Vitamin A is an essential micronutrient for pregnant women and their unborn babies, and its status during pregnancy has been associated with neonatal outcomes(Reference Hanson, Lyden and Anderson-Berry1–Reference Ma, Chen and Liu3). It is well-established that vitamin A is essential for the maintenance of vision, immune function, cell differentiation, fetal organ development and skeletal growth(Reference Bastos Maia, Rolland Souza and Costa Caminha2,Reference El-Khashab, Hamdy and Maher4,Reference Ishaq, Kunwar and Qadeer5) . Vitamin A deficiency (VAD) is a public health challenge affecting about 15 % of pregnant women in low-and middle-income countries(Reference Gernand, Xu, West, Karakochuk, Zimmermann, Moretti and Kraemer6,7) . VAD in pregnancy has been associated with intrauterine growth restriction, compromised fetal and maternal immune function, preterm delivery, low birthweight, maternal night blindness and increased risk of morbidity(7–Reference Kumwenda, Miotti and Taha10). Even sub-clinical VAD with no clinical signs (i.e. plasma retinol < 20 µg/dl) in the third trimester has been observed to increase the risk of preterm delivery by 74 % and maternal anaemia by 82 % among pregnant women in India(Reference Radhika, Bhaskaram and Balakrishna11). VAD has been linked to increased risk of anaemia in pregnancy, as vitamin A influences Hb concentration by regulating iron storage, mobilisation and transport and erythropoiesis(Reference Gernand, Xu, West, Karakochuk, Zimmermann, Moretti and Kraemer6,Reference Neves, Castro and Oliveira12) . Additionally, VAD leads to iron sequestration in the liver and spleen, reducing its availability for erythropoiesis and ultimately causing anaemia and suboptimal iron status(Reference Semba and Bloem13).
With the ongoing nutrition transition from a diet rich in indigenous foods to more westernised dietary patterns, coupled with urbanisation, the adult population of South Africa is grappling with a burden of multiple micronutrient deficiencies(Reference Steyn, Bradshaw and Norman14,Reference Vorster, Kruger and Margetts15) . It is therefore not surprising that approximately 22 % of South African women of reproductive age are vitamin A deficient(Reference Harika, Faber and Samuel16). As part of efforts to curb the prevalence of VAD and its detrimental effects on morbidity and mortality in the general population, the South African government implemented the mandatory fortification of maize meal and wheat flour with vitamin A in 2003(17). Additionally, a national vitamin A supplementation programme was implemented in 2002, which provides high-dose vitamin A to children aged 12–59 months (200 000 IU) and to children aged 6–11 months (100 000 IU) at all public health facilities(17). Nonetheless, the deficiency persists in some population groups, especially among women of reproductive age(Reference Harika, Faber and Samuel16). Contrary to the national VAD prevalence of 22 % reported among women of reproductive age in South Africa, a study conducted in a low socio-economic community in the Northern Cape Province of South Africa observed no cases of VAD among female caregivers below 50 years, who reported high liver consumption(Reference van Stuijvenberg, Schoeman and Lombard18). Similarly, in the KwaZulu-Natal Province, dietary intake of vitamin A among pregnant women in a rural community was reported to be above the daily recommended intake(Reference Napier, Warriner and Sibiya19). This indicates that there may be variabilities in the prevalence of VAD across the different provinces of South Africa, and such provincial differences must be taken into consideration when designing vitamin A supplementation programmes, especially in light of the teratogenic effects prenatal vitamin A toxicity can have on fetal development(Reference Bastos Maia, Rolland Souza and Costa Caminha2).
During pregnancy, nutrient requirements are increased, and vitamin A is one essential micronutrient whose requirement is increased during this period. Thus, VAD is likely to occur among women with insufficient dietary vitamin A intake during pregnancy. Information on the vitamin A intake and status of pregnant women in the Free State Province of South Africa is limited. Considering the significance of vitamin A in pregnancy, its link to anaemia and the adverse consequences of both deficiency and toxicity on maternal and fetal health, it is crucial to accurately assess the prevalence of VAD among high-risk populations. This evaluation is essential for determining the necessity of targeted supplementation and nutrition education. Therefore, this study aimed to investigate the vitamin A intake and status of pregnant women residing in Bloemfontein and the possible associations of vitamin A status with iron status, anaemia and birth outcomes.
Methods
Ethical statement
This study was approved by the University of the Free State Health Sciences Research Ethics Committee (Ethics No. UFS-HSD2017/0969) and the Free State Department of Health. During the study, all procedures followed the guidelines of the Declaration of Helsinki. The research was conducted on a voluntary basis, and all participants gave written informed consent.
