Introduction
Parasitic worms have a significant impact on humans, livestock and important crop cultures. It is estimated that a minimum of 2 billion people are infected by at least 1 parasitic worm, such as soil-transmitted helminths (Wright et al., Reference Wright, Werkman, Dunn and Anderson2018), flukes (Fürst et al., Reference Fürst, Duthaler, Sripa, Utzinger and Keiser2012; Wright et al., Reference Wright, Werkman, Dunn and Anderson2018) or tapeworms (Scholz and Kuchta, Reference Scholz and Kuchta2016; Eichenberger et al., Reference Eichenberger, Thomas, Gabriël, Bobić, Devleesschauwer, Robertson, Saratsis, Torgerson, Braae, Dermauw and Dorny2020). Nevertheless, the diseases they cause – helminthiasis – are severely understudied, and many belong to the group of neglected tropical diseases. Geographically, cases of helminthiasis are not distributed evenly, as most human infections by helminths occur in low- and middle-income countries (Weatherhead et al., Reference Weatherhead, Hotez and Mejia2017; James et al., Reference James, Abate, Abate, Abay, Abbafati, Abbasi, Abbastabar, Abd-Allah, Abdela, Abdelalim, Abdollahpour, Abdulkader, Abebe, Abera, Abil, Abraha, Abu-Raddad, Abu-Rmeileh, Accrombessi, Acharya, Acharya, Ackerman, Adamu, Adebayo, Adekanmbi, Adetokunboh, Adib, Adsuar, Afanvi, Afarideh, Afshin, Agarwal, Agesa, Aggarwal, Aghayan, Agrawal, Ahmadi, Ahmadi, Ahmadieh, Ahmed, Aichour, Aichour, Aichour, Akinyemiju, Akseer, Al-Aly, Al-Eyadhy, Al-Mekhlafi, Al-Raddadi, Alahdab, Alam, Alam, Alashi, Alavian, Alene, Alijanzadeh, Alizadeh-Navaei, Aljunid, Alkerwi, Alla, Allebeck, Alouani, Altirkawi, Alvis-Guzman, Amare, Aminde, Ammar, Amoako, Anber, Andrei, Androudi, Animut, Anjomshoa, Ansha, Antonio, Anwari, Arabloo, Arauz, Aremu, Ariani, Armoon, Ärnlöv, Arora, Artaman, Aryal, Asayesh, Asghar, Ataro, Atre, Ausloos, Avila-Burgos, Avokpaho, Awasthi, Ayala Quintanilla, Ayer, Azzopardi, Babazadeh and Badali2018). Helminthiases are particularly prevalent among children, which led to the launch of so-called Mass Drug Administration programs that aim to provide chemotherapy to school-aged children (Weatherhead et al., Reference Weatherhead, Hotez and Mejia2017; Majid et al., Reference Majid, Kang and Hotez2019). Helminths also impose a substantial economic burden on the livestock industry with €1·8 billion in Europe alone (Charlier et al., Reference Charlier, Rinaldi, Musella, Ploeger, Chartier, Vineer, Hinney, von Samson-himmelstjerna, Băcescu, Mickiewicz, Mateus, Martinez-Valladares, Quealy, Azaizeh, Sekovska, Akkari, Petkevicius, Hektoen, Höglund, Morgan, Bartley and Claerebout2020), and plant food production with $80 billion worldwide (Jones et al., Reference Jones, Haegeman, Danchin, Gaur, Helder, Jones, Kikuchi, Manzanilla-López, Palomares-Rius, Wesemael and Perry2013). This has led to the widespread use of the anthelmintic drugs, which, unfortunately, resulted in the emergence of drug resistance to all classes of anthelmintics (Fissiha and Kinde, Reference Fissiha and Kinde2021).
Given the importance of helminths and the emerging threat of drug resistance, numerous studies have examined various aspects of parasitic worms’ biology, including distribution, epidemiology and host–parasite interactions. However, one important aspect – worm ploidy – remains understudied. Ploidy is expected to play an important role in the evolution of host–parasite interactions and drug resistance, both of which are essential aspects of parasitic organisms. First, ploidy is hypothesized to affect host–parasite interaction by altering the number of antigens to be detected by hosts (M’Gonigle and Otto, Reference M’Gonigle and Otto2011). However, this effect was shown theoretically only in comparison of haploid and diploid pathogens, without any extrapolation to higher levels of ploidy. Second, theoretical studies suggest that polyploidy affects the rate of adaptation, as increased ploidy can lead to a higher number of beneficial mutations (Otto and Whitton, Reference Otto and Whitton2000). Unfortunately, only a limited number of experimental studies test theoretical predictions or even consider the effect of polyploidy on host–parasite dynamics (King et al., Reference King, Seppälä and Neiman2012), especially from the parasitic ‘point of view’.
While little is known about the prevalence of polyploidy in parasitic worms – hence, the reason for this study – parasitic worms with higher ploidy, mostly triploid, have already been observed, coexisting with their diploid conspecifics and congeners. The presence of such triploid parasitic species, usually reproducing through mitotic parthenogenesis, is interesting for several reasons. First, mitotic parthenogenesis means that oocytes are produced without recombination, and a new organism develops without fertilization. Lack of recombination in parasitic worms contradicts theoretical models suggesting that recombination (associated with sexual reproduction) is beneficial for parasites under the strong selection they experience from their hosts (Salathé et al., Reference Salathé, Kouyos, Regoes and Bonhoeffer2008), as hosts can develop resistance against asexual lineages rapidly. Moreover, without recombination, asexual lineages are supposed to accumulate deleterious mutations and are therefore considered evolutionary ‘dead ends’. On the other hand, resistance to anthelmintics can arise from multiple loci (Doyle et al., Reference Doyle, Laing, Bartley, Morrison, Holroyd, Maitland, Antonopoulos, Chaudhry, Flis, Howell, McIntyre, Gilleard, Tait, Mable, Kaplan, Sargison, Britton, Berriman, Devaney and Cotton2022), and the lack of recombination observed in several triploid parasitic worms can actually help preserve multigenic traits. Thus, one may hypothesize that anthelmintic resistance spreads faster in a population through asexual triploids, a hypothesis that is yet to be tested.
Second, triploidy, as a type of polyploidy, can affect traits related to reproduction, physiology and life history. For instance, polyploid individuals develop faster under harsher environments compared to diploid conspecifics, as was shown on Daphnia pulex (Dufresne and Hebert, Reference Dufresne and Hebert1998). Extrapolated to triploid parasites, this observation suggests that they can outcompete diploids coinfecting the same host due to higher virulence. Another comparison on free-living flatworms reported triploid individuals to produce 58% more offspring in comparison to tetraploids (D’souza et al., Reference D’souza, Storhas and Michiels2005). However, we still lack similar studies comparing different ploidy levels in terms of reproductive output and life-history traits in the context of host–parasite systems from the parasite perspective.
One of the first reviews mentioning polyploidy in animals was published in the context of parthenogenesis. For example, Suomalainen et al. (Reference Suomalainen, Saura, Lokki, Hecht, Steere and Wallace1976) provide a list of parthenogenetic polyploids among insects, molluscs and crustaceans. Suomalainen et al. (Reference Suomalainen, Saura, Lokki, Hecht, Steere and Wallace1976) expanded the list of triploid parthenogenetic insects by including other orders. Otto and Whitton (Reference Otto and Whitton2000) reported triploidy and tetraploidy cases across insects and several vertebrate taxa. However, Otto and Whitton (Reference Otto and Whitton2000) do not mention any groups of parasitic worms. A more recent study by Román-Palacios et al. (Reference Román‐Palacios, Medina, Zhan and Barker2021) provides estimates of polyploidy incidence among animals, including Platyhelminthes and Nematodes. However, because the main aim of the study was to compare chromosome counts between plants and animals, the authors did not specify whether the species included in the database are parasitic. Spakulova et al. (Reference Spakulová, Orosová, Mackiewicz, Rollinson, Hay, Rollinson and Hay2011) have focused specifically on parasitic flatworms, reviewing karyotypes of Cestoda species. According to this study, 7 species of Cestoda are either exclusively triploid or have triploid populations (Spakulová et al., Reference Spakulová, Orosová, Mackiewicz, Rollinson, Hay, Rollinson and Hay2011). Triploidy has also been shown for several species of plant-parasitic root-knot nematodes (RKNs), members of the Meloidogyne genus (Janssen et al., Reference Janssen, Karssen, Topalović, Coyne and Bert2017), that are known for their noticeable economic impact, infecting essential crops (Jones et al., Reference Jones, Haegeman, Danchin, Gaur, Helder, Jones, Kikuchi, Manzanilla-López, Palomares-Rius, Wesemael and Perry2013).
