acid-gastric reflux. These include alcohol consumption(Reference Grad, Abenavoli and L Dumitrascu48) and tobacco use(Reference Pandolfino and Kahrilas49), obesity(Reference Yang, Lin and Li50), particularly abdominal obesity(Reference Zhan, Yuan and Zhao51), central nervous system depressants(Reference MacFarlane52), pregnancy(Reference Ali, Hassan and Egan53), hiatal hernia(Reference Sfara and Dumitrașcu54), delayed gastric emptying(Reference Fass, McCallum and Parkman55) and increased gastric volume(Reference Newberry and Lynch13).
The impact of obesity
Obesity is an independent risk factor for GERD(Reference Nirwan, Hasan and Babar4,Reference Emerenziani56) and its associated complications, such as erosive oesophagitis and oesophageal adenocarcinoma(Reference Hampel, Abraham and El-Serag57). In individuals with GERD, higher BMI correlates with increased frequency and severity of pyrosis, regurgitation and oesophagitis(Reference Nocon, Labenz and Jaspersen58). In fact, more than one-third of patients with overweight report GERD symptoms proportional to their BMI, with improvement in symptoms observed following weight loss. These findings support the role of obesity treatment in managing GERD(Reference Kröner, Cortés and Lukens59).
A study with thirty-four participants with overweight and GERD symptoms found a significant association between weight loss and symptom improvement, leading the authors to recommend weight loss as a first-line treatment(Reference Fraser-Moodie, Norton and Gornall60). Another study, involving 10 545 women, detected that even those with a baseline BMI within the normal range had an increased risk of frequent reflux symptoms with a BMI increase of more than 3·5 kg/m2(Reference Jacobson, Somers and Fuchs61).
The relationship between abdominal obesity and increased risk of oesophagitis was investigated in a meta-analysis of forty-two observational studies, which found a significant association between abdominal obesity and oesophagitis, especially with waist circumference exceeding 87 cm(Reference Zhan, Yuan and Zhao51). Even in individuals with a normal BMI, abdominal obesity was associated with a higher risk of oesophageal adenocarcinoma.(Reference O’Doherty, Freedman and Hollenbeck62) These findings highlight the impact of not only excess weight, as assessed through BMI, but also body fat distribution on GERD(Reference Kröner, Cortés and Lukens59).
Several mechanisms by which obesity contributes to reflux have been proposed, including mechanical and humoral factors as well as gastrointestinal motility disorders. Among these mechanisms, increased intra-abdominal pressure combined with LES relaxation and prolonged exposure of the oesophageal mucosa to gastric acid content are significant(Reference Emerenziani56).
GERD is often associated with other components of the metabolic syndrome, such as type 2 diabetes mellitus(Reference Sun63) and metabolic dysfunction-associated steatotic liver disease(Reference Fukunaga, Mukasa and Nakano64). Although the exact causal mechanism remains unclear, insulin resistance, a hallmark of obesity, appears to play an important role in GERD pathophysiology. A positive relationship has been demonstrated between HOMA-IR values, components of metabolic syndrome and GERD symptoms, with higher insulin resistance correlating with increased severity of GERD symptoms and a higher risk of erosive oesophagitis(Reference Hsu, Wang and Chen65,Reference Budiyani, Purnamasari and Simadibrata66) . Furthermore, gastroparesis, a feature of autonomic neuropathy caused by poorly controlled type 2 diabetes mellitus(Reference Krishnasamy and Abell67), can predispose individuals to GERD symptoms(Reference Egboh and Abere68). Conversely, weight loss not only reduces GERDF symptoms but also improves insulin resistance, with a reduction of at least 5 % promoting improvements in hepatic and muscular insulin sensitivity and pancreatic β-cell function. Greater benefits are observed with weight losses above 5 %, following a dose-response relationship(Reference Magkos, Fraterrigo and Yoshino69).