Study design and participants
This cross-sectional study was a sub-study conducted as part of the larger Nutritional Status of Expectant Mothers and their newborn Infants (NuEMI) study, which included 700 pregnant women visiting the Pelonomi Tertiary Hospital in the urban Mangaung District of Bloemfontein, in the Free State Province of South Africa for routine antenatal appointments. Pelonomi Tertiary Hospital serves as a referral centre for high-risk pregnancies in Bloemfontein, managing cases of obesity, hypertension, diabetes, teenage or advanced maternal age, multiple caesarean sections, multiple pregnancies and poor previous pregnancy histories. Participants were conveniently recruited between May 2018 and April 2019. To participate in the larger NuEMI study, the pregnant women needed to provide informed consent and be at least 18 years old. Those expecting multiple babies were excluded. In this sub-study, only participants with plasma samples available for vitamin A and iron status assessment were included.
Data collection and measurements
Sociodemographic and health data
Trained fieldworkers conducted structured interviews with participants to collect sociodemographic information using a questionnaire adapted from the Assuring Health for All in the Free State (AHA-FS) study(Reference Walsh and van Rooyen20). Sociodemographic information collected included age, employment status, level of education, monthly income and marital status. Information on tobacco use and smoking was also collected. Information on the gestational age at enrolment and HIV status was extracted from the participants’ medical record booklets.
Anthropometry
Participants’ weight and height were measured using standard anthropometric techniques, as published elsewhere, to calculate gestational body mass index (GBMI)(Reference Ngounda, Baumgartner and Nel21). The weight and height of each participant, along with gestational age in weeks, were inputted into an algorithm used by Davies et al.(Reference Davies, Visser and Tomlinson22) to determine GBMI. GBMI was classified as underweight (GBMI ≥ 10 to < 19·8 kg/m2), normal weight (GBMI ≥ 19·8 to < 26·1 kg/m2), overweight (GBMI ≥ 26·1 to < 29 kg/m2) and obese (GBMI ≥ 29 kg/m2)(Reference Cruz, Harris and Read23).
Dietary vitamin A intake
Dietary intake was obtained in a structured interview using a previously validated quantified Food frequency questionnaire (FFQ)(Reference Symington, Baumgartner and Malan24) administered by trained fieldworkers in the preferred language of the participants. The daily amount of vitamin A consumed through dietary intake was calculated using the most recent South African food composition database(25), which contains the vitamin A content of foods fortified with vitamin A as part of the National Food Fortification Program at the South African Medical Research Council. The vitamin A intake (retinol activity equivalent (RAE)) was calculated using both retinol and β-carotene intake. The dietary intake was assessed over the previous 28 d; hence, the vitamin A intake values were divided by 28 to determine daily intake. To determine the possible intake of vitamin A from supplements, information on the type of prenatal supplements participants used, where they received them and how often they took them was collected. All the participants reported using the routine 65 mg elemental iron, 5 mg folic acid and 1000 mg calcium antenatal supplements provided as part of antenatal care in the South African public health system(26).
Household food security
The household food security of participants was evaluated using the Household Food Insecurity Access Scale(Reference Coates, Swindale and Bilinsky27). This scale comprises nine questions that determine whether household members have had sufficient food or had to change their food consumption due to resource limitations in the past 4 weeks. Household food security was categorised as ‘secure’, ‘mildly insecure’, ‘moderately insecure’ and ‘severely insecure’(Reference Coates, Swindale and Bilinsky27).
Biochemical analysis of vitamin A, inflammatory and anaemia status
Approximately 70–100 µl of capillary blood was consistently drawn from participants in the morning via finger prick into heparin tubes for biochemical analysis. Using the HemoCue Hb 201 + System(Reference Carboo, Dolman-Macleod and Uyoga28), Hb concentrations were measured in a drop of blood immediately after the finger prick. The blood samples were then centrifuged, and plasma samples were prepared and aliquoted into Eppendorf tubes (safe-lock, Eppendorf) onsite. The plasma samples were initially stored onsite at –20 °C for up to 4 d and later transported to the University of the Free State and stored at −80 °C. The samples were then transported on dry ice to the Centre of Excellence for Nutrition, North-West University, and stored at −80°C prior to analyses.
