Highlights
-
• Head trauma and early post-neurological-insult seizures are significant TLE risk factors in Canadian epilepsy clinic cohort
-
• Only 5.6% of TLE patients have a history of febrile seizures.
-
• Almost 50% of TLE patients exhibit unique EEG findings of mid/posterior temporal maximum of epileptiform discharges.
Introduction
Temporal lobe epilepsy (TLE) remains the most common type of focal onset epilepsy. This is especially true among people with pharmaco-resistant epilepsy.Reference Soeder, Gleissner and Urbach1,Reference Engel2 TLE is associated with significant morbidity and can severely impact the quality of life and functional outcome of those affected. Thus, understanding its antecedents and risk factors is crucial for early identification, prevention and improved management. While the understanding of this type of epilepsy is well-developed, with a substantial body of literature addressing its clinical and therapeutic dimensions, most work focuses on treatment outcomes and their determinants.Reference Rathore, George, Kesavadas, Sankara Sarma and Radhakrishnan3–Reference Uijl, Leijten, Arends, Parra, Van Huffelen and Moons5 Moreover, much of the published research has been conducted in surgical cohorts or outside of Canada, limiting their generalizability to the broader TLE population.
Despite the wealth of research on TLE, detailed evaluation of causal factors has not been adequately addressed. Between 1993 and 2022, eleven studies reported findings on one or a few risk factors for TLE, with sample sizes ranging between 30 and 484 patients.Reference Soeder, Gleissner and Urbach1,Reference Klein, Dingledine and Aronica6–Reference Özkara, Uzan and Benbir15 For instance, Harvey et al. in their paper published in 1997 found that 29% of the study’s pediatric population had experienced a significant illness preceding the onset of TLE, which included febrile status epilepticus, meningitis, respiratory arrest and head injury. In a 2019 prospective study, Tubi et al. identified early seizures (Occurring within 7 days) following traumatic brain injury (TBI) as a significant risk factor for the later development of post-traumatic epilepsy, predominantly TLE.
There remains a knowledge gap in understanding the prevalence and the relative contribution of specific risk factors within the TLE population in Canada and globally. This underscores the need for well-designed studies to systematically evaluate potential etiological risk factors and antecedents of TLE.Reference Rathore, George, Kesavadas, Sankara Sarma and Radhakrishnan3,Reference French, Williamson and Thadani7 Such clinical information might open the doors to investigate and develop treatment as well as preventive intervention strategies.Reference Klein, Dingledine and Aronica6 This study explores potential etiologic risk factors for TLE patients attending a Canadian epilepsy referral clinic. We compare this population to a control group with genetic generalized epilepsy (GGE) to better understand the specificity of these risk factors. Using a structured data collection template and including all patients with TLE attending the clinic over a three-year period, including non-surgical population, our study aimed at providing a more representative analysis of TLE risk factors. Key risk factors included head trauma, early seizures following acute neurological insults, neuroinfectious, febrile seizures and participation in outdoor sports.
Methods
Following approval from our institutional ethics board, this retrospective chart-review-based study was conducted using our epilepsy clinic database. Our comprehensive epilepsy center is part of an academic hospital group affiliated with Queen’s University in Kingston, Ontario, Canada.
In this retrospective study, consecutive patients from our epilepsy clinics who were diagnosed with TLE by a single senior epileptologist (GS), over a 3-year period (2018–2021), were included. The control group was consecutive patients diagnosed with GGE by the same epileptologist during the same period. Both diagnoses were based on a combination of detailed structured clinical evaluation, electroencephalographic (EEG) and neuroimaging findings.
TLE was diagnosed based on studies previously published by our group, primarily based on typical seizure semiology, including epigastric, olfactory or experiential aura, orofacial and/or manual automatisms and behavioral arrest, as well as history of antecedent illness followed by stuttering course and infrequent episodes of bilateral tonic-clonic evolution. EEG and MRI data were used to further corroborate the diagnosis.Reference Rai, Shukla and Afsar16–Reference Garg, Charlesworth and Shukla18
For this purpose, charts from all clinic visits were reviewed along with the results of all investigations patients underwent prior to inclusion in the study. Those with uncertain confirmation of diagnosis of epilepsy (only having presented with history of spells, and not receiving anti-seizure medications), or an unclear classification into focal onset versus generalized epilepsy, were excluded.