The role of macronutrients
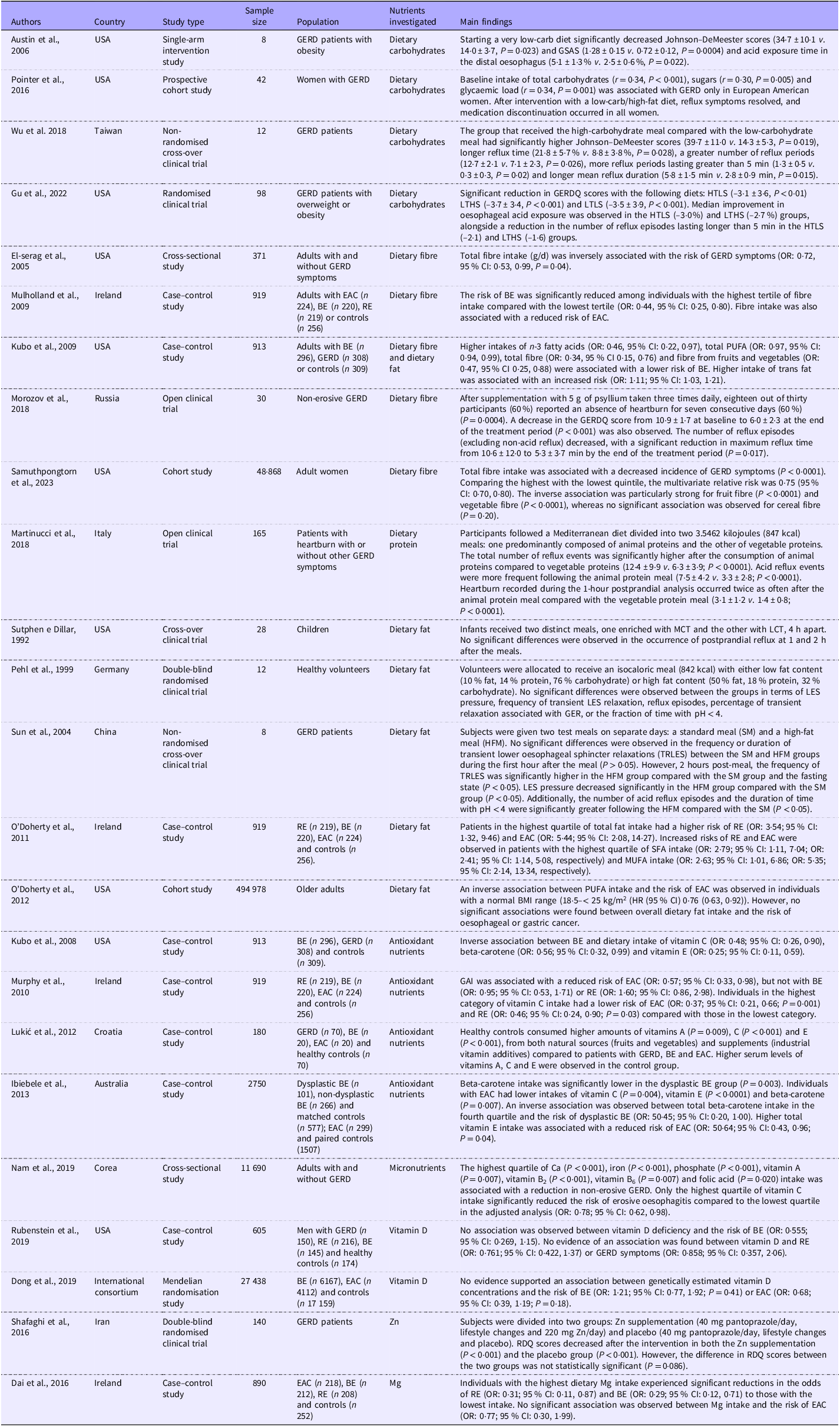
The appropriate distribution of macronutrients in the diet can play a fundamental role in controlling GERD symptoms. As described earlier, gastroduodenal content contains several mucosal irritants whose secretion depends on food intake and can be altered by changes in the nutritional composition of the diet(Reference Newberry and Lynch13). Table 1 provides a summary of studies that have evaluated the role of dietary components in GERD.
Summary of studies evaluating the impact of macronutrients, micronutrients and bioactive compounds on GERD and related conditions

GERD, gastroesophageal reflux disease; GSAS, Gastroesophageal Reflux Disease Symptom Assessment Scale; GERDQ, Gastroesophageal Reflux Disease Questionnaire; HTLS, high-total/low-simple carbohydrates; LTHS, low-total/high-simple carbohydrates; LTLS, low-total/low-simple carbohydrates; LES, lower oesophageal sphincter; GER, gastroesophageal reflux; TRLES, transient relaxation of the lower oesophageal sphincter; SM, standard meal; HFM, high-fat meal; EAC, oesophageal adenocarcinoma; BE, Barrett’s oesophagus; RE, reflux oesophagitis; MCT, medium-chain TAG; LCT, long-chain TAG; LCGAI, general antioxidant index; RDQ, Reflux Disease Questionnaire.
