Highlights
-
Cannabidiol is a novel anti-seizure medication that can produce meaningful seizure reductions in individuals with various forms of drug-resistant epilepsy.
-
Cannabidiol is generally well tolerated, and treatment discontinuation due to adverse effects is uncommon.
-
Cannabidiol may offer additional benefits beyond seizure frequency reduction.
Introduction
Globally, approximately 70 million individuals live with epilepsy. Reference Ngugi, Bottomley, Kleinschmidt, Sander and Newton1 Although most achieve seizure control with anti-seizure medications (ASMs), approximately one-third develop drug-resistant epilepsy (DRE). Reference Kwan and Brodie2 DRE is defined as failure of adequate trials of two tolerated, appropriately selected ASM regimens to achieve sustained seizure freedom. Reference Kwan, Arzimanoglou and Berg3 Patients with DRE face heightened risks, including premature mortality, injuries, psychosocial difficulties and diminished quality of life. Reference Kwan, Schachter and Brodie4 The relationship between ASMs tried and seizure control likelihood is well documented. After trialing two ASMs, the probability of achieving optimal seizure control with a third ASM (4.4%) or subsequent ASMs (2.12%) is small, underscoring the challenge of treating highly refractory patients. Reference Chen, Brodie, Liew and Kwan5
Recent research has explored cannabinoids’ therapeutic potential in epilepsy. Preclinical studies demonstrate that cannabinoids attenuate neuronal hyperexcitability through multiple mechanisms, including G protein-coupled receptor 55 receptor inhibition, Reference Yang, Zhou and Lehmann6 transient receptor potential vanilloid 1 channel desensitization Reference Iannotti and Vitale7 and adenosine reuptake transporter inhibition. Reference Borea, Gessi, Merighi and Varani8,Reference Williams-Karnesky, Sandau and Lusardi9 Cannabinoids also possess anti-inflammatory properties and exert effects on cannabinoid receptors 1 and 2. Reference Howlett, Breivogel, Childers, Deadwyler, Hampson and Porrino10
Key phase III trials demonstrated that adjunctive cannabinoids significantly reduce seizure frequency in Dravet syndrome (DS) (GWPCARE 1 and 2), Lennox–Gastaut syndrome (LGS) (GWPCARE 3 and 4) Reference Thiele, Marsh and French11,Reference Devinsky, Cross and Laux12 and tuberous sclerosis complex (TSC) (GWPCARE 6). Reference Thiele, Bebin and Bhathal13 Patients showed continued seizure reduction in an open-label extension (GWPCARE 5). Reference Thiele, Marsh and Mazurkiewicz-Beldzinska14,Reference Devinsky, Nabbout and Miller15 Health Canada approved Epidiolex, the first pharmaceutical-grade, plant-derived purified cannabidiol (CBD), for seizures associated with LGS, DS and TSC in children aged two years and older on November 15, 2023.
This study aims to retrospectively evaluate the effectiveness of adjunctive CBD therapy in reducing seizure frequency among patients with highly refractory DRE at a single tertiary care center during the initial period when Epidiolex became available for prescription. As is typical with newly available ASMs, it was initially prescribed to the most treatment-refractory patients.
Methods
We conducted a retrospective single-center study of all patients who were started on Epidiolex (purified CBD oral solution, 100 mg/mL, Jazz Pharmaceuticals, Inc.) as adjunctive therapy between June 2024 to April 2025, at BC Children’s Hospital in Vancouver, Canada, the sole tertiary epilepsy center serving a province of 5.7 million people and treating 2500 children with complex epilepsy annually. Patients were identified through the Jazz Assist Program, a support initiative facilitating drug access following its Canadian introduction, with consent from treating physicians. Patients were included if they received CBD for at least four months and had adequate follow-up data.
Medical records were reviewed to extract clinical data including age, sex, family history, seizure history, epilepsy etiology and syndrome and epilepsy medical and surgical management (including epilepsy surgery, number of prior ASMs, concomitant ASMs and adverse effects [AEs]). Seizure type and etiology were classified using the 2017 ILAE criteria. Reference Fisher, Cross and French16 Baseline seizure frequency was defined as the mean monthly frequency during the 3–6 months preceding CBD initiation. For each patient, the age at initiation, maximum dose, treatment duration and AEs were collected.
