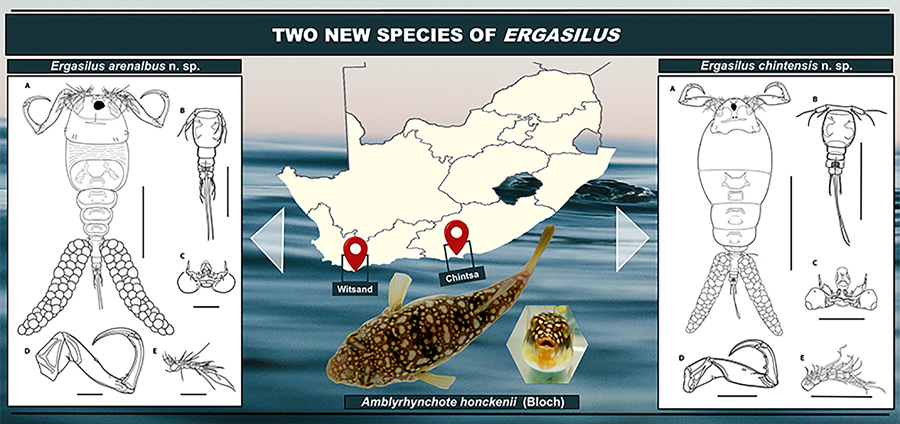
Introduction
Exploring diversity within marine ecosystems has become increasingly important in recent years, revealing a previously unnoticed level of species richness and genetic variation. This is particularly true for parasitic copepods within the family Ergasilidae Burmeister, 1835 (Boxshall and Defaye, Reference Boxshall and Defaye2008). Among the various genera in this family, Ergasilus Nordmann, 1832, stands out as one of the most speciose and widely distributed (Oldewage and van As, Reference Oldewage and van As1988; Oldewage and Avenant-Oldewage, Reference Oldewage and Avenant-Oldewage1993; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023). However, the extent of this diversity remains largely unexplored in Africa (Killian and Avenant-Oldewage, Reference Killian and Avenant-Oldewage2013; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023), with merely 19 reported species, 12% (19/163), of the global diversity (Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; WoRMS, 2024).
The lack of comprehensive studies in Africa limits our understanding of Ergasilus diversity, where unique environmental conditions and host communities may foster an even higher diversity of distinct Ergasilus species. While some investigations have provided insights into Ergasilus species in African freshwater systems (Oldewage and van As, Reference Oldewage and van As1988; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023), marine or brackish environments in this region remain largely unexplored, hosting only 5 known species and a single species from the South African coastline (Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; WoRMS, 2024). Additionally, the limited knowledge regarding their diversity and the lack of genetic data, compared to those of other well-studied organisms, hinder comprehensive genomic analyses, creating further challenges in gaining deeper insights into their phylogeny and biology (Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023).
The discovery of new Ergasilus species, especially in unexplored regions would aid in filling these crucial gaps and understanding these parasites' biogeography and evolution (Boxshall and Halsey, Reference Boxshall and Halsey2004; Song et al., Reference Song, Wang, Yao, Gao and Nie2008; Míč et al., Reference Míč, Řehulková and Seifertová2023). Furthermore, marine regions across the Atlantic and Indian oceans represent hotspots for parasitic diversity due to their wide range of marine habitats (Everett et al., Reference Everett, Groeneveld, Fennessy, Porter, Munga, Dias, Filipe, Zacarias, Igulu, Kuguru, Kimani, Rabarison and Razafindrakoto2015; Miller et al., Reference Miller, Hayashi, Song and Wiens2018). Hence, given the rich diversity of Ergasilus species described from the southern Atlantic and Indian Ocean regions (see Table 1), South Africa could provide an ideal setting to study marine Ergasilus species and their host associations, possibly yielding a hidden marine parasite diversity within these shores. Understanding the distribution and diversity of Ergasilus species across South Africa can provide valuable insights into evolutionary patterns, connectivity between populations and the impact of environmental factors on parasite distribution (Boxshall and Halsey, Reference Boxshall and Halsey2004). Additionally, such comparisons can aid in identifying potential host-switching events and the emergence of novel host–parasite associations (Boxshall and Halsey, Reference Boxshall and Halsey2004; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023).
Updated information for all marine species of Ergasilus Nordmann, 1832 described within the South Atlantic and Indian oceans, with information on host species, host families, distribution and available GenBank data
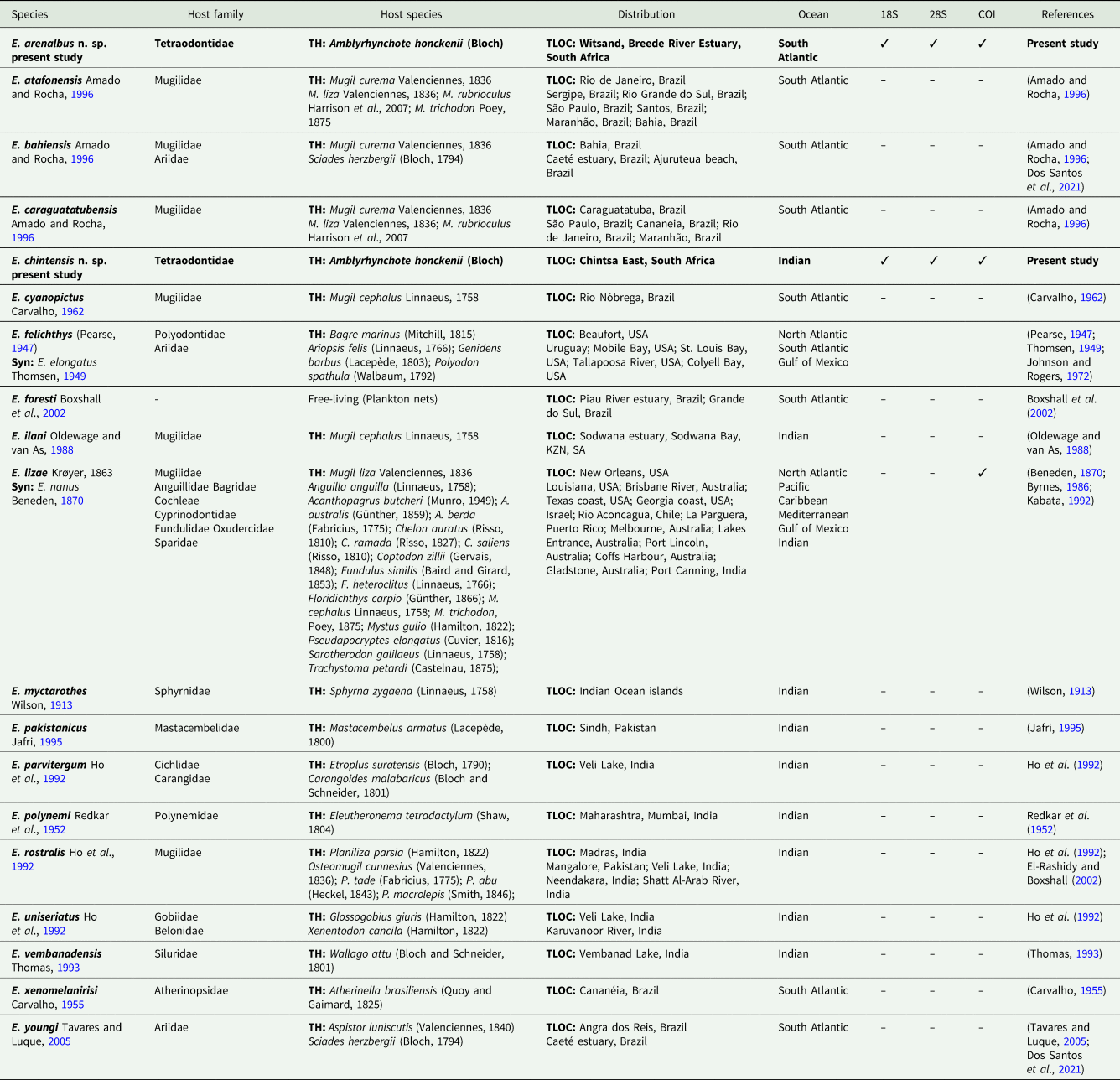
Information from the present study is represented in bold.
Syn, synonym; TH, type host; TLOC, type locality.
This study aimed to start filling this gap by investigating the presence, diversity and molecular characteristics of Ergasilus copepods associated with the Evileye blaasop, Amblyrhynchote honckenii (Bloch), along the South African coastline. Combining morphological examination and molecular analyses based on partial ribosomal RNA (rRNA) gene regions (18S and 28S), and 1 mitochondrial DNA gene region (COI), 2 new marine Ergasilus species were found and described. Revealing and documenting these new species enhances our understanding of the marine parasite diversity within this region, revealing new host–parasite interactions and evolutionary links.
Materials and methods
Sampling
As part of a larger study on the biodiversity of marine fish parasites in southern Africa, 25 A. honckenii specimens were collected from 2 coastal localities. Using rod and reel, 15 specimens of A. honckenii (13 males and 2 females) were collected from the Breede River Estuary, Witsand (−34.397323; 20.837474) in November 2021 and 10 specimens (1 male, 8 females and 1 juvenile) from the intertidal rocky shore at Chintsa East (−32.836538; 28.116997) in July 2022 (Fig. 1). Following capture, the fish were transported in aerated water containers to a nearby field station for dissection. The specimens were then identified, photographed, weighed, measured and humanely killed using percussive stunning followed by pithing (ethics committee approved standard operation procedure NWU-00267-17-A5). Ethical approval for this project was received from the AnimCare Ethics Committee of the North-West University with ethics number NWU-00565-19-A5. Permits for collecting A. honckenii were issued by Cape Nature, Western Cape Province and the South African Department of Agriculture, Forestry and Fisheries (permit no. CN44-87-18289 and RES2022-44, respectively).
Map indicating the sampling localities of specimens of Amblyrhynchote honckenii (Bloch).
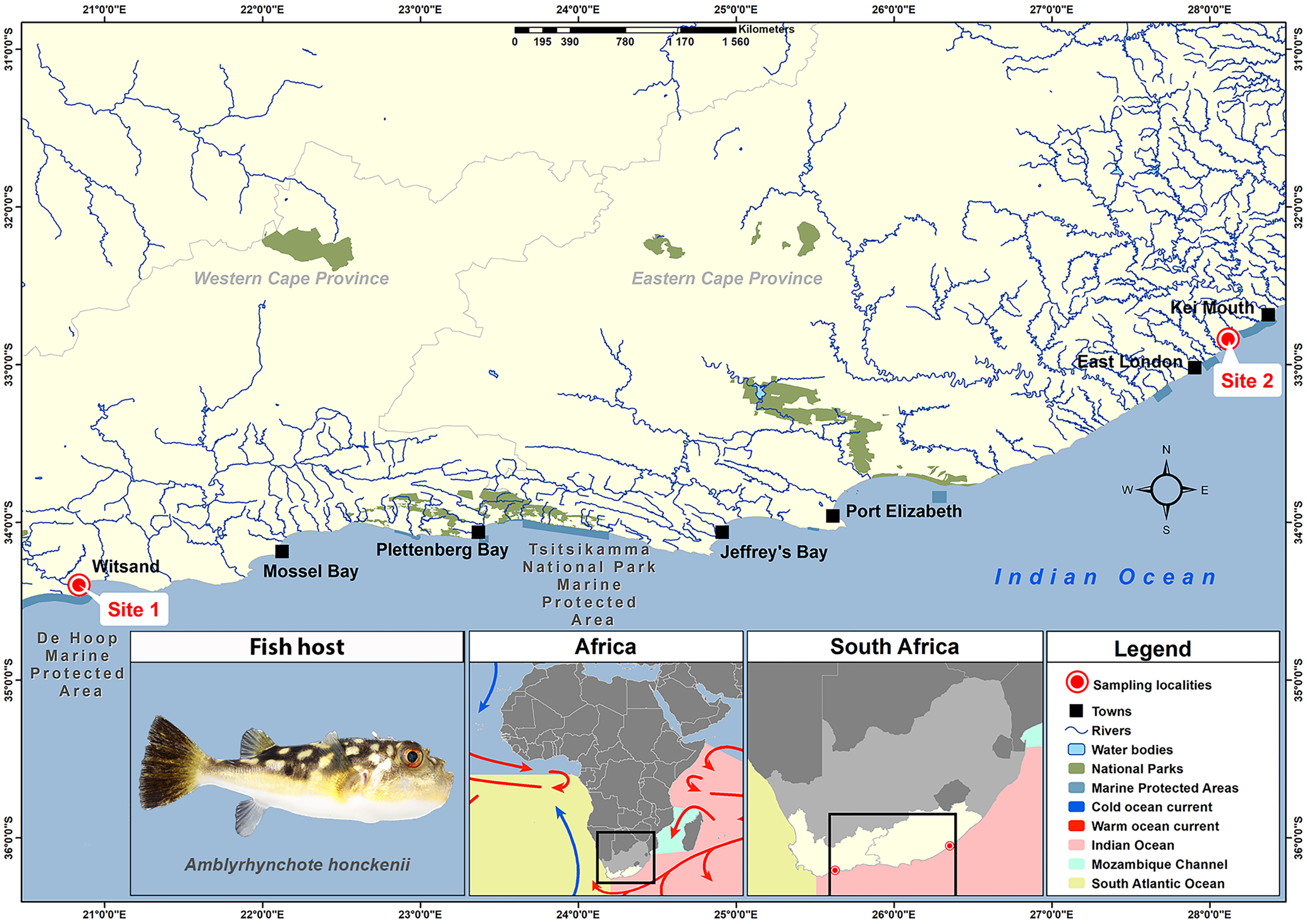
Fish were identified using Smith Sea Fishes (Smith and Heemstra, Reference Smith and Heemstra2012), with fish nomenclature following FishBase (Froese and Pauly, Reference Froese and Pauly2024) and Eschmeyer's Catalog of Fishes (Fricke et al., Reference Fricke, Eschmeyer and Van der Laan2024). Host authorities are not included in the text or references.