Carbohydrates and fibre
While the intake of simple sugars and starch can exacerbate reflux symptoms, dietary fibre has shown protective and therapeutic effects. These findings underscore the importance of carbohydrate quality(Reference Newberry and Lynch13). Wu and colleagues(Reference Wu, Kuo and Yao70) investigated the effect of dietary carbohydrates on GERD. Twelve patients diagnosed with GERD were given 500 ml of a liquid meal with identical protein and fat content but varying carbohydrate levels: one group received 84·8 g of carbohydrates, while the other received 178·8 g. Individuals with higher carbohydrate intake had worse symptom scores, longer total reflux times and more frequent reflux episodes.
A study involving participants with obesity and GERD found that a very low-carbohydrate diet, starting with less than 20 g/d, decreased oesophageal exposure to gastric acid and reduced symptoms(Reference Austin, Thiny and Westman71). The effects of a low-carbohydrate diet were also evaluated in a pilot study with forty-two women with obesity, showing reduced GERD symptoms and medication use(Reference Pointer, Rickstrew and Slaughter72). In a prospective, randomised, single-blind, controlled dietary intervention study involving ninety-eight individuals, reducing carbohydrate intake – particularly simple sugars – improved pH monitoring results and GERD symptoms(Reference Gu, Olszewski and King73).
A recent meta-analysis assessing the effectiveness of dietary interventions in GERD showed that low-carbohydrate diets significantly reduced oesophageal acid exposure time(Reference Lakananurak, Pitisuttithum and Susantitaphong74). Additionally, a higher glycaemic index was associated with an increased risk of oesophageal adenocarcinoma, with each 10-unit increase in glycaemic index amplifying the risk(Reference Mulholland, Cantwell and Anderson75). The effect of carbohydrates on GERD symptoms is primarily attributed to their ability to reduce LES tone(Reference Newberry and Lynch13).
Regarding dietary fibre, El-Serag and colleagues(Reference El-Serag76) demonstrated an inverse association between higher fibre intake and GERD symptoms. Similarly, Mulholland and colleagues(Reference Mulholland, Cantwell and Anderson75) found that increased fibre intake was associated with a decreased risk of Barrett’s oesophagus and oesophageal adenocarcinoma. Importantly, fibre source appears to play an important role, with fibre from fruits and vegetables being associated with a lower risk of Barrett’s oesophagus, whereas no significant association was observed for fibre from other sources(Reference Kubo, Block and Quesenberry77). A cohort study from the Nurses’ Health Study II reinforced these findings, showing that higher total fibre intake was associated with a decreased incidence of GERD symptoms, with the strongest associations observed for fibre from fruits and vegetables, but not from cereals(Reference Samuthpongtorn, Mehta and Ma78).
Supplementation with 15 g/d of psyllium significantly improved reflux symptoms by increasing LES resting pressure and decreasing both the number of GER episodes and the frequency of pyrosis. Participants in this study had an average baseline fibre intake of 6 g/d, and the supplementation brought their intake closer to dietary reference values. This highlights the importance of adjusting fibre consumption in individuals whose intake falls below the recommended guidelines(Reference Morozov, Isakov and Konovalova79).
Protein
The role of dietary protein in GERD is not well described in the scientific literature, with studies yielding heterogeneous results(Reference Zhang, Hou and Huang80). While the exact relationship between protein intake and GERD symptoms is uncertain(Reference Newberry and Lynch13), some evidence suggests that high-protein diets may reduce reflux symptoms(Reference Kröner, Cortés and Lukens59). The presence of oligopeptides from protein digestion in the stomach stimulates gastrin release, which enhances LES constriction(Reference Zeng, Ou and Wang81–Reference Soll and Walsh83). Of note, plant-based proteins are associated with fewer reflux episodes, particularly acid reflux and a reduced number of symptoms in the first postprandial hour(Reference Martinucci, Guidi and Savarino84).
Lipids
In addition to being energy-dense, dietary fats require the secretion of mucosal irritants, such as bile salts and hormonal mediators like CCK, for digestion and absorption(Reference Newberry and Lynch13). CCK plays a multifaceted role in GERD pathophysiology: it inhibits gastric emptying, promotes gallbladder contraction and relaxes the LES, thereby contributing to reflux symptoms(Reference Newberry and Lynch13,Reference Koeppen and Stanton85) .
Comparing individuals with and without GERD symptoms, those with reflux had higher daily intakes of total fat, saturated fat and cholesterol. A dose–response correlation was observed between fat and cholesterol intake and GERD risk. However, after adjusting for BMI, the impact of dietary fat on GERD became statistically nonsignificant(Reference El-Serag76).