Treatment response and AEs were extracted from documented patient-reported seizure frequency and AEs during follow-up visits in the electronic medical record. Responder rates were calculated as the percentage of patients achieving seizure reductions of ≥50%, 25%–49% and <25%. Seizure freedom (or near-seizure freedom) was defined as ≥99% reduction. A worsening of seizures was defined as a ≥25% increase in seizures from baseline. Treatment-emergent AEs, concomitant ASM adjustments and reasons for CBD discontinuation were analyzed. Following Chen et al., Reference Chen, Brodie, Liew and Kwan5 ASM discontinuation due to AEs was classified as treatment failure. When documented, we collected patient- or parent-reported number of seizure-free days and changes in seizure intensity (seizure severity or duration), mood, behavior and quality of sleep following CBD initiation. Institutional Research Ethics approval was obtained on March 12, 2025 (H25-00392-A003).
Statistical analysis
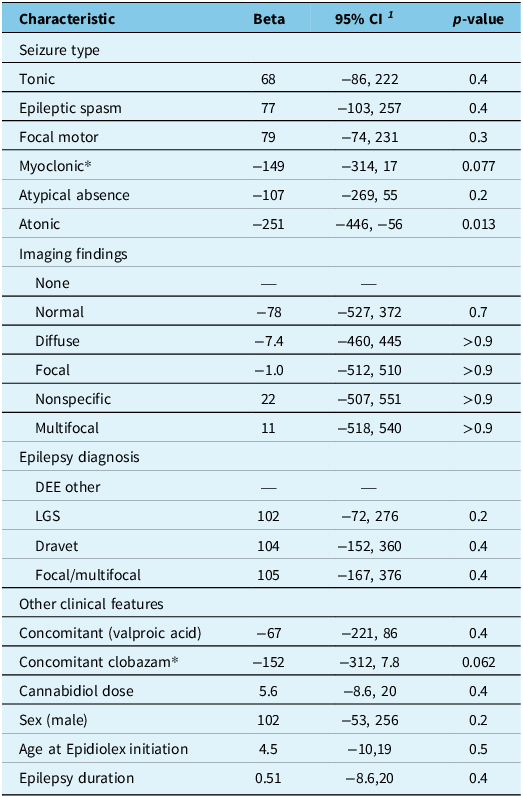
Descriptive statistics were calculated for all variables, with categorical data presented as frequencies and percentages and continuous variables as medians with interquartile ranges (Q1, Q3). The primary outcome measure was change in reported monthly seizure frequency, calculated as the difference between post-treatment and pretreatment seizure rates (negative values indicating improvement). Univariable linear regression analyses were performed to identify associations between clinical variables and seizure frequency change, with results reported as beta coefficients with 95% confidence intervals and p-values. Clinical predictors examined included seizure types, epilepsy syndrome, neuroimaging findings, concomitant ASMs, CBD dose and patient sex. Treatment response was categorized as >99% reduction, ≥50% reduction, 25%–49%, <25% reduction or worsening of seizures with >25% increase in seizure frequency. Among the subset of patients with <50% seizure reduction who continued treatment, we explored the reported clinical benefits, including seizure intensity reduction, seizure-free days in those with daily seizures and improvements in behavior and sleep. Statistical significance was set at p < 0.05, with p-values between 0.05 and 0.10 noted as trends.
Results
Cohort characteristics
Sixty-one patients met the inclusion criteria, of whom 37 (61%) were male. Median age was 10.5 years (range: 3–24 years), with median seizure onset at 8 months (IQR: 0–84 months). The median duration of treatment with CBD was 172 days (IQR: 131 days–219 days). Table 1 summarizes patient demographic and clinical characteristics. The cohort had highly refractory epilepsy, with a median of 8.0 prior ASM trials (IQR: 6.0–10.0). Fifty-two patients (85%) had previously tried more than 4 ASMs. Ten patients (16%) had undergone epilepsy surgery. Nineteen patients (31%) had previously tried the ketogenic diet, with 5 continuing it at CBD initiation. Four patients had vagal nerve stimulators prior to initiation of CBD. Nine patients switched from other formulations of CBD to Epidiolex, and six patients had tried another CBD formulation at least six months before Epidiolex initiation.
Table 1. Demographic and clinical data

Note: Number of patients (n) described in percentages and interquartile ranges. CBD = cannabidiol; ASM = anti-seizure medication; VNS = vagal nerve stimulator; DEE = developmental and epileptic encephalopathy; LGS = Lennox–Gastaut syndrome.
Epilepsy diagnoses were classified as focal/multifocal epilepsy in 7 patients (11%), LGS in 23 (38%), DS in 7 (11%) and other developmental and epileptic encephalopathies (DEEs) due to genetic, structural or metabolic causes in 24 (39%). Seizure types comprised generalized tonic seizures in 25 patients (41%), focal tonic seizures in 15 (25%), bilateral tonic-clonic seizures in 26 (43%), atypical absence seizures in 19 (31%), atonic seizures in 10 (16%), focal motor seizures in 28 (46%), epileptic spasms in 14 (23%) and myoclonic seizures in 17 (28%). Eighteen patients (30%) experienced drop seizures or ground-level falls, posing injury risk. Forty-nine patients (80%) experienced multiple seizure types.