Vitamin A status of participants was measured as the concentration of plasma retinol-binding protein 4 (RBP4). RBP4 (i.e. the carrier of retinol in blood) is one of the developed biomarkers for assessing vitamin A status(Reference Sombié, Zeba and Somé29). RBP4 is synthesised in the liver and is responsible for transporting vitamin A from the liver to other tissues in a 1:1 complex with retinol(Reference Baeten, Richardson and Bankson30). RBP4 is more stable against light and heat and cheaper to measure than retinol. Therefore, it is easier to measure RBP4 concentration in surveys and in resource-limited settings(Reference Sombié, Zeba and Somé29). RBP4, markers of iron status, that is, ferritin and soluble transferrin receptor (sTfR) concentration, and markers of inflammation, that is, C-reactive protein and α-1-acid glycoprotein, were measured in 50 µL of plasma using the Q-Plex™ Human Micronutrient Array (7-plex) (Quansys Bioscience)(Reference Chimhashu, Verhoef and Symington31) at the micronutrient laboratory of the Centre of Excellence for Nutrition. Ferritin and sTfR concentrations are influenced by inflammation and, thus, were adjusted for C-reactive protein and α-1-acid glycoprotein, using the BRINDA correction method(Reference Luo, Geng and Zeiler32).
Vitamin A status was categorised as ‘adequate’ if the plasma RBP4 was ≥1·05 µmol/l, ‘insufficient’ if plasma RBP4 was 0·7–1·05 µmol/l and ‘deficient’ if RBP4 concentration was <0·7 µmol/l(7,Reference Baeten, Richardson and Bankson30,Reference Ross, Hodges, Wei, Prasad and Brewer33) . RBP4 < 1·05 µmol/l was termed as low vitamin A status in this article. C-reactive protein > 5 mg/l and α-1-acid glycoprotein > 1 g/l were indicative of elevated inflammation (Reference Thurnham, Northrop-Clewes and Knowles34). After adjusting Hb values for altitude (Bloemfontein is 1300 metres above sea level)(Reference Silubonde, Baumgartner and Ware35), anaemia was defined as Hb <11 g/dl(36). Iron deficiency (ID) was defined as a ferritin concentration <15 µg/l after adjusting for inflammation. ID anaemia was defined as adjusted ferritin <15 µg/l and Hb <11 g/dl(Reference Thurnham, Northrop-Clewes and Knowles34). ID erythropoiesis was defined as adjusted sTfR > 8·3 mg/l(Reference Symington, Baumgartner and Malan37,Reference Erhardt, Estes and Pfeiffer38) .
Birth outcomes
Participants were requested to present the medical record booklet of their neonates to the study dietitian for assessment of birth outcomes after delivery. Data regarding birth outcomes, including gestational age at birth and birth weight, were extracted from the medical record of each baby after delivery. Infants delivered at 37 weeks of gestation or beyond were categorised as term, while those born before 37 weeks of gestation were classified as premature(39). In accordance with WHO criteria, newborns weighing less than 2500 g were classified as low birth weight (LBW)(40). The neonates were further classified as small, appropriate or large for gestational age based on their gestational age and birth weight and sex using the INTERGROWTH-21st standards for Size at Birth, version 1.0.6257.25111. Weight at gestational age <10th percentile, between 10th and 90th percentiles and > 90th percentile was classified as small, appropriate and large for gestational age, respectively(Reference Falcão, Ribeiro-Silva and de, Almeida41). Mothers who did not provide birth outcome information after delivery were followed up with reminder text messages to obtain the necessary details.
Statistical analyses
All data analyses were conducted using the Statistical Package for Social Sciences (SPSS) software, version 27. Normality of distribution of data was tested using histograms and the Shapiro–Wilk test. Descriptive statistics, including frequencies and percentages for categorical data and medians (25th percentile (Q1), 75th percentile (Q3)) and means (sd) for numerical data, were calculated. Comparisons of continuous variables between the different groups were performed using an independent t test if normally distributed, the Mann–Whitney U test for non-normally distributed data between two groups and the Kruskal–Wallis test for non-normally distributed data across three or more groups. The comparisons of categorical variables were performed using the χ 2 test. Linear regression models were used to determine the association of vitamin A status with birth outcomes and anaemia and iron status. Potential confounding factors, including maternal age, HIV status, GBMI and gestational age, which were significantly associated with birth outcomes, anaemia and iron status, were adjusted for in the adjusted models. P-values of < 0·05 were considered statistically significant.