A systematic review by Zhang et al. (Reference Zhang, Hou and Huang80) assessed the influence of dietary and lifestyle factors on GERD and found a significant correlation between high-fat diets and reflux. Nevertheless, a literature review by Heidarzadeh-Esfahani et al. (Reference Heidarzadeh-Esfahani, Soleimani and Hajiahmadi86) revealed highly variable outcomes among studies investigating the effect of dietary fat on GERD.
Pehl et al. (Reference Pehl, Waizenhoefer and Wendl87) examined the effects of an isocaloric liquid-solid meal with low (10 %) or high-fat content (50 %) on LES motility and GER in healthy individuals. The study found no discernible differences across the analysed parameters, including LES pressure, frequency of transient LES relaxation, reflux episodes, percentage of transient relaxation with GER and fraction of time at pH < 4. In contrast, a study by Sun et al. (Reference Sun, Ke and Wang88) assessed the impact of two test meals (standard v. high-fat) in individuals with GERD. They found a notable increase in transient LES relaxation frequency following both meals, with no substantial differences observed within the initial hour. However, 2 hours after consuming the high-fat meal, they observed a significant increase in transient LES relaxations, acid reflux episodes and prolonged periods of pH < 4, alongside a reduction in LES pressure.
It is important to highlight that the impact of different types and sources of fats on GERD symptoms can vary. For example, PUFA, particularly n-3 fatty acids, are associated with a lower risk of Barrett’s oesophagus, while increased consumption of trans fats has been linked to an elevated risk(Reference Kubo, Block and Quesenberry77). Additionally, PUFA may exhibit a protective effect against adenocarcinoma, especially in individuals with normal BMI(Reference O’Doherty, Freedman and Hollenbeck89). On the other hand, medium-chain TAG, unlike long-chain fatty acids, do not require bile acids for digestion and absorption and do not stimulate CCK secretion(Reference Symersky, Vu and Frölich90–Reference Hopman, Jansen and Rosenbusch92), which can be advantageous for managing GERD. Furthermore, medium-chain TAG intake, even in the presence of long-chain fatty acids, inhibits CCK secretion and gallbladder contraction, potentially reducing reflux symptoms(Reference Murata, Harada and Kishino93). However, these benefits require further confirmation(Reference Sutphen and Dillard94).
Micronutrients and bioactive compounds in modulating the antioxidant and inflammatory response
Dietary micronutrients and bioactive compounds are important modulators of antioxidant(Reference Halliwell95) and anti-inflammatory(Reference Gothai, Ganesan and Park96) responses. As previously described, oxidative stress and inflammation play critical roles in GERD(Reference Lee, Oh and Ahn47). Individuals with GERD have significantly lower levels of antioxidant enzymes, such as superoxide dismutase, glutathione peroxidase and catalase, underscoring the importance of reducing oxidative stress in managing this condition(Reference Bulut, Tetiker and Çelikkol97). Nutrients capable of modulating antioxidant and anti-inflammatory pathways are therefore important(Reference Herdiana98).
Retinoids and carotenoids
Retinoic acid, an active metabolite of vitamin A, promotes homeostasis and mitigates inflammatory responses in mucous membranes and tissues by increasing the expression of IL-10 and IL-22(Reference Oliveira, Teixeira and Sato99), two key anti-inflammatory cytokines(Reference Keir, Yi and Lu100,Reference Wei, Wang and Li101) . Carotenoids, some of which are vitamin A precursors, exhibit significant anti-inflammatory and antioxidant effects. These pigments become more bioavailable when consumed with lipids and when plant cell walls are broken down during preparation, such as through heating(Reference Maoka102,Reference Hammond and Renzi103) .