Seizure etiologies were structural in 12 patients (20%), combined genetic and structural in 8 (13%, including TSC), genetic in 30 (49%), metabolic in 2 (3.3%), autoimmune in 1 (1.6%) and unknown in 8 (13%).
Among 30 patients (49%) with pathogenic genetic mutations, the most common were SCN1A (n = 7), TSC1/2 (n = 5), IDIC-15 (n = 2) and MECP2 (n = 2). Other mutations included KCNK2, RHOBBTB2, TPRS, KIAA2022, QARS, APT1A2, ANKRD11, KIF1A, GRIN2A, TOP2B, GABRA1, SCN2A, KCNA7 and ATP2B1, as well as 1q36 deletion and ring chromosome 17.
Structural etiologies comprised hypoxic-ischemic encephalopathy (n = 4), lissencephaly (n = 2), polymicrogyria (n = 1), non-accidental injury (n = 2), neonatal hypoglycemia (n = 1) and focal cortical dysplasia (n = 2). Combined genetic and structural etiologies included TSC (n = 5), Pallister–Killian syndrome (n = 1), band heterotopia from doublecortin mutation (n = 1) and focal cortical dysplasia with IFIH1 mutation (n = 1). Among 25 patients with abnormal neuroimaging, 21 (34%) had diffuse abnormalities, 5 (8.2%) had focal abnormalities and 5 (8.2%) had TSC with multifocal tubers.
Cannabidiol dosage
CBD was initiated at 2.5 mg/kg daily for two weeks and then increased by 2.5 mg/kg every two weeks. Target doses were determined by the treating epileptologist based on optimal seizure reduction with minimal AEs. For patients transitioning from alternative CBD formulations, Epidiolex was increased by 25% weekly, while the previous formulation was decreased by 25% until the target dose was reached. In patients receiving concomitant valproic acid, serum transaminases were measured at baseline and one and three months after achieving the target CBD dose. Significant transaminase elevation was defined as aspartate aminotransferase or alanine aminotransferase exceeding three times the upper limit of normal.
The final median (IQR) CBD dosage was 15 (2.5–20) mg/kg/day. The median duration of treatment with CBD was 172 days (IQR: 131 days, 219 days). The median number of concomitant ASMs with CBD is 2 (IQR: 1–7).
Efficacy
One patient was seizure-free on another form of CBD before initiating Epidiolex and was therefore excluded from the seizure outcome analysis. Among all patients, 29 (48%) achieved a < 25% reduction in seizures (22 with <25% reduction and 7 with worsening seizure frequency), 18 (30%) achieved a 25%–49% reduction and 13 (22%) reached a ≥50% reduction, with 4 (7%) achieving seizure freedom. Table 2 demonstrates clinical variables in relation to seizure outcome.
Table 2. Clinical features and response to cannabidiol

Note: DEE = developmental and epileptic encephalopathy; LGS = Lennox–Gastaut syndrome.
* An association was observed, but it did not reach statistical significance.
Epilepsy diagnosis
Compared to the reference group (DEE other), the epilepsy diagnosis was not significantly associated with outcome response. Refer to Figure 1. No significant differences were observed for LGS (β = 102, 95% CI: −72 to 276, p = 0.2), DS (β = 104, 95% CI: −152 to 360, p = 0.4) or focal/multifocal epilepsy (β = 105, 95% CI: −167 to 376, p = 0.4).

Figure 1. Epilepsy diagnosis and seizure frequency after cannabidiol treatment. No significant difference was observed in seizure frequency after treatment with cannabidiol, based on the epilepsy diagnosis. DEE = developmental and epileptic encephalopathy; LGS = Lennox–Gastaut syndrome.
Seizure outcomes by diagnostic category are shown in Figure 1. Among the 6 (11 %) patients with focal/multifocal epilepsy, 2 (33%) achieved a seizure reduction of <25 %, 2 (33%) achieved a 25%–49 % reduction and 2 (33 %) achieved a > 50 % reduction. None of these patients achieved seizure freedom or worsening of seizures.
Among the 23 (38 %) of patients with LGS, 13 (57 %) had a seizure reduction of <25 %, of whom 5 (22%) had a worsening of seizures. Eight (35 %) achieved a 25%–49 % reduction, and 2 (9 %) achieved a ≥ 50% reduction, although none were seizure-free.