Reports on triploidy in parasitic worms are worth considering also through the lens of the evolution of drug resistance. However, no studies to date have tested whether triploidy, or polyploidy in general, affects the evolution of resistance. Moreover, it is not clear how prevalent triploidy is among parasitic worms and what its physiological consequences are. Apart from the 2 overviews of triploidy in Cestoda and the RKN, information on the prevalence of triploidy across helminths is still missing.
To address this gap, a systematic review was conducted searching for all published cases of triploidy in parasitic worms. The aim of the study is to (1) provide data on the prevalence and distribution of triploidy, listing all reported cases among parasitic worms, (2) present information when available on relevant aspects of triploid parasitic worm biology, such as their origin, reproduction mode, coexistence with other ploidies, drug resistance and observed physiological differences. We believe this review will shed light on the importance of ploidy and serve as a starting point for further helminth-related studies, especially those focused on drug resistance evolution or host–parasite interactions.
Materials and methods
A systematic literature search was carried out on March 26, 2025, with the follow-up search on March 28, 2025 (see details below). Web of Science and Scopus platforms were used to search for articles published before the search dates. The aim of the literature search was to find published English-language sources reporting triploidy in species from 2 animal phyla: Platyhelminthes (flatworms) and Nematoda (roundworms). For this, 2 sets of keywords were used. The first set covered terminology describing triploidy, such as ‘triploid*’, ‘autotriploid*’ and ‘allotriploid*’. The second set included scientific and common names of main taxonomic groups from the 2 mentioned phyla (e.g. ‘cestod*’, ‘trematod*’, ‘nematod*’). See SI 1 for the complete list of search terms and search strings for each platform.
The initial search (conducted on March 26) included keywords covering all roundworms and flatworms with an emphasis on parasitic groups (such as cestodes and trematodes). The search returned 168 abstracts in Web of Science and 355 abstracts in Scopus. After removing duplicates, 396 abstracts remained. The second search run (conducted on March 28) included the same ‘triploidy’ keyword set as the initial search, but only 2 keywords covering 2 taxonomic groups, ‘monogenea*’ and ‘turbellaria*’. The rationale for this decision is that the initial list of ‘taxonomic’ keywords (which included the broad term ‘flatworm’) did not specify a separate class of parasitic worms – Monogenea. The initial list of keywords did not specify any groups of free-living flatworms either. Although they are not considered a monophyletic group (Schockaert et al., Reference Schockaert, Hooge, Sluys, Schilling, Tyler and Artois2008), it was still decided to use the currently deprecated term ‘Turbellaria’, specifically to address the older literature. The search resulted in 81 abstracts (21 in Web of Science, 60 in Scopus). After combining abstracts from both searches and checking for duplicates, the final number of abstracts was 416.
During the initial abstract screening, 143 papers were included for the full-text screening. A paper was marked as relevant if it was a published scientific material (an original paper, as well as a review) with an explicit report on triploidy of a species from the taxonomic groups defined above. See SI 2 for more details on the inclusion criteria for abstract screening and full-text reading. In total, 100 papers were marked as relevant and used for data extraction. Whenever possible, the following information was extracted: the species name, reproductive system and mode, origin of triploidy, other ploidy levels detected for a given species and any potential benefits of triploidy compared to diploidy. See SI 3 for the PRISMA flow diagram. When any of this information was not reported for a given species in selected papers, an additional search was conducted. However, all the additional papers were not included in the primary dataset, but were cited in the Results section.
Results
In total, 121 cases of triploidy were reported for Platyhelminthes and Nematoda in 100 articles. All these triploid cases have been reported in 52 species. Hereafter, by a triploid species is meant a species with populations that consist exclusively of triploid individuals or with a proportion of triploid individuals sampled at all life-cycle stages (i.e. eggs, larval stages, mature individuals).
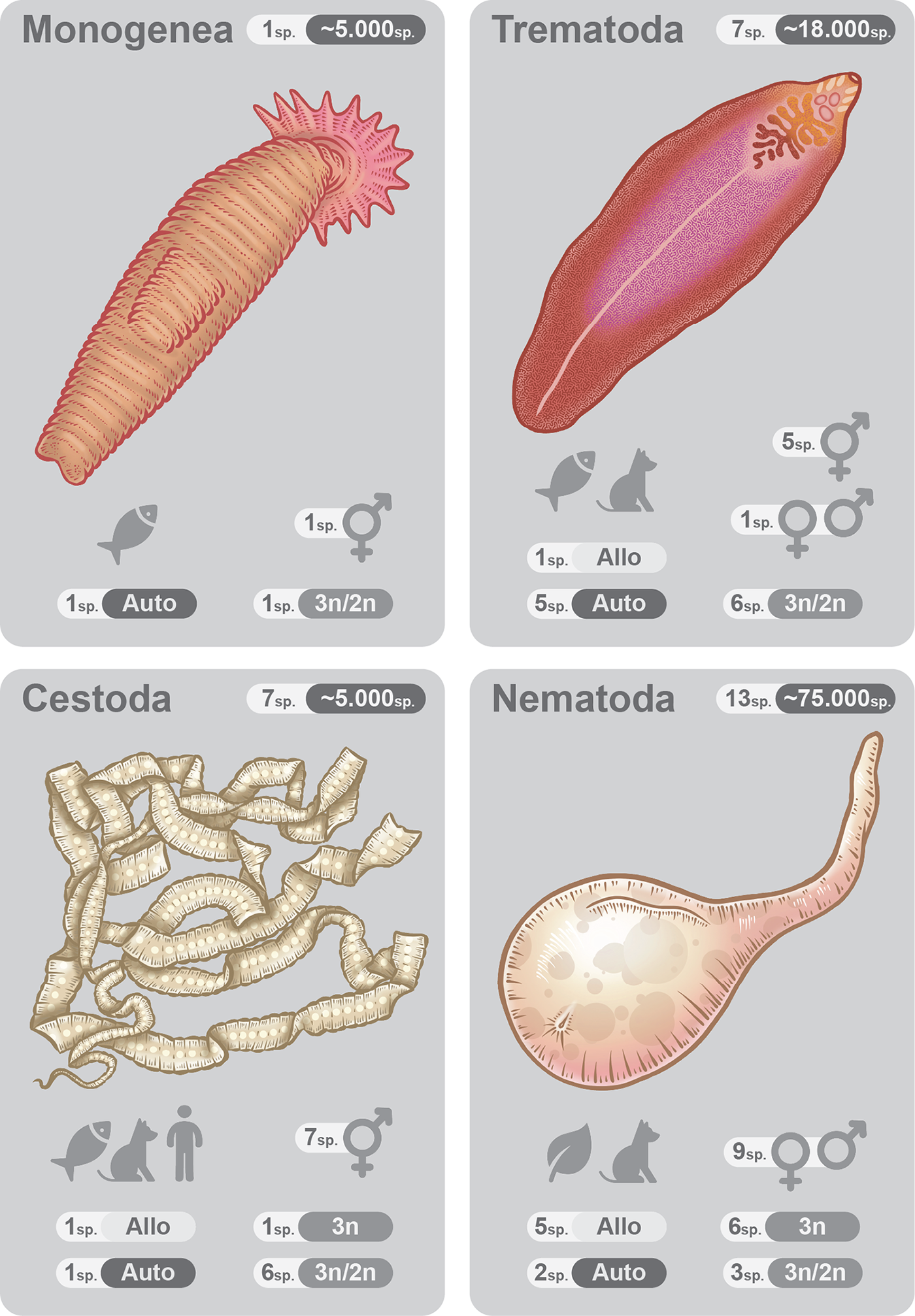
In the following subsections, the paper focuses on parasitic Nematoda and 3 parasitic classes of Platyhelminthes (Cestoda, Trematoda and Monogenea). For each group, the number of reported triploid species is provided. For each species, the information (when available) on the origin of triploidy, the presence of other ploidy levels, and reproductive mode is provided. Information on the phenotypic effects of triploidy includes any available information for a given species on the difference between triploids and individuals of other ploidy, as well as on potential drug resistance. See Table 1 for the complete list of triploid parasitic species within each taxonomic group. Table 1 also contains information on the method applied to identify triploidy for each species. Figure 1 provides a summary of each taxonomic group on the main aspects of triploid parasitic worm biology. For information on non-parasitic triploid worms, see the chapter ‘Non-parasitic triploid species’ and SI 4.
