Introduction
Malaria, for its rapid reproduction and its concentration in tropical/subtropical regions where access to intervention and treatment is hindered, remains a major global health threat today – in 2022 alone, c. 250 million cases occurred globally, with 610 000 people dying, most of which were subadults in sub-Saharan Africa (CDC, 2024a). It is a mosquito-borne infectious disease caused by Plasmodium parasites, different species are endemic in different regions: P. falciparum, i.e. malaria tropica, is prevalent in the African continent, causing the highest mortality; P. vivax is the most commonly encountered species elsewhere, causing lower mortality than its African counterpart but toll nonetheless, especially in both children younger than 5 as well as pregnant women.
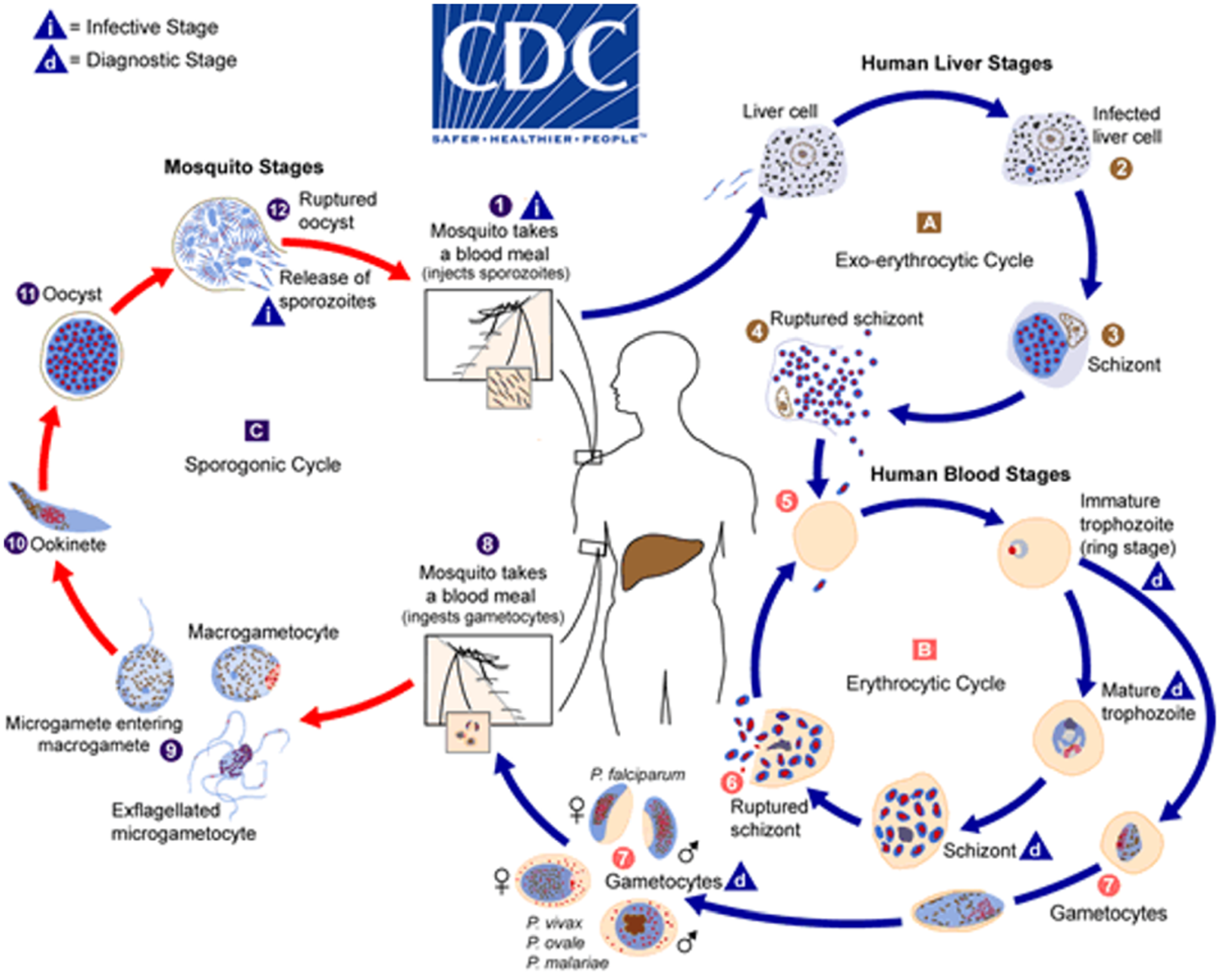
A full malaria lifecycle (Figure 1) – complex on its own for its sexual as well as asexual reproduction stages – involves 2 hosts, Anopheles mosquito and human. The bite of a mosquito inoculates sporozoites into human host, which would infect liver cells then red blood cells (RBCs), the latter would be destroyed post-reproduction. This very destruction of RBCs is what causes the symptoms, from mild ones like chills, nausea, headache, etc., to fatal outcomes.
Lifecycle of the genus Plasmodium. After CDC (2024b).

The selection pressure imposed by malaria is strong enough to shape the path of human evolution (Newfield, Reference Newfield2017) – as the evidence from the European and African continents suggests (Schats, Reference Schats2023), malaria had long troubled ancient societies and caused mayhem. This is not to say that, sheltered by more advanced modern medical technologies, we are now exempted from such pressure: that P. simium has recently transmitted from monkey to human host in Brazil (Brasil et al., Reference Brasil, Zalis, de Pina-costa, Siqueira, Júnior, Silva, Areas, Pelajo-Machado, de Alvarenga, da Silva Santelli, Albuquerque, Cravo, Santos de Abreu, Peterka, Zanini, Suárez Mutis, Pissinatti, Lourenço-de-oliveira, de Brito, de Fátima Ferreira-da-cruz, Culleton and Daniel-Ribeiro2017), along with the aforementioned global toll, constantly warns us the nature of malaria: it has historically been, and will still be, one of the most imposing diseases. However, one may stress that we humans distinguish ourselves from the rest of animal kingdom by our capacity to alter the environment we dwell in – to such an extent that some even name the era from the mid-20th century onwards as ‘Anthropocene’ (Yan et al., Reference Yan, Han, An, Lei, Zhao, Zhao, Liu and Capo2024), as we seemingly have changed the environment far enough from what it used to be. Regardless of the validity of this concept, in our case of malaria transmission, the impact of human activities is evident: a successful malarial infection hinges upon the bite of mosquito host, thus any human behaviour that may proliferate or impede the reproduction of mosquito could lead to wax or wane in malarial infections. Anthropogenic factors linked to high prevalence of malaria infection, as summarized by Shin et al. (Reference Shin, Seo, Hong and Le2018: 4–5), include irrigation facilities expansion, wetland reclamation for farming, economic specialization in agriculture, deforestation, etc. Again, that humans change local environments that are conducive to malaria transmission is not exclusive to ancient societies: during the British colonial period in India, British colonizers extensively cleared forests to boost tea cultivation, dramatically altering Assam’s ecosystem. While this environmental shift initially boosted the tea industry, it inadvertently created ideal breeding grounds for Aedes mosquitoes, thereby exacerbating malaria transmission. Notably, studies of the 1920s had revealed a strong correlation between deforestation and the dramatic surge in malaria cases (Friederich, Reference Friederich2021).