Following an initial training phase (conducted specifically for the student investigator (JK)), the study database was populated through chart review, using the hospital information system. Patient clinical encounter notes as well as EEG and imaging reports were reviewed. Data were then entered manually into an MS Excel® spreadsheet, which was used later for analysis and comparison between groups. This data collection was initially conducted by JK, and later independently checked for accuracy and completeness by a neurology resident (HM). All concerns pertaining to missing data, unclear diagnostic categorization and appropriate categorization of EEG data were resolved after discussion with the epileptologist (GS).
The study database compiles detailed information that was prospectively collected during initial and follow-up visits to the epilepsy clinic. Using a pre-structured database template, the collected information included demographic and epilepsy classification details, birth history, developmental history, detailed history of past medical events and potential antecedents (like head trauma, febrile seizures and/or febrile status epilepticus, neuroinfections, early seizures following neurological insult as well as birth-related complications), along with seizure semiology and epilepsy course (initial remission, latent period between initial seizure and later recurrences). Family history, social history and detailed epilepsy comorbidity (sleep-related, psychiatric, cognitive morbidity and focal neurological deficits) were also part of the assessment template. EEG and MRI details were entered into the study database.
Definitions
A Prior history of head trauma was defined as any prior history of involvement in motor vehicle accidents and/or physical assault and/or injury through outdoor sport participation or by impact with objects (sports equipment, solid surfaces and others), associated with any loss of awareness and/or headache, vomiting or neurological deficit.Reference Ding, Gupta, Diaz-Arrastia, Laskowitz and Grant19 Concussion diagnosed following admission to any hospital facility was also included.Reference Ding, Gupta, Diaz-Arrastia, Laskowitz and Grant19
Major neurological insults included head trauma, stroke, neuroinfection or metabolic encephalopathy.Reference Herman20,Reference Lolk, Dreier, Sun and Christensen21 Seizures occurring within 3 months of a major neurological insult were considered “early.”
Birth-related brain injury included hypoxemia, pathological hyperbilirubinemia, hypoglycemia or other perinatal events that required extended hospitalization (with or without the need for neonatal intensive care unit management).Reference Lolk, Dreier, Sun and Christensen21 Neuroinfections included a history of diagnosed meningitis or encephalitis.Reference Vezzani, Fujinami and White22
EEG data categorization
Findings from the most recent EEG recordings (routine and/or sleep-deprived EEG with time-locked video and/or prolonged video-EEG monitoring) were entered into the study database. As a routine, anterior temporal electrodes (T1, T2) were applied for all patients undergoing EEG evaluation at our EEG laboratory. Additionally, sub-temporal electrodes (T9, P9 [left] and T10, P10 [right]) were applied for patients in whom there was high suspicion of a temporal region focus and/or if a previous study was found unremarkable.
Findings were categorized according to morphological abnormality (e.g., slowing, sharp waves or spike discharges, polyspikes) and distribution (generalized; focal – temporal/extratemporal [frontal, central, parietal, occipital]) of abnormal activity. Temporal lobe abnormalities were subclassified as:
-
– Mesial/anterior, if the maximal involvement was over channels including anterior temporal electrodes (T1, F7 on left; T2, F8 on right); and
-
– Middle or posterior, if abnormalities involved mid or posterior electrodes (T3, T5, T9, P9 – on the left and T4, T6, T10, P10 on the right).
MRI data categorization
MRI data were categorized as normal or abnormal. Abnormalities were further categorized based on topographical distribution and etiological possibilities. For MRI conducted at centers other than ours, images were procured from other centers following patients’ consent. If images were unavailable, text reports were used. Patients’ accounts and/or charts from other centers were used for the rare case where none of the above (images or formal reports) were available.
Statistical analysis
Data analysis was conducted to compare different documented antecedents and other characteristics of included patients in the TLE versus GGE groups. After running tests of normality of distribution, different demographic and epilepsy characteristics, including means of age, age at epilepsy onset and epilepsy duration in years, were compared between both groups. This comparison was conducted using a two-tailed, independent t-test, while accounting for unequal variances between the groups. Additionally, the prevalence of epilepsy etiology and comorbidities was compared between the two groups using the chi-square test. Prevalence of reported antecedents and potential etiological risk factors was also compared between the TLE and GGE groups using the chi-square test.