Morphological analyses
Fish gills were removed and screened for parasites using a Zeiss Stemi 305 compact stereomicroscope (Zeiss, Oberkochen, Germany). Copepod specimens were removed from the gills and preserved in 80% ethanol for morphology and 96% for molecular analysis. Twelve selected specimens underwent morphological observations after being cleared in lactic acid, dissected and temporarily mounted onto slides with glycerine. Photomicrographs of various body structures were captured using a Nikon Y-TV55 video camera mounted on a Nikon ECLIPSE Ni microscope (Nikon, Tokyo, Japan). Image analysis software, Image-Pro Express (Nikon), facilitated obtaining all necessary measurements for descriptive analyses. All measurements and terminology for describing body somites and cephalic appendages follow Boxshall and Montú's (Reference Boxshall and Montú1997) guidelines. Measurements are provided in text descriptions and tabular form, with text descriptions including average measurements followed by the range and number of specimens in parentheses. Table 2 presents metrical data as the mean, followed by the standard deviation and the number of specimens examined. Measurements are in micrometres unless otherwise specified. Pencil drawings of specimens and dissected appendages were created using a drawing tube attached to a Nikon ECLIPSE Ni microscope (Nikon). Final digital illustrations were made with Adobe Photoshop version 23.0.1 software using a Wacom Intuos Pro tablet (Wacom, Saitama, Japan).
Metrical information of the new species of Ergasilus Nordmann, 1832
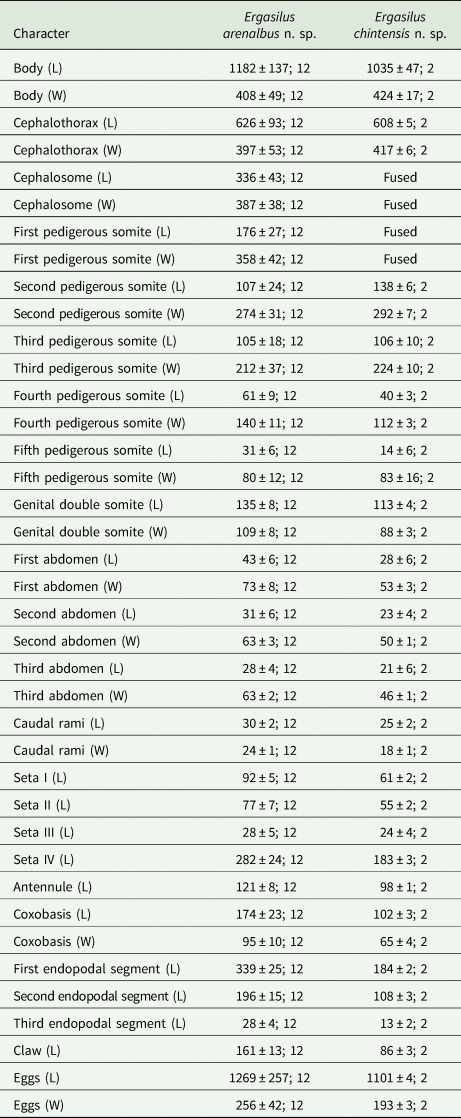
L, length; W, width.
Information is presented as the mean, followed by the standard deviation and the number of specimens examined.
All measurements are in micrometres.
Furthermore, 6 adult specimens collected from the Breede River Estuary, Witsand, were used for scanning electron microscopy (SEM). SEM could not be performed on specimens from Chintsa East due to the limited availability of specimens. Each specimen selected for SEM observation was cleaned by lightly brushing the surface. Cleaned specimens were dehydrated, placed in hexamethyldisilisane (HMDS), mounted onto carbon tape, placed on aluminium stubs and sputter-coated with carbon (Emscope TB500, Quorum Technologies, Puslinch, ON, USA), followed by 20–30 nm gold/palladium (Eiko IB2 ion coater, Eiko, Japan). Specimens were examined with a FEI Nova NanoSEM 450 scanning electron microscope (FEI, Hillsboro, OR, USA). Images were taken of various characteristic body structures to aid in the morphological description and for comparisons among species.
Molecular analyses
Genomic DNA extraction was conducted using egg strings from 2 copepod specimens from the Breede River Estuary, Witsand, and 1 copepod specimen from Chintsa East. Extraction followed the Macherey-Nagel NucleoSpin® Tissue extraction kit protocol (GmbH and Co. KG, Sandton, South Africa), with an adapted 4 h pre-lysis period and adding 50% more buffer BE. Partial gene amplification targeted 3 gene regions: 2 rRNA gene regions (18S and 28S) and 1 mitochondrial DNA gene region (cytochrome c oxidase I or COI), using primers (18SF, 18SR; 28SF, 28SR) prepared by Song et al. (Reference Song, Wang, Yao, Gao and Nie2008) for 18S and 28S, and universal mitochondrial primers (LCO1490 and HCO2198) (Folmer et al., Reference Folmer, Black, Hoeh, Lutz and Vrijenhoek1994) for COI (Table 3). Amplification reactions were conducted in 25 μL volumes, made up of 12.5 μL of DreamTaq PCR Master Mix (Thermo Fischer Scientific, Waltham, MA, USA), 1.25 μL of 10 μm of each primer, 3 μL of DNA product and 7 μL of double distilled water. Thermocycling conditions followed adapted protocols established by Folmer et al. (Reference Folmer, Black, Hoeh, Lutz and Vrijenhoek1994) and Song et al. (Reference Song, Wang, Yao, Gao and Nie2008). The PCR thermocycling profile followed adapted conditions: 94°C for 5 min, followed by 40 amplification cycles of 95°C for 30 s, 47°C for 30 s and 72°C for 1 min with a final extension at 72°C for 7 min. Positive PCR products were verified via 1% agarose gel electrophoresis and then sent for purification and sequencing in both forward and reverse directions to Inqaba Biotechnical Industries (Pty) Ltd. (Pretoria, South Africa).
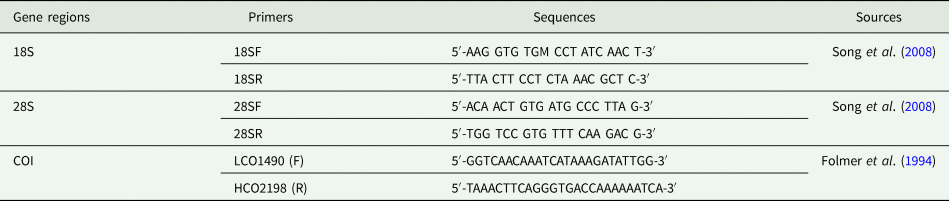
List of primers used for DNA amplification of parasitic copepods with sequences and references, used to amplify partial 18S, 28S and COI genes in this study
Sequences were assembled, aligned, edited and trimmed using Geneious Prime version 2023.1.2 (Biomatters, Auckland, New Zealand). Additionally, the nucleotide Basic Local Alignment Search Tool (BLAST) was used to select Paracyclopina nana Smirnov, 1935 (Cyclopettidae Martínez Arbizu, 2000), as the outgroup of the study (Table 4). Considering the limited availability of COI sequences, unpublished sequences of Ergasilus species, sourced from the Barcode of Life Database (BOLD), were also included in the COI alignment (Table 4). Alignments for the novel sequences were generated and trimmed using default parameters of MAFFT version 7.4.9 (Katoh et al., Reference Katoh, Misawa, Kuma and Miyata2002; Katoh and Standley, Reference Katoh and Standley2013). Genetic divergences among aligned specimens were calculated within Geneious Prime version 2023.1.2, presenting percentage similarities and differences in base numbers.
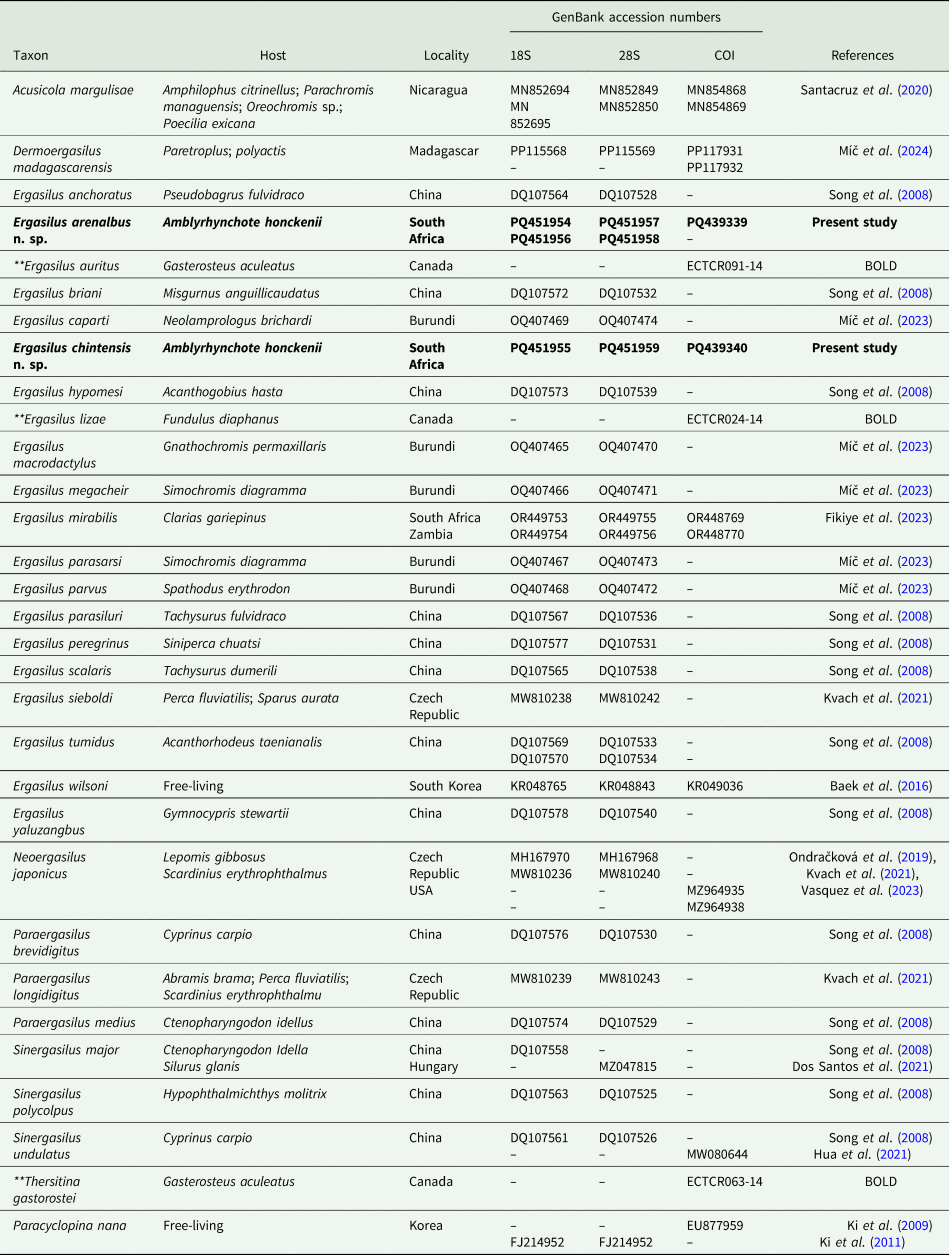
List of GenBank and Barcode of Life Database (BOLD) Ergasilidae sequences included in the phylogenetic analyses
The taxa in bold fonts are sequences generated from the present study. Paracyclopina nana (in the grey shade) was used as the outgroup.