The historical human–mosquito–malaria interplay has been well studied in regions like Europe and Africa (Shin et al., Reference Shin, Seo, Hong and Le2018: 4–5). However, evidence for such grand scale interplay from Asia, especially those eastern region, has somewhat attracted less attention, despite the region’s endemic nature and plethora of written records on this very parasite. We aim to address this knowledge gap through systematically reviewing available evidence in East, Southeast and South Asia regarding: 1) the origin and further dispersal of Plasmodium parasites, 2) anthropogenic factors of malaria endemics and 3) the malarial health consequences to, and societal reactions of, ancient Chinese societies. For 1), the reviewing emphasis is placed upon genetic studies of such parasites; for 2), that upon how lifestyle changes and social-economic activities on humans’ end had an impact on malarial transmission. For 3), we ought to stress our focus on Chinese materials. This is due to its continuance of written records on malaria parasites – the earliest of which can be dated back to the oracle bone script era – and a striking dearth of literature coverage on these records as well as potential bioarchaeological evidence for malaria, i.e. porotic skeletal lesions such as cribra orbitalia (CO). While in recent years ancient Chinese medical/historical descriptions on malaria have gained some deserved attention (Miao, Reference Miao2025), there remains no review concerning the cases of CO in China as of yet. We have compiled the available evidence of CO in ancient China, which would in turn be the prerequisite for future analysis on the geological relationship among, and regional ecologies of, these CO sites – a viable research pathway for studying ancient malaria as suggested by Schats (Reference Schats2023).
Genetic insights on Plasmodium origin and dispersal
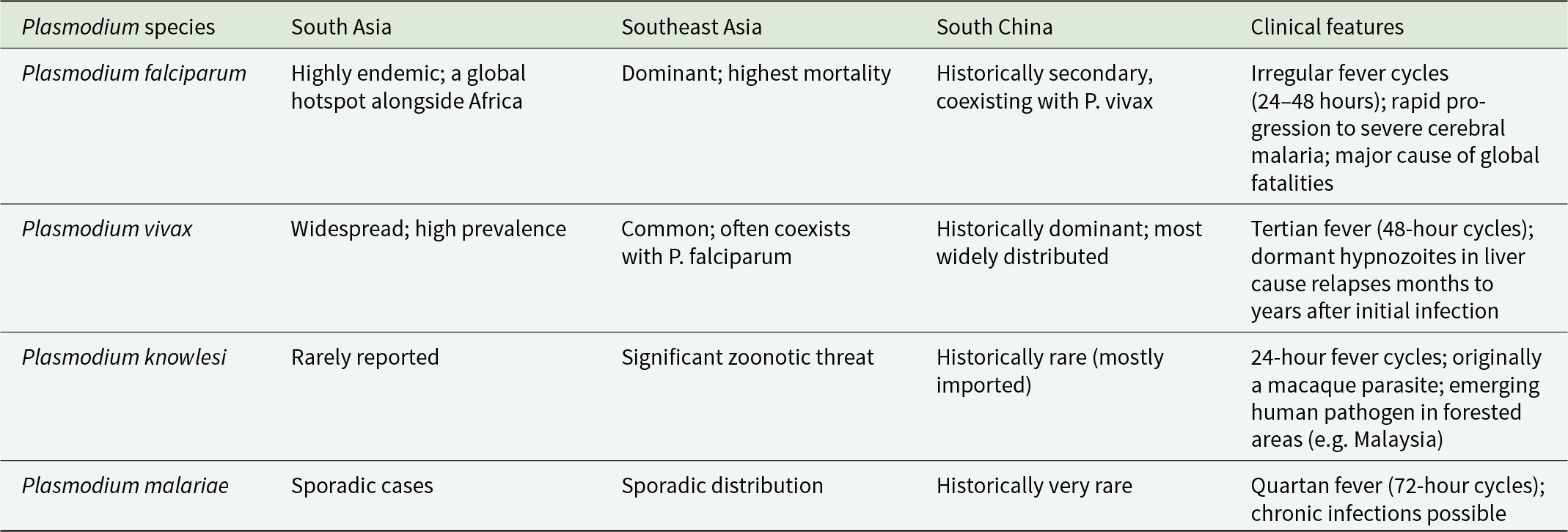
To understand health in the past, one has to determine whether the source of disease had originated in Africa and infected humans and our ancestors throughout the evolution; or it was acquired by humans on our travels to other parts of the globe. For parasites, the former is termed ‘heirloom parasites’, while the latter ‘souvenir parasites’ (Mitchell, Reference Mitchell2013). P. falciparum and P. vivax fall into the heirloom category, both share African origins, yet the specific times for their African origin and exodus might differ. Interestingly, P. vivax was once believed to be of Asian origin, with scholars such as Daron et al. (Reference Daron, Boissièr, Boundenga, Ngoubangoye, Houze, Arnathau, Sidobre, Trape, Durand, Renaud, Fontaine, Prugnolle and Rougeron2021) still proposing this notion. Nevertheless, it is now mostly believed to have travelled with human groups outside of Africa prior to 30 000 years ago, with all extant human P. vivax being traced back to a single ancestor (Liu et al., Reference Liu, Li, Shaw, Learn, Plenderleith, Malenke, Sundararaman, Ramirez, Crystal, Smith, Bibollet-Ruche, Ayouba, Locatelli, Esteban, Mouacha, Guichet, Butel, Ahuka-Mundeke, Inogwabini, Ndjango, Speede, Sanz, Morgan, Gonder, Kranzusch, Walsh, Georgiev, Muller, Piel, Stewart, Wilson, Pusey, Cui, Wang, Färnert, Sutherland, Nolder, Hart, Hart, Bertolani, Gillis, LeBreton, Tafon, Kiyang, Djoko, Schneider, Wolfe, Mpoudi-Ngole, Delaporte, Carter, Culleton, Shaw, Rayner, Peeters, Hahn and Sharp2014; Sharp et al., Reference Sharp, Plenderleith, Culleton and Hahn2024). Interestingly, ancestral P. vivax might have been able to infect humans as well as gorillas and chimpanzees in Africa, until a mutation that favoured P. vivax protection occurred in humans and eliminated infection cases there – this might explain why such protection to P. vivax is prevalent in the continent where this very parasite is absent (Liu et al., Reference Liu, Li, Shaw, Learn, Plenderleith, Malenke, Sundararaman, Ramirez, Crystal, Smith, Bibollet-Ruche, Ayouba, Locatelli, Esteban, Mouacha, Guichet, Butel, Ahuka-Mundeke, Inogwabini, Ndjango, Speede, Sanz, Morgan, Gonder, Kranzusch, Walsh, Georgiev, Muller, Piel, Stewart, Wilson, Pusey, Cui, Wang, Färnert, Sutherland, Nolder, Hart, Hart, Bertolani, Gillis, LeBreton, Tafon, Kiyang, Djoko, Schneider, Wolfe, Mpoudi-Ngole, Delaporte, Carter, Culleton, Shaw, Rayner, Peeters, Hahn and Sharp2014). Nonetheless, P. falciparum, on the other hand, had likely emerged in Africa c. 60 000 to 40 000 years ago – its success in infecting humans, however, might be a more recent event, i.e. around 5000 years ago when P. falciparum underwent a genetic bottleneck (Otto et al., Reference Otto, Gilabert, Crellen, Böhme, Arnathau, Sanders, Oyola, Okouga, Boundenga, Willaume, Ngoubangoye, Moukodoum, Paupy, Durand, Rougeron, Ollomo, Renaud, Newbold, Berriman and Prugnolle2018). In our context, both species along with P. knowlesi and P. malariae are endemic in Asia and have imposed selection pressure on the populations (Table 1). The latter 2 species, though covered with less literature due to lower infection frequency, still infect humans to date (Jeyaprakasam et al., Reference Jeyaprakasam, Liew, Low, Wan-Sulaiman and Vythilingam2020; Zhang et al., Reference Zhang, Yi, Zhou, Xia and Yin2023). P. knowlesi is thought to be in Southeast Asia prior to human settlement, infecting mainly macaques, underwent population expansion c. 30 000–40 000 years ago (Lee et al., Reference Lee, Divis, Zakaria, Matusop, Julin, Conway, Cox-Singh and Singh2011); P. malariae, on the other hand, of zoonic origin from African apes (Plenderleith et al., Reference Plenderleith, Liu, Li, Loy, Mollison, Connell, Ayouba, Esteban, Peeters, Sanz, Morgan, Wolfe, Ulrich, Sachse, Calvignac-Spencer, Leendertz, Shaw, Hahn and Sharp2022).