Results
Demographic details and epilepsy characteristics
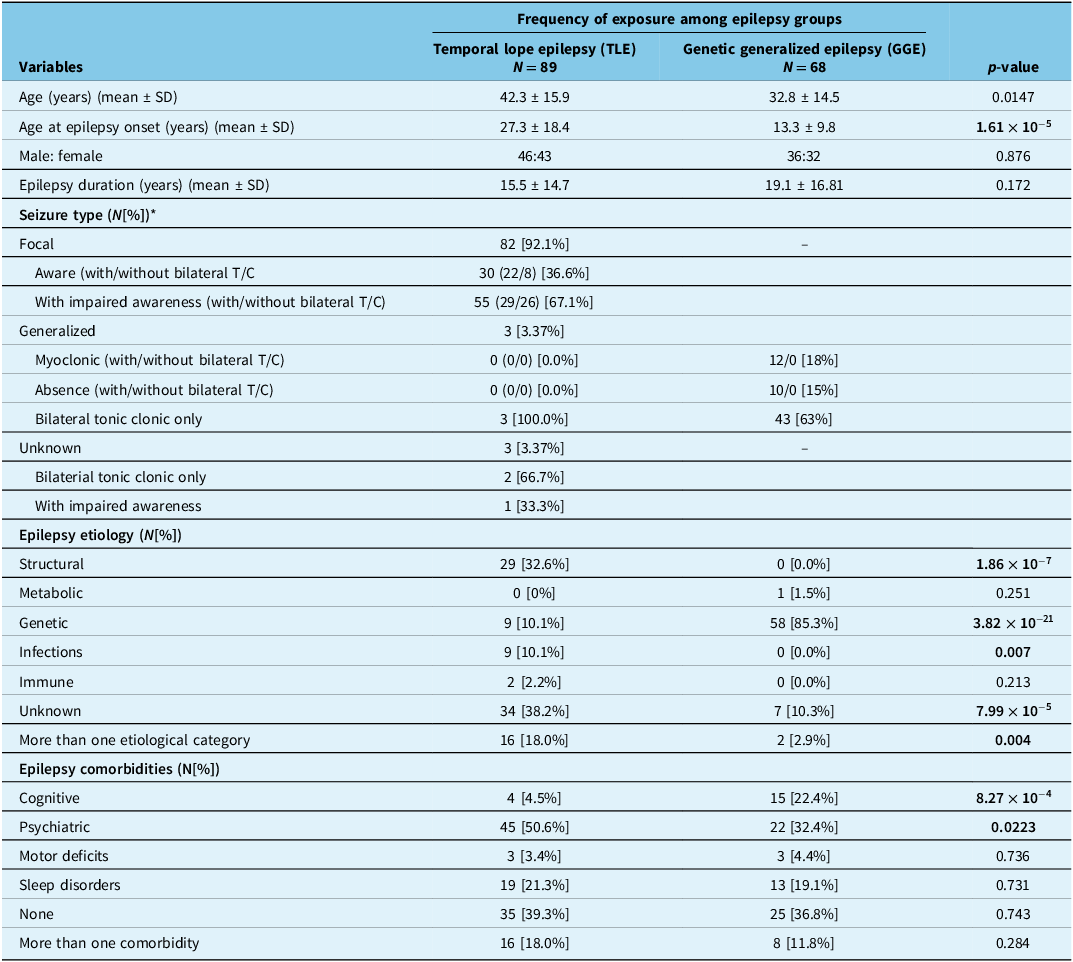
Among 505 epilepsy patients seen during the study period, a total of 89 patients with average age of 42.3 ± 15.9 years (46 males, 43 females) fulfilled inclusion criteria and comprised the TLE group. In comparison, 68 patients with average age of 32.8 ± 14.5 years (36 males, 32 females) were included in the GGE group. The average age at epilepsy onset in the TLE group was 27.3 ± 18.4 years, while the GGE group had a significantly lower average age at epilepsy onset of 13.1 ± 9.6 years (p = 1.61×10−5). The average epilepsy duration was 15.5 ± 14.7 years in the TLE group and 19.13 ± 16.81 years in the GGE group (p = 0.172) (Table 1).
Demographic details and epilepsy characteristics of patients with temporal lobe epilepsy (TLE) (N = 89) compared to those with genetic generalized epilepsy (GGE) (N = 68)

* Statistical significance not reported as seizure types are evidently different.