Summary of the main findings for each taxonomic group of parasitic worms included in the systematic review. Numbers at the top-right indicate the number of triploid species (white background) and the estimate of the total number of species. Icons at the bottom-left represent definitive hosts – fish, mammals (a dog), humans and plants (a leaf). Auto – number of triploid species with alleged autotriploid origin. Allo – number of triploid species with alleged allotriploid origin. The hermaphrodite sign shows the number of hermaphrodite species. Venus and Mars signs show the number of dioecious species. 3n – a species is exclusively triploid. 3n/2n – both triploid and diploid populations can be found. Note that information on origin, reproductive system and ploidy levels was available for not all triploid species.

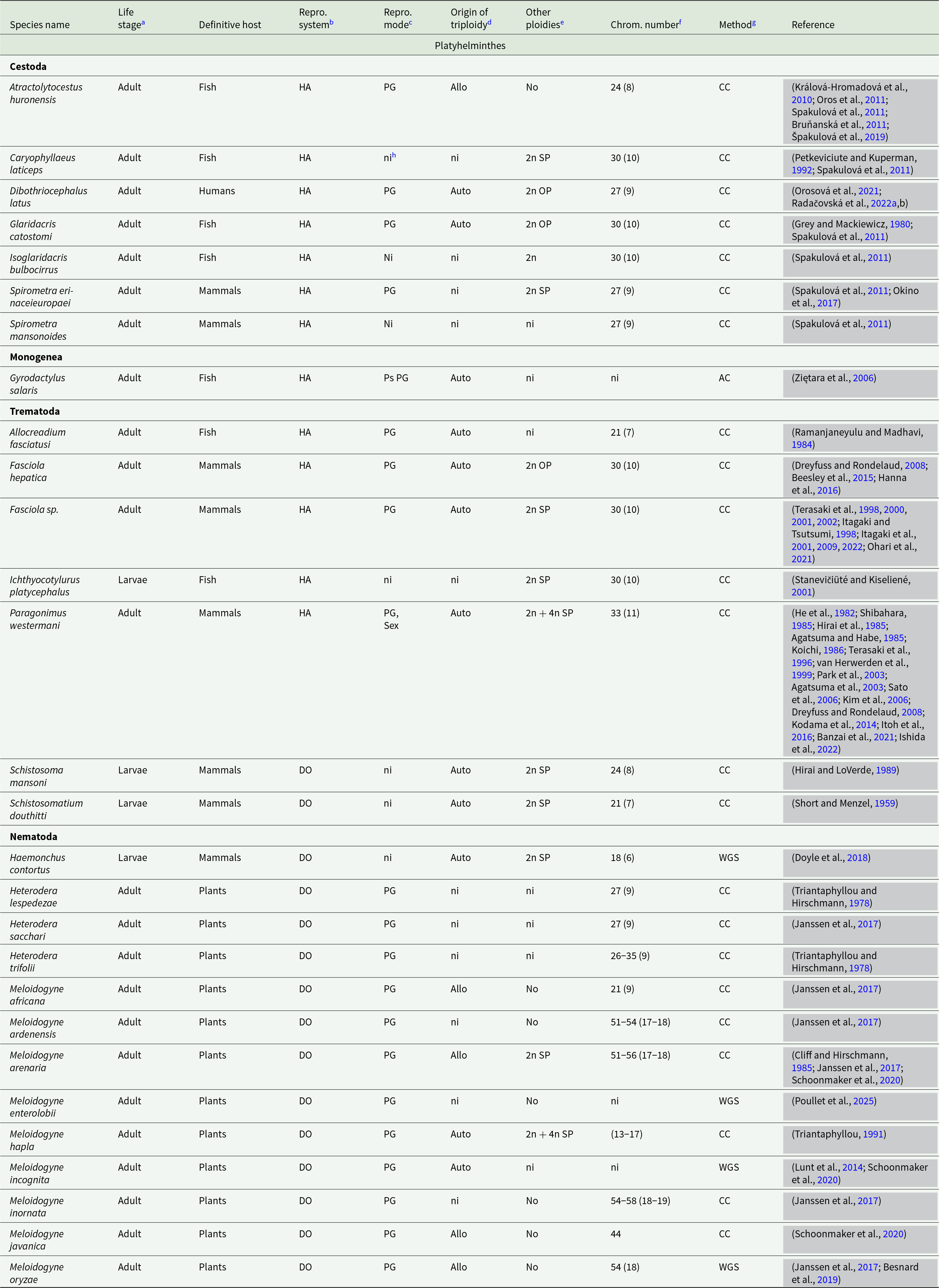
List of species of parasitic worms from 2 classes – Platyhelminthes and Nematoda – with reported triploidy

Notes:
a Life stage at which triploidy was detected. Adult – triploidy was detected at the adult stage; Larvae – triploidy was reported at a larval stage in any of the intermediate hosts.
b Reported reproductive system. HA – hermaphroditism – adult individuals are hermaphroditic; DO – dioecious – adult individuals are dioecious (gonochoric).
c Reported reproductive mode of triploids. PG – parthenogenesis – triploid individuals reproduce through parthenogenesis; Ps PG – pseudogamous parthenogenesis – a triploid egg is activated by sperm without incorporating sperm genetic material; Sex – a triploid egg can fuse with haploid sperm to produce a zygote.
d Proposed origin of triploidy. Allo – allotriploidy, the hybrid origin of triploidy; Auto – autotriploidy, triploidy origins within 1 diploid species.
e Information, whether other ploidies level exist within a triploid species. No – a species is supposed to be exclusively triploid; 2n – both triploidy and diploids are reported with no information whether both are from the same population; 2n OP – diploids are present in other populations; 2n SP – diploids are present in the same population with triploids; 2n + 4n SP – diploids and tetraploids are reported in the same population with triploids.
f A triploid chromosome count (a haploid chromosome count).
g A method how triploidy was confirmed for this species. AC – allele counting; CC – chromosome count; WGS – whole-genome sequencing.
h ‘ni’ indicates that no information was found.
Triploidy among parasitic flatworms
Triploidy was reported for 37 species of Platyhelminthes, 22 of which are non-parasitic flatworms, and 15 species are parasitic. Parasitic triploid species belong to 3 classes: Cestoda (7 species), Trematoda (7 species) and Monogenea (1 species).
Cestoda
Before describing results, it is worth mentioning the taxonomy of the genus Spirometra, as it is not fully established yet (Scholz et al., Reference Scholz, Kuchta and Brabec2019). Below, species names as they were reported in the original studies are provided. At the same time, some species may not be valid. Thus, following Kuchta et al. (Reference Kuchta, Kołodziej-Sobocińska, Brabec, Młocicki, Sałamatin and Scholz2021), potential new names are suggested based on the sampling area. In the case of Spirometra mansoni, it was decided to use the short name Sp. mansoni to avoid confusion with Schistosoma mansoni (S. mansoni).
Reproduction
All triploid species of Cestoda originate from a hermaphroditic diploid state. Parthenogenesis, as a reproductive mode, was mentioned for 4 species out of 7. No information is available on reproductive mode for the next species: Caryophyllaeus laticeps, Isoglaridacris bulbocirrus and Spirometra mansonoides (∼Sp. mansoni). An additional search did not reveal any information. Apart from the parthenogenetic reproductive mode, 3 triploid species (A. huronensis, D. latus and G. catostomi) were reported to have abnormal spermatogenesis.
Origin of triploidy
Suggestions for the origin of triploidy are available for 3 triploid species out of 7. A. huronensis is supposed to be of a hybrid origin (allopolyploidy) with another congeneric species, A. tenuicollis, as a potential candidate for an ancestral role (Spakulová et al., Reference Spakulová, Orosová, Mackiewicz, Rollinson, Hay, Rollinson and Hay2011). Two other species, D. latus (Orosová et al., Reference Orosová, Marková, Provazníková, Oros, Radačovská, Čadková and Marec2021) and G. catostomi (Grey and Mackiewicz, Reference Grey and Mackiewicz1980; Oros et al., Reference Oros, Králová-Hromadová, Hanzelová, Bruňanská, Orosová, Sanders and Peterson2011), are assumed to have an autopolyploid origin. No data were found on C. laticeps, I. bulbocirrus, Spirometra erinaceieuropaei (∼Sp. mansoni) and S. mansonoides (∼Sp. mansoni).