Of 7 patients with DS, 3 (43%) had a seizure reduction of <25 %, of which one had a worsening of seizures. Two patients (29 %) achieved a 25%–49% reduction, and 2 (29 %) achieved a ≥ 50% seizure reduction, of which 1 (14 %) achieved seizure freedom.
Of the 24 (39 %) patients with other DEEs, 11 (46%) had a seizure reduction of ≤25 %, of whom 1 (4%) had worsening of seizures. Six (25 %) achieved a 25%–49 % reduction, and 7 (29 %) achieved a ≥ 50% reduction, of whom 3 (12 %) achieved near-seizure freedom. Notably, one patient with Doose syndrome became seizure-free.
Pretreatment seizure frequency
Pretreatment seizure burden was highest in those with moderate response (25%–49%) reduction: median 90 seizures/month, IQR: 16–150) and lowest in those achieving seizure freedom (median 2, IQR: 0–127). Refer to Table 3.
Table 3. Seizure outcome and baseline seizure frequency

1Median monthly seizure frequency (n) and interquartile ranges.
Note: Number of patients (n) described with median and interquartile ranges.
Concomitant ASMs
In our cohort, the median number of concomitant ASMs (excluding CBD) at baseline was 3 (IQR: 2–3): 5 patients (8%) were on 1 ASM, 12 (20 %) on 2 ASMs, 31 (51%) on 3 ASMs and 13 (21%) on 4 or more ASMs. At follow-up, 8 (30 %) of patients discontinued one ASM after initiation of CBD. Concomitant clobazam showed a trend toward better outcome response (β = −152, 95% CI: −312 to 7.8, p = 0.062). All patients who continued CBD despite <50% seizure reduction had highly refractory epilepsy, with median prior ASMs trials of 7.5 (IQR: 5.0–9.0) in the no worthwhile improvement group, 8.5 (IQR: 8.0–10.0) in the 25%–50% reduction group and 9.0 (IQR: 7.0–14.0) in those who worsened.
Seizure type
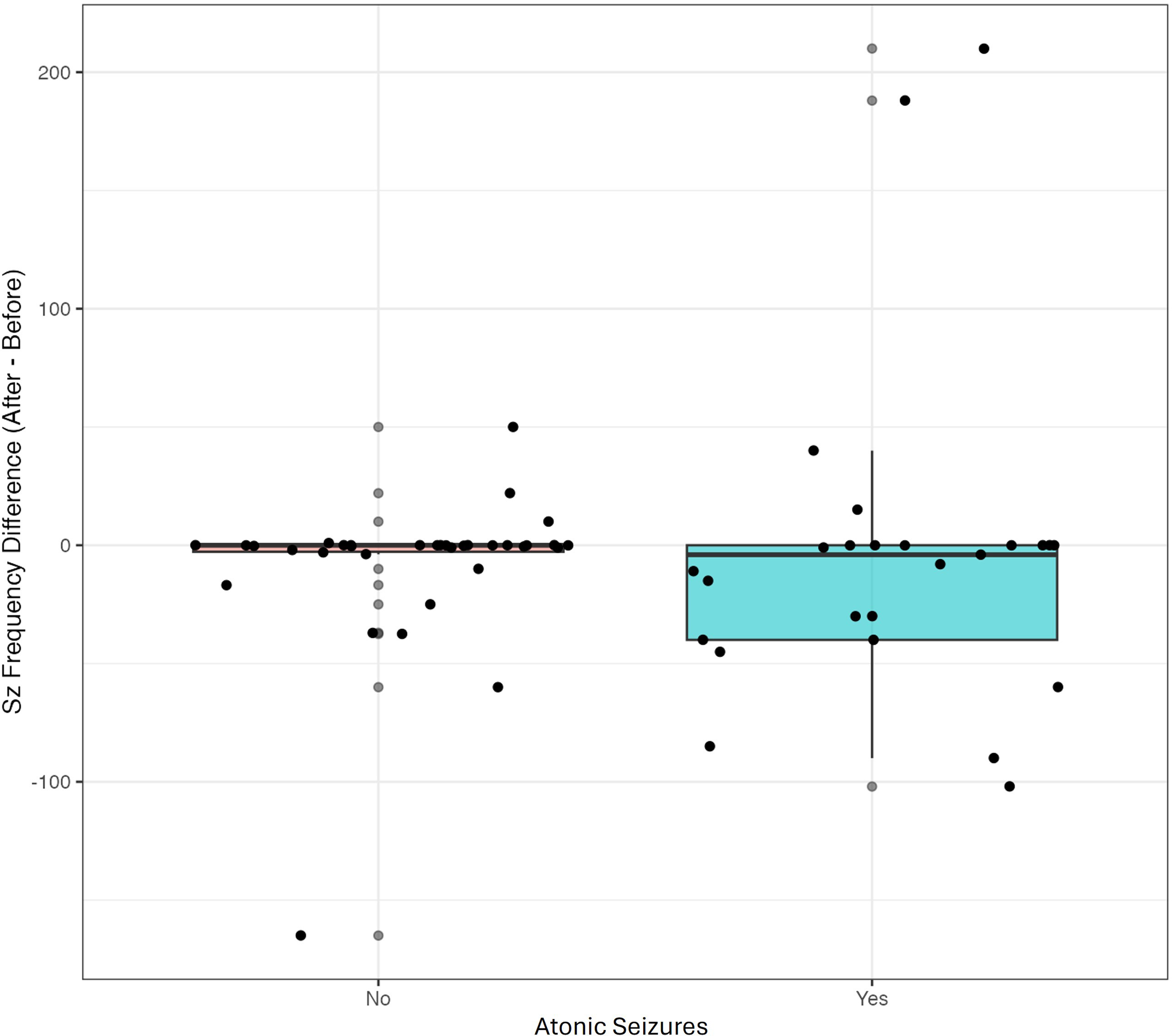
Patients with atonic seizures demonstrated significantly greater seizure reduction following CBD initiation (β = −251, 95% CI: −446 to −56, p = 0.013). Refer to Figure 2. No significant associations were found between outcome response and epilepsy syndrome, neuroimaging findings or other seizure types.

Figure 2. Difference in atonic seizure frequency pre- and post-cannabidiol. Y axis: seizure frequency difference before and after initiation of cannabidiol. X axis: Patients without (No) and with (Yes) atonic seizures. Box-whisker plots with the median [IQR] for the axis. Those with atonic seizures experienced fewer seizures at follow-up than at baseline after starting cannabidiol treatment.
Previous use of cannabidiol