Antecedents in TLE versus GGE groups
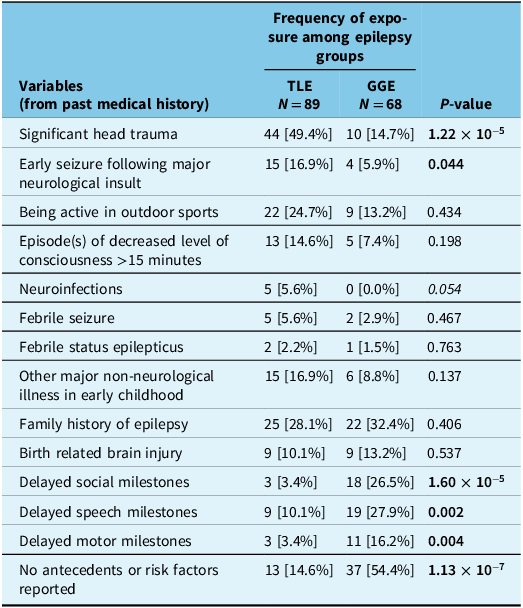
Factors more strongly associated with TLE than GGE included a history of head trauma (p < 0.0001) and a history of early seizure(s) following major neurological illness (p = 0.04). In addition, previous diagnosis of neuroinfection (p = 0.05) was also much more commonly reported in the TLE group. Another notable observation is that nearly a quarter of this population had a history of participation in outdoor sports, though this difference was not statistically significant compared to patients with GGE (p = 0.44).
Other clinically relevant factors that did not differ significantly between the two groups included: history of febrile status epilepticus (p = 0.77); major non-neurological illness in early childhood (p = 0.14), and episode(s) of decreased level of consciousness for more than 15 minutes (p = 0.20); family history of epilepsy (p = 0.41) and history of birth related brain insult (p = 0.54). Interestingly, delayed social, speech and motor developmental milestones were found significantly more common in the GGE group (p < 0.0001, p = 0.003 and p = 0.004, respectively) but not with TLE (Table 2).
Antecedents and potential etiological risk factors reported for patients with temporal lobe epilepsy (TLE) (N = 89) compared to those with genetic generalized epilepsy (GGE) (N = 68)

TLE characteristics
In our cohort of 89 patients, most (92.1%, n = 82) had a clear history of focal onset seizure semiology. In the entire TLE group, 61.8% (n = 55) experienced impaired awareness, while 33.7% (n = 30) reported no impairment of awareness. Fifty-one patients in the TLE group had a history of at least one seizure that progressed to bilateral tonic-clonic seizure. Among those without impaired awareness, 29 patients reported progression to bilateral tonic-clonic seizures, while 26 did not.
Five patients in the TLE group (5.6%) presented with a history suggestive of only bilateral tonic-clonic seizures, two of whom had seizures exclusively during sleep. The seizure type was classified as “unknown” in two patients.
Clinical course and seizure semiology
Only nine patients in the TLE group experience an initial period of remission following the first seizure. The median latent period for remission was 14.1 years with a range spanning 2 to 43 years. Based on description by family members or other witnesses, seizure semiology largely included loss of awareness/behavioral arrest (n = 52, 58%), typical aura of epigastric rising sensation and/or nausea (n = 14, 16%), aura of déjà vu, feelings of fear, confusion, anxiety and others (n = 40, 45%), auditory, visual and/or olfactory hallucinations or auras (n = 25, 28%) and oral and/or distal manual automatisms (n = 27, 30%).
Epilepsy comorbidities
In the TLE group, diagnosed cognitive comorbidities were significantly less prevalent compared to the GGE group (4.5% vs 22.4%, p < 0.001). Conversely, physician-diagnosed psychiatric comorbidities were significantly more common in the TLE group than in the GGE group (50.6% vs 32.4%, p = 0.022). There were no significant differences between the two groups in terms of motor deficits (3.4% vs 4.4%, p = 0.736) and sleep disorders (21.3% vs 19.1%, p = 0.731). Furthermore, the proportions of patients with no comorbidities (39.3% vs 36.8%, p = 0.743) and those with more than one comorbidity (18.0% vs 11.8%, p = 0.284) were similar between the TLE and GGE groups.