**Taxon from the Barcode of Life Database (BOLD).
The optimal nucleotide substitution model for each dataset was estimated using the Akaike Information Criterion (AIC) in jModelTest 2.1.4 (Posada, Reference Posada2008; Darriba et al., Reference Darriba, Taboada, Doallo and Posada2012). The general time-reversible model with invariant sites and gamma-distributed rate variation (GTR + I + G) was recommended for all datasets (18S, 28S and COI). Phylogenetic analyses were performed using Maximum Likelihood (ML) and Bayesian Inference (BI) methods with this suggested model. BI analyses were conducted on the CIPRES Science Gateway version 3.3 (Miller et al., Reference Miller, Pfeiffer and Schwartz2010) using MrBayes version 3.2.7a (Ronquist et al., Reference Ronquist, Teslenko, van der Mark, Ayres, Darling, Höhna, Larget, Liu, Suchard and Huelsenbeck2012), with 2 independent Markov Chain Monte Carlo (MCMC) runs of 4 chains for 10 million generations, sampling every 1000 generations, and a burn-in of the first 25 000 generations. ML analyses were performed using PhyML version 3.0 (Guindon et al., Reference Guindon, Dufayard, Lefort, Anisimova, Hordijk and Gascuel2010) on the ATGC bioinformatics platform, with model parameters estimated and 1000 bootstrap repetitions for nodal support. The resulting phylogenetic trees from BI and ML analyses were visualized using TreeViewer version 2.2.0 (Bianchini and Sánchez-Baracaldo, Reference Bianchini and Sánchez-Baracaldo2024).
Results
Two distinct gill-associated parasitic species, morphologically and molecularly differentiated, were obtained from subsets of 10–15 A. honckenii specimens, ranging from 95 to 185 mm in length and weighing 30 to 140 g. Both morphotypes were classified as Ergasilus (Ergasilidae) based on specific characteristics, such as body typically cyclopiform with clear segmentation, biramous legs IV with 2-segmented exopods and 3-segmented endopods, 6-segmented antennules, antennas featuring a single claw and the absence of maxillipeds in females, following descriptions by Boxshall and Montú (Reference Boxshall and Montú1997) and Boxshall and Halsey (Reference Boxshall and Halsey2004). Notably, only 1 morphotype was found and described from each location, highlighting the uniqueness of both morphotypes in their respective collection sites.
Taxonomy
Ergasilus arenalbus n. sp.: Figures 2–5
ZooBank LSID: urn:lsid:zoobank.org:act:4B5580B2-C5BA-4B8E-99C3-14E9BBF32D3D
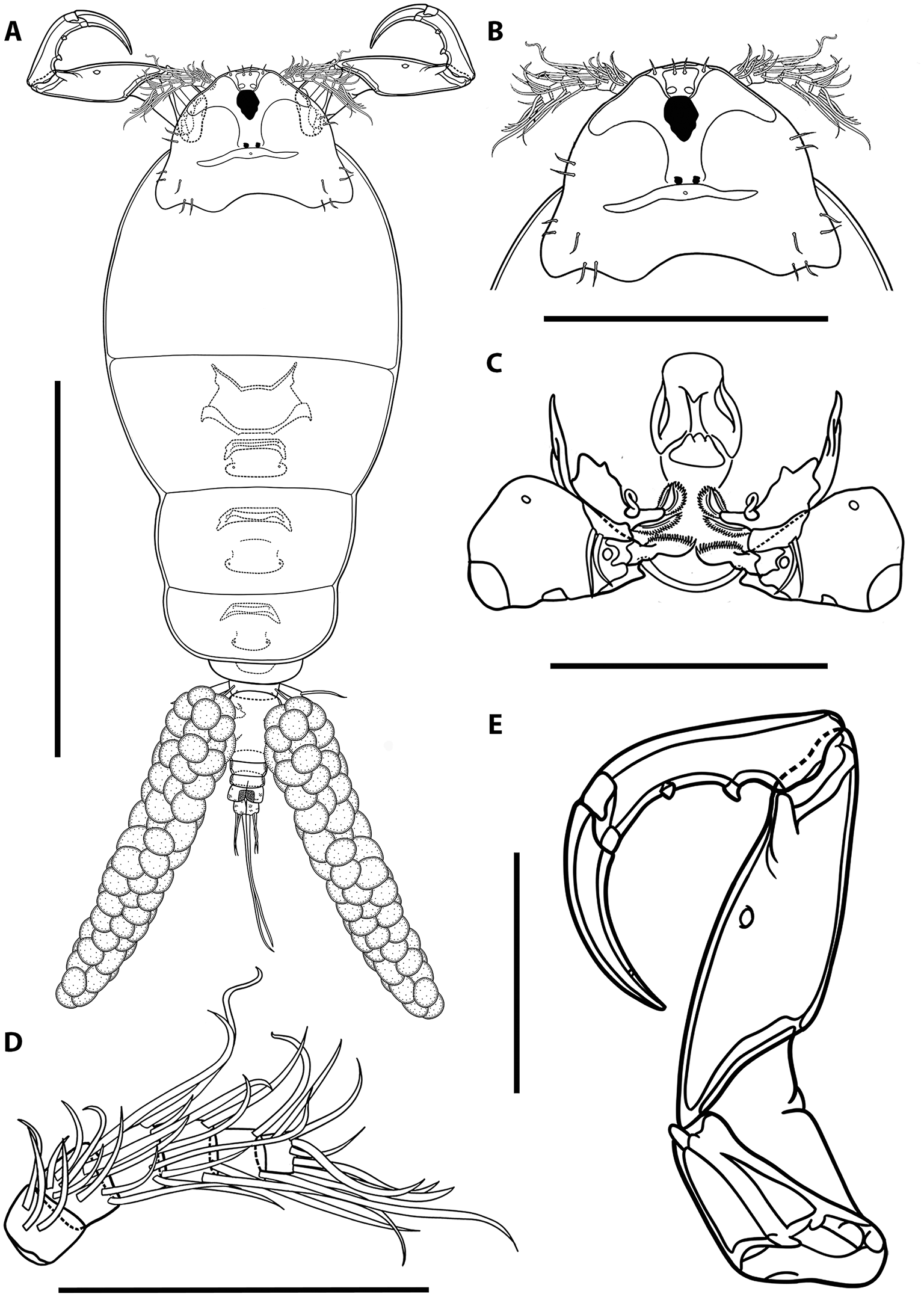
Illustrations of adult female of Ergasilus arenalbus n. sp: (A) entire specimen, dorsal view; (B) detail of cephalosome, dorsal view; (C) mouth, mandible, maxillule and maxilla; (D) antenna; (E) antennule. Scale bars: A – 500 μm; B – 250 μm; C–E – 100 μm.
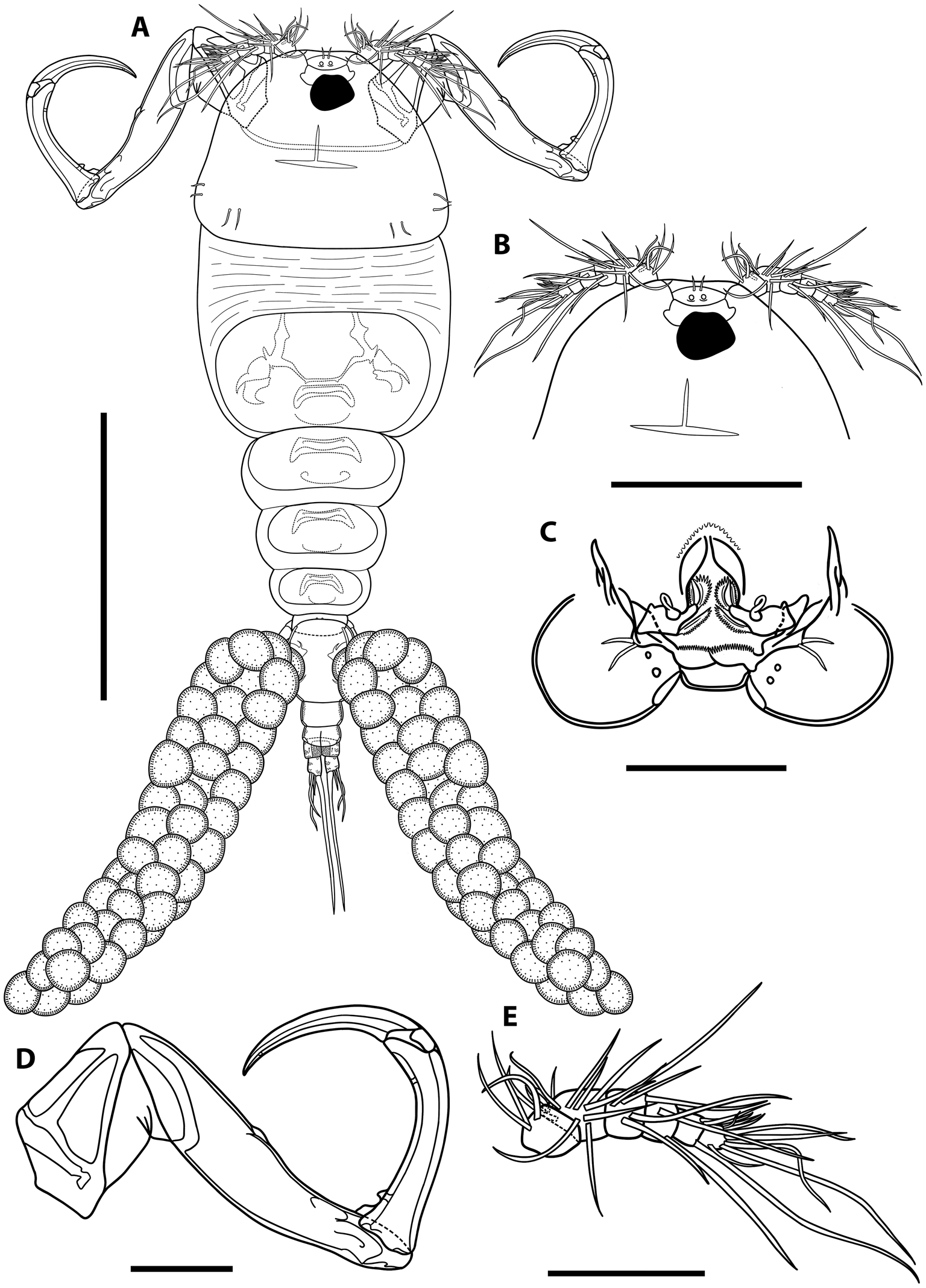
Illustrations of adult female of Ergasilus arenalbus n. sp: (A) urosome, dorsal view; (B) intercoxal sclerites and interpodal plates; (C) leg 1; (D) leg 2 and leg 3; (E) leg 4; (F) leg 5. Scale bars: A – 200 μm; B – 100 μm; C–F – 50 μm.
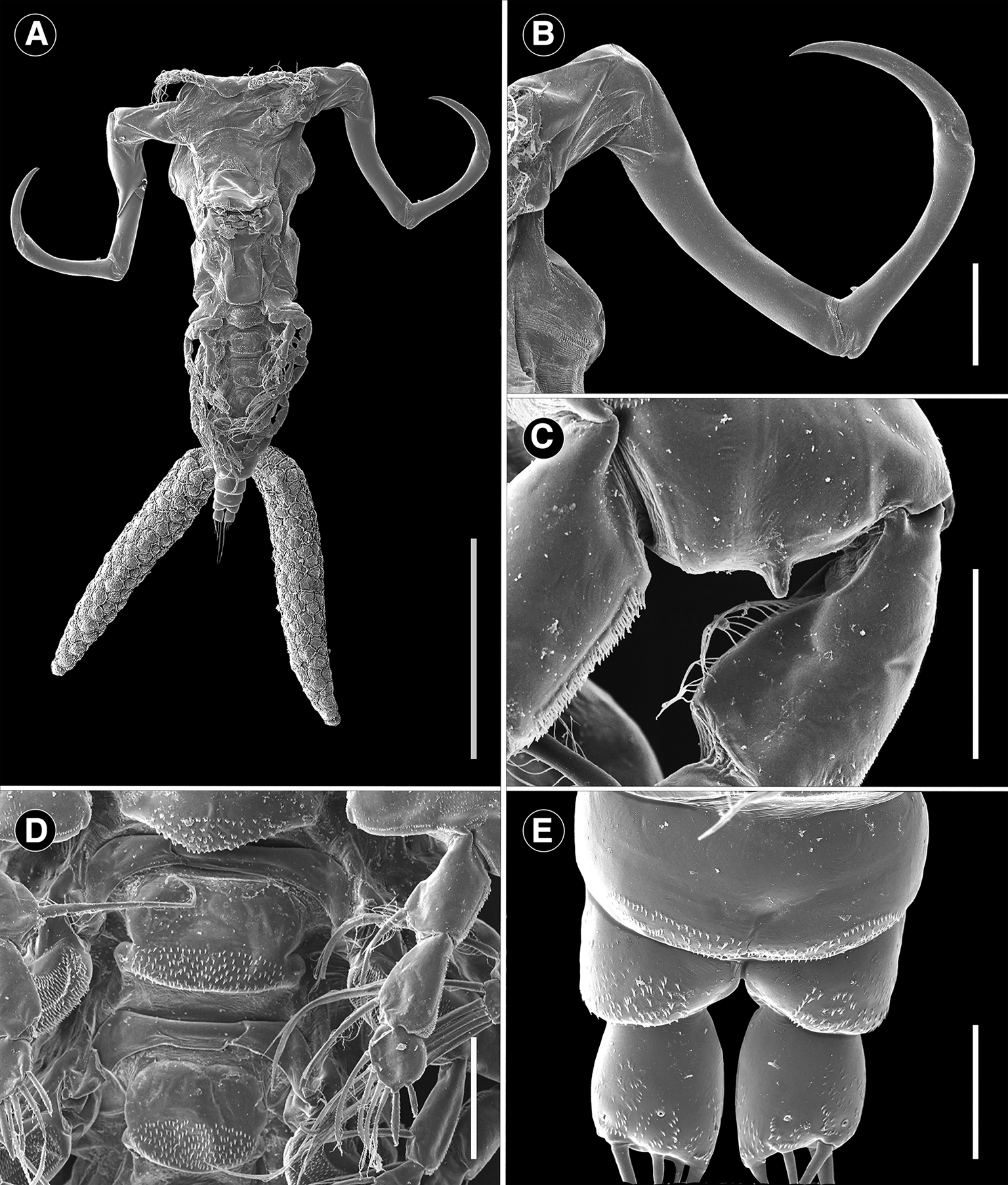
Scanning electron microscope photomicrographs of adult female Ergasilus arenalbus n. sp. showing features from the ventral and dorsal view: (A) entire specimen; (B) antenna; (C) base of first leg; (D) ventral view of interpodal plates; (E)– dorsal view of ornamentation on caudal rami. Scale bars: A – 500 μm; B – 100 μm; C–E – 25 μm.