Plasmodium distribution and clinical features in South, Southeast and East Asia

Detection of malaria aDNA has been somewhat capricious – this problem becomes more evident when studying P. vivax, for the lower parasitic load it causes than does P. falciparum (>5% in the latter while < 2% in the former, Anstey et al., Reference Anstey, Russell, Yeo and Price2009). Another contributing factor to the dearth of aDNA evidence for malaria parasites is the lack of studied samples: in China, for instance, almost no archaeological site to date has been found positive with malarian genome. If we were to extend our scope to the entirety of Asia, we may find the following remarks, however disarrayed:
1. A study on mt/api genome of P. falciparum of China–Myanmar border isolates (Tian et al., Reference Tian, Ye, Zhang and Zhang2024) collected from 37 symptomatic malaria patients finds that this parasite had dwelled in this region long before being introduced into other Southeast Asian places, expanded slowly with population expansion between c. 12 500–20 000 ybp.
2. Studies on ancient Plasmodium genomes suggest that there seems to be affinities between European and South Asian P. falciparum populations, but more data are required to elucidate (de-Dios et al., Reference de-Dios, van Dorp, Gelabert, Carøe, Sandoval-Velasco, Fregel, Escosa, Aranda, Huijben, Balloux, Gilbert and Lalueza-Fox2019; Michel et al., Reference Michel, Skourtanioti, Pierini, Guevara, Mötsch, Kocher, Barquera, Bianco, Carlhoff, Coppola Bove, Freilich, Giffin, Hermes, Hiß, Knolle, Nelson, Neumann, Papac, Penske, Rohrlach, Salem, Semerau, Villalba-Mouco, Abadie, Aldenderfer, Beckett, Brown, Campus, Chenghwa, Cruz Berrocal, Damašek, Duffett Carlson, Durand, Ernée, Fântăneanu, Frenzel, García Atiénzar, Guillén, Hsieh, Karwowski, Kelvin, Kelvin, Khokhlov, Kinaston, Korolev, Krettek, Küßner, Lai, Look, Majander, Mandl, Mazzarello, McCormick, de Miguel Ibáñez, Murphy, Németh, Nordqvist, Novotny, Obenaus, Olmo-Enciso, Onkamo, Orschiedt, Patrushev, Peltola, Romero, Rubino, Sajantila, Salazar-García, Serrano, Shaydullaev, Sias, Šlaus, Stančo, Swanston, Teschler-Nicola, Valentin, Van de Vijver, Varney, Vigil-Escalera Guirado, Waters, Weiss-Krejci, Winter, Lamnidis, Prüfer, Nägele, Spyrou, Schiffels, Stockhammer, Haak, Posth, Warinner, Bos, Herbig and Krause2024); also, modern P. falciparum displays lower genetic diversity than P. vivax (Michel et al., Reference Michel, Skourtanioti, Pierini, Guevara, Mötsch, Kocher, Barquera, Bianco, Carlhoff, Coppola Bove, Freilich, Giffin, Hermes, Hiß, Knolle, Nelson, Neumann, Papac, Penske, Rohrlach, Salem, Semerau, Villalba-Mouco, Abadie, Aldenderfer, Beckett, Brown, Campus, Chenghwa, Cruz Berrocal, Damašek, Duffett Carlson, Durand, Ernée, Fântăneanu, Frenzel, García Atiénzar, Guillén, Hsieh, Karwowski, Kelvin, Kelvin, Khokhlov, Kinaston, Korolev, Krettek, Küßner, Lai, Look, Majander, Mandl, Mazzarello, McCormick, de Miguel Ibáñez, Murphy, Németh, Nordqvist, Novotny, Obenaus, Olmo-Enciso, Onkamo, Orschiedt, Patrushev, Peltola, Romero, Rubino, Sajantila, Salazar-García, Serrano, Shaydullaev, Sias, Šlaus, Stančo, Swanston, Teschler-Nicola, Valentin, Van de Vijver, Varney, Vigil-Escalera Guirado, Waters, Weiss-Krejci, Winter, Lamnidis, Prüfer, Nägele, Spyrou, Schiffels, Stockhammer, Haak, Posth, Warinner, Bos, Herbig and Krause2024).
3. P. vivax seems to have been more prevalent than P. falciparum and P. malariae in Eurasia. Studies such as Michel et al. (Reference Michel, Skourtanioti, Pierini, Guevara, Mötsch, Kocher, Barquera, Bianco, Carlhoff, Coppola Bove, Freilich, Giffin, Hermes, Hiß, Knolle, Nelson, Neumann, Papac, Penske, Rohrlach, Salem, Semerau, Villalba-Mouco, Abadie, Aldenderfer, Beckett, Brown, Campus, Chenghwa, Cruz Berrocal, Damašek, Duffett Carlson, Durand, Ernée, Fântăneanu, Frenzel, García Atiénzar, Guillén, Hsieh, Karwowski, Kelvin, Kelvin, Khokhlov, Kinaston, Korolev, Krettek, Küßner, Lai, Look, Majander, Mandl, Mazzarello, McCormick, de Miguel Ibáñez, Murphy, Németh, Nordqvist, Novotny, Obenaus, Olmo-Enciso, Onkamo, Orschiedt, Patrushev, Peltola, Romero, Rubino, Sajantila, Salazar-García, Serrano, Shaydullaev, Sias, Šlaus, Stančo, Swanston, Teschler-Nicola, Valentin, Van de Vijver, Varney, Vigil-Escalera Guirado, Waters, Weiss-Krejci, Winter, Lamnidis, Prüfer, Nägele, Spyrou, Schiffels, Stockhammer, Haak, Posth, Warinner, Bos, Herbig and Krause2024) and Sikora et al. (Reference Sikora, Canteri, Fernandez-Guerra, Oskolkov, Ågren, Hansson, Irving-Pease, Mühlemann, Holtsmark Nielsen, Scorrano, Allentoft, Valeur Seersholm, Schroeder, Gaunitz, Stenderup, Vinner, Jones, Nystedt, Sjögren, Parkhill, Fugger, Racimo, Kristiansen, Iversen and Willerslev2025) have found aDNA evidence for P. vivax in burials from the Neolithic and Bronze Age, with all 3 malarial species becoming more common over the last 1000 years.
We thus could state with confidence that malaria parasites have accompanied humans during our evolution in Africa, along with our migration to the rest of the world. However, their genetic potential for infecting humans should not have been fulfilled had we not modified the environment to our benefit. To better understand this apparent paradoxical interaction, we should direct our attention to the other party of interest, i.e. us humans.
Anthropogenic factors in malaria endemicity
While malaria parasites have been established to be heirlooms, co-evolving with us since our African origins, we should consider whether human activities have impacted our relationship with these parasites. Has early human behaviour altered the risk of malarial transmission? Evidence for ancient malaria in East and Southeast Asia can be seen to shed light on this question as humans marked their transition to agriculture.