EEG findings
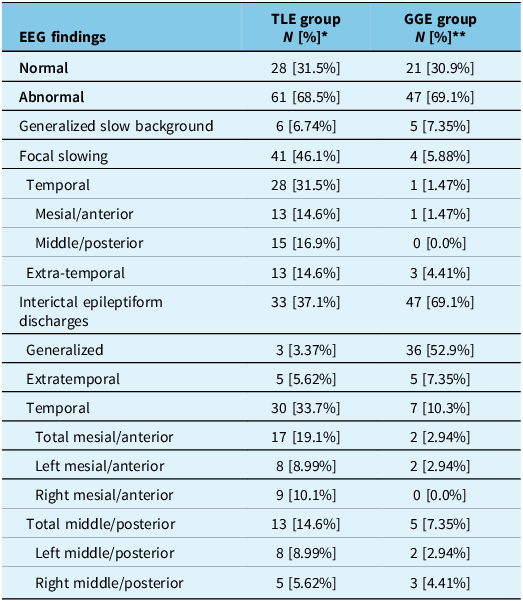
In the TLE group, left temporal EEG abnormalities were found in 28 patients, right temporal abnormalities in 18 patients and bilateral temporal lobes involvement in 8 patients. In this group, 61 patients out of 89 patients (68.5%) had abnormal EEG findings. Most commonly, focal temporal region slowing was found in 41 patients’ EEGs, accounting for 67.2% of those with abnormal findings. The second most common EEG finding was focal temporal epileptiform discharges, found in 33 patients, accounting for 54.1% of those with abnormal findings.
Extratemporal slowing was found in 13 patients (31%), often occurring alongside temporal abnormalities; among those, five patients also had extratemporal epileptiform discharges. Only eight patients were found to have generalized (or non-lateralized) findings (slowing and/or epileptiform discharges). Of note, 34 patients (38%) had a combination of two or more of the above-mentioned findings. Twenty-eight (31.5%) patients of the TLE group had a normal EEG.
Of those who had temporal lobe abnormalities on EEG, epileptiform discharges were detected in anterior channels (F7, F8, T1, T2) in 17 patients (19.1%) and middle/posterior temporal (T3, T4, T9, T10, P9, P10, T5, T6) channels in 13 patients (14.6%), thus demonstrating a similar percentage of patients having anterior/mesial abnormalities and middle/posterior temporal abnormalities. In the GGE group, a similar proportion of patients exhibited abnormal EEG findings (n = 47, 69.1%), with the majority being generalized interictal epileptiform discharges (n = 36) (see Table 3).
EEG findings reported for patients with temporal lobe epilepsy (TLE) (N = 89) compared to those with genetic generalized epilepsy (GGE) (N = 68)

* All percentages are calculated from the total number of TLE group of 89.
** All percentages are calculated from the total number of GGE group of 68.
Neuroimaging findings (MRI)
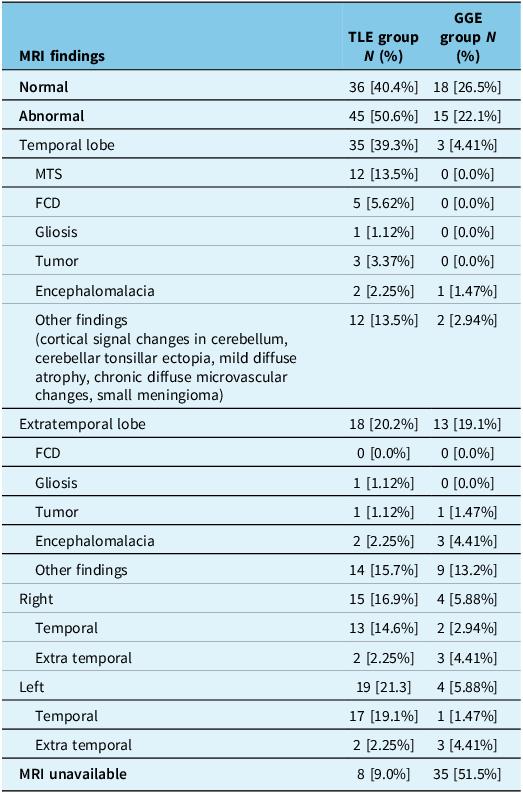
In the TLE group, a total of 13 patients were found to have an abnormality in the right temporal lobe, 17 patients in the left temporal lobe and 1 patient had abnormalities involving both temporal lobes (hyperintensities over bilateral hippocampi). Among these, 12 patients (13.5%) demonstrated clear evidence of mesial temporal sclerosis (unilateral n = 11, bilateral n = 1). In the remaining patients, other lesions, including focal cortical dysplasia (n = 5), tumor (n = 3), encephalomalacia (n = 2) and gliosis (n = 1), were found. A total of 36 patients (40.4%) showed normal MRI findings, while MRI images or formal reports were unavailable for 8 (9.0%).