The number of patients who had previously received another form of CBD or directly switched from another preparation to the highly purified form was small. Nine patients were switched from artisanal CBD to Epidiolex; of these, three had no worthwhile improvement, three experienced a > 50% reduction in seizure frequency and three had a 25%–49% reduction. Among the six patients who had tried another form of CBD at least six months prior, three had no worthwhile improvement, one had a > 50% reduction and two had a 25%–49% reduction in seizure frequency. One patient was seizure-free on another form of CBD before switching to Epidiolex. Overall, there appeared to be no clear difference in response rates between patients who were new to CBD and those who had prior exposure or switched formulations.
Safety and tolerability
Eighteen patients (30%) experienced AEs. There was no observed worsening of status epilepticus in the cohort. The most common AEs included lethargy (n = 7); gastrointestinal symptoms (n = 4), of which two had diarrhea; seizure worsening (n = 3); tremor (n = 2); and behavioral changes, cognitive effects and sleep disturbances (n = 1 each). Among the 34 (56%) patients receiving concomitant valproic acid, none experienced significantly elevated transaminases. CBD was discontinued in 18 (30 %) patients and temporarily discontinued but later restarted in 1 patient. Reasons for discontinuation included seizure worsening (7), lack of efficacy (5), AEs (4) and cost (1).
Comorbidities and retention
Although comorbidity data were not systematically collected in this study, documented patient-reported comorbidity changes after CBD treatment were predominantly positive. Mood improved in 16 patients (26%) and worsened in 3 (4.9%); behavior improved in 19 patients (31%) and worsened in 2 (3.3%), and sleep improved in 11 patients (18%) and worsened in 2 (3.3%). Patients continuing CBD despite limited seizure frequency reduction often reported meaningful improvements in these epilepsy comorbidities. Among those with 25%–50% reduction, 15 patients remained on CBD, as 8 (53%) noted reduced seizure intensity, 5 (33%) gained seizure-free days despite ongoing daily seizures, 7 (47%) experienced behavioral improvements and 4 (27%) had better sleep. Even among those with no worthwhile improvement, 11 remained on CBD with reported benefits in behavior (27%) or seizure intensity (18%). Refer to Table 4.
Table 4. Subset no improvement but still on cannabidiol

Note: Number of patients (n).