Results
Characteristics of study participants
Table 1 presents the characteristics of the study participants. A total of 427 pregnant women were included in this sub-study, out of which only 205 and 197 provided the birth weight and gestational age of their neonates after birth, respectively, due to loss to follow-up. The 427 participants had a median (Q1, Q3) age of 32 (27, 37) years and were at a median (Q1, Q3) gestational age of 32 (26, 36) weeks at enrolment. The majority of participants (73 %) were in their third trimester at the time of enrolment. More than half (52·0 %) of the participants were unemployed, while 43·0 % had a monthly household income of R3000 (∼$168) or less. The median (Q1, Q3) GBMI was 31·3 kg/m2 (24·6, 37·7), with the majority (54·5 %) being obese and 6·5 % being underweight. Only 26·6 % of participants were food secure, while 61·4 % were moderately and severely food insecure. Additionally, 30·9 % of the participants were living with HIV infection. Almost 42 % of the participants were anaemic, while 28·3 % were iron deficient. Out of the 205 and 197 participants who provided birth weight and gestational age of their neonates, 11 % had LBW and 14 % preterm babies. There was no significant difference in the prevalence of anaemia (P = 0·980), ID (P = 0·453), ID anaemia (P = 0·897) and ID erythropoiesis (P = 0·088) status between participants who reported birth outcomes and those who did not (Supplementary Table 1).
Characteristics of participants

Q1, 25th percentile; Q3, 75th percentile; GBMI, gestational BMI; ID, iron deficiency; IDA, iron deficiency anaemia; IDE, iron deficiency erythropoiesis; sTfR, soluble transferrin receptor.
Vitamin A status and dietary intake of participants
Table 2 presents the vitamin A status and dietary intake of vitamin A, retinol, β-carotene and total carotenoids of participants. The median (interquartile range) plasma RBP4 concentration was 1·51 (0·78) µmol/l, with 86·7 % having adequate vitamin A status (i.e. RBP4 ≥ 1·05 µmol/l), while 12·2 % were vitamin A insufficient (i.e. RBP < 0·7 – <1·05 µmol/l), and 1·2 % were vitamin A deficient (i.e. RBP < 0·7 µmol/l). The median dietary intake of vitamin A was 1007 µgRAE/d. Approximately 19 % of participants had a daily dietary intake of vitamin A below the estimated average requirement of 550 µgRAE, while 7 % consumed more than the tolerable upper limit of 3000 µgRAE/d(42). Additionally, the median concentration of plasma RBP4 among participants living with HIV was significantly higher than those without HIV (1·63) (1·27, 2·08 µmol/l) v. (1·47) (1·18, 1·89) (µmol/l, P = 0·026), despite similar daily dietary intake of vitamin A (i.e. 954 (642, 993) µgRAE/d, v. 1032 (631, 1754) µgRAE/d, P = 0·990). There was no significant difference in the median plasma RBP4 concentration and prevalence of low vitamin A status between participants who had birth outcome data (i.e. 1·50 (1·20, 2·04) µmol/l; 10·7 %) and those without (i.e. 1·51 (1·20, 1·95) µmol/l, P = 0·709; 15·8 %, P = 0·154).
Vitamin A status and dietary vitamin A intake of participants (n 427)

Q1, 25th percentile; Q3, 75th percentile; RBP4, retinol-binding protein 4; RAE, retinol activity equivalent; EAR, estimated average requirement; UL, tolerable upper limit. *Median (Q1, Q3) reported.
The contribution of commonly consumed vitamin A-rich foods to overall daily vitamin A intake
Table 3 presents the contribution of commonly consumed vitamin A-containing foods to the pregnant women’s total daily vitamin A intake. All participants reportedly consumed some form of vitamin A fortified cereal and/or cereal products, which accounted for approximately 24·9 % of their total daily vitamin A intake. Furthermore, liver and carrots/pumpkin accounted for 32·7 and 28·0 %, respectively, of the total daily vitamin A intake. Participants within the highest quartile of daily vitamin A intake (i.e. >1785·7 µgRAE/d) had a significantly lower contribution of their daily vitamin A intake from fortified cereal (7·5 %) but significantly higher daily contributions from liver (46·3 %) and carrot/pumpkin (45·4 %) intake compared with those within the lower vitamin A quartiles who had higher vitamin A intake from cereal and lower intakes from liver and carrots/pumpkin (P < 0·001) (Table 3). Comparing the contributions of various foods to total daily vitamin A intake by household food security status shows that the food insecure participants derived a higher proportion of their daily vitamin A intake from fortified cereal, that is, 31·1 % compared with those who were food secure (i.e. 18·2 %, P < 0·001) (Table 3). Additionally, the food secure participants had a significantly higher contribution of their daily vitamin A intake from the consumption of carrots/pumpkin than the participants who were food insecure (P = 0·033).