In the GGE group, about half of the patients had accessible MRI images or formal reports (n = 33, 48.5%), 18 of which were normal. Most abnormal findings were non-specific, bilateral and of extratemporal distribution (n = 15, 86.7%) (Table 4).
MRI findings reported for patients with temporal lobe epilepsy (N = 89) compared to those with genetic generalized epilepsy (N = 68)

Discussion
In this study, we assessed antecedents and etiologic risk factors among patients diagnosed with TLE, compared to a control population of those with GGE. Our analysis revealed significant associations between TLE and a history of head trauma, early seizures following major neurological insults and neuroinfections.
Current understanding of risk factors in TLE
There is paucity of literature, especially from Canada, around the etiological risk factors of TLE, apart from surgical case series.Reference Shukla and Prasad4,Reference Uijl, Leijten, Arends, Parra, Van Huffelen and Moons5,Reference Junna, Buechler and Cohen-Gadol23,Reference Tassi, Meroni and Deleo24 Between 1993 and 2022, 11 studies reported findings on one or a few risk factors of TLE, with sample sizes ranging between 30 and 333 patients.Reference Soeder, Gleissner and Urbach1,Reference Klein, Dingledine and Aronica6–Reference Rai, Shukla and Afsar16 Four larger studies (N = 188–243) primarily investigated febrile seizures.Reference Heuser, Cvancarova, Gjerstad and Taubøll10–Reference Ferruzzi, Ferruzzi and Fernandes12,Reference Özkara, Uzan and Benbir15 Other studies investigated additional risk factors, including head trauma, early seizures following acute neurological insult, infections of the central nervous system (CNS) and family history.Reference Soeder, Gleissner and Urbach1,Reference Klein, Dingledine and Aronica6–Reference Sztriha, Gururaj, Bener and Nork9,Reference Tubi, Lutkenhoff and Blanco13,Reference Berg14
Most of the studies were retrospective and focused on post-surgical population. One prospective study evaluated epilepsy development in patients with significant head trauma,Reference Özkara, Uzan and Benbir15 along with two comprehensive review papers.Reference French, Williamson and Thadani7,Reference Tubi, Lutkenhoff and Blanco13 Significant head trauma emerged as the most common risk factor associated with TLE.Reference Klein, Dingledine and Aronica6–Reference Sztriha, Gururaj, Bener and Nork9,Reference Tubi, Lutkenhoff and Blanco13,Reference Berg14 Other factors identified in those studies included early onset seizures following an acute injury, febrile seizures and neuroinfections such as meningitis or encephalitis.Reference Soeder, Gleissner and Urbach1,Reference Shukla and Prasad4,Reference French, Williamson and Thadani7–Reference Berg, Langfitt and Shinnar11
The current study captures data collected through a structured template, applied to consecutive patients attending a referral epilepsy clinic, irrespective of patient’s candidacy for surgery, and hence is more representative of the general Canadian TLE population.
Head trauma
History of minor or major head trauma was reported in 2%–38% of patients with TLE in various studies,Reference Klein, Dingledine and Aronica6,Reference Sztriha, Gururaj, Bener and Nork9–Reference Berg, Langfitt and Shinnar11,Reference Tubi, Lutkenhoff and Blanco13–Reference Berg14 while in the current study, history of head trauma was recorded in 50% of patients with TLE. This was significantly higher proportion than in GGE (15%). This difference between our findings and those previously reported may be related to the study design with documentation limitations given varied research objectives of those studies, and may also reflect some socio-cultural variations, especially engagement in outdoor activity, as elaborated upon below.
Although participating in outdoor sports did not show a statistically significant difference between TLE and GGE groups, the frequency was notably higher in the TLE group (25%). On average, there is a higher percentage of Canadian population regularly engaging in outdoor activities (78%), with 15% reporting playing fast-paced sports such as football, soccer, hockey and others,Reference Berg14 compared to 44% of the European population25 and 54% of the US population,26 reporting some outdoor physical activity at least once a week.
Being active in fast-paced physical sports is frequently associated with repetitive TBI, which is usually mild and often underreported.27 Interestingly, we also noted a high percentage of patients with EEG abnormality originating from middle/posterior temporal regions (N = 28, accounting for 48% of all temporal EEG abnormalities), suggestive of lateral temporal lobe involvement.