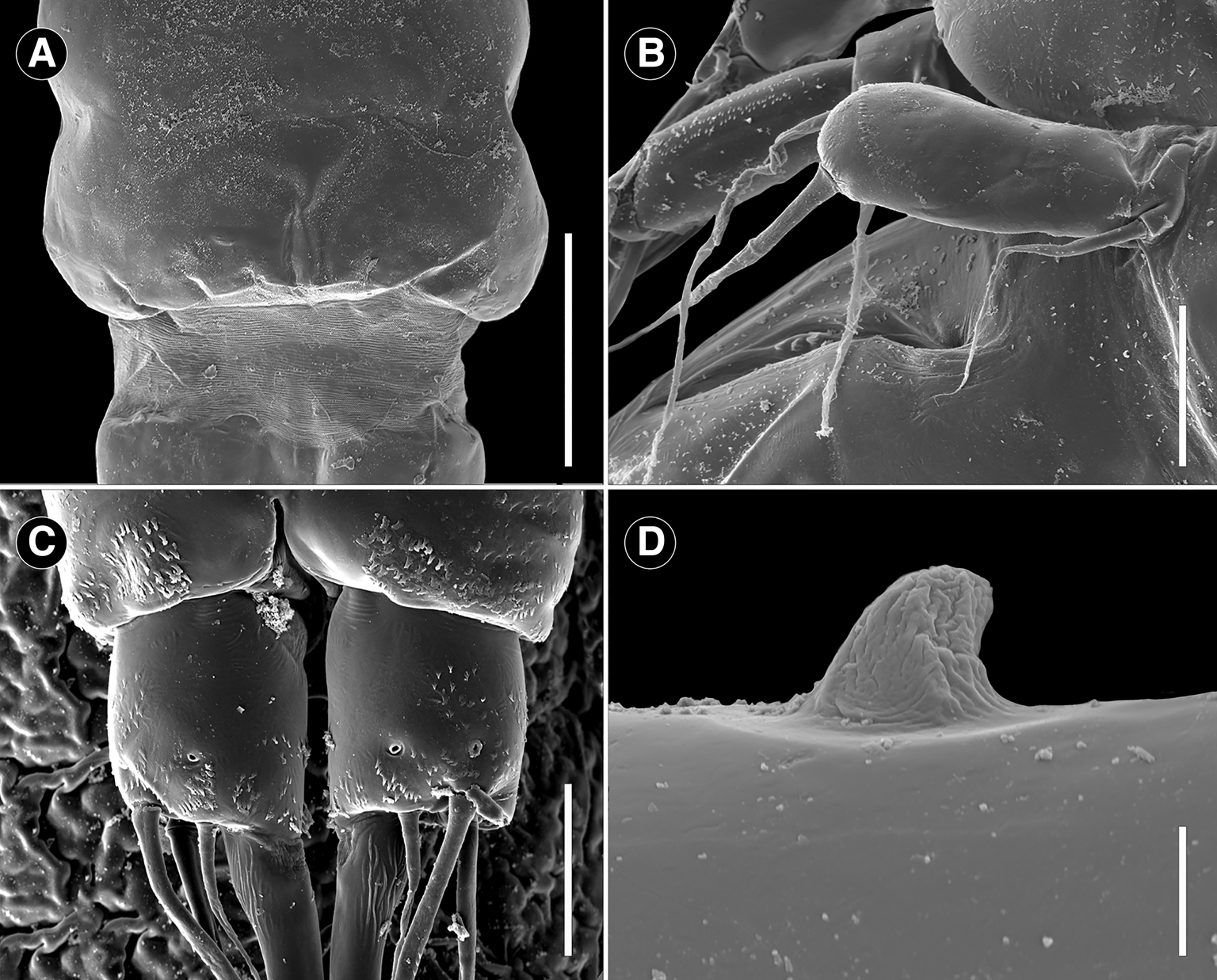
Scanning electron microscope photomicrographs of adult female Ergasilus arenalbus n. sp showing features from the ventral and dorsal view: (A) detail of the cuticular membrane of cephalothorax; (B) leg 5; (C) ventral view of detail of the caudal rami; (D) detail of the spine of the third antennal segment. Scale bars: A – 150 μm; B – 25 μm; C – 20 μm; D – 5 μm.
Type host: Amblyrhynchote honckenii (Bloch) (Tetraodontiformes: Tetraodontidae).
Type locality: Breede River Estuary, Witsand (−34.397323; 20.837474), Western Cape Province, South Africa.
Site on host: Gill filaments.
Prevalence of infection: 67% (10 of 15 pufferfish).
Type material: 151 Ergasilus specimens (adult females) were collected. Only adult females were examined: 6 were used for SEM; 2 for dissection; 12 for morphology; and 2 egg strings were used for DNA extraction. The hologenophores (NMB P 1044–NMB P 1045), holotype (NMB P 1042) and 11 paratypes (NMB P 1043) were deposited in the parasitological collections of the National Museum, Bloemfontein, South Africa; the remaining specimens are in the possession of the Water Research Group, North-West University, Potchefstroom, South Africa.
Representative DNA sequences: GenBank accession numbers and numbers of bases (bp) are given as follows: 18S: 1333 and 1344 bp long sequences of 2 specimens, accession numbers: PQ451954 and PQ451956; 28S: 668 and 664 bp long sequences of 2 specimens, accession numbers: PQ451957 and PQ451958; and COI: 701 bp long sequence of 1 specimen, accession number: PQ439339.
Etymology: The species name ‘arenalbus’ is derived from ‘arena albus’ meaning white sand (English) or wit sand (Afrikaans) in Latin. This refers to ‘Witsand’ the name of the type locality of this species.
Description
Adult female description (based on 12 specimens). Body length (measured from the anterior margin of cephalosome to posterior margin of caudal rami) 1182 (959–1370; n = 12). Body comprises prosome, urosome and caudal rami. Prosome 5-segmented, composed of cephalosome and 4 free pedigerous somites. Cephalothorax composed of cephalosome and first pedigerous somite; cephalosome separated dorsally from previous somite by flexible cuticle (Figs 2A and 5A). Cephalosome slightly shorter than wide, 336 (272–414; n = 12) long by 387 (316–460; n = 12) wide, oval to trapezoidal, with antennules and antenna visible in dorsal view. Cephalic ornamentation comprising of anterior circular eyespot and inverted T-shaped mark of thickened chitin situated medially on dorsal side (Fig. 2A and B). Paired sensory pores and papillae observed anterior to eyespot with numerous sensory papillae and pores scattered over dorsal surface of cephalosome. Rostrum well-developed, with truncated posterior margin. All pedigerous somites wider than long and progressively smaller. Paired sensory papillae observed mid-dorsally on segments 2–5. First pedigerous somite 176 (129–218; n = 12) long by 358 (297–428; n = 12) wide; second pedigerous somite 107 (82–154; n = 12) long by 274 (241–334; n = 12) wide; third pedigerous somite 105 (78–138; n = 12) long by 212 (163–268; n = 12) wide; fourth pedigerous somite 61 (44–82; n = 12) long by 140 (127–169; n = 12) wide.
Urosome comprising reduced fifth pedigerous somite, genital double somite and 3 free abdominal somites (Fig. 3A). Reduced fifth pedigerous somite 31 (20–39; n = 12) long by 80 (72–114; n = 12) wide. Genital double-somite longer than wide, 135 (120–152; n = 12) long by 109 (100–130; n = 12) wide (Fig. 3A), bearing a pair of multiseriate egg sacs dorsally (Figs 2A and 4A), measuring 1269 (989–1662; n = 12) long by 256 (222–340; n = 12) wide (Figs 2A and 4A). Abdomen 3-segmented; first abdominal somite widest, 43 (36–56; n = 12) long by 73 (64–92; n = 12) wide; second abdominal somite shorter, 31 (23–40; n = 12) long by 63 (55–67; n = 12) wide; third somite ( = anal somite) incised dorsoventrally ( = anal opening or anus) forming attachment for caudal rami, 28 (22–32; n = 12) long by 63 (59–68; n = 12) wide, ornamented with pair of pores on dorsal side; each pore located laterally to anal opening and carrying bristle (Figs 3A, 4E, 5C). All abdominal somites with posterior row of ventral spinules (Figs 4E and 5C).
Caudal rami slightly elongated, 30 (26–33; n = 12) long by 24 (22–28; n = 12) wide, with 4 setae (Fig. 3A). Innermost seta (IV) longest 282 (226–308; n = 12), followed by shortest seta (III) 28 (20–39; n = 12) and 2 longer setae (II and I) 77 (60–87; n = 12) and 92 (83–99; n = 12), respectively (Fig. 3A).
Antennule 6-segmented, armed with long and short setae (Fig. 2E). Setal formula from proximal to distal segments given as 3–12–6–2–3–8 (total 34). Antenna 4-segmented (Figs 2D and 4B) comprising coxobasis, 174 (110–196; n = 12) long by 95 (73–114; n = 12) wide; and 3-segmented endopod; armed with curved terminal claw (Figs 2D and 4B). First endopod segment longest 339 (288–366; n = 12), followed by second endopod segment 196 (152–215; n = 12) and small third endopod segment 28 (24–40; n = 12). Prominent spine observed on anterior second endopod segment (Figs 2D, 4B, 5D). Terminal claw pointed and smooth 161 (127–186; n = 12), with fossa on concave margin.
Mouth positioned ventrally on cephalosome. Labrum with internal teeth; teeth arranged in an arch. Mandible armed with 3 blades (anterior, medial and posterior blades); anterior blade thinner and shorter than others, ornamented along anterior margin; medial and posterior blades, both with teeth on opposite margin. Maxillule armed with 2 unequal setae; innermost seta shortest; ornamented with 1 pore and multiple spinules; pore lacking bristle (Fig. 2C). Maxilla 2-segmented, comprising syncoxa ( = first segment) and basis ( = second segment); syncoxa broad, with 2 distal pores; basis ornamented with multiple spinules on posterior margin. Labium broad, unornamented; mid-region produced posteriorly, with truncated posterior margin.
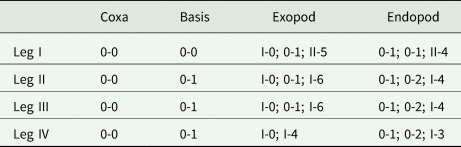
Swimming legs I to IV; each comprising coxa, basis and 2 segmented rami (i.e. exopod, endopod). Rami of all legs 3-segmented, except 2-segmented exopod of leg IV. Segments distinct, typical with similar basic morphology as in other species of Ergasilus. Armature on rami as Roman and Arabic numerals indicating spines and plumose setae, respectively, in Table 5.
Spine–setae formula on swimming legs of Ergasilus arenalbus n. sp.
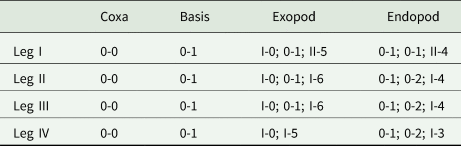
Number of spines in Roman numerals, number of setae in Arabic numerals.