The contribution of commonly consumed vitamin A-rich foods to total daily vitamin A intake, analysed by vitamin A intake quartiles and food security status

Q1, 25th percentile; Q3, 75th percentile; RAE, retinol activity equivalent; vitA Q1, vitamin A intake < 637.8 µgRAE/d; vitA Q2, vitamin A intake of 637.8 − 1006.9 µgRAE/d, vitA Q 3, vitamin A intake of 1007.0 – 1785.7 µgRAE/d; vitA Q4, vitamin A intake >1785.7 µgRAE/d. Kruskal–Wallis test was used to compare the percentage contribution of the commonly consumed vitamin A-rich foods to total daily vitamin A intake across the different vitamin A intake quartile groups and household food security categories. P <0.05 is considered statistically significant. Post hoc test using Bonferroni correction (a, d, f: <0.001; b: 0.002; c: 0.022; e: 0.026; g: 0.012).
Comparison of daily vitamin A intake among adequate, insufficient and deficient participants
The daily intake of vitamin A was non-significantly higher in participants with adequate plasma vitamin A status (1046·24 µgRAE) than in those who were insufficient (889·53 µgRAE) or deficient (500·66 µgRAE) (P = 0·079) (Supplementary Table 2). Nonetheless, a significant positive association was observed between vitamin A intake and plasma RBP4, with a percent increase in vitamin A intake resulting in a 0·07 % increase in plasma RBP4 concentration (β = 0·068; 95 % CI 0·020, 0·116; P = 0·006). Additionally, the daily intake of retinol was positively associated with plasma RBP4, as a percent increase in retinol intake resulted in a 0·03 % rise in plasma RBP4 concentration (β = 0·029; 95 % CI 0·005, 0·054; P = 0·019). There was no significant association of daily intake of β-carotene (β = 0·005; 95 % CI −0·022, 0·033; P = 0·710) and total carotenoids (β = 0·008; 95 % CI –0·020, 0·035; P = 0·586) with plasma RBP4 concentration (Supplementary Table 3).
Association between vitamin A status, anaemia and iron status
The prevalence of anaemia was 54·4 % among the participants with low vitamin A status (i.e. RBP4 < 1·05 µmol/l) and 39·7 % in those with adequate vitamin A status (i.e. RBP4 ≥ 1·05 µmol/l) (P = 0·043). The concentration of Hb was significantly lower among participants with plasma RBP4 < 1·05 µmol/l compared with those with RBP4 ≥ 1·05 µmol/l (10·63 ± 1·63 g/dl v. 11·25 ± 1·45 g/dl, P = 0·003) (Table 4). After adjusting for maternal age, HIV status and GBMI, the difference in Hb concentration observed between participants with low and adequate vitamin A status remained significant (P < 0·001). Additionally, a significant positive association between plasma RBP4 and Hb concentration was observed, with a unit increase in RBP4 concentration resulting in a 0·41 g/dl increase in Hb concentration (β = 0·409; 95 % CI 0·208, 0·610; P < 0·001). After adjusting for maternal age, HIV status and BMI, the positive association remained significant (β = 0·42; 95 % CI 0·223, 0·618; P < 0·001). Furthermore, the median concentration of ferritin was significantly lower among participants with low vitamin A status (19·12 (11·90, 34·15 μg/l) compared with those who had adequate levels (24·34 (14·33, 41·09) μg/l, P = 0·001) (Table 4). A significant positive relationship was observed between plasma RBP4 and ferritin concentration, with a percent increase in RBP4 resulting in a 0·36 % (β = 0·359; 95 % CI 0·139, 0·579; P = 0·001) increase in ferritin concentration. Additionally, RBP4 was observed to be negatively associated with sTfR, with a percent increase in plasma RBP4 concentration being associated with a 0·13 % decrease in sTfR concentration (β = −0·125; 95 % CI −0·246, −0·005; P = 0·041).
Association of vitamin A status with Hb, ferritin and soluble transferrin receptor concentrations

Q1, 25th percentile; Q3, 75th percentile; RBP4, retinol-binding protein 4; sTfR, soluble transferrin receptor; +Independent t test. ^ Independent-samples Mann–Whitney U test. In the linear regression analysis, Hb, ferritin and soluble transferrin receptor concentrations were the outcome/dependent variables, and retinol-binding protein 4 concentration was the predictor variable * log-transformed ferritin, soluble transferrin receptor and retinol-binding protein 4 concentrations. P-value <0.05 is considered statistically significant. Maternal age, HIV and gestational BMI were adjusted in the adjusted model.