Coexisting with other ploidies
A. huronensis is found to have exclusively triploid populations with no diploid lines (Oros et al., Reference Oros, Králová-Hromadová, Hanzelová, Bruňanská, Orosová, Sanders and Peterson2011). S. erinaceieuropaei (∼Sp. mansoni) triploid individuals are present along with diploid individuals in the same populations (Okino et al., Reference Okino, Ushirogawa, Matoba, Nishimatsu and Saito2017). Similar to C. laticeps, where a triploid individual was found in the same location as diploid individuals (Petkeviciute and Kuperman, Reference Petkeviciute and Kuperman1992). Both diploid and triploid populations were reported for I. bulbocirrus. However, it is unclear whether both ploidy levels occur within the same population. G. catostomi and D. latus can exist in both diploid and triploid states (Grey and Mackiewicz, Reference Grey and Mackiewicz1980; Radačovská et al., Reference Radačovská, Čisovská Bazsalovicsová and Králová-Hromadová2022b). However, individuals with different ploidy levels have not been reported from the same population.
Phenotypic effects of triploidy
No information was found within the 100 papers on the difference between triploid and diploid tapeworms. Also, no studies mentioned resistance to any anthelminthic drugs. At the same time, one finding may suggest potential resistance in one of the Spirometra species (Bennett et al., Reference Bennett, Mok, Gkrania-Klotsas, Tsai, Stanley, Antoun, Coghlan, Harsha, Traini, Ribeiro, Steinbiss, Lucas, Allinson, Price, Santarius, Carmichael, Chiodini, Holroyd, Dean and Berriman2014). The authors detected amino acids in β-tubulin genes of Spirometra sp., identical to those that are associated with resistance to benzimidazole in another Cestoda species, Echinococcus multilocularis (Bennett et al., Reference Bennett, Mok, Gkrania-Klotsas, Tsai, Stanley, Antoun, Coghlan, Harsha, Traini, Ribeiro, Steinbiss, Lucas, Allinson, Price, Santarius, Carmichael, Chiodini, Holroyd, Dean and Berriman2014). However, the authors did not mention the ploidy of the Spirometra sp. sample, nor have they experimentally tested this alleged resistance. It is also worth noting that, while Bennet et al. (Reference Bennett, Mok, Gkrania-Klotsas, Tsai, Stanley, Antoun, Coghlan, Harsha, Traini, Ribeiro, Steinbiss, Lucas, Allinson, Price, Santarius, Carmichael, Chiodini, Holroyd, Dean and Berriman2014) used the name S. erinaceieuropaei, the latter study suggests that the sample may be Sp. mansoni (Kuchta et al., Reference Kuchta, Kołodziej-Sobocińska, Brabec, Młocicki, Sałamatin and Scholz2021).
Trematoda
Reproduction
Five species of trematodes that have been reported as triploid are hermaphroditic. Two species from the Schistosomatidae family with reported triploidy (Schistosoma mansoni and Schistosomatium douthitti) are dioecious. Parthenogenesis, as a reproductive mode, was mentioned for 4 species out of 7. There is no explicit information about the reproductive mode of triploid individuals for Ichthyocotylurus platycephalus and S. douthitti. In the case of I. platycephalus, the original paper concluded triploidy based on chromosomal preparations of the intramolluscan stage of the fluke. An additional search did not show any information about the reproductive mode. Despite this, parthenogenetic reproduction can be suggested, given that this species is hermaphroditic. It is worth mentioning that, for S. mansoni, triploidy was shown only for several cells at the intramolluscan stage of the parasite. No studies were found reporting triploidy in adults.
Triploidy of S. douthitti was shown on miracidial and cercarial embryos with XXY (a male) and XYY (a female) karyotypes. No adult triploid individuals were checked; therefore, no information is available about their reproductive mode. However, females of S. douthitti are known to produce eggs parthenogenetically (Short and Menzel, Reference Short and Menzel1959).
Triploid Paragonimus westermani reproduces parthenogenetically and has abnormal spermatogenesis. At the same time, it was shown that non-reduced eggs (3n) of triploid individuals can fuse with reduced sperm (1n) from diploid individuals (Terasaki et al., Reference Terasaki, Shibahara, Noda and Kayano1996) when both ploidy forms co-infect a definitive host. Such fusion leads to the origin of tetraploid individuals, supporting the hypothesis that the tetraploid form of P. westermani has an autopolyploid origin. This finding may indicate a possibility for triploids to reproduce sexually when there are individuals with functional sperm.
Origin of triploidy
Limited information is available regarding the origin of triploidy among Trematoda. Autotriploidization has been suggested for 3 species: Allocreadium fasciatusi, S. douthitti and P. westermani. The fusion of an unreduced gamete with a reduced gamete within 1 species was proposed both for A. fasciatusi (Ramanjaneyulu and Madhavi, Reference Ramanjaneyulu and Madhavi1984) and S. douthitti (Short and Menzel, Reference Short and Menzel1959). This suggestion, however, was not tested. The origin of triploidy for P. westermani is not entirely clear either (Blair, Reference Blair, Toledo and Fried2024). It was suggested that different triploid populations of P. westermani might have multiple origins, following autopolyploidization events, an idea that was supported genetically (Saijuntha et al., Reference Saijuntha, Tantrawatpan, Jarilla, Agatsuma, Andrews and Petney2016).
A hybrid origin was suggested only for a triploid line referred to in the literature as Fasciola sp. Studies on the genetic structure of triploid Fasciola sp. individuals suggest that triploidization followed 2 stages. First, an initial crossing between F. hepatica and F. gigantica produces a hybrid diploid. Then, a hybrid diploid backcrosses with either of the parental species, producing a triploid hybrid (Itagaki et al., Reference Itagaki, Hayashi and Ohari2022).
Coexisting with other ploidies
A. fasciatusi is the only reported species of Trematoda with exclusively triploid individuals (Ramanjaneyulu and Madhavi, Reference Ramanjaneyulu and Madhavi1984). Triploid individuals of F. hepatica were reported from 1 population only, where all sampled individuals possessed a triploid set of chromosomes (Fletcher et al., Reference Fletcher, Hoey, Orr, Trudgett, Fairweather and Robinson2004). Other populations of F. hepatica seem to have only diploid individuals. A hybrid line of Fasciola sp. occurs in both triploid and diploid forms, which are often present in the same populations. Moreover, both ploidy forms of Fasciola sp. are found along with both or either parental species.
Ichthyocotylurus platycephalus shows 2 ploidy forms, diploid and triploid, originating from the same population. P. westermani is present in 3 ploidy forms: diploid, triploid and tetraploid. All 3 forms can occur sympatrically, while there are also areas where only 1 ploidy form is present (Kim et al., Reference Kim, de Guzman, Kong and Chung2006). Both diploid and triploid individuals are shown for S. mansoni, which assumes that both ploidy forms may coexist. However, triploidy was reported only in a population maintained in a laboratory for several generations. In the case of S. douthitti, diploid and triploid forms were found among the progeny of bisexual infection by diploid individuals.
Phenotypic effects of triploidy
Several studies have reported differences in life history traits between diploid and triploid individuals. For example, triploid individuals of P. westermani develop faster than diploids (Blair, Reference Blair, Toledo and Fried2024). In addition, it was mentioned that triploid adult individuals, their metacercariae and eggs are larger compared to diploid individuals (Blair, Reference Blair2022). The triploid form of P. westermani is also more pathogenic than the diploid form (Blair et al., Reference Blair, Agatsuma, Watanobe, Okamoto and Ito1997).
No studies report a connection between drug resistance and triploidy in trematodes. At the same time, resistance was detected in diploid conspecifics for several species. For example, diploid F. hepatica is known to be resistant to several anthelmintics, such as triclabendazole (TCBZ), albendazole and clorsulon (Fairweather et al., Reference Fairweather, Brennan, Hanna, Robinson and Skuce2020). However, triploid individuals of F. hepatica were isolated from a population susceptible to TCBZ (Fletcher et al., Reference Fletcher, Hoey, Orr, Trudgett, Fairweather and Robinson2004). In the case of P. westermani, resistance to TCBZ was suspected in 1 medical case in which 2 courses of TCBZ treatment failed to cure a patient infected with P. westermani (Kyung et al., Reference Kyung, Cho, Kim, Park, Jeong, Lee, Sung and Lee2011). This observation, however, did not report any data on ploidy level.
Monogenea
One species of Monogenea, Gyrodactylus salaris, was reported to be triploid, the first triploid species in this genus. G. salaris is a hermaphrodite reproducing parthenogenetically. Ziętara et al. (Reference Ziętara, Kuusela and Lumme2006) assumed that the triploid form originated from a diploid egg fertilized by a reduced sperm, which coincided with a switch to a rainbow trout host. Their study also suggested that G. salaris produces diploid offspring, which revert to sexual reproduction (Ziętara et al., Reference Ziętara, Kuusela and Lumme2006). Diploid offspring are potentially produced via pseudogamous parthenogenesis, which involves the activation of a diploid egg by sperm from non-selfing hermaphrodites. However, there is no transfer of genetic material from sperm.