A study evaluating the consumption and serum levels of antioxidant vitamins, including vitamin A, found a relationship between serum vitamin levels and the severity of reflux disease ((Reference Lukic, Segec and Segec104). Nam and colleagues(Reference Nam, Park and Cho105) reported that a high intake of vitamin A and retinol was associated with a 22 % and 27 % reduction in the risk of non-erosive reflux disease, respectively, although no association was observed with erosive oesophagitis(Reference Nam, Park and Cho105). Beta-carotene intake has also been inversely associated with Barrett’s oesophagus(Reference Kubo, Levin and Block106,Reference Ibiebele, Hughes and Nagle107)
Vitamin D
Vitamin D is crucial for immune regulation and proper mucosal function(Reference Sun108). Its active metabolite, 1α,25-dihydroxy-vitamin D, modulates the inflammatory response by suppressing pro-inflammatory cytokines(Reference Martens, Gysemans and Verstuyf109) and upregulating anti-inflammatory ones(Reference Calton, Keane and Newsholme110). It inhibits NF-κB activity and increases the expression of NF-κB inhibitor, resulting in reduced expression of pro-inflammatory genes responsible for IL-6, IL-8, TNF-α and COX-2, thereby reducing prostaglandin levels(Reference Hassanshahi, Anderson and Sylvester111–Reference Liu, Zhang and Xu113). Additionally, Vitamin D acts as an antagonist in the Wnt/β-catenin pathway(Reference Shah, Islam and Dakshanamurthy114), interfering with the expression of genes linked to carcinogenesis(Reference El-Sharkawy and Malki115). It has also been shown to enhance the expression of the transmembrane glycoprotein E-cadherin(Reference Zhang, Wu and Sun116).
Despite these promising mechanisms, no consistent association was found between vitamin D levels and the presence of Barrett’s oesophagus, erosive oesophagitis, or GERD symptoms(Reference Rubenstein, McConnell and Beer117). A Mendelian randomisation study also found no link between vitamin D status and the risk of Barrett’s oesophagus or oesophageal adenocarcinoma ((Reference Dong, Gharahkhani and Chow118)). However, the vitamin D receptor may be overexpressed in precancerous lesions, especially in males(Reference Zhou, Xia and Bandla119). Individuals with polymorphisms in the vitamin D receptor gene associated with reduced receptor expression in oesophageal tissue have been found to have lower incidences of reflux oesophagitis, Barrett’s oesophagus and oesophageal adenocarcinoma(Reference Janmaat, van de Winkel and Peppelenbosch120). Further studies are needed to elucidate the role of vitamin D in GERD.
Vitamin E
Vitamin E, a lipid-soluble vitamin, functions as a primary antioxidant in cellular membranes, scavenging free radicals and preventing lipid peroxidation(Reference Herrera and Barbas121). It also exhibits significant anti-inflammatory effects(Reference Asbaghi, Sadeghian and Nazarian122), including the modulation of eicosanoids and the suppression of NF-κB, IL-6 and IL-8(Reference Jiang123). Its major dietary sources include vegetable oils, nuts and certain cereals(Reference Zaaboul and Liu124).
Although studies in experimental models have shown vitamin E’s beneficial effects(Reference Rao and Vijayakumar125,Reference Hao, Zhang and Liu126) , its role in preventing GERD-related complications in humans remains inconclusive. For instance, Kubo et al. (Reference Kubo, Levin and Block106) found that individuals in the highest quartile of vitamin E intake had a lower risk of Barrett’s oesophagus. However, Murphy et al. (Reference Murphy, Anderson and Ferguson127) reported no association between vitamin E intake and reflux oesophagitis, Barrett’s oesophagus or oesophageal adenocarcinoma.
Vitamin C
Vitamin C is a potent antioxidant due to its high electron-donating capacity(Reference Padayatty, Katz and Wang128). It regenerates vitamin E from the tocoferoxyl radical formed by α-tocopherol interaction with lipid peroxides in membranes(Reference Niki129). Furthermore, as a cofactor, vitamin C maintains proper epithelial barrier function(Reference Carr and Maggini130).
Nam et al. (Reference Murphy, Anderson and Ferguson127) reported that individuals in the highest quartile of vitamin C had a 22 % lower risk of erosive oesophagitis. Similar findings were described by Wu et al. (Reference Wu, Zhao and Ai131). Furthermore, Murphy et al. (Reference Nam, Park and Cho105) demonstrated that a higher dietary intake of vitamin C was associated with a reduced risk of oesophageal adenocarcinoma.
Zinc
Zn regulates the antioxidant response through multiple mechanisms. Low Zn levels correlate with increased cellular oxidants, disruptions in antioxidant defense and elevated markers of tissue oxidative stress(Reference Lee132). Additionally, Zn modulates the activity of glutathione, the most important low molecular weight antioxidant in cells,(Reference Oteiza133,Reference Forman, Zhang and Rinna134) and serves as a cofactor for numerous enzymes involved in cellular repair(Reference Lin, Sermersheim and Li135). Zn deficiency has been associated with increased expression of inflammatory factors in the pathogenesis of oesophageal cancer(Reference Taccioli, Chen and Jiang136), a process that can be reversed with supplementation(Reference Ma, Li and Fang137). In individuals with GERD, low Zn levels may pose an additional risk factor for oesophageal cancer(Reference Liu, Liang and Jin138). However, Zn supplementation in patients with GERD did not affect the severity of symptoms ((Reference Shafaghi, Hasanzadeh and Mansour-Ghanaei139)).