Molecular biology sequencing and zoonotic infectious disease research have shown that the biological origin of human malaria is far more complex than previously thought, extending beyond the commonly known mosquito vectors to encompass primates as potential reservoirs (Capasso, Reference Capasso1998; Hartl, Reference Hartl2004; Plenderleith et al., Reference Plenderleith, Liu, Li, Loy, Mollison, Connell, Ayouba, Esteban, Peeters, Sanz, Morgan, Wolfe, Ulrich, Sachse, Calvignac-Spencer, Leendertz, Shaw, Hahn and Sharp2022). Malaria transmission is intrinsically tied to the ecology of Anopheles mosquito vectors, which varies across Asia’s diverse landscapes. Vectors such as A. minimus and A. dirus thrive in tropical forests and irrigated landscapes, with their distribution influenced by climate patterns and agricultural practices. Ancient human populations residing in tropical regions, characterized by dense forests and abundant water bodies, frequently came into direct contact with these malaria vectors, thereby facing significant and persistent infection risks. However, the absence of direct aDNA evidence poses a significant challenge, leaving the actual prevalence of such infections within Pleistocene hunter-gatherer communities to be shrouded in uncertainty. Notably, the harsh and colder climatic conditions that prevailed during the Ice Age (spanning from approximately 100 000 to 10 000 years ago) also played a crucial role in significantly constraining the transmission of malaria, due to the reduced activity and limited survival range of both mosquitoes and primates (Rich and Ayala, Reference Rich, Ayala, Dronamraju and Arese2006; Nosten et al., Reference Nosten, Richard-Lenoble and Danis2022). Evidence from the Con Co Ngua site in northern Vietnam, dating to at least the early 7000 BP, suggests the presence of thalassemia, a genetic adaptation to malaria, among hunter-gatherer communities (Oxenham et al., Reference Oxenham, Trinh, Willis, Jones, Domett, Castillo, Wood, Bellwood, Tromp, Kells, Piper, Pham, Matsumura and Buckley2018). It is thus evident that Plasmodium parasites had been exerting evolutionary pressure on human groups during the pre-agricultural era (Wang et al., Reference Wang, Dittmar, Inskip, Cessford and Mitchell2023a). These complex hunter-gatherers lived in forested areas near water sources, which was conducive to mosquito vectors like A. dirus, therefore faced year-round transmission risks (Vlok et al., Reference Vlok, Buckley, Miszkiewicz, Walker, Domett, Willis, Trinh, Minh, Nguyen, Nguyen, Matsumura, Wang, Nghia and Oxenham2021). The tropical climate of Southeast Asia would have permitted year-round transmission, even if at a lower intensity compared to later periods. However, the generally lower population density and higher mobility characteristic of many hunter-gatherer societies likely limited the sustained transmission of malaria, confining it to a more endemic rather than epidemic state. On the other hand, such sustained transmission, in combination with mobile hosts, has rendered the genetics of P. vivax highly diversified in Southeast Asia – indeed, studies using microsatellite markers indicate that diversity is highest in Southeast Asia compared to other regions, reflecting both endemicity and historical spread (Koepfli et al., Reference Koepfli, Robinson, Rarau, Salib, Sambale, Wampfler, Betuela, Nuitragool, Barry, Siba, Felger and Mueller2015). In Papua New Guinea and Thailand, P. vivax populations are more genetically diverse than sympatric P. falciparum, highlighting different evolutionary pressures (Jennison et al., Reference Jennison, Arnott, Tessier, Tavul, Koepfli, Felger, Siba, Reeder, Bahlo, Mueller and Barry2015).
As the weather gradually became warmer on a grand scale, agricultural subsistence patterns, whether endogenous or exogenous, successively emerged across diverse regions of Asia (Moore and Hillman, Reference Moore and Hillman1992). In comparison to our earlier hunter-gatherer days, humans had changed the environment more drastically, in which process made it more malaria conducive: Agriculture, mainly in the form of monsoon paddy fields, promoted population prosperity and settlement size, and indirectly led to social problems such as increased population density, ecological destruction and tension between people and land, providing a breeding ground for the epidemic of malaria. Thus, the transitional stage from hunting-gathering to agriculture is a crucial turning point and a central research focus for the study of malaria evolution in prehistory (Bellwood, Reference Bellwood and Bellwood2005; Sabbatani et al., Reference Sabbatani, Manfredi and Fiorino2010).
The Neolithic transition to agriculture occurred in mainland Southeast Asia around 4500–3100 BP and involved significant environmental modification, including land clearance and the creation of irrigation systems such as rice paddies and reservoirs (Bellwood, Reference Bellwood and Bellwood2005). The Khok Phanom Di site in northern Thailand provides a vivid case. By reconstructing its ancient environment, researchers discovered the site’s proximity to a marshland ecosystem. This unique setting served dual purposes: it was ideal for cultivating monsoon-dependent paddy fields while simultaneously creating a breeding ground for malaria. When the site’s inhabitants initiated agricultural practices around 2000 BCE, the resulting malaria outbreaks caused massive neonatal mortality (Higham and Thosarat, Reference Higham and Thosarat1994; Tayles, Reference Tayles1999). These natural or paddy-related aquatic environments provided ideal breeding grounds for anopheline mosquitoes, notably Anopheles minimus, the primary malaria vector in the region.
The history of malaria in ancient Korea could help illustrate how the flourishing of agriculture could have led to a heavier parasitic load (Shin et al., Reference Shin, Seo, Hong and Le2018: 3–5). Whilst close to China, the first written case of malaria only occurred in the Goryeo Dynasty (918–1392 CE) – this is not to state that the disease was somewhat less severe. On the contrary, even a Joseon King’s mother was heavily infected by malaria and eventually died of it. Between the 15th and 19th centuries, agricultural endeavours were highly regarded by the Joseon people, with their investments and innovations drastically changing the environment, despite unintentionally to the favour of malaria transmission. Virtually all lands available had been cultivated, and by the end of the Joseon Dynasty, rice had been raised as the preferred crop – farmlands were changed into rice paddies, supporting irrigation systems purposefully constructed, thus population expanded. An environment inductive to malaria transmission was hence produced by the intermingling of these factors.
From the Korean example, we may tell that, with the advancement of social progress, the specialization of human livelihoods – agriculture, in this case – has evidently had a profound impact on malaria transmission. Yet agriculture is not the sole factor responsible for that – various economic factors, such as the expansion of commercial activities and the increasing density of populations, have played pivotal roles in shaping human mobility patterns and demographic trends. It is noteworthy that the dynamics of both economic activities and population growth continue to be critical determinants of malaria prevalence, not only historically but also in modern times. A striking example of this interplay is the said colonial-era tea industry in the Assam region, where intense economic pressures ultimately resulted in severe labour shortages and significant livelihood challenges for the local population, thereby exacerbating the spread of malaria (Friederich, Reference Friederich2021).
The mobility of populations driven by various economic activities serves as a crucial transmission pathway for malaria, facilitating its spread from primary epidemic zones to surrounding regions. During the Northern Song period in ancient China, there were 59 major epidemic outbreaks, the majority of which were concentrated in the economically prosperous Yangtze River Delta (Wang, Reference Wang2014). A particularly notable case of a malaria epidemic occurred in the year 1087, when renowned figures Su Shi (苏轼) and Shen Kuo (沈括) documented the widespread prevalence of malaria in Zhejiang province. Their records indicate that even the highest-ranking officials, who had access to medical support, were not spared from infection (Gong and Liu, Reference Gong and Liu2011). These historical cases effectively demonstrate the significant overlap between malaria-endemic areas and regions that are economically developed, highlighting the intricate relationship between economic activities, population dynamics, and the spread of malaria.