Triploidy among parasitic roundworms
Among Nematoda, 15 species are found to be triploid, with 13 of them parasitic. All triploid parasitic Nematoda species are dioecious.
Reproduction
Diploid individuals of Haemonchus contortus reproduce sexually. The reproductive mode of triploid individuals is unknown because triploidy was detected postmortem at the larval stage (Doyle et al., Reference Doyle, Laing, Bartley, Britton, Chaudhry, Gilleard, Holroyd, Mable, Maitland, Morrison, Tait, Tracey, Berriman, Devaney, Cotton and Sargison2018). Another study reports an adult triploid female of H. contortus (Bremner, Reference Bremner1954), suggesting that triploid individuals can at least reach maturity.
Three triploid species of plant parasites in the Heterodera genus (H. lespedezae, H. sacchari and H. trifolii) reproduce parthenogenetically (Triantaphyllou and Hirschmann, Reference Triantaphyllou and Hirschmann1978). However, males, while rare and without details on their ploidy, can be found in populations (Triantaphyllou and Hirschmann, Reference Triantaphyllou and Hirschmann1978).
Triploidy was detected in 9 species of RKN, members of the Meloidogyne genus. All triploid RKN species are reported to reproduce via mitotic parthenogenesis (Janssen et al., Reference Janssen, Karssen, Topalović, Coyne and Bert2017). Despite this, studies have shown that males are present in triploid populations of RKN species. Males are reported, among others, for M. africana (Janssen et al., Reference Janssen, Karssen, Topalović, Coyne and Bert2017), M. inornata (Carneiro et al., Reference Carneiro, de Lourdes Mendes, Almeida, Dos Santos, Gomes and Karssen2008) and M. arenaria (Cliff and Hirschmann, Reference Cliff and Hirschmann1985). In the case of M. ardenensis, males are not only present but also appear to be sexually active, as the spermathecae of triploid females from sampled populations were filled with sperm (Janssen et al., Reference Janssen, Karssen, Topalović, Coyne and Bert2017). For 2 triploid species, M. hapla and M. javanica, it was shown that spermatozoa are able to enter an oocyte, but without further transfer of genetic material (Janssen et al., Reference Janssen, Karssen, Topalović, Coyne and Bert2017). Males were also found together with triploid females in infected roots for M. oryzae (da Mattos et al., Reference da Mattos, Cares, Gomes, Gomes, Monteiro, Gomez, Castagnone-Sereno and Carneiro2018). However, this species reproduces exclusively parthenogenetically (Besnard et al., Reference Besnard, Thi-Phan, Ho-Bich, Dereeper, Trang Nguyen, Quénéhervé, Aribi and Bellafiore2019). In addition, there is some evidence for genome reshuffling in M. oryzae, potentially due to a few meiotic events soon after original polyploidization (Besnard et al., Reference Besnard, Thi-Phan, Ho-Bich, Dereeper, Trang Nguyen, Quénéhervé, Aribi and Bellafiore2019).
No information on males was found for triploid M. incognita and M. enterolobii from papers selected for the systematic review. However, it is known that M. incognita females can reverse sex into males (Papadopoulou and Triantaphyllou, Reference Papadopoulou and Triantaphyllou1982).
Apart from parasitic nematodes, several free-living species of the genus Panagrolaimus are found to be exclusively triploid and reproducing parthenogenetically (Villegas et al., Reference Villegas, Ferretti, Wiehe, Waldvogel and Schiffer2024). Eggs of triploid Panagrolaimus develop without sperm, with all offspring being females (Villegas et al., Reference Villegas, Ferretti, Wiehe, Waldvogel and Schiffer2024).
Origin of triploidy
Triploid individuals of H. contortus are highly likely to originate from an autotriploidization event, as triploid larvae were detected during the experimental infection by 2 lines of H. contortus (Doyle et al., Reference Doyle, Laing, Bartley, Britton, Chaudhry, Gilleard, Holroyd, Mable, Maitland, Morrison, Tait, Tracey, Berriman, Devaney, Cotton and Sargison2018). Two potential mechanisms are proposed: (1) polyspermy, when an egg is fertilized by 2 sperm cells, or (2) fertilization of a non-reduced egg. At the same time, it is not clear whether triploidy is prevalent in the field and whether triploid larvae are viable and fertile after reaching maturity (Doyle et al., Reference Doyle, Laing, Bartley, Britton, Chaudhry, Gilleard, Holroyd, Mable, Maitland, Morrison, Tait, Tracey, Berriman, Devaney, Cotton and Sargison2018). However, an adult triploid female of H. contortus has been described earlier (Bremner, Reference Bremner1954), which suggests that triploid individuals are at least able to reach maturity.
The origin of triploid species from the genus Meloidogyne is still under investigation. There are data available on the potential origin of the members of the Meloidogyne incognita group (MIG), that includes M. incognita, M. javanica and M. arenaria (Schoonmaker et al., Reference Schoonmaker, Hao, Bird and Conant2020). Initially, it was suggested that M. incognita is a double-hybrid between the ancestor of M. floridensis and another unknown species (Lunt et al., Reference Lunt, Kumar, Koutsovoulos and Blaxter2014). Later, however, it was shown that M. floridensis rather belongs to the MIG group (Schoonmaker et al., Reference Schoonmaker, Hao, Bird and Conant2020). While all MIG species diverged from M. hapla with the following radiation (Schoonmaker et al., Reference Schoonmaker, Hao, Bird and Conant2020).
In 1 study, triploid females of M. hapla resulted from a cross between diploid females and tetraploid males of the same species (Triantaphyllou, Reference Triantaphyllou1991). Triploid females were able to produce normal-sized eggs. However, most of the eggs did not hatch (Triantaphyllou, Reference Triantaphyllou1991), which raises uncertainty about whether triploidy in M. hapla is stable.
One study examines the genetic relatedness of 2 RKN species, triploid M. oryzae and diploid M. graminicola, suggesting that M. graminicola is a putative parent of M. oryzae (Besnard et al., Reference Besnard, Thi-Phan, Ho-Bich, Dereeper, Trang Nguyen, Quénéhervé, Aribi and Bellafiore2019). No information was found on the possible origin of other triploid RKN species. There was 1 study claiming that M. enterolobii has a hybrid origin. However, the paper was retracted by the authors due to contamination of sequencing data with another species (Koutsovoulos et al., Reference Koutsovoulos, Poullet, Elashry, Kozlowski, Sallet, Da Rocha, Perfus-Barbeoch, Martin-Jimenez, Frey, Ahrens, Kiewnick and Danchin2025).
Free-living triploid species of Panagrolaimus are shown to be a monophyletic group that originates from a single triploidization event (Schiffer et al., Reference Schiffer, Danchin, Burnell, Creevey, Wong, Dix, O’Mahony, Culleton, Rancurel, Stier, Martínez-Salazar, Marconi, Trivedi, Kroiher, Thorne, Schierenberg, Wiehe and Blaxter2019; Villegas et al., Reference Villegas, Ferretti, Wiehe, Waldvogel and Schiffer2024).
No information was found on the origin of triploidy in triploid species from the Heterodera genus.
Other ploidies
Six RKN species are assumed to be exclusively triploid. These species are M. africana, M. ardenensis, M. enterolobii, M. inornata, M. oryzae and M. javanica. The last species itself is rather a hypotriploid (or paleotriploid) with 43–46 chromosomes, which does not correspond to the full triple set (Cenis, Reference Cenis1992; Schoonmaker et al., Reference Schoonmaker, Hao, Bird and Conant2020). No information is available about the exclusivity of triploidy or the presence of other ploidies for M. incognita.
M. arenaria is shown to have a hybrid origin (Schoonmaker et al., Reference Schoonmaker, Hao, Bird and Conant2020). Despite this, 1 study describes the presence of diploid individuals in 1 sampled population (Cliff and Hirschmann, Reference Cliff and Hirschmann1985).
Two stable ploidy lines are reported for M. hapla: diploid and tetraploid. Triploid individuals appear due to outcrossing between diploids and tetraploids when both lineages coinfect the same plant (Triantaphyllou, Reference Triantaphyllou1991). While its origin is plausible, triploidy does not seem to be stable given the low viability of eggs (Triantaphyllou, Reference Triantaphyllou1991).