Selenium
Se’s primary functions are antioxidant(Reference Kiełczykowska, Kocot and Paździor140), involving enzymes that maintain redox homeostasis, a process influenced by its organic status(Reference Mehdi, Hornick and Istasse141). Additionally, Se’s exhibits chemopreventive properties(Reference Ahsan, Liu and Su142). In line with this, Cai et al. (Reference Cai, Wang and Yu143), in their meta-analysis investigating the relationship between Se exposure and the risk of various types of cancers, demonstrated an association between this micronutrient and a reduced risk of oesophageal cancer. However, studies specifically evaluating Se intake or supplementation in patients with GERD are limited.
Magnesium
Mg is the fourth most abundant mineral in the body, influencing directly and indirectly approximately 800 metabolic reactions(Reference Gröber, Schmidt and Kisters144,Reference de Baaij, Hoenderop and Bindels145) . Mg deficiency is associated with increased inflammation and oxidative stress(Reference Nielsen146). Due to its involvement in DNA and RNA synthesis, as well as mitochondrial membrane stabilisation, Mg may play a crucial role in cellular repair processes and the resolution of inflammation in GER(Reference Volpe147). Individuals with the highest dietary Mg intake had significantly reduced odds of reflux oesophagitis and Barrett’s oesophagus compared with those with the lowest intake. However, no significant association was observed between Mg intake and the risk of oesophageal adenocarcinoma ((Reference Dai, Cantwell and Murray148)).
Bioactive compounds
Dietary bioactive compounds or phytochemicals are substances produced by the secondary metabolism of plants in response to environmental stressors(Reference Kumar, P and Kumar149). Within the human body, these compounds can modulate various metabolic pathways, acting as direct antioxidants(Reference Zhang, Gan and Li150) and influencing the expression or activity of antioxidant enzymes(Reference Lee and Park151). They also play a beneficial role in regulating inflammatory pathways(Reference Zhu, Du and Xu152).
The cytoprotective effects of dietary bioactive compounds are partly mediated through the activation of the transcription factor NRF2(Reference Eggler and Savinov153). When activated, NRF2 induces the expression of key antioxidant enzymes, including superoxide dismutase, glutathione peroxidase and peroxiredoxin while simultaneously downregulating NF-κB-mediated expression of pro-inflammatory cytokines(Reference Saha, Buttari and Panieri154). This dual action reduces oxidative stress and inflammation, protecting the gastrointestinal mucosa from damage. Indeed, several phytochemicals have been shown to upregulate the expression of antioxidant enzymes, providing protection against oxidative damage to the gastrointestinal mucosa(Reference Cheng, Lu and Yen155). A prominent example is curcumin, which inhibits the NF-κB signalling pathway activated by bile acids and genes associated with carcinogenesis in human hypopharyngeal cells(Reference Vageli, Doukas and Spock156). However, it is important to note that curcumin exhibits a potent cholecystokinetic effect, with a 40 mg dose causing up to a 50 % contraction of the gallbladder(Reference Rasyid, Rahman and Jaalam157). This highlights the need for consideration of individual tolerance and clinical context when recommending curcumin supplementation.
The role of dietary patterns in gastroesophageal reflux disease
Dietary patterns play a crucial role in the risk and management of non-communicable chronic diseases, as evidenced by multiple observational and intervention studies(Reference Wang, Song and Eliassen158,Reference Schulze, Martínez-González and Fung159) . Given that obesity is closely linked to GERD, dietary pattern-focused interventions have gained increasing importance in addressing this condition(Reference Hwalla and Jaafar14).
A Western dietary pattern, characterised by a high intake of saturated fats, refined grains, sugar, salt, alcohol and other harmful components, along with reduced consumption of fruits and vegetables(Reference García-Montero, Fraile-Martínez and Gómez-Lahoz160), has been linked to reflux(Reference Khodarahmi, Azadbakht and Daghaghzadeh161). In contrast, dietary patterns such as the Mediterranean diet, rich in fruits, vegetables, whole grains and unsaturated fats(Reference Davis, Bryan and Hodgson162), may offer potential benefits for GERD(Reference Özenoğlu, Anul and Özçelikçi163).