In South China, the historical dominance of P. vivax is evidenced by historical and traditional medical records, though genetic data are sparse due to preservation challenges. As for vectors across regions, historical records document frequent population movements between South China and Vietnam, promoting cross-regional transmission. This also explains, to a certain extent, the similarity in the distribution of Plasmodium between South China and the Indochina Peninsula (Shetty et al., Reference Shetty, Jacob, Huang, Li, Agrawal, Saunders, Lon, Fukuda, Ringwald, Ashley, Han, Hlaing, Nyunt, Silva, Stewart, Plowe, O’Connor and Takala-Harrison2019). It is worth noting that with the development of navigation technology, starting from the 13th century, global trade gradually emerged from South China to Southeast Asia and even all over the coast of the Indian Ocean. Against this historical background, trade networks such as the Maritime Silk Road likely acted as conduits for Plasmodium dispersal. Yet genomic analyses reveal that South Asian P. falciparum shows closer affinity to African strains than to East Asian strains (Michel et al., Reference Michel, Skourtanioti, Pierini, Guevara, Mötsch, Kocher, Barquera, Bianco, Carlhoff, Coppola Bove, Freilich, Giffin, Hermes, Hiß, Knolle, Nelson, Neumann, Papac, Penske, Rohrlach, Salem, Semerau, Villalba-Mouco, Abadie, Aldenderfer, Beckett, Brown, Campus, Chenghwa, Cruz Berrocal, Damašek, Duffett Carlson, Durand, Ernée, Fântăneanu, Frenzel, García Atiénzar, Guillén, Hsieh, Karwowski, Kelvin, Kelvin, Khokhlov, Kinaston, Korolev, Krettek, Küßner, Lai, Look, Majander, Mandl, Mazzarello, McCormick, de Miguel Ibáñez, Murphy, Németh, Nordqvist, Novotny, Obenaus, Olmo-Enciso, Onkamo, Orschiedt, Patrushev, Peltola, Romero, Rubino, Sajantila, Salazar-García, Serrano, Shaydullaev, Sias, Šlaus, Stančo, Swanston, Teschler-Nicola, Valentin, Van de Vijver, Varney, Vigil-Escalera Guirado, Waters, Weiss-Krejci, Winter, Lamnidis, Prüfer, Nägele, Spyrou, Schiffels, Stockhammer, Haak, Posth, Warinner, Bos, Herbig and Krause2024), though it had also likely resulted from ancient human migration. This genetic connectivity underscores the role of regional networks in pathogen evolution.
Similar cases were noted in South Asia, too: Himalayan trade routes have historically facilitated gene flow of P. vivax in this region. Environmental drivers, including perennial breeding grounds created by Himalayan meltwater that sustain vector populations, mark the synergy between geography and disease spread. The case of P. falciparum in an individual from the high-altitude site of Chokhopani in Nepal, dating to 500–1000 BCE, illustrates how human mobility has the potential to spread malaria beyond endemic lowland regions. This individual likely contracted P. falciparum in the Terai lowlands, with vectors such as A. culicifacies (adapted to rural Indian rice paddies) and A. dirus (in Nepal’s Terai) facilitating transmission in riverine wetlands (Michel et al., Reference Michel, Skourtanioti, Pierini, Guevara, Mötsch, Kocher, Barquera, Bianco, Carlhoff, Coppola Bove, Freilich, Giffin, Hermes, Hiß, Knolle, Nelson, Neumann, Papac, Penske, Rohrlach, Salem, Semerau, Villalba-Mouco, Abadie, Aldenderfer, Beckett, Brown, Campus, Chenghwa, Cruz Berrocal, Damašek, Duffett Carlson, Durand, Ernée, Fântăneanu, Frenzel, García Atiénzar, Guillén, Hsieh, Karwowski, Kelvin, Kelvin, Khokhlov, Kinaston, Korolev, Krettek, Küßner, Lai, Look, Majander, Mandl, Mazzarello, McCormick, de Miguel Ibáñez, Murphy, Németh, Nordqvist, Novotny, Obenaus, Olmo-Enciso, Onkamo, Orschiedt, Patrushev, Peltola, Romero, Rubino, Sajantila, Salazar-García, Serrano, Shaydullaev, Sias, Šlaus, Stančo, Swanston, Teschler-Nicola, Valentin, Van de Vijver, Varney, Vigil-Escalera Guirado, Waters, Weiss-Krejci, Winter, Lamnidis, Prüfer, Nägele, Spyrou, Schiffels, Stockhammer, Haak, Posth, Warinner, Bos, Herbig and Krause2024).
In conclusion, the historical trajectory of malaria across Asia reveals that our relationship with Plasmodium parasites is a dynamic narrative shaped profoundly by human agency. While malaria is an ‘heirloom’ disease of African origins, the transition of humans from hunter-gatherer to agricultural sedentarism marked a fundamental shift in its transmission power. A recurring theme is that, as we humans modified the landscape in our favour, whether it was for agricultural prosperity or ease of communication, we also engineered ideal nurseries for malarial vectors of this infectious disease. The example of China in the subsequent section might serve as a case study, as it is among the regions where the earliest agricultural activities occurred. The surviving medical texts and human skeletal remains could help us understand what the ancients knew of malaria, and what was the health consequences of this disease.
Evidence for malaria and its consequences in ancient Chinese societies
China, despite its recent milestone of being certified as malaria-free by the World Health Organization in 2021 (WHO, 2021), has historically constituted a heartland of intense malaria transmission, especially in its southern regions. Chinese textual records illustrate a long and continuous history of human engagement with this disease spanning over 3 millennia.
Notwithstanding, one needs to justify the usage of historical texts for retrospective diagnosis, especially in times of translation – after all, ‘malaria’ is a 19th-century Italian term. How can we be certain that ‘疟疾’, the current Chinese translation of ‘malaria’, denotes this very disease in history, but not some others? Such diagnosis could only be possible when the materials allow it, i.e. where the evidence is specific and sufficiently detailed (Mitchell, Reference Mitchell2013: 9–17), and should only be performed ‘when the quality of the sources allow it, when the research team has sufficient understanding of the language, literature and cultural context to interpret the source appropriately, and when the research team knows enough about disease to determine which condition most closely matches the symptoms and signs described in those texts’. Our focus on Chinese written records on malaria in this section is due to the abundance of textual descriptions they contain.
We have now established that malaria has been endemic in China for several thousand years. The Chinese character used for malaria today ‘疟’, looks as if a tiger (on the right) is attacking a person (on the left). An example from early oracle bone script showing this can be seen in Figure 2. The association with a tiger shows the fear with which the people of the time regarded malaria. While the early texts that use this character are short in length and one might cast doubt on its meaning, written records from later periods provide more detailed accounts of symptoms that allow more confidence in linking the term with malaria.
Oracle bone script of the character ‘疟 (malaria)’, a rather intuitive hieroglyph depicting a human on the left being attacked by a tiger on the right.

For instance, more detailed description and treatment towards malaria had appeared in medical books ever since the Zhou dynasty commenced at c. 1046 BC.《周礼 · 疾医》(Rites of Zhou – Diseases and Medicines) writes ‘四时皆有疠疾:春时有瘠首疾,夏时有痒疥疾,秋时有疟寒疾,冬时有嗽上气疾 (There are diseases in all seasons: head illness in spring, itchy scabies in summer, malaria in autumn, and coughing in winter)’, suggesting that people believed malaria is associated with weathers in autumn (Li, Reference Li1999). By the 6th century BC, along with the advances in human societies, malaria had also become a threat to not only the general population but rulers as well. 左传 (Zuo Zhuan/The Commentary of Zuo) comprises 30 chapters covering the period from 722 to 468 BC. In 566 BC, it wrote:
‘子驷使贼夜弑僖公,而以疟疾赴于诸侯。’
‘Zisi sent assassin to kill Xigong in the evening, then lied to others that he died of malaria’.