For H. contortus, 7 individuals out of 41 progeny of a single female were identified as triploid at the larval stage (Doyle et al., Reference Doyle, Laing, Bartley, Britton, Chaudhry, Gilleard, Holroyd, Mable, Maitland, Morrison, Tait, Tracey, Berriman, Devaney, Cotton and Sargison2018). Ploidy for other progeny was not specified. However, diploidy can be assumed, as the authors reported only triploidy among polyploid cases (Doyle et al., Reference Doyle, Laing, Bartley, Britton, Chaudhry, Gilleard, Holroyd, Mable, Maitland, Morrison, Tait, Tracey, Berriman, Devaney, Cotton and Sargison2018).
Phenotypic effects of triploidy
H. contortus shows resistance to anthelmintics from different families, such as benzimidazoles, imidazothiazoles and macrocyclic lactones (Kotze and Prichard, Reference Kotze, Prichard, Gasser and Samson-Himmelstjerna2016). Triploid larvae of H. contortus were detected after co-infection with 2 genetically divergent strains, 1 fully susceptible and 1 resistant (Doyle et al., Reference Doyle, Laing, Bartley, Britton, Chaudhry, Gilleard, Holroyd, Mable, Maitland, Morrison, Tait, Tracey, Berriman, Devaney, Cotton and Sargison2018). However, no information is available on whether triploid individuals inherited alleles responsible for resistance in H. contortus. Although not related to anthelmintic resistance, it is worth mentioning that studies have reported that triploid M. enterolobii overcomes the Mi-1 resistance gene in breeding cultivars (Santos et al., Reference Santos, Abrantes and Maleita2019).
Non-parasitic triploid species
Among 100 selected papers, information on triploidy in 24 non-parasitic flatworms and nematodes was found. Non-parasitic species of flatworms with triploid individuals belong to 5 genera: Dolichoplana – 1 species, Dugesia – 15 species, Girardia – 3 species, Polycelis – 2 species, Schmidtea – 1 species. Non-parasitic triploid flatworms reproduce parthenogenetically and by fission (splitting their body into 2 parts). In addition, some triploid species (e.g. Dugesia ryukyuensis) can reproduce sexually, switching reproductive modes seasonally (Nodono and Matsumoto, Reference Nodono and Matsumoto2022). Another species (Schmidtea polychroa) can produce fertile haploid sperm (D’souza and Michiels, Reference D’souza and Michiels2008). Among Nematoda, the only non-parasitic triploid species are Caenorhabditis elegans and members of the Panagrolaimus genus. C. elegans is the only reported triploid Nematoda species with an androdioecious reproductive system. See SI 4 for the full list of non-parasitic triploid species
Discussion
In the systematic review, 28 species of parasitic worms were identified in which triploid individuals or populations have been detected. The number of known parasitic species with triploidy makes up only a tiny fraction of the total number of estimated species within each taxonomic group of parasitic worms. For example, Cestoda includes around 5000 known species (Scholz and Kuchta, Reference Scholz and Kuchta2022), and Trematoda includes at least 18 000 species (Kostadinova and Pérez-del-Olmo, Reference Kostadinova, Pérez-del-Olmo, Toledo and Fried2014). There are around 3400 species of plant-parasitic nematodes (Singh et al., Reference Singh, Hodda and Ash2013) and up to 28 000 estimated species of vertebrate-parasitic nematodes (Carlson et al., Reference Carlson, Dallas, Alexander, Phelan and Phillips2020).
The small number of known triploid species among parasitic worms, relative to their total number, could be explained by 2 main factors. The first, and very likely one, is a sampling bias and the lack of available information. Triploidy in animals, or polyploidy in the broader context, has been investigated over the last few decades, primarily from genetic and physiological perspectives (Choleva and Janko, Reference Choleva and Janko2013; Small et al., Reference Small, Benfey and Crawford2022). However, parasitic worms were rarely present in reviews discussion polyploid animals. In addition, only a few studies have addressed the evolutionary and ecological consequences of polyploidy (but see Wertheim et al., Reference Wertheim, Beukeboom and van de Zande2013), especially in the context of host–parasite interactions from a parasite perspective. The most recent overview of tapeworm cytogenetics (Spakulová et al., Reference Spakulová, Orosová, Mackiewicz, Rollinson, Hay, Rollinson and Hay2011) provided karyological data for 115 species of parasitic worms. However, this study does not cover most of the species, because, as the authors mentioned, only up to 2% of known Cestoda species were studied karyologically (Spakulová et al., Reference Spakulová, Orosová, Mackiewicz, Rollinson, Hay, Rollinson and Hay2011). From our limited observation, triploid tapeworms make up around 6% of the total number of Cestoda species for which ploidy information is available. Can one then expect to have a similar ratio of triploids out of the total number of tapeworm species?
The second explanation is the actual rarity of triploidy. There is a general perception that triploid animals are either rare or that triploid lines are ‘evolutionary dead-ends’ (Mable, Reference Mable2004). Their populations are thus expected to be unstable and prone to dying out within a shorter time. However, such a perception may rely on assumptions that do not always hold. For example, connected with polyploidy, parthenogenesis and the consequent accumulation of deleterious mutations (see more in the following subsections).
Below, separate chapters focus on several aspects of triploid parasitic worms, including the origin of triploidy, reproduction, comparison with diploid conspecifics and how these factors can affect host–parasite dynamics and adaptation rates, particularly in the context of drug resistance evolution.
Emergence of triploidy
The origin of triploid parasitic worms can be broadly classified into 2 modes: hybrid origin (allotriploidy) and non-hybrid origin (autotriploidy). In the first case, a triploid lineage arises from hybridization between 2 sexual diploid species. Autotriploids are formed from a mating within 1 diploid species. In both cases, triploidy may result from the fusion of either 2 or 3 gametes. When 2 gametes are involved, 1 is diploid (usually an ovum), the other is haploid (usually a spermatozoon). With 3 gametes, a haploid egg is fertilized by 2 sperm. See Choleva and Janko (Reference Choleva and Janko2013) for an informative review of the mechanisms by which triploidy arises. While mechanisms describing the origin of triploidy are academically interesting, Soltis et al. (Reference Soltis, Buggs, Doyle and Soltis2010) raised an important question as to whether the mode of origin makes any evolutionary difference. This question can be formulated even more broadly: whether there is any difference between allo- and autotriploids that their origin mode can explain.
Choleva and Janko (Reference Choleva and Janko2013) have reviewed scenarios of polyploidization among animals, both allo- and autopolyploidy, with many examples illustrating each scenario. However, it remains unclear which type of triploidy prevails in animals. This may be partly due to the challenges in identifying pathways to polyploidy, even in recently evolved lineages (Choleva and Janko, Reference Choleva and Janko2013). As a result, the number of autopolyploids may be underestimated (Soltis et al., Reference Soltis, Buggs, Doyle and Soltis2010).
On the other hand, studies of polyploidy in plants are more prevalent. For a long time in plant polyploidy studies, there was a traditional view that autopolyploids are rare and less fit than allopolyploids, as discussed by Soltis et al. (Reference Soltis, Buggs, Doyle and Soltis2010). Contrary to this, Ramsey and Schemske (Reference Ramsey and Schemske1998) have estimated that the rate of autopolyploid formation among flowering plants is higher than the rate of allopolyploid formation. They have also stated that triploids play an important role in the formation of tetraploids among plants through the so-called ‘triploidy bridge’ (Ramsey and Schemske, Reference Ramsey and Schemske1998). Later, Barker et al. (Reference Barker, Arrigo, Baniaga, Li and Levin2016) showed near parity between auto- and allopolyploids within plant genera. While such results clearly contrast with the traditional view on the rarity of autopolyploids, Barker et al. (Reference Barker, Arrigo, Baniaga, Li and Levin2016) have concluded that allopolyploids do have an evolutionary advantage. Such a conclusion is based on the suggestion that autopolyploid plants should outnumber allopolyploids with at least a 5-fold difference (Barker et al., Reference Barker, Arrigo, Baniaga, Li and Levin2016), which is not the case.
It is hypothesized that the reproductive system of original diploid lines can predispose triploids to be formed in a particular way. For example, many flatworm species (with several exceptions among Trematoda) are hermaphrodites with the potential for self-fertilization. This raises an important question: Can one expect more cases of triploidy among hermaphrodites? Data on the origin of triploidy is available for 10 species of flatworms. Only 2 of them have supposedly a hybrid origin, Cestoda species Atractolytocestus huronensis and Trematoda Fasciola sp. complex. On the other hand, most triploid nematodes are found within the RKN group, which includes exclusively dioecious species. Most RKN triploid species (5 out of 6) show a hybrid origin, resulting from a cross between 2 parental species.