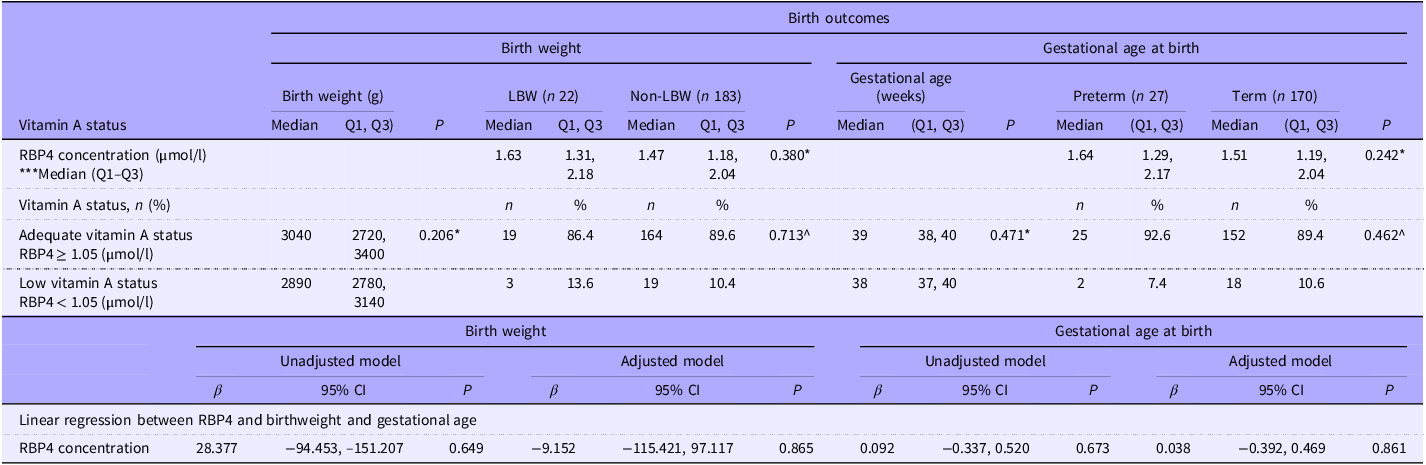
Association between vitamin A status, birth weight and gestational age
Table 5 presents the vitamin A status of participants according to birth outcomes. Neonates born to mothers with low vitamin A status (RBP4 < 1·05 µmol/l) had a median birth weight of 2890 g. Those born to mothers with adequate vitamin A status (RBP4 ≥ 1·05 µmol/) had a median birth weight of 3040 g. The incidence of LBW did not differ between participants with low vitamin A status and those with adequate status (P = 0·713). There was no significant association between plasma RBP4 concentration and birth weight (β = −9·152; 95 % CI −115·421, 97·117; P = 0·865). Similarly, there was no significant difference in the median gestational age of babies born to participants with low vitamin A status compared with those who were vitamin A replete (i.e. 38 (37, 40) weeks v. 39 (38, 40), P = 0·471). Furthermore, there was no significant difference in the incidence of preterm birth between participants with low and adequate vitamin A status (P = 0·462).
Association between vitamin A status, birth weight and preterm delivery

Q1, 25th percentile; Q3, 75th percentile; LBW, low birth weight; RBP4, retinol-binding protein 4. * Mann–Whitney U test comparing birth weight and gestational age at birth between the adequate vitamin A status group and low vitamin A status group; ^ χ 2 test. Maternal age and GBMI were adjusted in the adjusted model.
Discussion
In the present study, we assessed vitamin A intake and status and its association with iron status and anaemia, as well as birth outcomes among pregnant women. Our findings indicate a relatively low prevalence of VAD (RBP4 < 0·7 µmol/l) of no public health significance in this group of pregnant women(7), in addition to a positive association between RBP4 and the concentrations of Hb and ferritin and a negative association with sTfR.