Nevertheless, though in current endemic settings, malaria does have ‘seasonal peaks’, its pattern is less marked (Roca-Feltrer et al., Reference Roca-Feltrer, Schellenberg, Smith and Carneiro2009). Thus, there could still be room for doubt on whether ‘疟’ in ancient texts genuinely means what we understand as malaria. However, notes on symptoms of ‘疟’ in ancient Chinese medical texts should make it clear that, while ‘疟’ of ancient China and malaria may not be exact synonyms, the former should in worst cases be a hypernym as it includes characteristic symptoms of malaria. 黄帝内经 (Inner Canon of the Yellow Emperor) and 五十二病方 (52 Ailments) – some of the earliest medical books – both have detailed descriptions and treatments for malaria. The former described that the symptoms of malaria include:
‘……寒栗鼓颈,腰脊俱痛,寒去则内外皆热,颈痛如破,渴欲冷饮。’
‘…… A chill runs through the neck, and pain in the lower back and spine. When the chills subside, heat returns both internally and externally. The neck pain feels like it’s about to burst, and there is a thirst that craves cold drinks’.
Fifty-Two Ailments refers to malaria as ‘痎疟 (malaria that occurs every two days)’ and records treatments using Artemisia species (Zhang, Reference Zhang and Zhang2017). As noted, the destruction of RBCs by malarian parasites would cause symptoms of chills and fevers; in addition, the symptoms would occur in a consistent and cyclical way – i.e. every 2–3 days – following the release of parasites into the bloodstream from the destroyed RBCs (Loomis, Reference Loomis and Loomis2020). Such description of symptoms matches that of malaria as we understand.
An even more convincing argument for connecting ‘疟’ in ancient China with malaria comes from the fact that medical treatments for the former had directly inspired a Nobel Prize winner to successfully extract a powerful antimalarial compound. The Wei, Jin, and the Northern and Southern Dynasties (AD 220–589) saw a great amount of migration, with northerners moving to the south to avoid wars; thus, more people were at risk of contracting malaria. 肘后备急方 (A Handbook of Prescriptions for Emergencies), a medical handbook characterized by its ‘easiness, handiness, shortness, and effectiveness (简、便、廉、验)’, was written during this time by Ge Hong (East Jin Dynasty per se, around AD 317–420). It stood out as one of the first to systematically prescribe Artemisia carvifolia to treat malaria symptoms. A. carvifolia contains artemisinin, one of the most effective compounds to eliminate Plasmodium parasites from the bloodstream. Tu Youyou was awarded the Nobel Prize in Physiology or Medicine 2015 for extracting this compound, and she stated that Ge Hong’s masterpiece was the very source of her inspiration (The Noble Prize, 2025). Artemisinin was known to be heat-unstable (Bilia et al., Reference Bilia, Gabriele, Bergonzi, de Malgalhaes and Vincieri2006). In Ge’s A. carvifolia prescription, he wrote (Figure 3):
‘又有青蒿一握,以水二升渍,绞取汁,尽服之。’
‘Grab a handful of Artemisia carvifolia, soak it with two litres of water, wring out the juice from the plant, drink it all’.
Ge Hong’s A Handbook of Prescriptions for Emergencies. Quotation appears in the fifth column counted from the right. Photo: Nobel Prize Museum.

Tu noted that ‘wring out the juice’ implies that heating might impair the effectiveness of the useful ingredients, hence she used ether – of which boiling point is merely 34·5℃ – to successfully extract artemisinin (National Administration of Traditional Chinese Medicine, 2019).
The Song Dynasty (AD 960–1279) was a time when malaria became more common, thus people’s understanding of this disease became more sophisticated. Chen Wuze, in his famous 三因极一病证方论 (Treatise on Three Categories of Pathogenic Factors), stated that there are 3 categories of causes of malaria: 1) external causes, such as warm and cold malaria; 2) internal causes, such as liver malaria, heart malaria, spleen malaria, lung malaria and kidney malaria; and 3) malaria without the influence of either internal or external causes, for instance ghost malaria. A deeper dive into the literature will help us better understand the reasoning behind such categorization: virtually all ‘external causes’ are in fact describing the cyclical shivering and chills. For example, ‘温疟 (warm malaria)’ is ‘病者先热后寒……故先热而后寒,名曰温疟 (The patient experiences fever followed by chills… Therefore, the condition followed by chills is called warm malaria)’ – whereas ‘寒疟 (cold malaria)’ ‘病者先寒后热,寒则汤火不能温,热则冰水不能寒 (The patient experiences chills followed by fever; when chilled, hot water could not warm the body, and when feverish, ice water could not cool it)’. On the other hand, ‘internal causes’ are the attempts of the author linking Yin-yang and the Five Elements with the 5 human organs, with all the diagnoses still mentioning the characteristic malarial symptom of chills and fevers. For instance, ‘脾疟 (spleen malaria)’ patients suffer from ‘……寒多,腹中热痛 (… excessive coldness with abdominal pain and heat)’, and is caused by ‘……以思伤脾,气郁涎结 (… a damage to spleen by excessive thoughts and melancholy)’. Finally, the third ‘without the influence of either internal or external causes’ malaria category mostly describes cases that cannot be cured for a prolonged period, hence attributed to factors such as ‘鬼 (ghost)’ or ‘劳 (attrition)’.
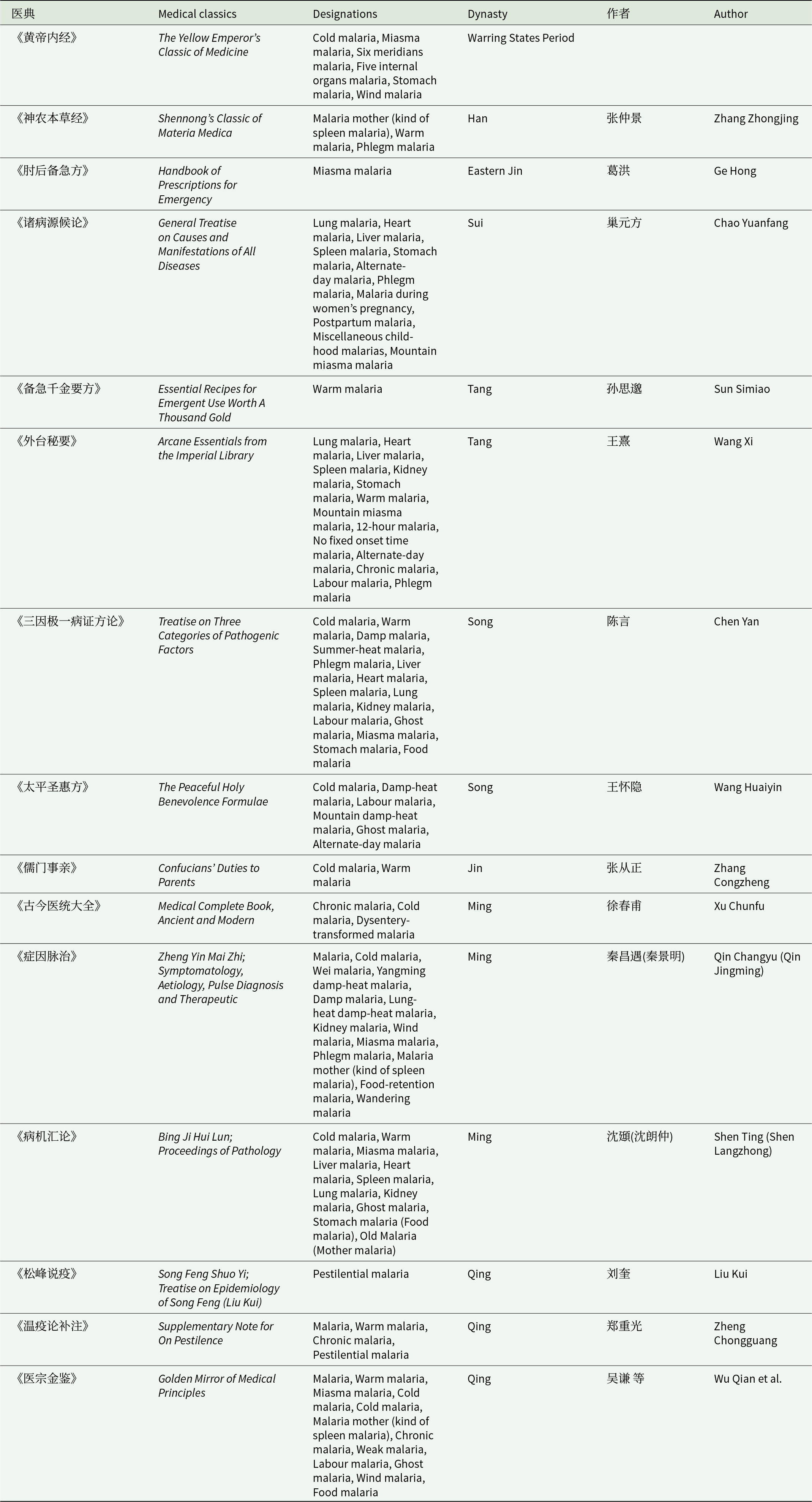
Prior to the late Qing Dynasty (AD 1840), there was no substantial change to how people viewed or treated malaria, and nor advances in treatment were developed (Table 2).