Discussion
This study is among the first evaluations of highly purified medicinal CBD in Canadian pediatric and young adult populations. The 22% responder rate and 7% seizure freedom rate, though lower than clinical trial results, Reference Thiele, Marsh and French11–Reference Thiele, Bebin and Bhathal13 exceed typical responses after three or more failed ASMs, Reference Chen, Brodie, Liew and Kwan5 suggesting CBD’s value as adjunctive therapy for highly DRE. CBD was well tolerated, with only 6.5% discontinuing due to AEs, such as lethargy and gastrointestinal symptoms.
Many patients experienced benefits beyond seizure reduction. Among those with 25%–49% seizure reduction, over half reported decreased seizure severity, one-third achieved seizure-free days, nearly half experienced behavioral improvements and over a quarter noted better sleep.
Efficacy
The overall response rates in our cohort were lower than those reported in pivotal clinical trials. Among our patients, 22% achieved a > 50% reduction in seizure frequency (including 7% who became seizure-free), compared with 29%–56% reported in randomized controlled trials (RCTs) for LGS, DS and TSC. Several factors likely contributed to this difference.
First, our cohort was more treatment-refractory. Patients had tried a median of eight prior ASMs, compared with four in DS and TSC trials and six in LGS trials. Reference Thiele, Marsh and French11–Reference Thiele, Bebin and Bhathal13 Additionally, our patients had extensive treatment histories, including prior surgical interventions and neuromodulation, reflecting great disease severity. A comparable US pediatric real-world study reported higher responder rates ( ≥50% seizure reduction), likely given a less refractory cohort: 12% had primary generalized epilepsy, and 19.2% had trialed more than four ASMs, versus 85% in our cohort. Reference Aizara, Robin and Hanna17 Our findings align with a systematic review of CBD in children with DEEs, where CBD led to a ≥ 50% reduction in seizure frequency in at least 20% of patients included in 11 studies. Reference Saranti, Dragoumi, Pavlogiannis, Pavlou and Zafeiriou18 This similarity likely reflects comparable treatment refractoriness, with patients in those studies having tried up to nine ASMs.
Second, the median CBD dose in our study was 15 mg/kg/day (range: 2.5–20 mg/kg/day), which may have contributed to lower efficacy. In RCTs, doses of 20 mg/kg/day were used for DS and LGS and 25–50 mg/kg/day for TSC, with comparable efficacy between doses but more AEs at higher doses. Real-world studies and expanded-access programs have used doses as high as 50–112 mg/kg/day. Reference Aizara, Robin and Hanna17,Reference Talwar, Estes, Aparasu and Reddy19 Some evidence supports the potential benefit of higher doses in specific contexts. Reference Lattanzi, Brigo and Trinka20–Reference Szaflarski, Hernando and Bebin22 However, the dose-response relationship for CBD is incompletely understood and not consistently observed. Reference Talwar, Estes, Aparasu and Reddy19
Third, concomitant clobazam use was substantially lower in our cohort (31%) compared with clinical trials (48% in LGS, 65% in DS and 23%–26% in TSC). Reference Thiele, Marsh and French11–Reference Thiele, Bebin and Bhathal13 Enhanced efficacy with concomitant clobazam use has been consistently observed across epilepsy syndromes, including in meta-analyses of LGS and DS trials. Reference Devinsky, Thiele and Wright23 Likely to mitigate potential AEs of somnolence from CBD-mediated increased N-desmethylclobazam metabolite levels, Reference Lattanzi, Zaccara, Russo, La Neve, Lodi and Striano24 in this cohort, clobazam was preemptively reduced in five patients, while eight patients had their clobazam reduced after starting CBD, despite only four experiencing AEs from this combination.
Fourth, a subset of our patients were not CBD-naïve, which may have influenced outcomes. Nine patients were switched from other CBD formulations to Epidiolex. Those who switched could have already benefited from initial CBD exposure, so the switch might not yield a substantial additional reduction in seizure frequency. Six patients had used CBD and discontinued CBD at least six months prior to starting Epidiolex. Prior exposure and discontinuation may reflect waning responsiveness or a lower likelihood of benefit upon re-initiation. These individuals likely represent a particularly refractory subgroup, having exhausted other CBD products and multiple therapies before moving to a pharmaceutical-grade formulation, potentially biasing outcomes toward lower efficacy. Although the sample size is small, these findings highlight the importance of accounting for prior CBD exposure when interpreting treatment outcomes.
The median follow-up duration of four months likely did not negatively affect efficacy outcomes. Although this duration exceeded the 14–16 weeks typical of pivotal RCTs, it may have allowed for more accurate capture of natural disease fluctuations and treatment plateaus not evident in shorter trials. Expanded-access program results over 144 weeks Reference Laux, Bebin and Checketts25,Reference Weinstock, Bebin and Checketts26 have demonstrated sustained seizure reduction with CBD.