Adherence to anti-inflammatory diets, like the Mediterranean diet, has been shown to reduce the risk of non-communicable chronic diseases(Reference Ahmad, Moorthy and Demler164), while pro-inflammatory dietary patterns increase these risks(Reference Shivappa165). In the context of GERD, adherence to a pro-inflammatory diet has been correlated with increased risks of reflux oesophagitis, Barrett’s oesophagus(Reference Shivappa, Hebert and Anderson166) and oesophageal adenocarcinoma(Reference Marx, Veronese and Kelly167). This is likely mediated by the upregulation of the inflammation–metaplasia–adenocarcinoma pathway in oesophageal carcinogenesis(Reference Shivappa, Hebert and Anderson166).
Similarly, the dietary approaches to stop hypertension diet, which emphasises a high intake of fruits and vegetables, low-fat dairy, reduced saturated and total fat and low cholesterol, along with moderate consumption of whole grains, nuts, poultry and fish, have proven effective in managing non-communicable chronic diseases(Reference Suri, Kumar and Kumar168) Evidence suggests that it may also benefit GERD patients. For instance, a cross-sectional study involving 5141 adolescents aged 13–14 years found that those with higher adherence to the dietary approaches to stop hypertension diet were less likely to develop GERD(Reference Beigrezaei, Sasanfar and Nafei169).
In addition, various studies have explored the impact of specific food groups on GERD prevalence. A cross-sectional study of 1146 participants compared adherence to an omnivorous diet v. a vegan diet and found a twofold higher prevalence of GERD among those following an omnivorous diet, suggesting that a diet high in animal-derived foods may increase GERD risk(Reference Baroni, Bonetto and Solinas170). Supporting this notion, a case–control study conducted among Irish adults examined the associations between fat and meat consumption and the risks of reflux oesophagitis, Barrett’s oesophagus and oesophageal adenocarcinoma. The study revealed that participants in the highest quartile of fresh red meat consumption faced a significantly greater risk of oesophageal adenocarcinoma, whereas those in the highest quartile of processed meat consumption had a higher risk of reflux oesophagitis(Reference O’Doherty, Cantwell and Murray171). In contrast, with respect to dairy, no significant differences in common GERD symptoms such as heartburn and acid regurgitation were observed between individuals consuming higher amounts of full- or low-fat dairy (3 servings/day) and those following a diet with limited dairy intake(Reference Fernando, Schmidt and Cromer172).
The efficacy of dietary interventions like the low-FODMAP diet has also been explored. Rivière et al. (Reference Rivière, Vauquelin and Rolland173) found no significant advantage of this diet compared with standard dietary counselling in GERD treatment. However, in individuals with irritable bowel syndrome overlapping with GERD, high-FODMAP meals were associated with a higher frequency of symptoms compared to low-FODMAP meals(Reference Plaidum, Patcharatrakul and Promjampa174). Similarly, Patcharatrakul et al. (Reference Patcharatrakul, Linlawan and Plaidum175) demonstrated that postprandial reflux symptoms were more pronounced after consuming wheat noodles (high in FODMAP) compared with rice noodles (low in FODMAP). These results support the utility of low-FODMAP diets for individuals with overlapping irritable bowel syndrome and GERD, emphasising the need for personalised nutritional interventions based on individual food sensitivities and intolerances.
Additionally, histamine-free diets have shown promise in managing laryngopharyngeal reflux symptoms. A case study reported substantial improvements in symptoms, such as persistent cough and throat clearing, in a patient who underwent Nissen fundoplication and followed a histamine-free diet. This suggests a potential link between laryngopharyngeal reflux and food sensitivities, particularly in patients unresponsive to standard treatment(Reference Alnouri, Cha and Sataloff176).
The role of oesophageal mucosal irritants
Several lifestyle and dietary risk factors have been implicated in GERD symptoms, with alcohol emerging as a significant contributor(Reference Taraszewska177). Alcohol exhibits a dose–response relationship with GERD risk, serving as a predisposing factor for symptom exacerbation(Reference Pan, Cen and Chen178). Consequently, individuals experiencing reflux symptoms after alcohol consumption are advised to limit their intake(Reference Hungin, Yadlapati and Anastasiou179). The direct contact of alcohol with the mucosal lining of the upper GI tract induces numerous metabolic and functional alterations, which may lead to a broad spectrum of acute and chronic ailments. Additionally, alcohol influences oesophageal motility by reducing LES tone, further predisposing individuals to reflux symptoms(Reference Bode and Bode180).