Leg I (Fig. 3C). Coxa ornamented with spinules on outer margin. Basis armed with bare outer seta, ornamented with spinules on both sides; posterior margin protrudes posteriorly forming 1 spinous process; spines located between rami (Figs 3C and 4C). Exopod 3-segmented; first endopodal segment with distal spine, ornamented with patch of spinules on outer margin; spinules located just above distal spine; and bristles along outer margin; second exopodal segment with 1 plumose seta, unornamented; third exopodal segment armed with 2 serrated spines (inner and outer spine); inner spine about 2.0 times longer than outer spine; and 5 plumose setae. Endopod 3-segmented; all segments with spinules along inner margin; first and second endopodal segment, each with 1 plumose seta on inner margin; third endopodal segment armed with 2 serrated spines (inner and outer spine); inner spine about 2.0 times longer than outer spine; and 4 plumose setae.
Leg II (Fig. 3D). Coxa ornamented with spinules on outer margin. Basis armed with bare outer seta, ornamented with spinules on both sides. Exopod 3-segmented; first exopodal segment with distal spine, ornamented with patch of spinules on outer margin; spinules located just above distal spine; and bristles along inner margin; second exopodal segment armed with 1 plumose seta; third exopodal segment with simple spine (or non-serrated) and 6 plumose setae. Endopod 3-segmented; first and second endopodal segments with 1 and 2 plumose setae, respectively; third endopodal segment with serrated spine and 4 plumose setae. Leg III with the same armament and ornamentation described for leg II.
Leg IV (Fig. 3E). Coxa ornamented with spinules on both sides. Basis armed with bare outer seta, with spinules scattered across surface. Exopod 2-segmented; first exopodal segment armed with distal spine, ornamented with spinules and bristles on outer and inner margin, respectively; second exopodal segment with 1 spine and 5 plumose setae. Endopod 3-segmented, all segments lacking ornaments on both margins; first and second endopodal segment with 1 and 2 plumose setae, respectively; third endopodal segment with 1 serrated spine and 3 plumose setae.
Leg V (Fig. 3F) with single ramus. Ramus 2-segmented; proximal segment rectangular, without any armaments or ornaments; distal segment about 3.0 times longer than previous segment, with spinules scattered across surface, armed with 2 bare setae.
Intercoxal sclerites and interpodal plates of all legs, present (Figs 2A, 3B, 4D). Intercoxal sclerites unornamented, with both ends directed posteriorly. Interpodal plates present; first to third plate with spinules; fourth plate absent (Figs 2A and 3B).
Remarks
The detailed morphological description of E. arenalbus n. sp. sheds light on its distinctiveness among the recognized species of Ergasilus worldwide. With 163 valid species known, comparisons were only made with other marine Ergasilus species from the southern Atlantic and Indian Ocean regions. Among the 17 marine species from these regions (Table 1), E. arenalbus n. sp. stands out in several key morphological aspects, notably in size; armature of the antenna; the segmentation in the body (free vs fused prosome somites); and general ornamentation throughout the entire body. Firstly, its larger body size, with a length averaging 1182 μm, sets it apart from species such as E. atafonensis Amado and Rocha, Reference Amado and Rocha1996, E. bahiensis Amado and Rocha, Reference Amado and Rocha1996, E. caraguatatubensis Amado and Rocha, Reference Amado and Rocha1996, E. ilani Oldewage and van As, Reference Oldewage and van As1988, E. myctarothes Wilson, Reference Wilson1913, E. parvitergum Ho et al., Reference Ho, Jayarajan and Radhakrishnan1992, E. rostralis Ho et al., Reference Ho, Jayarajan and Radhakrishnan1992 and E. uniseriatus Ho et al., Reference Ho, Jayarajan and Radhakrishnan1992 which typically have a smaller total length, below 1054 μm. Conversely, E. felichthys (Pearse, Reference Pearse1947) and E. youngi Tavares and Luque, Reference Tavares and Luque2005 present larger body sizes, approximately 1400 μm, emphasizing the distinctive size range of the newly described species. The segmentation of the body, particularly the free vs fused prosome somites, is another distinguishing factor. For instance, E. arenalbus n. sp. shows variations in abdominal somite dimensions, contrasting with the more uniform structures seen in species like E. atafonensis. This variability extends to cephalosome characteristics, with E. caraguatatubensis exhibiting an inflated shape absent in E. arenalbus n. sp. The spine-setae formulae on the swimming legs of E. arenalbus n. sp. further differentiate it, particularly when compared to all other marine congeners, except for E. lizae Krøyer, 1863. Ergasilus atafonensis, E. bahiensis, E. myctarothes, E. parvitergum and E. xenomelanirisi Carvalho, Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León1955, have a spine on the outer margin of the second exopodite of leg I, which is absent in the new species. In addition, E. ilani lacks a certain number of armaments that are common on the legs of the species in the group, for example, spines and setae on the first segments of the exopod and endopod, respectively. In E. caraguatatubensis, E. felichthys, E. foresti Boxshall et al., Reference Boxshall, Araujo and Montu2002, E. ilani, E. parvitergum and E. youngi, leg V is extremely reduced and is represented by 1 or 2 setae. While the setae formula of the antennule is also a clear distinguishing factor, species like E. ilani, E. rostralis and E. uniseriatus were further excluded due to their antennules being described as only 5-segmented, unlike the 6-segmented antennules observed in the other Ergasilus species. Although E. arenalbus n. sp. morphologically resembles E. lizae in many aspects, it differs notably in the armature of the antenna, with E. arenalbus n. sp. exhibiting a single spine on the anterior second endopod segment, unlike E. lizae. Additionally, a distinctive feature of the new species is the spine projections on the posterior margin of the basis of the first leg, a characteristic absent in all other species examined. This comprehensive morphological analysis of E. arenalbus n. sp. provides a clear understanding of its unique features within the Ergasilus genus, emphasizing size, body segmentation, appendage armature and ornamentation as crucial factors in species differentiation.
Ergasilus chintensis n. sp.: Figures 6–7
ZooBank LSID: urn:lsid:zoobank.org:act:E51F54EC-9CF7-47DA-B4B7-61DEC73B430F.
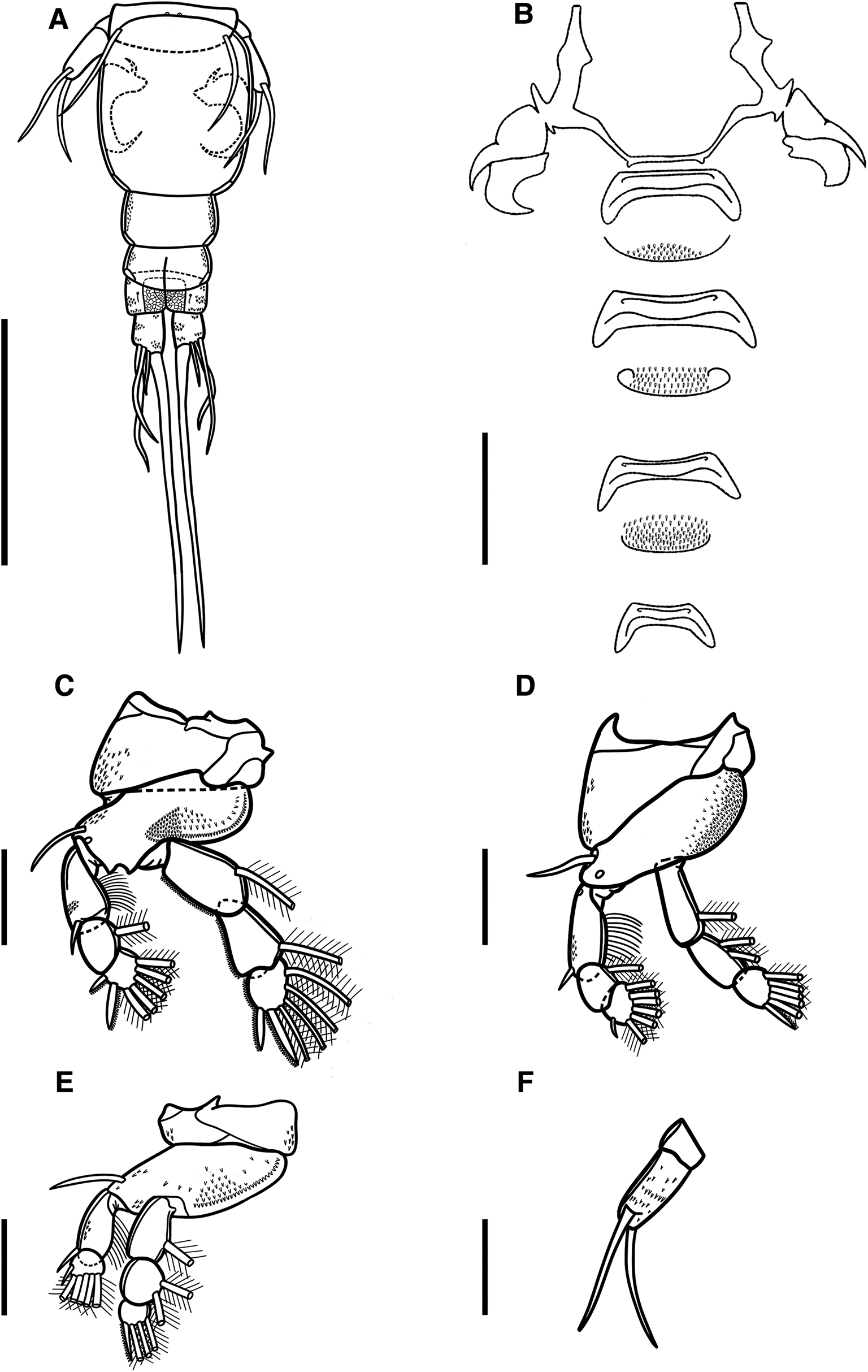
Illustrations of adult female of Ergasilus chintensis n. sp: (A) entire specimen, dorsal view; (B) detail of the cephalosome, dorsal view; (C) mouth, mandible, maxillule and maxilla; (D) antennule; (E) antenna. Scale bars: A – 500 μm; B – 250 μm; C–E – 100 μm.
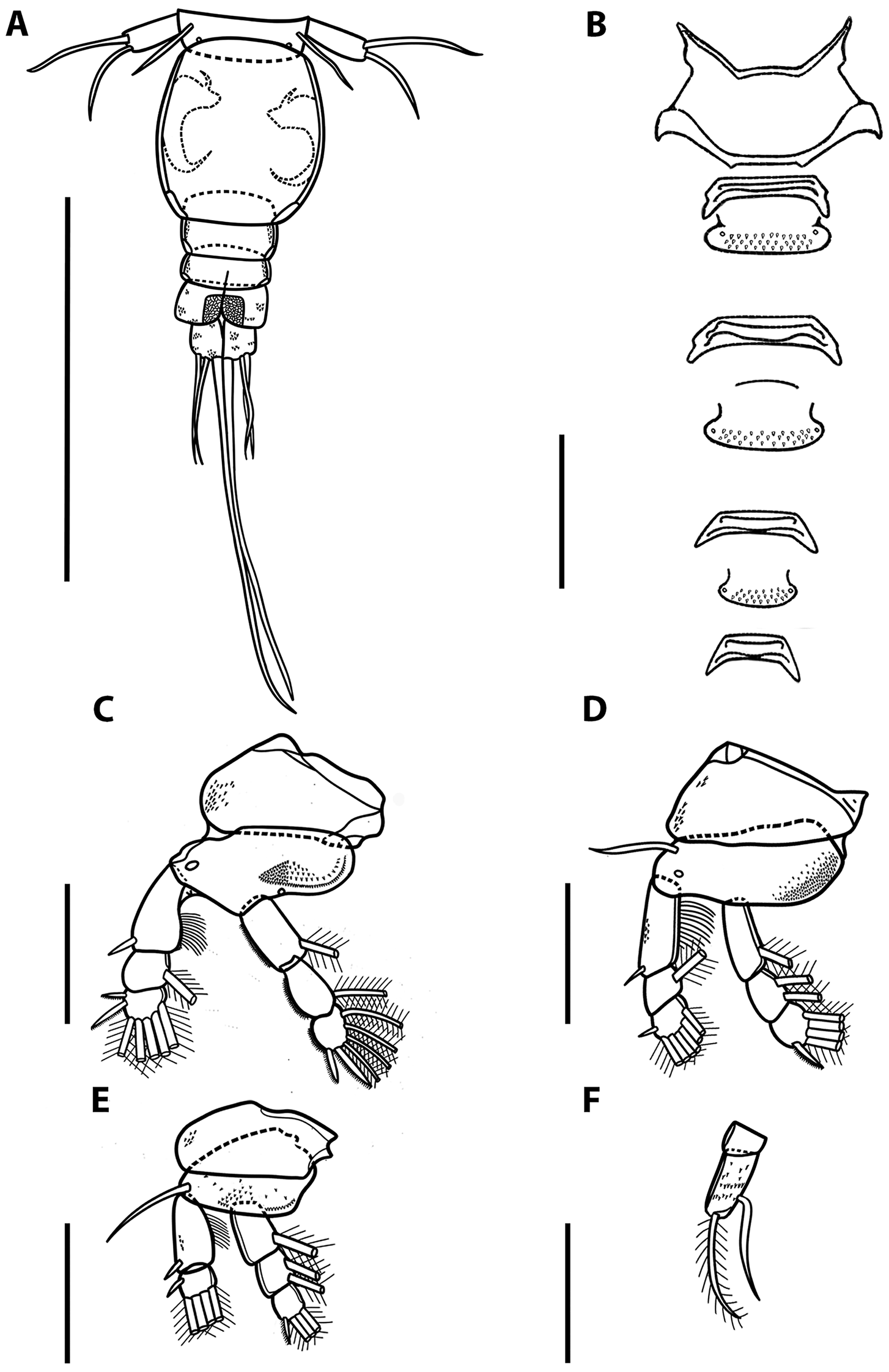
Illustrations of adult female of Ergasilus chintensis n. sp: (A) urosome, dorsal view; (B) intercoxal sclerites and interpodal plates; (C) leg 1; (D) leg 2 and leg 3; (E) leg 4; (F) leg 5. Scale bars: A – 200 μm; B – 100 μm; C–F – 50 μm.