In this study, the prevalence of low vitamin A status (RBP4 < 1·05 µmol/l) was 13·3 %, which is comparatively higher than the 4 % prevalence reported in another cohort of pregnant women at <18 weeks of gestation in Johannesburg, South Africa(Reference Chimhashu, Melse-Boonstra and Symington43). The authors noted, however, that vitamin A levels decreased substantially as the pregnancy progressed(Reference Chimhashu, Melse-Boonstra and Symington43). In contrast to the 22 % prevalence of VAD (retinol < 0·7 µmol/l) reported in a systematic review among South African women of reproductive age, which included national data and studies across five provinces(Reference Harika, Faber and Samuel16), our study found a deficiency prevalence of 1·2 %, which is substantially lower. The high prevalence of adequate plasma vitamin A concentration in our study population likely stems from the high daily intake of vitamin A, which was 183 % of the estimated average requirement for pregnant women, with 81 % of the participants meeting and exceeding the daily estimated average requirement(42). This may be partly due to the high consumption of fortified maize meal and wheat flour, which are major components of the diet of this population. Fortified cereal and cereal products accounted for 25 % of the daily vitamin A intake in our study. Additionally, liver and carrot/pumpkin consumption contributed significantly to the daily vitamin A intake, accounting for 33% and 27 %, respectively. For participants in the highest quartile of vitamin A intake, liver consumption contributed over 46 % of their daily vitamin A intake. As reported by van Stuijvenberg et al.(Reference van Stuijvenberg, Schoeman and Lombard18) in a study conducted in the Northern Cape Province of South Africa, regular liver consumption was associated with the absence of VAD. Although liver is a good and relatively inexpensive source of preformed vitamin A, excessive consumption during pregnancy could lead to vitamin A toxicity, which is known to potentially have deleterious consequences on the growing fetus(Reference Bastos Maia, Rolland Souza and Costa Caminha2). In our study, 7 % of the pregnant women exceeded the tolerable upper limit of daily vitamin A intake. Excessive vitamin A intake, especially during early pregnancy, can lead to congenital malformations affecting the central nervous and cardiovascular systems, as well as an increased risk of spontaneous abortion(44).
In the present study, the positive relationship observed between RBP4 and Hb concentration aligns with findings from other clinical and experimental studies, which have demonstrated an association between VAD and anaemia(Reference Hamdy, Abdel Aleem and El-Shazly45–Reference Dantham, Gaddam and Vadakattu48). These studies have suggested that VAD impairs haematopoiesis and contributes to anaemia, a condition that often improves with vitamin A supplementation, regardless of iron levels(Reference Zimmermann, Biebinger and Rohner46,Reference McCauley, van den Broek and Dou49) . Evidence from previous animal studies indicates that vitamin A plays a crucial role in the mobilisation of iron from the liver and spleen(Reference Mehdad, Siqueira and Arruda50,Reference Beynen, Sijtsma and Van Den Berg51) . Hence, VAD leads to retention of iron in these organs, making it less available for erythropoiesis and reducing its uptake into the bone marrow and incorporation into the red blood cells(Reference Beynen, Sijtsma and Van Den Berg51–Reference Sijtsma, Van Den Berg and Lemmens54). In a systematic review involving 3818 pregnant women, the risk of anaemia was significantly reduced by 36 % with vitamin A supplementation(Reference McCauley, van den Broek and Dou49). Additionally, studies conducted in Egypt, Brazil and Bangladesh have reported significantly lower Hb concentrations in pregnant women with VAD compared with those with adequate levels. These studies also found a significant positive association between maternal serum retinol and Hb concentration, along with a negative relationship between VAD and Hb concentration(Reference Radhika, Bhaskaram and Balakrishna11,Reference Neves, Castro and Oliveira12,Reference Hamdy, Abdel Aleem and El-Shazly45,Reference Ahmed, Mahmuda and Sattar55) . The results of these studies reinforce our finding that plasma RBP4 is positively associated with Hb concentration and participants with low vitamin A status had lower Hb concentration, amidst the routine high-dose iron supplementation provided to all pregnant women as part of standard antenatal care in South Africa(26).
Additionally, participants with adequate vitamin A status (plasma RBP4 ≥ 1·05 µmol/l) in our study had significantly higher ferritin concentrations, an indicator of iron stores, compared with those with low vitamin A status (plasma RBP4 < 1·05 µmol/l). Plasma RBP4 was positively associated with ferritin concentration. Among lactating mothers in Kenya, a positive association between serum retinol and ferritin has also been reported(Reference Ettyang, van Marken Lichtenbelt and Oloo56). Further evidence from a meta-analysis of vitamin A supplementation trials indicated that vitamin A supplementation significantly increased serum ferritin concentrations among pregnant and lactating women, but not in children and teenagers(Reference da Cunha, Campos Hankins and Arruda57). Furthermore, RBP4 was inversely associated with sTfR, a biomarker that reflects tissue iron demand and erythropoiesis, which increases in ID anaemia(Reference Beguin58). This inverse relationship suggests a potential role of vitamin A in improving tissue iron status, concurring with evidence from a vitamin A supplementation trial, which significantly reduced sTfR concentration among Moroccan children(Reference Zimmermann, Biebinger and Rohner46). The positive association between RBP4 and Hb and ferritin, as well as the inverse RBP4-sTfR relationship observed in this study and previous supplementation trials, suggests that vitamin A should be considered alongside iron and folic acid for the effective prevention and management of anaemia and ID in pregnant women who are vitamin A deficient. Notably, despite the low prevalence of VAD in this study, a high proportion of the participants were anaemic. This may be attributed to the high prevalence of HIV infection in the study population. As reported elsewhere, participants living with HIV had more than twice the odds of being anaemic and iron deficient(Reference Carboo, Ngounda and Baumgartner59).