Similarly, coffee consumption has been extensively studied for its gastrointestinal effects(Reference Iriondo-DeHond, Uranga and del Castillo181). While coffee stimulates gastrin release–primarily through its caffeine content–thereby increasing gastric acid secretion, its components also reduce LES tone, potentially contributing to reflux(Reference Nehlig182). However, a meta-analysis investigating the association between coffee consumption and GERD risk yielded inconclusive results(Reference Kim, Oh and Myung183).
The role of carbonated beverages in GERD remains controversial(Reference Heidarzadeh-Esfahani, Soleimani and Hajiahmadi86). Cuomo et al. (Reference Cuomo, Savarese and Sarnelli184) found that carbonated and sweetened beverages did not significantly alter upper digestive tract physiology in healthy individuals. Johnson et al. (Reference Johnson, Gerson and Hershcovici185) further argued that these beverages neither directly cause oesophageal damage nor are consistently associated with GERD.
Citrus fruit consumption between meals has been linked to increased GERD(Reference Eslami, Shahraki and Bahari186). Some studies indicate that citrus increases the risk of GERD recurrence in individuals undergoing PPI treatment(Reference López-Colombo, Pacio-Quiterio and Jesús-Mejenes187). These findings were validated by a systematic review assessing the relationship between dietary habits and GERD risk(Reference Heidarzadeh-Esfahani, Soleimani and Hajiahmadi86). Although these effects are believed to be partially due to the reduction in oesophageal pH caused by citrus fruit consumption, a dietary strategy involving acidic pH foods has been associated with symptom reduction and even resolution(Reference Langella, Naviglio and Marino188).
Chocolate has also been investigated for its impact on GERD. While most studies systematically reviewed by Heidarzadeh-Esfahani et al. (Reference Heidarzadeh-Esfahani, Soleimani and Hajiahmadi86) found no direct association between chocolate consumption and GERD risk, chocolate was shown to significantly lower LES mean basal pressure(Reference Wright and Castell20). Moreover, in individuals with reflux oesophagitis, chocolate consumption significantly increased acid exposure during the first postprandial hour(Reference Murphy and Castell189). This effect is primarily attributed to methylxanthines, such as theobromine, which induces LES relaxation through a mechanism similar to caffeine(Reference Fredholm190).
In general, several foods have been reported to precipitate GERD symptoms(Reference Choe, Joo and Kim191), and the elimination of entire categories of foods or beverages is a common practice in primary care and gastroenterology clinics(Reference Newberry and Lynch13). However, studies have shown conflicting associations for most foods(Reference Heidarzadeh-Esfahani, Soleimani and Hajiahmadi86). Therefore, it is more prudent to recommend that dietary adjustments be made on a personalised basis, taking into account each patient’s individual response.
Behavioural measures associated with diet
Behavioural measures, in addition to dietary factors, play a significant role in GERD. A case-control study involving forty-seven GERD patients and 294 age- and sex-matched controls found that a shorter interval between dinner and bedtime (< 3 h) significantly increased GERD risk compared to longer intervals (≥ 4 h), even after adjusting for smoking, alcohol consumption and BMI(Reference Fujiwara, Machida and Watanabe192). Similar findings were confirmed in subsequent studies, which also associated behaviours such as skipping breakfast, midnight snacking, rapid eating and consuming very hot foods with higher GERD prevalence(Reference Zhang, Hou and Huang80).
Conclusion
Numerous studies have examined the impact of diet and specific nutritional components on GERD, but findings often remain inconclusive. Among macronutrients, carbohydrates – particularly refined sources – have been consistently linked to GERD. Conversely, dietary fibre from fruits and vegetables appears protective and even therapeutic. Overall, encouraging weight loss in individuals with overweight and obese, along with promoting adherence to healthy dietary patterns emphasising minimally processed plant-based foods, while reducing ultra-processed foods, refined carbohydrates and unhealthy fats, should be prioritised in GERD management.
Acknowledgements
This research received no specific grant from any funding agency or from commercial or not-for-profit sectors.
D. F. S. and P. M. L. M. conceptualised the article and developed the overall research approach. D. F. S., R. L. V. L. and P. M. L. M. conducted the investigation. D. F. S. curated the data and led the formal analysis with contributions from D. R. M., P. M. L. M. and P. C-B. Methodology was developed by D. F. S., R. L. V. L. and D. R. M. Project administration was overseen by D. F. S. and R. L. V. L.
P. C-B. supervised the project. D. F. S. and P. M. L. M. drafted the original manuscript, while R. L. V. L., D. R. M. and P. C-B. contributed to the review and critical editing of the manuscript. All authors have reviewed and approved the final manuscript and take full responsibility for its content.
The authors declare that there are no conflicts of interest.