Type host: Amblyrhynchote honckenii (Bloch) (Tetraodontiformes: Tetraodontidae).
Type locality: Chintsa East (−32.836538; 28.116997), Eastern Cape Province, South Africa.
Site on host: Gill filaments.
Prevalence of infection: 20% (2 of 10 pufferfish observed).
Type material: 2 ergasilids (adult females) were collected. Only adult females were examined: 2 were used for morphology, and 1 egg string was used for DNA extraction. The hologenophore (NMB P 1047) and holotype (NMB P 1046) were deposited in the parasitological collections of the National Museum, Bloemfontein, South Africa.
Representative DNA sequences: GenBank accession numbers and numbers of bases (bp) are given as follows: 18S: 1353 bp long sequence of 1 specimen, accession number: PQ451955; 28S: 668 bp long sequence of 1 specimen, accession number: PQ451959; and COI: 692 bp long sequence of 1 specimen, accession number: PQ439340.
Etymology: The species name ‘chintensis’ is derived from Chintsa, representing the type locality of the species.
Description
Adult female description (based on 2 specimens). Body length (measured from the anterior margin of cephalosome to the posterior margin of caudal rami) 1035 (1002–1068; n = 2). Body comprises prosome, urosome and caudal rami. Prosome composed of cephalosome, fused somites (first to third pedigerous somites) and fourth free somite (Fig. 6A). Cephalosome slightly shorter than wide, oval to trapezoidal, with antennules and antenna visible in dorsal view. Cephalic ornamentation comprising of anterior circular eyespot and an inverted T-structure of thickened chitin situated medially on dorsal side (Fig. 6A and B). Paired sensory pores and papillae observed anterior to eyespot with numerous sensory papillae and pores scattered over the dorsal surface of cephalosome. Rostrum well-developed, with truncated posterior margin. All pedigerous somites wider than long and progressively smaller. Second pedigerous somite 104 (98–110; n = 2) long by 292 (287–297; n = 2) wide; third pedigerous somite 106 (99–113; n = 2) long by 224 (217–231; n = 2) wide; fourth pedigerous somite 40 (38–42; n = 2) long by 112 (110–114; n = 2) wide.
Urosome comprising reduced fifth pedigerous somite, non-pedigerous barrel-shaped genital double somite, and 3 free abdominal somites (Fig. 7A). Reduced fifth pedigerous somite 14 (10–18; n = 2) long by 83 (72–94; n = 2) wide. Genital double-somite longer, 113 (110–115; n = 2), than wide, 88 (86–90; n = 2) (Fig. 7A), bearing a pair of multiseriate egg sacs dorsally, measuring 1098 (1101–1095; n = 2) long by 190 (192–188; n = 2) wide (Fig. 6A). Abdomen 3-segmented, first abdominal somite widest, 28 (24–32; n = 2) long by 53 (51–55; n = 2) wide, second abdominal somite shorter, 23 (20–25; n = 2) long by 50 (49–51; n = 2) wide; third somite ( = anal somite) incised dorsoventrally ( = anal opening or anus) forming attachment for caudal rami, 21 (17–25; n = 2) long by 46 (45–47; n = 2) wide (Fig. 7A). All abdominal somites with posterior row of ventral spinules.
Caudal rami slightly elongated, 23 (22–25; n = 2) long by 18 (17–18; n = 2) wide, with 4 setae (Fig. 7A). Innermost seta (IV) longest 184 (182–186; n = 2), followed by shortest seta (III) 24 (23–24; n = 2) and 2 longer setae (II and I) 55 (54–56; n = 2) and 59 (57–60; n = 2), respectively (Fig. 7A). Two sensory pores on posterior ventral margins on each ramus.
Antennule 6-segmented, armed with long and short setae (Fig. 6D). Setal formula from proximal to distal segments given as 3–10–6–3–2–6 (total 30). Antenna 4-segmented, comprising of coxobasis, 99 (97–101; n = 2) long by 61 (58–65; n = 2) wide; and 3-segmented endopod, armed with curved terminal claw (Fig. 6E). First endopod segment longest 182 (181–184; n = 2), followed by second endopod segment 110 (108–112; n = 2), and small third endopod segment 14 (13–15; n = 2). Two spines observed on second endopod segment. Terminal claw pointed and smooth 83 (81–85; n = 2), with fossa on inner margin (Fig. 6E).
Mouth positioned ventrally on cephalosome. Mandible armed with 3 blades (anterior, medial and posterior blades); anterior blade thinner and shorter than others, ornamented along anterior margin; medial and posterior blades, both with teeth on opposite margin (Fig. 6C). Maxillule armed with 2 bare setae. Maxilla 2-segmented, comprising syncoxa ( = first segment) and basis ( = second segment); syncoxa broad with large maxillary pore; basis distally ornamented with numerous teeth on convex margin. Labium broad, unornamented; mid-region produced posteriorly, with rounded posterior margin.
Swimming legs I to IV; each comprising coxa, basis and 2 segmented rami (i.e. exopod, endopod). Rami of all legs 3-segmented, except 2-segmented exopod of leg IV. Segments distinct, typical with similar basic morphology as in other species of Ergasilus. Armature on rami as follows (Roman and Arabic numerals indicating spines and plumose setae, respectively) in Table 6.
Spine–setae formula on swimming legs of Ergasilus chintensis n. sp.
Number of spines in Roman numerals, number of setae in Arabic numerals.
Leg I (Fig. 7C). Coxa ornamented with spinules on outer margin. Basis lacking outer setae, ornamented with spinules on inner margin. Exopod 3-segmented; first segment with small outer spine; second segment with 1 inner plumose seta, lacking spine; third segment with small spine on outer corner, longer apical spine; both spines with serrated margins; and 5 plumose setae. Endopod 3-segmented; first and second segment each with 1 plumose seta; third segment with 4 plumose setae and 2 distal serrated spines.
Leg II (Fig. 7D). Coxa ornamented with spinules on outer margin. Basis with outer seta and pore, ornamented with multiple spinules on inner margin. Exopod 3-segmented; first segment with small outer spine; second segment with 1 plumose seta, lacking spine; third segment with small spine on outer corner and 6 plumose setae. Endopod 3-segmented; first segment with 1 plumose seta; second segment with 2 plumose setae; third segment with 4 plumose setae and 1 distal serrated spine (Fig. 7D). Leg III (Fig. 7D) with same ornamentation and armament described for leg 2.
Leg IV (Fig. 7E). Coxa ornamented with spinules on outer margin. Basis with outer seta, ornamented with multiple spinules on inner margin. Exopod 2-segmented; first segment with small outer spine; second segment with small spine on outer corner and 4 plumose setae. Endopod 3-segmented; first segment with 1 plumose seta; second segment with 2 plumose setae; third segment with 3 plumose setae and 1 distal serrated spine.
Leg V (Fig. 7F) with single ramus. Ramus 2-segmented: proximal segment rectangular, without any armaments or ornaments; distal segment about 2.5 times longer than previous segment, with spinules scattered across surface, bearing 2 setae (lateral and inner setae); lateral seta plumose.
Intercoxal sclerites of all legs, present (Figs 6A and 7B); each sclerite with both ends directed posteriorly. Interpodal plates of leg I to III ornamented with spinules; fourth plate absent (Fig. 7B).
Remarks
The detailed description of E. chintensis n. sp. highlights its unique characteristics among Ergasilus species worldwide, especially compared to marine congeners from these geographic regions. Notably, similar to E. arenalbus n. sp., its body size averaging 1035 μm sets it apart from both larger species like E. felichthys and E. youngi, ranging around 1400 μm, and also from species with smaller sizes below 1054 μm, such as E. atafonensis, E. bahiensis, E. caraguatatubensis, E. ilani, E. myctarothes, E. parvitergum, E. rostralis and E. uniseriatus. Similarities emerge due to their shared size range when comparing the 2 newly described South African species; however, subtle proportional variations in body segments and appendages are key to their differentiation upon closer examination. Additionally, E. chintensis n. sp. displays a more intricate armature on its antenna segments than E. arenalbus n. sp., where the second endopod segment in this species shows 2 developed spines rather than just 1 as present in E. arenalbus n. sp. The most striking feature of E. chintensis n. sp. is its body segmentation, characterized by a fused 2-segmented prosome. This completely contrasts with the free prosome somites observed not only in E. arenalbus n. sp. but also concerning all the other compared marine ergasilid species. Ergasilus caraguatatubensis also exhibits a fused prosome structure, although, to a lesser degree than E. chintensis n. sp. The antennule setae formula also contributes to this distinction, with E. chintensis n. sp. differing from E. ilani, E. rostralis and E. uniseriatus in antennule segmentation (6- vs 5-segmented). These distinguishing features collectively characterize E. chintensis n. sp. within the Ergasilus genus, highlighting body segmentation, appendage armature and ornamentation as key characteristics for species differentiation.
Molecular characterization and phylogenetic position of African marine Ergasilus species
The molecular analyses revealed distinct genetic profiles for the newly described Ergasilus species. This study successfully generated a total of 8 sequences. For E. arenalbus n. sp., 5 sequences were produced: 2 18S, 2 28S and 1 COI sequence. For E. chintensis n. sp., 3 sequences were obtained: 1 18S, 1 28S and 1 COI sequence. Nucleotide comparisons of the 2 new species against the partial 18S rDNA, 28S rDNA and COI mtDNA gene sequences of the genus Ergasilus were performed, as detailed in Tables 7–9, respectively. Both ML and BI analyses were conducted on the partial 18S rDNA, 28S rDNA and COI gene alignments, producing phylogenetic trees with congruent topologies. Thus, only the ML tree of the 28S and COI gene regions are presented (Figs 8 and 9, respectively).