A recent study on the dioecy-polyploidy association among plants revealed that polyploids are rare in genera where the majority of the species are dioecious (Osterman et al., Reference Osterman, Hill, Hagan, Whitton, Bacon and Bjorkman2024). The authors even suggested a reverse relationship, in which polyploidization in hermaphrodites favours a transition to dioecy. However, the authors considered a species as a polyploid only if its chromosome count was typical of autopolyploids (Osterman et al., Reference Osterman, Hill, Hagan, Whitton, Bacon and Bjorkman2024). This may suggest that hermaphrodites do transit to polyploids more often than dioecious plants, but only through the non-hybrid pathway. Otto and Whitton formulated a similar idea for animals. They proposed that polyploidy should be more common, among others, in taxa with hermaphroditic or asexual reproduction (Otto and Whitton, Reference Otto and Whitton2000). Again, this suggestion considers only potential autopolyploids. Thus, it is not yet clear whether allopolyploids are more common in taxa with predominantly dioecious species.
After their emergence, triploid animals can maintain reproductive connectivity with parental species, especially in cases of hybrid origin. One example is a hybrid species of the edible frog Pelophylax esculentus with triploid lines. Triploid individuals are fertile and, for reproduction, rely on gametes from one of the parental species (Biriuk et al., Reference Biriuk, Shabanov, Korshunov, Borkin, Lada, Pasynkova, Rosanov and Litvinchuk2016). Among all triploid worm species reported here, only 1 case resembles such a relationship – the triploid Fasciola sp. complex. Molecular studies revealed a potential scenario of hybrid Fasciola emergence. It was suggested that triploid lines have emerged via backcrossing between initial diploid hybrids and either parental species (Itagaki et al., Reference Itagaki, Hayashi and Ohari2022). An experimental crossing between F. hepatica and F. gigantica indicated that the first hybrid generation is fertile and can produce viable offspring (Itagaki et al., Reference Itagaki, Ichinomiya, Fukuda, Fusyuku and Carmona2011). It is not clear, however, whether backcrossing of Fasciola hybrids keeps occurring in natural populations. At the same time, the experimental study suggests that a reproductive connection between triploid Fasciola and parental species is possible during co-infection.
Reproduction of triploids
Polyploidy in animals has long been associated with parthenogenesis (Suomalainen, Reference Suomalainen and Demerec1950; Suomalainen et al., Reference Suomalainen, Saura, Lokki, Hecht, Steere and Wallace1976). Despite limitations in available data, our findings show that the association between triploidy and parthenogenesis has been suggested for most triploid parasitic worms. An ability to reproduce sexually (along with parthenogenesis) has been suggested only for 1 species from our list of triploids – P. westermani, where triploid individuals had sperm cells in their reproductive tracts when coinfected with diploids (Terasaki et al., Reference Terasaki, Shibahara, Noda and Kayano1996).
Given that triploidy is associated with parthenogenesis, the difference between triploids and diploids in terms of reproduction can be explained partially by the difference between asexuals and sexuals. Exploring such a difference between asexual triploids and sexual diploids could be useful in explaining (alleged) stability of some triploid lines. For example, triploid parthenogens can benefit from reduced costs associated with sex. The classic approach states that sexual reproduction imposes a 2-fold cost due to either the production of males or genome dilution (Maynard Smith, Reference Maynard Smith1971). Thus, parthenogens are likely to avoid any costs related to the production of males (for dioecious organisms) or the development of male gonads (for hermaphrodites).
Avoiding costs can explain why several triploid hermaphroditic species have abnormal testes or non-functional sperm. As in D. latus and A. huronensis from Cestoda (Bruňanská et al., Reference Bruňanská, Nebesářová and Oros2011; Orosová et al., Reference Orosová, Marková, Provazníková, Oros, Radačovská, Čadková and Marec2021), or Fasciola sp. from Trematoda (Itagaki et al., Reference Itagaki, Hayashi and Ohari2022). When a triploid line can reproduce without sperm, male gonads play no role. Thus, there is no need to invest in male gonads. At the same time, it remains unclear to what extent costs related to the production of male gonads (or individuals) contribute to the origin or stability of triploidy. One can assume that any cost reduction follows triploidization events, without playing a primary selective factor in establishing triploid lines. A similar idea was proposed by Lundmark and Saura (Reference Lundmark and Saura2006), stating that asexuality per se did not play a primary role in the success of asexual species over sexual ones. Our assumption is also supported by a few observations from the RKN species complex, where triploid species include both females and males, despite the predominant occurrence of mitotic parthenogenesis as in M. africana (Janssen et al., Reference Janssen, Karssen, Topalović, Coyne and Bert2017). It should be mentioned, however, that Meloidogyne species seem to have environmental sex determination, as in M. hapla and M. incognita (Papadopoulou and Triantaphyllou, Reference Papadopoulou and Triantaphyllou1982; Triantaphyllou, Reference Triantaphyllou1991). This observation suggests that the type of sex determination should be taken into account when comparing asexual parasitic worms with sexual ones.
Apart from the direct cost related to sex, parthenogens can have an advantage over sexual populations in populations with lower density. A recent study on the prevalence of D. latus in Alpine lakes shows that this species is usually found as a single plerocercoid per fish, a second intermediate host (Radačovská et al., Reference Radačovská, Bazsalovicsová, Costa, Orosová, Gustinelli and Králová-Hromadová2020). This often leads to infection of a definitive host by a single worm (Bazsalovicsová et al., Reference Bazsalovicsová, Koleničová, Králová-Hromadová, Minárik, Šoltys, Kuchta and Štefka2018; Orosová et al., Reference Orosová, Marková, Provazníková, Oros, Radačovská, Čadková and Marec2021). Under such conditions, parthenogens can reproduce without relying on mating. In general, the probability of encounters between individuals (a proxy for population density) appears to play an important role in the transition to female-dominated parthenogenetic populations. Schwander et al. (Reference Schwander, Vuilleumier, Dubman and Crespi2010) have developed an analytical model to investigate the conditions under which males are lost in populations with parthenogenetic females. They found that females dominate in a population under a low encounter rate and a medium-to-high proportion of eggs that develop without fertilization (Schwander et al., Reference Schwander, Vuilleumier, Dubman and Crespi2010). However, this model implicitly assumes that sex is determined genetically, an assumption that cannot be applied to all parasitic worms, as mentioned earlier. Thus, it might be useful to investigate the spread of parthenogenesis in a population with environmental sex determination, in which males are present and fertile.
Discussion of cost comparison between sexuals and asexuals often includes recombination as a double-edged sword. Asexual triploid parasitic worms reproduce through mitotic parthenogenesis, in which an oocyte develops without meiosis and, consequently, without recombination. On the one hand, the lack of recombination is one of the reasons why parthenogenetic lineages are considered evolutionary dead-ends (Lodé, Reference Lodé2013). In theory, deleterious mutations accumulate in the absence of recombination. This accumulation eventually limits the long-term persistence of asexual lineages, a phenomenon known as ‘Muller’s ratchet’ (Muller, Reference Muller1964). Thus, alternative mechanisms might be required to sustain asexuality. For example, Vogt (Reference Vogt2017) has suggested that epigenetic mechanisms can increase phenotypic diversity within genetically identical clonal lines, thereby contributing to environmental adaptation. It is not clear, however, whether asexual triploid lines adapt such a mechanism.
On the other hand, recombination is assumed to be the most general cost of sex (in the short-term perspective), because it breaks up beneficial gene combinations (Maynard Smith, Reference Maynard Smith1971; Lehtonen et al., Reference Lehtonen, Jennions and Kokko2012). When, for example, heterozygosity confers higher fitness than homozygosity, asexual lineages maintain this difference by preserving the same level of heterozygosity (assuming a negligible mutational load). In contrast, already the first generation of sexuals will experience at least 50% heterozygosity decrease (Lewis, Reference Lewis and Stearns1987).
Comparison between diploids and triploids
Stability of triploid parasitic species, or even their expansion, relies on the competitive advantages triploidy provides compared to diploidy. In their review, Otto and Whitton (Reference Otto and Whitton2000) proposed a useful framework for comparing polyploids with diploids, splitting all effects into 2 categories: phenotypic and genetic. One of the most common phenotypic effects of polyploidy is increased cell size and, consequently, increased body size (Otto and Whitton, Reference Otto and Whitton2000; Choleva and Janko, Reference Choleva and Janko2013). These effects were shown for 1 species from our review – P. westermani. Triploid individuals have larger eggs, metacercariae and adult body size compared to diploids (Blair, Reference Blair, Toledo and Fried2024). In addition, triploid individuals of P. westermani reach maturity in 2 months in infected cats and dogs, while diploid flukes develop in almost two and a half months (Habe et al., Reference Habe, Lai, Agatsuma, Ow-Yang and Kawashima1996). All these life-history traits, while potentially associated with increased metabolic costs (Milosavljevic et al., Reference Milosavljevic, Kauai, Mortier, Van de Peer and Bonte2024), may provide selective advantages for triploids in competition with diploids for the host.