Discontinuation of concomitant ASM
Thirty percent of patients reduced at least one concomitant ASM, even though only 22% achieved >50% seizure reduction. CBD may have replaced another ASM, or it may have reduced seizure intensity enough to enable tapering. Notably, 23% reported less severe seizures, and 16% of those with daily seizures had intermittent seizure-free days, indicating meaningful benefits beyond the standard responder definition.
Other DEEs
A substantial proportion of our cohort (39%) had DEE other than DS, LGS or TSC, syndromes represented in regulatory CBD trials. Efficacy did not differ significantly by epilepsy diagnosis, indicating potential benefit across diverse epilepsy syndromes, consistent with prior experimental evidence. Reference Howlett, Breivogel, Childers, Deadwyler, Hampson and Porrino10,Reference Consroe and Wolkin27 Our results concur with other open-label and observational studies in both children and adults with epilepsies beyond those included in trials. Reference Lattanzi, Trinka and Striano28
Safety and tolerability
Our study demonstrated more favorable tolerability compared to pivotal clinical trials and prior real-world studies. AEs occurred in 30% of patients, substantially lower than the safety profile reported in previous studies, which is relatively homogeneous with a percentage of AEs between 74% and 93%. Reference Devinsky, Cross and Laux12,Reference Devinsky, Patel and Thiele29 The most commonly reported AEs were lethargy (11%), gastrointestinal symptoms (7%) and seizure worsening (5%), with isolated cases of tremor, behavioral changes, cognitive effects and sleep disturbances, which is similar to other pediatric studies. Reference Aizara, Robin and Hanna17,Reference Talwar, Estes, Aparasu and Reddy19
Our withdrawal rate (30%) was within the range of previous studies (7%–28.4%). Reference Devinsky, Nabbout and Miller15,Reference Devinsky, Marsh and Friedman30 Withdrawals were primarily due to CBD inefficacy and rarely to AEs (7%). This low rate of discontinuation due to AEs likely reflects our conservative titration protocol, targeting 10–20 mg/kg/day over at least four weeks compared to the 20–25 mg/kg/day achieved in 2–4 weeks in clinical trials. Gradual dose escalation may improve tolerance, including with concomitant clobazam, without compromising efficacy. Reference D’Onofrio, Kuchenbuch and Hachon-Le Camus31 In addition, a decrease in AE over time may have occurred due to better patient tolerance due to slow titration of CBD. Similarly, another pediatric study showed lower AEs and higher retention with slower titration rates. Reference D’Onofrio, Kuchenbuch and Hachon-Le Camus31
Liver function test elevations occurred during CBD treatment in clinical trials, Reference Thiele, Marsh and French11–Reference Devinsky, Nabbout and Miller15,Reference Devinsky, Patel and Thiele29,Reference Miller, Scheffer and Gunning32 predominantly in patients taking concomitant valproic acid. Since CBD does not affect systemic concentrations of Valproic acid (VPA) or its metabolite 4-ene-VPA, Reference Ben-Menachem, Gunning and Arenas Cabrera33 the interaction is likely pharmacodynamic rather than pharmacokinetic, possibly mediated through effects on hepatic mitochondria. Reference Lattanzi, Brigo and Trinka20 In our study, two patients had VPA reduced before initiating CBD, and three had VPA lowered during treatment, though only one was due to lethargy. Notably, no patients developed clinically significant transaminase elevations, contrasting with the 7%–23% incidence in previous studies. The absence of hepatotoxicity in our cohort, despite 56% receiving concomitant valproate, is perhaps because slow titration mitigates this risk. Similarly, another pediatric study using slow titration reported no discontinuations due to hepatotoxicity. Reference D’Onofrio, Kuchenbuch and Hachon-Le Camus31
Non-seizure benefits
Despite only 22% achieving >50% seizure reduction, 70% continued CBD therapy, suggesting meaningful benefits beyond seizure control. Patients typically discontinue ineffective ASMs, yet our high retention rate indicates value in dimensions not captured by seizure frequency alone. Although likely underestimated due to non-systematic assessment, families reported improvements in mood (26%), behavior (31%) and sleep (18%).