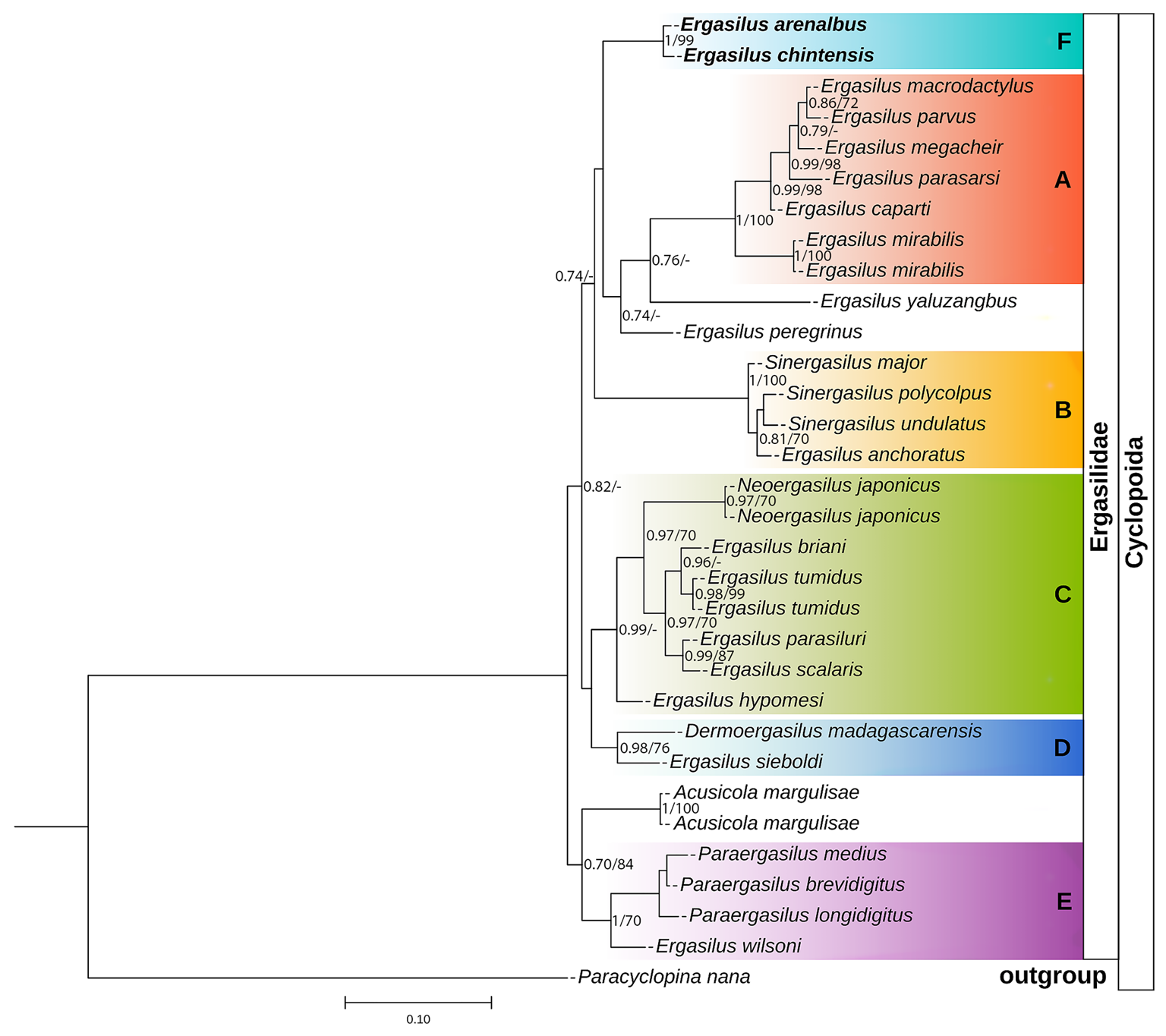
Phylogenetic tree of Ergasilidae copepods based on partial 28S rRNA gene alignments. Newly generated sequences for Ergasilus arenalbus n. sp. and Ergasilus chintensis n. sp. are provided in bold. Nodal support presented above or below branches for Bayesian Inference (>0.7) and Maximum Likelihood (>70%) analyses (BI/ML). Dashes indicate values below 0.7 and 70%, respectively. Paracyclopina nana Smirnov, 1935, was used as the outgroup.
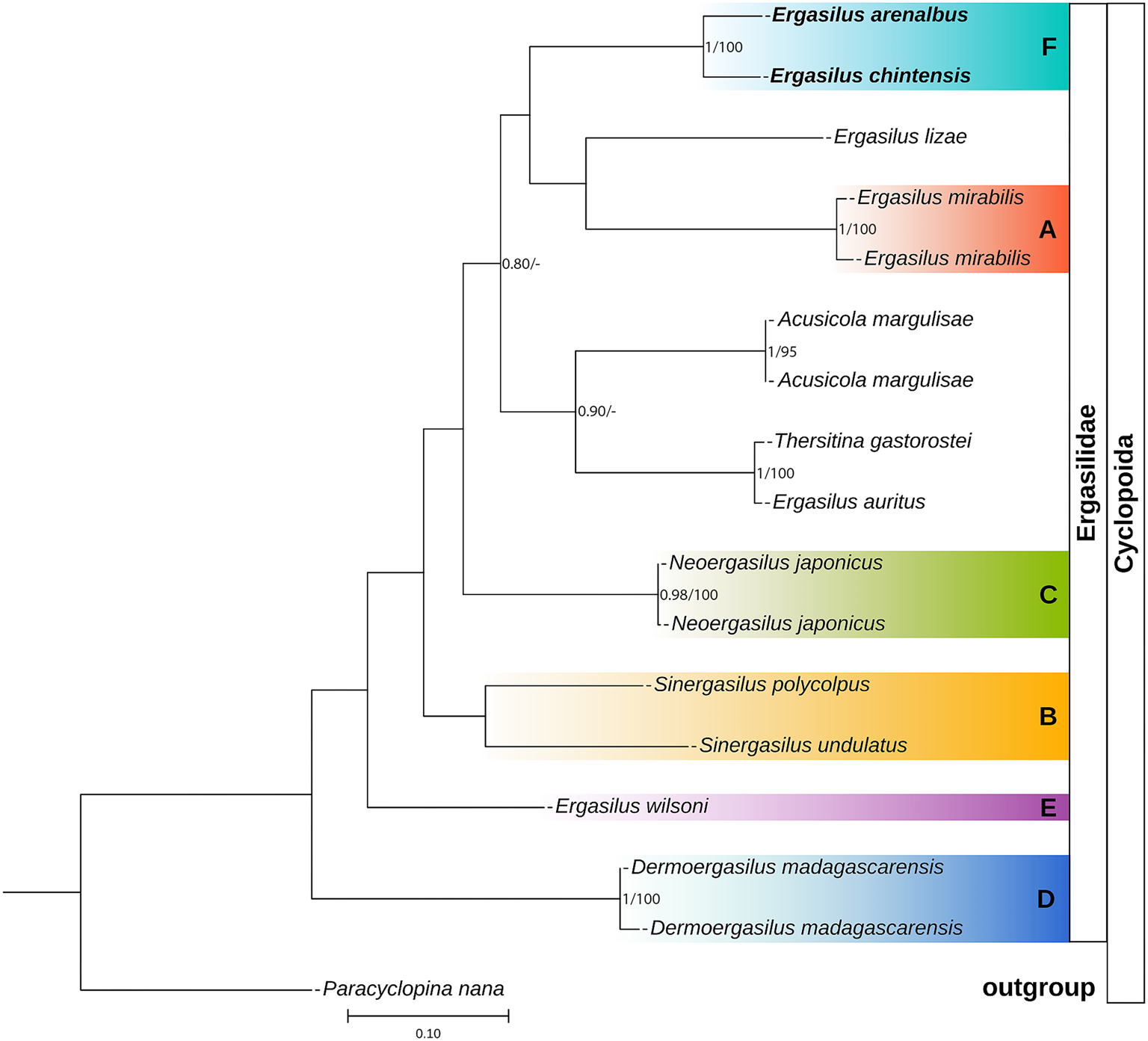
Phylogenetic tree of Ergasilidae copepods based on partial COI mtDNA gene alignments. Newly generated sequences for Ergasilus arenalbus n. sp. and Ergasilus chintensis n. sp. are provided in bold. Nodal support presented above or below branches for Bayesian Inference (>0.7) and Maximum Likelihood (>70%) analyses (BI/ML). Dashes indicate values below 0.7 and 70%, respectively. Paracyclopina nana Smirnov, 1935, was used as the outgroup.
Nucleotide comparison of the partial 18S rDNA sequences of the genus Ergasilus Nordman, 1832, based on 1354 bp-long alignment.
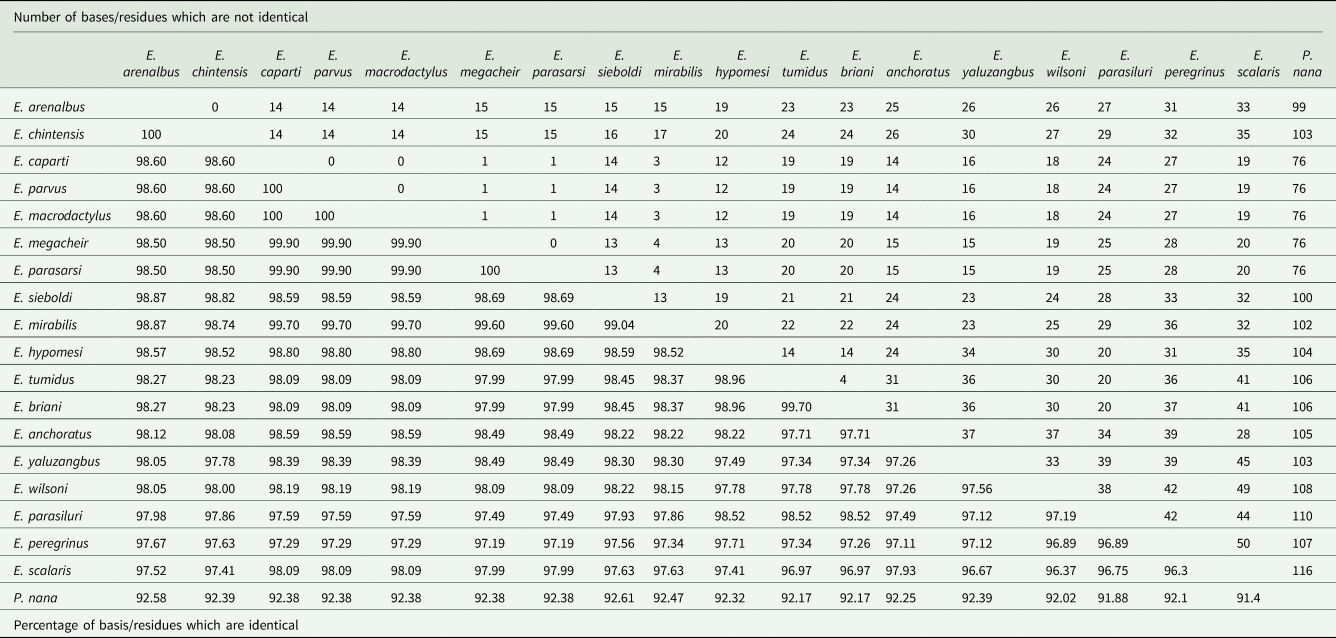
Nucleotide comparison of the partial 28S rDNA sequences of genus Ergasilus Nordman, 1832, based on 682 bp-long alignment

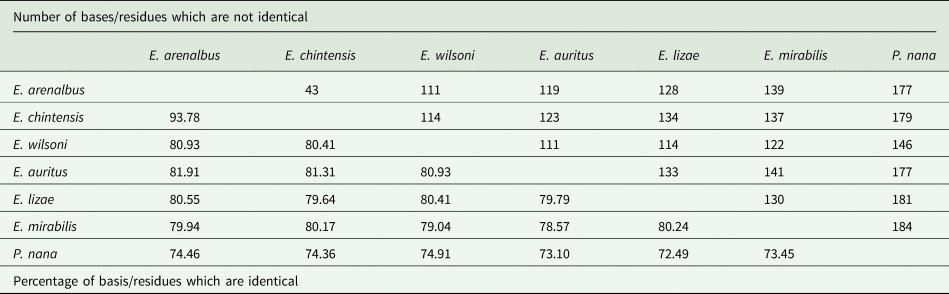
Nucleotide comparison of the mtDNA COI gene sequences of genus Ergasilus Nordman, 1832, based on 700 bp-long alignment.
The 18S phylogenetic analyses yielded a final alignment consisting of 1354 bases. The 18S sequences exhibited no interspecific variability (0 bp difference) since no differences were found among the 18S rDNA sequences of E. arenalbus n. sp. and E. chintensis n. sp. (Table 7). The analysis revealed E. scalaris Markevich, 1940, as the most genetically distant species from E. arenalbus n. sp. (33 bp/2.48%) and E. chintensis n. sp. (35 bp/2.59%) (Table 7). The lowest interspecific differences (1.13–1.18%) were observed between the new species and E. sieboldi von Nordmann, 1832 (Table 7).
The 28S phylogenetic analysis produced a final alignment of 682 bases. These 28S sequences displayed minor interspecific variability (6 bp) among the 28S rDNA sequences of E. arenalbus n. sp. and E. chintensis n. sp. (Table 8). The analysis showed E. parasarsi Míč et al., Reference Míč, Řehulková and Seifertová2023, and E. yaluzangbus Kuang and Qian, 1985, as the most genetically distant species from E. arenalbus n. sp. (71 bp/10.70%) and E. chintensis n. sp. (72 bp/10.68%), respectively (Table 8). The smallest interspecific differences (5.39–5.60%) were noted between the new species and E. wilsoni Markevich, 1933 (Table 8). ML and BI analyses using rDNA alignment that included partial 28S sequences of Ergasilidae produced trees with consistent topologies and similar nodal support values. Consistent with previous phylogenetic studies on ergasilids (Song et al., Reference Song, Wang, Yao, Gao and Nie2008; Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024), the analyses identified 5 well-supported polyphyletic Ergasilus groups (Fig. 8): (A) the African freshwater Ergasilus species, (B) the Sinergasilus Yin, 1949, species and the E. anchoratus Markevich, 1946, group, (C) the Asian Ergasilus species and the Neoergasilus japonicus (Harada, 1930) group, (D) the recently described Dermoergasilus madagascarensis Míč et al. , Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024, and E. sieboldi group and (E) the Paraergasilus Markevich, 1937, species and the E. wilsoni group (Fig. 8). Despite forming a distinct subclade (F), the newly described species still clustered within the larger clade that includes subclade (A) comprising African freshwater species, along with non-African species like E. yaluzangbus and E. peregrinus Heller, 1865 (Fig. 8).