Regarding the association between maternal vitamin A status and birth outcomes, we observed no significant association between vitamin A and birth weight as well as gestational age. Additionally, there was no difference in the incidence of LBW and preterm delivery between the mothers with RBP4 < 1·05 µmol/l and those with RBP4 ≥ 1·05 µmol/l. Contrary to our findings, a study in Brazil involving 488 mother–newborn pairs reported that VAD in pregnancy was negatively associated with neonatal birth weight (β: −0·10 kg; 95 % CI 0·20, −0·00), but the significance of the relationship was lost after adjusting for maternal ferritin concentration, and there was no association between retinol and birth weight z-scores(Reference Neves, Castro and Oliveira12). Evidence from meta-analyses and observational studies has consistently failed to demonstrate a clear association of vitamin A biomarkers and birth weight and the effectiveness of vitamin A interventions during pregnancy in reducing the risk of LBW in resource-limited settings but has shown to be beneficial in reducing the risk of maternal anaemia, clinical infections and night blindness (Reference McCauley, van den Broek and Dou49,Reference Thorne-Lyman and Fawzi60,Reference Gebremedhin, Enquselassie and Umeta61) . The evidence on the association between prenatal vitamin A status and birth weight and preterm birth is inconclusive.
It is worth highlighting the significantly higher RBP4 concentration observed among the pregnant women living with HIV compared with their counterparts without the infection, despite similar dietary intake. This could likely be attributed to antiretroviral therapy‑related mechanisms, which alter retinol metabolism and RBP dynamics in the liver(Reference Toma, Devost and Lan62). Alterations such as elevated retinoic acid associated with antiretroviral therapy can potentially decrease RBP turnover or increase RBP gene transcription, leading to higher RBP concentration(Reference Toma, Devost and Lan62,Reference Napoli63) .
This study has some limitations. The cross-sectional design of the study did not allow for the assessment of participants’ vitamin A status across different trimesters and a potential causal relationship with birth outcomes. Additionally, the study was carried out in a tertiary hospital that predominantly manages high-risk pregnancies; thus, the results may not fully reflect the general population of pregnant women with minimal or no risk factors and should be interpreted with this context in mind. Furthermore, the dietary vitamin A intake estimation was based on a quantified FFQ over a 28-d period, which is subject to recall bias. Despite this limitation, the findings highlight the importance of adequate vitamin A intake and status for improving iron status and preventing anaemia in pregnant women and emphasise beneficial food-based sources of vitamin A that can be leveraged to improve the vitamin A status of pregnant women in resource-limited areas. Additionally, our study was limited by the high loss to follow-up, leading to a significant amount of missing birth outcome data, which restricted the association studies between vitamin A and birth outcomes.
Conclusion
The prevalence of VAD in this sample of pregnant women in Bloemfontein is of no public health significance. However, RBP4 positively associated with Hb and ferritin concentrations, indicating the importance of adequate vitamin A status in preventing maternal anaemia and ID in pregnancy. Plasma vitamin A status was however not associated with birth weight and preterm delivery. Pregnant women, particularly in regions with high prevalence of VAD, might benefit from the consumption of food-based sources of vitamin A, including fortified cereal, liver and carrots/pumpkins. However, intake of preformed vitamin A-rich foods like liver should be moderated, especially in populations with a low VAD prevalence, to avoid the risk of vitamin A toxicity.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/S0007114526106473.
Acknowledgements
We appreciate the contribution of all the fieldworkers, Michelle Du Plooy, Desiré Brand and Khanyi Khumalo, for their hard work and dedication during data collection. We also thank the staff of the antenatal clinic at Pelonomi Regional Hospital and the participants for their support. We are grateful to Professor Gina for her guidance on cleaning the data.
This project was financially supported by the Department of Nutrition and Dietetics, University of the Free State, Bloemfontein, South Africa.
J. A. C.: conceptualisation, data analysis, interpretation of results and manuscript preparation. J. N.: conceptualisation, methodology, data collection, review of manuscript. J. B.: laboratory analysis, interpretation of results, review of manuscript. L. R.: coding and interpretation of dietary data, review of manuscript. M. J.: coding and interpretation of dietary data and birth outcome data, review of manuscript. C. M. W.: conceptualisation, methodology, principal investigator, review and finalisation of manuscript.
The authors declare that there are no conflicts of interest.