Description of different types of malaria in ancient Chinese medical books

Wu Kun, a medical practitioner in the late Ming dynasty (edited in Reference Wu and Hong1998), once stated:
‘疟之理难言矣。知五运、六气、十四经络,始能粗知其证;知阴阳进退、
消长之理,然后知夫疟疾变迁、之妙。’
‘The principles of malaria are difficult to explain. Only by understanding the Five Elements, Six Qi and Fourteen Meridians can one have a rough understanding of its symptoms; only by understanding the principles of Yin and Yang’s advance and retreat, waxing and waning, can one truly grasp the subtle changes in malaria’.
‘瘴气 (miasma)’ would often be used as synonym with ‘疟 (malaria)’, though the former denotes the hot and humid weather that is often seen in forests of southern regions. Analogous to the western counterparts, the odorous nature of miasma convinced many traditional Chinese medical practitioners that it is among the causes of malaria – along with diets and superstitious sources (Jiang, Reference Jiang2020).
An ancient disease like malaria, the knowledge of Chinese people on its concept and aetiology – just as many other aspects of society in general – was changed fairly recently in late Qing dynasty by the break in of western civilization. Miao (Reference Miao2025: 1) marked 3 phases of conceptual changes: 1) early missionary dictionaries (1822s–1860s) translated symptoms of malaria; 2) the 1870s–1920s, a period of terminological competition between ‘疟疾 (malaria)’ and ‘瘴气 (miasma)’, reflecting the clashes between traditional Chinese and western understandings of malaria that were inevitably encounter during the earlier endeavour; and 3) 1930s–1940s saw the term ‘疟疾 (malaria)’ became dominant through institutional standardization.
So how might modern bioarchaeological techniques help unveil the impact of malaria in ancient China? Should there be some direct aDNA evidence for malaria in China, we might have a clearer picture of its effect on ancient populations. Yet, evidence for malaria infection in ancient populations is hard to find, partially due to the fact that molecular evidence of malaria does not preserve well, and partially because acute death from malaria would not normally leave skeletal lesions, as explored in the osteological paradox (Wood et al., Reference Wood, Milner, Harpending, Weiss, Cohe, Eisenberg, Hutchinson, Jankauskas, Cesnys, Katzenberg, Lukacs, McGrath, Roth, Ubelaker and Wilkinson1992). Nonetheless, it is established that porotic lesions, such as CO, can be caused by malaria – thus, despite the risk of type 2 error, studying such lesions could shed light upon our understanding towards our interactions with malaria.
CO refers to the porotic lesions manifesting in orbital roofs (Welcker, Reference Welcker1888). Sufficient expansion of the bone marrow, along with atrophy of the cortical bone, will lead to perforation of the orbital roof and the outer table of the skull. Cortical bone forming the orbital roof is relatively thin, hence its atrophy becomes more significant at this location (Brickley, Reference Brickley2018). Anaemia is considered a primary factor in the development of CO, as in various forms of anaemia, including haemolytic (e.g. sickle cell), macrocytic (e.g. megaloblastic) and iron-deficiency anaemia, there is ineffective RBC production, hence the expansion of bone marrow to facilitate production of RBCs. Expanding marrow causes pressure atrophy, eventually leads to the thinning of cortical bone (McFadden and Oxenham, Reference McFadden and Oxenham2020). Therefore, in bioarchaeological literature, it is established that anaemia is among potential contributors to the development of CO (Oxenham and Cavill, Reference Oxenham and Cavill2010).
Nevertheless, anaemia does not produce porotic skeletal lesions in both current and past communities in many cases (Brickley, Reference Brickley2018). It has been argued that only severe congenital anaemia is sufficient to cause skull changes (Stuart-Macadam, Reference Stuart-Macadam1985). Research on the medieval population of Cambridge shows that evidence of roundworm and whipworm was found, but statistically, they had no correlation with CO, though these parasites are known to cause anaemia in clinical studies. Therefore, it is possible that only parasites capable of inducing severe anaemia contribute to the formation of CO (Wang et al., Reference Wang, McFadden, Buckley, Domett, Willis, Trinh, Matsumura, Vlok and Oxenham2023b).
Severe anaemia has been identified as a leading cause of morbidity and mortality in patients with malaria. Insufficient erythropoiesis constitutes a major factor in the development of malarial anaemia. The rapid multiplication of Plasmodium parasites can cause massive RBC destruction. Parasites, together with immune and non-immune mechanisms, jointly induce RBC loss, which ultimately leads to severe malarial anaemia (Chang and Stevenson, Reference Chang and Stevenson2004). Malaria infection triggers haemolysis – the process of an abnormal breakdown of RBCs. An individual becomes anaemic once haemolysis exceeds the rate of erythropoiesis (Gowland and Western, Reference Gowland and Western2012). Malarial haemolysis may liberate chemicals that impact the skeleton, while severe malarial anaemia can also induce extramedullary erythropoiesis, which is known to cause cortical bone atrophy (Smith-Guzmán, Reference Smith-Guzmán2015).
Research indicates a deep history of malaria in Mainland Southeast Asia, as evidenced by thalassemia, which is still endemic today. The only archaeological proof of hereditary anaemia in the region dates from approximately 4000–3500 BP from Khok Phanom Di, Thailand. Skeletal remains from the Man Bac and Con Co Ngua sites, both of which belong to the Da But Culture, exhibited bone marrow expansion – a clinical consequence manifestation of thalassemia (Vlok et al., Reference Vlok, Buckley, Miszkiewicz, Walker, Domett, Willis, Trinh, Minh, Nguyen, Nguyen, Matsumura, Wang, Nghia and Oxenham2021). The Da But Culture and the Dingshishan Culture in Guangxi, China, exhibit multiple similarities. The environmental factors of high temperature and humidity, along with cultural similarities such as high disease loads, pottery and access to stable sources of carbohydrates and protein (Oxenham et al., Reference Oxenham, Trinh, Willis, Jones, Domett, Castillo, Wood, Bellwood, Tromp, Kells, Piper, Pham, Matsumura and Buckley2018), lead to the conclusion that numerous cases of CO in Southeast Asia are highly correlated with malaria, as thalassemia is considered an adaptive response to the threat of this disease (Oxenham et al., Reference Oxenham, Trinh, Willis, Jones, Domett, Castillo, Wood, Bellwood, Tromp, Kells, Piper, Pham, Matsumura and Buckley2018). Pre-agricultural sedentism in forested environments may have facilitated malaria transmission, thereby driving natural selection for thalassemia-associated genes (Vlok et al., Reference Vlok, Buckley, Miszkiewicz, Walker, Domett, Willis, Trinh, Minh, Nguyen, Nguyen, Matsumura, Wang, Nghia and Oxenham2021).