COI sequences were aligned using invertebrate mitochondrial translation, resulting in an alignment length of 700 bases. The sequences included GenBank and BOLD sequences submitted from Canada (Table 4). These results displayed substantial interspecific variability (43 bp) among the COI sequences of E. arenalbus n. sp. and E. chintensis n. sp. and more than 110 bases from all other Ergasilus congeners (Table 9). The analysis showed E. mirabilis Oldewage and van As, 1987, and E. lizae as the most genetically distant species from E. arenalbus n. sp. (139 bp/20.06%) and E. chintensis n. sp. (134 bp/20.36%), respectively (Table 9). The smallest interspecific differences (19.07–19.59%) were noted between the new species and E. wilsoni (Table 9). Similar to the 28S tree topology, the novel sequences of E. arenalbus n. sp. and E. chintensis n. sp. formed the same subclade (F), within the larger clade that includes subclade (A) comprising African freshwater species, E. mirabilis, and the morphologically similar marine species E. lizae, which is also found within the Indian Ocean (Fig. 9).
Based on these analyses, this study proposes the existence of a sixth clade (F) consisting of African marine Ergasilus species (Figs 8 and 9). However, this proposition remains speculative as their precise phylogenetic placement within Ergasilidae remains unresolved due to low support values and limited molecular data, concerning marine ergasilids.
Discussion
The discovery of 2 new Ergasilus species, E. arenalbus n. sp. and E. chintensis n. sp., from the Evileye blaasop, A. honckenii, significantly enhances our understanding of marine parasite diversity in South Africa. These findings highlight the underexplored nature of marine parasites in this region, particularly within the genus Ergasilus, known for its rich diversity in global freshwater and marine environments (Boxshall and Defaye, Reference Boxshall and Defaye2008; Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; Míč et al., Reference Míč, Řehulková and Seifertová2023). To date, only a limited number of Ergasilus species have been reported from African marine environments, with only 5 documented, including just 1 from South Africa (Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023; WoRMS, 2024). Moreover, despite Ergasilus being found in a wide range of fish host families (see Table 1), only a single species, E. colomesus Thatcher and Boeger, Reference Thatcher and Boeger1983, has been described from the family Tetraodontidae Bonaparte, 1832 (Thatcher and Boeger, Reference Thatcher and Boeger1983) in the Amazon River, Brazil.
The addition of E. arenalbus n. sp. and E. chintensis n. sp. not only introduces new host records but also suggests a higher hidden diversity of Ergasilus within South Africa's coastal region and the Tetraodontidae family. This hints at a potentially broader copepod diversity and novel host–parasite relationships yet to be explored, aligning with global trends revealing extensive species diversity in under-studied marine ecosystems (Boxshall and Defaye, Reference Boxshall and Defaye2008). The presence of these new species along the South African coastline highlights the region's rich marine biodiversity and emphasizes the importance of investigating lesser-known areas and hosts for hidden parasite diversity.
Taxonomically, detailed morphological examinations of these species, focusing on size, body segmentation, appendage armature and ornamentation, provide crucial insights into their distinctiveness from known congeners. Integrating traditional morphological taxonomy with molecular techniques has been instrumental in characterizing these new Ergasilus species. Molecular analyses alongside morphological assessments confirm the uniqueness of these species with greater accuracy (Míč et al., Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024; Walter and Boxshall, Reference Walter and Boxshall2024). This integration is valuable as morphological characters alone often yield conflicting results in distinguishing new species and understanding their placement within Ergasilidae lineages (Míč et al., Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024; Walter and Boxshall, Reference Walter and Boxshall2024).
The nomenclatural history of ergasilids emphasizes the significant challenge of formulating generic diagnoses that effectively distinguish species. This complexity is evidenced by the synonymization of 33 Ergasilus species either with other previously described Ergasilus species or with species from different genera within the Ergasilidae family (Walter and Boxshall, Reference Walter and Boxshall2024). Moreover, genera such as Acusicola Cressey, in Cressey and Collette, 1970, Dermoergasilus Ho and Do, 1982, Neoergasilus Yin, 1956, Paraergasilus and Sinergasilus, have consistently been confirmed as monophyletic (Song et al., Reference Song, Wang, Yao, Gao and Nie2008; Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Kvach et al., Reference Kvach, Tkachenko, Seifertová and Ondračková2021; Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024). However, these genera render Ergasilus polyphyletic, with certain species, like E. anchoratus, E. sieboldi and E. wilsoni, showing closer relationships to Sinergasilus, Dermoergasilus and Paraergasilus, respectively (Figs 8 and 9).
Phylogenetics pose significant challenges that hinder comprehensive genomic analyses of Ergasilus species, creating obstacles in gaining deeper insights into their biology. The limited knowledge regarding their diversity and the lack of genetic data, compared to those of other well-studied organisms, are major contributing factors. Previous studies (Song et al., Reference Song, Wang, Yao, Gao and Nie2008; Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Kvach et al., Reference Kvach, Tkachenko, Seifertová and Ondračková2021; Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024) have attempted to overcome these challenges through molecular characterization using rRNA genes, particularly the 18S and 28S rDNA regions. However, the effectiveness of these markers in species-level differentiation has been variable (Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024). The 18S rDNA region of the present study has shown minimal or even zero variation (0–2.59%) in some cases, making it unsuitable for distinguishing between closely related species. This is consistent with findings from earlier studies, which reported that the 18S rRNA gene is highly conserved and not suitable for identification at lower taxonomic levels (Taniguchi et al., Reference Taniguchi, Kanehisa, Sawabe, Christen and Ikeda2004; Huys et al., Reference Huys, Llewellyn-Hughes, Olson and Nagasawa2006; Tang et al., Reference Tang, Leasi, Obertegger, Kieneke, Barraclough and Fontaneto2012; Marrone et al., Reference Marrone, Lo Brutto, Hundsdoerfer and Arculeo2013). This lack of variation reinforces the limitations of the 18S rDNA marker for species-level differentiation within this genus.
In contrast, the 28S rDNA analyses have proven more effective in distinguishing between species (Song et al., Reference Song, Wang, Yao, Gao and Nie2008; Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Kvach et al., Reference Kvach, Tkachenko, Seifertová and Ondračková2021; Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024). The present study supports these findings to an extent, revealing higher, albeit minor interspecific divergence (6 bp between E. arenalbus n. sp. and E. chintensis n. sp.). The analysis also identified other Ergasilus species as genetically distant from the newly described taxa, highlighting the potential of 28S rDNA in elucidating phylogenetic relationships within the family Ergasilidae. However, despite its relative effectiveness, the genetic variation observed in the 28S rDNA is still limited, with little variation (0.90–10.70%), compared to other markers, raising questions about its adequacy for reliable species identification.
The COI gene, a widely used barcode for species-level differentiation (Tang et al., Reference Tang, Leasi, Obertegger, Kieneke, Barraclough and Fontaneto2012; Baek et al., Reference Baek, Jang, Choi, Ryu, Kim, Lee, Lim, Lee, Jun, Kwak, Lee, Hwang, Maran, Chang, Kim and Hwang2016; Mayor et al., Reference Mayor, Galimova, Sheveleva, Sukhanova and Kirilchik2017; Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024), demonstrated high resolution at the species level for the Ergasilus species described in this study and indicated significant interspecific variability. The COI analyses revealed substantial differences (43 bp) among the COI sequences of E. arenalbus n. sp. and E. chintensis n. sp., as well as notable variation from other Ergasilus congeners. This suggests that the COI gene may be a more suitable marker for species-level differentiation in this group. However, it is important to note that the limited availability of only 4 other Ergasilus COI sequences (see Fig. 9 and Table 9) means that drawing definitive conclusions from these results is premature. While COI shows promise for more precise species identification, further research is needed to expand the dataset and validate its effectiveness across a broader range of Ergasilus species. Moving forward, prioritizing the COI gene in future studies may provide a clearer understanding of the diversity and evolutionary relationships within the family Ergasilidae.
The phylogenetic relationships of ergasilid copepods remain largely unclear, with only 11% (31 out of 277) of the known species with any molecular data available (Míč et al., Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024; Walter and Boxshall, Reference Walter and Boxshall2024). Limited studies have examined the genetic characteristics of African Ergasilus species (Fikiye et al., Reference Fikiye, Smit, Van As, Truter and Hadfield2023, Míč et al., Reference Míč, Řehulková and Seifertová2023), and no genetic studies exist for the characterization of marine species. Notably, the present study provides the first marine sequences for this genus. The only available brackish sequences are for Ergasilus wilsoni and Ergasilus sieboldi (Walter and Boxshall, Reference Walter and Boxshall2024), both of which are primarily associated with freshwater environments rather than being strictly marine or brackish. Ergasilus wilsoni and E. sieboldi can inhabit fresh or brackish waters, while recognized as typical freshwater species found in the Palearctic region, particularly in rivers and lakes (Kvach et al., Reference Kvach, Tkachenko, Seifertová and Ondračková2021), distinguishing them from the newly discovered Ergasilus species, as close relationships among ergasilids may be influenced by the geographical origin of the species or the endemism of their hosts (Míč et al., Reference Míč, Řehulková and Seifertová2023). This means that parasite distribution is closely linked to the geographic distribution of their hosts (Morand and Guégan, Reference Morand and Guégan2000), suggesting potential coevolution between parasites and hosts. The discovery of 2 genetically similar species from the same host species suggests that the 2 newly identified Ergasilus species associated with pufferfish along the South African coast reflect coevolutionary patterns and host-specific endemism. These findings are consistent with conclusions drawn from previous phylogenetic studies (Song et al., Reference Song, Wang, Yao, Gao and Nie2008; Santacruz et al., Reference Santacruz, Morales-Serna, Leal-Cardín, Barluenga and Pérez-Ponce de León2020; Kvach et al., Reference Kvach, Tkachenko, Seifertová and Ondračková2021; Oliveira et al., Reference Oliveira, Corrêa, Adriano and Tavares-Dias2021). Therefore, the geographic separation between E. sieboldi and E. wilsoni from the newly discovered species further highlights the critical need for more comprehensive genetic data on marine Ergasilus species to enhance our understanding of their diversity, evolutionary relationships and distribution patterns.
Conclusion
The discovery and descriptions of E. arenalbus n. sp. and E. chintensis n. sp. in association with the Evileye blaasop represent a significant advance in our understanding of marine parasite diversity in South Africa. These results highlight the rich marine ecosystems of the region and emphasize the importance of investigating under-explored areas to uncover the hidden biodiversity. Furthermore, the new Ergasilus sequences and phylogenetic analyses presented in this study provide the first insight into the phylogenetic relationships of marine Ergasilus species within the South Atlantic and Indian Ocean regions. Alongside the studies by Míč et al. (Reference Míč, Řehulková and Seifertová2023, Reference Míč, Řehulková, Šimková, Razanabolana and Seifertová2024) and Fikiye et al. (Reference Fikiye, Smit, Van As, Truter and Hadfield2023), this research also offers a further understanding of the African clade lineage, making the molecular data presented here the first to elucidate the phylogenetic relationships of this genus in African and marine systems. Our phylogenetic analysis suggests that African marine ergasilids form a distinct monophyletic lineage separate from freshwater species, proposing the recognition of a sixth clade (F) for African marine Ergasilus species (Figs 8 and 9). Nonetheless, due to low support values and the scarcity of molecular data for marine ergasilids, their exact phylogenetic placement within the family Ergasilidae remains unresolved. Further studies that integrate both morphological and molecular data are essential to elucidate these relationships.
Data available statement
Sequence data are available on the NCBIGenBank database. All other necessary data are included in the article.
Acknowledgements
We would like to thank the Unit for Environmental Sciences and Management, North-West University (NWU) for the use of field equipment and laboratory facilities. We thank Anja Erasmus from NWU-WRG, for the creation of the study area map. Additional thanks go to our colleagues Francois Retief, Reese Alberts, Anja Erasmus, Marliese Truter, Anja Vermaak and Coret van Wyk, from the NWU who assisted with the collection of samples. This is contribution No. 920 from the NWU-Water Research Group.
Author contributions
L. van der Spuy: methodology, investigation, funding acquisition, writing – original draft. R. B. Narciso: methodology, investigation, drawing, writing – review and editing. K. A. Hadfield: methodology, funding acquisition, writing – review and editing. V. Wepener: conceptualization, data curation, funding acquisition, writing – review and editing. N. J. Smit: conceptualization, data curation, funding acquisition, writing – review and editing.
Financial support
This work is based on the research supported by the National Research Foundation (NRF) of South Africa (grant 132805; L. van der Spuy; grant 150623; N. J. Smit). Opinions, findings, conclusions and recommendations expressed in this publication are those of the authors, and the NRF accept no liability whatsoever in this regard. The authors acknowledge The Nippon Foundation-Nekton Ocean Census Programme (https://oceancensus.org/) for supporting the description of these species. These are Ocean Census Species Numbers 49–50.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.