Furthermore, it has been hypothesized that polyploid animals better tolerate stressful environments (Van de Peer et al., Reference Van de Peer, Ashman, Soltis and Soltis2021). Such a hypothesis has been tested experimentally with synthetic tetraploid C. elegans. Chauve et al. (Reference Chauve, Bazzani, Verdier, Butler, Atimise, McGarry and McLysaght2025) have shown that tetraploid individuals resist cold stress better than diploid conspecifics. Tetraploid individuals also produce more offspring under cold stress compared to diploid ones (Chauve et al., Reference Chauve, Bazzani, Verdier, Butler, Atimise, McGarry and McLysaght2025). Irrespective of whether abiotic stressors limit the dispersal of triploid parasitic worms, the general ability to tolerate stress can be beneficial for triploids.
Genetic effects of triploidy can also provide a selective advantage over diploids. A theoretical model of Otto and Whitton (Reference Otto and Whitton2000) showed that newly formed polyploids have an initial advantage of masking deleterious mutations. This advantage, however, diminishes later as mutational load in a stable state correlates with ploidy level (Otto, Reference Otto2007). In the same model, polyploids obtained beneficial mutations more often when the population was small, and mutations had partial dominance (Otto and Whitton, Reference Otto and Whitton2000). While this model did not provide a parasitic perspective, it may be useful to apply a similar framework for comparing host–parasite dynamics between diploids and triploids.
The effect of triploidy on host–parasite interaction
One aspect of polyploidy that, to the best of our knowledge, has been addressed quite rarely is parasitism. Namely, whether polyploidy of parasites affects the host–parasite dynamic. A few theoretical models have tested the effect of ploidy on the evolution of parasitism (Nuismer and Otto, Reference Nuismer and Otto2004; M’Gonigle and Otto, Reference M’Gonigle and Otto2011). However, they are limited to 2 ploidy levels only – haploidy and diploidy. The main conclusion of the mentioned models is that parasites benefit from being haploid as it limits an array of antigens that could be detected by a host (Nuismer and Otto, Reference Nuismer and Otto2004; M’Gonigle and Otto, Reference M’Gonigle and Otto2011). While diploidy is favoured among hosts as it helps to recognize a broader range of antigens (Nuismer and Otto, Reference Nuismer and Otto2004; M’Gonigle and Otto, Reference M’Gonigle and Otto2011). Applying this framework, can one expect triploids to be more easily detected by a host than diploid conspecifics? Which host recognition models, following the description of Nuismer and Otto (Reference Nuismer and Otto2004), will better detect triploids? Which role does the type of triploidy origin play in parasite recognition?
Sexual reproduction is expected to be maintained in systems with antagonistic interactions between hosts and parasites, a notion formulated as the Red Queen Hypothesis (Hamilton et al., Reference Hamilton, Axelrod and Tanese1990; Ladle, Reference Ladle1992). The main prediction of the Red Queen Hypothesis (that hosts increase recombination rate in the presence of coevolving virulent parasites) has been supported empirically in many systems, see examples in Gibson and Fuentes (Reference Gibson and Fuentes2015). Theoretically, similar logic applies to parasites (Galvani et al., Reference Galvani, Coleman and Ferguson2003; Salathé et al., Reference Salathé, Kouyos, Regoes and Bonhoeffer2008), as they experience at least an equivalent level of selective pressure. A recent empirical study even shows that asexuality is less likely to arise on parasitic lineages of Nematoda compared to free-living ones (Gibson and Fuentes, Reference Gibson and Fuentes2015). Thus, there is a potential contradiction to theoretical predictions. There are examples of asexual triploids that, as in A. huronensis (Špakulová et al., Reference Špakulová, Bombarová, Miklisová, Nechybová and Langrová2019), expand their host range. The problem of asexual parasites and host ranges has been reviewed recently by Gibson (Reference Gibson2019). In addition, some triploids overcome resistance against congenerics, as in M. enterolobii (Poullet et al., Reference Poullet, Konigopal, Rancurel, Sallaberry, Lopez-Roques, Mota, Lledo, Kiewnick and Danchin2025). One should also not forget that parasitic worms can be infected by hyperparasites. For example, Pasteuria penetrans is an obligate parasite of phytonematodes that is used as a biocontrol agent (Mohan et al., Reference Mohan, Kiran Kumar, Sutar, Saha, Rowe and Davies2020). As a result, asexual triploid parasitic worms experience simultaneous selective pressure from their hosts and their hyperparasites. Such contradictions open new prospects for further theoretical studies. One direction, for example, is to investigate conditions that allow asexual triploid lines to sustain themselves without recombination under selection from 2 sides.
Triploidy and resistance
Triploid parasitic worms pose a noticeable threat to health and economy, which could intensify as drug resistance has been reported for all classes of anthelmintics (Fissiha and Kinde, Reference Fissiha and Kinde2021). Several species are reported to have triploid individuals in some populations, as well as cases of drug resistance in others. However, no data indicate that both are linked. At the same time, given examples of successful adaptation to new hosts, one should address questions related to anthelmintic resistance. One question of particular importance is whether triploid worms evolve resistance faster than diploids. What exactly can triploidy provide for resistance to evolve faster? As Doyle et al. (Reference Doyle, Laing, Bartley, Britton, Chaudhry, Gilleard, Holroyd, Mable, Maitland, Morrison, Tait, Tracey, Berriman, Devaney, Cotton and Sargison2018) stated, resistance to some anthelmintics has a multigenic nature. In this case, can one suppose that a lack of recombination will lead to a faster spread of a multigenic trait after its emergence? As was mentioned before, polyploids can receive more beneficial mutations under certain conditions. Would this factor play any role in the emergence of resistance? Given the importance of some triploid parasitic worms (e.g. members of the RKN species), there might be a need to investigate the effect of polyploidy on the evolution of drug resistance. Theoretical models can serve as a first step, identifying which parameters should be considered in subsequent experimental studies.
Conclusion
Polyploidy in animals has received noticeable attention with the development of, broadly speaking, molecular methods, as polyploidy has been detected in a wider selection of taxa. Thanks to this, we have a general understanding of how often polyploidy appears among animals compared to plants. In addition, there are studies reporting the effects of polyploidy on genomics, physiological traits and life history in animals. Partially due to such substantial progress, one can identify gaps in the understanding of the general effect of polyploidy on evolutionary dynamics and formulate new questions. For example, whether there are any consequences of triploidization of parasitic worms on the host–parasite dynamic remains an open question. This is one of the aspects of polyploidy that was not investigated thoroughly before.
With this systematic review, 2 main aims were addressed. The first aim was to attract attention to a parasitic aspect of triploidy (and polyploidy in general). Despite a perception that polyploid lineages are evolutionary ‘dead-ends’, it is clear that some triploid species can have a noticeable effect on livestock health and crop production. The second aim was to collect and report available information on the distribution of triploidy, its origin, the reproduction of triploid lineages and any phenotypic effects of triploidy. This information, as well as the questions raised, will motivate further studies investigating various aspects of triploidy. Such as the effects of triploidy on parasite life history, host–parasite interactions or the evolution of anthelminthic resistance. Any potential findings from these research areas may be of both academic and practical importance.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S003118202610198X.
Acknowledgements
The authors are grateful to the members of the Pathogen Evolution group and the Department of Aquatic Ecology for their comments, questions and suggestions during group meetings and departmental seminars at the earlier stages of this project. The authors are grateful to Prof Dr Jukka Jokela for reading and commenting on the original draft. As well as to the anonymous reviewer for providing constructive comments that improved the manuscript. Special thanks to Dr Giacomo Moggioli for the artwork in this manuscript.
Author contributions
Conceptualization: VK and BT. Literature search and screening: VK. Synthesis and writing: VK and BT.
Financial support
This work was supported by the Swiss National Science Foundation Starting Grant TMSGI3_218475: Drug resistance evolution across taxonomic boundaries: investigating the effects of ploidy, lifestyle and reproductive strategy.
Competing interests
The authors declare no conflicts of interest.
Ethical standards
Not applicable.