This temporal neocortical involvement is commonly seen in both major head trauma and repetitive concussions among individuals engaged in high-impact sports.Reference Mez, Daneshvar and Kiernan28 Our EEG findings align with those of Gupta et al who reported lateral (neocortical) localization in 56% of 115 patients with focal post-traumatic epilepsy.Reference Diaz-Arrastia, Agostini and Frol29 Moreover, only 12% of TLE patients in the our study had mesial temporal sclerosis on brain MRI studies, with 40% showing no apparent abnormality, 12% had non-specific findings and only 3% demonstrated clear encephalomalacia or gliosis (indicative of prior major trauma or stroke); these results possibly indicate some association with prior minor (but likely repetitive) head trauma.
Neuroinfections
In our study, 5.6% of TLE patients had a documented history of neuroinfections, compared to none in the GGE group. This is higher than the cumulative range of 3% reported in three studies on TLE patients.Reference Rathore, George, Kesavadas, Sankara Sarma and Radhakrishnan3,Reference Heuser, Cvancarova, Gjerstad and Taubøll10,Reference Berg14 The differences between our findings and other studies could be attributed to the difference in study population and study design. For instance, Harvey et al reported only new onset TLE in a pediatric population,Reference Heuser, Cvancarova, Gjerstad and Taubøll10 while Ozkara et al specifically reported patients with mesial TLE (mTLE) and hippocampal sclerosis,Reference Rathore, George, Kesavadas, Sankara Sarma and Radhakrishnan3 and Feruzzi et al reported a broader epilepsy population from Brazil, 19 of which had TLE.Reference Berg14 In a comprehensive review by Klein et al, data from multiple studies indicated that CNS infections, among other acute brain injuries, share many commonalities, particularly inflammatory processes that can eventually lead to epilepsy.Reference Rai, Shukla and Afsar16
Early seizure(s) following neurological insult
While the definition of “early seizures” varies across the literature,Reference Harvey, Berkovic, Wrennall and Hopkins8,Reference Berg14 with timeframes ranging between one week and six months following an acute neurological insult, in the current study, we defined early-onset seizures as those occurring within 3 months of an acute brain injury.
In our study, approximately 16% of the TLE group had a history of early seizures following a neurological insult. In comparison, a recent study reported a higher rate of 32%.Reference Özkara, Uzan and Benbir15 It is important to note that in 53% of those patients in that study, early seizures occurred within one week of the insult, while the remainder developed seizures within six months. Several factors may explain the differences between our findings and those of Tubi et alReference Tubi, Lutkenhoff and Blanco13:
-
1) Although the majority of patients who developed post-traumatic epilepsy in Tubi et al. study had TLE, as evidenced by temporal lobe abnormalities on neuroimaging, recruited patients had various epilepsy syndromes, not exclusively TLE.
-
2) The Tubi et al. study included only patients with moderate to severe TBI, a population known to have a higher risk for early onset seizures.
-
3) In Tubi et al.’s study, all patients were subjected to a continuous EEG in the acute phase during the ICU admission, which increases the likelihood of seizure detection. In our study, we primarily relied on patient or family reported seizure history, which could be affected by memory inaccuracies or the misidentification of atypical seizures.