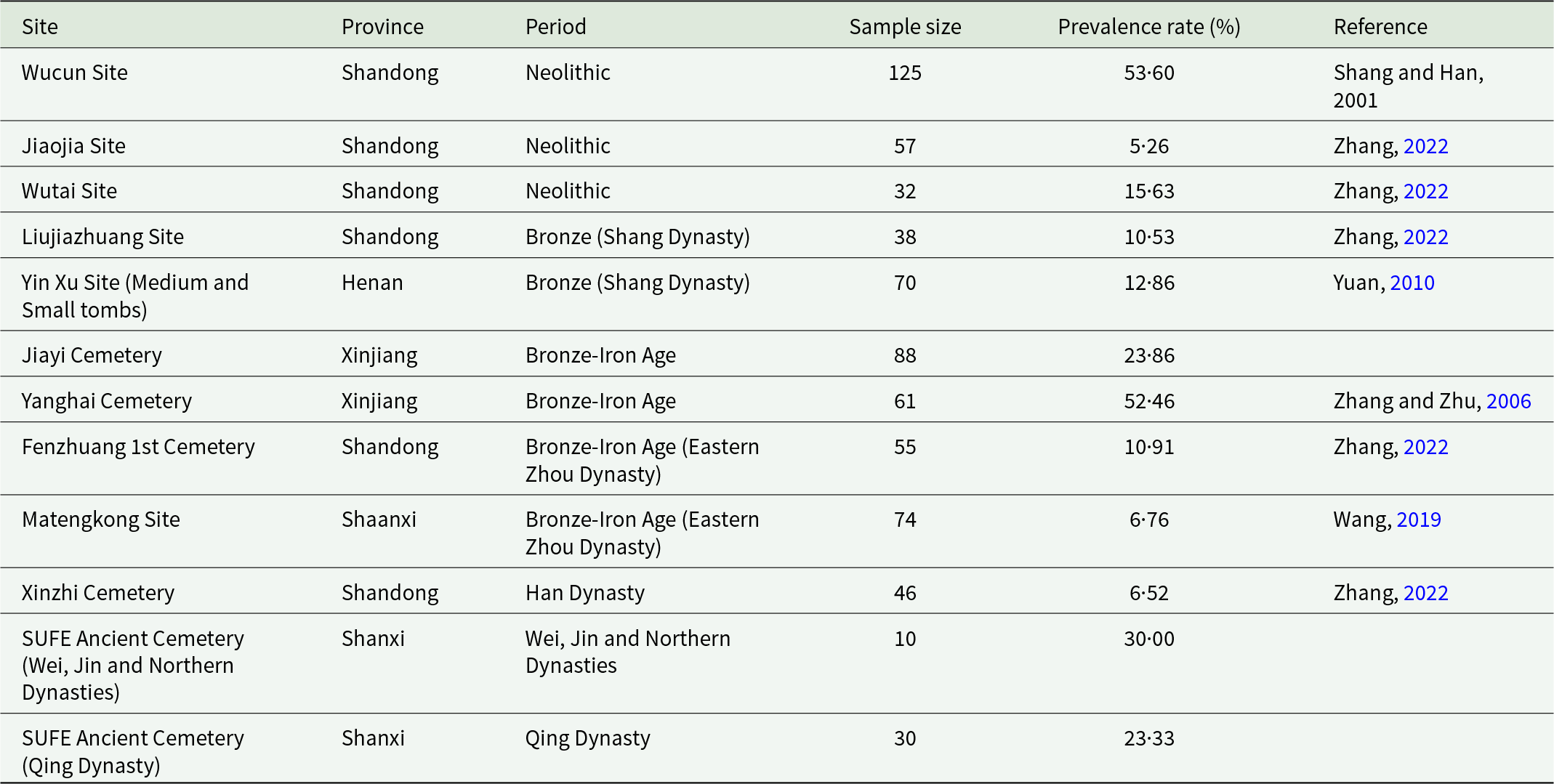
However, current understanding of palaeopathology in China is limited, calling for future studies. The palaeopathological records of CO in Chinese archaeological contexts (Table 3) are currently limited to certain regions, and there is no clear spatial/temporal distribution pattern. Research across multiple sites indicates that CO, a skeletal indicator often associated with anaemia, was present in ancient Chinese populations from the Neolithic to historical periods. Spatially, cases are concentrated in the Northwestern (Zhang and Zhu, Reference Zhang and Zhu2006; Wang, Reference Wang2019) and the Eastern region (Zhang, Reference Zhang2022; Song et al., Reference Song, Liu, You, Guo and Zhao2023) with fewer cases in the Central and Northeastern region (Yuan, Reference Yuan2010; Duan et al., Reference Duan, Fang, Quan, Wu, Lu, Zhao, Zhong and Cui2021). Take the Shandong Region as an example: from the Neolithic to the Han Dynasty, prevalence rates remained relatively stable, ranging from approximately 5% to 15%, without significant chronological fluctuation (Zhang, Reference Zhang2022). Scholarly interpretations primarily attribute CO to iron deficiency anaemia, resulting from factors such as dietary reliance on iron-poor cereals, parasitic infections and poor sanitary conditions (Shang and Han, Reference Shang and Han2001; Zhang and Zhu, Reference Zhang and Zhu2006). In pastoralist populations, like those from the Yanghai cemetery in Xinjiang, a diet rich in meat but lacking vitamin C may have impaired Fe absorption (Zhang and Zhu, Reference Zhang and Zhu2006). While some scholars have theorized a potential link between certain hereditary anaemias (like thalassemia) and malaria resistance, no direct archaeological evidence connects CO specifically to malaria infection in ancient China. The current understanding of these lesions underscores multifactorial aetiology tied to local subsistence economies and environmental conditions (Zhang, Reference Zhang2009; Wang, Reference Wang2019; Zhao and Li, Reference Zhao and Li2023). Following the work done by Gowland and Western (Reference Gowland and Western2012) and Schats (Reference Schats2026), scholars could investigate if CO is more prevalent in locations where the climates were more suitable for malaria transmission, even when the parasites themselves are hard to identify; grand-scale, population-level comparisons could also invoke interesting findings, as shown by Smith-Guzman (Reference Smith-Guzmán2015).
Palaeopathological records of cribra orbitalia in Chinese archaeological contexts

Conclusion
The historical trajectory of malaria across Asia reveals a dynamic narrative of co-evolution between Plasmodium parasites and their human hosts. While of African origin, the evolutionary path of malaria was fundamentally altered by the human transition to agriculture – East, Southeast and South Asia host some of its earliest occurrences. Cases covered in this review have shown that social and environmental modifications placed by humans had inadvertently yet explicitly expedited the transmission of malaria – from the medical and historical texts of ancient China, one can tell the persistent impact of malaria on the societies, general population and rulers alike.
Ultimately, the history of malaria in Asia underscores that human progress – whether through agricultural innovation, colonial industries or commercial trade – has never been ecologically neutral. By continuously redefining the parasite’s ecological niche, humanity has ensured that this ancient co-evolutionary struggle remains a constant force in regional health regulation.
Author contributions
TW conceived and designed the study. GL, XY and TW conducted data gathering. GL, XY and TW wrote the article.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
The authors declare there are no conflicts of interest.
Ethical standards
Not applicable.