These benefits were particularly evident among patients with suboptimal reduction in seizure frequency. In the 25%–50% seizure reduction group (n = 15), 53% reported reduced seizure intensity, 47% experienced behavioral improvements, 33% gained seizure-free days and 27% had better sleep. Even among those without meaningful seizure improvement (n = 11), some noted benefits in behavior (27%) or seizure intensity (18%), suggesting CBD’s therapeutic value extends to seizure severity, duration and recovery time, factors highly meaningful to families. These findings align with a caregiver survey where 93% planned to continue CBD treatment, citing reduced seizure burden and improvements in non-seizure outcomes, even among patients with unchanged or worsened seizure frequency. Reference Marshall, Skrobanski, Moore-Ramdin, Kornalska, Swinburn and Bowditch34
Emerging data support CBD’s effects on epilepsy comorbidities. One adult study found 71.5% of patients with drug-resistant focal epilepsy reported significant improvements in anxiety (71%) and quality of life (68%). Reference Lamonarca, Mintz, Bayarres, Kochen and Oddo35 While one study found no quality-of-life improvement, possibly due to profound intellectual disabilities, Reference Kim, Choi and Koo36 a prospective pediatric study demonstrated significant increases in Quality of Life in Childhood Epilepsy scores after 12 weeks, with improvements in memory, cognition, social interactions and behavior. Reference Rosenberg, Louik, Conway, Devinsky and Friedman37 These effects may result from CBD-induced changes in connectivity related to executive functioning and emotional control, Reference Allendorfer, Nenert and Bebin38–Reference Gaston, Allendorfer and Nair40 potentially mediated through endocannabinoid system facilitation of neurotransmission at 5HT1A and CB1 receptors. Reference Sartim, Guimarães and Joca41
Limitations
While our study demonstrates CBD’s efficacy in a highly refractory epilepsy cohort, exceeding expected outcomes after three or more failed ASMs, several limitations warrant consideration. The variability in seizure etiology, types and treatment histories may introduce confounding factors affecting CBD efficacy. All patients had a minimum of four months of follow-up, therefore restricting assessment of long-term efficacy and tolerability. Our population had highly refractory epilepsy with CBD initiated late in the disease course, potentially limiting generalizability to other cohorts. The Jazz Assist program may have facilitated treatment continuation among patients experiencing partial benefits, such as reduced seizure intensity or improvements in comorbidities, that might not have been sustained if cost had been prohibitive. This could bias retention rates upward; however, families typically discontinue medications that are clearly ineffective or poorly tolerated regardless of financial support. The retrospective design limits control for potential biases, including varying baseline seizure frequencies and concomitant ASM use. Additionally, while our findings suggest improvements in epilepsy comorbidities, seizure-free days and seizure intensity, a lack of use of validated questionnaires and systematic documentation may have affected these observations, potentially under- or overestimating the benefits.
Conclusion
This study represents one of the first evaluations of highly purified medicinal CBD in Canadian pediatric and young adult populations. While the 22% responder rate and 7% seizure freedom rate were lower than clinical trial results, they exceed typical responses after three or more failed ASMs, supporting CBD’s value as an adjunctive therapy for highly DRE. CBD provides meaningful improvements in mood, behavior and sleep even when seizure reduction is not ≥50%, indicating multifaceted benefits and strong acceptability with a 70% retention rate. Future real-world studies should include broader epilepsy populations and validated quality-of-life, mood/behavior and caregiver-burden measures to define CBD’s role in comprehensive epilepsy management.
Acknowledgments
We would like to thank Akshdeep Sandhu for his assistance with statistics.
Author contributions
KK: Data collection, editing. RG: Data collection, editing. CB: Data collection, editing. LH: Data collection, editing. MC: Data collection, editing. AD: Conceptualization, data collection, writing, editing.
Funding statement
This study received no funding.
Competing interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Linda Huh has served on the advisory board for Jazz Pharmaceuticals and served as chair of the BC Epilepsy Society. Mary Connolly is part of clinical trials for Jazz Pharmaceuticals and GRIN Therapeutics. She has received honoraria for speaking for Jazz Pharmaceuticals. Anita Datta has received honoraria for speaking for Jazz and BioMarin Pharmaceuticals. She has served on the advisory board for Acadia and Pendopharm pharmaceuticals. Kyah Kang, Ria Grewal and Cyrus Boelman have no declaration of competing interests. This study was conducted independently without any involvement from Jazz Pharmaceuticals.
Ethical standard
Approved by the University of British Columbia/Children’s and Women’s Health Centre of British Columbia Research Ethics Board: UBC C&W REB: H25-00392-A003






Target article
Highly Purified Cannabidiol Treatment Outcomes in Pediatric Patients with Highly Refractory Epilepsy at a Tertiary Center
Related commentaries (1)
Reviewer Comment on Kang et al. “Highly Purified Cannabidiol Treatment Outcomes in Pediatric Patients with Highly Refractory Epilepsy at a Tertiary Center”