Our findings are similar to those found in a large systematic review, in which Klein et al reported data on a total of 10,211 patients across five studies conducted between 1935 and 2010 in Europe and the USA.Reference Harvey, Berkovic, Wrennall and Hopkins8 These studies investigated epilepsy development following acute brain injury, specifically TBI, stroke, intracerebral hemorrhage and CNS infection. Early-onset seizures were found to increase the risk of later developing epilepsy, with incidence rates ranging from 13% to 27%. These authors propose that the acute activation of the innate immune system in the brain following an acute injury and the subsequent changes in the blood-brain barrier and the local structures trigger an inflammatory cascade affecting areas of the brain that are prone to seizures. It is possible that the inflammation itself promotes brain damage, leading to hyperexcitability and lowered seizures threshold, and resulting in early seizures that can further progress to epilepsy.Reference Harvey, Berkovic, Wrennall and Hopkins8
Febrile seizures and febrile status epilepticus
Febrile seizures are among the most frequently reported antecedents and risk factors for TLE, particularly mTLE, with prevalence estimates ranging from 3.3% to as high as 70–78%.Reference Rathore, George, Kesavadas, Sankara Sarma and Radhakrishnan3,Reference Tubi, Lutkenhoff and Blanco13
Interestingly, Heuser, in a 2011 study, considered TLE with childhood febrile seizure (TLE-FS) to be a distinct category of TLE, potentially pointing toward a common genetic ground.Reference Ferruzzi, Ferruzzi and Fernandes12 In the current study, we found that only 5.6% of TLE patients had a history of febrile seizures, with 2% having a history of confirmed febrile status epilepticus. Our study included a wider TLE population and not just those with mTLE with hippocampal sclerosis (mTLE-HS). A large population-based study from Denmark found an increased risk of epilepsy (not necessarily TLE) in children with recurrent febrile seizures, with the risk rising as the number of hospital admissions for febrile seizures increased. It is plausible that many of these patients had prolonged febrile seizures/febrile status epilepticus (not elaborated upon in the study), rather than simple febrile seizures alone.Reference Gupta, Sayed and Ding30 Independent risk of epilepsy in general, associated with febrile seizures (simple, complex or status epilepticus), remains unclear.Reference Dreier, Li, Sun and Christensen31 However, the FEBSTAT study did not identify a clear association between febrile status epilepticus and hippocampal injury on imaging.Reference Lewis, Shinnar and Hesdorffer32
Strengths and limitations
Our study provides systematically collected data with the objective of identifying common antecedents and potential risk factors for TLE in a Canadian population. Having an appropriate control population of GGE is an important strength of this study as most previous studies have reported frequency of different risk factors in TLE populations, without including a comparison group. Another strength of this study is the use of structured data collection template by a single epileptologist, ensuring consistency and reliability across cases.
Limitations include the relatively small sample size, along with some missing data, which could have been more complete through prospective data collection, although the retrospective design minimizes risk of bias. Additionally, we were unable to further stratify TLE, as categorizing subtypes was challenging given the high percentage of patients with normal MRI (40%) and the small proportion with specific TLE syndromes such as mTLE-HS (13.5%).
Nonetheless, this research lays the groundwork for broader prevention and early identification strategies and serves as a steppingstone toward larger, multicenter studies incorporating prospective assessments with advanced structured interviews, neurophysiologic and neuroimaging evaluation, which could yield deeper insights into the optimal management of TLE.
Conclusion
This retrospective study from a Canadian epilepsy center, exploring antecedents and potential risk factors for TLE, provides important insights into the etiological basis of this condition. The study identifies head trauma, neuroinfections and early seizures following a past neurological insult as the most significantly associated risk factors among Canadian patients with TLE. Further, larger multi-center prospective studies, incorporating the evaluation strategies highlighted here, could provide valuable new information regarding TLE etiopathology.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/cjn.2025.10517.
Acknowledgements
We express our gratitude to the patients whose data made this study possible; their participation is invaluable in advancing clinical research in temporal lobe epilepsy. We sincerely acknowledge the support from KHSC Epilepsy Secretary Ann Norton, Epilepsy nurse Sandra Weatherby and all KHSC EEG technologists. We also express our appreciation to the reviewer for their valuable and detailed input, which helped improve this manuscript.
Author contributions
HM: Data extraction, data organization, manuscript writing. JK: Ethics application submission, data extraction, data analysis, manuscript editing
LBL: EEG interpretation, manuscript review. GPW: EEG interpretation, manuscript review and editing. GS: Study conceptualization, data collection in clinic, ethics application review and submission, manuscript writing and editing.
Funding statement
No funding was received to support this study.
Competing interests
HM – None. JK – None. LBL – Payment from International League against Epilepsy Education Grant to Canadian League against epilepsy. Consulting fees, honoraria and advisory board participation fees from Knight Pharmaceuticals and Jazz Pharmaceuticals. President Elect, Canadian League against Epilepsy. GPW – Research grants from Canadian Institutes of Health Research, PSI Foundation, SEAMO Innovation Fund, Canada First Research Excellence Fund, University Hospitals Kingston Foundation and Department of Medicine, Queen’s University, Kingston, ON. GS – Research grants from Canadian Foundation for Innovation, PSI Foundation, University Hospitals Kingston Foundation and Department of Medicine, Queen’s University, Kingston, ON.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the authors used ChatGPT in parts of the paper to improve language and readability. After using this service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.